Bigels as Delivery Systems of Bioactive Fatty Acids Present in Functional Edible Oils: Coconut, Avocado, and Pomegranate
Abstract
1. Introduction
2. Results and Discussion
2.1. Optimization of Bigel Production
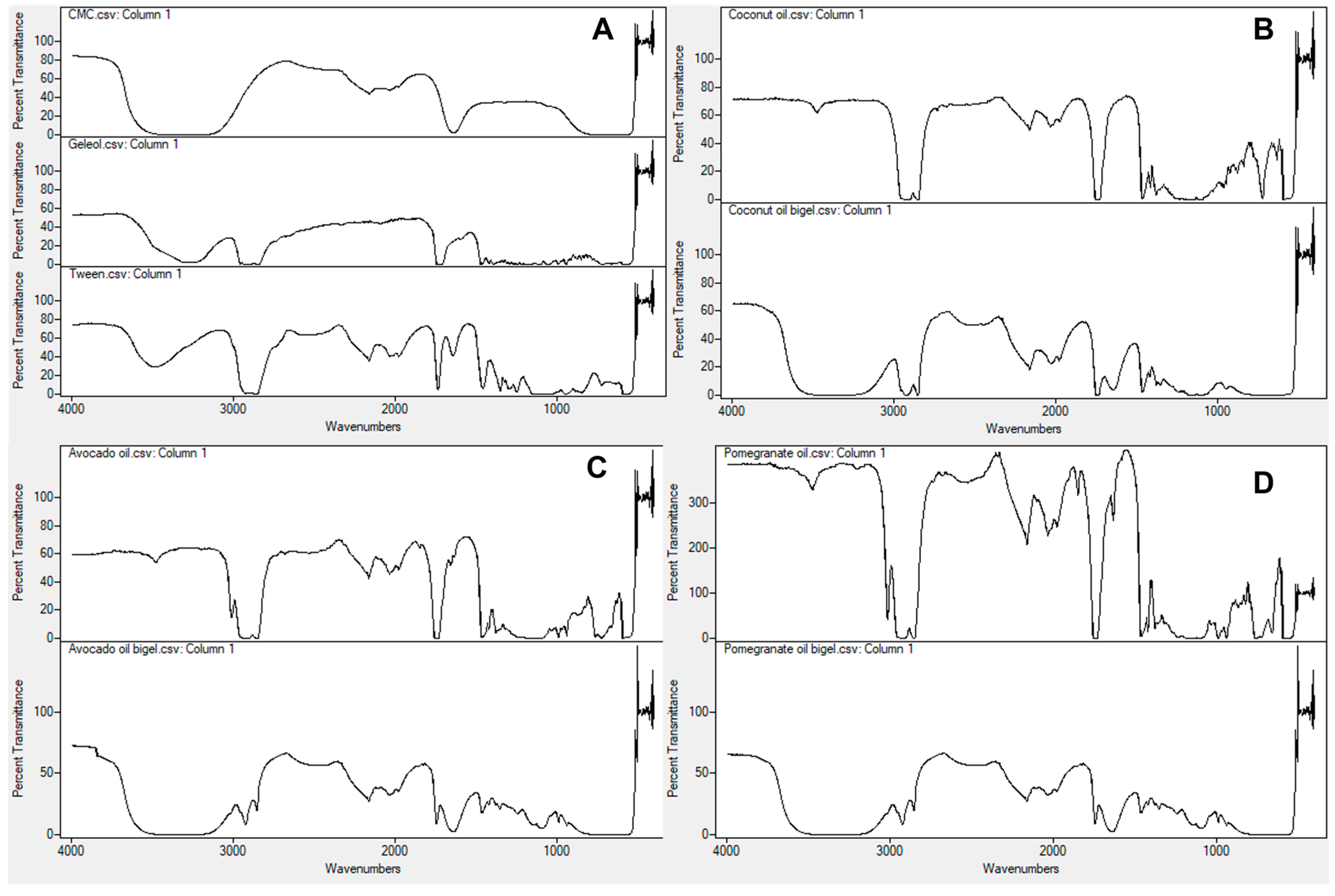
2.2. Structural Characterization of Bigels
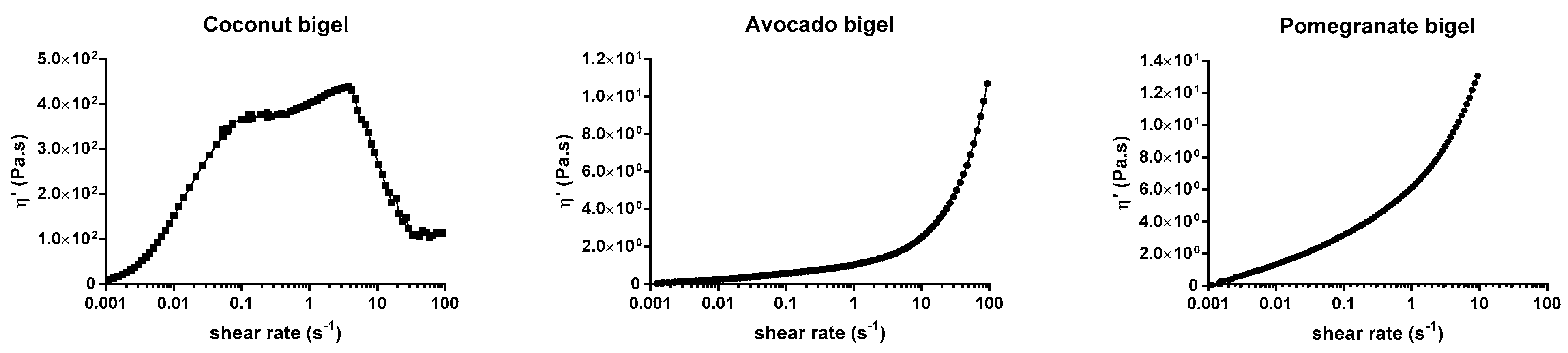
2.3. Rheological Properties
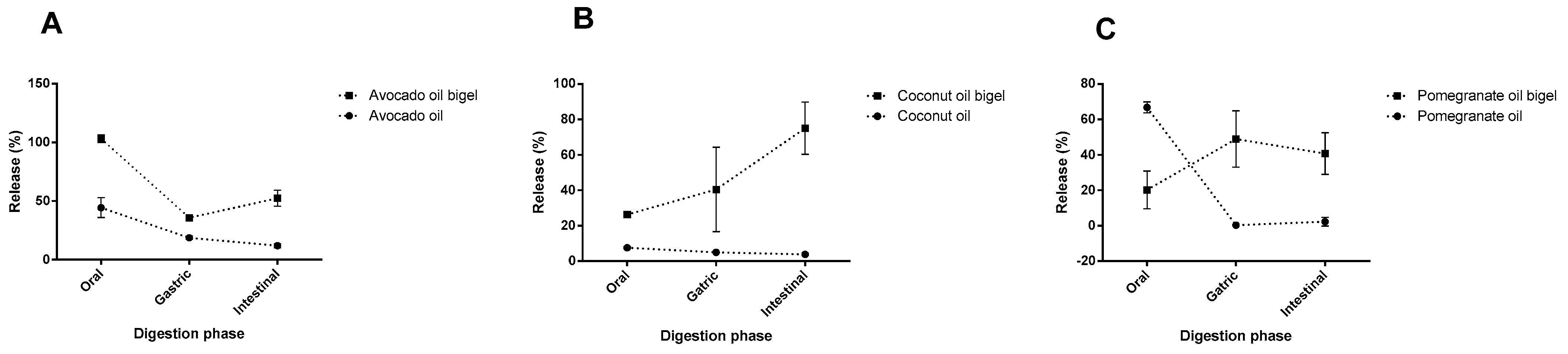
2.4. In Vitro GIT Survival of Bioactive Fatty Acids within Bigels
3. Conclusions
4. Materials and Methods
4.1. Materials and Reagents
4.2. Experimental Design, Modelling, and Optimization
4.3. Bigel Preparation
4.4. Oil Binding Capacity
4.5. ATR-FTIR Spectra
4.6. Rheological Measurements
4.7. In Vitro Gastrointestinal Tract Simulation
4.8. Bigels Fatty Acids Profile
4.9. Statistical Analysis
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- McClements, D.J.; Öztürk, B. Utilization of Nanotechnology to Improve the Handling, Storage and Biocompatibility of Bioactive Lipids in Food Applications. Foods 2021, 10, 365. [Google Scholar] [CrossRef]
- McClements, D.J. Nanoscale Nutrient Delivery Systems for Food Applications: Improving Bioactive Dispersibility, Stability, and Bioavailability. J. Food Sci. 2015, 80, N1602–N1611. [Google Scholar] [CrossRef] [PubMed]
- Salminen, H.; Gömmel, C.; Leuenberger, B.H.; Weiss, J. Influence of encapsulated functional lipids on crystal structure and chemical stability in solid lipid nanoparticles: Towards bioactive-based design of delivery systems. Food Chem. 2016, 190, 928–937. [Google Scholar] [CrossRef] [PubMed]
- Shakeel, A.; Lupi, F.R.; Gabriele, D.; Baldino, N.; De Cindio, B. Bigels: A unique class of materials for drug delivery applications. Soft Mater. 2018, 16, 77–93. [Google Scholar] [CrossRef]
- Mao, L.; Lu, Y.; Cui, M.; Miao, S.; Gao, Y. Design of gel structures in water and oil phases for improved delivery of bioactive food ingredients. Crit. Rev. Food Sci. Nutr. 2020, 60, 1651–1666. [Google Scholar] [CrossRef] [PubMed]
