Food-Grade Oleogels: Trends in Analysis, Characterization, and Applicability
Abstract
1. Introduction
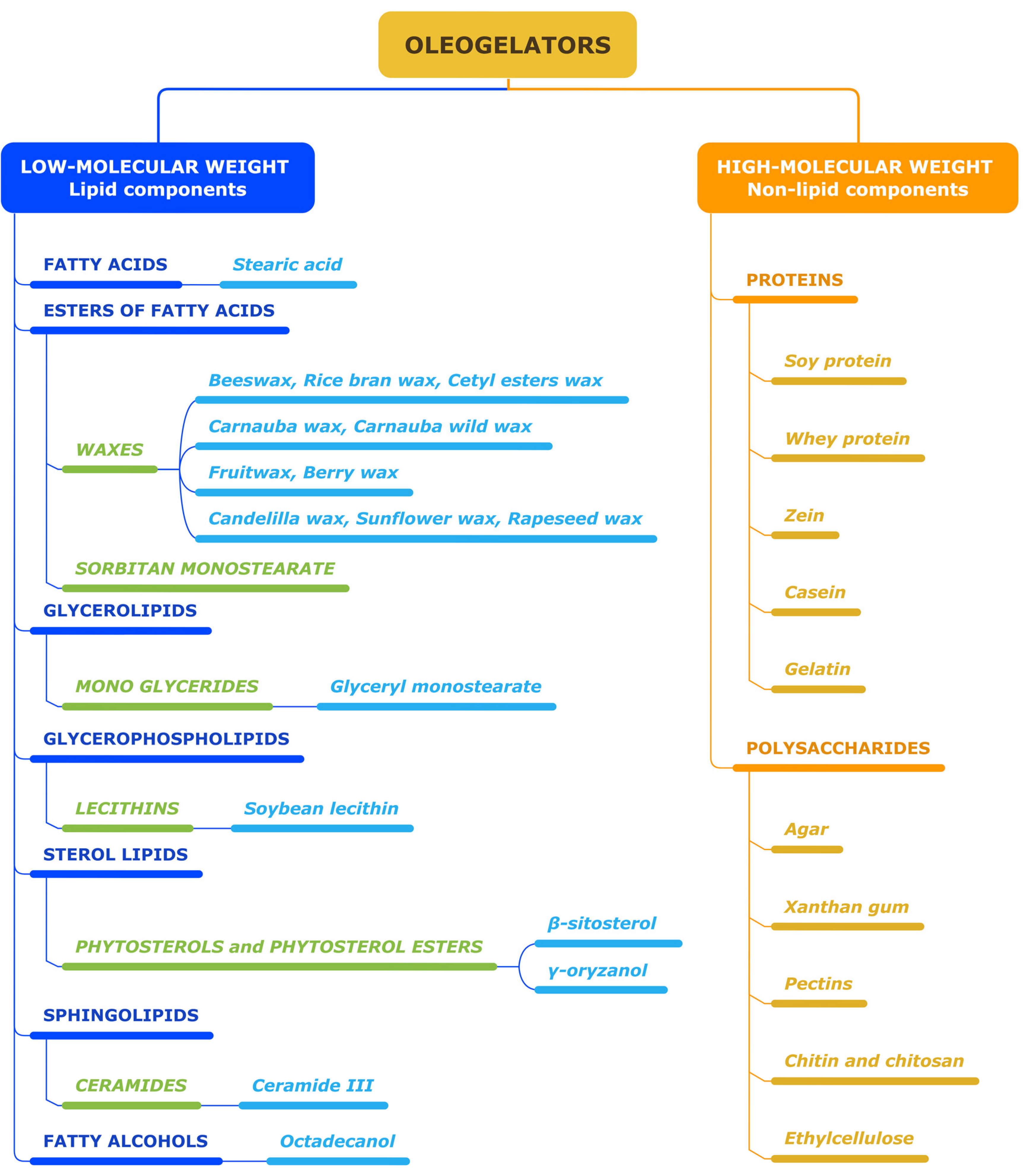
2. Oleogelators
2.1. Low-Molecular-Weight Oleogelators
2.1.1. Waxes
Beeswax
Carnauba Wax
Sunflower Wax
Rice Bran Wax
2.1.2. Phytosterols and Phytosterol Esters
β-Sitosterol
γ-Oryzanol
2.1.3. Fatty Acids and Monoglycerides
2.2. High-Molecular-Weight Oleogelators
2.2.1. Proteins
Soy Protein
Whey Protein
Zein
Gelatin
2.2.2. Polysaccharides
Agar
Xanthan Gum
Pectins
Chitin and Chitosan
Ethylcellulose
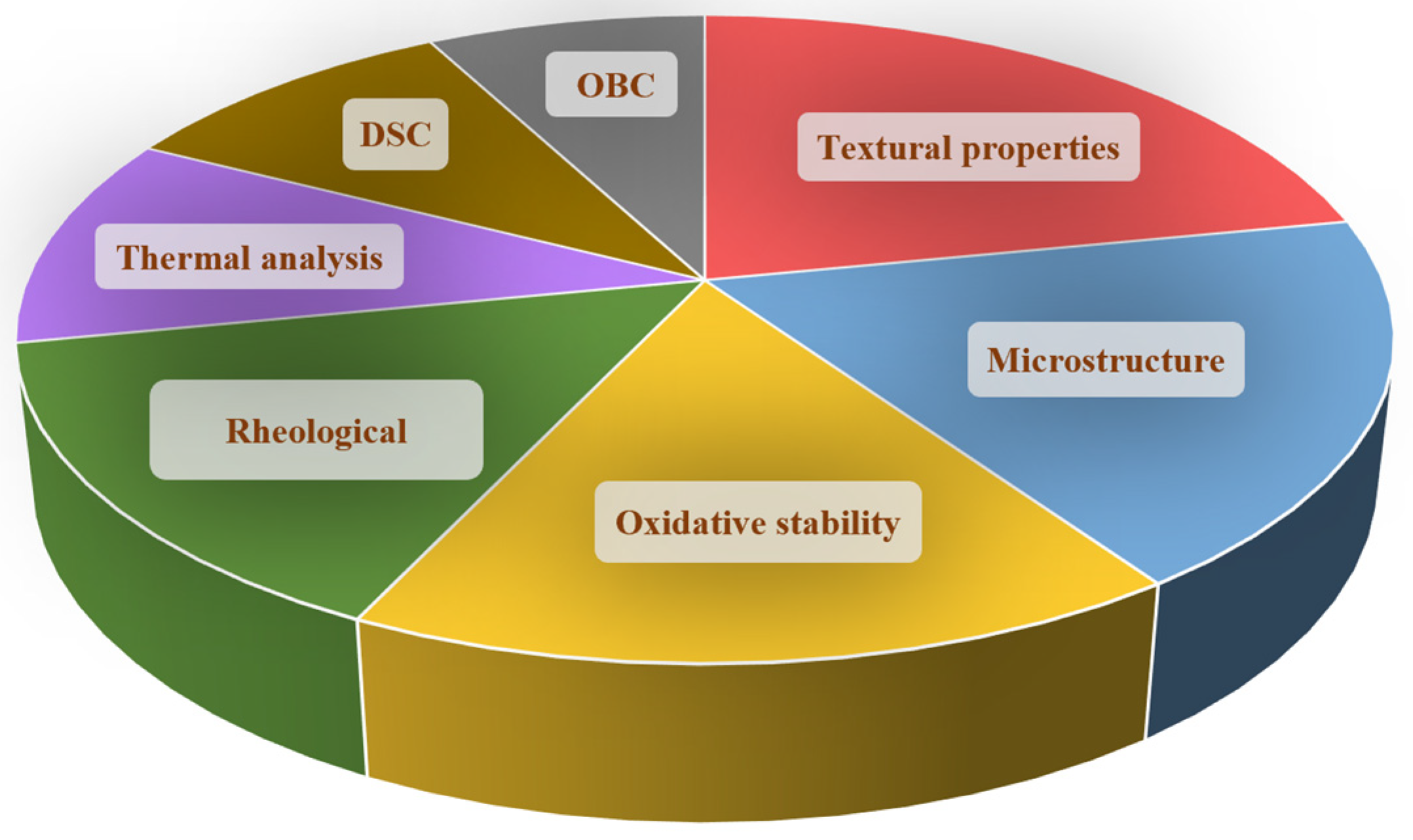
3. Food-Grade Oleogels: Analysis and Characterization
3.1. Microstructure
3.2. Rheological Properties
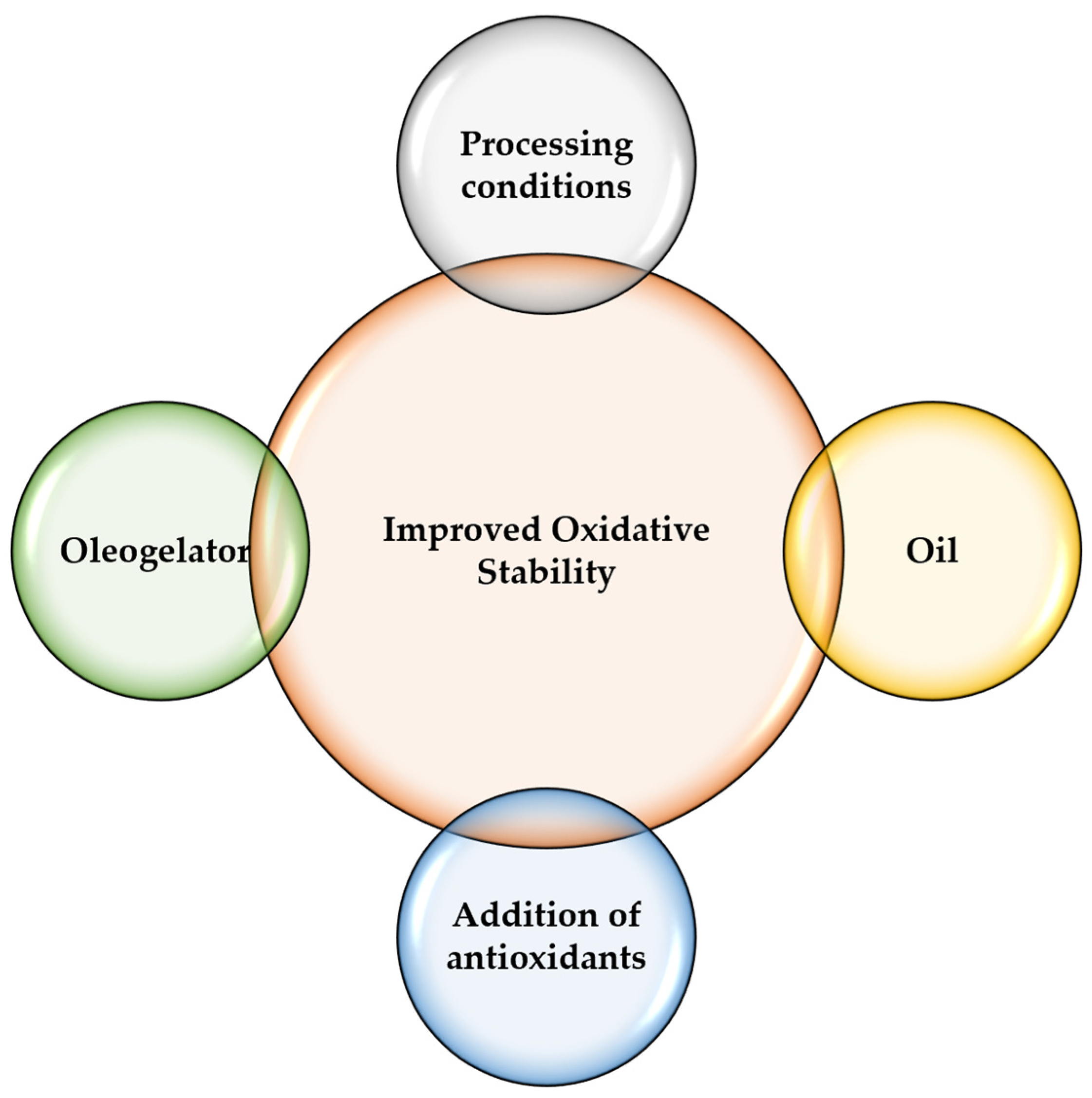
3.3. Oxidative Stability
3.4. Textural Properties
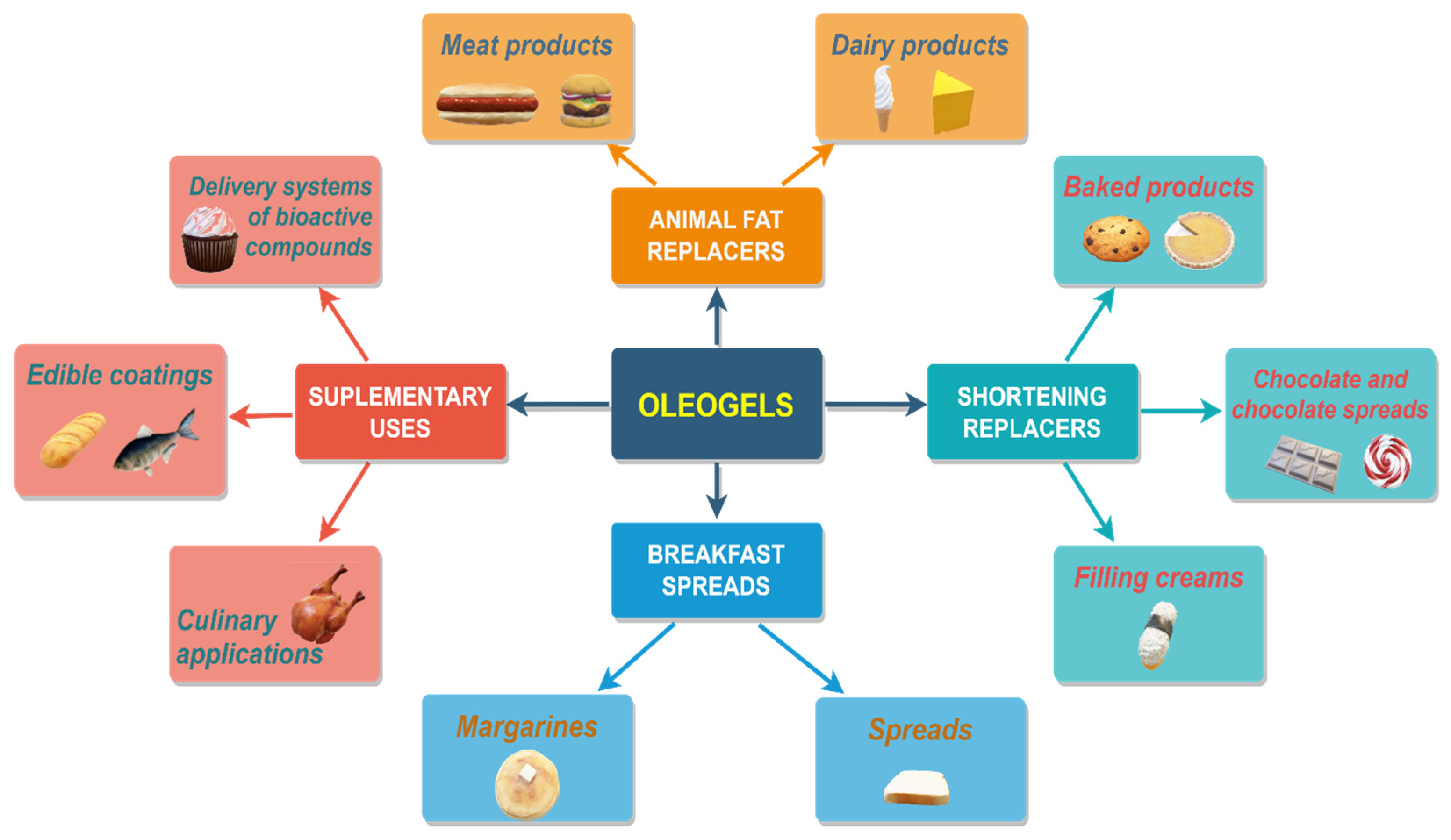
4. Applicability of Oleogels in Food
4.1. Oleogels Used as Animal Fat Replacers
4.1.1. Meat Products
4.1.2. Dairy Products
Ice Cream
Vegan Creams
Imitation Cheese
4.2. Oleogels Used as Shortening Replacers
4.2.1. Baked Products
4.2.2. Chocolate and Chocolate Spreads
4.2.3. Filling Creams
4.3. Oleogels Used in Breakfast Spread Developing
Spreads and Margarines
4.4. Supplementary Uses of Oleogels in Foods
4.4.1. Edible Coatings
4.4.2. Delivery Systems of Bioactive Compounds
4.4.3. Culinary Applications
5. Conclusions and Future Perspectives
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Jeong, S.; Lee, S.; Oh, I. Development of Antioxidant-Fortified Oleogel and Its Application as a Solid Fat Replacer to Muffin. Foods 2021, 10, 3059. [Google Scholar] [CrossRef] [PubMed]
- Pinto, T.C.; Martins, A.J.; Pastrana, L.; Pereira, M.C.; Cerqueira, M.A. Oleogel-Based Systems for the Delivery of Bioactive Compounds in Foods. Gels 2021, 7, 86. [Google Scholar] [CrossRef] [PubMed]
- Alizadeh, L.; Abdolmaleki, K.; Nayebzadeh, K.; Bahmaei, M. Characterization of sodium caseinate/Hydroxypropyl methylcellulose concentrated emulsions: Effect of mixing ratio, concentration and wax addition. Int. J. Biol. Macromol. 2019, 128, 796–803. [Google Scholar] [CrossRef] [PubMed]
- Brykczynski, H.; Hetzer, B.; Floter, E. An Attempt to Relate Oleogel Properties to Wax Ester Chemical Structures. Gels 2022, 8, 579. [Google Scholar] [CrossRef]
- Buitimea-Cantua, G.V.; Serna-Saldivar, S.O.; Perez-Carrillo, E.; Silva, T.J.; Barrera-Arellano, D.; Buitimea-Cantua, N.E. Effect of quality of carnauba wax (Copernica cerifera) on microstructure, textural, and rheological properties of soybean oil-based organogels. LWT Food Sci. Technol. 2021, 136, 110267. [Google Scholar] [CrossRef]
- De Freitas, C.A.S.; de Sousa, P.H.M.; Soares, D.J.; da Silva, J.Y.G.; Benjamin, S.R.; Guedes, M.I.F. Carnauba wax uses in food—A review. Food Chem. 2019, 291, 38–48. [Google Scholar] [CrossRef]
- Holey, S.A.; Sekhar, K.P.C.; Mishra, S.S.; Kanjilal, S.; Nayak, R.R. Effect of oil unsaturation and wax composition on stability, properties and food applicability of oleogels. J. Am. Oil Chem. Soc. 2021, 98, 1189–1203. [Google Scholar] [CrossRef]
- Mattice, K.D.; Marangoni, A.G. New Insights into Wax Crystal Networks in Oleogels. In Edible Oil Structuring: Concepts, Methods and Applications; Patel, A.R., Ed.; The Royal Society of Chemistry: Cambridge, UK, 2017. [Google Scholar] [CrossRef]
- Noonim, P.; Rajasekaran, B.; Venkatachalam, K. Effect of Palm Oil-Carnauba Wax Oleogel That Processed with Ultrasonication on the Physicochemical Properties of Salted Duck Egg White Fortified Instant Noodles. Gels 2022, 8, 487. [Google Scholar] [CrossRef]
- Zhang, R.F.; Zhang, T.; Hu, M.Y.; Xue, Y.; Xue, C.H. Effects of oleogels prepared with fish oil and beeswax on the gelation behaviors of protein recovered from Alaska Pollock. LWT Food Sci. Technol. 2021, 137, 110423. [Google Scholar] [CrossRef]
- Patel, A.R.; Cludts, N.; Bin Sintang, M.D.; Lesaffer, A.; Dewettinck, K. Edible oleogels based on water soluble food polymers: Preparation, characterization and potential application. Food Funct. 2014, 5, 2833–2841. [Google Scholar] [CrossRef]
- Piekarska, K.; Sikora, M.; Owczarek, M.; Jozwik-Pruska, J.; Wisniewska-Wrona, M. Chitin and Chitosan as Polymers of the Future-Obtaining, Modification, Life Cycle Assessment and Main Directions of Application. Polymers 2023, 15, 793. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.Y.; Zhou, P.W.; Song, C.F.; Jin, G.Y.; Wei, L.J. An approach to manufacturing heat-stable and bloom-resistant chocolate by the combination of oleogel and sweeteners. J. Food Eng. 2022, 330, 111064. [Google Scholar] [CrossRef]
- Wang, X.; Wang, S.; Nan, Y.; Liu, G. Production of Margarines Rich in Unsaturated Fatty Acids Using Oxidative-stable Vitamin C-Loaded Oleogel. J. Oleo Sci. 2021, 70, 1059–1068. [Google Scholar] [CrossRef] [PubMed]
- Contreras-Ramirez, J.I.; Acosta-Gurrola, E.; Rosas-Flores, W.; Gallegos-Infante, J.A.; Toro-Vazquez, J.F. Microstructuring process in oleogels formulated with vegetable oils and monoglycerides: A comparison of non-isothermal nucleation kinetics by spectrophotometric and DSC analysis. J. Am. Oil Chem. Soc. 2023, 12685. [Google Scholar] [CrossRef]
- Contreras-Ramirez, J.I.; Patel, A.R.; Gallegos-Infante, J.A.; Toro-Vazquez, J.F.; Perez-Martinez, J.D.; Rosas-Flores, W.; Gonzalez-Laredo, R.F. Organogel-Based Emulsified Systems, Food Applications, Microstructural and Rheological Features—A Review. Biointerface Res. Appl. Chem. 2022, 12, 1601–1627. [Google Scholar] [CrossRef]
- Du, L.Y.; Jiang, Q.B.; Li, S.Y.; Zhou, Q.; Tan, Y.Q.; Meng, Z. Microstructure evolution and partial coalescence in the whipping process of oleofoams stabilized by monoglycerides. Food Hydrocoll. 2021, 112, 106245. [Google Scholar] [CrossRef]
- Giacintucci, V.; Di Mattia, C.D.; Sacchetti, G.; Flamminii, F.; Gravelle, A.J.; Baylis, B.; Dutcher, J.R.; Marangoni, A.G.; Pittia, P. Ethylcellulose oleogels with extra virgin olive oil: The role of oil minor components on microstructure and mechanical strength. Food Hydrocoll. 2018, 84, 508–514. [Google Scholar] [CrossRef]
- Gu, X.Y.; Du, L.Y.; Meng, Z. Comparative study of natural wax-based W/O emulsion gels: Microstructure and macroscopic properties. Food Res. Int. 2023, 165, 112509. [Google Scholar] [CrossRef]
- Martin-Alfonso, J.E.; Franco, J.M. Influence of polymer reprocessing cycles on the microstructure and rheological behavior of polypropylene/mineral oil oleogels. Polym. Test. 2015, 45, 12–19. [Google Scholar] [CrossRef]
- Matheson, A.B.; Koutsos, V.; Dalkas, G.; Euston, S.; Clegg, P. The microstructure of β-sitosterol: γ-oryzanol edible organogels. Langmuir 2017, 33, 4537–4542. [Google Scholar] [CrossRef]
- Saffold, A.C.; Acevedo, N.C. Development of Novel Rice Bran Wax/Gelatin-Based Biphasic Edible Gels and Characterization of their Microstructural, Thermal, and Mechanical Properties. Food Bioprocess Technol. 2021, 14, 2219–2230. [Google Scholar] [CrossRef]
- Tavernier, I.; Doan, C.D.; Van der Meeren, P.; Heyman, B.; Dewettinck, K. The Potential of Waxes to Alter the Microstructural Properties of Emulsion-Templated Oleogels. Eur. J. Lipid Sci. Technol. 2018, 120, 1700393. [Google Scholar] [CrossRef]
- Wijarnprecha, K.; de Vries, A.; Santiwattana, P.; Sonwai, S.; Rousseau, D. Microstructure and rheology of oleogel-stabilized water-in-oil emulsions containing crystal-stabilized droplets as active fillers. LWT Food Sci. Technol. 2019, 115, 108058. [Google Scholar] [CrossRef]
