Revisiting Species Distribution and Antifungal Susceptibility of Candida Bloodstream Isolates from Latin American Medical Centers
Abstract
1. Introduction
2. Methods
3. Results
Temporal Trends of Antifungal Resistance to Azoles and Echinocandins
- (i)
- Trends in resistance to fluconazole and voriconazole among C. albicans, C. tropicalis, and C. parapsilosis isolates.
- (ii)
- Trends in resistance to echinocandins among Candida spp.
4. Discussion
5. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Magill, S.S.; Edwards, J.R.; Bamberg, W.; Beldavs, Z.G.; Dumyati, G.; Kainer, M.A.; Lynfield, R.; Maloney, M.; McAllister-Hollod, L.; Nadle, J.; et al. Multistate point-prevalence survey of health care-associated infections. N. Engl. J. Med. 2014, 370, 1198–1208. [Google Scholar] [CrossRef] [PubMed]
- Colombo, A.L.; Guimaraes, T.; Sukienik, T.; Pasqualotto, A.C.; Andreotti, R.; Queiroz-Telles, F.; Nouer, S.A.; Nucci, M. Prognostic factors and historical trends in the epidemiology of candidemia in critically ill patients: An analysis of five multicenter studies sequentially conducted over a 9-year period. Intensive Care Med. 2014, 40, 1489–1498. [Google Scholar] [CrossRef] [PubMed]
- Delaloye, J.; Calandra, T. Invasive candidiasis as a cause of sepsis in the critically ill patient. Virulence 2014, 5, 161–169. [Google Scholar] [CrossRef] [PubMed]
- Bassetti, M.; Righi, E.; Ansaldi, F.; Merelli, M.; Trucchi, C.; De Pascale, G.; Diaz-Martin, A.; Luzzati, R.; Rosin, C.; Lagunes, L.; et al. A multicenter study of septic shock due to candidemia: Outcomes and predictors of mortality. Intensive Care Med. 2014, 40, 839–845. [Google Scholar] [CrossRef] [PubMed]
- Lortholary, O.; Renaudat, C.; Sitbon, K.; Madec, Y.; Denoeud-Ndam, L.; Wolff, M.; Fontanet, A.; Bretagne, S.; Dromer, F.; French Mycosis Study, G. Worrisome trends in incidence and mortality of candidemia in intensive care units (Paris Area, 2002–2010). Intensive Care Med. 2014, 40, 1303–1312. [Google Scholar] [CrossRef] [PubMed]
- Tedeschi, S.; Tumietto, F.; Giannella, M.; Bartoletti, M.; Cristini, F.; Cioni, G.; Ambretti, S.; Carretto, E.; Sambri, V.; Sarti, M.; et al. Epidemiology and outcome of candidemia in internal medicine wards: A regional study in italy. Eur. J. Intern. Med. 2016, 34, 39–44. [Google Scholar] [CrossRef] [PubMed]
- Nucci, M.; Queiroz-Telles, F.; Alvarado-Matute, T.; Tiraboschi, I.N.; Cortes, J.; Zurita, J.; Guzman-Blanco, M.; Santolaya, M.E.; Thompson, L.; Sifuentes-Osornio, J.; et al. Epidemiology of candidemia in Latin America: A laboratory-based survey. PLoS ONE 2013, 8, e59373. [Google Scholar] [CrossRef] [PubMed]
- Pfaller, M.A.; Jones, R.N.; Castanheira, M. Regional data analysis of candida non-albicans strains collected in united states medical sites over a 6-year period, 2006–2011. Mycoses 2014, 57, 602–611. [Google Scholar] [CrossRef] [PubMed]
- Pfaller, M.A.; Andes, D.R.; Diekema, D.J.; Horn, D.L.; Reboli, A.C.; Rotstein, C.; Franks, B.; Azie, N.E. Epidemiology and outcomes of invasive candidiasis due to non-albicans species of candida in 2,496 patients: Data from the prospective antifungal therapy (path) registry 2004–2008. PLoS ONE 2014, 9, e101510. [Google Scholar] [CrossRef] [PubMed]
- Guinea, J. Global trends in the distribution of candida species causing candidemia. Clin. Microbiol. Infect. 2014, 20, 5–10. [Google Scholar] [CrossRef] [PubMed]
- Ruggero, M.A.; Topal, J.E. Development of echinocandin-resistant candida albicans candidemia following brief prophylactic exposure to micafungin therapy. Transpl. Infect. Dis 2014, 16, 469–472. [Google Scholar] [CrossRef] [PubMed]
- Cuervo, G.; Garcia-Vidal, C.; Nucci, M.; Puchades, F.; Fernandez-Ruiz, M.; Obed, M.; Manzur, A.; Gudiol, C.; Peman, J.; Aguado, J.M.; et al. Breakthrough candidaemia in the era of broad-spectrum antifungal therapies. Clin. Microbiol. Infect. 2016, 22, 181–188. [Google Scholar] [CrossRef] [PubMed]
- Ben-Ami, R.; Olshtain-Pops, K.; Krieger, M.; Oren, I.; Bishara, J.; Dan, M.; Wiener-Well, Y.; Weinberger, M.; Zimhony, O.; Chowers, M.; et al. Antibiotic exposure as a risk factor for fluconazole-resistant Candida bloodstream infection. Antimicrob. Agents Chemother. 2012, 56, 2518–2523. [Google Scholar] [CrossRef] [PubMed]
