Assessing the Importance of Native Mycorrhizal Fungi to Improve Tree Establishment after Wildfires
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Site and Target Species
2.2. Root Sampling and Fungal Inoculum
2.3. Assessment of the Survival and Photosynthesis in the Inoculated and Non-Inoculated Trees
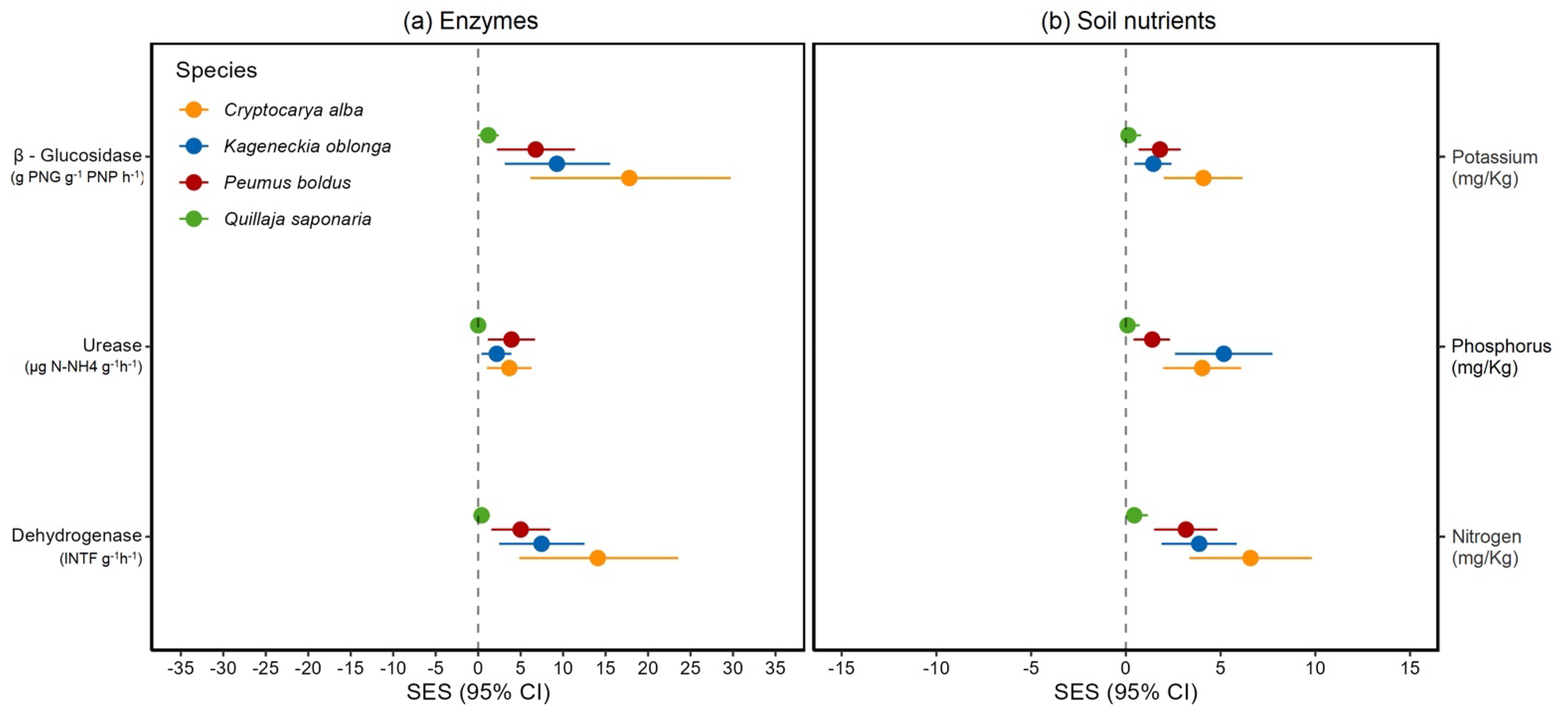
2.4. Enzymatic Activity and Nutritional Effect of the Mycorrhizal Inoculation on the Soil
2.5. Statistical Analyses
3. Results
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Brundrett, M.C.; Tedersoo, L. Evolutionary history of mycorrhizal symbioses and global host plant diversity. New Phytol. 2018, 220, 1108–1115. [Google Scholar] [CrossRef] [PubMed]
- Smith, S.E.; Read, D.J. Mycorrhizal Symbiosis, 3rd ed.; Elsevier Ltd.: London, UK, 2008; p. 800. [Google Scholar]
- Jiang, Y.; Wang, W.; Xie, Q.; Liu, N.; Liu, L.; Wang, D.; Zhang, X.; Yang, C.; Chen, X.; Tang, D.; et al. Plants transfer lipids to sustain colonization by mutualistic mycorrhizal and parasitic fungi. Science 2017, 356, 1172–1175. [Google Scholar] [CrossRef] [PubMed]
- Keymer, A.; Pimprikar, P.; Wewer, V.; Huber, C.; Brands, M.; Bucerius, S.L.; Delaux, P.-M.; Klingl, V.; Röpenack-Lahaye, E.v.; Wang, T.L.; et al. Lipid transfer from plants to arbuscular mycorrhiza fungi. eLife 2017, 6, e29107. [Google Scholar] [CrossRef]
- Luginbuehl, L.H.; Menard, G.N.; Kurup, S.; Van Erp, H.; Radhakrishnan, G.V.; Breakspear, A.; Oldroyd, G.E.D.; Eastmond, P.J. Fatty acids in arbuscular mycorrhizal fungi are synthesized by the host plant. Science 2017, 356, 1175–1178. [Google Scholar] [CrossRef]
- Li, J.; Meng, B.; Chai, H.; Yang, X.; Song, W.; Li, S.; Lu, A.; Zhang, T.; Sun, W. Arbuscular mycorrhizal fungi alleviate drought stress in C3 (Leymus chinensis) and C4 (Hemarthria altissima) grasses via altering antioxidant enzyme activities and photosynthesis. Front. Plant Sci. 2019, 10, 499. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Wu, X.; Li, G.; Qin, P. Interactions between arbuscular mycorrhizal fungi and phosphate-solubilizing fungus (Mortierella sp.) and their effects on Kostelelzkya virginica growth and enzyme activities of rhizosphere and bulk soils at different salinities. Biol. Fertil. Soils 2011, 47, 543–554. [Google Scholar] [CrossRef]
- Bowles, T.M.; Jackson, L.E.; Cavagnaro, T.R. Mycorrhizal fungi enhance plant nutrient acquisition and modulate nitrogen loss with variable water regimes. Glob. Chang. Biol. 2018, 24, e171–e182. [Google Scholar] [CrossRef]
- van der Heijden, M.G.A.; Martin, F.M.; Selosse, M.A.; Sanders, I.R. Mycorrhizal ecology and evolution: The past, the present, and the future. New Phytol. 2015, 205, 1406–1423. [Google Scholar] [CrossRef] [PubMed]
