Characterization of Collagen Fibers (I, III, IV) and Elastin of Normal and Neoplastic Canine Prostatic Tissues
Abstract
1. Introduction
2. Materials and Methods
2.1. The Subjects
2.2. Picrosirius (PSR)
2.3. Immunohistochemistry (IHC)
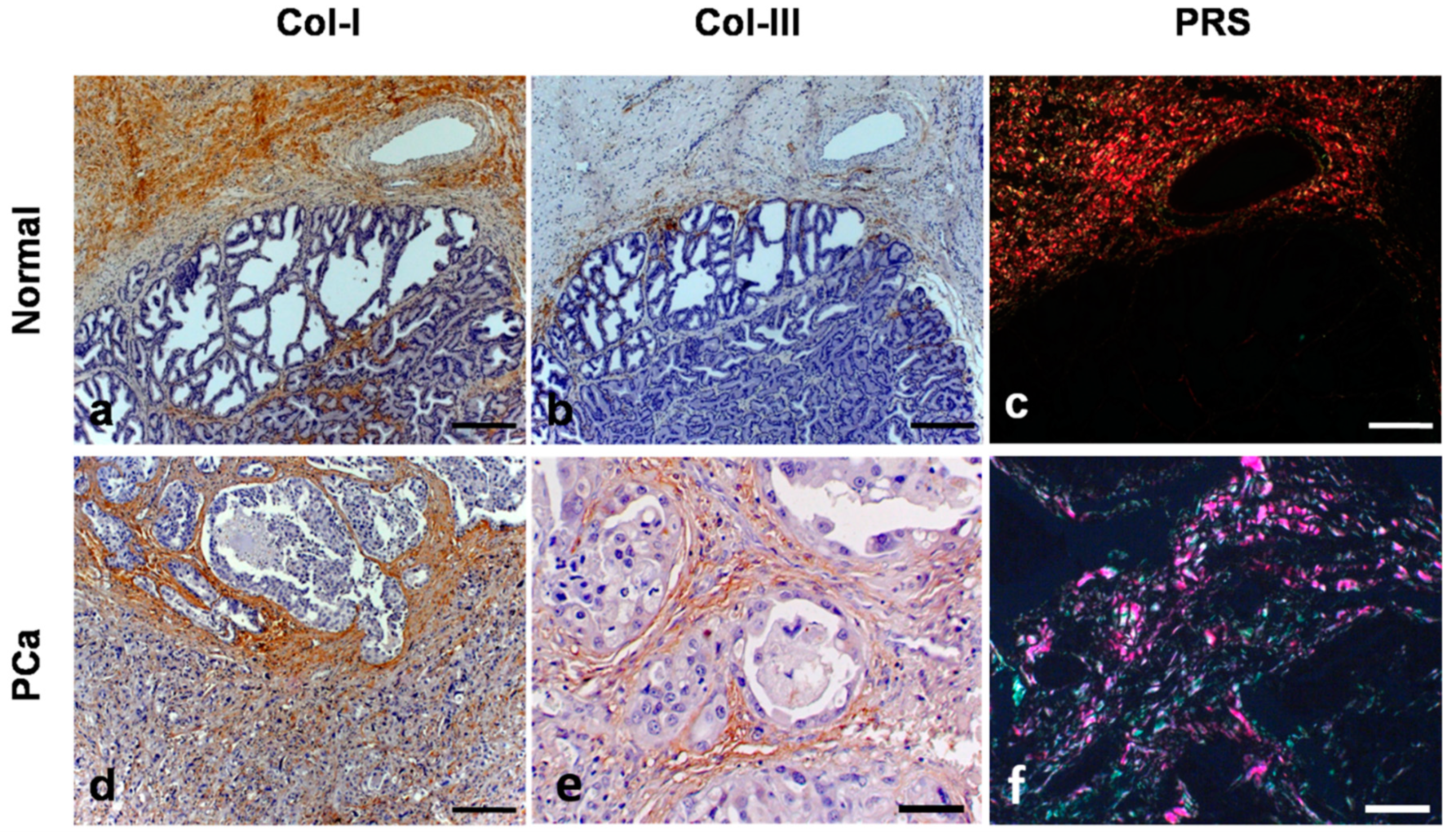
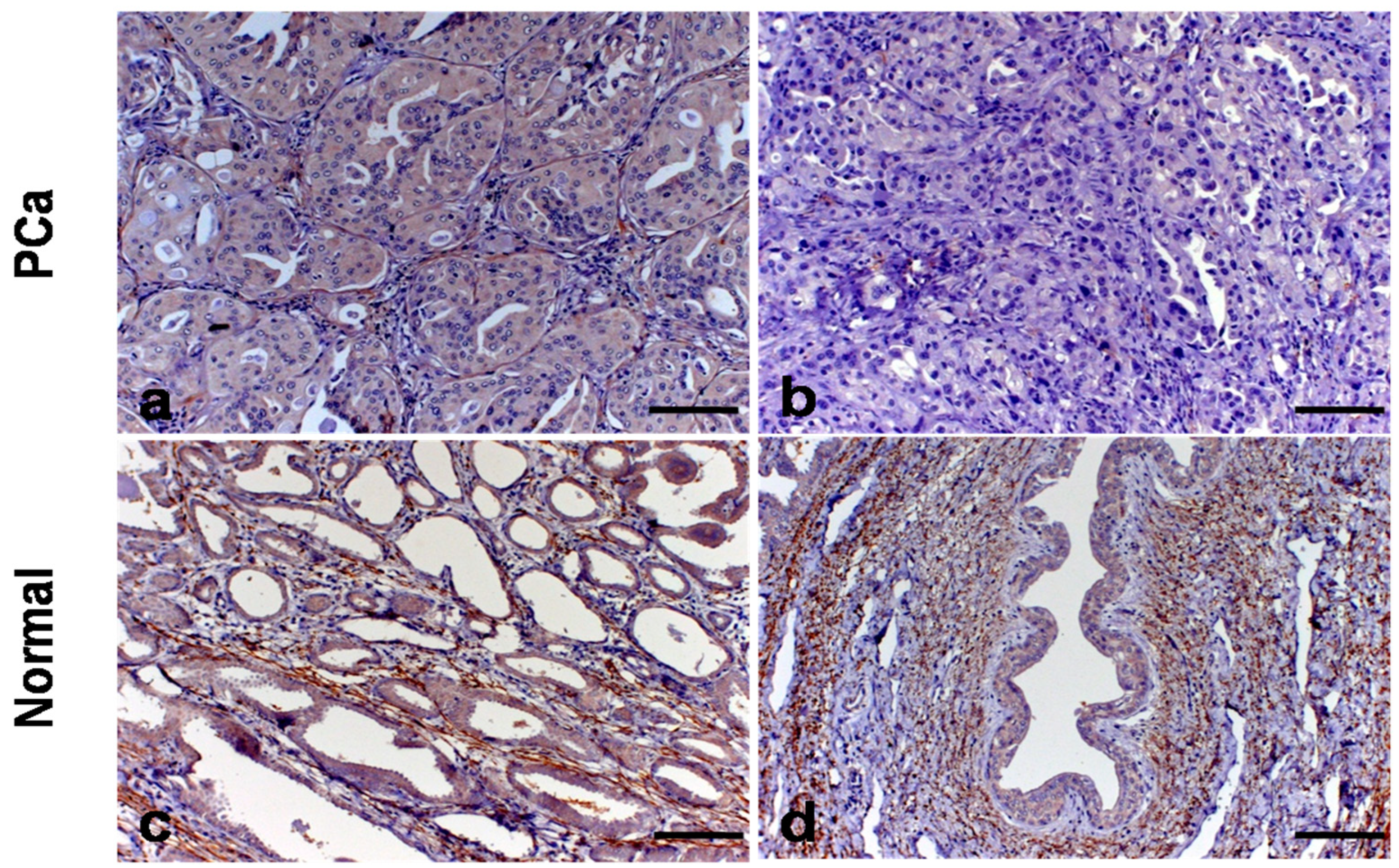
2.4. Interpretation of PSR and IHC Staining
2.5. Data Analysis
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Ferlay, J.; Soerjomataram, I.; Ervik, M.; Dikshit, R.; Eser, S.; Mathers, C.; Rebelo, M.; Parkin, D.M.; Forman, D.; Bray, F. Cancer incidence and mortality worldwide: Source, methods and major patterns in GLOBOCAN 2012. Int. J. Cancer 2014, 136. [Google Scholar] [CrossRef] [PubMed]
- Mundy, G. Metastasis to bone: Causes, consequences and therapeutic opportunities. Nat. Rev. Cancer 2002, 2, 584–593. [Google Scholar] [CrossRef] [PubMed]
- Leroy, B.R.; Northrup, N. Prostate cancer in dogs: Comparative and clinical aspects. Vet. J. 2009, 180, 149–162. [Google Scholar] [CrossRef] [PubMed]
- Palmieri, C.; Lean, F.Z.; Akter, S.H.; Romussi, S.; Grieco, V. A retrospective analysis of 111 canine prostatic samples: Histopathological findings and classification. Res. Vet. Sci. 2014, 97, 568–573. [Google Scholar] [CrossRef]
- Argyle, D.J. Prostate cancer in dogs and men: Unique opportunity to study the disease. Vet. J. 2009, 180, 137–138. [Google Scholar] [CrossRef] [PubMed]
- Palmieri, C.; Grieco, V. Proposal of Gleason-like grading system of canine prostate carcinoma in veterinary pathology practice. Res. Vet. Sci. 2015, 103, 11–15. [Google Scholar] [CrossRef] [PubMed]
- Fonseca-Alves, C.E.; Kobayashi, P.E.; Rivera Calderón, L.G.; Felisbino, S.L.; Rinaldi, J.C.; Drigo, S.A.; Rogatto, S.R.; Laufer-Amorim, R. Immunohistochemical panel to characterize canine prostate carcinomas according to aberrant p63 expression. PLoS ONE 2018, 12, e0199173. [Google Scholar] [CrossRef] [PubMed]
- Palumbo, A., Jr.; Ferreira, L.B.; de Souza, P.A.R.; Oliveira, F.L.; Pontes, B.; Viana, N.B.; Machado, D.E.; Palmero, C.Y.; Alves, L.M.; Gimba, E.R.; et al. Extracellular matrix secreted by reactive stroma is a main inducer of pro-tumorigenic features on LNCaP prostate cancer cells. Cancer Lett. 2012, 321, 55–64. [Google Scholar] [CrossRef] [PubMed]