- Mikulska, M.; Del Bono, V.; Ratto, S.; Viscoli, C. Occurrence, presentation and treatment of candidemia. Expert Rev. Clin. Immunol. 2012, 8, 755–765. [Google Scholar] [CrossRef] [PubMed]
- Colombo, A.L.; Garnica, M.; Aranha Camargo, L.F.; Da Cunha, C.A.; Bandeira, A.C.; Borghi, D.; Campos, T.; Senna, A.L.; Valias Didier, M.E.; Dias, V.C.; et al. Candida glabrata: An emerging pathogen in Brazilian tertiary care hospitals. Med. Mycol. 2013, 51, 38–44. [Google Scholar] [CrossRef] [PubMed]
- Yang, Z.T.; Wu, L.; Liu, X.Y.; Zhou, M.; Li, J.; Wu, J.Y.; Cai, Y.; Mao, E.Q.; Chen, E.Z.; Lortholary, O. Epidemiology, species distribution and outcome of nosocomial Candida spp. Bloodstream infection in Shanghai. BMC Infect. Dis. 2014, 14, 241. [Google Scholar] [CrossRef] [PubMed]
- Farmakiotis, D.; Kyvernitakis, A.; Tarrand, J.J.; Kontoyiannis, D.P. Early initiation of appropriate treatment is associated with increased survival in cancer patients with Candida glabrata fungaemia: A potential benefit from infectious disease consultation. Clin. Microbiol. Infect. 2015, 21, 79–86. [Google Scholar] [CrossRef] [PubMed]
- Cowen, L.E.; Sanglard, D.; Howard, S.J.; Rogers, P.D.; Perlin, D.S. Mechanisms of antifungal drug resistance. Cold Spring Harb. Perspect. Med. 2014, 5, a019752. [Google Scholar] [CrossRef] [PubMed]
- Goncalves, S.S.; Souza, A.C.; Chowdhary, A.; Meis, J.F.; Colombo, A.L. Epidemiology and molecular mechanisms of antifungal resistance in Candida and Aspergillus. Mycoses 2016, 4, 198–219. [Google Scholar]
- Arendrup, M.C.; Bruun, B.; Christensen, J.J.; Fuursted, K.; Johansen, H.K.; Kjaeldgaard, P.; Knudsen, J.D.; Kristensen, L.; Moller, J.; Nielsen, L.; et al. National surveillance of fungemia in Denmark (2004 to 2009). J. Clin. Microbiol. 2011, 49, 325–334. [Google Scholar] [CrossRef] [PubMed]
- Peman, J.; Canton, E.; Minana, J.J.; Florez, J.A.; Echeverria, J.; Ortega, D.N.; Alarcon, J.M.; Fontanals, D.; Sard, B.G.; Moreno, B.B.; et al. Changes in the epidemiology of fungaemia and fluconazole susceptibility of blood isolates during the last 10 years in spain: Results from the fungemyca study. Rev. Iberoam. Micol. 2011, 28, 91–99. [Google Scholar] [PubMed]
- Lockhart, S.R.; Iqbal, N.; Cleveland, A.A.; Farley, M.M.; Harrison, L.H.; Bolden, C.B.; Baughman, W.; Stein, B.; Hollick, R.; Park, B.J.; et al. Species identification and antifungal susceptibility testing of Candida bloodstream isolates from population-based surveillance studies in two U.S. cities from 2008 to 2011. J. Clin. Microbiol. 2012, 50, 3435–3442. [Google Scholar] [CrossRef] [PubMed]
- Ericsson, J.; Chryssanthou, E.; Klingspor, L.; Johansson, A.G.; Ljungman, P.; Svensson, E.; Sjolin, J. Candidaemia in Sweden: A nationwide prospective observational survey. Clin. Microbiol. Infect. 2013, 19, E218–E221. [Google Scholar] [CrossRef] [PubMed]
- Pinhati, H.M.; Casulari, L.A.; Souza, A.C.; Siqueira, R.A.; Damasceno, C.M.; Colombo, A.L. Outbreak of candidemia caused by fluconazole resistant Candida parapsilosis strains in an intensive care unit. BMC Infect. Dis. 2016, 16, 433. [Google Scholar] [CrossRef] [PubMed]
- Bizerra, F.C.; Jimenez-Ortigosa, C.; Souza, A.C.; Breda, G.L.; Queiroz-Telles, F.; Perlin, D.S.; Colombo, A.L. Breakthrough candidemia due to multidrug-resistant Candida glabrata during prophylaxis with a low dose of micafungin. Antimicrob. Agents Chemother. 2014, 58, 2438–2440. [Google Scholar] [CrossRef] [PubMed]
- Vallabhaneni, S.; Cleveland, A.A.; Farley, M.M.; Harrison, L.H.; Schaffner, W.; Beldavs, Z.G.; Derado, G.; Pham, C.D.; Lockhart, S.R.; Smith, R.M. Epidemiology and risk factors for echinocandin nonsusceptible Candida glabrata bloodstream infections: Data from a large multisite population-based candidemia surveillance program, 2008–2014. Open Forum Infect. Dis. 2015, 2, ofv163. [Google Scholar] [CrossRef] [PubMed]
- Alexander, B.D.; Johnson, M.D.; Pfeiffer, C.D.; Jimenez-Ortigosa, C.; Catania, J.; Booker, R.; Castanheira, M.; Messer, S.A.; Perlin, D.S.; Pfaller, M.A. Increasing echinocandin resistance in Candida glabrata: Clinical failure correlates with presence of fks mutations and elevated minimum inhibitory concentrations. Clin. Infect. Dis. 2013, 56, 1724–1732. [Google Scholar] [CrossRef] [PubMed]
