Abstract
Inoculation of selected non-Saccharomyces yeasts with Saccharomyces cerevisiae as means to produce Merlot wines with reduced ethanol contents was investigated. Fermentations of grape musts (25.4° Brix, pH 3.50, and 4.23 g/L titratable acidity) were conducted in stainless steel tanks inoculated with Metschnikowia pulcherrima strains P01A016 or NS-MP or Meyerozyma guilliermondii P40D002 with S. cerevisiae Syrah added after three days. After fermentation, wines with Mt. pulcherrima contained 13.8% (P01A016) or 13.9% (NS-MP) v/v ethanol, respectively, amounts which were lower than in wines with S. cerevisiae alone (14.9% v/v). Delayed inoculation of must with S. cerevisiae (day 3) or musts with My. guilliermondii contained elevated concentrations of ethyl acetate (145 and 148 mg/L, respectively), concentrations significantly higher than those with S. cerevisiae inoculated on day 0 or with either strain of Mt. pulcherrima. Descriptive sensory analysis revealed a significant effect due to panelist but not due to Mt. pulcherrima or My. guilliermondii. This research indicates the potential for commercial application of these yeasts towards the production of reduced alcohol wines but without imparting negative sensory attributes.
1. Introduction
The use of non-Saccharomyces yeasts, in particular species of Metschnikowia, have gained popularity within the wine industry globally as a means to alter sensory profiles [1,2]. These yeasts can positively contribute to wine quality and complexity [3,4,5] through synthesis of compounds responsible for ‘fruity’ and/or ‘floral’ aromas [6,7,8]. In addition, many synthesize glycosidases and/or pectinases that can release varietal grape aromas as well as hydrolyze polysaccharides [9,10]. Conversely, however, wine quality can also be decreased through production of ethyl acetate, acetic acid, acetaldehyde, and/or other undesirable aroma or flavor compounds [6,11]. Growth of these yeasts in grape musts may also consume key nutrients important for Saccharomyces cerevisiae, resulting in increased risks associated with sluggish or stuck fermentations [12,13,14]. Several commercial preparations of Metschnikowia are now available to winemakers [2].
More recently, a number of studies have identified certain species of non-Saccharomyces yeasts, in particular, Metschnikowia pulcherrima, whose early growth can produce wines with less alcohol [2,5,8,15,16,17,18,19,20,21]. Canonico et al. [16] noted that these yeasts consume sugar through respiro-fermentative metabolisms (Crabtree negative) whereas metabolized carbon from sugars can be diverted to by-products other than ethanol. However, differences in response exist as reported by García et al. [22] and Hranilovic et al. [23] who found that ≤50% of Mt. pulcherrima strains examined yielded lower ethanol concentrations. Evaluating a number of different species isolated from Washington State vineyards [24], Aplin et al. [20] and Aplin and Edwards [21] reported that Mt. pulcherrima as well as a strain of Meyerozyma guilliermondii produced lower alcohol wines without large amounts of residual sugar (≤0.6 g/L) or acetic acid (≤0.25 g/L). Subsequent inoculation of S. cerevisiae is still required to complete fermentation because non-Saccharomyces yeasts are typically incapable of consuming all the available sugar in a grape must [1,9,20].
As many studies regarding use of Mt. pulcherrima were conducted in smaller laboratory-scale fermentations, often <1 L [5,15,19,25,26], few studies have been conducted under larger, pilot winery-scale conditions. Furthermore, while the sensory effects of different strains of Mt. pulcherrima have been reported [3,4,5,27], impacts of My. guilliermondii on wine quality have not been described. Thus, the objectives of this research were to evaluate the influence of Mt. pulcherrima and My. guilliermondii on ethanol production and quality of wines produced under pilot-scale winery conditions.
2. Materials and Methods
2.1. Yeast Strains
Mt. pulcherrima P01A016 and My. guilliermondii P40D002 were previously isolated from vineyards located at the Irrigated Research and Extension Center (Prosser, WA, USA) as described by Bourret et al. [24]. Additional strains of Mt. pulcherrima (NS-MP) and S. cerevisiae (Enoferm Syrah) were acquired from Lallemand Inc. (Montréal, QC, Canada). All yeasts were maintained on yeast peptone dextrose (YPD) agar plates containing 10 g/L yeast extract (Becton, Dickinson, and Company, Sparks, MD, USA), 20 g/L peptone (Becton, Dickinson, and Company, Sparks, MD, USA), 20 g/L glucose (Thermo Fisher Scientific, Waltham, MA, USA), and 15 g/L agar (Acros Organics, Morris, NJ, USA).