- Penet, M.F.; Kakkad, S.; Pathak, A.P.; Krishnamachary, B.; Mironchik, Y.; Raman, V.; Solaiyappan, M.; Bhujwalla, Z.M. Structure and function of prostate cancer dissemination-permissive extracellular matrix. Clin. Cancer Res. 2016, 23, 2245–2254. [Google Scholar] [CrossRef] [PubMed]
- Frantz, C.; Stewart, K.M.; Weaver, V.M. The extracellular matrix at a glance. J. Cell. Sci. 2010, 123, 4195–4200. [Google Scholar] [CrossRef] [PubMed]
- Theocharis, A.D.; Skandalis, S.S.; Gialeli, C.; Karamanos, N.K. Extracellular matrix structure. Adv. Drug Deliv. Rev. 2016, 97, 4–27. [Google Scholar] [CrossRef] [PubMed]
- Rozario, T.; DeSimone, D.W. The extracellular matrix in development and morphogenesis: A dynamic view. Dev. Biol. 2010, 341, 126–140. [Google Scholar] [CrossRef] [PubMed]
- Tuxhorn, J.A.; Ayala, G.E.; Rowley, D.R. Reactive stroma prostate cancer progression. J. Urol. 2001, 166, 2472–2483. [Google Scholar] [CrossRef]
- Zhang, K.; Corsa, C.A.; Ponik, S.M.; Prior, J.L.; Piwnica-Worms, D.; Eliceiri, K.W.; Keely, P.J.; Longmore, G.D. The collagen receptor discoidin domain receptor 2 stabilizes SNAIL1 to facilitate breast cancer metastasis. Nat. Cell Biol. 2013, 15, 677–687. [Google Scholar] [CrossRef] [PubMed]
- Zeng, Z.S.; Cohen, A.M.; Guillem, J.G. Loss of basement membrane type IV collagen is associated with increased expression of metalloproteinases 2 and 9 during human colorectal tumorigenesis. Carcinogenesis 1999, 20, 749–755. [Google Scholar] [CrossRef] [PubMed]
- Tanjore, H.; Kalluri, R. The role of type IV collagen and basement membranes in cancer progression and metastasis. Am. J. Pathol. 2006, 168, 715–717. [Google Scholar] [CrossRef] [PubMed]
- Provenzano, P.P.; Inman, D.R.; Eliceiri, K.W.; Knittel, J.G.; Yan, L.; Rueden, C.T.; White, J.G.; Keely, P.J. Collagen density promotes mammary initiation and progression. BMC Med. 2008, 6, 11. [Google Scholar] [CrossRef] [PubMed]
- Li, A.; Zhou, T.; Guo, L.; Si, J. Collagen type I regulates ß-catenin tyrosine phosphorylation and nuclear translocation to promote migration and proliferation of gastric carcinoma cells. Oncol. Rep. 2010, 23, 1247–1255. [Google Scholar] [PubMed]