- Martins, A.J.; Silva, P.; Maciel, F.; Pastrana, L.M.; Cunha, R.L.; Cerqueira, M.A.; Vicente, A.A. Hybrid gels: Influence of oleogel/hydrogel ratio on rheological and textural properties. Food Res. Int. 2019, 116, 1298–1305. [Google Scholar] [CrossRef]
- Shakeel, A.; Farooq, U.; Gabriele, D.; Marangoni, A.G.; Lupi, F.R. Bigels and multi-component organogels: An overview from rheological perspective. Food Hydrocoll. 2021, 111, 106190. [Google Scholar] [CrossRef]
- Rehman, K.; Amin, M.C.I.M.; Zulfakar, M.H. Development and Physical Characterization of Polymer-Fish Oil Bigel (Hydrogel/Oleogel) System as a Transdermal Drug Delivery Vehicle. J. Oleo Sci. 2014, 63, 961–970. [Google Scholar] [CrossRef]
- Zheng, H.; Mao, L.; Cui, M.; Liu, J.; Gao, Y. Development of food-grade bigels based on κ-carrageenan hydrogel and monoglyceride oleogels as carriers for β-carotene: Roles of oleogel fraction. Food Hydrocoll. 2020, 105, 105855. [Google Scholar] [CrossRef]
- Zhu, Q.; Gao, J.; Han, L.; Han, K.; Wei, W.; Wu, T.; Li, J.; Zhang, M. Development and characterization of novel bigels based on monoglyceride-beeswax oleogel and high acyl gellan gum hydrogel for lycopene delivery. Food Chem. 2021, 365, 130419. [Google Scholar] [CrossRef]
- Zulfakar, M.H.; Chan, L.M.; Rehman, K.; Wai, L.K.; Heard, C.M. Coenzyme Q10-Loaded Fish Oil-Based Bigel System: Probing the Delivery Across Porcine Skin and Possible Interaction with Fish Oil Fatty Acids. AAPS Pharm. Sci. Tech. 2017, 19, 1116–1123. [Google Scholar] [CrossRef] [PubMed]
- Gumus, C.E.; Gharibzahedi, S.M.T. Yogurts supplemented with lipid emulsions rich in omega-3 fatty acids: New insights into the fortification, microencapsulation, quality properties, and health-promoting effects. Trends Food Sci. Technol. 2021, 110, 267–279. [Google Scholar] [CrossRef]