- Rodero, L.; Davel, G.; Soria, M.; Vivot, W.; Cordoba, S.; Canteros, C.E.; Saporiti, A.; Emifn. Multicenter study of fungemia due to yeasts in Argentina. Rev. Argent. Microbiol. 2005, 37, 189–195. [Google Scholar] [PubMed]
- Cordoba, S.; Vivot, W.; Bosco-Borgeat, M.E.; Taverna, C.; Szusz, W.; Murisengo, O.; Isla, G.; Davel, G.; Red Nacional De Laboratorios De, M. Species distribution and susceptibility profile of yeasts isolated from blood cultures: Results of a multicenter active laboratory-based surveillance study in Argentina. Rev. Argent. Microbiol. 2011, 43, 176–185. [Google Scholar] [PubMed]
- Lopez Moral, L.; Tiraboschi, I.N.; Schijman, M.; Bianchi, M.; Guelfand, L.; Cataldi, S.; integrantes de la Red de Micología de la Ciudad de Buenos Aires. Fungemia in hospitals of the city of Buenos aires, Argentina. Rev. Iberoam. Micol. 2012, 29, 144–149. [Google Scholar] [CrossRef] [PubMed]
- Riera, F.; Medeot, M.; Sartori, L.; Bergallo, C.; Minoli, J.; Vilchez, V.; Sanchez, P.; Abiega, C.; Pincheira, C.; Correa, S.; et al. Candidemia epidemiology in Cordoba Argentina. Surveillance study of five institutions. Rev. Fac. Cien. Med. Univ. Nac. Cordoba. 2014, 71, 89–93. [Google Scholar] [PubMed]
- Colombo, A.L.; Nucci, M.; Salomao, R.; Branchini, M.L.; Richtmann, R.; Derossi, A.; Wey, S.B. High rate of non-albicans candidemia in brazilian tertiary care hospitals. Diagn. Microbiol. Infect. Dis. 1999, 34, 281–286. [Google Scholar] [CrossRef]
- Costa, S.F.; Marinho, I.; Araujo, E.A.; Manrique, A.E.; Medeiros, E.A.; Levin, A.S. Nosocomial fungaemia: A 2-year prospective study. J. Hosp. Infect. 2000, 45, 69–72. [Google Scholar] [CrossRef] [PubMed]
- Colombo, A.L.; Nakagawa, Z.; Valdetaro, F.; Branchini, M.L.; Kussano, E.J.; Nucci, M. Susceptibility profile of 200 bloodstream isolates of Candida spp. Collected from Brazilian tertiary care hospitals. Med. Mycol. 2003, 41, 235–239. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Goldani, L.Z.; Mario, P.S. Candida tropicalis fungemia in a tertiary care hospital. J. Infect. 2003, 46, 155–160. [Google Scholar] [CrossRef] [PubMed]
- Antunes, A.G.; Pasqualotto, A.C.; Diaz, M.C.; d’Azevedo, P.A.; Severo, L.C. Candidemia in a Brazilian tertiary care hospital: Species distribution and antifungal susceptibility patterns. Rev. Inst. Med. Trop. Sao Paulo 2004, 46, 239–241. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Aquino, V.R.; Lunardi, L.W.; Goldani, L.Z.; Barth, A.L. Prevalence, susceptibility profile for fluconazole and risk factors for candidemia in a tertiary care hospital in Southern Brazil. Braz. J. Infect. Dis. 2005, 9, 411–418. [Google Scholar] [CrossRef] [PubMed]
- Colombo, A.L.; Nucci, M.; Park, B.J.; Nouer, S.A.; Arthington-Skaggs, B.; da Matta, D.A.; Warnock, D.; Morgan, J.; Brazilian Network Candidemia, S. Epidemiology of candidemia in brazil: A nationwide sentinel surveillance of candidemia in eleven medical centers. J. Clin. Microbiol. 2006, 44, 2816–2823. [Google Scholar] [CrossRef] [PubMed]
- Medrano, D.J.A.; Brilhante, R.S.N.; Cordeiro, R.A.; Rocha, M.F.G.; Rabenhorst, S.H.B.; Sidrim, J.J. Candidemia in a Brazilian hospital: The importance of Candida parapsilosis. Rev. Inst. Med. Trop. Sao Paulo 2006, 48, 4. [Google Scholar] [CrossRef]
- Colombo, A.L.; Guimaraes, T.; Silva, L.R.; de Almeida Monfardini, L.P.; Cunha, A.K.; Rady, P.; Alves, T.; Rosas, R.C. Prospective observational study of candidemia in Sao Paulo, Brazil: Incidence rate, epidemiology, and predictors of mortality. Infect. Control. Hosp. Epidemiol. 2007, 28, 570–576. [Google Scholar] [CrossRef] [PubMed]
- Da Matta, D.A.; de Almeida, L.P.; Machado, A.M.; Azevedo, A.C.; Kusano, E.J.; Travassos, N.F.; Salomao, R.; Colombo, A.L. Antifungal susceptibility of 1000 candida bloodstream isolates to 5 antifungal drugs: Results of a multicenter study conducted in Sao Paulo, Brazil, 1995–2003. Diagn. Microbiol. Infect. Dis. 2007, 57, 399–404. [Google Scholar] [CrossRef] [PubMed]
