A Novel Effector FlSp1 Inhibits the Colonization of Endophytic Fusarium lateritium and Increases the Resistance to Ralstonia solanacearum in Tobacco
Abstract
1. Introduction
2. Materials and Methods
2.1. Plant Growing Conditions
2.2. Strain Growth Conditions
2.3. Bioinformatic Prediction of Effectors in F. lateritium
2.4. Plasmid Construction
2.5. Genetic Transformation
2.6. Transient Expression of FlSp1
2.7. Phenotypic Analysis
2.8. F. lateritium Co-Cultured with Tobacco
2.9. R. solanacearum Inoculation Treatment of Tobacco
2.10. Histological Staining of Tobacco
2.11. RNA Extraction and RT-qPCR
2.12. Data Processing
3. Results
3.1. Prediction of F. lateritium Effectors
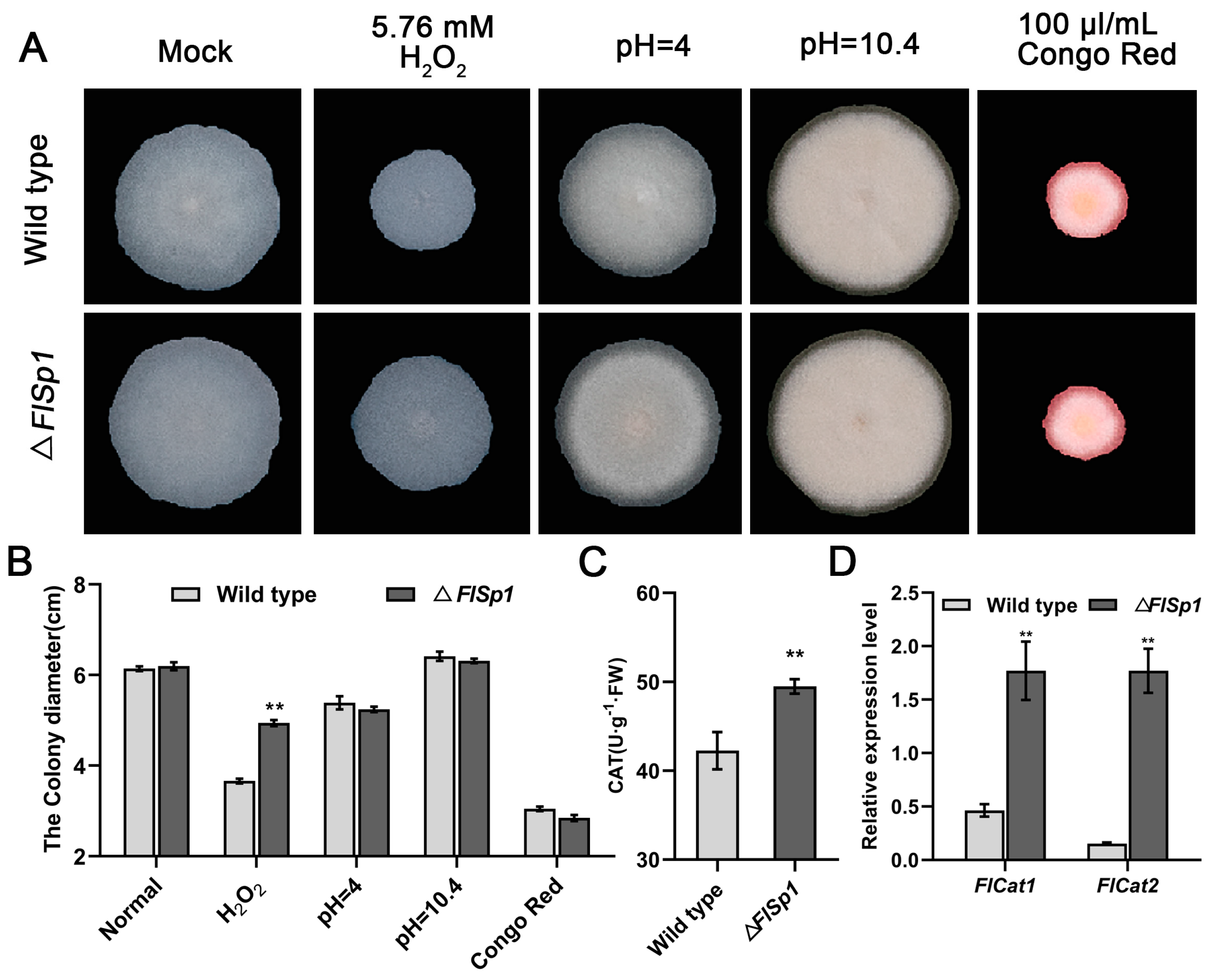
3.2. FlSp1 Negatively Mediates the Oxidative Stress Response in F. lateritium
3.3. FlSp1 Stimulates Plant Immune Defense Response and Accumulates ROS
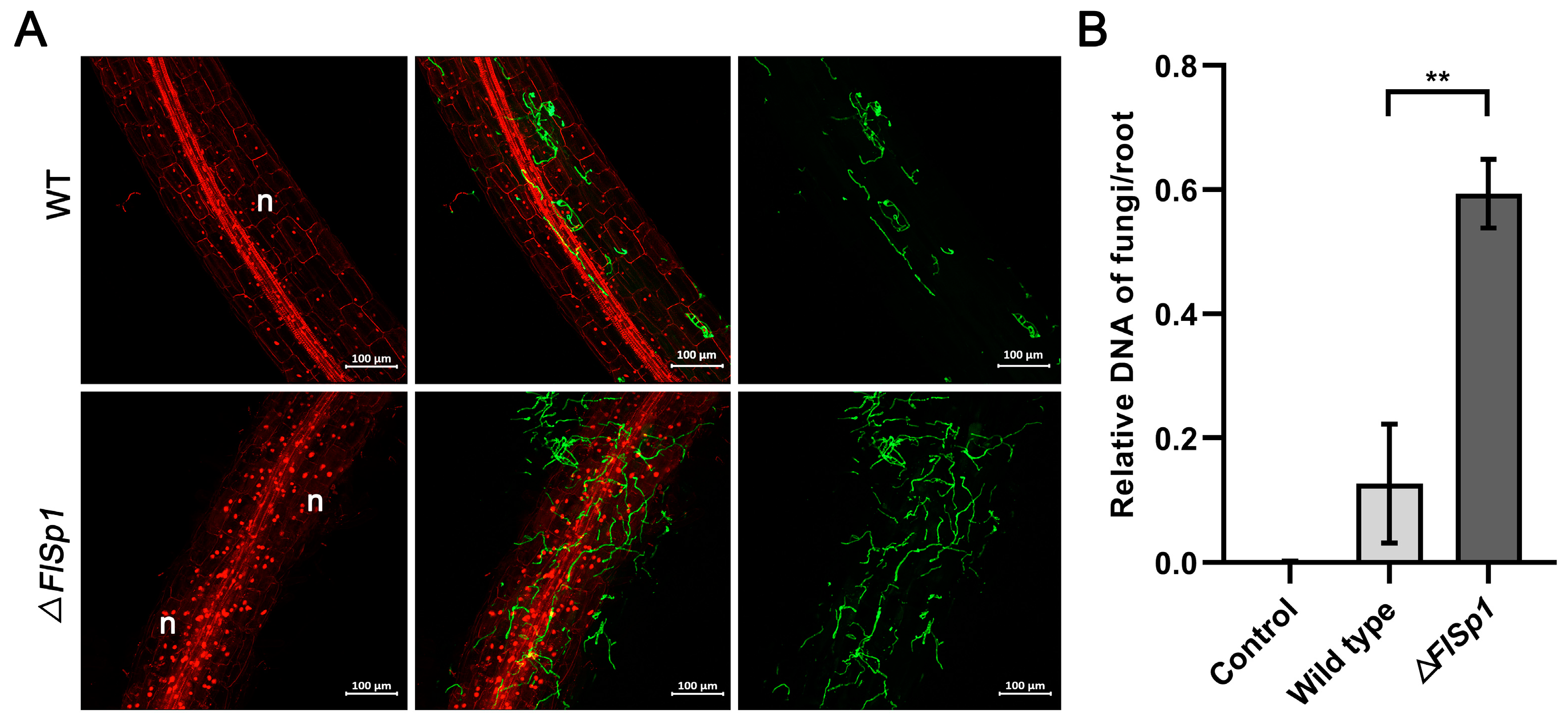
3.4. Disruption of FlSp1 Enhances the Colonization Rate of F. lateritium in Plants
3.5. FlSp1 Negatively Mediates the Resistance of Tobacco to R. solanacearum
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Martel, A.; Ruiz-Bedoya, T.; Breit-McNally, C.; Laflamme, B.; Desveaux, D.; Guttman, D.S. The ETS-ETI Cycle: Evolutionary Processes and Metapopulation Dynamics Driving the Diversification of Pathogen Effectors and Host Immune Factors. Curr. Opin. Plant Biol. 2021, 62, 102011. [Google Scholar] [CrossRef]
- Mittler, R.; Zandalinas, S.I.; Fichman, Y.; Van Breusegem, F. Reactive Oxygen Species Signalling in Plant Stress Responses. Nat. Rev. Mol. Cell Biol. 2022, 23, 663–679. [Google Scholar] [CrossRef]
- Ju, S.; Go, Y.S.; Choi, H.J.; Park, J.M.; Suh, M.C. DEWAX Transcription Factor Is Involved in Resistance to Botrytis Cinerea in Arabidopsis Thaliana and Camelina Sativa. Front. Plant Sci. 2017, 8, 1210. [Google Scholar] [CrossRef] [PubMed]
- Zhao, P.; Zhang, F.; Liu, D.; Imani, J.; Langen, G.; Kogel, K.-H. Matrix Metalloproteinases Operate Redundantly in Arabidopsis Immunity against Necrotrophic and Biotrophic Fungal Pathogens. PLoS ONE 2017, 12, e0183577. [Google Scholar] [CrossRef]