- De Godoi, K.R.R.; Basso, R.C.; Ming, C.C.; da Selva, A.A.; Cardoso, L.P.; Ribeiro, A.P.B. Crystallization, microstructure and polymorphic properties of soybean oil organogels in a hybrid structuring system. Food Res. Int. 2020, 137, 109460. [Google Scholar] [CrossRef] [PubMed]
- Doan, C.D.; Van de Walle, D.; Dewettinck, K.; Patel, A.R. Evaluating the Oil-Gelling Properties of Natural Waxes in Rice Bran Oil: Rheological, Thermal, and Microstructural Study. J. Am. Oil Chem. Soc. 2015, 92, 801–811. [Google Scholar] [CrossRef]
- Gomez-Mascaraque, L.G.; Tran, C.; O’Callaghan, T.; Hogan, S.A. Use of confocal Raman imaging to understand the microstructure of anhydrous milk fat-based oleogels. Food Struct. 2021, 30, 100228. [Google Scholar] [CrossRef]
- Valoppi, F.; Salmi, A.; Ratilainen, M.; Puranen, T.; Tommiska, O.; Hyvonen, J.; Heikkila, J.; Haeggstrom, E. Ultrasonic standing wave chamber for engineering microstructures of water- and lipid-based materials. Eng. Res. Express 2021, 3, 016002. [Google Scholar] [CrossRef]
- Airoldi, R.; da Silva, T.L.T.; Ract, J.N.R.; Foguel, A.; Colleran, H.L.; Ibrahim, S.A.; da Silva, R.C. Potential use of carnauba wax oleogel to replace saturated fat in ice cream. J. Am. Oil Chem. Soc. 2022, 99, 1085–1099. [Google Scholar] [CrossRef]
- Espert, M.; Wang, Q.; Sanz, T.; Salvador, A. Sunflower Oil-based Oleogel as Fat Replacer in Croissants: Textural and Sensory Characterisation. Food Bioprocess Technol. 2023. [Google Scholar] [CrossRef]
- Franco, D.; Martins, A.J.; Lopez-Pedrouso, M.; Cerqueira, M.A.; Purrinos, L.; Pastrana, L.M.; Vicente, A.A.; Zapata, C.; Lorenzo, J.M. Evaluation of linseed oil oleogels to partially replace pork backfat in fermented sausages. J. Sci. Food Agric. 2020, 100, 218–224. [Google Scholar] [CrossRef]
- Gao, Y.; Li, M.; Zhang, L.; Wang, Z.; Yu, Q.; Han, L. Preparation of rapeseed oil oleogels based on beeswax and its application in beef heart patties to replace animal fat. LWT 2021, 149, 111986. [Google Scholar] [CrossRef]
- Gómez-Estaca, J.; Herrero, A.M.; Herranz, B.; Álvarez, M.D.; Jiménez-Colmenero, F.; Cofrades, S. Characterization of ethyl cellulose and beeswax oleogels and their suitability as fat replacers in healthier lipid pâtés development. Food Hydrocoll. 2019, 87, 960–969. [Google Scholar] [CrossRef]
- Li, Q.N.; Zhang, J.Y.; Zhang, G.J.; Xu, B.C. L-Lysine-Based Gelators for the Formation of Oleogels in Four Vegetable Oils. Molecules 2022, 27, 1369. [Google Scholar] [CrossRef] [PubMed]
- Siraj, N.; Shabbir, M.A.; Ahmad, T.; Sajjad, A.; Khan, M.R.; Khan, M.I.; Butt, M.S. Organogelators as a Saturated Fat Replacer for Structuring Edible Oils. Int. J. Food Prop. 2015, 18, 1973–1989. [Google Scholar] [CrossRef]
- Wolfer, T.L.; Acevedo, N.C.; Prusa, K.J.; Sebranek, J.G.; Tarte, R. Replacement of pork fat in frankfurter-type sausages by soybean oil oleogels structured with rice bran wax. Meat Sci. 2018, 145, 352–362. [Google Scholar] [CrossRef] [PubMed]
- Babu, A.; Sivakumar, G.; Das, A.; Bharti, D.; Qureshi, D.; Habibullah, S.K.; Satheesan, A.; Mohanty, B.; Pal, K.; Maji, S. Preparation and Characterization of Novel Oleogels Using Jasmine Floral Wax and Wheat Germ Oil for Oral Delivery of Curcumin. ACS Omega 2022, 7, 30125–30136. [Google Scholar] [CrossRef]
- Behera, B.; Singh, V.K.; Kulanthaivel, S.; Bhattacharya, M.K.; Paramanik, K.; Banerjee, I.; Pal, K. Physical and mechanical properties of sunflower oil and synthetic polymers based bigels for the delivery of nitroimidazole antibiotic—A therapeutic approach for controlled drug delivery. Eur. Polym. J. 2015, 64, 253–264. [Google Scholar] [CrossRef]
- Kanelaki, A.; Zampouni, K.; Mourtzinos, I.; Katsanidis, E. Hydrogels, Oleogels and Bigels as Edible Coatings of Sardine Fillets and Delivery Systems of Rosemary Extract. Gels 2022, 8, 660. [Google Scholar] [CrossRef]
- Kavimughil, M.; Leena, M.M.; Moses, J.A.; Anandharamakrishnan, C. 3D printed MCT oleogel as a co-delivery carrier for curcumin and resveratrol. Biomaterials 2022, 287, 121616. [Google Scholar] [CrossRef]
- Kodela, S.P.; Pandey, P.M.; Nayak, S.K.; Uvanesh, K.; Anis, A.; Pal, K. Novel agar–stearyl alcohol oleogel-based bigels as structured delivery vehicles. Int. J. Polym. Mater. Polym. Biomater. 2017, 66, 669–678. [Google Scholar] [CrossRef]
- Lupi, F.R.; Gabriele, D.; Baldino, N.; Mijovic, P.; Parisi, O.I.; Puoci, F. Olive oil/policosanol organogels for nutraceutical and drug delivery purposes. Food Funct. 2013, 4, 1512–1520. [Google Scholar] [CrossRef] [PubMed]
- Xu, Y.; Sun, H.; Lv, J.; Wang, Y.; Zhang, Y.; Wang, F. Effects of polysaccharide thickening agent on the preparation of walnut oil oleogels based on methylcellulose: Characterization and delivery of curcumin. Int. J. Biol. Macromol. 2023, 232, 123291. [Google Scholar] [CrossRef]
- Zhao, W.; Wei, Z.; Xue, C.; Meng, Y. Development of food-grade oleogel via the aerogel-templated method: Oxidation stability, astaxanthin delivery and emulsifying application. Food Hydrocoll. 2023, 134, 108058. [Google Scholar] [CrossRef]
- Issara, U. Improvement of Thai Sweet Sausage (Goon Chiang) Properties by Oleogel Made of Rice Bran Wax and Rice Bran Oil: A Textural, Sensorial, and Nutritional Aspect. In Proceedings of the Sriwijaya Conference on Sustainable Environment, Agriculture and Farming System, Palembang, Indonesia, 29 September 2021; IOP: Bristol, UK, 2022; Volume 995. [Google Scholar] [CrossRef]
- Fattore, E.; Massa, E. Dietary fats and cardiovascular health: A summary of the scientific evidence and current debate. Int. J. Food Sci. Nutr. 2018, 69, 916–927. [Google Scholar] [CrossRef] [PubMed]
- Teicholz, N. A short history of saturated fat: The making and unmaking of a scientific consensus. Curr. Opin. Endocrinol. Diabetes Obes. 2023, 30, 65–71. [Google Scholar] [CrossRef] [PubMed]
- Demirkesen, I.; Mert, B. Recent developments of oleogel utilizations in bakery products. Crit. Rev. Food Sci. Nutr. 2020, 60, 2460–2479. [Google Scholar] [CrossRef]
- Moon, K.; Choi, K.O.; Jeong, S.; Kim, Y.W.; Lee, S. Solid Fat Replacement with Canola Oil-Carnauba Wax Oleogels for Dairy-Free Imitation Cheese Low in Saturated Fat. Foods 2021, 10, 1351. [Google Scholar] [CrossRef] [PubMed]
- Yilmaz, E.; Toksoz, B. Flaxseed oil-wax oleogels replacement for tallowfat in sucuk samples provided higher concentrations of polyunsaturated fatty acids and aromatic volatiles. Meat Sci. 2022, 192, 108875. [Google Scholar] [CrossRef]
- Liu, J.; Willför, S.; Xu, C. A review of bioactive plant polysaccharides: Biological activities, functionalization, and biomedical applications. Bioact. Carbohydr. Diet. Fibre 2015, 5, 31–61. [Google Scholar] [CrossRef]
- Nieri, P.; Carpi, S.; Esposito, R.; Costantini, M.; Zupo, V. Bioactive Molecules from Marine Diatoms and Their Value for the Nutraceutical Industry. Nutrients 2023, 15, 464. [Google Scholar] [CrossRef]
- Wang, S.; Chen, K.; Liu, G. Monoglyceride oleogels for lipophilic bioactive delivery—Influence of self-assembled structures on stability and in vitro bioaccessibility of astaxanthin. Food Chem. 2022, 375, 131880. [Google Scholar] [CrossRef] [PubMed]
- Aliasl Khiabani, A.; Tabibiazar, M.; Roufegarinejad, L.; Hamishehkar, H.; Alizadeh, A. Preparation and characterization of carnauba wax/adipic acid oleogel: A new reinforced oleogel for application in cake and beef burger. Food Chem. 2020, 333, 127446. [Google Scholar] [CrossRef] [PubMed]