- Bray, S.R.; Kitajima, K.; Sylvia, D.M. Mycorrhizae differentially alter growth, physiology, and competitive ability of an invasive shrub. Ecol. Appl. 2003, 13, 565–574. [Google Scholar] [CrossRef]
- Sielaff, A.C.; Polley, H.W.; Fuentes-Ramirez, A.; Hofmockel, K.; Wilsey, B.J. Mycorrhizal colonization and its relationship with plant performance differs between exotic and native grassland plant species. Biol. Invasions 2019, 21, 1981–1991. [Google Scholar] [CrossRef]
- Bever, J.D.; Mangan, S.A.; Alexander, H.M. Maintenance of plant species diversity by pathogens. Annu. Rev. Ecol. Evol. Syst. 2015, 46, 305–325. [Google Scholar] [CrossRef]
- Neuenkamp, L.; Prober, S.M.; Price, J.N.; Zobel, M.; Standish, R.J. Benefits of mycorrhizal inoculation to ecological restoration depend on plant functional type, restoration context and time. Fungal Ecol. 2019, 40, 140–149. [Google Scholar] [CrossRef]
- Wardle, D.A.; Bardgett, R.D.; Klironomos, J.N.; Setälä, H.; van der Putten, W.H.; Wall, D.H. Ecological linkages between aboveground and belowground biota. Science 2004, 304, 1629–1633. [Google Scholar] [CrossRef] [PubMed]
- Gerrits, G.M.; Waenink, R.; Aradottir, A.L.; Buisson, E.; Dutoit, T.; Ferreira, M.C.; Fontaine, J.B.; Jaunatre, R.; Kardol, P.; Loeb, R.; et al. Synthesis on the effectiveness of soil translocation for plant community restoration. J. Appl. Ecol. 2023. [Google Scholar] [CrossRef]
- Underwood, E.C.; Viers, J.H.; Klausmeyer, K.R.; Cox, R.L.; Shaw, M.R. Threats and biodiversity in the mediterranean biome. Divers. Distrib. 2009, 15, 188–197. [Google Scholar] [CrossRef]
- Armesto, J.J.; Arroyo, M.; Hinojosa, L. The Mediterranean environment of central Chile. In The Physical Geography of South America; Veblen, T.T., Young, K.R., Orme, A.R., Eds.; Oxford University Press, Inc.: New York, NY, USA, 2007; pp. 184–199. [Google Scholar]
- Úbeda, X.; Sarricolea, P. Wildfires in Chile: A review. Glob. Planet. Chang. 2016, 146, 152–161. [Google Scholar] [CrossRef]
- Becerra, P.I. Invasión de árboles alóctonos en una cuenca pre-andina de Chile central. Gayana Botánica 2006, 63, 161–174. [Google Scholar] [CrossRef]
- Montoya-Tangarife, C.; De La Barrera, F.; Salazar, A.; Inostroza, L. Monitoring the effects of land cover change on the supply of ecosystem services in an urban region: A study of Santiago-Valparaíso, Chile. PLoS ONE 2017, 12, e0188117. [Google Scholar] [CrossRef]
- Instituto Nacional de Estadísticas. Available online: https://www.ine.cl (accessed on 15 December 2022).
