Aromatic Potential of Diverse Non-Conventional Yeast Species for Winemaking and Brewing
Abstract
1. Introduction
2. Materials and Methods
2.1. Strains
2.2. Microscale Fermentations
2.3. Residual Sugars and Ethanol Analysis by High-Performance Liquid Chromatography (HPLC)
2.4. Aroma Analysis by GC-MS
2.5. Comparison of Microscale to Labscale
2.5.1. Biomass Analysis
2.5.2. Fermentation Kinetics and Aroma Profiles
2.6. Statistical Analysis
3. Results and Discussion
3.1. Microwine
3.1.1. Fermentation Performance
3.1.2. Aroma Profiles
Higher Alcohols
Acetate Esters
Ethyl Esters
3.2. Microbeer
3.2.1. Fermentation Performance
3.2.2. Aroma Profiles
Vicinal Diketones
Higher Alcohols
Acetate Esters
Ethyl Esters
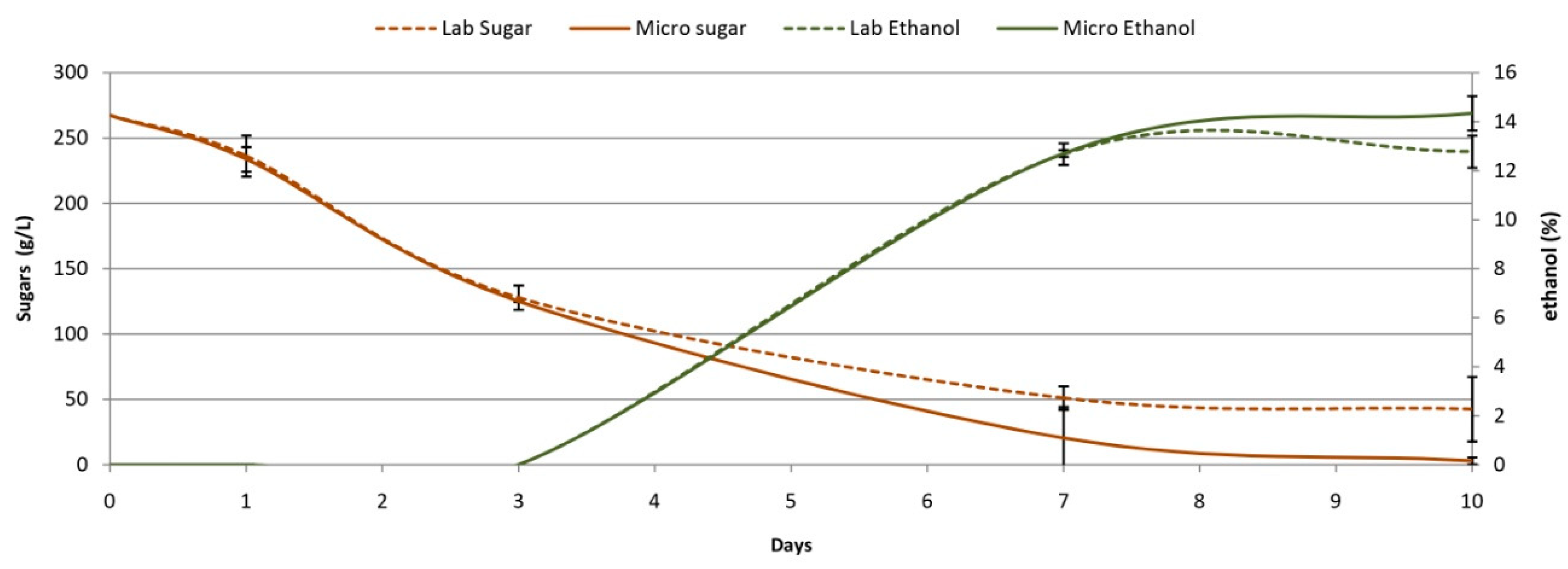
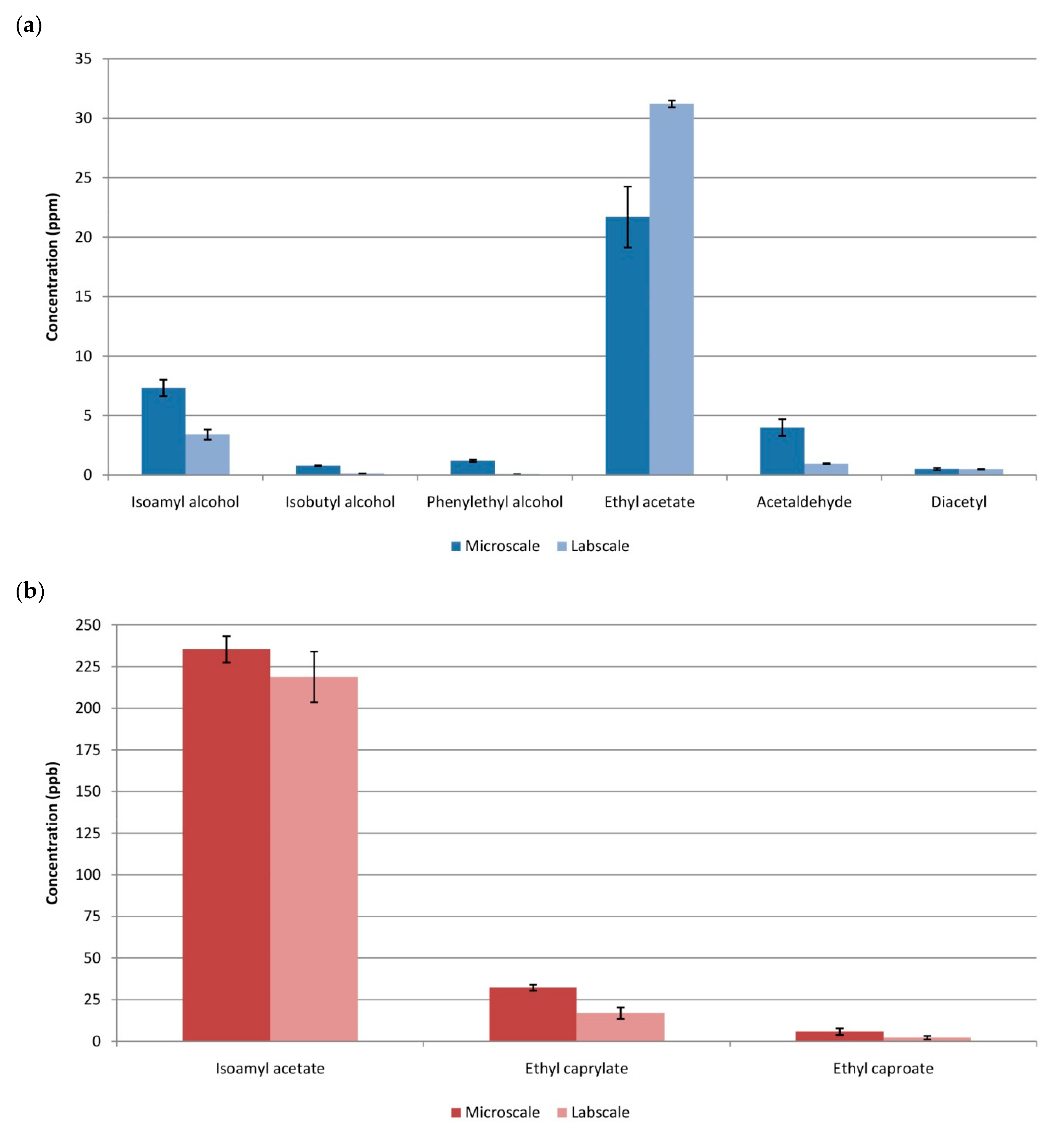
3.3. Comparison Microscale vs.Labscale
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Aslankoohi, E.; Herrera-Malaver, B.; Rezaei, M.N.; Steensels, J.; Courtin, C.M.; Verstrepen, K.J. Non-Conventional Yeast Strains Increase the Aroma Complexity of Bread. PLoS ONE 2016, 11, e0165126. [Google Scholar] [CrossRef] [PubMed]
- Varela, C. The impact of non-Saccharomyces yeasts in the production of alcoholic beverages. Appl. Microbiol. Biotechnol. 2016, 100, 9861–9874. [Google Scholar] [CrossRef] [PubMed]
- Dashko, S.; Zhou, N.; Tinta, T.; Sivilotti, P.; Lemut, M.S.; Trost, K.; Gamero, A.; Boekhout, T.; Butinar, L.; Vrhovsek, U.; et al. Use of non-conventional yeast improves the wine aroma profile of Ribolla Gialla. J. Ind. Microbiol. Biotechnol. 2015, 42, 997–1010. [Google Scholar] [CrossRef] [PubMed]
- Jood, I.; Hoff, J.W.; Setati, M.E. Evaluating fermentation characteristics of Kazachstania spp. and their potential influence on wine quality. World J. Microbiol. Biotechnol. 2017, 33, 129. [Google Scholar] [CrossRef] [PubMed]
- Varela, C.; Sengler, F.; Solomon, M.; Curtin, C. Volatile flavour profile of reduced alcohol wines fermented with the non-conventional yeast species Metschnikowia pulcherrima and Saccharomyces uvarum. Food Chem. 2016, 209, 57–64. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.-C.; Li, A.-H.; Dizy, M.; Ullah, N.; Sun, W.-X.; Tao, Y. Evaluation of aroma enhancement for “Ecolly” dry white wines by mixed inoculation of selected Rhodotorula mucilaginosa and Saccharomyces cerevisiae. Food Chem. 2017, 228, 550–559. [Google Scholar] [CrossRef]