- Brito, G.B.; Peixoto, V.O.D.; Martins, M.T.; Rosario, D.K.A.; Ract, J.N.; Conte, C.A.; Torres, A.G.; Castelo-Branco, V.N. Development of chitosan-based oleogels via crosslinking with vanillin using an emulsion templated approach: Structural characterization and their application as fat-replacer. Food Struct. 2022, 32, 100264. [Google Scholar] [CrossRef]
- Ghorghi, Z.B.; Yeganehzad, S.; Hesarinejad, M.A.; Faezian, A.; Kutsenkova, V.; Gao, Z.; Nishinari, K.; Nepovinnykh, N. Fabrication of novel hybrid gel based on beeswax oleogel: Application in the compound chocolate formulation. Food Hydrocolloids 2023, 140, 108599. [Google Scholar] [CrossRef]
- Choi, K.O.; Hwang, H.S.; Jeong, S.; Kim, S.; Lee, S. The thermal, rheological, and structural characterization of grapeseed oil oleogels structured with binary blends of oleogelator. Food Sci. 2020, 85, 3432–3441. [Google Scholar] [CrossRef]
- Davidovich-Pinhas, M.; Barbut, S.; Marangoni, A.G. Development, Characterization, and Utilization of Food-Grade Polymer Oleogels. Annu. Rev. Food Sci. Technol. 2016, 7, 65–91. [Google Scholar] [CrossRef]
- Garcia-Ortega, M.L.; Toro-Vazquez, J.F.; Ghosh, S. Development and characterization of structured water-in-oil emulsions with ethyl cellulose oleogels. Food Res. Int. 2021, 150, 15. [Google Scholar] [CrossRef]
- Li, J.X.; Guo, R.H.; Wang, M.X.; Bi, Y.L.; Zhang, H.; Xu, X.B. Development and Characterization of Compound Oleogels Based on Monoglycerides and Edible Waxes. ACS Food Sci. Technol. 2022, 2, 302–314. [Google Scholar] [CrossRef]
- Luo, S.Z.; Hu, X.F.; Jia, Y.J.; Pan, L.H.; Zheng, Z.; Zhao, Y.Y.; Mu, D.D.; Zhong, X.Y.; Jiang, S.T. Camellia oil-based oleogels structuring with tea polyphenol-palmitate particles and citrus pectin by emulsion-templated method: Preparation, characterization and potential application. Food Hydrocoll. 2019, 95, 76–87. [Google Scholar] [CrossRef]
- Martin-Alfonso, J.; Franco, J.M. Formulation and characterization of oleogels based on high-oleic sunflower oil and ethylene vinyl acetate copolymer/polypropylene blends. Polym. Eng. Sci. 2015, 55, 1429–1440. [Google Scholar] [CrossRef]
- Patel, A.R.; Schatteman, D.; De Vos, W.H.; Lesaffer, A.; Dewettinck, K. Preparation and rheological characterization of shellac oleogels and oleogel-based emulsions. J. Colloid Interface Sci. 2013, 411, 114–121. [Google Scholar] [CrossRef] [PubMed]
- Sun, H.; Xu, J.; Lu, X.Z.; Xu, Y.Y.; Regenstein, J.M.; Zhang, Y.; Wang, F.J. Development and characterization of monoglyceride oleogels prepared with crude and refined walnut oil. LWT-Food Sci. Technol. 2022, 154, 112769. [Google Scholar] [CrossRef]
- Sun, P.; Xia, B.; Ni, Z.J.; Wang, Y.; Elam, E.; Thakur, K.; Ma, Y.L.; Wei, Z.J. Characterization of functional chocolate formulated using oleogels derived from beta-sitosterol with gamma-oryzanol/lecithin/stearic acid. Food Chem. 2021, 360, 130017. [Google Scholar] [CrossRef]
- Tsung, K.L.; Ilavsky, J.; Padua, G.W. Formation and Characterization of Zein-Based Oleogels. J. Agric. Food Chem. 2020, 68, 13276–13281. [Google Scholar] [CrossRef]
- Uslu, E.K.; Yilmaz, E. Preparation and characterization of oleogels with tallow and partially hydrolyzed tallow as organogelators. Grasas Aceites 2021, 72, 1192. [Google Scholar] [CrossRef]
- Doan, C.D.; To, C.M.; De Vrieze, M.; Lynen, F.; Danthine, S.; Brown, A.; Dewettinck, K.; Patel, A.R. Chemical profiling of the major components in natural waxes to elucidate their role in liquid oil structuring. Food Chem. 2017, 214, 717–725. [Google Scholar] [CrossRef]
- Gupta, G.; Anjali, K. Environmentally Friendly Beeswax: Properties, Composition, Adulteration, and its Therapeutic Benefits. In Proceedings of the International Conference on Advance Earth Sciences & Foundation Engineering, Mohali, India, 23–24 June 2022; IOP: Bristol, UK, 2023; Volume 1110. [Google Scholar] [CrossRef]
- Mandu, C.C.; Barrera-Arellano, D.; Santana, M.H.A.; Fernandes, G.D. Waxes used as structuring agents for food organogels: A Review. Grasas Aceites 2020, 71, 918. [Google Scholar] [CrossRef]
- Sarkisyan, V.; Frolova, Y.; Sobolev, R.; Kochetkova, A. On the Role of Beeswax Components in the Regulation of Sunflower Oil Oleogel Properties. Food Biophys. 2022. [Google Scholar] [CrossRef]
- Silva, P.M.; Martins, A.J.; Fasolin, L.H.; Vicente, A.A. Modulation and Characterization of Wax-Based Olive Oil Organogels in View of Their Application in the Food Industry. Gels 2021, 7, 12. [Google Scholar] [CrossRef]
- Medic, J.; Atkinson, C.; Hurburgh, C.R. Current Knowledge in Soybean Composition. J. Am. Oil Chem. Soc. 2014, 91, 363–384. [Google Scholar] [CrossRef]
- Hammond, E.G.; Johnson, L.A.; Murphy, P.A. Soybean | Grading and Marketing. In Reference Module in Food Science; Elsevier: Amsterdam, The Netherlands, 2016. [Google Scholar] [CrossRef]
- Madureira, A.R.; Pereira, C.I.; Gomes, A.M.P.; Pintado, M.E.; Xavier Malcata, F. Bovine whey proteins—Overview on their main biological properties. Food Res. Int. 2007, 40, 1197–1211. [Google Scholar] [CrossRef]
- Rabiey, L.; Britten, M. Effect of protein composition on the rheological properties of acid-induced whey protein gels. Food Hydrocoll. 2009, 23, 973–979. [Google Scholar] [CrossRef]
- Marshall, K. Therapeutic applications of whey protein. Altern. Med. Rev. 2004, 9, 136–156. [Google Scholar] [PubMed]
- Giteru, S.G.; Ali, M.A.; Oey, I. Recent progress in understanding fundamental interactions and applications of zein. Food Hydrocoll. 2021, 120, 106948. [Google Scholar] [CrossRef]
- Reineccius, G.; Meng, Y. Chapter 20—Gelatin and other proteins for microencapsulation. In Microencapsulation in the Food Industry, 2nd ed.; Sobel, R., Ed.; Academic Press: Cambridge, MA, USA, 2022; pp. 293–308. [Google Scholar] [CrossRef]
- Mikhailov, O.V. Molecular structure design and soft template synthesis of aza-, oxaaza- and thiaazamacrocyclic metal chelates in the gelatin matrix. Arab. J. Chem. 2017, 10, 47–67. [Google Scholar] [CrossRef]
- Rather, J.A.; Akhter, N.; Ashraf, Q.S.; Mir, S.A.; Makroo, H.A.; Majid, D.; Barba, F.J.; Khaneghah, A.M.; Dar, B.N. A comprehensive review on gelatin: Understanding impact of the sources, extraction methods, and modifications on potential packaging applications. Food Packag. Shelf Life 2022, 34, 100945. [Google Scholar] [CrossRef]
- Wang, Z.; Chandrapala, J.; Truong, T.; Farahnaky, A. Oleogels prepared with low molecular weight gelators: Texture, rheology and sensory properties, a review. Crit. Rev. Food Sci. Nutr. 2022, 1–45. [Google Scholar] [CrossRef]
- Yilmaz, E.; Keskin Uslu, E.; Öz, C. Oleogels of Some Plant Waxes: Characterization and Comparison with Sunflower Wax Oleogel. J. Am. Oil Chem. Soc. 2021, 98, 643–655. [Google Scholar] [CrossRef]
- Tinto, W.F.; Elufioye, T.O.; Roach, J. Waxes. In Pharmacognosy; Badal, S., Delgoda, R., Eds.; Elsevier: Amsterdam, The Netherlands, 2017; pp. 443–455. [Google Scholar] [CrossRef]
- EFSA Panel on Food Additives and Nutrient Sources added to Food (ANS). Scientific Opinion on the re-evaluation of carnauba wax (E 903) as a food additive. EFSA J. 2012, 10, 2880. [Google Scholar] [CrossRef]
- Revised Compendium of Food Additive Specifications Addendum 13. In Proceedings of the Joint FAO/WHO Expert Committee on Food Additives (JECFA) 65th Meeting, Geneva, Switzerland, 7–16 June 2005; ISBN 92-5-105355-3.