- Girao, E.; Levin, A.S.; Basso, M.; Gobara, S.; Gomes, L.B.; Medeiros, E.A.; Costa, S.F. Seven-year trend analysis of nosocomial candidemia and antifungal (fluconazole and caspofungin) use in intensive care units at a Brazilian university hospital. Med. Mycol. 2008, 46, 581–588. [Google Scholar] [CrossRef] [PubMed]
- Franca, J.C.; Ribeiro, C.E.; Queiroz-Telles, F. Candidemia in a Brazilian tertiary care hospital: Incidence, frequency of different species, risk factors and antifungal susceptibility. Rev. Soc. Bras. Med. Trop. 2008, 41, 23–28. [Google Scholar] [PubMed]
- Motta, A.L.; Almeida, G.M.; Almeida Junior, J.N.; Burattini, M.N.; Rossi, F. Candidemia epidemiology and susceptibility profile in the largest Brazilian teaching hospital complex. Braz. J. Infect. Dis. 2010, 14, 441–448. [Google Scholar] [CrossRef]
- Sampaio Camargo, T.Z.; Marra, A.R.; Silva, C.V.; Cardoso, M.F.; Martino, M.D.; Camargo, L.F.; Correa, L. Secular trends of candidemia in a tertiary care hospital. Am. J. Infect. Control. 2010, 38, 546–551. [Google Scholar] [CrossRef] [PubMed]
- Bonfietti, L.X.; Szeszs, M.W.; Chang, M.R.; Martins, M.A.; Pukinskas, S.R.; Nunes, M.O.; Pereira, G.H.; Paniago, A.M.; Purisco, S.U.; Melhem, M.S. Ten-year study of species distribution and antifungal susceptibilities of candida bloodstream isolates at a Brazilian tertiary hospital. Mycopathologia 2012, 174, 389–396. [Google Scholar] [CrossRef] [PubMed]
- Hoffmann-Santos, H.D.; Paula, C.R.; Yamamoto, A.C.; Tadano, T.; Hahn, R.C. Six-year trend analysis of nosocomial candidemia and risk factors in two intensive care hospitals in Mato Grosso, Midwest region of Brazil. Mycopathologia 2013, 176, 409–415. [Google Scholar] [CrossRef] [PubMed]
- Moretti, M.L.; Trabasso, P.; Lyra, L.; Fagnani, R.; Resende, M.R.; de Oliveira Cardoso, L.G.; Schreiber, A.Z. Is the incidence of candidemia caused by Candida glabrata increasing in Brazil? Five-year surveillance of candida bloodstream infection in a university reference hospital in Southeast Brazil. Med. Mycol. 2013, 51, 225–230. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Santos, E.R.; Dal Forno, C.F.; Hernandez, M.G.; Kubica, T.F.; Venturini, T.P.; Chassot, F.; Santurio, J.M.; Alves, S.H. Susceptibility of Candida spp. Isolated from blood cultures as evaluated using the M27-A3 and new M27-S4 approved breakpoints. Rev. Inst. Med. Trop. Sao Paulo 2014, 56, 477–482. [Google Scholar] [CrossRef] [PubMed]
- Da Costa, V.G.; Quesada, R.M.; Abe, A.T.; Furlaneto-Maia, L.; Furlaneto, M.C. Nosocomial bloodstream candida infections in a tertiary-care hospital in South Brazil: A 4-year survey. Mycopathologia 2014, 178, 243–250. [Google Scholar] [CrossRef] [PubMed]
- Doi, A.M.; Pignatari, A.C.; Edmond, M.B.; Marra, A.R.; Camargo, L.F.; Siqueira, R.A.; da Mota, V.P.; Colombo, A.L. Epidemiology and microbiologic characterization of nosocomial candidemia from a brazilian national surveillance program. PLoS ONE 2016, 11, e0146909. [Google Scholar] [CrossRef] [PubMed]
- Cortes, J.A.; Reyes, P.; Gomez, C.; Buitrago, G.; Leal, A.L.; Group, G. Fungal bloodstream infections in tertiary care hospitals in Colombia. Rev. Iberoam. Micol. 2011, 28, 74–78. [Google Scholar] [CrossRef] [PubMed]
- Cortes, J.A.; Jaimes, J.A.; Leal, A.L. Incidence and prevalence of candidemia in critically ill patients in Colombia. Rev. Chil. Infectol. 2013, 30, 599–604. [Google Scholar] [CrossRef] [PubMed]
- Cortes, J.A.; Reyes, P.; Gomez, C.H.; Cuervo, S.I.; Rivas, P.; Casas, C.A.; Sanchez, R. Clinical and epidemiological characteristics and risk factors for mortality in patients with candidemia in hospitals from Bogota, Colombia. Braz J. Infect. Dis. 2014, 18, 631–637. [Google Scholar] [CrossRef] [PubMed]
- Ortiz Ruiz, G.; Osorio, J.; Valderrama, S.; Alvarez, D.; Elias Diaz, R.; Calderon, J.; Ballesteros, D.; Franco, A. Risk factors for candidemia in non-neutropenic critical patients in Colombia. Med. Intensiva 2016, 40, 139–144. [Google Scholar] [CrossRef] [PubMed]
- Villalobos, J.M.; Castro, J.A.; Aviles, A.; Pelaez, M.C.; Somogyi, T.; Sandoval, L. Candida parapsilosis: A major cause of bloodstream infection in a tertiary care hospital in Costa Rica. Rev. Chil. Infectol. 2016, 33, 159–165. [Google Scholar] [CrossRef] [PubMed]