- Capece, A.; Romaniello, R.; Siesto, G.; Romano, P. Conventional and Non-Conventional Yeasts in Beer Production. Fermentation 2018, 4, 38. [Google Scholar] [CrossRef]
- Michel, M.; Kopecká, J.; Meier-Dörnberg, T.; Zarnkow, M.; Jacob, F.; Hutzler, M. Screening for new brewing yeasts in the non-Saccharomyces sector with Torulaspora delbrueckii as model. Yeast 2016, 33, 129–144. [Google Scholar] [CrossRef]
- Rijswijck, I.M.H.; Rooijackers, J.C.M.W.; Abee, T.; Smid, E.J. Performance of non-conventional yeasts in co-culture with brewers’ yeast for steering ethanol and aroma production. Microb. Biotechnol. 2017, 10, 1591–1602. [Google Scholar] [CrossRef]
- Holt, S.; Mukherjee, V.; Lievens, B.; Verstrepen, K.J.; Thevelein, J.M. Bioflavoring by non-conventional yeasts in sequential beer fermentations. Food Microbiol. 2018, 72, 55–66. [Google Scholar] [CrossRef]
- Gutiérrez, A.; Boekhout, T.; Gojkovic, Z.; Katz, M. Evaluation of non-Saccharomyces yeasts in the fermentation of wine, beer and cider for the development of new beverages. J. Inst. Brew. 2018, 124, 389–402. [Google Scholar] [CrossRef]
- Ravasio, D.; Carlin, S.; Boekhout, T.; Groenewald, M.; Vrhovsek, U.; Walther, A.; Wendland, J. Adding Flavor to Beverages with Non-Conventional Yeasts. Fermentation 2018, 4, 15. [Google Scholar] [CrossRef]
- Swiegers, J.H.; Bartowsky, E.; Henschke, P.; Pretorius, I. Yeast and bacterial modulation of wine aroma and flavour. Aust. J. Grape Wine Res. 2005, 11, 139–173. [Google Scholar] [CrossRef]
- Michel, M.; Meier-Dörnberg, T.; Jacob, F.; Methner, F.J.; Wagner, S.; Hutzler, M. Review: Pure non-Saccharomyces starter cultures for beer fermentation with a focus on secondary metabolites and practical applications. J. Inst. Brew. 2016, 122, 569–587. [Google Scholar] [CrossRef]
- Šmogrovičová, D.; Dömény, Z. Beer volatile by-product formation at different fermentation temperature using immobilised yeasts. Process. Biochem. 1999, 34, 785–794. [Google Scholar] [CrossRef]
- Zhou, N.; Schifferdecker, A.J.; Gamero, A.; Compagno, C.; Boekhout, T.; Piškur, J.; Knecht, W. Kazachstania gamospora and Wickerhamomyces subpelliculosus: Two alternative baker’s yeasts in the modern bakery. Int. J. Food Microbiol. 2017, 250, 45–58. [Google Scholar] [CrossRef]