- EFSA. Beeswax (E 901) as a glazing agent and as carrier for flavours—Scientific Opinion of the Panel on Food additives, Flavourings, Processing aids and Materials in Contact with Food (AFC). EFSA J. 2007, 5, 615. [Google Scholar] [CrossRef]
- Council, E.P.A.O.T. Regulation (EC) no 1333/2008 of the European Parliament and of the Council. 2008. Available online: https://www.legislation.gov.uk/eur/2008/1333/contents (accessed on 31 March 2023).
- Sarkisyan, V.; Sobolev, R.; Frolova, Y.; Vorobiova, I.; Kochetkova, A. A Study of the Quantitative Relationship between Yield Strength and Crystal Size Distribution of Beeswax Oleogels. Gels 2022, 8, 39. [Google Scholar] [CrossRef] [PubMed]
- Gómez-Estaca, J.; Pintado, T.; Jiménez-Colmenero, F.; Cofrades, S. Assessment of a healthy oil combination structured in ethyl cellulose and beeswax oleogels as animal fat replacers in low-fat, PUFA-enriched pork burgers. Food Bioprocess Technol. 2019, 12, 1068–1081. [Google Scholar] [CrossRef]
- Mohd Hassim, N.A. Characteristics of Sunflower Wax, Carnauba Wax and Beeswax in Palm Superolein Blended Oil. J. Oil Palm Res. 2022, 55. [Google Scholar] [CrossRef]
- Yi, B.; Kim, M.J.; Lee, S.Y.; Lee, J. Physicochemical properties and oxidative stability of oleogels made of carnauba wax with canola oil or beeswax with grapeseed oil. Food Sci. Biotechnol. 2017, 26, 79–87. [Google Scholar] [CrossRef]
- Qiu, H.; Qu, K.; Eun, J.B.; Zhang, H. Analysis of thermal oxidation of different multi-element oleogels based on carnauba wax, beta-sitosterol/lecithin, and ethyl cellulose by classical oxidation determination method combined with the electronic nose. Food Chem. 2023, 405, 134970. [Google Scholar] [CrossRef] [PubMed]
- Martini, S.; An, M.C. Determination of Wax Concentration in Sunflower Seed Oil. J. Am. Oil Chem. Soc. 2000, 77, 1087–1093. [Google Scholar] [CrossRef]
- Doan, C.D.; Tavernier, I.; Sintang, M.D.B.; Danthine, S.; Van de Walle, D.; Rimaux, T.; Dewettinck, K. Crystallization and Gelation Behavior of Low- and High Melting Waxes in Rice Bran Oil: A Case-Study on Berry Wax and Sunflower Wax. Food Biophys. 2016, 12, 97–108. [Google Scholar] [CrossRef]
- Scharfe, M.; Niksch, J.; Floter, E. Influence of Minor Oil Components on Sunflower, Rice Bran, Candelilla, and Beeswax Oleogels. Eur. J. Lipid Sci. Technol. 2022, 124, 2100068. [Google Scholar] [CrossRef]
- Winkler-Moser, J.K.; Anderson, J.; Felker, F.C.; Hwang, H.S. Physical Properties of Beeswax, Sunflower Wax, and Candelilla Wax Mixtures and Oleogels. J. Am. Oil Chem. Soc. 2019, 96, 1125–1142. [Google Scholar] [CrossRef]
- Dassanayake, L.S.K.; Kodali, D.R.; Ueno, S.; Sato, K. Physical Properties of Rice Bran Wax in Bulk and Organogels. J. Am. Oil Chem. Soc. 2009, 86, 1163. [Google Scholar] [CrossRef]
- Marangoni, A.G. Organogels: An Alternative Edible Oil-Structuring Method. J. Am. Oil Chem. Soc. 2012, 89, 749–780. [Google Scholar] [CrossRef]
- Srikaeo, K. Organic Rice Bran Oils in Health. In Wheat and Rice in Disease Prevention and Health; Watson, R.R., Preedy, V.R., Zibadi, S., Eds.; Elsevier: Amsterdam, The Netherlands, 2014; pp. 453–465. [Google Scholar] [CrossRef]
- Ito, J.; Sawada, K.; Ogura, Y.; Xinyi, F.; Rahmania, H.; Mohri, T.; Kohyama, N.; Kwon, E.; Eitsuka, T.; Hashimoto, H.; et al. Definitive evidence of the presence of 24-methylenecycloartanyl ferulate and 24-methylenecycloartanyl caffeate in barley. Sci. Rep. 2019, 9, 12572. [Google Scholar] [CrossRef] [PubMed]
- Chanioti, S.; Katsouli, M.; Tzia, C. β-Sitosterol as a functional bioactive. In A Centum of Valuable Plant Bioactives; Mushtaq, M., Anwar, F., Eds.; Elsevier: Amsterdam, The Netherlands, 2021; pp. 193–212. [Google Scholar] [CrossRef]
- Babu, S.; Jayaraman, S. An update on beta-sitosterol: A potential herbal nutraceutical for diabetic management. Biomed. Pharmacother. 2020, 131, 110702. [Google Scholar] [CrossRef] [PubMed]
- Yang, R.; Xue, L.; Zhang, L.; Wang, X.; Qi, X.; Jiang, J.; Yu, L.; Wang, X.; Zhang, W.; Zhang, Q.; et al. Phytosterol Contents of Edible Oils and Their Contributions to Estimated Phytosterol Intake in the Chinese Diet. Foods 2019, 8, 334. [Google Scholar] [CrossRef]
- Lerma-García, M.J.; Herrero-Martínez, J.M.; Simó-Alfonso, E.F.; Mendonça, C.R.B.; Ramis-Ramos, G. Composition, industrial processing and applications of rice bran γ-oryzanol. Food Chem. 2009, 115, 389–404. [Google Scholar] [CrossRef]
- Bot, A.; Flöter, E. Edible Oil Oleogels Based on Self-assembled β-Sitosterol + γ-Oryzanol Tubules. In Edible Oleogels; Marangoni, A.G., Garti, N., Eds.; Elsevier: Amsterdam, The Netherlands, 2018; pp. 31–63. [Google Scholar] [CrossRef]
- Cantrill, R. Phytosterols, Phytostanols and Their Esters. Chemical and Technical Assessment. In Proceedings of the 69th Joint FAO/WHO Expert Committee on Food Additives (JECFA) Meeting—Chemical and Technical Assessment (CTA), Rome, Italy, 17–26 June 2008. [Google Scholar]
- Bot, A.; Agterof, W.G.M. Structuring of Edible Oils by Mixtures of γ-Oryzanol with β-Sitosterol or Related Phytosterols. J. Am. Oil Chem. Soc. 2006, 83, 513–521. [Google Scholar] [CrossRef]
- Bin Sayeed, M.S.; Karim, S.M.R.; Sharmin, T.; Morshed, M.M. Critical Analysis on Characterization, Systemic Effect, and Therapeutic Potential of Beta-Sitosterol: A Plant-Derived Orphan Phytosterol. Medicines 2016, 3, 29. [Google Scholar] [CrossRef]
- Sawalha, H.; den Adel, R.; Venema, P.; Bot, A.; Floter, E.; van der Linden, E. Organogel-emulsions with mixtures of beta-sitosterol and gamma-oryzanol: Influence of water activity and type of oil phase on gelling capability. J. Agric. Food Chem. 2012, 60, 3462–3470. [Google Scholar] [CrossRef]
- Scharfe, M.; Flöter, E. Oleogelation: From Scientific Feasibility to Applicability in Food Products. Eur. J. Lipid Sci. Technol. 2020, 122, 2000213. [Google Scholar] [CrossRef]
- European Food Safety Authority. Scientific Opinion on the safety of stigmasterol-rich plant sterols as food additive. EFSA J. 2012, 10, 2659. [Google Scholar] [CrossRef]
- Bot, A. Phytosterols. In Encyclopedia of Food Chemistry; Melton, L., Shahidi, F., Varelis, P., Eds.; Academic Press: Oxford, UK, 2019; pp. 225–228. [Google Scholar]
- Patel, M.; Naik, S.N. Gamma-oryzanol from rice bran oil—A review. J. Sci. Ind. Res. 2004, 63, 569–578. [Google Scholar]
- Den Adel, R.; Heussen, P.C.M.; Bot, A. Effect of water on self-assembled tubules in β-sitosterol + γ-oryzanol-based organogels. In Proceedings of the XIV International Conference on Small-Angle Scattering (SAS09), Oxford, UK, 13–18 September 2009; IOP: Bristol, UK, 2010; Volume 247. [Google Scholar] [CrossRef]
- Martins, A.J.; Cerqueira, F.; Vicente, A.A.; Cunha, R.L.; Pastrana, L.M.; Cerqueira, M.A. Gelation Behavior and Stability of Multicomponent Sterol-Based Oleogels. Gels 2022, 8, 37. [Google Scholar] [CrossRef] [PubMed]
- Rosen-Kligvasser, J.; Davidovich-Pinhas, M. The role of hydrogen bonds in TAG derivative-based oleogel structure and properties. Food Chem. 2021, 334, 127585. [Google Scholar] [CrossRef] [PubMed]
- Sagiri, S.S.; Singh, V.K.; Pal, K.; Banerjee, I.; Basak, P. Stearic acid based oleogels: A study on the molecular, thermal and mechanical properties. Mater. Sci. Eng. C Mater. Biol. Appl. 2015, 48, 688–699. [Google Scholar] [CrossRef]
- Sivakanthan, S.; Fawzia, S.; Madhujith, T.; Karim, A. Synergistic effects of oleogelators in tailoring the properties of oleogels: A review. Compr. Rev. Food. Sci. Food Saf. 2022, 21, 3507–3539. [Google Scholar] [CrossRef]
- Feichtinger, A.; Nibbelink, D.G.; Poppe, S.; Bozzo, L.; Landman, J.; Scholten, E. Protein oleogels prepared by solvent transfer method with varying protein sources. Food Hydrocoll. 2022, 132, 107821. [Google Scholar] [CrossRef]
- Tang, C.-H. Globular proteins as soft particles for stabilizing emulsions: Concepts and strategies. Food Hydrocoll. 2020, 103, 105664. [Google Scholar] [CrossRef]
- Li, Y.; Zou, Y.; Que, F.; Zhang, H. Recent advances in fabrication of edible polymer oleogels for food applications. Curr. Opin. Food Sci. 2022, 43, 114–119. [Google Scholar] [CrossRef]
- Nicolai, T. Gelation of food protein-protein mixtures. Adv. Colloid Interface Sci. 2019, 270, 147–164. [Google Scholar] [CrossRef]
- Alipal, J.; Mohd Pu’ad, N.A.S.; Lee, T.C.; Nayan, N.H.M.; Sahari, N.; Basri, H.; Idris, M.I.; Abdullah, H.Z. A review of gelatin: Properties, sources, process, applications, and commercialisation. Mater. Today Proc. 2021, 42, 240–250. [Google Scholar] [CrossRef]
- Zhang, T.; Dou, W.; Zhang, X.; Zhao, Y.; Zhang, Y.; Jiang, L.; Sui, X. The development history and recent updates on soy protein-based meat alternatives. Trends Food Sci. Technol. 2021, 109, 702–710. [Google Scholar] [CrossRef]
- Ma, Y.; Ye, F.; Chen, J.; Ming, J.; Zhou, C.; Zhao, G.; Lei, L. The microstructure and gel properties of linseed oil and soy protein isolate based-oleogel constructed with highland barley β-glucan and its application in luncheon meat. Food Hydrocoll. 2023, 140, 108666. [Google Scholar] [CrossRef]
- Minj, S.; Anand, S. Whey Proteins and Its Derivatives: Bioactivity, Functionality, and Current Applications. Dairy 2020, 1, 233–258. [Google Scholar] [CrossRef]
- Ganju, S.; Gogate, P.R. A review on approaches for efficient recovery of whey proteins from dairy industry effluents. J. Food Eng. 2017, 215, 84–96. [Google Scholar] [CrossRef]
- Meissner, P.M.; Keppler, J.K.; Stockmann, H.; Schwarz, K. Cooxidation of proteins and lipids in whey protein oleogels with different water amounts. Food Chem. 2020, 328, 127123. [Google Scholar] [CrossRef]
- Wang, G.S.; Chen, H.Y.; Wang, L.J.; Zou, Y.; Wan, Z.L.; Yang, X.Q. Formation of protein oleogels via capillary attraction of engineered protein particles. Food Hydrocoll. 2022, 133, 107912. [Google Scholar] [CrossRef]
- Lv, L.-C.; Huang, Q.-Y.; Ding, W.; Xiao, X.-H.; Zhang, H.-Y.; Xiong, L.-X. Fish gelatin: The novel potential applications. J. Funct. Foods 2019, 63, 103581. [Google Scholar] [CrossRef]
- Labowska, M.B.; Cierluk, K.; Jankowska, A.M.; Kulbacka, J.; Detyna, J.; Michalak, I. A Review on the Adaption of Alginate-Gelatin Hydrogels for 3D Cultures and Bioprinting. Materials 2021, 14, 858. [Google Scholar] [CrossRef]
- Shao, P.; Feng, J.; Sun, P.; Xiang, N.; Lu, B.; Qiu, D. Recent advances in improving stability of food emulsion by plant polysaccharides. Food Res. Int. 2020, 137, 109376. [Google Scholar] [CrossRef]
- Ullah, S.; Khalil, A.A.; Shaukat, F.; Song, Y. Sources, Extraction and Biomedical Properties of Polysaccharides. Foods 2019, 8, 304. [Google Scholar] [CrossRef]
- Davidovich-Pinhas, M. Oil structuring using polysaccharides. Curr. Opin. Food Sci. 2019, 27, 29–35. [Google Scholar] [CrossRef]
- Vélez-Erazo, E.M.; Bosqui, K.; Rabelo, R.S.; Kurozawa, L.E.; Hubinger, M.D. High internal phase emulsions (HIPE) using pea protein and different polysaccharides as stabilizers. Food Hydrocoll. 2020, 105, 105775. [Google Scholar] [CrossRef]
- Yang, X.; Li, A.; Li, X.; Sun, L.; Guo, Y. An overview of classifications, properties of food polysaccharides and their links to applications in improving food textures. Trends Food Sci. Technol. 2020, 102, 1–15. [Google Scholar] [CrossRef]