Non-Saccharomyces yeast starter cultures were prepared by inoculating yeast/mold (YM) broth (Becton, Dickinson, and Company, Sparks, MD) from single colonies grown on YPD agar. Upon reaching the late exponential phase, cells were harvested by centrifugation at 2000× g for 20 min, washed twice with 0.2 M Na2HPO4 (pH 7.0) buffer, then resuspended in buffer prior to inoculation. Active dry yeast cultures of S. cerevisiae were rehydrated according to manufacturer’s instructions.
2.2. Merlot Grape Must
Merlot grapes were hand-harvested 22 September 2016 from vineyards located at the Irrigated Agriculture Research and Extension Center (Washington State University, Prosser, WA, USA) and processed using a stainless steel crusher-destemmer (approximately 1300 kg/h). Immediately after crushing, 20 mg/L total SO2 were added as K2S2O5 to 37.5 kg of must which had been placed in 300 L stainless steel, jacketed tanks. The must contained 264 g/L glucose and fructose, pH 3.50, 4.23 g/L titratable acidity (as tartaric acid), 198 mg N/L (yeast assimilable nitrogen or YAN). Diammonium phosphate was added at a rate of 0.2 g/kg to increase available nitrogen to 250 mg N/L. Musts were then either uninoculated or inoculated at 106 cfu/mL with Mt. pulcherrima P01A016, Mt. pulcherrima NS-MP, My. guilliermondii P40D002, or S. cerevisiae, in triplicate. After three days, S. cerevisiae (106 cfu/mL) into those treatments not previously inoculated with this yeast. Fermentations were conducted at cellar temperature (approximately 20 °C), without heating or chilling, with 1-min daily punch-downs using a stainless-steel punch-down tool. At approximately 18° Brix, 0.2 g/kg Fermaid-K (Lallemand Inc.) was added as a nitrogen supplement to all tanks while fermentations were pressed at approximately 0° Brix using a hydraulic bladder press (Speidel, Ofterdingen, Germany). Once dry (<2 g/L reducing sugars), wines were racked and moved to 3–4 °C after addition of 50 mg/L total SO2. After 9 months of storage, enough K2S2O5 was added to achieve 0.8 mg/L molecular SO2 prior to bottling without filtering. Bottled wines were kept at 4 °C to limit potential microbial spoilage for at least 4 months before analyses.
2.3. Chemical and Microbiological Analyses
Yeast culturabilities were monitored by spiral plating using an Autoplate 4000 (Spiral Biotech, Bethesda, MD, USA) and both Wallenstein Laboratory nutrient agar for total yeast populations (Becton, Dickinson, and Company, Franklin Lakes, NJ, USA) and lysine agar for non-Saccharomyces yeasts (Oxoid, Hampshire, UK). Plates were incubated at 28 °C for two days prior to enumeration. S. cerevisiae populations were estimated as differences between culturabilities on the two media.
Fermentations were monitored by measuring soluble solids with an Anton Paar DMA35 density meter (Anton Paar USA Inc., Ashland, VA, USA). Volatile acidity (Cash still), titratable acidity, residual sugars (Clinitest® method), and free and total SO2 (aeration/oxidation) were measured while molecular SO2 was calculated following standard methods [28]. Yeast assimilable nitrogen (YAN) was calculated as the sum of primary amino nitrogen according to Dukes and Butzke [29] and ammonium by an ion-selective probe (Denver Instruments, Orville, NY, USA).
Concentrations of glucose, fructose, glycerol, and organic acids were quantified by an Agilent 1100 HPLC system equipped with an Aminex HPX-87H column (300 × 7.8 mm, BIO-RAD, Hercules, CA, USA) equilibrated at 60 °C with 0.005M H2SO4 as mobile phase flowing at 0.6 mL/min.
Volatile aroma compounds were analyzed by headspace solid-phase microextraction coupled with gas chromatography-mass spectroscopy as previously described [20]. Briefly, compounds were analyzed with an Agilent HP 6890 gas chromatograph fitted with a DB-1MS column (0.32 mm × 60.0 m, 1.0 μm thickness, Phenomenex, Torrence, CA, USA) linked to an HP 5973 Mass Selective Detector. Fibers were desorbed for 3 min at 250 °C with the injection inlet operating in splitless mode and a carrier gas (He) flow of 0.7 mL/min. The temperature program was as follows: 33 °C for 5 min, increase to 50 °C at 2.0 °C/min, increase to 225 °C as 5.0 °C/min and hold for 5 min. The mass spectrometer was operating in electron impact mode at 70 eV (150 °C ion source). Compounds were identified using the Wiley/NIST library while quantification was accomplished using 4-methyl-2-pentanol and 2-octanol as internal standards.