- Gonzalez, G.M.; Elizondo, M.; Ayala, J. Trends in species distribution and susceptibility of bloodstream isolates of candida collected in Monterrey, Mexico, to seven antifungal agents: Results of a 3-year (2004 to 2007) surveillance study. J. Clin. Microbiol. 2008, 46, 2902–2905. [Google Scholar] [CrossRef] [PubMed]
- Corzo-Leon, D.E.; Alvarado-Matute, T.; Colombo, A.L.; Cornejo-Juarez, P.; Cortes, J.; Echevarria, J.I.; Guzman-Blanco, M.; Macias, A.E.; Nucci, M.; Ostrosky-Zeichner, L.; et al. Surveillance of Candida spp. bloodstream infections: Epidemiological trends and risk factors of death in two Mexican tertiary care hospitals. PLoS ONE 2014, 9, e97325. [Google Scholar] [CrossRef] [PubMed]
- Bustamante, B.; Martins, M.A.; Bonfietti, L.X.; Szeszs, M.W.; Jacobs, J.; Garcia, C.; Melhem, M.S. Species distribution and antifungal susceptibility profile of candida isolates from bloodstream infections in Lima, Peru. J. Med. Microbiol. 2014, 63, 855–860. [Google Scholar] [CrossRef] [PubMed]
- Conde-Rosa, A.; Amador, R.; Perez-Torres, D.; Colon, E.; Sanchez-Rivera, C.; Nieves-Plaza, M.; Gonzalez-Ramos, M.; Bertran-Pasarell, J. Candidemia distribution, associated risk factors, and attributed mortality at a university-based medical center. PR Health Sci. J. 2010, 29, 26–29. [Google Scholar]
- Franco, M.E.D.; Reviakina, V.; Panizo, M.M.; Macero, C.; Moreno, X.; Calvo, A.; Selgrad, S.; Papatzikos, J.; Vergara, V.; Mendoza, M.J. Distribution and antifungal susceptibility of candida clinical isolations coming from six health care centers in the metropolitan area of Caracas, Venezuela (years 2003–2005). Rev. Iberoam Micol. 2008, 25, 5. [Google Scholar]
- Pfaller, M.A.; Jones, R.N.; Doern, G.V.; Sader, H.S.; Messer, S.A.; Houston, A.; Coffman, S.; Hollis, R.J. Bloodstream infections due to candida species: Sentry antimicrobial surveillance program in North America and Latin America, 1997–1998. Antimicrob. Agents Chemother. 2000, 44, 747–751. [Google Scholar] [CrossRef] [PubMed]
- Pfaller, M.A.; Diekema, D.J.; Jones, R.N.; Sader, H.S.; Fluit, A.C.; Hollis, R.J.; Messer, S.A.; Group, S.P. International surveillance of bloodstream infections due to candida species: Frequency of occurrence and in vitro susceptibilities to fluconazole, ravuconazole, and voriconazole of isolates collected from 1997 through 1999 in the sentry antimicrobial surveillance program. J. Clin. Microbiol. 2001, 39, 3254–3259. [Google Scholar] [PubMed]
- Godoy, P.; Tiraboschi, I.N.; Severo, L.C.; Bustamante, B.; Calvo, B.; Almeida, L.P.; da Matta, D.A.; Colombo, A.L. Species distribution and antifungal susceptibility profile of Candida spp. Bloodstream isolates from Latin American hospitals. Mem. Inst. Oswaldo Cruz 2003, 98, 401–405. [Google Scholar] [CrossRef] [PubMed]
- Nishikaku, A.S.; Melo, A.S.A.; Colombo, A.L. Geographic trends in invasive candidiasis. Curr. Fungal Infect. Rep. 2010, 4, 210–218. [Google Scholar] [CrossRef]
- Pfaller, M.A.; Diekema, D.J. Epidemiology of invasive candidiasis: A persistent public health problem. Clin. Microbiol. Rev. 2007, 20, 133–163. [Google Scholar] [CrossRef] [PubMed]
- Pappas, P.G.; Kauffman, C.A.; Andes, D.R.; Clancy, C.J.; Marr, K.A.; Ostrosky-Zeichner, L.; Reboli, A.C.; Schuster, M.G.; Vazquez, J.A.; Walsh, T.J.; et al. Clinical practice guideline for the management of Candidiasis: 2016 update by the infectious diseases society of America. Clin. Infect. Dis. 2016, 62, e1–e50. [Google Scholar] [CrossRef] [PubMed]
- Colombo, A.L.; Guimaraes, T.; Camargo, L.F.; Richtmann, R.; Queiroz-Telles, F.; Salles, M.J.; Cunha, C.A.; Yasuda, M.A.; Moretti, M.L.; Nucci, M. Brazilian guidelines for the management of Candidiasis—A joint meeting report of three medical societies: Sociedade brasileira de infectologia, sociedade paulista de infectologia and sociedade brasileira de medicina tropical. Braz. J. Infect. Dis. 2013, 17, 283–312. [Google Scholar] [CrossRef] [PubMed]
- Merseguel, K.B.; Nishikaku, A.S.; Rodrigues, A.M.; Padovan, A.C.; e Ferreira, R.C.; de Azevedo Melo, A.S.; Briones, M.R.; Colombo, A.L. Genetic diversity of medically important and emerging candida species causing invasive infection. BMC Infect. Dis. 2015, 15, 57. [Google Scholar] [CrossRef] [PubMed]