- Nishinari, K.; Fang, Y. Relation between structure and rheological/thermal properties of agar. A mini-review on the effect of alkali treatment and the role of agaropectin. Food Struct. 2017, 13, 24–34. [Google Scholar] [CrossRef]
- Sousa, A.M.M.; Rocha, C.M.R.; Gonçalves, M.P. Chapter 24—Agar. In Handbook of Hydrocolloids, 3rd ed.; Phillips, G.O., Williams, P.A., Eds.; Woodhead Publishing: Sawston, UK, 2021; pp. 731–765. [Google Scholar] [CrossRef]
- BeMiller, J.N. 6—Starches: Molecular and Granular Structures and Properties. In Carbohydrate Chemistry for Food Scientists, 3rd ed.; BeMiller, J.N., Ed.; AACC International Press: Washington, DC, USA, 2019; pp. 159–189. [Google Scholar] [CrossRef]
- Zucca, P.; Fernandez-Lafuente, R.; Sanjust, E. Agarose and Its Derivatives as Supports for Enzyme Immobilization. Molecules 2016, 21, 1577. [Google Scholar] [CrossRef]
- Banas, K.; Harasym, J. Natural Gums as Oleogelators. Int. J. Mol. Sci. 2021, 22, 12977. [Google Scholar] [CrossRef]
- Habibi, H.; Khosravi-Darani, K. Effective variables on production and structure of xanthan gum and its food applications: A review. Biocatal. Agric. Biotechnol. 2017, 10, 130–140. [Google Scholar] [CrossRef]
- Kang, Y.; Li, P.; Zeng, X.; Chen, X.; Xie, Y.; Zeng, Y.; Zhang, Y.; Xie, T. Biosynthesis, structure and antioxidant activities of xanthan gum from Xanthomonas campestris with additional furfural. Carbohydr. Polym. 2019, 216, 369–375. [Google Scholar] [CrossRef]
- Riaz, T.; Iqbal, M.W.; Jiang, B.; Chen, J. A review of the enzymatic, physical, and chemical modification techniques of xanthan gum. Int. J. Biol. Macromol. 2021, 186, 472–489. [Google Scholar] [CrossRef]
- Nsengiyumva, E.M.; Alexandridis, P. Xanthan gum in aqueous solutions: Fundamentals and applications. Int. J. Biol. Macromol. 2022, 216, 583–604. [Google Scholar] [CrossRef]
- Berninger, T.; Dietz, N.; Gonzalez Lopez, O. Water-soluble polymers in agriculture: Xanthan gum as eco-friendly alternative to synthetics. Microb. Biotechnol. 2021, 14, 1881–1896. [Google Scholar] [CrossRef] [PubMed]
- Patel, J.; Maji, B.; Moorthy, N.; Maiti, S. Xanthan gum derivatives: Review of synthesis, properties and diverse applications. RSC Adv. 2020, 10, 27103–27136. [Google Scholar] [CrossRef] [PubMed]
- Patel, A.R.; Rajarethinem, P.S.; Cludts, N.; Lewille, B.; De Vos, W.H.; Lesaffer, A.; Dewettinck, K. Biopolymer-Based Structuring of Liquid Oil into Soft Solids and Oleogels Using Water-Continuous Emulsions as Templates. Langmuir 2015, 31, 2065–2073. [Google Scholar] [CrossRef]
- Gawkowska, D.; Cybulska, J.; Zdunek, A. Structure-Related Gelling of Pectins and Linking with Other Natural Compounds: A Review. Polymers 2018, 10, 762. [Google Scholar] [CrossRef] [PubMed]
- Lara-Espinoza, C.; Carvajal-Millan, E.; Balandran-Quintana, R.; Lopez-Franco, Y.; Rascon-Chu, A. Pectin and Pectin-Based Composite Materials: Beyond Food Texture. Molecules 2018, 23, 942. [Google Scholar] [CrossRef] [PubMed]
- Khamsucharit, P.; Laohaphatanalert, K.; Gavinlertvatana, P.; Sriroth, K.; Sangseethong, K. Characterization of pectin extracted from banana peels of different varieties. Food Sci. Biotechnol. 2018, 27, 623–629. [Google Scholar] [CrossRef]
- Mellinas, C.; Ramos, M.; Jimenez, A.; Garrigos, M.C. Recent Trends in the Use of Pectin from Agro-Waste Residues as a Natural-Based Biopolymer for Food Packaging Applications. Materials 2020, 13, 673. [Google Scholar] [CrossRef]
- Freitas, C.M.P.; Coimbra, J.S.R.; Souza, V.G.L.; Sousa, R.C.S. Structure and Applications of Pectin in Food, Biomedical, and Pharmaceutical Industry: A Review. Coatings 2021, 11, 922. [Google Scholar] [CrossRef]
- Thambiliyagodage, C.; Jayanetti, M.; Mendis, A.; Ekanayake, G.; Liyanaarachchi, H.; Vigneswaran, S. Recent Advances in Chitosan-Based Applications-A Review. Materials 2023, 16, 2073. [Google Scholar] [CrossRef]
- Hu, Z.; Ganzle, M.G. Challenges and opportunities related to the use of chitosan as a food preservative. J. Appl. Microbiol. 2019, 126, 1318–1331. [Google Scholar] [CrossRef]
- Rocha, M.A.M.; Coimbra, M.A.; Nunes, C. Applications of chitosan and their derivatives in beverages: A critical review. Curr. Opin. Food Sci. 2017, 15, 61–69. [Google Scholar] [CrossRef]
- Aranaz, I.; Alcantara, A.R.; Civera, M.C.; Arias, C.; Elorza, B.; Heras Caballero, A.; Acosta, N. Chitosan: An Overview of Its Properties and Applications. Polymers 2021, 13, 3256. [Google Scholar] [CrossRef] [PubMed]
- Kabanov, V.L.; Novinyuk, L.V. Chitosan Application in Food Technology: A Review of Rescent Advances. Food Syst. 2020, 3, 10–15. [Google Scholar] [CrossRef]
- Baraki, S.Y.; Jiang, Y.; Li, X.; Debeli, D.K.; Wang, B.; Feng, X.; Mao, Z.; Sui, X. Stable sunflower oil oleogel from oil/water pickering emulsion with regenerated chitin. LWT 2021, 146, 111483. [Google Scholar] [CrossRef]
- Puscas, A.; Muresan, V.; Muste, S. Application of Analytical Methods for the Comprehensive Analysis of Oleogels—A Review. Polymers 2021, 13, 1934. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.Y.; Yi, B.; Kim, M.-J.; Lee, J. Oxidative stability of solid fats containing ethylcellulose determined based on the headspace oxygen content. Food Sci. Biotechnol. 2014, 23, 1779–1784. [Google Scholar] [CrossRef]
- Opinion of the Scientific Panel on Food Additives, Flavourings, Processing Aids and Materials in Contact with Food (AFC) on a request from the Commission related to Ethyl Cellulose as a food additive. EFSA J. 2004, 2, 35. [CrossRef]
- Ahmadi, P.; Jahanban-Esfahlan, A.; Ahmadi, A.; Tabibiazar, M.; Mohammadifar, M. Development of Ethyl Cellulose-based Formulations: A Perspective on the Novel Technical Methods. Food Rev. Int. 2020, 38, 685–732. [Google Scholar] [CrossRef]
- Wasilewska, K.; Winnicka, K. Ethylcellulose-A Pharmaceutical Excipient with Multidirectional Application in Drug Dosage Forms Development. Materials 2019, 12, 3386. [Google Scholar] [CrossRef]
- Davidovich-Pinhas, M.; Barbut, S.; Marangoni, A.G. Physical structure and thermal behavior of ethylcellulose. Cellulose 2014, 21, 3243–3255. [Google Scholar] [CrossRef]
- Rogers, M.A.; Strober, T.; Bot, A.; Toro-Vazquez, J.F.; Stortz, T.; Marangoni, A.G. Edible oleogels in molecular gastronomy. Int. J. Gastron. Food Sci. 2014, 2, 22–31. [Google Scholar] [CrossRef]
- Fu, H.; Lo, Y.M.; Yan, M.; Li, P.; Cao, Y. Characterization of thermo-oxidative behavior of ethylcellulose oleogels. Food Chem. 2020, 305, 125470. [Google Scholar] [CrossRef] [PubMed]
- Perta-Crisan, S.; Ursachi, C.S.; Chereji, B.D.; Munteanu, F.D. Oleogels-Innovative Technological Solution for the Nutritional Improvement of Meat Products. Foods 2022, 12, 131. [Google Scholar] [CrossRef] [PubMed]
- Zetzl, A.K.; Marangoni, A.G.; Barbut, S. Mechanical properties of ethylcellulose oleogels and their potential for saturated fat reduction in frankfurters. Food Funct. 2012, 3, 327–337. [Google Scholar] [CrossRef]
- Scharfe, M.; Prange, D.; Floter, E. The composition of edible oils modifies beta-sitosterol/gamma-oryzanol oleogels. Part I: Stripped triglyceride oils. J. Am. Oil Chem. Soc. 2022, 99, 43–56. [Google Scholar] [CrossRef]
- Scharfe, M.; Prange, D.; Floter, E. The composition of edible oils modifies beta-sitosterol/gamma-oryzanol oleogels. Part II: Addition of selected minor oil components. J. Am. Oil Chem. Soc. 2022, 99, 57–77. [Google Scholar] [CrossRef]
- Li, F.; Gunenc, A.; Hosseinian, F. Developing emulsion gels by incorporating Jerusalem artichoke inulin and investigating their lipid oxidative stability. Food Prod. Process. Nutr. 2020, 2, 2. [Google Scholar] [CrossRef]
- Zampouni, K.; Soniadis, A.; Moschakis, T.; Biliaderis, C.G.; Lazaridou, A.; Katsanidis, E. Crystalline microstructure and physicochemical properties of olive oil oleogels formulated with monoglycerides and phytosterols. LWT 2022, 154, 112815. [Google Scholar] [CrossRef]
- Kesselman, E.; Shimoni, E. Imaging of Oil/Monoglyceride Networks by Polarizing Near-Field Scanning Optical Microscopy. Food Biophys. 2007, 2, 117–123. [Google Scholar] [CrossRef]
- Chen, C.H.; Van Damme, I.; Terentjev, E.M. Phase behavior of C18 monoglyceride in hydrophobic solutions. Soft Matter 2009, 5, 432–439. [Google Scholar] [CrossRef]
- Giacomozzi, A.S.; Carrín, M.E.; Palla, C.A. Storage Stability of Oleogels Made from Monoglycerides and High Oleic Sunflower Oil. Food Biophys. 2021, 16, 306–316. [Google Scholar] [CrossRef]
- Pang, M.; Shi, Z.; Lei, Z.; Ge, Y.; Jiang, S.; Cao, L. Structure and thermal properties of beeswax-based oleogels with different types of vegetable oil. Grasas Aceites 2020, 71, 11. [Google Scholar] [CrossRef]
- Öğütcü, M.; Arifoğlu, N.; Yılmaz, E. Preparation and Characterization of Virgin Olive Oil-Beeswax Oleogel Emulsion Products. J. Am. Oil Chem. Soc. 2015, 92, 459–471. [Google Scholar] [CrossRef]
- Frolova, Y.; Sarkisyan, V.; Sobolev, R.; Makarenko, M.; Semin, M.; Kochetkova, A. The Influence of Edible Oils’ Composition on the Properties of Beeswax-Based Oleogels. Gels 2022, 8, 48. [Google Scholar] [CrossRef] [PubMed]
- Da Silva, T.L.T.; Arellano, D.B.; Martini, S. Effect of Water Addition on Physical Properties of Emulsion Gels. Food Biophys. 2019, 14, 30–40. [Google Scholar] [CrossRef]
- Liao, Z.Y.; Guo, S.L.; Lu, M.W.; Xiao, J.; Cao, Y.; Lan, Y.Q. Tailoring Water-Induced Multi-Component (Ceramide and Lecithin) Oleogels: Influence of Solute Added in Water. Food Biophys. 2022, 17, 84–92. [Google Scholar] [CrossRef]
- Papadaki, A.; Cipolatti, E.P.; Aguieiras, E.C.G.; Cerqueira Pinto, M.C.; Kopsahelis, N.; Freire, D.M.G.; Mandala, I.; Koutinas, A.A. Development of Microbial Oil Wax-Based Oleogel with Potential Application in Food Formulations. Food Bioprocess Technol. 2019, 12, 899–909. [Google Scholar] [CrossRef]
- Fayaz, G.; Calligaris, S.; Nicoli, M.C. Comparative Study on the Ability of Different Oleogelators to Structure Sunflower Oil. Food Biophys. 2019, 15, 42–49. [Google Scholar] [CrossRef]
- Jiang, Z.B.; Bai, X.P. Effects of Polysaccharide Concentrations on the Formation and Physical Properties of Emulsion-Templated Oleogels. Molecules 2022, 27, 5391. [Google Scholar] [CrossRef]
- Yao, Y.P.; Zhou, H.; Liu, W.T.; Li, C.M.; Wang, S. The Effect of Cooling Rate on the Microstructure and Macroscopic Properties of Rice Bran Wax Oleogels. J. Oleo Sci. 2021, 70, 135–143. [Google Scholar] [CrossRef]
- Liu, W.T.; Liu, D.; Yao, Y.P.; Li, C.M. Effects of Low-melting-point Fractions of Cocoa Butter on Rice Bran Wax-corn Oil Mixtures: Thermal, Crystallization and Rheological Properties. J. Oleo Sci. 2021, 70, 491–502. [Google Scholar] [CrossRef] [PubMed]
- Patel, A.R.; Babaahmadi, M.; Lesaffer, A.; Dewettinck, K. Rheological Profiling of Organogels Prepared at Critical Gelling Concentrations of Natural Waxes in a Triacylglycerol Solvent. J. Agric. Food Chem. 2015, 63, 4862–4869. [Google Scholar] [CrossRef] [PubMed]
- Floter, E.; Wettlaufer, T.; Conty, V.; Scharfe, M. Oleogels-Their Applicability and Methods of Characterization. Molecules 2021, 26, 1673. [Google Scholar] [CrossRef] [PubMed]
- Plazzotta, S.; Jung, I.; Schroeter, B.; Subrahmanyam, R.P.; Smirnova, I.; Calligaris, S.; Gurikov, P.; Manzocco, L. Conversion of Whey Protein Aerogel Particles into Oleogels: Effect of Oil Type on Structural Features. Polymers 2021, 13, 4063. [Google Scholar] [CrossRef]