2.4. Sensory Analyses
Merlot wines were evaluated by a trained panel (n = 10, four males and six females aged 24 to 40) consisting of regular wine drinkers recruited from the Washington State University community. Panelists received 12 h of training across six weeks using feedback calibration through Compusense Cloud ver. 8.8 sensory acquisition software (Guelph, ON, Canada). All sensory attributes were illustrated with appropriate reference standards for aroma or flavor (‘estery,’ ‘pungency,’ ‘fruity,’ ‘dried fruit,’ ‘green,’ ‘yeasty,’ ‘ethanol,’ ‘berry,’ ‘earthy,’ ‘woody,’ ‘sulfur,’ ‘sweaty,’ ‘chemical,’ ‘vegetal’ or ‘barn’), taste (‘sweet,’ ‘bitter’ or ‘sour’), and mouthfeel (‘viscosity,’ ‘hot,’ ‘astringent’ or ‘roughness’) as described by Aplin [30]. Samples (40 mL) were presented to panelist in covered three-digit coded ISO standard wine glasses at room temperature in individual tasting booths under white light at the Washington State University Sensory Evaluation Facility. For the final evaluations, ten wines were assessed in duplicate in a randomized order over four evaluation sessions. Five wines were evaluated each session and fresh bottles were opened each day to prevent oxidative changes to wines. To avoid fatigue, panelists were required to take a five-minute break between samples and instructed to rinse their palate with water and an unsalted cracker. Data was collected on a 15 cm unstructured line scale with anchor points ‘low’ (10% of the scale) and ‘high’ (90% of the scale) using Compusense software.
2.5. Statistical Analyses
Statistical analyses of chemical analyses were performed by ANOVA while means separations were accomplished by Fisher’s LSD using XLSTAT software (Addinsoft, New York, NY, USA). For sensory data, three-way ANOVA was performed to analyze panelist, treatment, and replicate interactions while means were separated using Fisher’s Least Significant Difference (LSD) post-hoc test. Differences were considered significant when p ≤ 0.05.
3. Results
Merlot grape musts were inoculated with S. cerevisiae alone at days 0 or 3 (Figure 1) or with Mt. pulcherrima P01A016 (Figure 2A), Mt. pulcherrima NS-MP (Figure 2B), or My. guilliermondii P40D002 (Figure 2C) at day 0 with S. cerevisiae added on day 3. Populations of S. cerevisiae nearly reached ≈ 108 cfu/mL by day 3 in ferments inoculated with this yeast alone on day 0 (Figure 1A) or by day 7 when inoculation was delayed (Figure 1B). Native non-Saccharomyces present in these ferments did not exceed 106 cfu/mL with inoculation of S. cerevisiae on day 0 (Figure 1A). However, when S. cerevisiae was inoculated on day 3, unidentified non-Saccharomyces present in the must reached populations approaching 107 cfu/mL before entering decline to undetectable levels by day 10 (Figure 1B). In comparison, total populations of non-Saccharomyces yeasts in musts inoculated with Mt. pulcherrima P01A016, Mt. pulcherrima NS-MP, or My. guilliermondii P40D002 approached one log higher, 108 cfu/mL, by day 3 prior to decreases to ≈ 103 cfu/mL by day 10 (Figure 2). In these fermentations, S. cerevisiae approached 108 cfu/mL by day 7 and remained culturable at day 16.
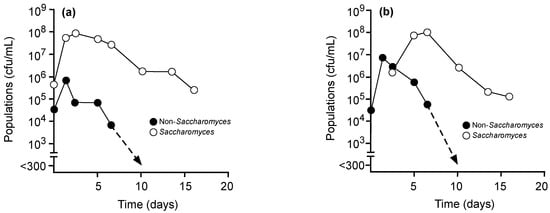
Figure 1.
Culturable populations of uninoculated with non-Saccharomyces yeasts (●) but with Saccharomyces cerevisiae (○) inoculated either on day 0 (a) or day 3 (b) in Merlot grape musts.
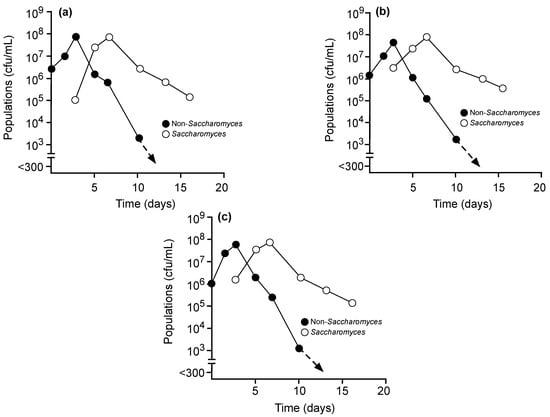
Figure 2.