- Martinez, R.M.; Magalhães, W.V.; Sufi, B.D.S.; Padovani, G.; Nazato, L.I.S.; Velasco, M.V.R.; Lannes, S.C.D.S.; Baby, A.R. Vitamin E-loaded bigels and emulsions: Physicochemical characterization and potential biological application. Colloids Surf. B Biointerfaces 2021, 201, 111651. [Google Scholar] [CrossRef]
- Fayaz, G.; Goli, S.A.H.; Kadivar, M.; Valoppi, F.; Barba, L.; Calligaris, S.; Nicoli, M.C. Potential application of pomegranate seed oil oleogels based on monoglycerides, beeswax and propolis wax as partial substitutes of palm oil in functional chocolate spread. LWT 2017, 86, 523–529. [Google Scholar] [CrossRef]
- Yu, H.; Shi, K.; Liu, D.; Huang, Q. Development of a food-grade organogel with high bioaccessibility and loading of curcuminoids. Food Chem. 2012, 131, 48–54. [Google Scholar] [CrossRef]
- Oh, I.K.; Lee, S. Utilization of foam structured hydroxypropyl methylcellulose for oleogels and their application as a solid fat replacer in muffins. Food Hydrocoll. 2018, 77, 796–802. [Google Scholar] [CrossRef]
- Hamdan, M.A.; Ramli, N.A.; Othman, N.A.; Amin, K.N.M.; Adam, F. Characterization and property investigation of microcrystalline cellulose (MCC) and carboxymethyl cellulose (CMC) filler on the carrageenan-based biocomposite film. Mater. Today Proc. 2021, 42, 56–62. [Google Scholar] [CrossRef]
- Amit; Jamwal, R.; Kumari, S.; Kelly, S.; Cannavan, A.; Singh, D.K. Rapid detection of pure coconut oil adulteration with fried coconut oil using ATR-FTIR spectroscopy coupled with multivariate regression modelling. LWT 2020, 125, 109250. [Google Scholar] [CrossRef]
- Amit; Jamwal, R.; Kumari, S.; Dhaulaniya, A.S.; Balan, B.; Singh, D.K. Application of ATR-FTIR spectroscopy along with regression modelling for the detection of adulteration of virgin coconut oil with paraffin oil. LWT 2020, 118, 108754. [Google Scholar] [CrossRef]
- Rehman, K.; Zulfakar, M.H. Novel Fish Oil-based Bigel System for Controlled Drug Delivery and its Influence on Immunomodulatory Activity of Imiquimod Against Skin Cancer. Pharm. Res. 2017, 34, 36–48. [Google Scholar] [CrossRef]