- Van Veen, S.Q.; Claas, E.C.; Kuijper, E.J. High-throughput identification of bacteria and yeast by matrix-assisted laser desorption ionization-time of flight mass spectrometry in conventional medical microbiology laboratories. J. Clin. Microbiol. 2010, 48, 900–907. [Google Scholar] [CrossRef] [PubMed]
- Pulcrano, G.; Iula, D.V.; Vollaro, A.; Tucci, A.; Cerullo, M.; Esposito, M.; Rossano, F.; Catania, M.R. Rapid and reliable MALDI-TOF mass spectrometry identification of candida non-albicans isolates from bloodstream infections. J. Microbiol. Methods 2013, 94, 262–266. [Google Scholar] [CrossRef] [PubMed]
- Calvo, B.; Melo, A.S.; Perozo-Mena, A.; Hernandez, M.; Francisco, E.C.; Hagen, F.; Meis, J.F.; Colombo, A.L. First report of candida auris in America: Clinical and microbiological aspects of 18 episodes of candidemia. J. Infect. 2016, 73, 369–374. [Google Scholar] [CrossRef] [PubMed]
- Morales-Lopez, S.E.; Parra-Giraldo, C.M.; Ceballos-Garzon, A.; Martinez, H.P.; Rodriguez, G.J.; Alvarez-Moreno, C.A.; Rodriguez, J.Y. Invasive infections with multidrug-resistant yeast Candida auris, Colombia. Emerg. Infect. Dis. 2017, 23, 162–164. [Google Scholar] [CrossRef] [PubMed]
- Chowdhary, A.; Voss, A.; Meis, J.F. Multidrug-resistant candida auris: New kid on the block in hospital-associated infections? J. Hosp. Infect. 2016, 94, 209–212. [Google Scholar] [CrossRef] [PubMed]
- Asmundsdottir, L.R.; Erlendsdottir, H.; Gottfredsson, M. Nationwide study of candidemia, antifungal use, and antifungal drug resistance in Iceland, 2000 to 2011. J. Clin. Microbiol. 2013, 51, 841–848. [Google Scholar] [CrossRef] [PubMed]
- Cleveland, A.A.; Farley, M.M.; Harrison, L.H.; Stein, B.; Hollick, R.; Lockhart, S.R.; Magill, S.S.; Derado, G.; Park, B.J.; Chiller, T.M. Changes in incidence and antifungal drug resistance in candidemia: Results from population-based laboratory surveillance in Atlanta and Baltimore, 2008–2011. Clin. Infect. Dis. 2012, 55, 1352–1361. [Google Scholar] [CrossRef] [PubMed]
- Minea, B.; Nastasa, V.; Moraru, R.F.; Kolecka, A.; Flonta, M.M.; Marincu, I.; Man, A.; Toma, F.; Lupse, M.; Doroftei, B.; et al. Species distribution and susceptibility profile to fluconazole, voriconazole and MXP-4509 of 551 clinical yeast isolates from a romanian multi-centre study. Eur J. Clin. Microbiol. Infect. Dis. 2015, 34, 367–383. [Google Scholar] [CrossRef] [PubMed]
- Guinea, J.; Zaragoza, O.; Escribano, P.; Martin-Mazuelos, E.; Peman, J.; Sanchez-Reus, F.; on behalf of the CANDIPOP Project, GEIH-GEMICOMED (SEIMC), and REIPI. Molecular identification and antifungal susceptibility of yeast isolates causing fungemia collected in a population-based study in Spain in 2010 and 2011. Antimicrob. Agents Chemother. 2014, 58, 1529–1537. [Google Scholar] [CrossRef] [PubMed]
- Souza, A.C.; Fuchs, B.B.; Pinhati, H.M.; Siqueira, R.A.; Hagen, F.; Meis, J.F.; Mylonakis, E.; Colombo, A.L. Candida parapsilosis resistance to fluconazole: Molecular mechanisms and in vivo impact in infected Galleria mellonella larvae. Antimicrob. Agents Chemother. 2015, 59, 6581–6587. [Google Scholar] [CrossRef] [PubMed]
- Maubon, D.; Garnaud, C.; Calandra, T.; Sanglard, D.; Cornet, M. Resistance of Candida spp. to antifungal drugs in the ICU: Where are we now? Intensive Care Med. 2014, 40, 1241–1255. [Google Scholar] [CrossRef] [PubMed]
- Arendrup, M.C.; Pfaller, M.A.; Danish Fungaemia Study Group. Caspofungin etest susceptibility testing of candida species: Risk of misclassification of susceptible isolates of C. glabrata and C. krusei when adopting the revised CLSI caspofungin breakpoints. Antimicrob. Agents Chemother. 2012, 56, 3965–3968. [Google Scholar] [CrossRef] [PubMed]
- Arendrup, M.C.; Perlin, D.S. Echinocandin resistance: An emerging clinical problem? Curr. Opin. Infect. Dis. 2014, 27, 484–492. [Google Scholar] [CrossRef] [PubMed]
- Forastiero, A.; Garcia-Gil, V.; Rivero-Menendez, O.; Garcia-Rubio, R.; Monteiro, M.C.; Alastruey-Izquierdo, A.; Jordan, R.; Agorio, I.; Mellado, E. Rapid development of Candida krusei echinocandin resistance during caspofungin therapy. Antimicrob. Agents Chemother. 2015, 59, 6975–6982. [Google Scholar] [CrossRef] [PubMed]