Culturable populations of total non-Saccharomyces yeasts (●) or Saccharomyces cerevisiae (○) in Merlot grape musts where Mt. pulcherrima P01A016 (a), Mt. pulcherrima NS-MP (b), or My. guilliermondii P40D002 (c) were inoculated on day 0 with Saccharomyces cerevisiae added on day 3.
Chemical compositions of final wines depended on the yeast species present and populations achieved. While ferments inoculated on day 0 with S. cerevisiae alone achieved dryness by day 13 (≤2 g/L reducing sugar), wines inoculated with non-Saccharomyces yeasts or S. cerevisiae added on day 3 required approximately three additional days (data not shown). In support, residual concentrations of glucose and fructose reached ≤0.13 g/L and were not significantly different between wines (Table 1). Furthermore, amounts of glycerol (10.0 to 10.1 g/L) and succinic acid (1.73 to 1.91 g/L) did not vary depending on the yeast present. While wines inoculated with non-Saccharomyces yeasts contained slightly higher titratable acidities and decreased pH, both strains of Mt. pulcherrima metabolized some of the malic acid present, approximately 36 to 40%. Concentrations of volatile acidity were similar (Mt. pulcherrima P01A016 and My. guilliermondii P40D002) or less (Mt. pulcherrima NS-MP) than wines inoculated with S. cerevisiae alone. In fact, wines inoculated on day 3 with S. cerevisiae alone contained the highest concentrations of volatile acidity (0.45 g/L), potentially due to the high populations of unidentified non-Saccharomyces yeasts naturally present (Figure 1B). However, wines inoculated with either strain of Mt. pulcherrima contained 1.0 to 1.1% v/v less ethanol than ferments inoculated with S. cerevisiae alone (14.9% v/v). The addition of My. guilliermondii to grape musts did not affect final alcohol concentration (15.0% v/v).

Table 1.
Concentration of volatile compounds in Merlot wines inoculated with or without non-Saccharomyces yeasts followed by S. cerevisiae.
Besides changes to composition previously described, Merlot wines inoculated with non-Saccharomyces yeasts exhibited different concentrations of various aroma and/or flavor compounds (Table 1). In general, higher alcohols were more abundant in wines inoculated with non-Saccharomyces yeasts, primarily due to increased levels of 2-methyl-1-propanol (61.4 to 70.4 mg/L) and 2- and 3-methyl-1-butanols (137 to 165 mg/L). Wines only inoculated with S. cerevisiae at day 0 contained 179 mg/L total higher alcohols, compared to wines with Mt. pulcherrima NS-MP (272 mg/L) or My. guilliermondii (266 mg/L). Higher concentrations of ethyl acetate were noted where addition of S. cerevisiae was delayed until day 3 (145 mg/L) or in the presence of My. guilliermondii (145 mg/L) in contrast other wines (52.3 to 73.1 mg/L). Significant differences were not observed regarding concentrations of some esters and medium-chain fatty acids (e.g., hexyl acetate, ethyl butanoate, ethyl hexanoate, and octanoic acid) while a limited number of differences were noted for others (e.g., 2- and 3-methylbutyl acetates, diethyl succinate, 2-phenylethyl acetate, ethyl octanoate, and octanoic acid).
Analysis of variance results for sensory attributes revealed no significant differences attributed to yeast but primarily to panelists (Table 2). In fact, significant panelist effects (p < 0.05) were noted for all attributes except ‘green,’ ‘earthy,’ ‘sweaty,’ and ‘vegetal’ (aromas) or for ‘earthy’ and ‘vegetal’ (flavors) with very few differences between aroma, taste, mouthfeel, or flavor attributes (data not shown).

Table 2.
Significance and F ratios from analysis of variance of trained panel evaluations of aroma, taste, mouthfeel, and flavor sensory attributes in Merlot wines inoculated with or without non-Saccharomyces yeasts followed by S. cerevisiae.
4. Discussion
In sequentially inoculated grape musts, the presence of Mt. pulcherrima or My. guilliermondii did not adversely affect alcoholic fermentation by S. cerevisiae. While observations related to the growth of Mt. pulcherrima were similar to small volume fermentations [8,20,25], this is the first report describing unsterilized musts inoculated with either Mt. pulcherrima or My. guilliermondii under larger-scale vinification conditions. Of concern was that initial growth of non-Saccharomyces yeasts may metabolize key nutrients present in grape must [12,13], thereby yielding incomplete alcoholic fermentations [8,17,19,31]. In fact, wines with inoculated non-Saccharomyces yeasts reached dryness within three days of those musts inoculated with only S. cerevisiae. However, an apparent suppression effect against unidentified, native non-Saccharomyces yeasts was observed by inoculation of S. cerevisiae on day 0. Here, inoculation of S. cerevisiae upon preparation of the must resulted in total populations of non-Saccharomyces yeasts a log lower compared to ferments with delayed inoculation. Antagonistic effects between yeasts have been thought to be due to competition for nutrients or the production of toxic metabolites [4,8,9,26,27].