- Behera, B.; Singh, V.; Kulanthaivel, S.; Bhattacharya, M.; Paramanik, K.; Banerjee, I.; Pal, K. Physical and mechanical properties of sunflower oil and synthetic polymers based bigels for the delivery of nitroimidazole antibiotic—A therapeutic approach for controlled drug delivery. Eur. Polym. J. 2015, 64, 253–264. [Google Scholar] [CrossRef]
- Nasirpour-Tabrizi, P.; Azadmard-Damirchi, S.; Hesari, J.; Heshmati, M.K.; Savage, G.P. Rheological and physicochemical properties of novel low-fat emulgels containing flaxseed oil as a rich source of ω-3 fatty acids. LWT 2020, 133, 110107. [Google Scholar] [CrossRef]
- Rohman, A.; Irnawati; Erwanto, Y.; Lukitaningsih, E.; Rafi, M.; Fadzilah, N.A.; Windarsih, A.; Sulaiman, A.; Zakaria, Z. Virgin Coconut Oil: Extraction, Physicochemical Properties, Biological Activities and Its Authentication Analysis. Food Rev. Int. 2021, 37, 46–66. [Google Scholar] [CrossRef]
- Cervantes-Paz, B.; Yahia, E.M. Avocado oil: Production and market demand, bioactive components, implications in health, and tendencies and potential uses. Compr. Rev. Food Sci. Food Saf. 2021, 20, 4120–4158. [Google Scholar] [CrossRef] [PubMed]
- Drinić, Z.; Mudrić, J.; Zdunić, G.; Bigović, D.; Menković, N.; Šavikin, K. Effect of pomegranate peel extract on the oxidative stability of pomegranate seed oil. Food Chem. 2020, 333, 127501. [Google Scholar] [CrossRef] [PubMed]
- Paul, A.; Radhakrishnan, M. Pomegranate seed oil in food industry: Extraction, characterization, and applications. Trends Food Sci. Technol. 2020, 105, 273–283. [Google Scholar] [CrossRef]
- Brodkorb, A.; Egger, L.; Alminger, M.; Alvito, P.; Assunção, R.; Ballance, S.; Bohn, T.; Bourlieu-Lacanal, C.; Boutrou, R.; Carrière, F.; et al. INFOGEST static in vitro simulation of gastrointestinal food digestion. Nat. Protoc. 2019, 14, 991–1014. [Google Scholar] [CrossRef]