- Pliscoff, P.; Fuentes-Castillo, T. Modelación de la distribución de especies y ecosistemas en el tiempo y en el espacio: Una revisión de las nuevas herramientas y enfoques disponibles. Rev. Geogr. Norte Gd. 2011, 48, 61–79. [Google Scholar] [CrossRef]
- Durán, A.P.; Casalegno, S.; Marquet, P.A.; Gaston, K.J. Representation of ecosystem services by terrestrial protected areas: Chile as a case study. PLoS ONE 2013, 8, e82643. [Google Scholar] [CrossRef]
- Bannister, J.R.; Vidal, O.J.; Teneb, E.; Sandoval, V. Latitudinal patterns and regionalization of plant diversity along a 4270-km gradient in continental Chile. Austral Ecol. 2012, 37, 500–509. [Google Scholar] [CrossRef]
- Rodriguez, R.; Marticorena, C.; Alarcón, D.; Baeza, C.; Cavieres, L.; Finot, V.L.; Fuentes, N.; Kiessling, A.; Mihoc, M.; Pauchard, A.; et al. Catálogo de las plantas vasculares de Chile. Gayana Botánica 2018, 75, 1–430. [Google Scholar] [CrossRef]
- Van De Water, K.M.; Safford, H.D. A summary of fire frequency estimates for california vegetation before euro-american settlement. Fire Ecol. 2011, 7, 26–58. [Google Scholar] [CrossRef]
- Reszka, P.; Fuentes, A. The great valparaiso fire and fire safety management in chile. Fire Technol. 2015, 51, 753–758. [Google Scholar] [CrossRef]
- Castillo, S.M.; Plaza, V.Á.; Garfias, S.R. A recent review of fire behavior and fire effects on native vegetation in Central Chile. Glob. Ecol. Conserv. 2020, 24, e01210. [Google Scholar] [CrossRef]
- Elia, M.; Giannico, V.; Spano, G.; Lafortezza, R.; Sanesi, G. Likelihood and frequency of recurrent fire ignitions in highly urbanised Mediterranean landscapes. Int. J. Wildland Fire 2020, 29, 120–131. [Google Scholar] [CrossRef]
- Syphard, A.D.; Radeloff, V.C.; Hawbaker, T.J.; Stewart, S.I. Conservation threats due to human-caused increases in fire frequency in Mediterranean-climate ecosystems. Conserv. Biol. 2009, 23, 758–769. [Google Scholar] [CrossRef]
- Valderrama, L.; Contreras-Reyes, J.E.; Carrasco, R. Ecological impact of forest fires and subsequent restoration in Chile. Resources 2018, 7, 26. [Google Scholar] [CrossRef]
- Reazin, C.; Morris, S.; Smith, J.E.; Cowan, A.D.; Jumpponen, A. Fires of differing intensities rapidly select distinct soil fungal communities in a Northwest US ponderosa pine forest ecosystem. For. Ecol. Manag. 2016, 377, 118–127. [Google Scholar] [CrossRef]
- Adams, M.A. Mega-fires, tipping points and ecosystem services: Managing forests and woodlands in an uncertain future. For. Ecol. Manag. 2013, 294, 250–261. [Google Scholar] [CrossRef]
- Ngole-Jeme, V.M. Fire-induced changes in soil and implications on soil sorption capacity and remediation methods. Appl. Sci. 2019, 9, 3447. [Google Scholar] [CrossRef]
- Vilà-Cabrera, A.; Saura-Mas, S.; Lloret, F. Effects of fire frequency on species composition in a Mediterranean shrubland. Ecoscience 2008, 15, 519–528. [Google Scholar] [CrossRef]
- Figueroa, J.A.; Cavieres, L.A. The effect of heat and smoke on the emergence of exotic and native seedlings in a Mediterranean fire-free matorral of central Chile. Rev. Chil. Hist. Nat. 2012, 85, 101–111. [Google Scholar] [CrossRef]
- Contreras, T.E.; Figueroa, J.A.; Abarca, L.; Castro, S.A. Fire regimen and spread of plants naturalized in central Chile. Rev. Chil. Hist. Nat. 2011, 84, 307–323. [Google Scholar] [CrossRef]
