Anatomy and Pathology of the Texel Sheep Larynx
Abstract
:1. Introduction
2. Materials and Methods
2.1. Ethical Approval
2.2. Larynx Selection
2.3. Carcase Measurements
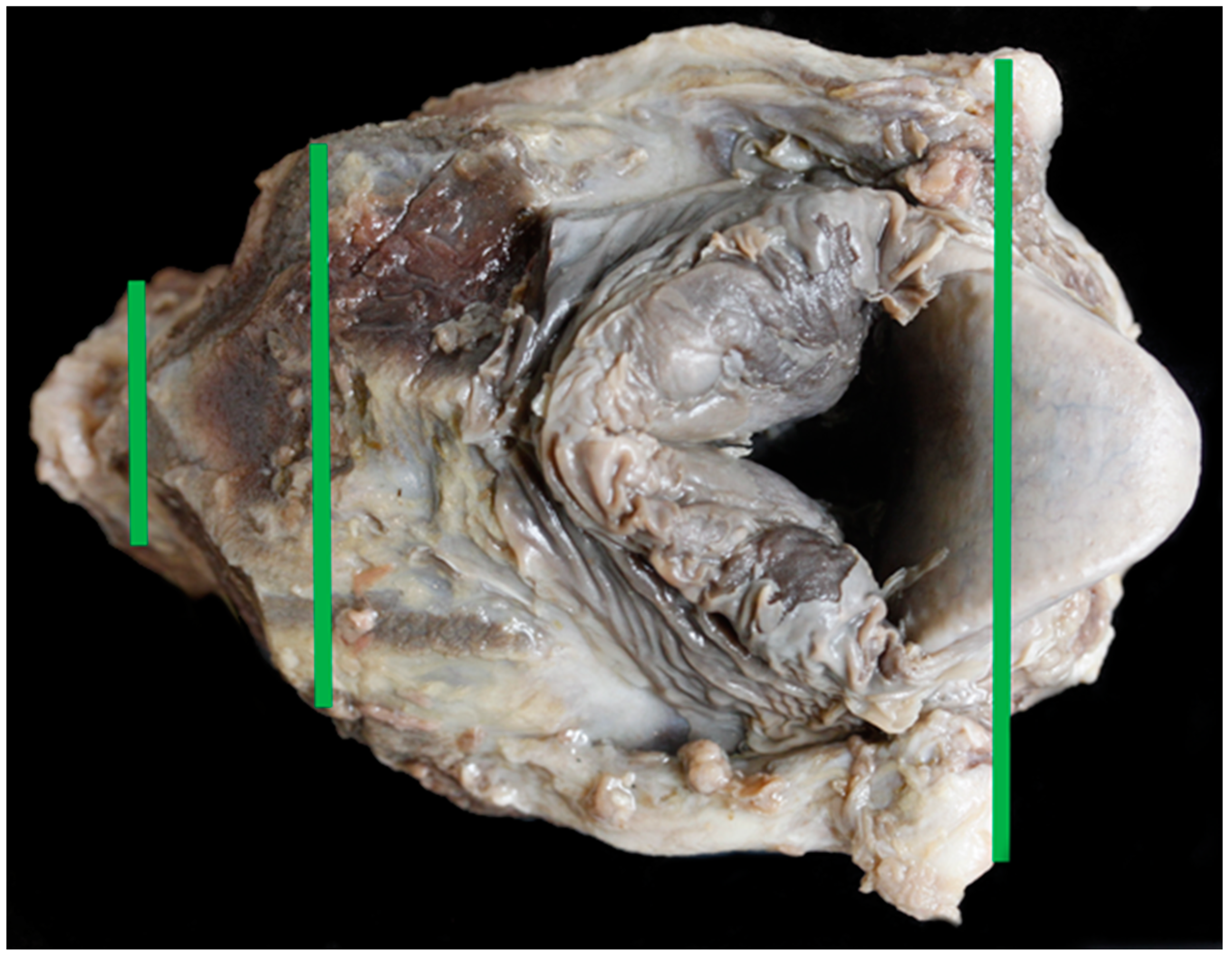
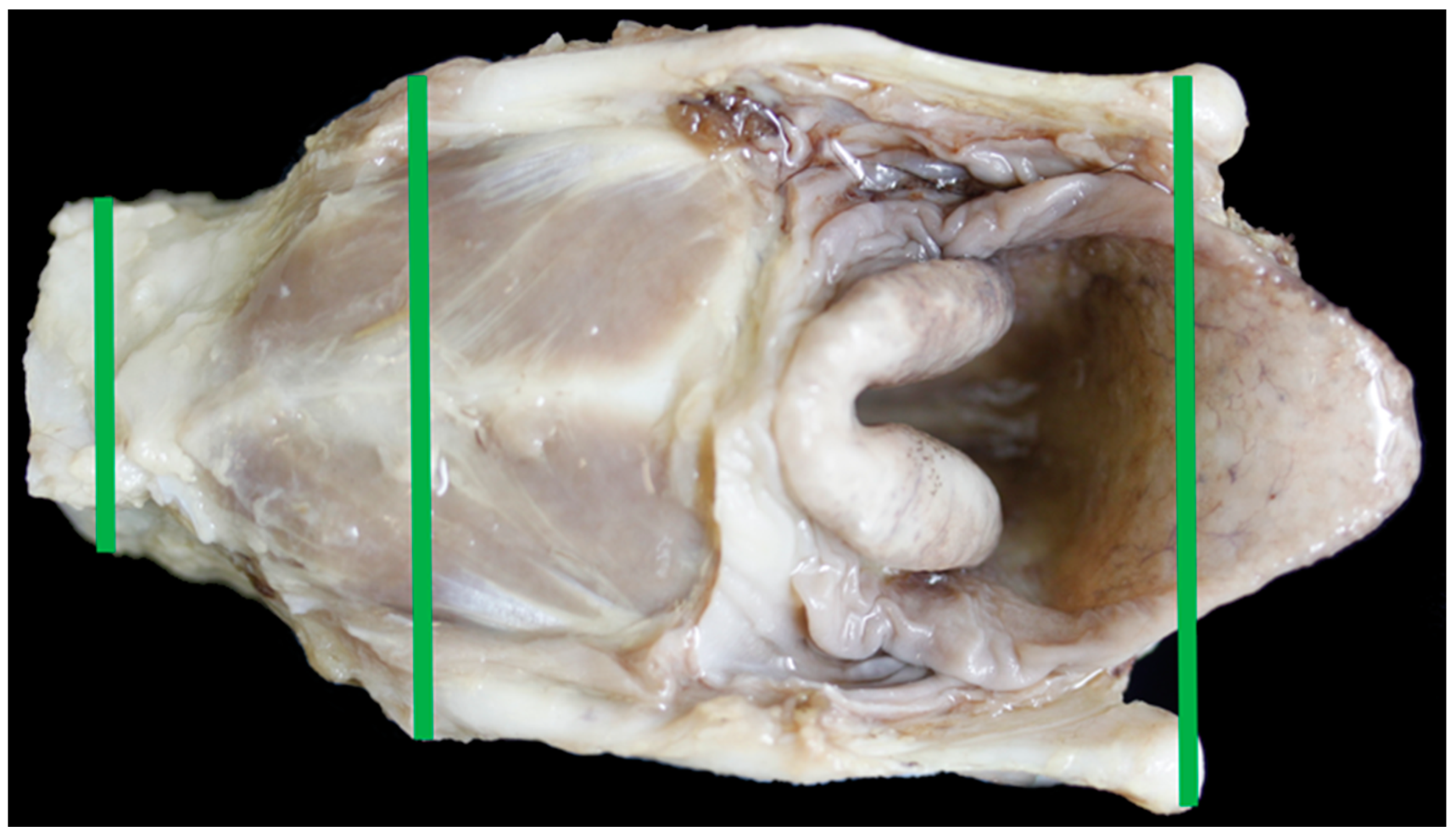
2.4. Larynx Collection, Measuring and Photography
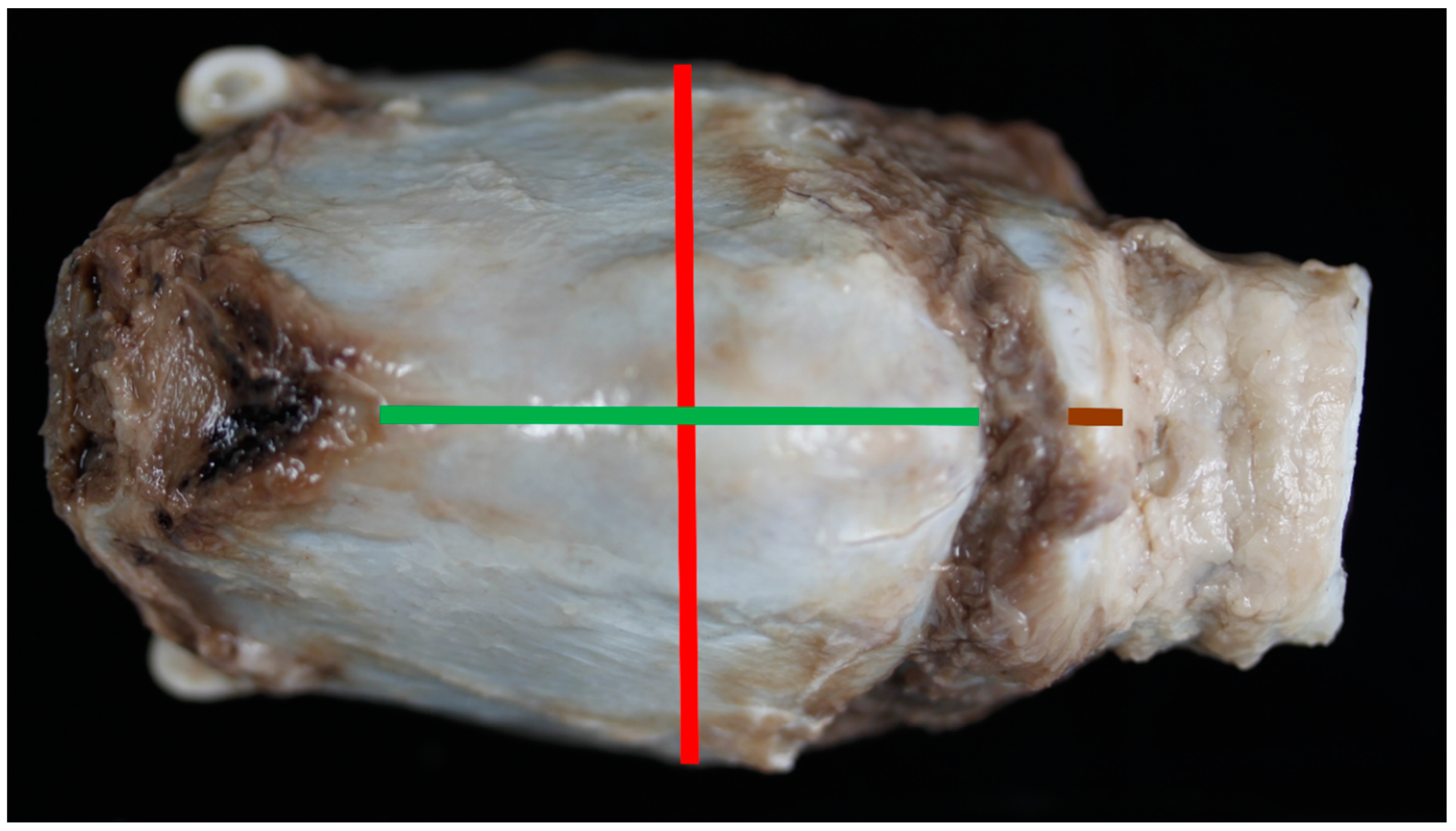
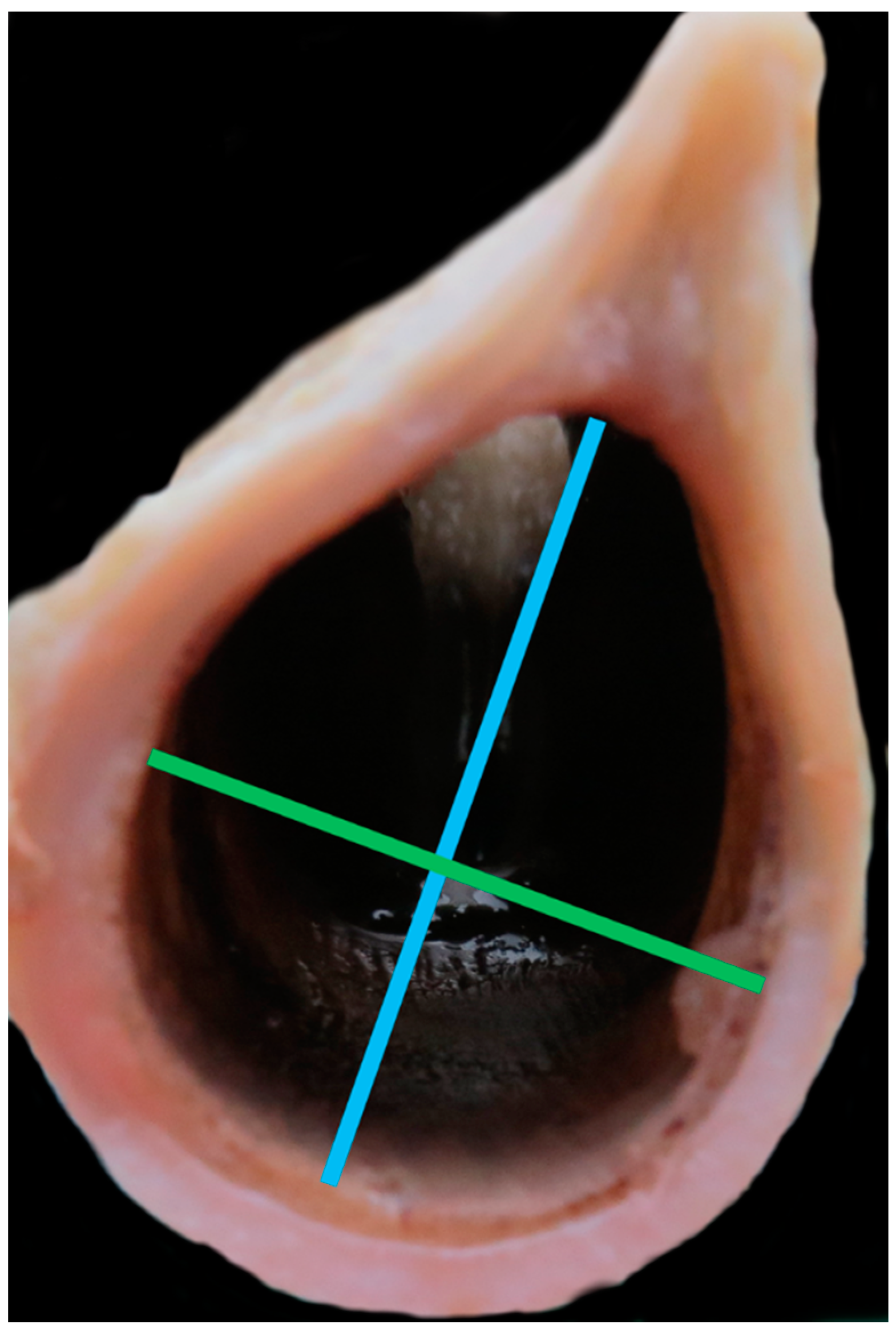
- Anterior thyroid length (distance from upper to lower thyroid incisure (mm)) (Figure 1)
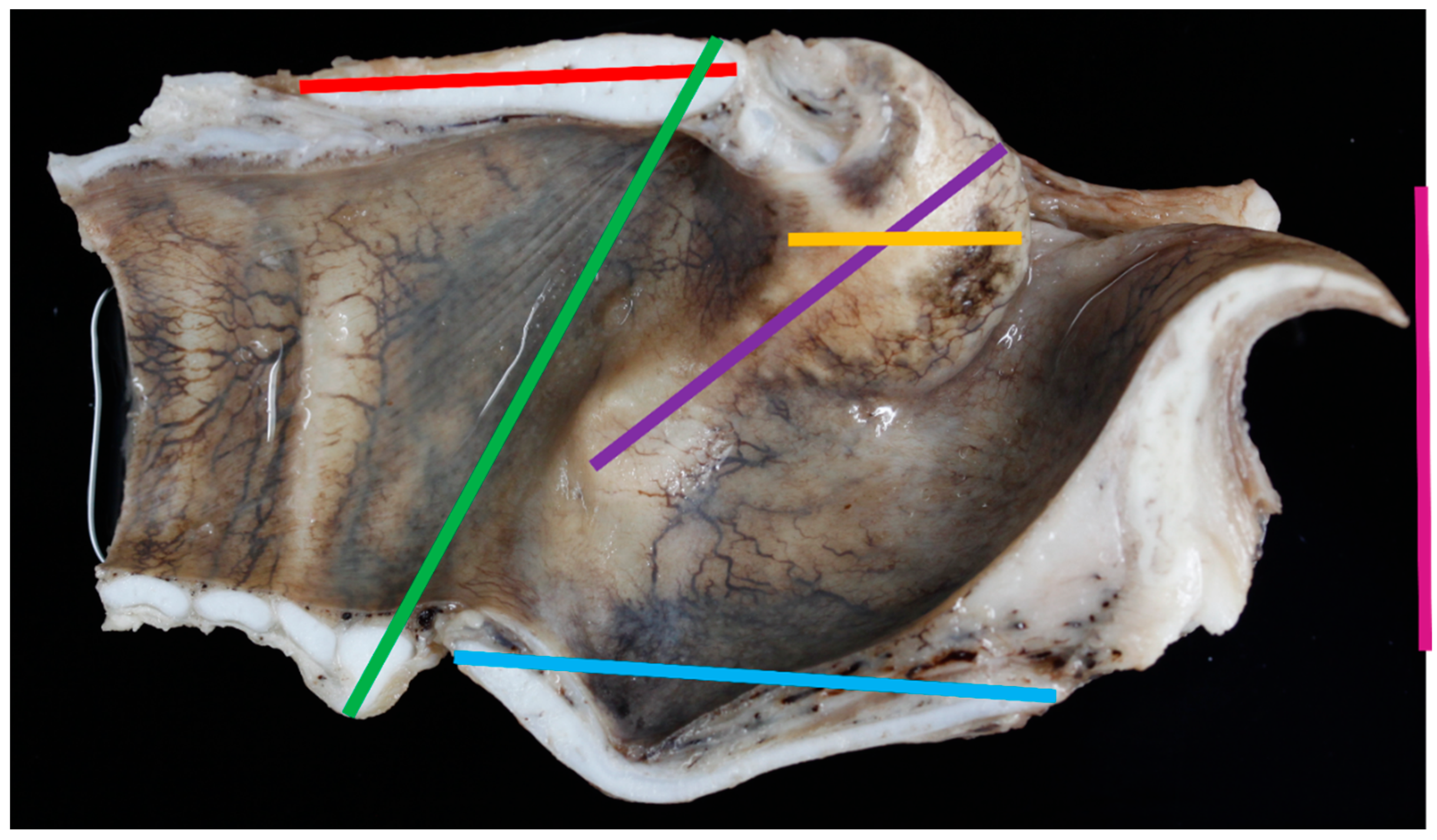