- Park, C.; Jimenez-Flores, R.; Maleky, F. Quantifications of Oleocolloid Matrices Made of Whey Protein and Oleogels. Foods 2020, 9, 1697. [Google Scholar] [CrossRef]
- Kupiec, M.; Zbikowska, A.; Marciniak-Lukasiak, K.; Kowalska, M. Rapeseed Oil in New Application: Assessment of Structure of Oleogels Based on their Physicochemical Properties and Microscopic Observations. Agriculture 2020, 10, 211. [Google Scholar] [CrossRef]
- Li, J.; Yu, H.; Yang, Y.; Drummond, C.J.; Conn, C.E. Effect of Crystallization State on the Gel Properties of Oleogels Based on β-sitosterol. Food Biophys. 2020, 16, 48–57. [Google Scholar] [CrossRef]
- Yang, S.; Zhu, M.P.; Wang, N.; Cui, X.T.; Xu, Q.; Saleh, A.S.M.; Duan, Y.M.; Xiao, Z.G. Influence of Oil Type on Characteristics of -Sitosterol and Stearic Acid Based Oleogel. Food Biophys. 2018, 13, 362–373. [Google Scholar] [CrossRef]
- Wang, Y.; Selomulya, C. Food rheology applications of large amplitude oscillation shear (LAOS). Trends Food Sci. Technol. 2022, 127, 221–244. [Google Scholar] [CrossRef]
- Wang, L.; Wen, Y.; Su, C.; Gao, Y.; Li, Q.; Du, S.; Yu, X. Effect of water content on the physical properties and structure of walnut oleogels. RSC Adv. 2022, 12, 8987–8995. [Google Scholar] [CrossRef]
- Toro-Vazquez, J.F.; Bello-Santillan, M.F.; De la Pena-Gil, A.; Aguilar-Zarate, M.; Charo-Alonso, M.A. Vegetable and mineral oil oleogels developed at different monoglyceride to lecithin molar ratios. J. Am. Oil Chem. Soc. 2022, 99, 951–969. [Google Scholar] [CrossRef]
- De Vries, A.; Jansen, D.; van der Linden, E.; Scholten, E. Tuning the rheological properties of protein-based oleogels by water addition and heat treatment. Food Hydrocoll. 2018, 79, 100–109. [Google Scholar] [CrossRef]
- Bin Sintang, M.D.; Rimaux, T.; Van de Walle, D.; Dewettinck, K.; Patel, A.R. Oil structuring properties of monoglycerides and phytosterols mixtures. Eur. J. Lipid Sci. Technol. 2017, 119, 1500517. [Google Scholar] [CrossRef]
- Doan, C.D.; Patel, A.R.; Tavernier, I.; De Clercq, N.; Van Raemdonck, K.; Van de Walle, D.; Delbaere, C.; Dewettinck, K. The feasibility of wax-based oleogel as a potential co-structurant with palm oil in low-saturated fat confectionery fillings. Eur. J. Lipid Sci. Technol. 2016, 118, 1903–1914. [Google Scholar] [CrossRef]
- Guo, S.L.; Song, M.Y.; Gao, X.Y.; Dong, L.L.; Hou, T.; Lin, X.K.; Tan, W.J.; Cao, Y.; Rogers, M.; Lan, Y.Q. Assembly pattern of multicomponent supramolecular oleogel composed of ceramide and lecithin in sunflower oil: Self-assembly or self-sorting? Food Funct. 2020, 11, 7651–7660. [Google Scholar] [CrossRef]
- Han, L.J.; Li, L.; Zhao, L.; Li, B.; Liu, G.Q.; Liu, X.Q.; Wang, X.D. Rheological properties of organogels developed by sitosterol and lecithin. Food Res. Int. 2013, 53, 42–48. [Google Scholar] [CrossRef]
- Heymans, R.; Tavernier, I.; Danthine, S.; Rimaux, T.; Van der Meeren, P.; Dewettinck, K. Food-grade monoglyceride oil foams: The effect of tempering on foamability, foam stability and rheological properties. Food Funct. 2018, 9, 3143–3154. [Google Scholar] [CrossRef]
- Li, S.Y.; Zhu, L.; Wu, G.C.; Jin, Q.Z.; Wang, X.G.; Zhang, H. Relationship between the microstructure and physical properties of emulsifier based oleogels and cookies quality. Food Chem. 2022, 377, 131966. [Google Scholar] [CrossRef]
- Naktiniene, M.; Eisinaite, V.; Kersiene, M.; Jasutiene, I.; Leskauskaite, D. Emulsification and gelation as a tool for iron encapsulation in food-grade systems. LWT Food Sci. Technol. 2021, 149, 111895. [Google Scholar] [CrossRef]
- Plazzotta, S.; Calligaris, S.; Manzocco, L. Structure of oleogels from kappa-carrageenan templates as affected by supercritical-CO2-drying, freeze-drying and lettuce-filler addition. Food Hydrocoll. 2019, 96, 1–10. [Google Scholar] [CrossRef]
- Trujillo-Ramirez, D.; Lobato-Calleros, C.; Vernon-Carter, E.J.; Alvarez-Ramirez, J. Cooling rate, sorbitan and glyceryl monostearate gelators elicit different microstructural, viscoelastic and textural properties in chia seed oleogels. Food Res. Int. 2019, 119, 829–838. [Google Scholar] [CrossRef] [PubMed]
- Fayaz, G.; Polenghi, O.; Giardina, A.; Cerne, V.; Calligaris, S. Structural and rheological properties of medium-chain triacylglyceride oleogels. Int. J. Food Sci. Technol. 2021, 56, 1040–1047. [Google Scholar] [CrossRef]
- Lim, J.; Hwang, H.-S.; Lee, S. Oil-structuring characterization of natural waxes in canola oil oleogels: Rheological, thermal, and oxidative properties. Appl. Biol. Chem. 2016, 60, 17–22. [Google Scholar] [CrossRef]
- Qiu, C.; Huang, Y.; Li, A.; Ma, D.; Wang, Y. Fabrication and Characterization of Oleogel Stabilized by Gelatin-Polyphenol-Polysaccharides Nanocomplexes. J. Agric. Food Chem. 2018, 66, 13243–13252. [Google Scholar] [CrossRef] [PubMed]
- Espert, M.; Hernandez, M.J.; Sanz, T.; Salvador, A. Rheological properties of emulsion templated oleogels based on xanthan gum and different structuring agents. Curr. Res. Food Sci. 2022, 5, 564–570. [Google Scholar] [CrossRef]
- Yılmaz, E.; Öğütcü, M. Properties and Stability of Hazelnut Oil Organogels with Beeswax and Monoglyceride. J. Am. Oil Chem. Soc. 2014, 91, 1007–1017. [Google Scholar] [CrossRef]
- Moradabbasi, M.; Goli, S.A.H.; Fayaz, G. Effect of biopolymers concentration and drying methods on physicochemical properties of emulsion-templated oleogel. J. Food Sci. Technol. 2022, 59, 1994–2003. [Google Scholar] [CrossRef]
- Ghan, S.Y.; Siow, L.F.; Tan, C.P.; Cheong, K.W.; Thoo, Y.Y. Influence of Soya Lecithin, Sorbitan and Glyceryl Monostearate on Physicochemical Properties of Organogels. Food Biophys. 2020, 15, 386–395. [Google Scholar] [CrossRef]
- Parmar, V.; Sharma, R.; Sharma, S.; Singh, B. Recent advances in fabrication of food grade oleogels: Structuring methods, functional properties and technical feasibility in food products. J. Food Meas. Charact. 2022, 16, 4687–4702. [Google Scholar] [CrossRef]
- López-Pedrouso, M.; Lorenzo, J.M.; Gullón, B.; Campagnol, P.C.B.; Franco, D. Novel strategy for developing healthy meat products replacing saturated fat with oleogels. Curr. Opin. Food Sci. 2021, 40, 40–45. [Google Scholar] [CrossRef]
- Ferro, A.C.; de Souza Paglarini, C.; Rodrigues Pollonio, M.A.; Lopes Cunha, R. Glyceryl monostearate-based oleogels as a new fat substitute in meat emulsion. Meat Sci. 2021, 174, 108424. [Google Scholar] [CrossRef] [PubMed]
- Tarté, R.; Paulus, J.S.; Acevedo, N.C.; Prusa, K.J.; Lee, S.-L. High-oleic and conventional soybean oil oleogels structured with rice bran wax as alternatives to pork fat in mechanically separated chicken-based bologna sausage. LWT 2020, 131, 109659. [Google Scholar] [CrossRef]
- Zampouni, K.; Soniadis, A.; Dimakopoulou-Papazoglou, D.; Moschakis, T.; Biliaderis, C.G.; Katsanidis, E. Modified fermented sausages with olive oil oleogel and NaCl–KCl substitution for improved nutritional quality. LWT 2022, 158, 113172. [Google Scholar] [CrossRef]
- Gómez-Estaca, J.; Pintado, T.; Jiménez-Colmenero, F.; Cofrades, S. The effect of household storage and cooking practices on quality attributes of pork burgers formulated with PUFA- and curcumin-loaded oleogels as healthy fat substitutes. LWT 2020, 119, 108909. [Google Scholar] [CrossRef]
- Lopes, R.; Costa, V.; Costa, M.; Paiva-Martins, F. Olive oil oleogels as strategy to confer nutritional advantages to burgers. Food Chem. 2022, 397, 133724. [Google Scholar] [CrossRef] [PubMed]
- Moghtadaei, M.; Soltanizadeh, N.; Goli, S.A.H. Production of sesame oil oleogels based on beeswax and application as partial substitutes of animal fat in beef burger. Food Res. Int. 2018, 108, 368–377. [Google Scholar] [CrossRef]
- Ferrer-González, B.M.; García-Martínez, I.; Totosaus, A. Textural properties, sensory acceptance and fatty acid profile of cooked meat batters employing pumpkin seed paste or soybean oil oleogel as fat replacers. Grasas Aceites 2019, 70, 5182. [Google Scholar] [CrossRef]
- Oh, I.; Lee, J.; Lee, H.G.; Lee, S. Feasibility of hydroxypropyl methylcellulose oleogel as an animal fat replacer for meat patties. Food Res. Int. 2019, 122, 566–572. [Google Scholar] [CrossRef]
- Martins, A.J.; Lorenzo, J.M.; Franco, D.; Pateiro, M.; Dominguez, R.; Munekata, P.E.S.; Pastrana, L.M.; Vicente, A.A.; Cunha, R.L.; Cerqueira, M.A. Characterization of Enriched Meat-Based Pate Manufactured with Oleogels as Fat Substitutes. Gels 2020, 6, 17. [Google Scholar] [CrossRef]
- Manzoor, S.; Masoodi, F.A.; Rashid, R.; Naqash, F.; Ahmad, M. Oleogels for the development of healthy meat products: A review. Appl. Food Res. 2022, 2, 100212. [Google Scholar] [CrossRef]
- Silva-Avellaneda, E.; Bauer-Estrada, K.; Prieto-Correa, R.E.; Quintanilla-Carvajal, M.X. The effect of composition, microfluidization and process parameters on formation of oleogels for ice cream applications. Sci. Rep. 2021, 11, 7161. [Google Scholar] [CrossRef] [PubMed]
- Moriano, M.E.; Alamprese, C. Organogels as novel ingredients for low saturated fat ice creams. LWT 2017, 86, 371–376. [Google Scholar] [CrossRef]
- Nazarewicz, S.; Kozłowicz, K.; Kobus, Z.; Gładyszewska, B.; Matwijczuk, A.; Ślusarczyk, L.; Skrzypek, T.; Sujka, M.; Kozłowicz, N. The Use of Ultrasound in Shaping the Properties of Ice Cream with Oleogel Based on Oil Extracted from Tomato Seeds. Appl. Sci. 2022, 12, 9165. [Google Scholar] [CrossRef]
- Szymanska, I.; Zbikowska, A.; Kowalska, M.; Golec, K. Application of Oleogel and Conventional Fats for Ultrasound-assisted Obtaining of Vegan Creams. J. Oleo Sci. 2021, 70, 1495–1507. [Google Scholar] [CrossRef] [PubMed]
- Colla, K.; Costanzo, A.; Gamlath, S. Fat Replacers in Baked Food Products. Foods 2018, 7, 192. [Google Scholar] [CrossRef] [PubMed]
- Patel, A.R.; Rajarethinem, P.S.; Gredowska, A.; Turhan, O.; Lesaffer, A.; De Vos, W.H.; Van de Walle, D.; Dewettinck, K. Edible applications of shellac oleogels: Spreads, chocolate paste and cakes. Food Funct. 2014, 5, 645–652. [Google Scholar] [CrossRef] [PubMed]
- Bharti, D.; Banerjee, I.; Makowska, A.; Jarzębski, M.; Kowalczewski, P.Ł.; Pal, K. Evaluation of the Effect of Stearyl Alcohol and Span-60 Tuned Sunflower Wax/Sunflower Oil Oleogel on Butter Replacement in Whole Wheat Cake. Appl. Sci. 2023, 13, 1063. [Google Scholar] [CrossRef]
- Malvano, F.; Laudisio, M.; Albanese, D.; d’Amore, M.; Marra, F. Olive Oil-Based Oleogel as Fat Replacer in a Sponge Cake: A Comparative Study and Optimization. Foods 2022, 11, 2643. [Google Scholar] [CrossRef]
- Sim, S.Y.J.; Wong, K.X.; Henry, C.J. Healthier pineapple tart pastry using oleogel-based solid fat replacement. Malays. J. Nutr. 2021, 27, 327–333. [Google Scholar] [CrossRef]
- Wettlaufer, T.; Floter, E. Wax based oleogels and their application in sponge cakes. Food Funct. 2022, 13, 9419–9433. [Google Scholar] [CrossRef]
- Kim, D.; Oh, I. The Characteristic of Insect Oil for a Potential Component of Oleogel and Its Application as a Solid Fat Replacer in Cookies. Gels 2022, 8, 355. [Google Scholar] [CrossRef] [PubMed]
- Li, S.Y.; Wu, G.C.; Li, X.J.; Jin, Q.Z.; Wang, X.G.; Zhang, H. Roles of gelator type and gelation technology on texture and sensory properties of cookies prepared with oleogels. Food Chem. 2021, 356, 129667. [Google Scholar] [CrossRef] [PubMed]