- Morales, N.S.; Fernández, I.C.; Duran, L.P.; Venegas-González, A. Community-driven post-fire restoration initiatives in Central Chile: When good intentions are not enough. Restor. Ecol. 2021, 29, e13389. [Google Scholar] [CrossRef]
- Bahadur, A.; Batool, A.; Nasir, F.; Jiang, S.; Mingsen, Q.; Zhang, Q.; Pan, J.; Liu, Y.; Feng, H. Mechanistic insights into arbuscular mycorrhizal fungi-mediated drought stress tolerance in plants. Int. J. Mol. Sci. 2019, 20, 4199. [Google Scholar] [CrossRef]
- Stoof, C.R.; Ferreira, A.J.D.; Mol, W.; Van den Berg, J.; De Kort, A.; Drooger, S.; Slingerland, E.C.; Mansholt, A.U.; Ferreira, C.S.S.; Ritsema, C.J. Soil surface changes increase runoff and erosion risk after a low–moderate severity fire. Geoderma 2015, 239–240, 58–67. [Google Scholar] [CrossRef]
- Stoof, C.R.; Wesseling, J.G.; Ritsema, C.J. Effects of fire and ash on soil water retention. Geoderma 2010, 159, 276–285. [Google Scholar] [CrossRef]
- Booth, E.M.; Creacy, G.; Fowler, N.L. Burn severity and post-fire erosion control affect post-fire woody plant recruitment. Nat. Areas J. 2019, 39, 188, 189–196. [Google Scholar] [CrossRef]
- Van Der Heijden, M.G.A.; Horton, T.R. Socialism in soil? The importance of mycorrhizal fungal networks for facilitation in natural ecosystems. J. Ecol. 2009, 97, 1139–1150. [Google Scholar] [CrossRef]
- Mahmoudi, N.; Dias, T.; Mahdhi, M.; Cruz, C.; Mars, M.; Caeiro, M.F. Does arbuscular mycorrhiza determine soil microbial functionality in nutrient-limited mediterranean arid ecosystems? Diversity 2020, 12, 234. [Google Scholar] [CrossRef]
- Muneer, M.A.; Wang, P.; Lin, C.; Ji, B. Potential role of common mycorrhizal networks in improving plant growth and soil physicochemical properties under varying nitrogen levels in a grassland ecosystem. Glob. Ecol. Conserv. 2020, 24, e01352. [Google Scholar] [CrossRef]
- Yu, L.; Zhang, H.; Zhang, W.; Liu, K.; Liu, M.; Shao, X. Cooperation between arbuscular mycorrhizal fungi and plant growth-promoting bacteria and their effects on plant growth and soil quality. PeerJ 2022, 10, e13080. [Google Scholar] [CrossRef] [PubMed]
- Godoy, R.; Silva-Flores, P.; Aguilera, P.; Marín, C. Microbial interactions in the plant-soil continuum: Research results presented at the Workshop “Mycorrhizal Symbiosis in the Southern Cone of South America”. J. Soil Sci. Plant Nutr. 2017, 17, 1–3. [Google Scholar]
- Soudzilovskaia, N.A.; Vaessen, S.; Barcelo, M.; He, J.; Rahimlou, S.; Abarenkov, K.; Brundrett, M.C.; Gomes, S.I.F.; Merckx, V.; Tedersoo, L. FungalRoot: Global online database of plant mycorrhizal associations. New Phytol. 2020, 227, 955–966. [Google Scholar] [CrossRef]
- Zang, X.; Liu, M.; Fan, Y.; Xu, J.; Xu, X.; Li, H. The structural and functional contributions of β-glucosidase-producing microbial communities to cellulose degradation in composting. Biotechnol. Biofuels 2018, 11, 51. [Google Scholar] [CrossRef]
- Witte, C.P. Urea metabolism in plants. Plant Sci. 2011, 180, 431–438. [Google Scholar] [CrossRef]
- Araújo, W.L.; Tohge, T.; Ishizaki, K.; Leaver, C.J.; Fernie, A.R. Protein degradation—An alternative respiratory substrate for stressed plants. Trends Plant Sci. 2011, 16, 489–498. [Google Scholar] [CrossRef]
- Bell, C.; Carrillo, Y.; Boot, C.M.; Rocca, J.D.; Pendall, E.; Wallenstein, M.D. Rhizosphere stoichiometry: Are C:N:P ratios of plants, soils, and enzymes conserved at the plant species-level? New Phytol. 2014, 201, 505–517. [Google Scholar] [CrossRef]