- Case, A.; Brisson, B.K.; Durham, A.C.; Rosen, S.; Monslow, J.; Buza, E.; Salah, P.; Gillem, J.; Ruthel, G.; Veluvolu, S.; et al. Identification of prognostic collagen signatures and potential therapeutic stromal targets in canine mammary gland carcinoma. PLoS ONE 2017, 12, e0180448. [Google Scholar] [CrossRef] [PubMed]
- Eble, J.N.; Sauter, G.; Epstein, I.A. Sesterhenn Pathology and Genetics of Tumours of the Urinary System and Male Genital Organs; International Agency for Research on Cancer: Lyon, France, 2004. [Google Scholar]
- Coleman, R. Picrosirius red staining revisited. Acta Histochem. 2011, 113, 231–233. [Google Scholar] [CrossRef] [PubMed]
- Ruetten, H.; Wegner, K.A.; Romero, M.F.; Wood, M.W.; Marker, P.C.; Strand, D.; Colopy, S.A.; Vezina, C.M. Prostatic collagen architecture in neutered and intact canines. Prostate 2018, 78, 839–848. [Google Scholar] [CrossRef] [PubMed]
- Bauman, T.M.; Nicholson, T.M.; Abler, L.L.; Eliceiri, K.W.; Huang, W.; Vezina, C.M.; Ricke, W.A. Characterization of fibrillar collagens and extracellular matrix of glandular benign prostatic hyperplasia nodules. PLoS ONE 2014, 9, e109102. [Google Scholar] [CrossRef] [PubMed]
- Yang, F.; Tuxhorn, J.A.; Ressler, S.J.; McAlhany, S.J.; Dang, T.D.; Rowley, D.R. Stromal expression of connective tissue growth factor promotes angiogenesis and prostate cancer tumorigenesis. Cancer Res. 2005, 65, 8887–8895. [Google Scholar] [CrossRef] [PubMed]
- Martin, M.; Pujuguet, P.; Martin, F. Role of stromal myofibroblasts infiltrating colon cancer in tumor invasion. Pathol. Res. Pract. 1996, 192, 712–717. [Google Scholar] [CrossRef]
- Tuxhorn, J.A.; McAlhany, S.J.; Dang, T.D.; Ayala, G.E.; Rowley, D.R. Stromal cells promote angiogenesis and growth of human prostate tumors in a differential reactive stroma (DRS) xenograft model. Cancer Res. 2002, 62, 3298–3307. [Google Scholar] [PubMed]
- Valastyan, S.; Weinberg, R.A. Tumor metastasis: Molecular insights and evolving paradigms. Cell 2011, 147, 275–292. [Google Scholar] [CrossRef] [PubMed]
- Jones, C.E.; Hammer, A.M.; Cho, Y.; Sizemore, G.M.; Cukierman, E.; Yee, L.D.; Ghadiali, S.N.; Ostrowski, M.C.; Leight, J.L. Stromal PTEN Regulates Extracellular Matrix Organization in the Mammary Gland. Neoplasia 2019, 21, 132–145. [Google Scholar] [CrossRef] [PubMed]