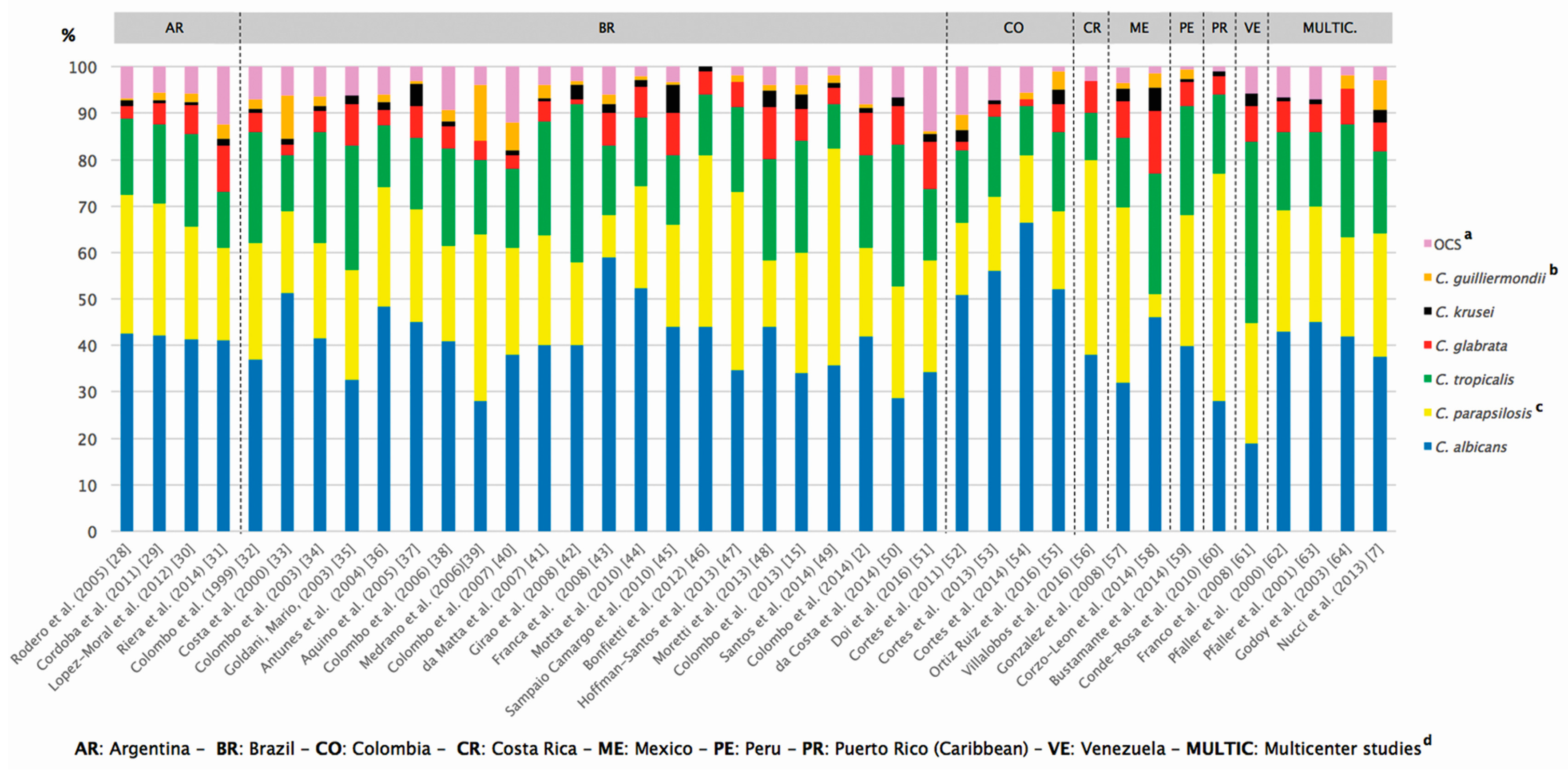
| Country/Territory | Reference | Period | n e (No. of Centers) | Species Distribution (%) | ||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Ca a | Cp a | Ct a | Cgla a | Ck a | C. gui a | OCS b | ||||
| Argentina | Rodero et al. (2005) [28] c | 1999–2000 | 253 (36) | 42.5 | 29.9 | 16.5 | 2.7 | 1.18 | 0.39 | 6.83 |
| Argentina | Cordoba et al. (2011) [29] c | 2007–2008 | 420 (47) | 42.1 | 28.5 | 16.9 | 4.7 | 0.47 | 1.66 | 5.67 |
| Argentina | Lopez Moral et al. (2012) [30] c | 2005–2008 | 683 (16) | 41.3 | 24.3 | 19.9 | 6.3 | 0.59 | 1.76 | 5.85 |
| Argentina | Riera et al. (2014) [31] | 2010–2012 | 158 (4) | 41 | 20 | 12 | 10 | 1.5 | 3 | 12.5 |
| Brazil | Colombo et al. (1999) [32] | 1995–1996 | 145 (6) | 37 | 25 | 24 | 4 | 1 | 2 | 7 |
| Brazil | Costa et al. (2000) [33] c | 1994–1996 | 84 (1) | 51.2 | 17.8 | 11.9 | 2.3 | 1.19 | 9.5 | 6.11 |
| Brazil | Colombo et al. (2003) [34] | 1996–1998 | 200 (5) | 41.5 | 20.5 | 24 | 4.5 | 1 | 2 | 6.5 |
| Brazil | Goldani and Mario (2003) [35] c | 1996–1999 | 101 (1) | 32.6 | 23.7 | 26.7 | 8.9 | 1.9 | 0 | 6.2 |
| Brazil | Antunes et al. (2004) [36] | 2002–2003 | 120 (1) | 48.3 | 25.8 | 13.3 | 3.3 | 1.7 | 1.6 | 6 |
| Brazil | Aquino et al. (2005) [37] | 1998–2004 | 131 (1) | 45 | 24.4 | 15.3 | 6.9 | 4.6 | 0.76 | 3.04 |
| Brazil | Colombo et al. (2006) [38] | 2003–2004 | 712 (11) | 40.9 | 20.5 | 20.9 | 4.9 | 1.1 | 2.4 | 9.3 |
| Brazil | Medrano et al. (2006) [39] | 2000–2002 | 50 (1) | 28 | 36 | 16 | 4 | 0 | 12 | 4 |
| Brazil | Colombo et al. (2007) [40] | 2002–2003 | 282 (4) | 38 | 23 | 17 | 3 | 1 | 6 | 12 |
| Brazil | da Matta et al. (2007) [41] | 1995–2003 | 1000 (4) | 40 | 23.8 | 24.3 | 4.4 | 0.6 | 3 | 3.9 |
| Brazil | Girao et al. (2008) [42] | 1999–2006 | 108 (1) | 40 | 18 | 34 | 1 | 3 | 1 | 3 |
| Brazil | Franca et al. (2008) [43] | 2001–2004 | 100 (1) | 59 | 9 | 15 | 7 | 2 | 2 | 6 |
| Brazil | Motta et al. (2010) [44] | 2006 | 136 (1) | 52.2 | 22.1 | 14.7 | 6.6 | 1.6 | 0.8 | 2 |
| Brazil | Sampaio Camargo et al. 2010 [45] | 1997–2007 | 151 (1) | 44 | 22 | 15 | 9 | 6 | 0.66 | 3.34 |
| Brazil | Bonfietti et al. (2012) [46] | 1998–2006 | 100 (1) | 44 | 37 | 13 | 5 | 1 | 0 | 0 |
| Brazil | Hoffmann-Santos et al. (2013) [47] | 2006–2011 | 130 (2) | 34.6 | 38.4 | 18.4 | 5.3 | 0 | 1.5 | 1.8 |
| Brazil | Moretti et al. (2013) [48] | 2006–2010 | 313 (1) | 44 | 14.4 | 21.7 | 11.2 | 3.5 | 1.3 | 3.9 |
| Brazil | Colombo et al. (2013) [15] | 2006–2007 | 300 (9) | 34 | 26 | 24 | 7 | 3 | 2 | 4 |
| Brazil | Santos et al. (2014) [49] | 1995–2009 | 422 (1) | 35.7 | 46.6 | 9.7 | 3.5 | 0.94 | 1.65 | 1.91 |
| Brazil | Colombo et al. (2014) [2] | 2003–2012 | 1392 (22) | 42 | 19 | 20 | 9 | 1.14 | 0.86 | 8 |
| Brazil | da Costa et al. (2014) [50] | 2006–2011 | 108 (1) | 28.7 | 24.1 | 30.5 | 8.3 | 1.8 | 0 | 6.6 |
| Brazil | Doi et al. (2016) [51] | 2007–2010 | 137 (16) | 34.3 | 24.1 | 15.3 | 10.2 | 1.5 | 0.7 | 13.9 |
| Colombia | Cortes et al. (2011) [52] c | 2001–2007 | 1622 (27) | 50.9 | 15.5 | 15.5 | 2 | 2.36 | 3.3 | 10.44 |
| Colombia | Cortes et al. (2013) [53] | 2004–2008 | 382 (7) | 56 | 16 | 17.3 | 2.6 | 0.8 | 0 | 7.3 |
| Colombia | Cortes et al. (2014) [54] | 2008–2009 | 131 (7) | 66.4 | 14.5 | 10.6 | 1.5 | 0 | 1.5 | 5.5 |
| Colombia | Ortiz Ruiz et al. (2016) [55] | 2008–2012 | 81 (3) | 52 | 17 | 17 | 6 | 3 | 4 | 1 |
| Costa Rica | Villalobos et al. (2016) [56] | 2007–2011 | 210 (1) | 38 | 42 | 10 | 7 | NR f | NR | 3 |
| Mexico | Gonzalez et al. (2008) [57] | 2004–2007 | 398 (5) | 31.9 | 37.9 | 14.8 | 8 | 2.7 | 1.3 | 3.3 |
| Mexico | Corzo-Leon et al. (2014) [58] | 2008–2010 | 74 (2) | 46 | 5 | 26 | 13.5 | 5 | 3 | 1.5 |
| Peru | Bustamante et al. (2014) [59] | 2009–2011 | 153 (1) | 39.9 | 28.1 | 23.5 | 5.2 | 0.7 | 2 | 0.6 |
| Puerto Rico | Conde-Rosa et al. (2010) [60] | 2005–2006 | 85 (1) | 28 | 49 | 17 | 4 | 1 | 0 | 1 |
| Venezuela | Franco et al. (2008) [61] | 2003–2005 | 154 (6) | 18.8 | 26 | 39 | 7.8 | 2.6 | 0 | 5.8 |
| Multic d | Pfaller et al. (2000) [62] | 1997–1998 | 107 (7) | 43 | 26.1 | 16.8 | 6.6 | 0.9 | 0 | 6.6 |