- Dorsal thyroid length (distance between tips of upper and lower horns (mm)) (Figure 3)
- Maximum thyroid breadth (length of thyroid tubercula (mm)) (Figure 1)
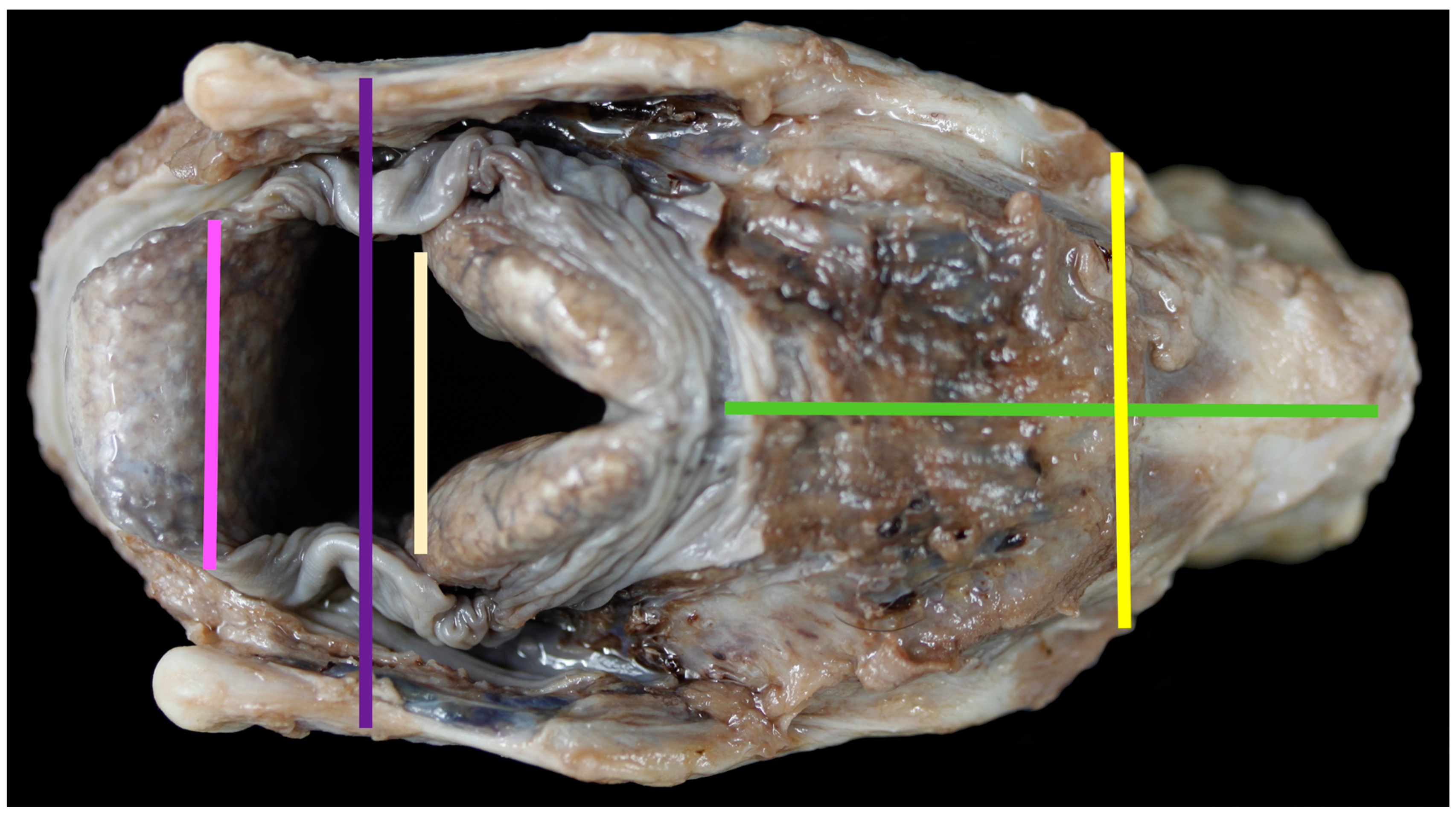
- Upper thyroid breadth (length of thyroid tubercula (mm)) (Figure 2)
- Lower thyroid breath (out distance of inferior horns (mm)) (Figure 2)
- Length of thyroid lamina (distance of notches prior to horns (mm)) (Figure 3)
- Anteroposterior dimension (distance between laryngeal prominence and plane thought posterior margins of laminae (mm)) (Figure 3)