Sequential inoculation of S. cerevisiae three days after addition of Mt. pulcherrima strain P01A016 or NS-MP produced Merlot wines which contained less ethanol (13.8% and 13.9% v/v, respectively) compared to wines inoculated only with S. cerevisiae (14.9% v/v). In agreement, other studies have reported reductions of 0.9 to 2.5% v/v by addition of Mt. pulcherrima, the amount depending on strain and inoculation protocol [5,8,16,19,23]. Studying six strains of Mt. pulcherrima, García et al. [22] noted that only two resulted in wines with lower ethanol concentrations, in agreement with Hranilovic et al. [23] who reported three of six strains yielded lower amounts of ethanol. The study by Hranilovic et al. [23] also highlighted the impact of inoculation timing where addition of S. cerevisiae two days after Mt. pulcherrima yielded the greatest decrease in ethanol yield, approximately 2.5% v/v, compared to additions up to seven days. Earlier work by Contreras et al. [8] studied inoculation of S. cerevisiae once 50% of the sugar in Shiraz and Chardonnay grape musts had been consumed by Mt. pulcherrima, a protocol which delayed addition until day 9 or 17, respectively. Of concern is that long delays before S. cerevisiae inoculation may be difficult for wineries to manage because extensive growth of other non-Saccharomyces could decrease important nutrients or produce by-products which decrease quality [13,32].
Unlike Mt. pulcherrima, presence of My. guilliermondii did not affect the final ethanol content of wines. This finding was in contrast to Aplin et al. [20] who had conducted smaller volume fermentations (3 L) using a sterile-filtered, grape juice made from concentrate (Merlot). Later work by Aplin and Edwards [21] utilizing a high sugar must (310 g/L) also reported a decrease in ethanol production. Here, addition of My. guilliermondii resulted in wines containing 15.5% v/v ethanol compared to control wines that contained 16.1% v/v. Despite reaching total populations of non-Saccharomyces yeasts of almost 107 cfu/mL, the metabolism of My. guilliermondii may have been adversely affected by naturally-present microflora. Though studies on My. guilliermondii are lacking, Contreras et al. [25] noted that growth of Mt. pulcherrima was inhibited due to the presence of Hanseniaspora uvarum, Pichia kluyveri, and Torulaspora delbrueckii.
To date, the fate of carbon from metabolized sugar by Mt. pulcherrima remains unclear. For example, Merlot wines produced with either strain of Mt. pulcherrima P01A016 and NS-MP contained similar concentrations of glycerol and succinic acid as those produced by S. cerevisiae alone, in agreement with some reports [19] but in disagreement to others [5,23]. In fact, succinic acid can be formed by at least three additional pathways besides tricarboxylic acid cycle such as the γ-amino butyric acid bypass (shunt), glyoxylic acid bypass, and/or the methylcitric acid cycle [33] which may or may not be active in Mt. pulcherrima under vinification conditions. Besides glycerol and succinic acid, Hranilovic et al. [23] suggested that another metabolic product of the tricarboxylic acid cycle, fumaric acid, may also partially explain the fate of metabolized sugar.
Few reports are available regarding the impacts of My. guilliermondii on chemical composition or wine sensory properties. Contreras et al. [26] noted their strain consumed more sugar than Mt. pulcherrima (40.5% vs. 21.2%) yet expressed similar ethanol yield (0.38 vs. 0.36 g ethanol/g sugar). Despite high concentrations of ethyl acetate (148 mg/L), these wines were not sensorily different from those produced with S. cerevisiae alone (52.3 mg/L ethyl acetate). Ethyl acetate is associated with the off-aroma of ‘nail-polish remover’ when present at concentrations ≥150 mg/L [34]. In addition, wines with My. guilliermondii were characterized by increased concentrations of 2- and 3-methyl-butyl acetates, 2-phenylethyl acetate, and/or higher alcohols associated with ‘fruity’ and/or ‘floral’ descriptors [34]. In one of the few studies involving My. guilliermondii (synonym Pichia guilliermondii), Benito et al. [35] noted elevated amounts of vinylphenolic pyranoanthocyanins, highly stable wine pigments which increase color stability, as well as increased ethyl acetate and higher alcohols due to this yeast.