- Comitini, F.; Gobbi, M.; Domizio, P.; Romani, C.; Lencioni, L.; Mannazzu, I.; Ciani, M. Selected non-Saccharomyces wine yeasts in controlled multistarter fermentations with Saccharomyces cerevisiae. Food Microbiol. 2011, 28, 873–882. [Google Scholar] [CrossRef]
- Jolly, N.P.; Varela, C.; Pretorius, I.S. Not your ordinary yeast: Non-Saccharomyces yeasts in wine production uncovered. FEMS Yeast Res. 2013, 14, 215–237. [Google Scholar] [CrossRef]
- Minnaar, P.; Du Plessis, H.; Jolly, N.; Van Der Rijst, M.; Du Toit, M. Non-Saccharomyces yeast and lactic acid bacteria in co-inoculated fermentations with two Saccharomyces cerevisiae yeast strains: A strategy to improve the phenolic content of Syrah wine. Food Chem. 2019, 4, 100070. [Google Scholar] [CrossRef]
- Canonico, L.; Solomon, M.; Comitini, F.; Ciani, M.; Varela, C. Volatile profile of reduced alcohol wines fermented with selected non-Saccharomyces yeasts under different aeration conditions. Food Microbiol. 2019, 84, 103247. [Google Scholar] [CrossRef]
- Gamero, A.; Ren, X.; Lamboni, Y.; de Jong, C.; Smid, E.J.; Linnemann, A.R. Development of a low-alcoholic fermented beverage employing cashew apple Juice and non-conventional yeasts. Fermentation 2019, 5, 71. [Google Scholar] [CrossRef]
- Gamero, A.; Quintilla, R.; Groenewald, M.; Alkema, W.; Boekhout, T.; Hazelwood, L. High-throughput screening of a large collection of non-conventional yeasts reveals their potential for aroma formation in food fermentation. Food Microbiol. 2016, 60, 147–159. [Google Scholar] [CrossRef] [PubMed]
- Sabel, A.; Martens, S.; Petri, A.; König, H.; Claus, H. Wickerhamomyces anomalus AS1: A new strain with potential to improve wine aroma. Ann. Microbiol. 2013, 64, 483–491. [Google Scholar] [CrossRef]
- Gamero, A.; Tronchoni, J.; Querol, A.; Belloch, C. Production of aroma compounds by cryotolerant Saccharomyces species and hybrids at low and moderate fermentation temperatures. J. Appl. Microbiol. 2013, 114, 1405–1414. [Google Scholar] [CrossRef] [PubMed]
- Arroyo-Lopez, F.N.; Torrado, R.P.; Querol, A.; Barrio, E. Modulation of the glycerol and ethanol syntheses in the yeast Saccharomyces kudriavzevii differs from that exhibited by Saccharomyces cerevisiae and their hybrid. Food Microbiol. 2010, 27, 628–637. [Google Scholar] [CrossRef]