- Wegner, K.A.; Keikhosravi, A.; Eliceiri, K.W.; Venzina, C.M. Fluorescence of picrosirius red multiplexed with immunohistochemistry for the quantitative assessment of collagen in tissue sections. J. Histochem. Cytochem. 2017, 65, 479–490. [Google Scholar] [CrossRef] [PubMed]
- Benazzi, C.; Sarli, G.; Galeotti, M.; Marcato, P.S. Basement membrane components in mammary tumours of the dog and cat. J. Comp. Pathol. 1993, 109, 241–252. [Google Scholar] [CrossRef]
- Murakami, M.; Sakai, H.; Kodama, A.; Yanai, T.; Mori, T.; Maruo, K.; Masegi, T. Activation of matrix metalloproteinase (MMP-2) by membrane type 1-MMP and abnormal immunolocalization of the basement membrane components laminin and type IV collagen in canine spontaneous hemangiosarcomas. Histol. Histopathol. 2009, 24, 437–446. [Google Scholar] [CrossRef] [PubMed]
- Bedoya, S.A.O.; Conceição, L.G.; Viloria, M.I.V.; Loures, F.H.; Valente, F.L.; Amorim, R.L.; Silva, F.F. Caracterização de colágenos tipos I e III no estroma do carcinoma de células escamosas cutâneo em cães. Arquivo Brasileiro de Medicina Veterinária e Zootecnia 2016, 68, 147–154. [Google Scholar] [CrossRef]
- Koutsilieris, M.; Sourla, A.; Pelletier, G.; Doillon, C.J. Three-dimensional type I collagen gel system for the study of osteoblastic metastases produced by metastatic prostate cancer. J. Bone Miner. Res. 1994, 9, 1823–1832. [Google Scholar] [CrossRef] [PubMed]
- Hall, C.L.; Dai, J.; van Golen, K.L.; Keller, E.T.; Long, M.W. Type I collagen receptor (alpha 2 beta 1) signaling promotes the growth of human prostate cancer cells within the bone. Cancer Res. 2006, 66, 8648–8654. [Google Scholar] [CrossRef] [PubMed]
- Kylmälä, T.; Tammela, T.L.; Risteli, L.; Risteli, J.; Kontturi, M.; Elomaa, I. Type I collagen degradation product (ICTP) gives information about the nature of bone metastases and has prognostic value in prostate cancer. Br. J. Cancer 1995, 71, 1061–1064. [Google Scholar] [CrossRef] [PubMed]
- Burns-Cox, N.; Avery, N.C.; Gingell, J.C.; Bailey, A.J. Changes in collagen metabolism in prostate cancer: A host response that may alter progression. J. Urol. 2001, 166, 1698–1701. [Google Scholar] [CrossRef]
- Duarte, A.H.; Colli, S.; Alves-Pereira, J.L.; Martins, M.P.; Sampaio, F.J.B.; Ramos, C.F. Collagen I and III metalloproteinase gene and protein expression in prostate cancer in relation to Gleason score. Int. Braz. J. Urol. 2012, 38, 341–354. [Google Scholar] [CrossRef] [PubMed]