- Main length of larynx (extension of distance 6 to lower margin of cricoid cartilage (mm))
- Length of cricoid lamina (mm) (Figure 2)
- Anterior length of cricoid arch (mm) (Figure 1)
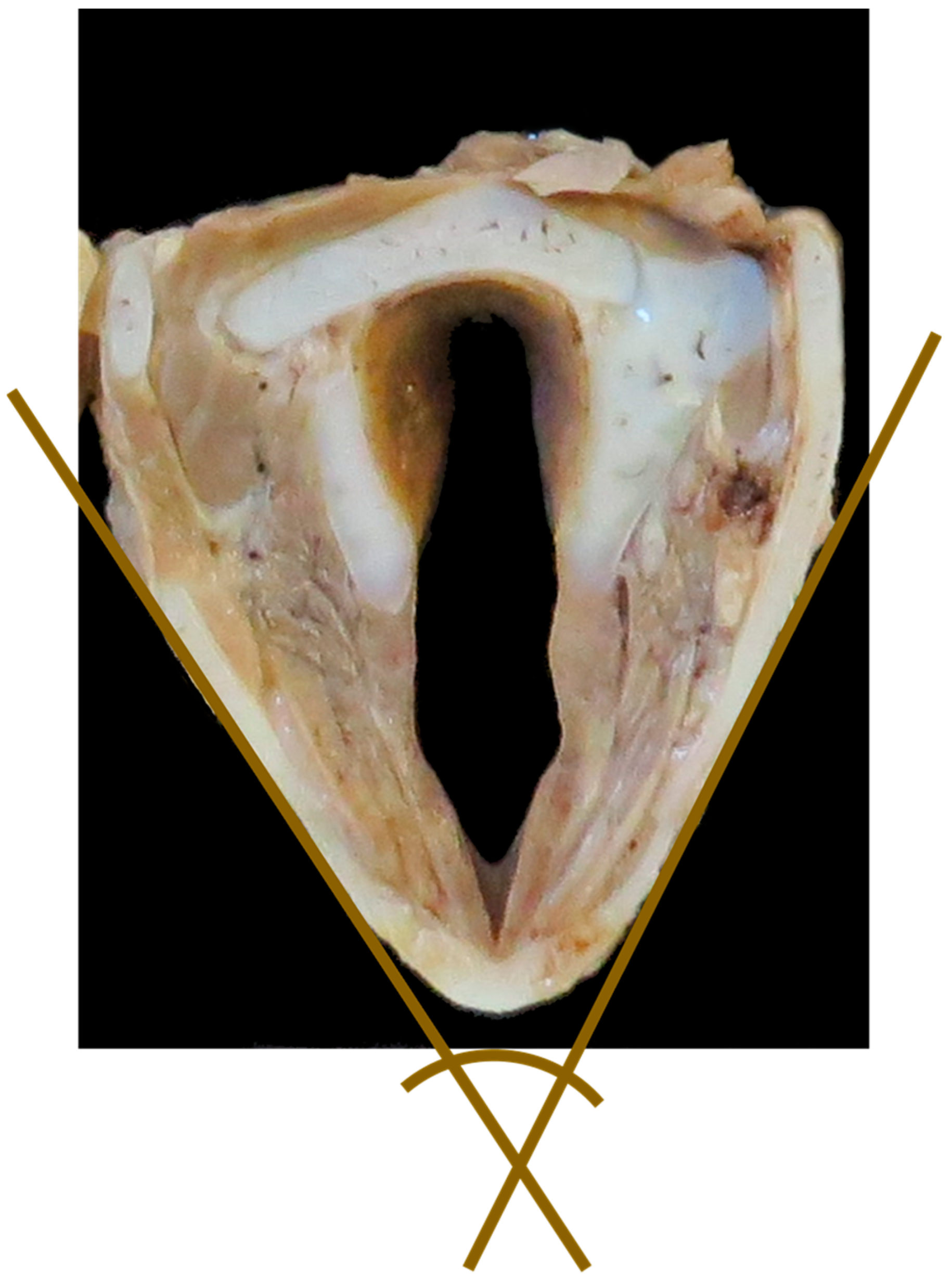
- Angle between thyroid laminae (°) (Figure 6)
- Width of epiglottis at attachment of soft tissue (mm) (Figure 2)
- Width of trachea at second tracheal ring (as felt by palpation) (mm) (Figure 5)