- Torrado, R.P.; Oliveira, B.M.; Zemančíková, J.; Sychrova, H.; Querol, A. Alternative Glycerol Balance Strategies among Saccharomyces Species in Response to Winemaking Stress. Front. Microbiol. 2016, 7, 628. [Google Scholar] [CrossRef]
- Erten, H.; Tangüler, H.; Cakiroz, H. The Effect of Pitching Rate on Fermentation and Flavour Compounds in High Gravity Brewing. J. Inst. Brew. 2007, 113, 75–79. [Google Scholar] [CrossRef]
- Verbelen, P.J.; Dekoninck, T.M.L.; Saerens, S.M.G.; Van Mulders, S.E.; Thevelein, J.M.; Delvaux, F.R. Impact of pitching rate on yeast fermentation performance and beer flavour. Appl. Microbiol. Biotechnol. 2009, 82, 155–167. [Google Scholar] [CrossRef]
- Bartowsky, E.J.; Henschke, P.A. The ‘buttery’ attribute of wine—Diacetyl—Desirability, spoilage and beyond. Int. J. Food Microbiol. 2004, 96, 235–252. [Google Scholar] [CrossRef]
- Yakobson, C. Pure Culture Fermentation Characteristics of Brettanomyces Yeast Species and Their Use in the Brewing Industry. Ph.D. Thesis, Heriot-Watt University, Edinburgh, UK, 2010. [Google Scholar]
- Liccioli, T.; Tran, T.M.T.; Cozzolino, D.; Jiranek, V.; Chambers, P.J.; Schmidt, S.A. Microvinification—How small can we go? Appl. Microbiol. Biotechnol. 2010, 89, 1621–1628. [Google Scholar] [CrossRef]
- Valero, E.; Moyano, L.; Millan, M.; Medina, M.; Ortega, J. Higher alcohols and esters production by Saccharomyces cerevisiae. Influence of the initial oxygenation of the grape must. Food Chem. 2002, 78, 57–61. [Google Scholar] [CrossRef]

| Strain | Species | Substrate | Origin |
|---|---|---|---|
| CBS 772.71 | Galactomyces geotrichum | Soil | Puerto Rico |
| CBS 2567 | Hanseniaspora guilliermondii | Juice | Israel |
| CBS 5552 | Wickerhamomyces subpelliculosus | Sugar | Unknown |
| CBS 6641 | Torulaspora microellipsoides | Plant | USA |
| CBS 7692 | Starmera caribaea | Plant | West Indies |
| CBS 8031 | Hanseniaspora vineae | Plant | Canada |
| CBS 8849 | Zygosaccharomyces kombuchaensis | Fungus | Russia |
| CBS 8860 | Barnettozyma californica | Fruit | Russia |
| CBS 9716 | Zygosaccharomyces rouxii | Insect | Germany |
| CBS 10400 | Kazachstania gamospora | Unknown | Japan |
| Compound | Aroma | ST Wine * (mg/L) | ST Beer ** (mg/L) |
|---|---|---|---|
| Vicinal diketones | |||
| Diacetyl | buttery | - | 0.10–0.15 |
| 2,3-Pentanedione | toffee | - | 0.90 |
| Aldehydes | |||
| Acetaldehyde | herbaceous, sherry, nutty, bruised apple | 100 | 20–25 |