- R CoreTeam. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2022. [Google Scholar]
- Wickham, H.; Averick, M.; Bryan, J.; Chang, W.; McGowan, L.D.; François, R.; Grolemund, G.; Hayes, A.; Henry, L.; Hester, J.; et al. Welcome to the tidyverse. J. Open Source Softw. 2019, 43, 1686. [Google Scholar] [CrossRef]
- Ben-Shachar, M.S.; Lüdecke, D.; Makowski, D. effectsize: Estimation of effect size indices and standardized parameters. J. Open Source Softw. 2020, 56, 2815. [Google Scholar] [CrossRef]
- Ouahmane, L.; Hafidi, M.; Thioulouse, J.; Ducousso, M.; Kisa, M.; Prin, Y.; Galiana, A.; Boumezzough, A.; Duponnois, R. Improvement of Cupressus atlantica Gaussen growth by inoculation with native arbuscular mycorrhizal fungi. J. Appl. Microbiol. 2007, 103, 683–690. [Google Scholar] [CrossRef]
- Richard, F.; Selosse, M.-A.; Gardes, M. Facilitated establishment of Quercus ilex in shrub-dominated communities within a Mediterranean ecosystem: Do mycorrhizal partners matter? FEMS Microbiol. Ecol. 2009, 68, 14–24. [Google Scholar] [CrossRef] [PubMed]
- Luebert, F.; Pliscoff, P. Sinopsis Bioclimática y Vegetacional de Chile, 2nd ed.; Universitaria: Santiago, Chile, 2018; p. 384. [Google Scholar]
- Zhang, L.; Feng, G.; Declerck, S. Signal beyond nutrient, fructose, exuded by an arbuscular mycorrhizal fungus triggers phytate mineralization by a phosphate solubilizing bacterium. ISME J. 2018, 12, 2339–2351. [Google Scholar] [CrossRef] [PubMed]
- Augé, R.M.; Toler, H.D.; Saxton, A.M. Arbuscular mycorrhizal symbiosis alters stomatal conductance of host plants more under drought than under amply watered conditions: A meta-analysis. Mycorrhiza 2015, 25, 13–24. [Google Scholar] [CrossRef] [PubMed]
- Chandrasekaran, M.; Chanratana, M.; Kim, K.; Seshadri, S.; Sa, T. Impact of arbuscular mycorrhizal fungi on photosynthesis, water status, and gas exchange of plants under salt stress—A meta-analysis. Front. Plant Sci. 2019, 10, 457. [Google Scholar] [CrossRef]
- Wang, Y.; Wang, J.; Yan, X.; Sun, S.; Lin, J. The effect of arbuscular mycorrhizal fungi on photosystem II of the host plant under salt stress: A meta-analysis. Agronomy 2019, 9, 806. [Google Scholar] [CrossRef]
- Francis, R.; Read, D.J. Mutualism and antagonism in the mycorrhizal symbiosis, with special reference to impacts on plant community structure. Can. J. Bot. 1995, 73, 1301–1309. [Google Scholar] [CrossRef]
- Jones, M.D.; Smith, S.E. Exploring functional definitions of mycorrhizas: Are mycorrhizas always mutualisms? Can. J. Bot. 2004, 82, 1089–1109. [Google Scholar] [CrossRef]
- Müller, J. Artificial infection by endophytes affects growth and mycorrhizal colonisation of Lolium perenne. Funct. Plant Biol. 2003, 30, 419–424. [Google Scholar] [CrossRef]
- Mack, K.M.L.; Rudgers, J.A. Balancing multiple mutualists: Asymmetric interactions among plants, arbuscular mycorrhizal fungi, and fungal endophytes. Oikos 2008, 117, 310–320. [Google Scholar] [CrossRef]
- Liu, Q.; Parsons, A.J.; Xue, H.; Fraser, K.; Ryan, G.D.; Newman, J.A.; Rasmussen, S. Competition between foliar Neotyphodium lolii endophytes and mycorrhizal Glomus spp. fungi in Lolium perenne depends on resource supply and host carbohydrate content. Funct. Ecol. 2011, 25, 910–920. [Google Scholar] [CrossRef]