- Jiamjariyatam, R. Rice bran wax shortening process for application in biscuit sticks. Int. Food Res. J. 2022, 29, 814–827. [Google Scholar] [CrossRef]
- Tanislav, A.E.; Pușcaș, A.; Păucean, A.; Mureșan, A.E.; Semeniuc, C.A.; Mureșan, V.; Mudura, E. Evaluation of Structural Behavior in the Process Dynamics of Oleogel-Based Tender Dough Products. Gels 2022, 8, 317. [Google Scholar] [CrossRef]
- Bascuas, S.; Morell, P.; Quiles, A.; Salvador, A.; Hernando, I. Use of Oleogels to Replace Margarine in Steamed and Baked Buns. Foods 2021, 10, 1781. [Google Scholar] [CrossRef] [PubMed]
- Bascuas, S.; Espert, M.; Llorca, E.; Quiles, A.; Salvador, A.; Hernando, I. Structural and sensory studies on chocolate spreads with hydrocolloid-based oleogels as a fat alternative. LWT 2021, 135, 110228. [Google Scholar] [CrossRef]
- Puscas, A.; Tanislav, A.E.; Muresan, A.E.; Farcas, A.C.; Muresan, V. Walnut Oil Oleogels as Milk Fat Replacing System for Commercially Available Chocolate Butter. Gels 2022, 8, 613. [Google Scholar] [CrossRef]
- Espert, M.; Hernández, M.J.; Sanz, T.; Salvador, A. Reduction of saturated fat in chocolate by using sunflower oil-hydroxypropyl methylcellulose based oleogels. Food Hydrocoll. 2021, 120, 106917. [Google Scholar] [CrossRef]
- Tirgarian, B.; Yadegari, H.; Bagheri, A.; Neshagaran, E.; Mardani, M.; Farmani, J. Reduced-fat chocolate spreads developed by water-in-oleogel emulsions. J. Food Eng. 2023, 337, 111233. [Google Scholar] [CrossRef]
- Kim, M.; Hwang, H.S.; Jeong, S.; Lee, S. Utilization of oleogels with binary oleogelator blends for filling creams low in saturated fat. LWT Food Sci. Technol. 2022, 155, 112972. [Google Scholar] [CrossRef]
- Salama, H.H.; Hashim, A.F. A functional spreadable canola and milk proteins oleogels as a healthy system for candy gummies. Sci. Rep. 2022, 12, 12619. [Google Scholar] [CrossRef] [PubMed]
- Puscas, A.; Muresan, V. The Feasibility of Shellac Wax Emulsion Oleogels as Low-Fat Spreads Analyzed by Means of Multidimensional Statistical Analysis. Gels 2022, 8, 749. [Google Scholar] [CrossRef] [PubMed]
- Chai, X.; Zhang, Y.; Shi, Y.; Liu, Y. Crystallization and Structural Properties of Oleogel-Based Margarine. Molecules 2022, 27, 8952. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Gavaliatsis, T.; Kuster, S.; Stadeli, C.; Fischer, P.; Windhab, E.J. Crust treatments to reduce bread staling. Curr. Res. Food Sci. 2021, 4, 182–190. [Google Scholar] [CrossRef] [PubMed]
- Cerqueira, M.A.; Valoppi, F.; Pal, K. Oleogels and Organogels: A Promising Tool for New Functionalities. Gels 2022, 8, 349. [Google Scholar] [CrossRef] [PubMed]
- Adrah, K.; Adegoke, S.C.; Nowlin, K.; Tahergorabi, R. Study of oleogel as a frying medium for deep-fried chicken. J. Food Meas. Charact. 2021, 16, 1114–1123. [Google Scholar] [CrossRef]
- Chauhan, D.S.; Khare, A.; Lal, A.B.; Bebartta, R.P. Utilising oleogel as a frying medium for deep fried Indian traditional product (Mathri) to reduce oil uptake. J. Indian Chem. Soc. 2022, 99, 100378. [Google Scholar] [CrossRef]
- Aydeniz-Guneser, B.; Yilmaz, E. Sunflower Oil-Polyglycerol Stearate Oleogels: Alternative Deep-fat Frying Media for Onion Rings. J. Oleo Sci. 2022, 71, 651–662. [Google Scholar] [CrossRef]



| Oleogelator | Structure/Composition |
|---|---|
| Low-molecular-weight oleogelators | |
| Fatty acids | |
| Stearic acid (Octadecanoic acid) n = 18 | 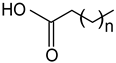 |
| Esters of fatty acids | |
| Sorbitan monostearate |  |
| Waxes | |
| Beeswax |
|
| Carnauba wax |
|
| Sunflower wax |
|
| Rice bran wax |
|
| Glycerolipids | |
| Monoglycerides | 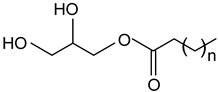 |
| Glycerophospholipids | |
| Lecithins (Phosphatidylcholine) | 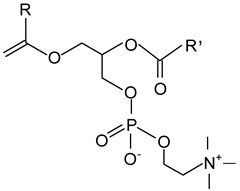 |
| Sterol lipids | |
| Phytosterols β-sitosterol | 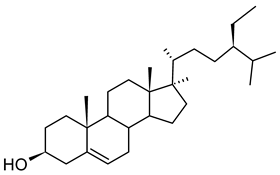 |
| Phytosterol esters γ-oryzanol |  |
| Sphingolipids | |
| Ceramides | 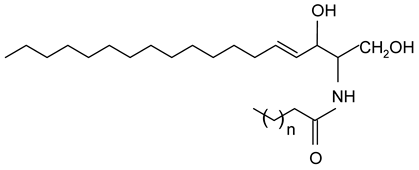 |
| Fatty alcohols | |
| Stearyl alcohol (1-octadecanol) n = 18 | 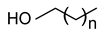 |
| High-molecular-weight oleogelators | |
| Proteins | |
| Soy protein |
|
| Whey protein |
|
| Zein |
|
| Gelatin |
|
| Polysaccharides | |
| Agar | 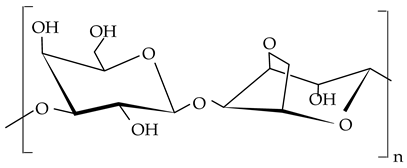 |
| Xanthan gum |  |
| Pectins |  |
| Chitin and chitosan |  |
| Ethylcellulose | 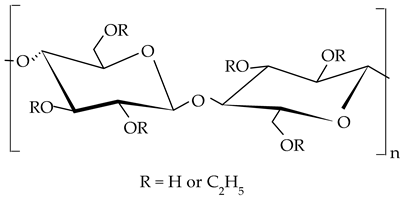 |
| Organogelator | Oil | Type of Microscopy | Results | Comments | Reference |
|---|---|---|---|---|---|
| Beeswax | Sunflower Flaxseed | Polarized light microscopy | Small number of small and rod-shaped crystals | Higher concentrations of beeswax contributed to formation of denser and larger crystals The type of oil did not have influence on the microstructure of the oleogel | [178] |
| Beeswax Beeswax hydrocarbons | Deodorized bleached sunflower | Polarized light microscopy | Needle-shaped crystals for oleogels prepared with beeswax and hexagonal crystals for beeswax-hydrocarbon-based oleogels | The cooling rate of the oleogels influences the size of the formed crystals | [88] |
| Beeswax | Virgin olive oil | Polarized light microscopy | Needle-like beeswax crystals | Preservation of the oleogels for 90 days at room temperature did not influence their structural stability | [179] |
| Beeswax | Sunflower oil | Polarized light microscopy | Needle-like crystals | The needle-like crystals are seen like this, but in reality, they have platelet-like shape due to the crystals perpendicular to the surface of the slides | [71] |
| Beeswax | Fish oil Sunflower oil Olive oil Linseed oil | Polarized light microscopy | Needle-like crystals, exception for linseed-oil-based oleogel where the crystals are mainly spherulitic in shape | The structure and the size of the crystal are related to the oil-binding capacity and textural performance | [180] |
| Candelilla wax Monoglycerides Hard fat | High-oleic sunflower | Polarized light microscopy | Spherulite-shaped crystals formed by radially oriented needle-like crystals | Addition of water changed the size and morphology of crystals | [181] |
| Carnauba wax | Refined soybean oil Refined peanut oil | Polarized light microscopy | Needle-like crystals | An increase in the carnauba wax content leads to the aggregation of the crystals | [29] |
| Ceramide III Soybean lecithin | Sunflower oil | Polarized light microscopy | Spherulite-like aggregates formed by radially oriented needle-like crystals | Addition of water to the system resulted in the formation of lamellar crystals due to the re-arrangement of ceramide and lecithin | [182] |
| Cetyl esters wax | Olive oil | Polarized light microscopy | Flake-like crystals | The cooling temperature influences the size of the crystals | [183] |
| Ethylcellulose Stearic acid Octadecanol Monoglyceride Beeswax Rice bran wax Sunflower wax Mixture of γ-oryzanol and β-sitosterol | Sunflower oil | Polarized light microscopy | Needle-like crystals In the case of rice bran wax oleogels, spherical crystals were observed, while for those with stearic acid and octadecanol, the crystals were platelet-like structures | Except for the mixture γ-oryzanol - β-sitosterol and monoglyceride-based oleogels, all the other oleogels did not show changes in their microstructure | [184] |
| Ethylcellulose Beeswax | Olive oil Linseed oil Fish oil | Polarized light microscopy | The ethylcellulose oleogels had polymeric structure with holes in which the oil is entrapped The beeswax oleogel has a needle-like structure | Needle-like crystals permit the entrapment of large amounts of oil | [89] |
| Hydroxypropyl methylcellulose with different concentrations of pectin | Medium-chain triglyceride oil | Optical microscopy | Small droplets | Addition of pectin contributes to the formation of smaller droplets | [185] |
| Monoglycerides | Olive oil | Polarized light microscopy | Large crystals | - | [173] |
| Nα, Nε-diacyl-L-lysines | Linseed oil Soybean oil gel Corn germ oil Olive oil | TEM | Needle-like crystals that have different lengths dependent on the used gelator | The type of used oil does not influence the microstructure of the oleogels. This depends on the carbon-chain length of gelators | [34] |
| Rice bran wax | Soybean oil | Polarized light microscopy | Dendritic crystals | An increase in rice bran wax content leads to an increase in the crystal size | [186] |
| Rice bran wax Added low-melting-point fractions of cocoa butter | Corn oil | Polarized light microscopy | Needle-like crystals | An increase in the content of low-melting-point fractions of cocoa butter contributes to the formation of crystallites with the possibility to form a crystallization network | [187] |
| Sunflower wax Carnauba wax Candelilla wax Beeswax Berry wax Fruit wax | Refined high-oleic sunflower oil | Normal and polarized light microscopy | Rod-like crystals | The high content of wax esters contributes to the formation of rod-like morphology | [188] |
| Sunflower wax Beeswax Rice bran wax Candelilla wax Carnauba Brazilian wax Carnauba wild wax Fruit wax Berry wax | Rice bran oil | Polarized light microscopy | Needle-like crystals | Beeswax in rice bran oil exhibited two types of crystals: needle-like crystals and spherulite crystals | [26] |
| Sunflower wax Berry wax | Rice bran oil | Polarized light microscopy Cryo-scanning electron microscopy | Needle crystals | The berry wax oleogels present higher transparency in their appearance due to the smaller size of the microcrystallites of berry wax oleogel | [94] |
| Sunflower wax | Canola oil | Polarized light microscopy Cryo-scanning electron microscopy | The PLM indicated the formation of needle-like crystals, while the cryo-SEM proves the formation of the platelets | - | [189] |
| Wax esters | Medium-chain triglycerides | Bright-field microscopy | Crystals of different sizes with angles of 110° and 70° | The total carbon number and molecular symmetry of the respective wax ester play an important role in the regularity and face length of the formed crystals | [4] |
| Whey protein | Castor oil, cod liver oil, corn oil Flaxseed oil MCT Peanut oil, sunflower oil | Optical microscopy | The microstructures are less dense, agglomerated, and fine-stranded because of the stabilization of the proteins close to isoelectric pint | The obtained microstructure is characteristic of the unfolding behavior of the globular proteins during the gelation process | [190] |
| Whey protein | High-oleic soybean oil | Polarized light microscopy | Protein aggregates were observed in the microstructure | Formation of whey protein aggregates is dependent on the concentration of the protein | [191] |
| White and yellow beeswax Candelilla wax Sunflower wax Monoacylglycerols | Rapeseed oil | Polarized and bright-field light microscopy | “Needle” networks of fatty crystals in structured oils | In the monoacylglycerol oleogels, the formed clusters were the largest | [192] |
| β-sitosterol and lecithin | Sunflower oil Liquid coconut oil Solid coconut oil | Optical and polarized optical microscopy | Spherulite-like crystals were obtained when used the sunflower oil Sparse spherulite crystals for the liquid coconut oil Radiolitic crystal structure for the case of solid coconut oil | Addition of lecithin promoted formation of needle-like crystals in the case of sunflower oil or decomposition of the radiolitic crystals into small fibrillar radiolitic crystals | [193] |