| Higheralcohols | |||
| Isobutyl alcohol | solvent, fusel, spiritous | 40 | 100 |
| Isoamyl alcohol | harsh, nail polish, banana | 30 | 50–65 |
| Phenylethyl alcohol | floral, rose, sweet | 10 | 40 |
| Acetateesters | |||
| Ethyl acetate | nail polish, fruity | 7.50 | 30–33 |
| Isoamyl acetate | banana, pear | 0.03 | 1.60–2.00 |
| Phenylethyl acetate | flowery, rose, fruity, honey | 0.25 | 3.80 |
| Ethyl Esters | |||
| Ethyl caproate | green apple, aniseed | 0.05 | 0.23 |
| Ethyl caprylate | sour apple, sweet soap | 0.02 | 0.50–0.90 |
| Ethyl caprate | apple, floral, soap | 0.20 | 1.50 |
| Strain 1 | Strain 2 | Ratio | Sugars (g/L) | Ethanol% (v/v) |
|---|---|---|---|---|
| Lalvin T73 | 11 ± 4.4 f | 20 ± 0.0 b | ||
| CBS 772.71 | 163 ± 7.8 a–d | 13 ± 0.3 d | ||
| CBS 772.71 | Lalvin T73 | 1:1 | 63 ± 1.7 d–f | 17 ± 1.1 b,c |
| CBS 2567 | 170 ± 1.4 a–c | 11 ± 0.1 d,e | ||
| CBS 2567 | Lalvin T73 | 1:1 | 30 ± 1.3 e,f | 21 ± 0.8 a,b |
| CBS 5552 | 20 ± 2.1 f | 18 ± 1.1 b,c | ||
| CBS 5552 | Lalvin T73 | 1:1 | 32 ± 0.6 e,f | 18 ± 0.3 b,c |
| CBS 6641 | 157 ± 2.7 a–d | 12 ± 0.7 d | ||
| CBS 6641 | Lalvin T73 | 1:1 | 34 ± 1.4 e,f | 24 ± 0.2 a |
| CBS 7692 | 235 ± 20 a | nd | ||
| CBS 7692 | Lalvin T73 | 1:1 | 59 ± 7.3 c–f | 19 ± 0.5 b,c |
| CBS 8031 | 173 ± 5.7 a,b | 13 ± 0.3 d | ||
| CBS 8031 | Lalvin T73 | 1:1 | 73 ± 2.3 b–f | 18 ± 0.8 b,c |
| CBS 8849 | 135 ± 1.7 b–f | 13 ± 0.0 d | ||
| CBS 8849 | Lalvin T73 | 1:1 | 30 ± 3.2 e,f | 21 ± 1.2 a,b |
| CBS 8860 | 250 ± 6.2 a | nd | ||
| CBS 8860 | Lalvin T73 | 1:1 | 75 ± 5.4 b–f | 22 ± 0.3 a,b |
| CBS 9716 | 138 ± 24.3 a–e | 15 ± 1.0 c,d | ||
| CBS 9716 | Lalvin T73 | 1:1 | 78 ± 2.0 b–f | 22 ± 0.6 a,b |
| CBS 10400 | 170 ± 4.5 a,b | 11 ± 0.4 d,e | ||
| CBS 10400 | Lalvin T73 | 1:1 | 74 ± 2.0 b–f | 18 ± 0.1 b,c |
| Strain 1 | Strain 2 | Ratio | Isobutyl Alcohol | Isoamyl Alcohol | Phenylethyl Alcohol | Ethyl acetate | Isoamyl Acetate | Phenylethyl Acetate | Ethyl Caproate | Ethyl Caprylate | Ethyl Caprate |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Lalvin T73 | 26 a–c | 24 a–c | 33i | 3 a | 1.54bc | 1.98a | 0.55a,b | 1.43e,f | 0.57c,d | ||
| CBS 772.71 | 12 a | 23 a–c | 17f,g | 4 a–c | 0.81ab | 23.79c | 0.12a,b | 0.22a,b | 0.26a–d | ||
| CBS 772.71 | Lalvin T73 | 1:1 | 78b–f | 30a–d | 3 a,b | nd | 0.09a | 2.49a | 0.87b | 0.29a,b | 0.42a–d |