- Wang, M.; Ji, Z.; Yan, H.; Xu, J.; Zhao, X.; Zhou, Z. Effector Sntf2 Interacted with Chloroplast-Related Protein Mdycf39 Promoting the Colonization of Colletotrichum Gloeosporioides in Apple Leaf. Int. J. Mol. Sci. 2022, 23, 6379. [Google Scholar] [CrossRef] [PubMed]
- Qi, T.; Guo, J.; Liu, P.; He, F.; Wan, C.; Islam, M.A.; Tyler, B.M.; Kang, Z.; Guo, J. Stripe Rust Effector PstGSRE1 Disrupts Nuclear Localization of ROS-Promoting Transcription Factor TaLOL2 to Defeat ROS-Induced Defense in Wheat. Mol. Plant 2019, 12, 1624–1638. [Google Scholar] [CrossRef] [PubMed]
- Gao, M.; He, Y.; Yin, X.; Zhong, X.; Yan, B.; Wu, Y.; Chen, J.; Li, X.; Zhai, K.; Huang, Y.; et al. Ca2+ Sensor-Mediated ROS Scavenging Suppresses Rice Immunity and Is Exploited by a Fungal Effector. Cell 2021, 184, 5391–5404.e17. [Google Scholar] [CrossRef]
- Zhu, L.; Li, T.; Wang, C.; Zhang, X.; Xu, L.; Xu, R.; Zhao, Z. The Effects of Dark Septate Endophyte (DSE) Inoculation on Tomato Seedlings under Zn and Cd Stress. Environ. Sci. Pollut. Res. 2018, 25, 35232–35241. [Google Scholar] [CrossRef]
- Mori, M.P.; Penjweini, R.; Knutson, J.R.; Wang, P.; Hwang, P.M. Mitochondria and Oxygen Homeostasis. FEBS J. 2022, 289, 6959–6968. [Google Scholar] [CrossRef]
- Vitale, S.; Santori, A.; Wajnberg, E.; Castagnone-Sereno, P.; Luongo, L.; Belisario, A. Morphological and Molecular Analysis of Fusarium Lateritium, the Cause of Gray Necrosis of Hazelnut Fruit in Italy. Phytopathology 2011, 101, 679–686. [Google Scholar] [CrossRef]
- Xiao, Q. Effects of an Endophytic Fusarium Lateritium on Growth and Disease Resistance of Tomato. J. Huazhong Agric. Univ. 2022, 41, 173–180. [Google Scholar]
- Wang, J. Endophyte Fusarium Lateritium Showing Potato Growth Promotion and Disease Resistanceand the Construction of Its Genetic Transformation System. Mycosystema 2021, 40, 2008–2023. [Google Scholar]
- Zha, X. A Strain of Endophytic Fusarium Lateritium Promotes Growth and Resistance to Bacteriawilt of Tobacco. Mycosystema 2022, 41, 1658–1671. [Google Scholar]