- Height of trachea at second tracheal ring (as felt by palpation) (mm) (Figure 5)
- Distance between midpoint cranial corniculate projections (mm) (Figure 2)
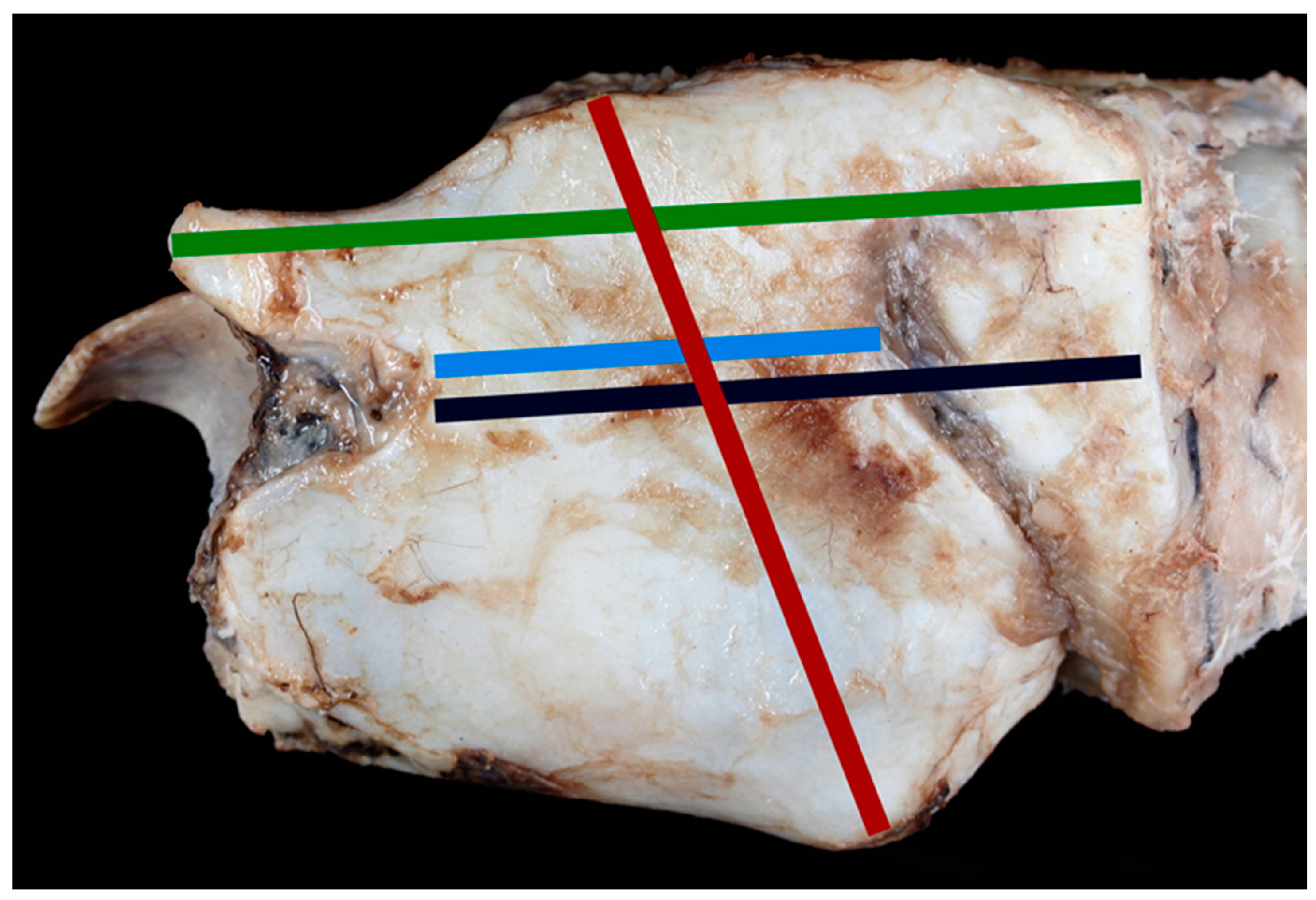
- Split cricoid length (mm) (Figure 4)
- Cricothyroid distance measuring to outer edges (mm) (Figure 4)
- Split thyroid length (mm) (Figure 4)
- Depth of the cranial AC projections (mm) (Figure 4)
- Flat length of epiglottis (mm) (Figure 4)
- Height of C-A cartilage (mm) (Figure 4)
2.5. Imaging
2.6. Statistical Analysis
3. Results