Descriptive sensory analysis revealed that wines produced with non-Saccharomyces and S. cerevisiae yeasts exhibited few significant differences in aroma, mouthfeel, taste and flavor characteristics. While positive sensory characteristics were not imparted, this observation further suggests that use of these yeasts does not hamper wine quality. In other studies, presence of different strains of Mt. pulcherrima were shown to increase ‘fruity’ and ‘floral’ aromas [5,27,36] as well as overall quality scores in Chenin blanc and Sauvignon blanc wines [3,4]. Although differences due to yeast were not noted, panelist responses were highly variable, as evidenced by the significant panelist effect for most sensory attributes (p < 0.05). Panelists are known to be a significant source of variation in descriptive analysis through differences in use of scale and attribute definition [37,38,39].
5. Conclusions
This work described Merlot wines produced with Mt. pulcherrima and My. guilliermondii yeasts under pilot-scale winery conditions. Inoculation of Mt. pulcherrima P01A016 and NS-MP generated wines did not impact sensory characteristics but did yield less alcohol in the final wines. Future research concerning other processing conditions (i.e., must temperature) is needed to optimize ethanol reduction in fermentations with these non-Saccharomyces yeasts.
Author Contributions
Conceptualization, J.J.A. and C.G.E.; methodology, J.J.A. and V.D.P.; formal analysis, J.J.A. and V.D.P.; writing—original draft preparation, C.G.E.; writing—review and editing, J.J.A., V.D.P., C.F.R., C.G.E.; supervision, C.F.R. and C.G.E.; funding acquisition, C.G.E. All authors have read and agreed to the published version of the manuscript.
Funding
This research was funded by Washington State Grape and Wine Research Program with additional support from Lallemand (Montréal, QC, Canada) and Washington State University through the School of Food Science (Pullman, WA, USA) and Irrigated Agriculture Research and Extension Center (Prosser, WA, USA). This work was also supported, in part, by the USDA National Institute of Food and Agriculture, Hatch project 1016366 and project No. 0846 of the Agricultural Research Center, Washington State University, Pullman, WA, USA, 99164-6376.
Institutional Review Board Statement
The study was conducted according to the guidelines of the Declaration of Helsinki, and approved by the Institutional Review Board (or Ethics Committee) of Washington State University (IRB#15370-004) on 16 August 2017.
Informed Consent Statement
Informed consent was obtained from all subjects involved in the study.
Data Availability Statement
Data are not available from other forums.
Acknowledgments
The authors gratefully acknowledge the Washington State Grape and Wine Research Program, Lallemand Inc., and the School of Food Science at Washington State University for financial and material support.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Jolly, N.P.; Varela, C.; Pretorius, I.S. Not your ordinary yeast: Non-Saccharomyces yeasts in wine production uncovered. FEMS Yeast Res. 2014, 14, 215–237. [Google Scholar] [CrossRef] [Green Version]
- Vicente, J.; Ruiz, J.; Belda, I.; Benito-Vázquez, I.; Marquin, D.; Calderón, F.; Santos, A.; Benito, S. The genus Metschnikowia in enology. Microorganisms 2020, 8, 1038. [Google Scholar] [CrossRef]
- Jolly, N.P.; Augustyn, O.P.H.; Pretorius, I.S. The effect of non-Saccharomyces yeasts on fermentation and wine quality. S. Afr. J. Enol. Vitic. 2003, 24, 55–62. [Google Scholar] [CrossRef]
- Jolly, N.P.; Augustyn, O.P.H.; Pretorius, I.S. The use of Candida pulcherrima in combination with Saccharomyces cerevisiae for the production of Chenin blanc wine. S. Afr. J. Enol. Vitic. 2003, 24, 63–69. [Google Scholar] [CrossRef] [Green Version]
- Varela, C.; Barker, A.; Tran, T.; Borneman, A.; Curtin, C. Sensory profile and volatile aroma composition of reduced alcohol Merlot wines fermented with Metschnikowia pulcherrima and Saccharomyces uvarum. Int. J. Food Microbiol. 2017, 252, 1–9. [Google Scholar] [CrossRef]