- Machado, M.; Costa, E.M.; Silva, S.; Rodriguez-Alcalá, L.M.; Gomes, A.M.; Pintado, M. Pomegranate Oil’s Potential as an Anti-Obesity Ingredient. Molecules 2022, 27, 4958. [Google Scholar] [CrossRef]
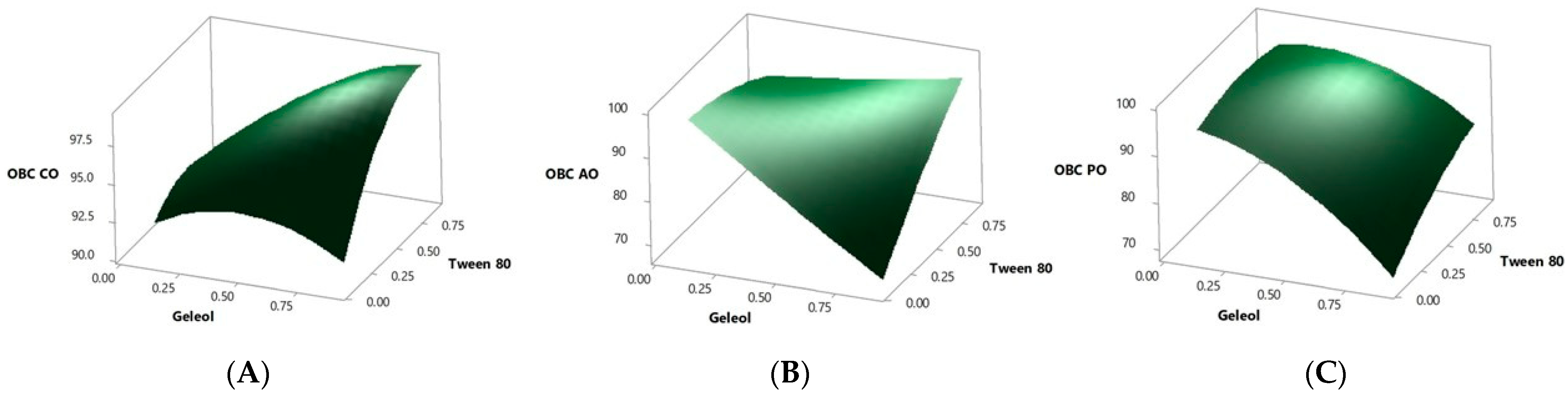


| Run Order | Pt Type | Blocks | Geleol | Tween 80 | OBC CO | OBC AO | OBC PO |
|---|---|---|---|---|---|---|---|
| 1 | 0 | 2 | 0.50 | 0.50 | 97.24 | 88.37 | 91.79 |
| 2 | 0 | 2 | 0.50 | 0.50 | 97.35 | 88.35 | 91.71 |
| 3 | −1 | 2 | 0.90 | 0.50 | 98.59 | 88.12 | 84.56 |
| 4 | −1 | 2 | 0.10 | 0.50 | 93.02 | 95.57 | 94.07 |
| 5 | −1 | 2 | 0.50 | 0.90 | 98.66 | 96.67 | 97.24 |
| 6 | 0 | 2 | 0.50 | 0.50 | 97.15 | 88.31 | 91.67 |
| 7 | −1 | 2 | 0.50 | 0.10 | 92.39 | 82.84 | 86.11 |
| 8 | −1 | 2 | 0.50 | 0.90 | 98.66 | 96.67 | 97.24 |
| 9 | 0 | 2 | 0.50 | 0.50 | 97.23 | 88.36 | 91.69 |
| 10 | −1 | 2 | 0.10 | 0.50 | 93.02 | 95.57 | 94.07 |
| 11 | −1 | 2 | 0.50 | 0.10 | 92.39 | 82.84 | 86.11 |
| 12 | −1 | 2 | 0.90 | 0.50 | 98.59 | 88.12 | 84.56 |
| 13 | 0 | 2 | 0.50 | 0.50 | 97.25 | 88.32 | 91.73 |
| 14 | 0 | 2 | 0.50 | 0.50 | 97.26 | 88.38 | 91.78 |
| 15 | 0 | 1 | 0.50 | 0.50 | 97.22 | 88.40 | 97.74 |
| 16 | 1 | 1 | 0.78 | 0.78 | 96.85 | 85.42 | 81.82 |
| 17 | 1 | 1 | 0.78 | 0.22 | 94.53 | 72.64 | 74.76 |
| 18 | 1 | 1 | 0.22 | 0.78 | 92.89 | 87.69 | 96.58 |
| 19 | 0 | 1 | 0.50 | 0.50 | 97.21 | 88.37 | 91.69 |
| 20 | 1 | 1 | 0.22 | 0.22 | 95.19 | 93.04 | 96.74 |
| 21 | 1 | 1 | 0.22 | 0.78 | 92.89 | 87.69 | 96.58 |
| 22 | 0 | 1 | 0.50 | 0.50 | 97.23 | 88.39 | 91.78 |
| 23 | 0 | 1 | 0.50 | 0.50 | 97.27 | 88.41 | 91.62 |
| 24 | 0 | 1 | 0.50 | 0.50 | 97.24 | 88.27 | 91.77 |
| 25 | 0 | 1 | 0.50 | 0.50 | 97.24 | 88.35 | 91.79 |
| 26 | 1 | 1 | 0.22 | 0.22 | 95.19 | 93.04 | 96.74 |
| 27 | 1 | 1 | 0.78 | 0.22 | 94.53 | 72.64 | 74.76 |
| 28 | 1 | 1 | 0.78 | 0.78 | 96.85 | 85.42 | 81.82 |