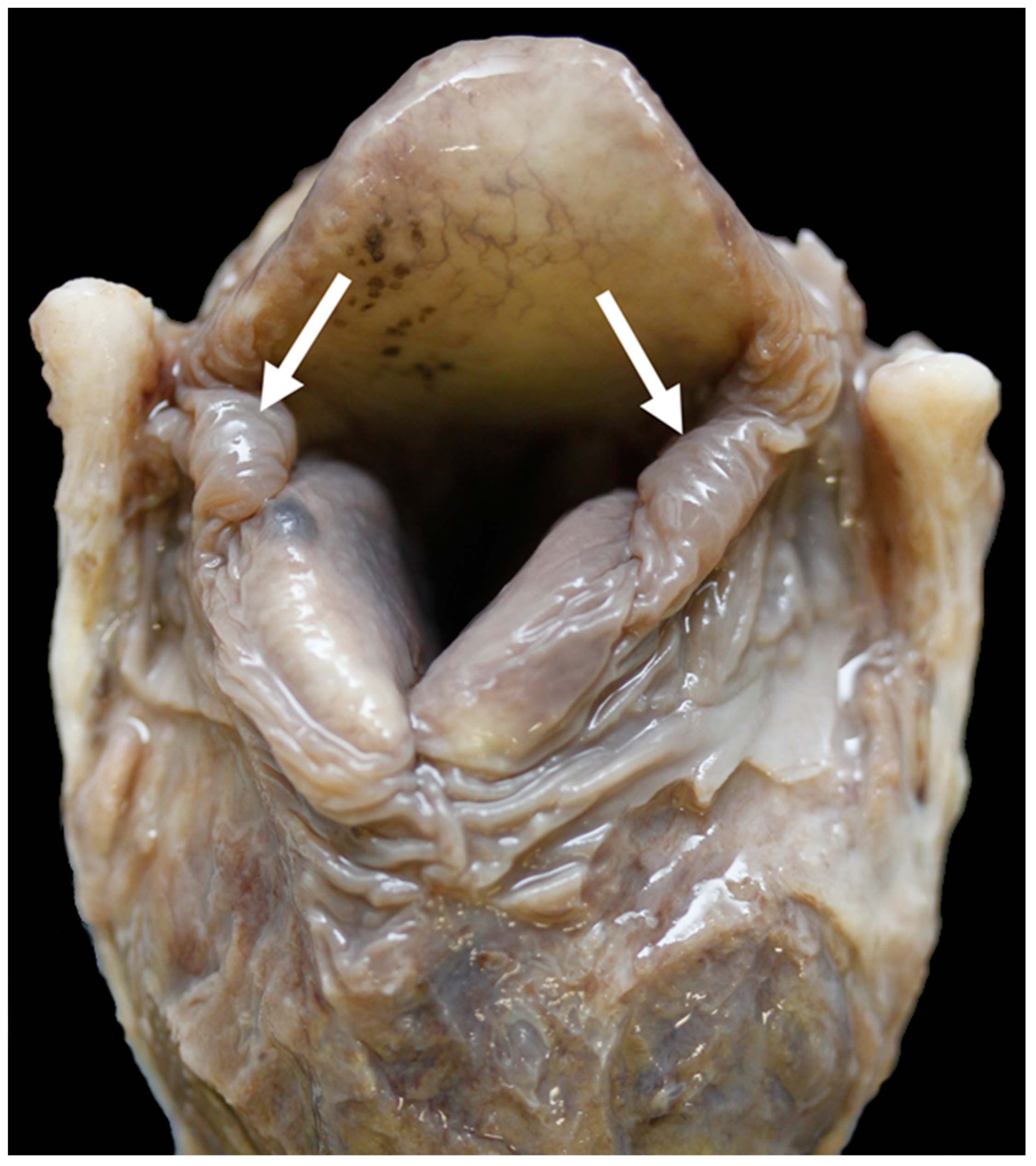
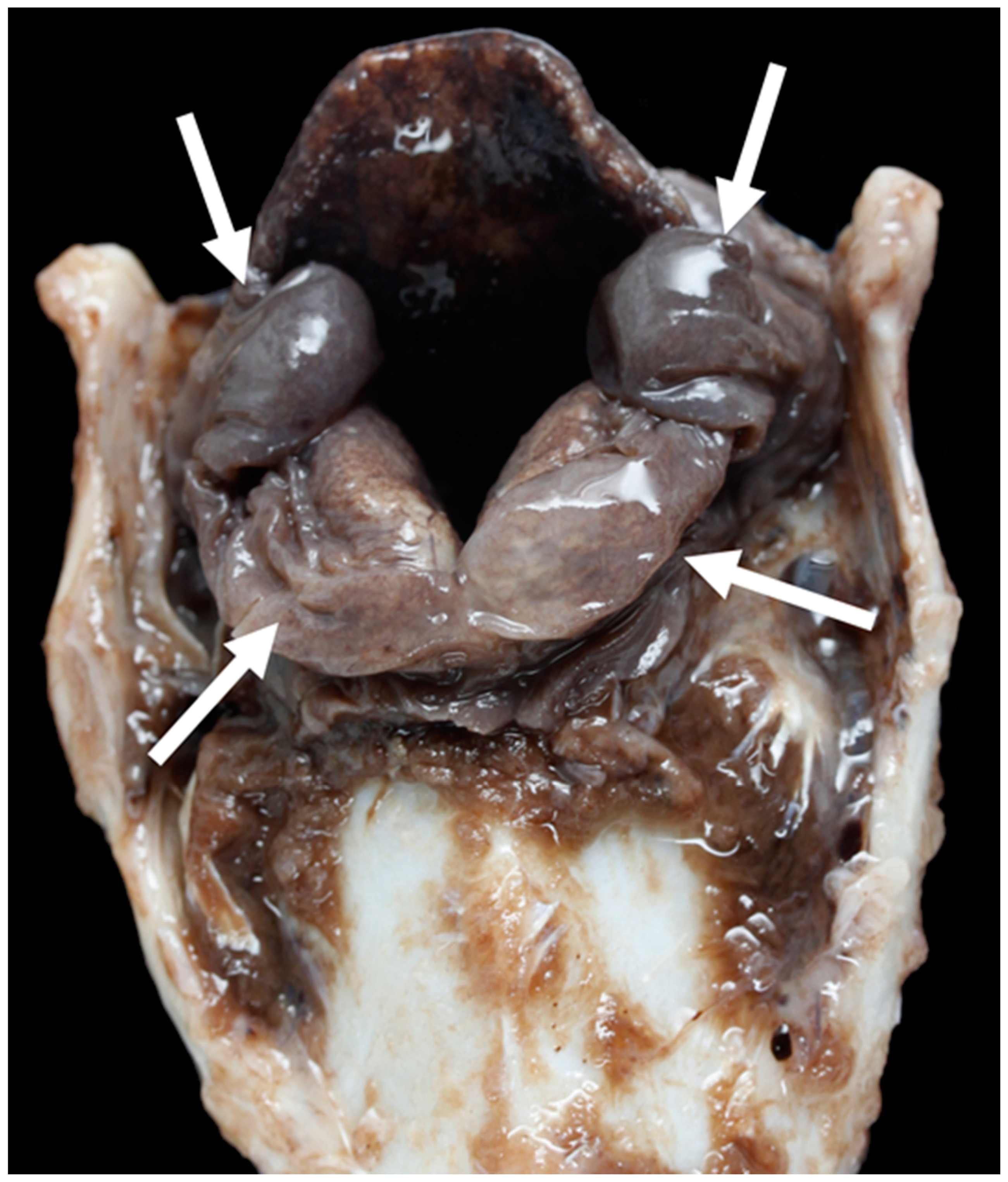
3.1. Anatomy of the Larynx
General Observations
3.2. Carcase Measurements
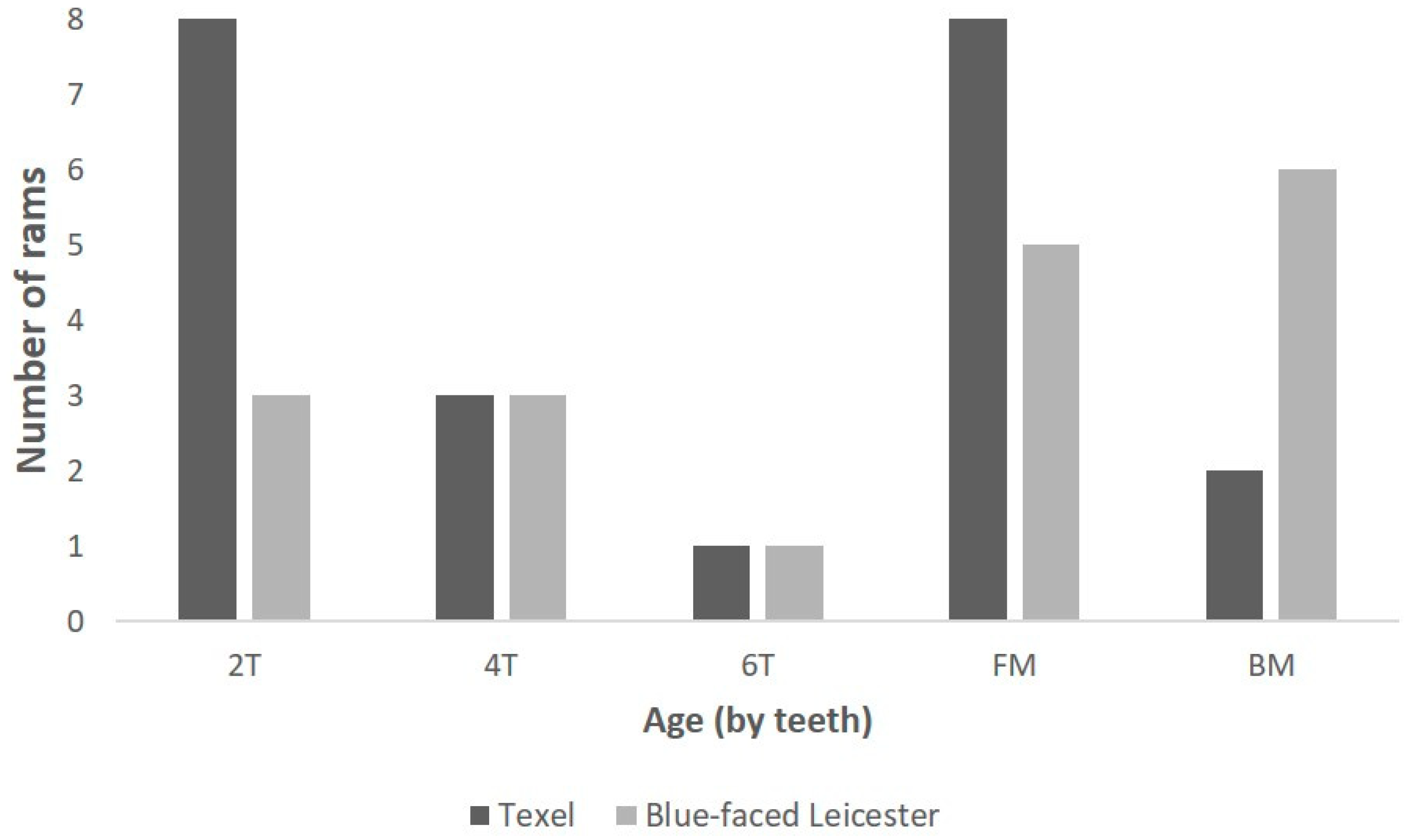
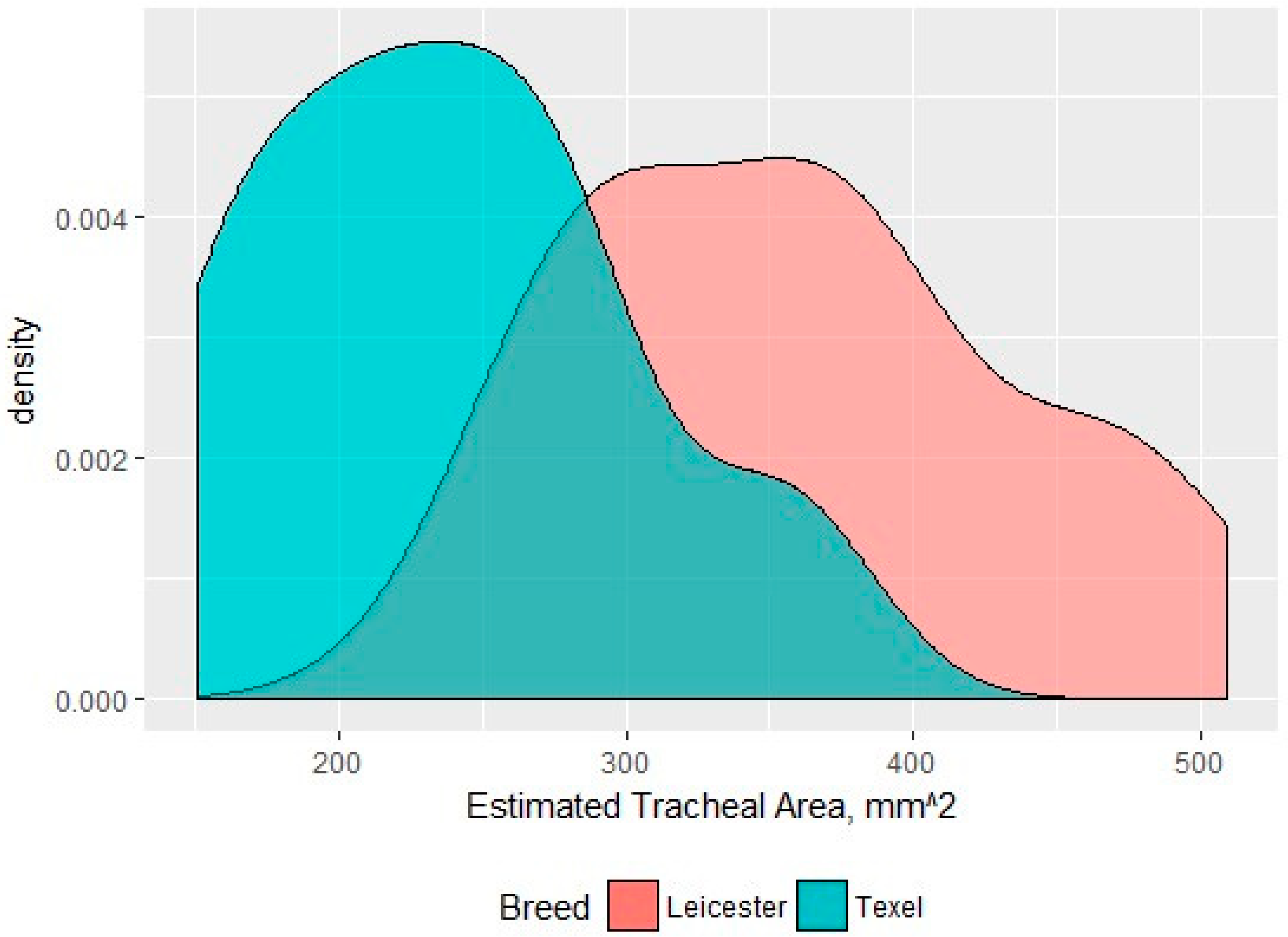
3.3. Larynx Measurements
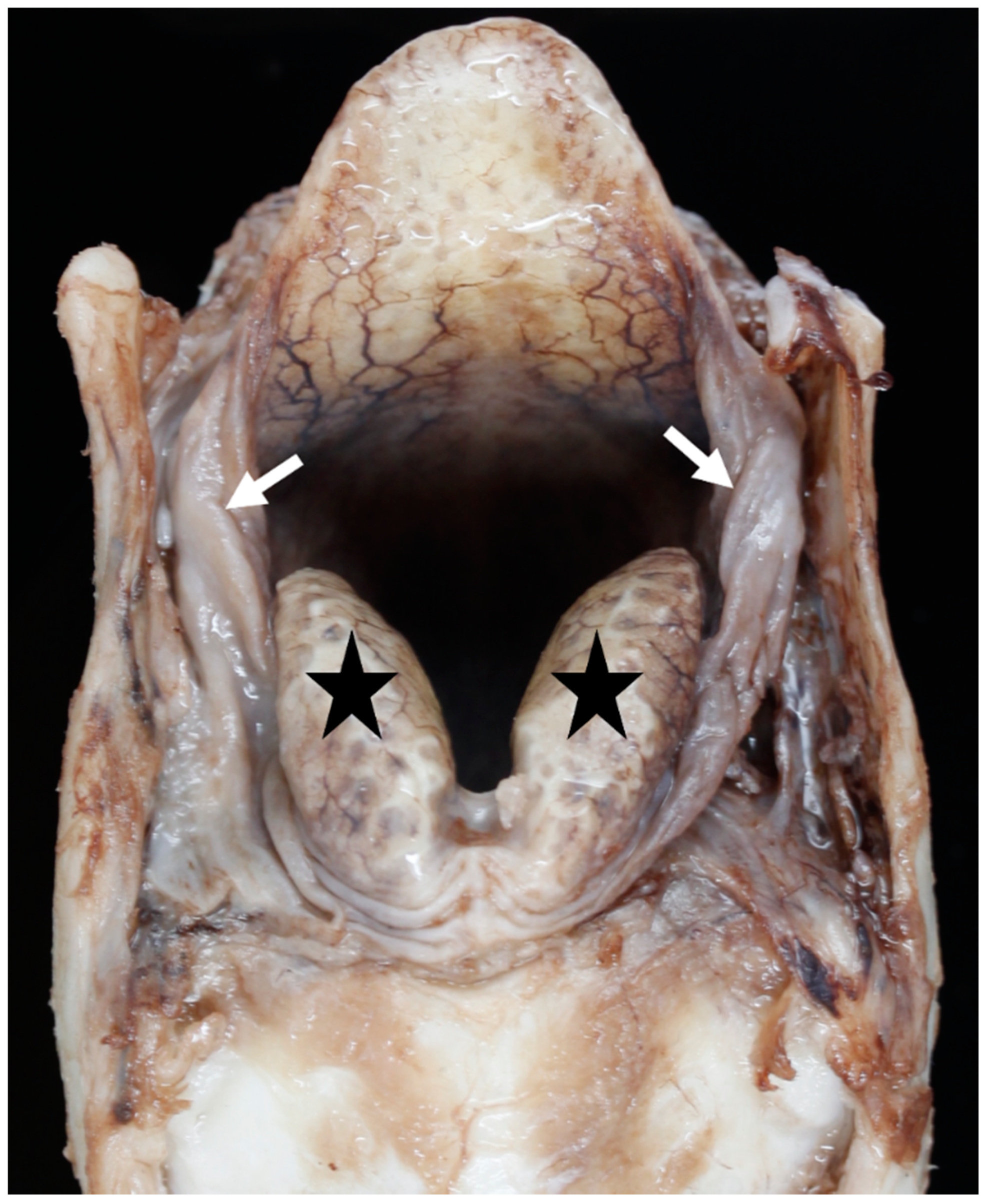
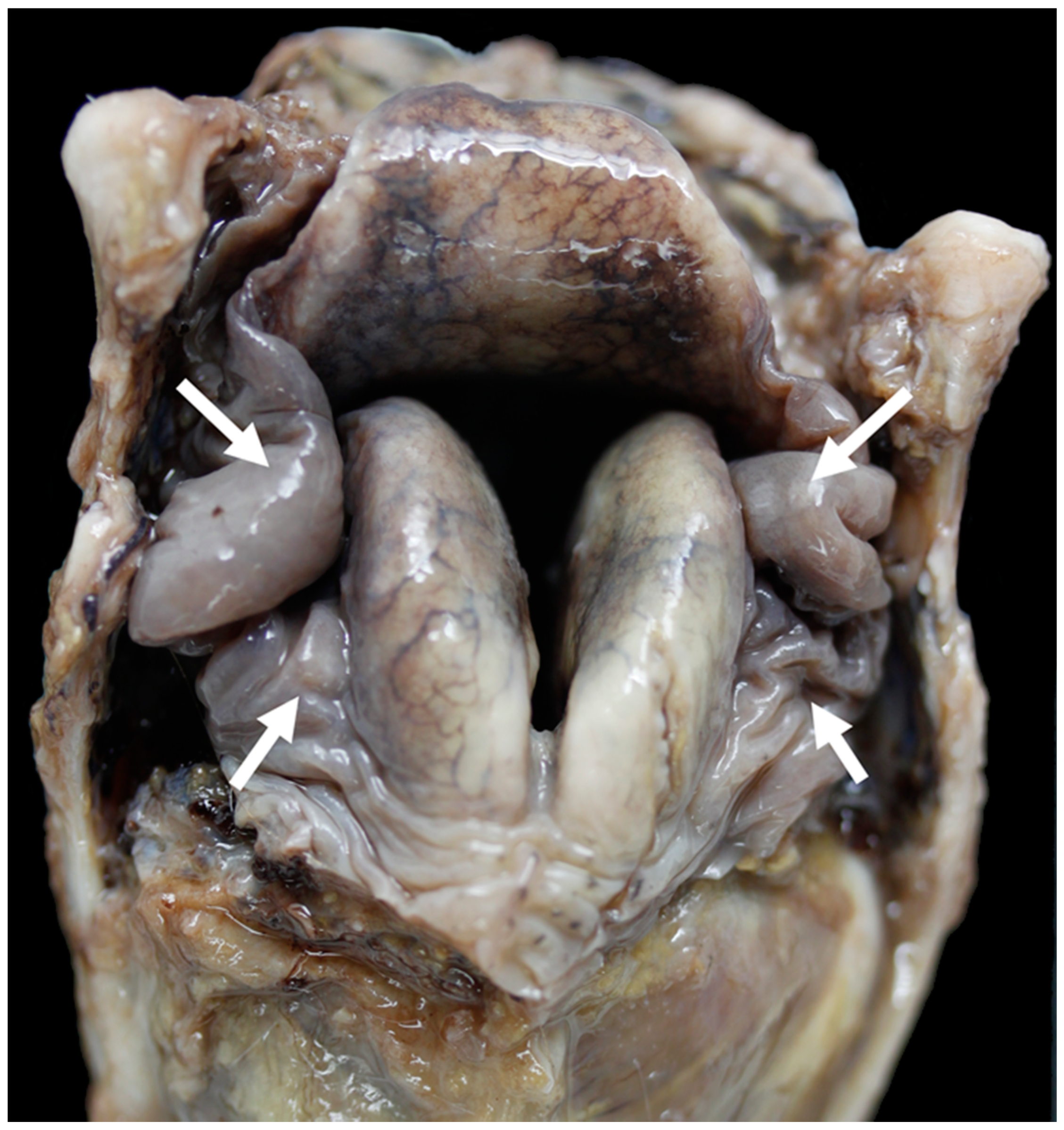
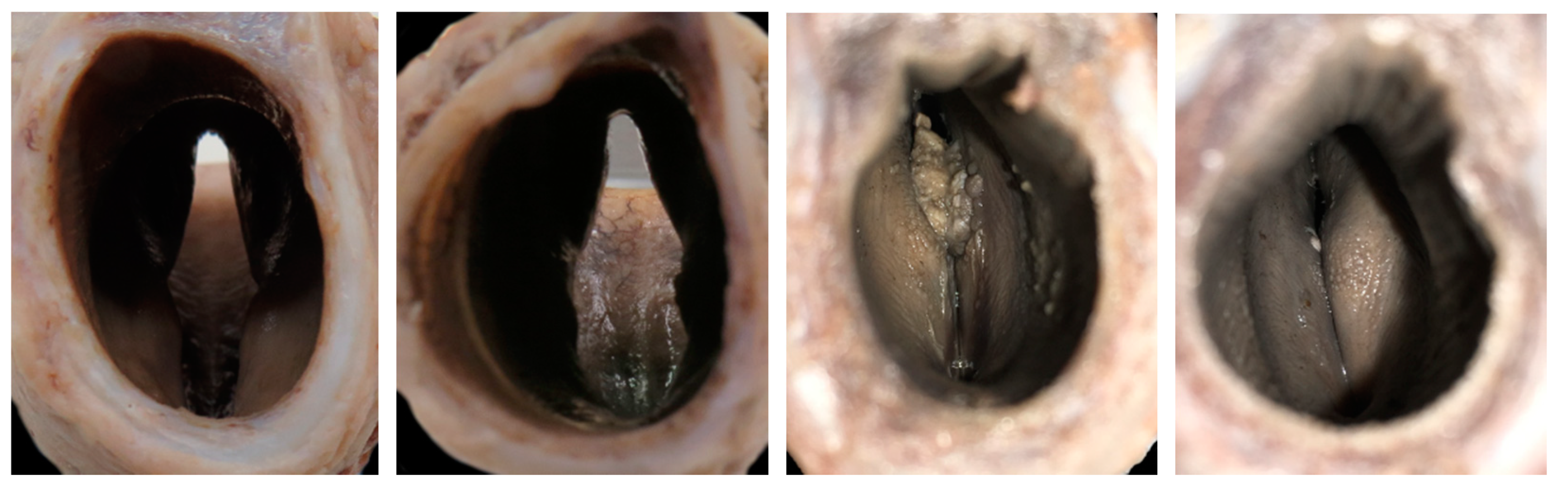
3.4. The Glottis
3.5. Diagnostic Imaging
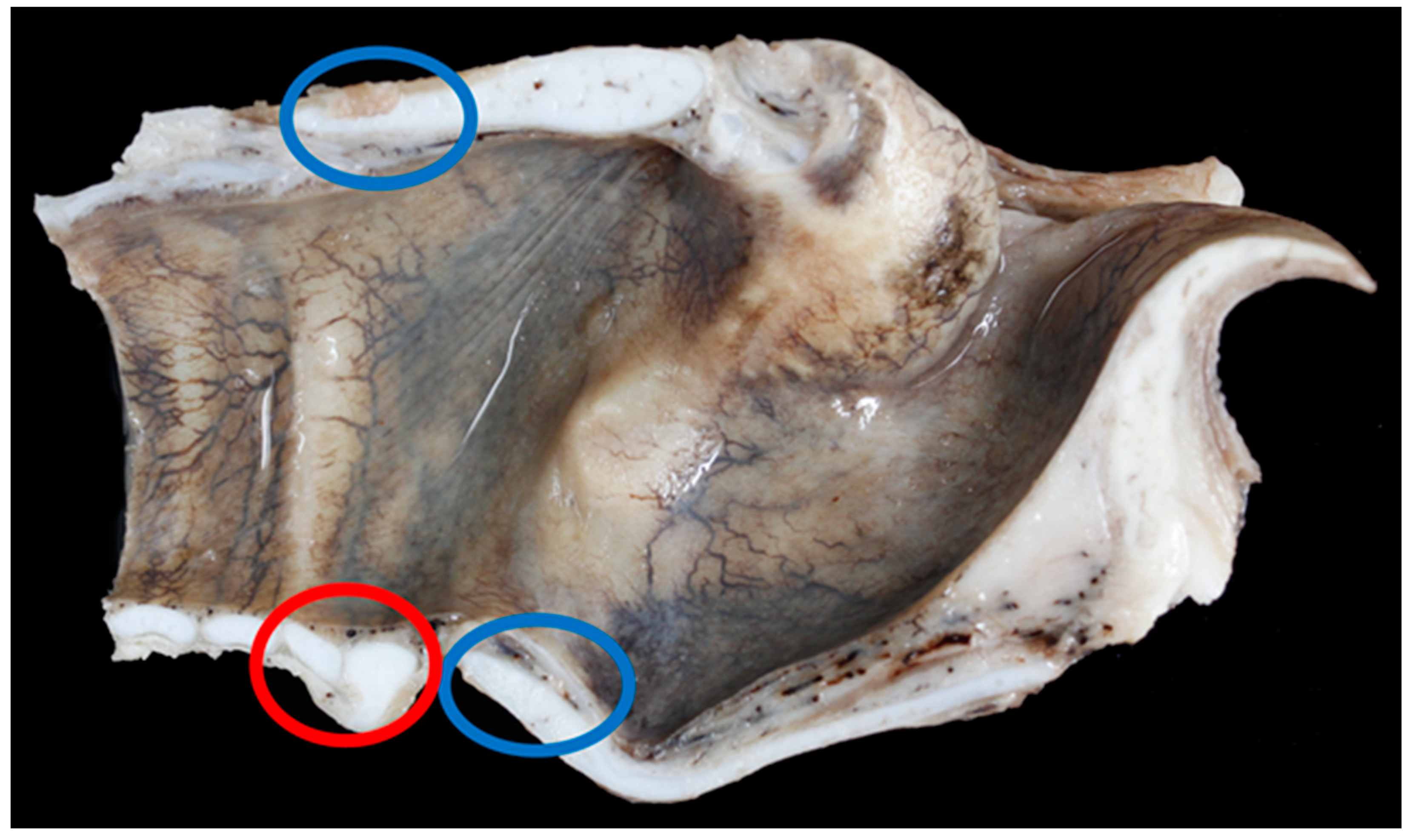
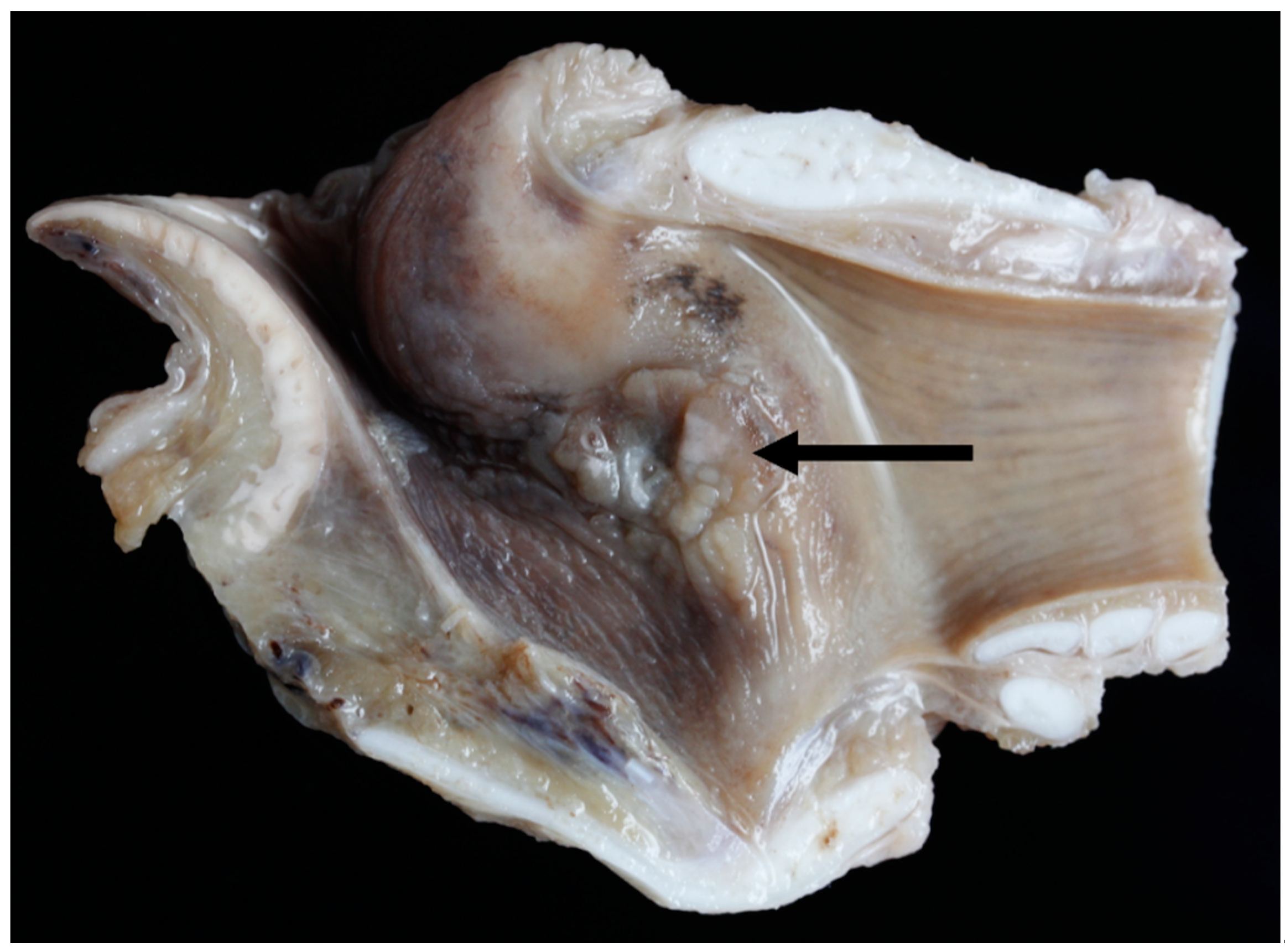