- Sperschneider, J.; Dodds, P.N. EffectorP 3.0: Prediction of Apoplastic and Cytoplasmic Effectors in Fungi and Oomycetes. Mol. Plant-Microbe Interact. 2022, 35, 146–156. [Google Scholar] [CrossRef]
- Jones, D.A.; Bertazzoni, S.; Turo, C.J.; Syme, R.A.; Hane, J.K. Bioinformatic Prediction of Plant–Pathogenicity Effector Proteins of Fungi. Curr. Opin. Microbiol. 2018, 46, 43–49. [Google Scholar] [CrossRef]
- Ma, J.-C.; Zhou, Q.; Zhou, Y.-H.; Liao, X.-G.; Zhang, Y.-J.; Jin, D.; Pei, Y. The Size and Ratio of Homologous Sequence to Non-Homologous Sequence in Gene Disruption Cassette Influences the Gene Targeting Efficiency in Beauveria Bassiana. Appl. Microbiol. Biotechnol. 2009, 82, 891–898. [Google Scholar] [CrossRef] [PubMed]
- Xiang, J.; Li, X.; Yin, L.; Liu, Y.; Zhang, Y.; Qu, J.; Lu, J. A Candidate RxLR Effector from Plasmopara Viticola Can Elicit Immune Responses in Nicotiana Benthamiana. BMC Plant Biol. 2017, 17, 75. [Google Scholar] [CrossRef]
- Zhang, C.; Chen, H.; Cai, T.; Deng, Y.; Zhuang, R.; Zhang, N.; Zeng, Y.; Zheng, Y.; Tang, R.; Pan, R.; et al. Overexpression of a Novel Peanut NBS-LRR Gene AhRRS5 Enhances Disease Resistance to Ralstonia Solanacearum in Tobacco. Plant Biotechnol. J. 2017, 15, 39–55. [Google Scholar] [CrossRef]
- Jing, Y.; Shen, N.; Zheng, X.; Fu, A.; Zhao, F.; Lan, W.; Luan, S. Danger-Associated Peptide Regulates Root Immune Responses and Root Growth by Affecting ROS Formation in Arabidopsis. Int. J. Mol. Sci. 2020, 21, 4590. [Google Scholar] [CrossRef]
- Zhang, L.; Yan, J.; Fu, Z.; Shi, W.; Ninkuu, V.; Li, G.; Yang, X.; Zeng, H. FoEG1, a Secreted Glycoside Hydrolase Family 12 Protein from Fusarium Oxysporum, Triggers Cell Death and Modulates Plant Immunity. Mol. Plant Pathol. 2021, 22, 522–538. [Google Scholar] [CrossRef]
- Palmieri, D.; Vitale, S.; Lima, G.; Di Pietro, A.; Turrà, D. A Bacterial Endophyte Exploits Chemotropism of a Fungal Pathogen for Plant Colonization. Nat. Commun. 2020, 11, 5264. [Google Scholar] [CrossRef]
- Zuccaro, A.; Lahrmann, U.; Güldener, U.; Langen, G.; Pfiffi, S.; Biedenkopf, D.; Wong, P.; Samans, B.; Grimm, C.; Basiewicz, M.; et al. Endophytic Life Strategies Decoded by Genome and Transcriptome Analyses of the Mutualistic Root Symbiont Piriformospora Indica. PLoS Pathog. 2011, 7, e1002290. [Google Scholar] [CrossRef]