- Rojas, V.; Gil, J.V.; Piñaga, F.; Manzanares, P. Acetate ester formation in wine by mixed cultures in laboratory fermentations. Int. J. Food Microbiol. 2003, 86, 181–188. [Google Scholar] [CrossRef]
- Viana, F.; Gil, J.V.; Genovés, S.; Vallés, S.; Manzanares, P. Rational selection of non-Saccharomyces wine yeasts for mixed starters based on ester formation and enological traits. Food Microbiol. 2008, 25, 778–785. [Google Scholar] [CrossRef] [PubMed]
- Contreras, A.; Hidalgo, C.; Henschke, P.A.; Chambers, P.J.; Curtin, C.; Varela, C. Evaluation of non-Saccharomyces yeasts for the reduction of alcohol content in wine. Appl. Environ. Microbiol. 2014, 80, 1670–1678. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Comitini, F.; Gobbi, M.; Domizio, P.; Romani, C.; Lencioni, L.; Mannazzu, I.; Ciani, M. Selected non-Saccharomyces wine yeasts in controlled multistarter fermentations with Saccharomyces cerevisiae. Food Microbiol. 2011, 28, 873–882. [Google Scholar] [CrossRef] [PubMed]
- Domizio, P.; Romani, C.; Lencioni, L.; Comitini, F.; Gobbi, M.; Mannazzu, I.; Ciani, M. Outlining a future for non-Saccharomyces yeasts: Selection of putative spoilage wine strains to be used in association with Saccharomyces cerevisiae for grape juice fermentation. Int. J. Food Microbiol. 2011, 147, 170–180. [Google Scholar] [CrossRef]
- Ciani, M.; Maccarelli, F. Oenological properties of non-Saccharomyces yeasts associated with wine-making. World J. Microbiol. Biotechnol. 1998, 14, 199–203. [Google Scholar] [CrossRef]
- Bataillon, M.; Rico, A.; Sablayrolles, J.-M.; Salmon, J.-M.; Barre, P. Early thiamin assimilation by yeasts under enological conditions: Impact on alcoholic fermentation kinetics. J. Ferment. Bioeng. 1996, 82, 145–150. [Google Scholar] [CrossRef]
- Medina, K.; Boido, E.; Dellacassa, E.; Carrau, F. Growth of non-Saccharomyces yeasts affects nutrient availability for Saccharomyces cerevisiae during wine fermentation. Int. J. Food Microbiol. 2012, 157, 245–250. [Google Scholar] [CrossRef]
- Rollero, S.; Bloem, A.; Ortiz-Julien, A.; Camarasa, C.; Divol, B. Altered fermentation performances, growth, and metabolic footprints reveal competition for nutrients between yeast species inoculated in synthetic grape juice-like medium. Front. Microbiol. 2018, 9, 196. [Google Scholar] [CrossRef]
- Quirós, M.; Rojas, V.; Gonzalez, R.; Morales, P. Selection of non-Saccharomyces yeast strains for reducing alcohol levels in wine by sugar respiration. Int. J. Food Microbiol. 2014, 181, 85–91. [Google Scholar] [CrossRef] [Green Version]
- Canonico, L.; Comitini, F.; Oro, L.; Ciani, M. Sequential fermentations with selected immobilized non-Saccharomyces yeast for reduction of ethanol content in wine. Front. Microbiol. 2016, 7, 278. [Google Scholar] [CrossRef] [Green Version]
- Englezos, V.; Rantsiou, K.; Cravero, F.; Torchio, F.; Ortiz-Julien, A.; Gerbi, V.; Rolle, L.; Cocolin, L. Starmerella bacillaris and Saccharomyces cerevisiae mixed fermentations to reduce ethanol content in wine. Appl. Microbiol. Biotechnol. 2016, 100, 5515–5526. [Google Scholar] [CrossRef]
- Rossouw, D.; Bauer, F. Exploring the phenotypic space of non-Saccharomyces wine yeast biodiversity. Food Microbiol. 2016, 55, 32–46. [Google Scholar] [CrossRef]
- Varela, C.; Sengler, F.; Solomon, M.; Curtin, C. Volatile flavor profile of reduced alcohol wines fermented with the non-conventional yeast species Metschnikowia pulcherrima and Saccharomyces uvarum. Food Chem. 2016, 209, 57–64. [Google Scholar] [CrossRef] [PubMed]
- Aplin, J.J.; White, K.P.; Edwards, C.G. Growth and metabolism of non-Saccharomyces yeasts isolated from Washington state vineyards in media and high sugar grape musts. Food Microbiol. 2019, 77, 158–165. [Google Scholar] [CrossRef] [PubMed]
- Aplin, J.A.; Edwards, C.G. Impacts of non-Saccharomyces species and aeration on sequential inoculation with Saccharomyces cerevisiae to produce lower alcohol Merlot wines. J. Sci. Food Agric. 2021, 101, 1715–1719. [Google Scholar] [CrossRef]
- García, M.; Esteve-Zarzoso, B.; Cabellos, J.M.; Teresa Arroyo, T. Sequential non-Saccharomyces and Saccharomyces cerevisiae fermentations to reduce the alcohol content in wine. Fermentation 2020, 6, 60. [Google Scholar] [CrossRef]