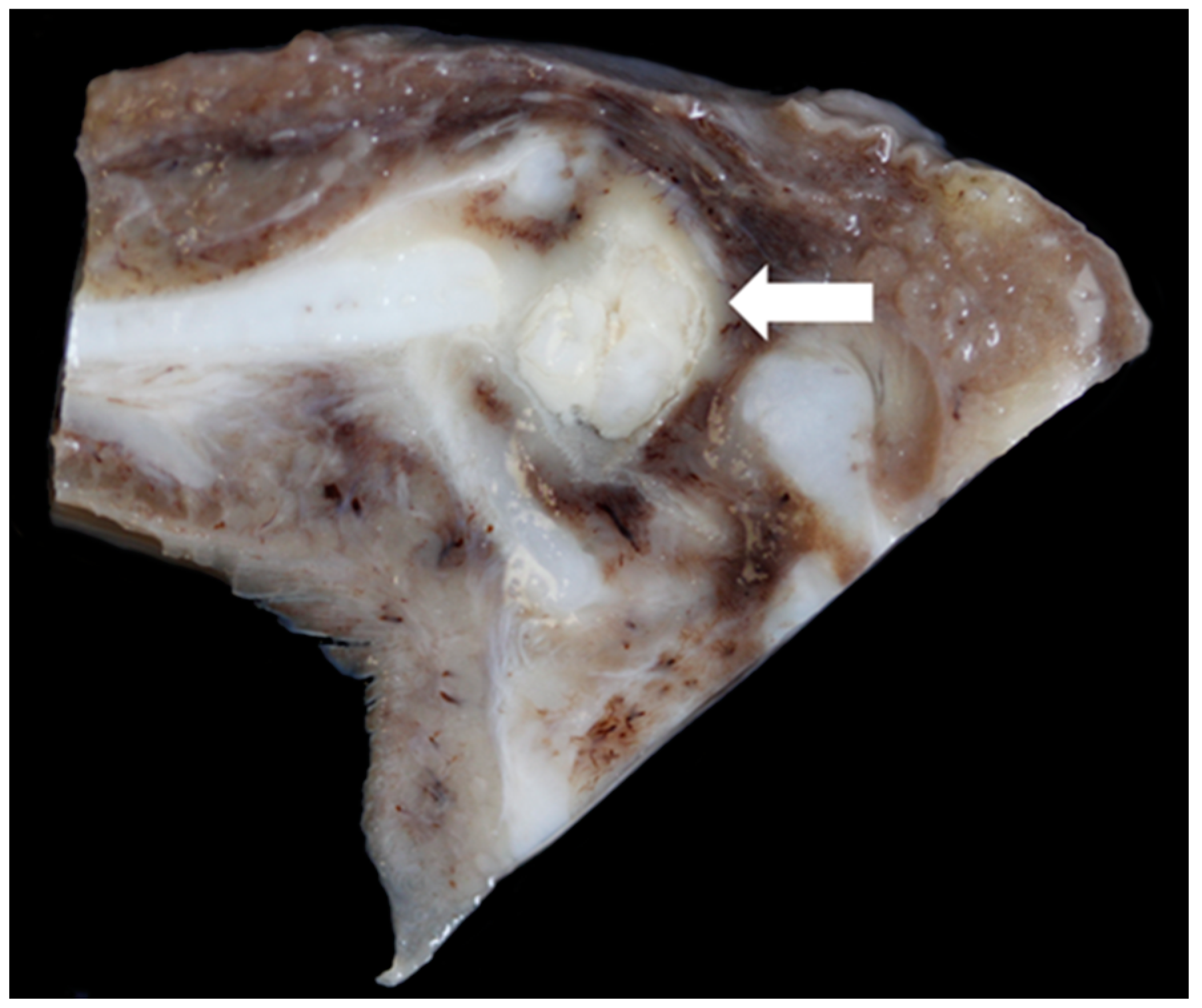
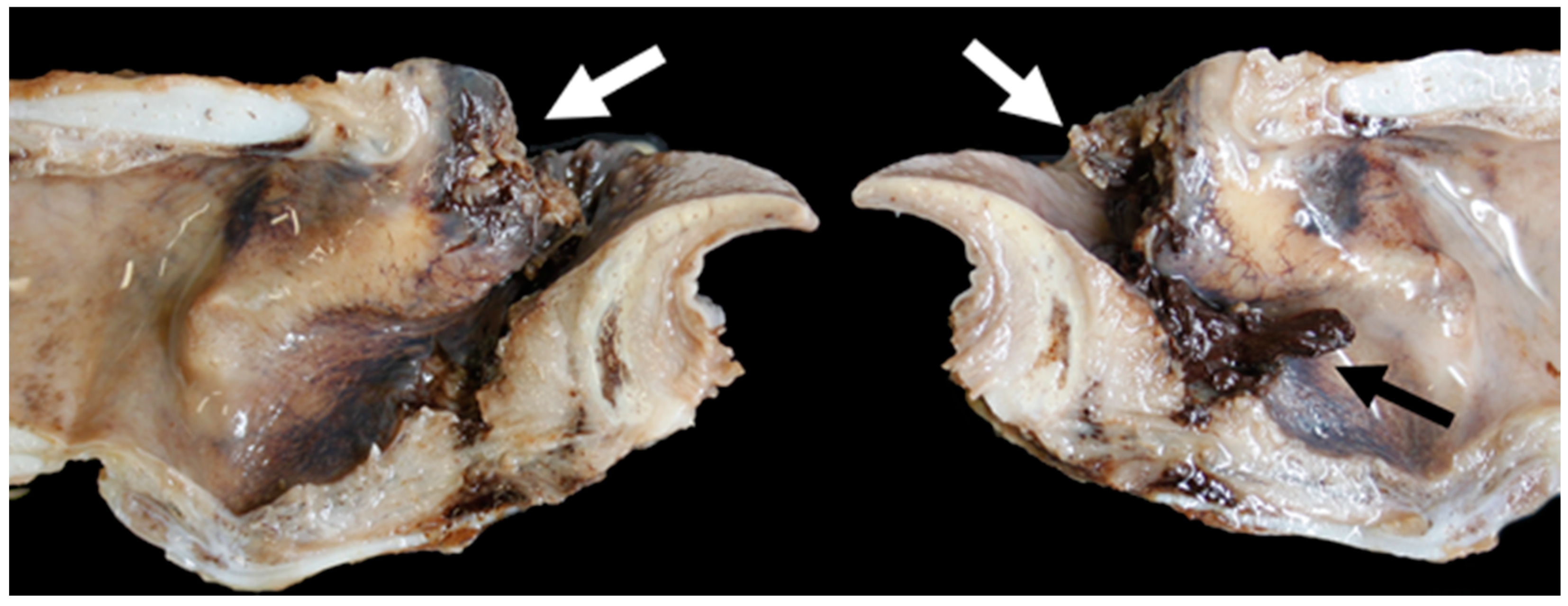
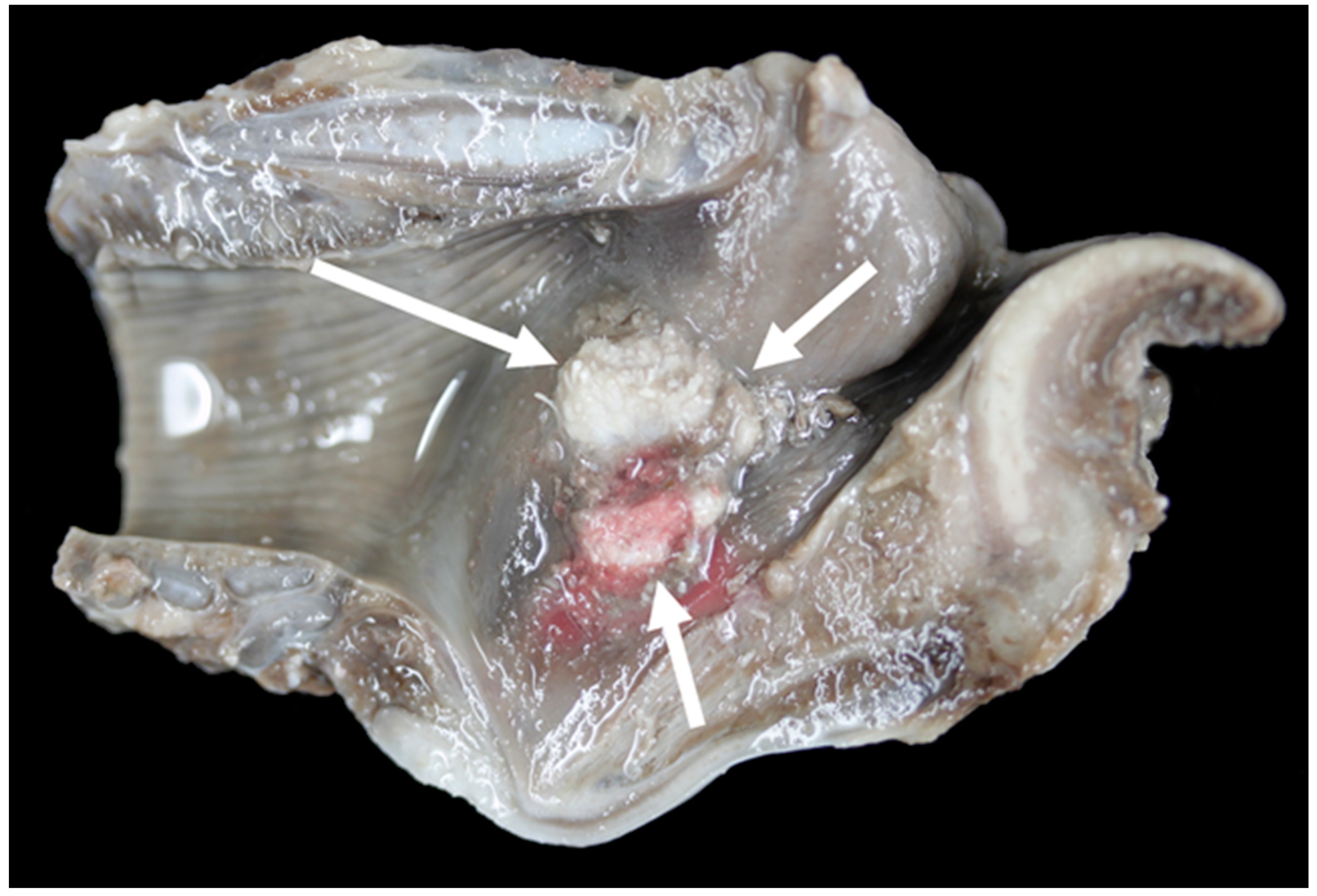
3.6. Pathology
4. Discussion
4.1. Imaging
4.2. Study Limitations
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Faull, W.B.; Scholes, S.F. Laryngeal chondritis in texel sheep. Vet. Rec. 1987, 121, 155. [Google Scholar] [CrossRef] [PubMed]
- Lane, J.G.; Brown, P.J.; Lancaster, M.L.; Todd, J.N. Laryngeal chondritis in Texel sheep. Vet. Rec. 1987, 121, 81–84. [Google Scholar] [CrossRef] [PubMed]