| CBS 2567 | 18 a,b | 11 a | 10b–e | 11c,d | 1.55bc | 13.35b | 0.06a | 0.10a | 0.08a | ||
| CBS 2567 | Lalvin T73 | 1:1 | 126e–g | 21 a–c | 6 a–d | nd | nd | 1.41a | 0.53a,b | 0.21a,b | 0.30a–d |
| CBS 5552 | 13 a | 19 a–c | 22g,h | 10b–d | 0.44ab | 11.88b | 0.22a,b | 1.26d–f | 0.60d | ||
| CBS 5552 | Lalvin T73 | 1:1 | 82c–g | 24 a–d | 6 a–d | nd | nd | 1.36a | 0.71a,b | 0.21a,b | 0.25a–d |
| CBS 6641 | 10 a | 16 a,b | 19g,h | 3 a,b | 0.25a | 0.95a | 0.14a,b | 0.36a–c | 0.30a–d | ||
| CBS 6641 | Lalvin T73 | 1:1 | 141g | 39b–d | 3 a | nd | nd | 2.19a | 0.78a,b | 0.23a,b | 0.40a–d |
| CBS 7692 | 14 a | 12 a | 9 a–d | 20e | 4.87d | 28.53d | 0.25a,b | 0.75b–d | 0.11a,b | ||
| CBS 7692 | Lalvin T73 | 1:1 | 76b–e | 41c,d | 3 a | nd | nd | 1.99a | 0.79a,b | 0.37a–c | 0.52a–d |
| CBS 8031 | 11 a | 22 a–c | 20g,h | 4 a,b | 0.76ab | 26.24c,d | 0.11a,b | 0.14a | 0.29a–d | ||
| CBS 8031 | Lalvin T73 | 1:1 | 75b–e | 29 a–d | 17f,g | nd | nd | 1.72a | 0.88b | 0.29a,b | 0.54b–d |
| CBS 8849 | 18 a,b | 25a–d | 25 h | 5 a–c | 0.88ab | 28.76d | 0.10a,b | 0.11a | 0.12a–c | ||
| CBS 8849 | Lalvin T73 | 1:1 | 138f,g | 26 a–d | 5 a–d | nd | nd | 2.42a | 0.67a,b | 0.22a,b | 0.29a–d |
| CBS 8860 | 20 a,b | 14 a | 11c–f | 4 a,b | 2.25c | 1.53a | 0.29a,b | 0.60a–c | 0.23a–d | ||
| CBS 8860 | Lalvin T73 | 1:1 | 125e–g | 48d | 4 a–c | nd | nd | 2.35a | 0.88b | 0.44a–c | 0.53a–d |
| CBS 9716 | 29 a–d | 25 a–d | 11d–f | 2 a | 1.56b,c | 1.49a | 0.39a,b | 1.61f | 0.69d | ||
| CBS 9716 | Lalvin T73 | 1:1 | 87d–g | 33a–d | 3 a | nd | nd | 1.85a | 0.81a,b | 0.28a,b | 0.59d |
| CBS 10400 | 10 a | 14 a | 16e–g | 14d | 3.85d | 33.71 e | 0.21a,b | 0.89c–e | 0.41a–d | ||
| CBS 10400 | Lalvin T73 | 1:1 | 76b–e | 32a–d | 7 a–d | nd | 0.06a | 2.71a | 0.83a,b | 0.29a,b | 0.58c,d |
| Strain 1 | Strain 2 | Ratio | Sugars (g/L) | Ethanol% (v/v) |
|---|---|---|---|---|
| WS34/70 | nd d | 5.9 ± 0.6 a | ||
| CBS 772.71 | WS34/70 | 5:1 | nd d | 5.6 ± 1.4 a |
| CBS 772.71 | WS34/70 | 20:1 | nd d | 6.5 ± 0.4 a |
| CBS 2567 | WS34/70 | 5:1 | 5.4 ± 1.4 a–c | 6.2 ± 1.1 a |
| CBS 2567 | WS34/70 | 20:1 | 2.4 ± 0.7 c,d | 6.0 ± 1.4 a |
| CBS 5552 | WS34/70 | 5:1 | nd d | 6.4 ± 0.3 a |
| CBS 5552 | WS34/70 | 20:1 | nd d | 5.4 ± 1.3 a |
| CBS 6641 | WS34/70 | 5:1 | 2.2 ± 0.4 c,d | 6.0 ± 0.6 a |
| CBS 6641 | WS34/70 | 20:1 | 5.2 ± 1.0 a–c | 6.4 ± 0.5 a |