- Jablonka, F.; Alves Bda, C.; de Oliveira, C.G.; Wroclawski, M.L.; Szwarc, M.; de Oliveira Vitória, W.; Fonseca, F.; Del Giglio, A. Serum crosslinked-N-terminal telopeptide of type I collagen (NTx) has prognostic implications for patients with initial prostate carcinoma (PCa): A pilot study. Clin. Chim. Acta 2014, 436, 316–318. [Google Scholar] [CrossRef] [PubMed]
- Brehmer, B.; Biesterfeld, S.; Jakse, G. Expression of matrix metalloproteinases (MMP-2 and -9) and their inhibitors (TIMP-1 and -2) in prostate cancer tissue. Prostate Cancer Prostatic Dis. 2003, 6, 217–222. [Google Scholar] [CrossRef] [PubMed]
- Faleiro, M.B.; Croce, G.B.; Toledo, D.C.; Rodrigues, M.M.; Batista, A.C.; Damasceno, A.D.; Brito, L.A.; Laufer-Amorim, R.; de Moura, V.M. Matrix metalloproteinases 2 and 9 expression in canine normal prostate and with proliferative disorders. Ciência Rural 2013, 43, 6. [Google Scholar] [CrossRef]
- Timpl, R. Macromolecular organization of basement membranes. Curr. Opin. Cell Biol. 1996, 8, 618–624. [Google Scholar] [CrossRef]
- Cassereau, L.; Miroshnikova, Y.A.; Ou, G.; Lakins, J.; Weaver, V.M. A 3D tension bioreactor platform to study the interplay between ECM stiffness and tumor phenotype. J. Biotechnol. 2015, 193, 66–69. [Google Scholar] [CrossRef] [PubMed]
- Haage, A.; Schneider, I.C. Cellular contractility and extracellular matrix stiffness regulate matrix metalloproteinase activity in pancreatic cancer cells. FASEB J. 2014, 28, 3589–3599. [Google Scholar] [CrossRef] [PubMed]
- Docheva, D.; Padula, D.; Schieker, M.; Clausen-Schaumann, H. Effect of collagen I and fibronectin on the adhesion, elasticity and cytoskeletal organization of prostate cancer cells. Biochem. Biophys. Res. Commun. 2010, 402, 361–366. [Google Scholar] [CrossRef] [PubMed]
- Kai, F.; Laklai, H.; Weaver, V.M. Force matters: Biomechanical regulation of cell Invasion and migration in disease. Trends Cell Biol. 2016, 26, 486–497. [Google Scholar] [CrossRef] [PubMed]
- Bissell, M.J.; Hines, W.C. Why don’t we get more cancer? A proposed role of the microenvironment in restraining cancer progression. Nat. Med. 2011, 17, 320–329. [Google Scholar] [CrossRef] [PubMed]