| β-sitosterol and stearic acid | Virgin olive oil Corn oil Sunflower oil Flaxseed oil | Optical microscopy | Crystals of different shapes | The shape of the crystals was influenced by the gelator’s concentration, composition, and polarity of the oil | [194] |
| Agar | Rice bran oil and stearyl alcohol | Bright-field microscopy | Globular-shaped particles dispersed in continuous matrix of agar gel | The globular structures might be attributed to the self-aggregated agar polymeric chains | [41] |
| Chitosan crosslinked with vanillin | Refined canola oil | Polarized light microscopy | Packed network with crystals of vanillin, depending on the used concentration | The presence of Tween60 (polysorbate) leads to unstable oleogels | [55] |
| Oleogelators | Oil | Results | Comments | Reference |
|---|---|---|---|---|
| Beeswax (BW) | MCT oil | When increasing the concentration to 10%, all the oleogels form a strong gel | BW is the strongest and elastic at 15% | [208] |
| Monoglyceride (MG) | MCT oil | Weak gel | ||
| Rapeseed wax (RAW) | MCT oil | At any concentration, RAW was not able to form stable structure with MCT oil | ||
| Rice bran wax (RW) | MCT oil | At 10%, it is a weak gel, but at 15%, it is elastic | ||
| Sunflower wax (SW) | MCT oil | Brittle structure | ||
| γ-oryzanol and β-sitosterol mixture | MCT oil | Increasing to 15% did not show modification | ||
| Candelilla wax (CDW) | Grapeseed oil | Form a harder gel | Greater viscoelasticity | [56] |
| Glyceryl monostearate (GMS) | ||||
| Candelilla wax | Canola oil | All the oleogel combinations form an elastic gel | Depending on the temperatures, the gels are more or less elastic | [209] |
| Carnauba wax | Canola oil | |||
| Beeswax | Canola oil | |||
| Sunflower wax (SW) | Rice bran oil (RBO) | Highest cross-over point | Highest critical stress Good tolerance of deformation | [26] |
| Beeswax (BW) | Strong gel property | Highest critical stress Good tolerance of deformation | ||
| Rice bran wax (RBW) | Weak and instability network formation | Weak gel strength Unstable at low amplitudes | ||
| Candelilla wax (CLW) | Strong gel property | Larger structural sensitivity | ||
| Carnauba Brazilian wax (CRBW) | Strong gel property | Unstable at low amplitudes | ||
| Carnauba wild wax (CRWW) | Strong gel property | Good tolerance of deformation | ||
| Fruit wax (FW) | Weak and instability network formation | Weaker gel strength Unstable at low amplitudes | ||
| Berry wax (BEW) | Strong gel property | Larger structural sensitivity | ||
| Ethylcellulose (EC) | Sunflower oil | Did not gel | EC alone cannot form gel | [184] |
| Stearic acid (ST) | Weaker gel | Weak network | ||
| Octadecanol (OD) | Weak gel | For multi-component food | ||
| Monoglyceride (MG) | Weak gel | |||
| Beeswax (BW) | The highest stress resistance | Best structure gel | ||
| Rice bran wax (RW) | Less gel strength | - | ||
| Sunflower wax (SW) | Hardness network | Undergoing changes at storage time | ||
| Mixture of γ-oryzanol and β-sitosterol (γ-β) | The highest stress resistance | Hard gel structure | ||
| Berry wax (BEW) | Rice bran oil (RBO) | Strong molecular network | Weaker gel strength Good tolerance of deformation | [94] |
| Sunflower wax (SW) | Strong molecular network More brittle than BEW | Better stability under applied stress Good tolerance of deformation Higher stability in the structure than BEW | ||
| Whey protein | Castor oil, cod liver oil, corn oil, flaxseed oil, MCT oil, peanut oil, sunflower oil | The oleogels showed a gel-like behavior, except for the one formulated with castor oil that lacked structuration | The type of oil did not have a major impact on the oleogel networking | [190] |
| Zein protein | Oleic acid | The results indicate formation of gels | For the gelation is necessary a high amount of ethanol–water | [66] |
| Colloidal solution of gelatin, tannic acid, and flaxseed gum | Soybean oil | Thermally stable oleogels that have the thixotropic recovery ability at high temperature | The obtained oleogel is appropriate for use in baked products | [210] |
| Xantham gum | High-oleic sunflower oil | The oleogels have high mechanical strengths | Addition of soy lecithin led to the lowering of the firmness of the oleogel | [211] |
| Organogelator | Oil | Method | Results | Comments | Reference |
| Candelilla waxCarnauba waxBeeswax | Canola oil | Measurement of the peroxide value for 18 days, accelerated conditions (60 °C) | The measured peroxide values for oleogels were higher than those for the fresh oil because of the higher temperature used for the oleogel preparation | The peroxide values increased over time for fresh oils and oleogels, but a faster rate of increasing was observed for fresh oils.The oxidative stability of the oleogels is influenced by the type of used wax: the peroxide value for candelilla wax < carnauba wax < beeswax.This behavior is correlated with the hardness of the oleogel | [209] |
| Sodium caseinateXanthan gum | Canola oil | Measurement of the peroxide and 2-thiobarbituric acid values | The measured values were similar for oleogel and the fresh canola oil | The xanthan gum, as a biopolymer, contributed to the protection of the oil, which could not be exposed to the prooxidants and oxidation | [213] |
| Sorbitan monostearate, Soy lecithin, Glyceryl monostearate | Palm olein | Differential scanning calorimetry | The thermal stability of the oleogel is directly proportional to the concentration of the organogelators | The lowest thermal stability was registered for the sorbitan monostearate | [214] |
| EthylcelluloseBeeswax | Mixture of olive, linseed, and fish oils | Measurement of the thiobarbituric acid values | For the oleogel formulated with ethylcellulose was registered the highest lipid oxidation because of the higher processing temperature and time during the obtaining of the oleogel | The measured oxidation values for both oleogels were below the values that contribute to the detection of unpleasant flavors | [89] |
| Monoglycerides | High-oleic sunflower oil | Measurement of the peroxide value | The measurement of the peroxide values for 8 weeks indicated low values for all samples | The use of monoglycerides as organogelators improved the oxidative stability of the high-oleic sunflower oil due to the obstruction of oxygen to the active sites by the monoglycerides network | [177] |
| Beeswax | Olive oil | Measurement of the peroxide value | The oxidative stability was measured for 90 days for samples kept at room and refrigerator temperature, and the results showed that all formulations were stable | The formulated oleogels might be compatible with commercial applications | [179] |
| Carnauba wax | Canola oil | Measurement of headspace oxygen content, conjugated dienoic acid, p-anisidine content, and headspace volatile compounds | The carnauba wax oleogels showed better oxidative stability than beeswax oleogels, at both studied temperatures 60 °C and 180 °C | The study indicates that carnauba wax contributes to the enhancing of the oleogel oxidative stability and that beeswax is an efficient oleogelator | [91] |
| Beeswax | Grapeseed oil |
| Oleogelators | Oil | Type ofAnalysis | Results | Comments | Reference |
|---|---|---|---|---|---|
| Rice bran wax (RBW) | Soybean oil (SBO) | Texture analyzer | Hard texture Good resistance to deformation | The main role in the texture of oleogels is represented by cooling rate | [186] |
| Candelilla wax (CLW) | Canola oil | Texture analyzer | Highest hardness Greater gumminess | The lowest adhesiveness and cohesiveness | [209] |
| Carnauba wax (CRW) | Second highest hardness | More gumminess than beeswax | |||
| Beeswax (BW) | The most adhesive and cohesive | Third highest hardness | |||
| Cetyl-derived wax esters | Extra virgin olive oil (VOO) | Texture analyzer | With 60%, the firmness was decreased | Stable texture | [183] |
| Beeswax (BW) | Sunflower oil (SFO) | Texture analyzer | Structured by fractions, showed the lowest firmness | In combinations of fractions has the highest firmness | [71] |
| Monoglycerides (MG) | High-oleic sunflower oil (HOSO) | Texture analyzer | Great stability texture just in the first 3 weeks of storage | Hardness decreases over time | [177] |
| β-Sitosterol (Sit) and Stearic acid (SA) | Olive oil (OLO) | Texture analyzer | Third highest firmness | The gelator mixture and the concentrations of all the oleogels affect the firmness and gel network formation | [194] |
| Corn oil (CRO) | Second highest firmness | ||||
| Sunflower oil (SFO) | The lower firmness | ||||
| Flaxseed oil (FSO) | The highest firmness | ||||
| Beeswax (BW) | Camellia oil (CO) | Texture analyzer | At 3%, the firmness was lower, and at 2%, it was a flowing liquid | For all oleogels, the storage temperature and oil type can affect the textural parameters | [178] |
| Soybean oil (SO) | At 2%, no gel formed | ||||
| Sunflower oil (SFO) | At 2%, no gel formed | ||||
| Flaxseed oil (FO) | The firmness was much greater than that of the others | ||||
| Beeswax (BW) | Sunflower oil (SO) | Texture analyzer | Spreadable texture | Satisfactory plastic | [180] |
| Olive oil (OO) | Spreadable texture | Satisfactory plastic | |||
| Linseed oil (LO) | No spreadable texture | Very soft | |||
| Fish oil (FO) | Spreadable texture | Satisfactory plastic | |||
| Glyceryl monostearate (GM) | Chia seed oil (ChSO) | Texture analyzer | The highest hardness The highest springiness | The highest gumminess Strong gel and more resistant | [207] |
| Sorbitan monostearate (SM) | The highest cohesiveness | Good texture gel | |||
| Colloidal solution of gelatin, tannic acid, and flaxseed gum | Soybean oil | Texture analyzer | Oven-dried oleogels are spreadable, while freeze-dried oleogels have higher hardness | Different textures observed for the freeze-dried and oven-dried oleogels due to different dehydration conditions | [210] |
| Chitosan crosslinked with vanillin | Refined canola oil | Texture analyzer | Hardness and adhesiveness were stable for 30 days | Presence of Tween60 affects the texture after 15 days | [55] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Perța-Crișan, S.; Ursachi, C.-Ș.; Chereji, B.-D.; Tolan, I.; Munteanu, F.-D. Food-Grade Oleogels: Trends in Analysis, Characterization, and Applicability. Gels 2023, 9, 386. https://doi.org/10.3390/gels9050386
Perța-Crișan S, Ursachi C-Ș, Chereji B-D, Tolan I, Munteanu F-D. Food-Grade Oleogels: Trends in Analysis, Characterization, and Applicability. Gels. 2023; 9(5):386. https://doi.org/10.3390/gels9050386
Chicago/Turabian StylePerța-Crișan, Simona, Claudiu-Ștefan Ursachi, Bianca-Denisa Chereji, Iolanda Tolan, and Florentina-Daniela Munteanu. 2023. "Food-Grade Oleogels: Trends in Analysis, Characterization, and Applicability" Gels 9, no. 5: 386. https://doi.org/10.3390/gels9050386
APA StylePerța-Crișan, S., Ursachi, C.-Ș., Chereji, B.-D., Tolan, I., & Munteanu, F.-D. (2023). Food-Grade Oleogels: Trends in Analysis, Characterization, and Applicability. Gels, 9(5), 386. https://doi.org/10.3390/gels9050386