- Aghababaei, F.; Raiesi, F.; Hosseinpur, A. The combined effects of earthworms and arbuscular mycorrhizal fungi on microbial biomass and enzyme activities in a calcareous soil spiked with cadmium. Appl. Soil Ecol. 2014, 75, 33–42. [Google Scholar] [CrossRef]
- Siasou, E.; Standing, D.; Killham, K.; Johnson, D. Mycorrhizal fungi increase biocontrol potential of Pseudomonas fluorescens. Soil Biol. Biochem. 2009, 41, 1341–1343. [Google Scholar] [CrossRef]
- Frey, S.D. Mycorrhizal fungi as mediators of soil organic matter dynamics. Annu. Rev. Ecol. Evol. Syst. 2019, 50, 237–259. [Google Scholar] [CrossRef]
- Johnson, N.C.; Graham, J.H.; Smith, F.A. Functioning of mycorrhizal associations along the mutualism-parasitism continuum. New Phytol. 1997, 135, 575–585. [Google Scholar] [CrossRef]
- Johnson, N.C.; Graham, J.H. The continuum concept remains a useful framework for studying mycorrhizal functioning. Plant Soil 2013, 363, 411–419. [Google Scholar] [CrossRef]
- Delavaux, C.S.; Smith-Ramesh, L.M.; Kuebbing, S.E. Beyond nutrients: A meta-analysis of the diverse effects of arbuscular mycorrhizal fungi on plants and soils. Ecology 2017, 98, 2111–2119. [Google Scholar] [CrossRef]
- Brundrett, M.C. Coevolution of roots and mycorrhizas of land plants. New Phytol. 2002, 154, 275–304. [Google Scholar] [CrossRef]
- Bueno, C.G.; Aldrich-Wolfe, L.; Chaudhary, V.B.; Gerz, M.; Helgason, T.; Hoeksema, J.D.; Klironomos, J.; Lekberg, Y.; Leon, D.; Maherali, H.; et al. Misdiagnosis and uncritical use of plant mycorrhizal data are not the only elephants in the room. New Phytol. 2019, 224, 1415–1418. [Google Scholar] [CrossRef]
- Zhao, R.; Guo, W.; Bi, N.; Guo, J.; Wang, L.; Zhao, J.; Zhang, J. Arbuscular mycorrhizal fungi affect the growth, nutrient uptake and water status of maize (Zea mays L.) grown in two types of coal mine spoils under drought stress. Appl. Soil Ecol. 2015, 88, 41–49. [Google Scholar] [CrossRef]
- Baum, C.; El-Tohamy, W.; Gruda, N. Increasing the productivity and product quality of vegetable crops using arbuscular mycorrhizal fungi: A review. Sci. Hortic. 2015, 187, 131–141. [Google Scholar] [CrossRef]
- Augé, R.M. Water relations, drought and vesicular-arbuscular mycorrhizal symbiosis. Mycorrhiza 2001, 11, 3–42. [Google Scholar] [CrossRef]
- Myers, N.; Mittermeier, R.A.; Mittermeier, C.G.; da Fonseca, G.A.B.; Kent, J. Biodiversity hotspots for conservation priorities. Nature 2000, 403, 853–858. [Google Scholar] [CrossRef] [PubMed]
- Turco, M.; von Hardenberg, J.; AghaKouchak, A.; Llasat, M.C.; Provenzale, A.; Trigo, R.M. On the key role of droughts in the dynamics of summer fires in Mediterranean Europe. Sci. Rep. 2017, 7, 81. [Google Scholar] [CrossRef] [PubMed]
- Urrutia-Jalabert, R.; González, M.E.; González-Reyes, Á.; Lara, A.; Garreaud, R. Climate variability and forest fires in central and south-central Chile. Ecosphere 2018, 9, e02171. [Google Scholar] [CrossRef]
- Garreaud, R.D.; Boisier, J.P.; Rondanelli, R.; Montecinos, A.; Sepúlveda, H.H.; Veloso-Aguila, D. The central Chile mega drought (2010–2018): A climate dynamics perspective. Int. J. Climatol. 2020, 40, 421–439. [Google Scholar] [CrossRef]
- Bowman, D.M.J.S.; Moreira-Muñoz, A.; Kolden, C.A.; Chávez, R.O.; Muñoz, A.A.; Salinas, F.; González-Reyes, Á.; Rocco, R.; De La Barrera, F.; Williamson, G.J.; et al. Human–environmental drivers and impacts of the globally extreme 2017 Chilean fires. Ambio 2019, 48, 350–362. [Google Scholar] [CrossRef]