| CBS 7692 | WS34/70 | 5:1 | 0.9 ± 1.2 d | 6.8 ± 0.3 a |
| CBS 7692 | WS34/70 | 20:1 | 3.5 ± 1.9 b–d | 6.2 ± 1.4 a |
| CBS 8031 | WS34/70 | 5:1 | nd d | 6.9 ± 1.2 a |
| CBS 8031 | WS34/70 | 20:1 | 1.0 ± 1.4 d | 6.2 ± 0.6 a |
| CBS 8849 | WS34/70 | 5:1 | 7.1 ± 0.6 a,b | 5.5 ± 1.2 a |
| CBS 8849 | WS34/70 | 20:1 | nd d | 6.5 ± 0.6 a |
| CBS 8860 | WS34/70 | 5:1 | nd d | 6.0 ± 0.4 a |
| CBS 8860 | WS34/70 | 20:1 | 0.8 ± 1.1 d | 6.8 ± 0.1 a |
| CBS 9716 | WS34/70 | 5:1 | 7.7 ± 0.4 a | 5.7 ± 1.1 a |
| CBS 9716 | WS34/70 | 20:1 | 3.9 ± 1.6 a–d | 6.6 ± 0.3 a |
| CBS 10400 | WS34/70 | 5:1 | nd d | 7.3 ± 0.4 a |
| CBS 10400 | WS34/70 | 20:1 | 1.6 ± 0.5 c,d | 6.6 ± 0.4 a |
| Strain 1 | Strain 2 | Ratio | Diacetyl | 2,3-Pentadione | Isobutyl Alcohol | Isoamyl Alcohol | Phenylethyl Alcohol | Ethyl Acetate | Isoamyl Acetate | Phenylethyl Acetate | Ethyl Caproate | Ethyl Caprylate | Ethyl Caprate |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| WS34/70 | 0.14a | 0.02 b,c | 32 d | 74e | 27 d–f | 27 a | 0.52 a | 0.84 a–e | 0.04 d | 0.12 g | 0.06 e,f | ||
| CBS 772.71 | WS34/70 | 5:1 | 0.13a | 0.02 a,b | 29 a–d | 67a–e | 23 a–e | 23 a | 0.41 a | 0.86 a–e | 0.04 c,d | 0.11 f,g | 0.05 c–f |
| CBS 772.71 | WS34/70 | 20:1 | 0.41c | 0.10 h | 29 a–d | 68b–e | 20 a–c | 20 a | 0.37 a | 1.22 e–g | 0.03 a–d | 0.10 e–g | 0.04 b–e |
| CBS 2567 | WS34/70 | 5:1 | 0.65d | 0.06 f,g | 27 a–c | 62a–d | 19 a–c | 19 a | 0.35 a | 1.04 b–f | 0.03 a–c | 0.03 a–c | 0.02 a–d |
| CBS 2567 | WS34/70 | 20:1 | 0.62d | 0.05 e,f | 25 a | 60a,b | 18 a,b | 18 a | 0.38 a | 1.53 f,g | 0.02 a,b | 0.02 a | 0.01 a |
| CBS 5552 | WS34/70 | 5:1 | 0.30b | 0.04 d | 27 a–c | 64a–d | 34 g,h | 34a | 0.45 a | 0.59 a,b | 0.03 b–d | 0.09 d–g | 0.02 a–d |
| CBS 5552 | WS34/70 | 20:1 | 0.08 a | 0.01 a,b | 30 b–d | 63a–d | 34 g,h | 34a | 0.33 a | 1.68 g | 0.03 a–c | 0.04 a–d | 0.01 a,b |
| CBS 6641 | WS34/70 | 5:1 | 0.09 a | 0.01 a,b | 28 a–d | 67a–e | 16 a | 16 a | 0.62 a | 0.61 a–c | 0.04 c,d | 0.12 g,h | 0.05 d–f |
| CBS 6641 | WS34/70 | 20:1 | 0.33b,c | 0.03 d | 28 a–d | 66a–e | 19 a–c | 19 a | 0.47 a | 1.20 d–g | 0.03 a–c | 0.03 a–c | 0.01 a,b |
| CBS 7692 | WS34/70 | 5:1 | 0.41c | 0.04 d,e | 25 a,b | 62a–d | 25 b–e | 25 a | 0.36 a | 1.01 b–e | 0.03 a–d | 0.09 e–g | 0.03 a–e |