| Avocado Bigel | Avocado Oil | Coconut Bigel | Coconut Oil | Pomegranate Bigel | Pomegranate Oil | |||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Fatty Acid | Sample | Digested | Sample | Digested | Sample | Digested | Sample | Digested | Sample | Digested | Sample | Digested |
| C6 | - | - | - | - | 0.95 ± 0.08 a | 0.56 ± 0.00 b | 0.17 ± 0.00 a | 0.06 ± 0.02 b | - | - | - | - |
| C8 | - | - | - | - | 16.39 ± 1.69 a | 10.71 ± 0.10 b | 2.26 ± 0.01 a | 2.43 ± 0.49 b | - | - | - | - |
| C10 | - | - | - | - | 15.52 ± 1.77 a | 11.02 ± 0.33 b | 2.26 ± 0.01 a | 1.30 ± 0.06 b | - | - | - | - |
| C12 | - | - | - | - | 161.12 ± 19.87 a | 119.45 ± 8.91 b | 23.62 ± 0.08 a | 0.92 ± 0.18 b | - | - | - | - |
| C14 | 0.18 ± 0.12 a | 0.08 ± 0.02 a | 0.13 ± 0.00 | n.d. | 79.71 ± 10.05 a | 5.15 ± 0.00 b | 11.50 ± 0.04 a | 0.68 ± 0.09 b | - | - | - | - |
| C16 | 41.28 ± 9.63 a | 23.96 ± 7.99 a | 64.50 ± 1.12 a | 9.30 ± 3.61 b | 93.49 ± 15.74 a | 59.63 ± 7.52 b | 5.33 ± 0.02 a | 0.36 ± 0.04 b | 19.87 ± 4.75 a | 10.76 ± 5.03 a | 6.44 ± 0.22 a | 1.16 ± 0.11 b |
| C16:1 c7 | 0.21 ± 0.04 a | 0.16 ± 0.05 a | 0.32 ± 0.01 a | 0.11 ± 0.04 b | - | - | - | - | - | - | - | - |
| C16:1 c9 | 8.94 ± 2.23 a | 5.22 ± 1.82 a | 14.95 ± 0.26 a | 1.75 ± 0.58 b | - | - | - | - | - | - | - | - |
| C17 | 0.12 ± 0.02 a | 0.09 ± 0.03 a | 0.18 ± 0.00 a | 0.13 ± 0.06 a | - | - | - | - | - | - | - | - |
| C17:1 c10 | 0.22 ± 0.05 a | 0.12 ± 0.04 a | 0.35 ± 0.00 a | 0.09 ± 0.01 b | - | - | - | - | - | - | - | - |
| C18 | 9.49 ± 2.03 a | 6.69 ± 2.18 a | 7.89 ± 0.13 a | 2.64 ± 1.17 b | 86.26 ± 24.41 a | 0.08 ± 0.01 b | 2.34 ± 0.01 a | 0.17 ± 0.02 b | 19.65 ± 5.19 a | 9.80 ± 4.78 a | 5.18 ± 0.18 a | 0.77 ± 0.07 b |
| C18.1 t12 | - | - | - | - | - | - | - | - | 0.28 ± 0.06 a | 0.20 ± 0.11 a | 0.12 ± 0.00 a | 0.11 ± 0.01 b |
| C18:1 c9 | 132.01 ± 29.55 a | 70.27 ± 24.59 b | 227.40 ± 3.96 a | 27.13 ± 3.43 b | 53.29 ± 2.82 a | 0.53 ± 0.31 b | 3.42 ± 0.01 a | 0.24 ± 0.02 b | 36.62 ± 6.98 a | 18.39 ± 9.29 b | 12.60 ± 0.43 a | 0.10 ± 0.0 b |