- Camprubi, A.; Zárate, I.A.; Adholeya, A.; Lovato, P.E.; Calvet, C. Field performance and essential oil production of Mycorrhizal rosemary in restoration low-nutrient Soils. Land Degrad. Dev. 2015, 26, 793–799. [Google Scholar] [CrossRef]
- Adamo, I.; Dashevskaya, S.; Alday, J.G. Fungal perspective of pine and oak colonization in Mediterranean degraded ecosystems. Forests 2022, 13, 88. [Google Scholar] [CrossRef]
- Elmostapha, O.; Hanane, D.; Rachid, B.; Lahcen, O. Application of arbuscular mycorrhizal fungi isolates from semi-arid Mediterranean ecosystems as biofertilizers in argan tree development. J. Soil Sci. Plant Nutr. 2022, 22, 944–955. [Google Scholar] [CrossRef]
- Silva-Flores, P.; Bueno, C.G.; Neira, J.; Palfner, G. Factors affecting arbuscular mycorrhizal fungi spore density in the chilean Mediterranean-type ecosystem. J. Soil Sci. Plant Nutr. 2019, 19, 42–50. [Google Scholar] [CrossRef]
- Maltz, M.R.; Treseder, K.K. Sources of inocula influence mycorrhizal colonization of plants in restoration projects: A meta-analysis. Restor. Ecol. 2015, 23, 625–634. [Google Scholar] [CrossRef]
- Fardella, C.; Oses, R.; Torres-Díaz, C.; Molina-Montenegro, M.A. Hongos endófitos antárticos como herramienta para la reintroducción de especies nativas en zonas áridas. Bosque 2014, 35, 235–239. [Google Scholar] [CrossRef]
- Molina-Montenegro, M.A.; Oses, R.; Torres-Díaz, C.; Atala, C.; Zurita-Silva, A.; Ruiz-Lara, S. Root-endophytes improve the ecophysiological performance and production of an agricultural species under drought condition. AoB Plants 2016, 8, plw062. [Google Scholar] [CrossRef]
- Rama, H.O.; Roberts, D.; Tignor, M.; Poloczanska, E.S.; Mintenbeck, K.; Alegría, A.; Craig, M.; Langsdorf, S.; Löschke, S.; Möller, V.; et al. Climate Change 2022: Impacts, Adaptation and Vulnerability Working Group II Contribution to the Sixth Assessment Report of the Intergovernmental Panel on Climate Change; Cambridge University Press: Cambridge, UK, 2022; p. 3056. [Google Scholar]


| Species | Factor | d.f. | F-Value | p-Value |
|---|---|---|---|---|
| Cryptocarya alba | Time | 4, 40 | 148.53 | <0.001 |
| Treatment | 1, 40 | 144.11 | <0.001 | |
| Time × Treatment | 4, 40 | 9.1 | <0.001 | |
| Kageneckia oblonga | Time | 4, 40 | 123.72 | <0.001 |
| Treatment | 1, 40 | 30.41 | <0.001 | |
| Time × Treatment | 4, 40 | 3.17 | 0.72 | |
| Peumus boldus | Time | 4, 40 | 110.96 | <0.001 |
| Treatment | 1, 40 | 7.04 | <0.05 | |
| Time × Treatment | 4, 40 | 4 | <0.05 | |
| Quillaja saponaria | Time | 4, 40 | 124.68 | <0.001 |
| Treatment | 1, 40 | 20.48 | <0.001 | |
| Time × Treatment | 4, 40 | 1.88 | 0.133 |
| Species | Factor | d.f. | F-Value | p-Value |
|---|---|---|---|---|
| Cryptocarya alba | Time | 4, 40 | 26.30 | <0.001 |
| Treatment | 1, 40 | 286.62 | <0.001 | |
| Time × Treatment | 4, 40 | 28.36 | <0.001 | |
| Kageneckia oblonga | Time | 4, 40 | 28.37 | <0.001 |
| Treatment | 1, 40 | 103.88 | <0.001 | |
| Time × Treatment | 4, 40 | 3.39 | 0.054 | |
| Peumus boldus | Time | 4, 40 | 13.42 | <0.001 |
| Treatment | 1, 40 | 22.62 | <0.001 | |
| Time × Treatment | 4, 40 | 7.02 | <0.001 | |
| Quillaja saponaria | Time | 4, 40 | 67.84 | <0.001 |
| Treatment | 1, 40 | 269.57 | <0.001 | |
| Time × Treatment | 4, 40 | 19.54 | <0.001 |
| (a) Cryptocarya alba | ||||
| Enzyme | Factor | d.f | F-value | p-value |
| Dehydrogenase | Time | 2, 12 | 212.64 | <0.001 |