- Miyazaki, K.; Oyanagi, J.; Hoshino, D.; Togo, S.; Kumagai, H.; Miyagi, Y. Cancer cell migration on elongate protrusions of fibroblasts in collagen matrix. Sci. Rep. 2019, 9, 292. [Google Scholar] [CrossRef] [PubMed]
- Babichenko, I.I.; Andriukhin, M.I.; Pulbere, S.; Loktev, A. Immunohistochemical expression of matrix metalloproteinase-9 and inhibitor of matrix metalloproteinase-1 in prostate adenocarcinoma. Int. J. Clin. Exp. Pathol. 2014, 7, 9090–9098. [Google Scholar] [PubMed]
- Sinha, A.A.; Gleason, D.F.; DeLeon, O.F.; Wilson, M.J.; Limas, C.; Reddy, P.K.; Furcht, L.T. Localization of type IV collagen in the basement membranes of human prostate and lymph nodes by immunoperoxidase and immunoalkaline phospatase. Prostate 1991, 18, 93–104. [Google Scholar] [CrossRef] [PubMed]
- Gonçalves, B.F.; Campos, S.G.; Costa, C.F.; Scarano, W.R.; Góes, R.M.; Taboga, S.R. Key participants of the tumor microenvironment of the prostate: An approach of the structural dynamic of cellular elements and extracellular matrix components during epithelial-stromal transition. Acta Histochem. 2015, 117, 4–13. [Google Scholar] [CrossRef] [PubMed]
- Marettová, E. Immunohistochemical localization of elastic system fibres in the canine prostate. Folia Veterinaria 2017, 6, 5–10. [Google Scholar] [CrossRef]
- Krušlin, B.; Ulamec, M.; Tomas, D. Prostate cancer stroma: An important factor in cancer growth and progression. Bosn. J. Basic Med. Sci. 2015, 15, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Scandolera, A.; Odoul, L.; Salesse, S.; Guillot, A.; Blaise, S.; Kawecki, C.; Maurice, P.; El Btaouri, H.; Romier-Crouzet, B.; Martiny, L.; et al. The Elastin Receptor Complex: A Unique Matricellular Receptor with High Anti-tumoral Potential. Front. Pharmacol. 2016, 7, 32. [Google Scholar] [CrossRef] [PubMed]
- Fülöp, T.; Larbi, A. Putative role of 67 kDa elastin-laminin receptor in tumor invasion. Semin. Cancer Biol. 2002, 12, 219–229. [Google Scholar] [CrossRef]
| Primary Antibody | Antigen Retrieval | Dilution | Incubation Period |
|---|---|---|---|
| Collagen I, rabbit, Novotec | Citrate buffer pH 6.0, microwave, twice for 5 min | 1:1000 | Overnight at 4 °C |