| C18:1 c11 | 9.89 ± 2.15 a | 5.26 ± 1.87 a | 16.77 ± 0.29 a | 1.87 ± 0.70 b | - | - | - | - | 1.65 ± 0.31 a | 0.67 ± 0.28 b | 0.93 ± 0.03 a | n.d. |
| C18:1 c6 | - | - | - | - | - | - | - | - | 0.89 ± 0.16 a | 0.34 ± 0.15 b | 0.63 ± 0.02 a | n.d. |
| C18:2 t9 c12 | 0.19 ± 0.04 a | 0.12 ± 0.04 a | 0.29 ± 0.00 a | 0.07 ± 0.00 b | - | - | - | - | - | - | - | - |
| C18:2 | 26.47 ± 6.67 a | 15.95 ± 5.52 a | 44.16 ± 0.77 a | 7.03 ± 2.74 b | 3.93 ± 0.48 a | 2.46 ± 0.22 b | 0.54 ± 0.00 a | 0.06 ± 0.01 b | 19.83 ± 3.52 a | 8.06 ± 3.26 b | 12.37 ± 0.43 a | 0.06 ± 0.0 b |
| γC18:3 | 0.98 ± 0.25 a | 0.65 ± 0.19 a | 1.58 ± 0.03 a | 0.24 ± 0.08 b | - | - | - | - | 0.08 ± 0.03 a | 0.03 ± 0.00 a | 1.13 ± 0.04 a | 0.0.6 ± 0.01 b |
| C20:1 | 0.79 ± 0.09 a | 0.38 ± 0.11 b | 1.39 ± 0.02 a | 0.13 ± 0.05 b | - | - | - | - | - | - | - | - |
| C18:3 | 0.56 ± 0.05 a | 0.27 ± 0.09 b | 0.93 ± 0.02 a | 0.14 ± 0.07 b | - | - | - | - | 2.91 ± 0.58 a | 0.07 ± 0.03 b | 1.68 ± 0.06 a | 0.08 ± 0.01 b |
| C22:1 | - | - | - | - | - | - | - | - | 0.50 ± 0.13 | n.d. | 0.04 ± 0.00 a | n.d. |
| C18:3 c9 t11 c13 | - | - | - | - | - | - | - | - | 220.25 ± 37.40 a | 92.26 ± 41.31 b | 140.77 ± 4.86 a | 3.03 ± 0.78 b |
| C18:3 c9 t11 t13 | - | - | - | - | - | - | - | - | 7.27 ± 1.01 a | 3.17 ± 1.29 b | 4.35 ± 0.15 a | 0.18 ± 0.15 b |
| C18:3 t9 t11 c13 | - | - | - | - | - | - | - | - | 0.78 ± 0.26 a | 0.63 ± 0.32 a | 1.96 ± 0.07 a | 0.14 ± 0.12 b |
| C18: 3 t9 t11 t13 | - | - | - | - | - | - | - | - | 3.61 ± 0.77 a | 0.64 ± 0.32 b | 0.49 ± 0.02 a | 0.09 ± 0.01 b |
| C20:3 | 0.27 ± 0.03 a | 0.11 ± 0.03 b | 0.47 ± 0.00 a | 0.13 ± 0.01 a | - | - | - | - | - | - | - | - |
| C20:5 | 0.18 ± 0.00 a | 0.12 ± 0.07 a | 0.29 ± 0.00 | n.d. | - | - | - | - | - | - | - | - |
| C24 | 0.20 ± 0.03 a | 0.08 ± 0.03 a | 0.36 ± 0.00 | n.d. | - | - | - | - | - | - | - | - |