| Treatment | 1, 12 | 591.50 | <0.001 | |
| Time × Treatment | 2, 12 | 240.93 | <0.001 | |
| β-Glucosidase | Time | 2, 12 | 730.94 | <0.001 |
| Treatment | 1, 12 | 1410.62 | <0.001 | |
| Time × Treatment | 2, 12 | 649.06 | <0.001 | |
| Urease | Time | 2, 12 | 87.30 | <0.001 |
| Treatment | 1, 12 | 90.00 | <0.001 | |
| Time × Treatment | 2, 12 | 29.10 | <0.001 | |
| (b) Kageneckia oblonga | ||||
| Enzyme | Factor | d.f. | F-value | p-value |
| Dehydrogenase | Time | 2, 12 | 126.25 | <0.001 |
| Treatment | 1, 12 | 126.87 | <0.001 | |
| Time × Treatment | 2, 12 | 63.42 | <0.001 | |
| β-Glucosidase | Time | 2, 12 | 871.79 | <0.001 |
| Treatment | 1, 12 | 368.58 | <0.001 | |
| Time × Treatment | 2, 12 | 220.28 | <0.001 | |
| Urease | Time | 2, 12 | 45.89 | <0.001 |
| Treatment | 1, 12 | 43.61 | <0.001 | |
| Time × Treatment | 2, 12 | 12.52 | <0.01 | |
| (c) Peumus boldus | ||||
| Enzyme | Factor | d.f. | F-value | p-value |
| Dehydrogenase | Time | 2, 12 | 204.97 | <0.001 |
| Treatment | 1, 12 | 72.00 | <0.001 | |
| Time × Treatment | 2, 12 | 42.84 | <0.001 | |
| β-Glucosidase | Time | 2, 12 | 401.77 | <0.001 |
| Treatment | 1, 12 | 47.69 | <0.001 | |
| Time × Treatment | 2, 12 | 51.80 | <0.001 | |
| Urease | Time | 2, 12 | 692.69 | <0.001 |
| Treatment | 1, 12 | 1508.10 | <0.001 | |
| Time × Treatment | 2, 12 | 900.46 | <0.001 | |
| (d) Quillaja saponaria | ||||
| Enzyme | Factor | d.f. | F-value | p-value |
| Dehydrogenase | Time | 2, 12 | 111.65 | <0.001 |
| Treatment | 1, 12 | 2.53 | 0.41 | |
| Time × Treatment | 2, 12 | 0.30 | 0.98 | |
| β-Glucosidase | Time | 2, 12 | 3678.07 | <0.001 |
| Treatment | 1, 12 | 399.87 | <0.001 | |
| Time × Treatment | 2, 12 | 286.62 | <0.001 | |
| Urease | Time | 2, 12 | 94.04 | <0.001 |
| Treatment | 1, 12 | 0.01 | 0.99 | |
| Time × Treatment | 2, 12 | 0.02 | 0.98 | |
| d.f. | t-Test | p-Value | |
|---|---|---|---|
| (a) Cryptocarya alba | |||
| Nitrogen | 8 | 18.62 | <0.0001 |
| Phosphorus | 8 | 11.41 | <0.0001 |
| Potassium | 8 | 11.56 | <0.0001 |
| (b) Kageneckia oblonga | |||
| Nitrogen | 8 | 10.96 | <0.0001 |
| Phosphorus | 8 | 14.62 | <0.0001 |
| Potassium | 8 | 4.10 | =0.0034 |
| (c) Peumus boldus | |||
| Nitrogen | 8 | 8.97 | <0.0001 |
| Phosphorus | 8 | 3.92 | =0.0044 |
| Potassium | 8 | 5.10 | <0.0001 |
| (d) Quillaja saponaria | |||
| Nitrogen | 8 | 1.25 | =0.2460 |
| Phosphorus | 8 | 0.25 | =0.8031 |
| Potassium | 8 | 0.39 | =0.7010 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Atala, C.; Reyes, S.A.; Molina-Montenegro, M.A. Assessing the Importance of Native Mycorrhizal Fungi to Improve Tree Establishment after Wildfires. J. Fungi 2023, 9, 421. https://doi.org/10.3390/jof9040421
Atala C, Reyes SA, Molina-Montenegro MA. Assessing the Importance of Native Mycorrhizal Fungi to Improve Tree Establishment after Wildfires. Journal of Fungi. 2023; 9(4):421. https://doi.org/10.3390/jof9040421
Chicago/Turabian StyleAtala, Cristian, Sebastián A. Reyes, and Marco A. Molina-Montenegro. 2023. "Assessing the Importance of Native Mycorrhizal Fungi to Improve Tree Establishment after Wildfires" Journal of Fungi 9, no. 4: 421. https://doi.org/10.3390/jof9040421
APA StyleAtala, C., Reyes, S. A., & Molina-Montenegro, M. A. (2023). Assessing the Importance of Native Mycorrhizal Fungi to Improve Tree Establishment after Wildfires. Journal of Fungi, 9(4), 421. https://doi.org/10.3390/jof9040421