- Park, E.; Cho, M.; Ki, C.-S. Correct Use of Repeated Measures Analysis of Variance. Ann. Lab. Med. 2009, 29, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Sperschneider, J.; Gardiner, D.M.; Dodds, P.N.; Tini, F.; Covarelli, L.; Singh, K.B.; Manners, J.M.; Taylor, J.M. E ffector P: Predicting Fungal Effector Proteins from Secretomes Using Machine Learning. New Phytol. 2016, 210, 743–761. [Google Scholar] [CrossRef] [PubMed]
- Kawano, Y.; Akamatsu, A.; Hayashi, K.; Housen, Y.; Okuda, J.; Yao, A.; Nakashima, A.; Takahashi, H.; Yoshida, H.; Wong, H.L.; et al. Activation of a Rac GTPase by the NLR Family Disease Resistance Protein Pit Plays a Critical Role in Rice Innate Immunity. Cell Host Microbe 2010, 7, 362–375. [Google Scholar] [CrossRef]
- Liberman, L.M. A Friend in Need (of Nutrients) Is A…. Cell 2016, 165, 269–271. [Google Scholar] [CrossRef] [PubMed]
- Guo, R.; Ji, S.; Wang, Z.; Zhang, H.; Wang, Y.; Liu, Z. Trichoderma Asperellum Xylanases Promote Growth and Induce Resistance in Poplar. Microbiol. Res. 2021, 248, 126767. [Google Scholar] [CrossRef]
- Pieterse, C.M.J.; Zamioudis, C.; Berendsen, R.L.; Weller, D.M.; Van Wees, S.C.M.; Bakker, P.A.H.M. Induced Systemic Resistance by Beneficial Microbes. Annu. Rev. Phytopathol. 2014, 52, 347–375. [Google Scholar] [CrossRef]
- Contreras-Cornejo, H.A.; Macías-Rodríguez, L.; Beltrán-Peña, E.; Herrera-Estrella, A.; López-Bucio, J. Trichoderma -Induced Plant Immunity Likely Involves Both Hormonal- and Camalexin-Dependent Mechanisms in Arabidopsis Thaliana and Confers Resistance against Necrotrophic Fungi Botrytis Cinerea. Plant Signal. Behav. 2011, 6, 1554–1563. [Google Scholar] [CrossRef]


| GeneID | Effector Probability | (aa) Position of Signal Peptide | (aa) Size of Peptide | Subcellular Localization | Integral Prediction of the Protein Location |
|---|---|---|---|---|---|
| EVM0011051.1 | 0.979 | 1–17 | 108 | Extracellular | 2.4 |
| EVM0014202.1 | 0.971 | 1–23 | 127 | Extracellular | 2.4 |