| Collagen III, rabbit, Novotec | Pepsin 2%, pH 1.4 in oven for 10 min at 60 °C after for 30 min at 37 °C. | 1:1000 | Overnight at 4 °C |
| Collagen IV, rabbit, Biorbyt | Pepsin 2%, pH 1.4, in oven for 10 min at 60 °C after for 30 min at 37 °C. | 1:1000 | Overnight at 4 °C |
| Elastin (BA-4), mouse, Santa Cruz. | Citrate buffer pH 6.0, pressure cooker (Pascal®, Dako, Carpinteria, CA, USA) | 1:100 | Overnight at 4 °C |
| PSR Test | IHC Test | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| 25% | Median | 75% | p | 25% | Median | 75% | p | ||
| Coll-I | N | 1.25 | 1.89 | 2.27 | 0.1298 | 2.85 | 4.73 | 8.03 | 0.3159 |
| PC | 2.09 | 2.24 | 2.43 | 3.31 | 6.18 | 8.56 | |||
| Coll-III | N | 1.33 | 1.64 | 2.06 | 0.001 | 1.81 | 3.22 | 5.03 | 0.05 |
| PC | 1.68 | 2.25 | 3.11 | 3.72 | 5.07 | 6.44 | |||
| Group | IHC Test | ||||
|---|---|---|---|---|---|
| 25% | Median | 75% | p | ||
| Coll-IV | Normal | 1.11 | 1.41 | 1.72 | 0.2135 |
| PC | 0.58 | 1.14 | 1.61 | ||
| Elastin | Normal | 0.25 | 0.26 | 0.42 | 0.00229 |
| PC | 0.28 | 0.43 | 0.51 | ||
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Calderón, L.G.R.; Kobayashi, P.E.; Vasconcelos, R.O.; Fonseca-Alves, C.E.; Laufer-Amorim, R. Characterization of Collagen Fibers (I, III, IV) and Elastin of Normal and Neoplastic Canine Prostatic Tissues. Vet. Sci. 2019, 6, 22. https://doi.org/10.3390/vetsci6010022
Calderón LGR, Kobayashi PE, Vasconcelos RO, Fonseca-Alves CE, Laufer-Amorim R. Characterization of Collagen Fibers (I, III, IV) and Elastin of Normal and Neoplastic Canine Prostatic Tissues. Veterinary Sciences. 2019; 6(1):22. https://doi.org/10.3390/vetsci6010022
Chicago/Turabian StyleCalderón, Luis Gabriel Rivera, Priscila Emiko Kobayashi, Rosemeri Oliveira Vasconcelos, Carlos Eduardo Fonseca-Alves, and Renée Laufer-Amorim. 2019. "Characterization of Collagen Fibers (I, III, IV) and Elastin of Normal and Neoplastic Canine Prostatic Tissues" Veterinary Sciences 6, no. 1: 22. https://doi.org/10.3390/vetsci6010022
APA StyleCalderón, L. G. R., Kobayashi, P. E., Vasconcelos, R. O., Fonseca-Alves, C. E., & Laufer-Amorim, R. (2019). Characterization of Collagen Fibers (I, III, IV) and Elastin of Normal and Neoplastic Canine Prostatic Tissues. Veterinary Sciences, 6(1), 22. https://doi.org/10.3390/vetsci6010022