| Multic | Pfaller et al. (2001) [63] | 1997–1999 | 132 (9) | 45 | 25 | 16 | 6 | 1 | NR | 7 |
| Multic | Godoy et al. (2003) [64] | 1999–2000 | 103 (5) | 42 | 21.3 | 24.2 | 7.7 | 0 | 2.9 | 1.9 |
| Multic | Nucci et al. (2013) [7] | 2008–2010 | 672 (21) | 37.6 | 26.5 | 17.6 | 6.3 | 2.7 | 6.5 | 2.8 |
| Country | Reference; Number of Isolates | Method | Species | Fluconazole | Voriconazole | ||
|---|---|---|---|---|---|---|---|
| SDD a | R b | SDD | R | ||||
| Argentina | Rodero et al. (2006) [28]; | CLSI c × | Ca e | NR i | 15.7% (1.8%) | NT j | NT |
| n = 265 | (EUCAST) | Cp f | NR | 0% (0%) | NT | NT | |
| Ct g | NR | 43% (5.4%) | NT | NT | |||
| Argentina | Cordoba et al. (2011) [29]; | EUCAST d | Ca | NR | 0% | NR | 0.5% |
| n = 420 | Cp | NR | 2.5% | NR | 0.8% | ||
| Ct | NR | 4.2% | NR | 4.2% | |||
| Brazil | Colombo et al. (2003) [34]; | CLSI | Ca | 1.2% | 0% | NT | NT |
| n = 200 | Cp | 0% | 0% | NT | NT | ||
| Ct | 0% | 0% | NT | NT | |||
| Brazil | Antunes et al. (2004) [36]; | CLSI | Ca | 0% | 0% | NT | NT |
| n = 120 | Cp | 0% | 0% | NT | NT | ||
| Ct | 0% | 0% | NT | NT | |||
| Brazil | Aquino et al. (2005) [37]; | CLSI | Ca | 0% | 0% | NT | NT |
| n = 131 | Cp | 0% | 0% | NT | NT | ||
| Ct | 0% | 0% | NT | NT | |||
| Brazil | Colombo et al. (2006) [38]; | CLSI | Ca | 0.3% | 0.3% | 0% | 0.3% |
| n = 712 | Cp | 0% | 0% | 0% | 0% | ||
| Ct | 1.3% | 0% | 0% | 0% | |||
| Brazil | Colombo et al. (2007) [40]; | CLSI | Ca | 0% | 0% | NT | NT |
| n = 282 | Cp | 0% | 0% | NT | NT | ||
| Ct | 0% | 0% | NT | NT | |||
| Brazil | da Matta et al. (2007) [41]; | CLSI | Ca | 0% | 0% | 0% | 0% |
| n = 1000 | Cp | 0% | 0% | 0% | 0% | ||
| Ct | 0% | 0% | 0% | 0% | |||
| Brazil | Bonfietti et al. (2012) [46]; | EUCAST | Ca | 0% | 0% | 0% | 0% |
| n = 100 | Cp | 6% | 0% | 0% | 3% | ||
| Ct | 0% | 0% | 0% | 0% | |||
| Brazil | Colombo et al. (2013) [15]; | CLSI | Ca | 0% | 0% | 0% | 0% |
| n = 300 | Cp | 1.3% | 0% | 0% | 0% | ||
| Ct | 2.5% | 0% | 0% | 0% | |||
| Brazil | Santos et al. (2014) [49]; | CLSI | Ca | 9.9% | 0% | 2.6% | 0% |
| n = 422 | Cp | 7% | 0% | 3.5% | 0% | ||
| Ct | 19.5% | 7.3% | 12% | 4.9% | |||
| Brazil | da Costa et al. (2014) [50]; | EUCAST | Ca | NR | 3.7% | NR | 3.7% |
| n = 108 | Cp | NR | 26.9% | NR | 0% | ||
| Ct | NR | 3,2% | NR | 3.2% | |||
| Mexico | Gonzalez et al. (2014) [57]; | CLSI | Ca | 0% | 0.8% | 0% | 0.8 |
| n = 398 | Cp | 0% | 0% | 0% | 0% | ||
| Ct | 0% | 0% | 0% | 0% | |||
| Mexico | Corzo-Leon et al. (2014) [58]; | CLSI | Ca | 0% | 0% | 0% | 0% |
| n = 74 | Cp | 0% | 0% | 0% | 0% | ||
| Ct | 0% | 0% | 0% | 0% | |||
| Peru | Bustamante et al. (2014) [59]; | CLSI | Ca | NR | NR | 0% | 5% |
| n = 153 | Cp | NR | 2.3% | NR | NR | ||
| Ct | NR | NR | NR | NR | |||
| Multicenter | Godoy et al. (2003) [64]; | CLSI | Ca | 0% | 0% | NT | NT |
| studies h | n = 103 | Cp | 0% | 0% | NT | NT | |
| Ct | 0% | 0% | NT | NT | |||
| Multicenter | Nucci et al. (2013) [7]; | CLSI | Ca | 0,4% | 0% | 0% | 0% |
| studies | n = 672 | Cp | 1,1% | 0% | 0% | 0% | |
| Ct | 0% | 0% | 0% | 0% | |||
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Da Matta, D.A.; Souza, A.C.R.; Colombo, A.L. Revisiting Species Distribution and Antifungal Susceptibility of Candida Bloodstream Isolates from Latin American Medical Centers. J. Fungi 2017, 3, 24. https://doi.org/10.3390/jof3020024
Da Matta DA, Souza ACR, Colombo AL. Revisiting Species Distribution and Antifungal Susceptibility of Candida Bloodstream Isolates from Latin American Medical Centers. Journal of Fungi. 2017; 3(2):24. https://doi.org/10.3390/jof3020024
Chicago/Turabian StyleDa Matta, Daniel Archimedes, Ana Carolina Remondi Souza, and Arnaldo Lopes Colombo. 2017. "Revisiting Species Distribution and Antifungal Susceptibility of Candida Bloodstream Isolates from Latin American Medical Centers" Journal of Fungi 3, no. 2: 24. https://doi.org/10.3390/jof3020024
APA StyleDa Matta, D. A., Souza, A. C. R., & Colombo, A. L. (2017). Revisiting Species Distribution and Antifungal Susceptibility of Candida Bloodstream Isolates from Latin American Medical Centers. Journal of Fungi, 3(2), 24. https://doi.org/10.3390/jof3020024