| EVM0003861.1 | 0.966 | 1–16 | 94 | Extracellular | 2.3 |
| EVM0007002.1 | 0.965 | 1–21 | 97 | Extracellular | 2.4 |
| EVM0005253.1 | 0.959 | 1–18 | 121 | Extracellular | 3.2 |
| EVM0002480.1 | 0.952 | 1–17 | 96 | Extracellular | 2.5 |
| EVM0001443.1 | 0.952 | 1–18 | 97 | Extracellular | 2.4 |
| EVM0000104.1 | 0.952 | 1–18 | 89 | Extracellular | 2.4 |
| EVM0005809.1 | 0.949 | 1–18 | 124 | Extracellular | 2.6 |
| EVM0004957.1 | 0.948 | 1–17 | 98 | Extracellular | 2.6 |
| EVM0001142.1 | 0.943 | 1–19 | 88 | Extracellular | 2.7 |
| EVM0005907.1 | 0.928 | 1–17 | 105 | Extracellular | 2.7 |
| EVM0013376.1 | 0.912 | 1–23 | 131 | Extracellular | 2.4 |
| EVM0007040.1 | 0.91 | 1–17 | 65 | Extracellular | 2.4 |
| EVM0011395.1 | 0.909 | 1–16 | 148 | Extracellular | 2.4 |
| EVM0001445.1 | 0.909 | 1–16 | 140 | Extracellular | 2.8 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Huang, J.; He, Z.; Wang, J.; Zha, X.; Xiao, Q.; Liu, G.; Li, Y.; Kang, J. A Novel Effector FlSp1 Inhibits the Colonization of Endophytic Fusarium lateritium and Increases the Resistance to Ralstonia solanacearum in Tobacco. J. Fungi 2023, 9, 519. https://doi.org/10.3390/jof9050519
Huang J, He Z, Wang J, Zha X, Xiao Q, Liu G, Li Y, Kang J. A Novel Effector FlSp1 Inhibits the Colonization of Endophytic Fusarium lateritium and Increases the Resistance to Ralstonia solanacearum in Tobacco. Journal of Fungi. 2023; 9(5):519. https://doi.org/10.3390/jof9050519
Chicago/Turabian StyleHuang, Jianming, Zhangjiang He, Jiankang Wang, Xingping Zha, Qing Xiao, Guihua Liu, Yongjie Li, and Jichuan Kang. 2023. "A Novel Effector FlSp1 Inhibits the Colonization of Endophytic Fusarium lateritium and Increases the Resistance to Ralstonia solanacearum in Tobacco" Journal of Fungi 9, no. 5: 519. https://doi.org/10.3390/jof9050519
APA StyleHuang, J., He, Z., Wang, J., Zha, X., Xiao, Q., Liu, G., Li, Y., & Kang, J. (2023). A Novel Effector FlSp1 Inhibits the Colonization of Endophytic Fusarium lateritium and Increases the Resistance to Ralstonia solanacearum in Tobacco. Journal of Fungi, 9(5), 519. https://doi.org/10.3390/jof9050519