| ∑Fatty acids | 231.99 ± 53.93 a | 129.51 ± 44.67 b | 381.96 ± 6.63 a | 50.77 ± 12.53 b | 510.67 ± 3.92 a | 209.59 ± 16.16 b | 51.45 ± 0.18 a | 6.19 ± 0.71 b | 334.48 ± 61.23 a | 145.10 ± 66.20 b | 188.68 ± 6.51 a | 6.95 ± 3.42 b |
| ∑SFAs | 51.27 ± 11.76 a | 30.89 ± 10.23 a | 73.06 ± 1.27 a | 12.07 ± 4.84 b | 453.44 ± 6.70 a | 20.59 ± 16.88 b | 47.48 ± 0.17 a | 5.88 ± 0.73 b | 39.82 ± 10.02 a | 20.63 ± 9.84 a | 11.62 ± 0.40 a | 1.93 ± 0.18 b |
| ∑MUFAs | 152.20 ± 34.17 a | 81.48 ± 28.52 b | 261.17 ± 4.54 a | 31.07 ± 4.81 b | 53.85 ± 2.58 a | 0.53 ± 0.31 b | 3.42 ± 0.01 a | 0.24 ± 0.03 b | 39.94 ± 7.64 a | 19.60 ± 9.83 a | 14.31 ± 0.45 a | 0.21 ± 0.01 b |
| ∑PUFAs | 26.65 ± 6.97 a | 17.21 ± 5.94 a | 47.72 ± 0.82 a | 7.61 ± 2.88 b | 3.93 ± 0.21 a | 2.46 ± 0.27 a | 0.54 ± 0.01 a | 0.07 ± 0.01 b | 254.72 ± 4.57 a | 104.07 ± 46.52 b | 162.75 ± 5.61 a | 4.80 ± 3.61 b |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Machado, M.; Sousa, S.C.; Rodríguez-Alcalá, L.M.; Pintado, M.; Gomes, A.M. Bigels as Delivery Systems of Bioactive Fatty Acids Present in Functional Edible Oils: Coconut, Avocado, and Pomegranate. Gels 2023, 9, 349. https://doi.org/10.3390/gels9040349
Machado M, Sousa SC, Rodríguez-Alcalá LM, Pintado M, Gomes AM. Bigels as Delivery Systems of Bioactive Fatty Acids Present in Functional Edible Oils: Coconut, Avocado, and Pomegranate. Gels. 2023; 9(4):349. https://doi.org/10.3390/gels9040349
Chicago/Turabian StyleMachado, Manuela, Sérgio Cruz Sousa, Luís Miguel Rodríguez-Alcalá, Manuela Pintado, and Ana Maria Gomes. 2023. "Bigels as Delivery Systems of Bioactive Fatty Acids Present in Functional Edible Oils: Coconut, Avocado, and Pomegranate" Gels 9, no. 4: 349. https://doi.org/10.3390/gels9040349
APA StyleMachado, M., Sousa, S. C., Rodríguez-Alcalá, L. M., Pintado, M., & Gomes, A. M. (2023). Bigels as Delivery Systems of Bioactive Fatty Acids Present in Functional Edible Oils: Coconut, Avocado, and Pomegranate. Gels, 9(4), 349. https://doi.org/10.3390/gels9040349