- Hranilovic, A.; Gambetta, J.M.; Jeffery, D.W.; Grbin, P.R.; Jiranek, V. Lower-alcohol wines produced by Metschnikowia pulcherrima and Saccharomyces cerevisiae co-fermentations: The effect of sequential inoculation timing. Int. J. Food Microbiol. 2020, 329, 108651. [Google Scholar] [CrossRef] [PubMed]
- Bourret, T.B.; Grove, G.G.; Vandemark, G.J.; Henick-Kling, T.; Glawe, D.A. Diversity and molecular determination of wild yeasts in a central Washington state vineyard. N. Am. Fungi 2013, 8, 1–32. [Google Scholar] [CrossRef] [Green Version]
- Contreras, A.; Curtin, C.; Varela, C. Yeast population dynamics reveal a potential ‘collaboration’ between Metschnikowia pulcherrima and Saccharomyces uvarum for the production of reduced alcohol wines during Shiraz fermentation. Appl. Microbiol. Biotechnol. 2015, 99, 1885–1895. [Google Scholar] [CrossRef]
- Contreras, A.; Hidalgo, C.; Schmidt, S.; Henschke, P.A.; Curtin, C.; Varela, C. The application of non-Saccharomyces yeast in fermentations with limited aeration as a strategy for the production of wine with reduced alcohol content. Int. J. Food Microbiol. 2015, 205, 7–15. [Google Scholar] [CrossRef]
- Rodríguez, M.E.; Lopes, C.A.; Barbagelata, R.J.; Barda, N.B.; Caballero, A.C. Influence of Candida pulcherrima Patagonian strain on alcoholic fermentation behaviour and wine aroma. Int. J. Food Microbiol. 2010, 138, 19–25. [Google Scholar] [CrossRef] [PubMed]
- Ough, C.S.; Amerine, M.A. Methods for Analysis of Musts and Wines; John Wiley & Sons: New York, NY, USA, 1988. [Google Scholar]
- Dukes, B.C.; Butzke, C.E. Rapid determination of primary amino acids in grape juice using an o-pthaldialdehyde/N-acetyl-L-cysteine spectrophotometric assay. Am. J. Enol. Vitic. 1998, 49, 125–134. [Google Scholar]
- Aplin, J.A. Use of Non-Saccharomyces Yeasts for Reducing the Ethanol Contents of Red Wine. Ph.D. Thesis, Washington State University, Pullman, WA, USA, 2019. [Google Scholar]
- Andorrà, I.; Berradre, M.; Rozès, N.; Mas, A.; Guillamón, J.M.; Esteve-Zarzoso, B. Effect of pure and mixed cultures of the main wine yeast species on grape must fermentations. Eur. Food Res. Technol. 2010, 231, 215–224. [Google Scholar] [CrossRef] [Green Version]
- Gobert, A.; Tourdot-Maréchal, R.; Morge, C.; Sparrow, C.; Liu, Y.; Quintanilla-Casas, B.; Vichi, S.; Alexandre, H. Non-Saccharomyces yeasts nitrogen source preference: Impacts on sequential fermentation and wine volatile compounds profile. Front. Microbiol. 2017, 8, 2175. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- De Klerk, J.L. Succinic Acid Production by Wine Yeasts. Master’s Thesis, Stellenbosch University, Stellenbosch, South Africa, 2010. [Google Scholar]
- Jackson, R.S. Wine Science: Principles and Applications, 3rd ed.; Elsevier Science: Amsterdam, The Netherlands, 2008. [Google Scholar]
- Benito, S.; Morata, A.; Palomero, F.; González, M.C.; Suárez-Lepe, J.A. Formation of vinylphenolic pyranoanthocyanins by Saccharomyces cerevisiae and Pichia guillermondii in red wines produced following different fermentation strategies. Food Chem. 2011, 124, 15–23. [Google Scholar] [CrossRef]
- Benito, S.; Hofmann, T.; Laier, M.; Lochbüler, B.; Schüttler, A.; Ebert, K.; Fritsch, S.; Röcker, J.; Rauhut, D. Effect on quality and composition of Riesling wines fermented by sequential inoculation with non-Saccharomyces and Saccharomyces cerevisiae. Eur. Food Res. Technol. 2015, 241, 707–717. [Google Scholar] [CrossRef]
- Bende, M.; Nordin, S. Perceptual learning in olfaction: Professional wine tasters versus controls. Physiol. Behav. 1997, 62, 1065–1070. [Google Scholar] [CrossRef]
- Landon, J.L.; Weller, K.; Harbertson, J.F.; Ross, C.F. Chemical and sensory evaluation of astringency in Washington state red wines. Am. J. Enol. Vitic. 2008, 59, 153–158. [Google Scholar]
- McMahon, K.M.; Diako, C.; Aplin, J.; Mattinson, D.S.; Culver, C.; Ross, C.F. Trained and consumer panel evaluation of sparkling wines sweetened to brut or demi sec residual sugar levels with three different sugars. Food Res. Int. 2017, 99, 173–185. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).