| CBS 7692 | WS34/70 | 20:1 | 0.60d | 0.07 g | 27 a–c | 61a–c | 19 a,b | 19 a | 0.53 a | 1.12 c–f | 0.03 a–d | 0.04 a–c | 0.02 a–d |
| CBS 8031 | WS34/70 | 5:1 | 0.08 a | 0.02 a,b | 26 a–c | 60a,b | 29 e–g | 29 a | 0.43 a | 2.25 h | 0.03 a–c | 0.02 ab | 0.01 abc |
| CBS 8031 | WS34/70 | 20:1 | 0.30b | 0.03 c,d | 26 a–c | 60a,b | 32 f–h | 32a | 0.23 a | 3.03 i | 0.02 a | 0.01 a | 0.01 a,b |
| CBS 8849 | WS34/70 | 5:1 | 0.08 a | 0.02 a,b | 30 b–d | 68b–e | 27 c–f | 27 a | 0.44 a | 0.72 a–e | 0.04 c,d | 0.10 e–g | 0.04 b–e |
| CBS 8849 | WS34/70 | 20:1 | 0.56d | 0.05 f | 29 a–d | 69c–e | 22 a–d | 22 a | 0.31 a | 0.43 a | 0.03 a–d | 0.04 a–c | 0.01 a,b |
| CBS 8860 | WS34/70 | 5:1 | 0.27b | 0.03 d | 29 a–d | 69c–e | 26 c–f | 26 a | 0.37 a | 0.74 a–e | 0.04 d | 0.16 h | 0.07 f |
| CBS 8860 | WS34/70 | 20:1 | 0.42c | 0.11 h | 31 c,d | 68b–e | 24 b–e | 24 a | 0.43 a | 0.80 a–e | 0.03 b–d | 0.10 e–g | 0.06 e,f |
| CBS 9716 | WS34/70 | 5:1 | 0.05 a | 0.01 a,b | 29 a–d | 70d,e | 35 g,h | 35a | 0.40 a | 0.60 a,b | 0.04 c,d | 0.10 e–g | 0.02 a–d |
| CBS 9716 | WS34/70 | 20:1 | 0.10a | 0.02 a,b | 28 a–d | 69b–e | 38 h | 38a | 0.38 a | 0.66 a–d | 0.03 a–d | 0.07 b–e | 0.01 a,b |
| CBS 10400 | WS34/70 | 5:1 | 0.10a | 0.02 a,b | 27 a–d | 61a–d | 20 a–c | 20 a | 0.24 a | 1.06 b–f | 0.03 a–d | 0.06 b–e | 0.05 d–f |
| CBS 10400 | WS34/70 | 20:1 | 0.05 a | 0.01 a | 25 a,b | 59a | 25 b–f | 25 a | 0.28 a | 1.66 g | 0.03 a–d | 0.07 c–f | 0.12 g |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gamero, A.; Dijkstra, A.; Smit, B.; de Jong, C. Aromatic Potential of Diverse Non-Conventional Yeast Species for Winemaking and Brewing. Fermentation 2020, 6, 50. https://doi.org/10.3390/fermentation6020050
Gamero A, Dijkstra A, Smit B, de Jong C. Aromatic Potential of Diverse Non-Conventional Yeast Species for Winemaking and Brewing. Fermentation. 2020; 6(2):50. https://doi.org/10.3390/fermentation6020050
Chicago/Turabian StyleGamero, Amparo, Annereinou Dijkstra, Bart Smit, and Catrienus de Jong. 2020. "Aromatic Potential of Diverse Non-Conventional Yeast Species for Winemaking and Brewing" Fermentation 6, no. 2: 50. https://doi.org/10.3390/fermentation6020050
APA StyleGamero, A., Dijkstra, A., Smit, B., & de Jong, C. (2020). Aromatic Potential of Diverse Non-Conventional Yeast Species for Winemaking and Brewing. Fermentation, 6(2), 50. https://doi.org/10.3390/fermentation6020050