- Sigurðardóttir, Ó.G.; Jörundsson, E.; Friðriksdóttir, V. Laryngeal Chondritis in Sheep in Iceland. J. Comp. Pathol. 2016, 155, 310–313. [Google Scholar] [CrossRef] [PubMed]
- Edmunds, J.; Roden, J.; Finch, J.; McEwan, N. Factors affecting the development of laryngeal chondritis in sheep. Large Anim. Rev. 2017, 23, 219–222. [Google Scholar]
- Carson, A. GB Emerging Threats Quarterly Report, Small Ruminant Diseases; Animal and Plant Health Agency: London, UK, 2017.
- Cameron, H.; Britton, J. Chronic ovine laryngitis. Cornell Vet. 1943, 33, 265–268. [Google Scholar]
- Salisbury, R. Chronic ovine laryngitis. N. Z. Vet. J. 1956, 4, 144–146. [Google Scholar] [CrossRef]
- Packer, R.M.A.; Hendricks, A.; Tivers, M.S.; Burn, C.C. Impact of Facial Conformation on Canine Health: Brachycephalic Obstructive Airway Syndrome. PLoS ONE 2015, 10, 137496. [Google Scholar] [CrossRef] [PubMed]
- Texel Breed Society Research and Development. Available online: https://texel.uk/research-and-development/ (accessed on 8 January 2019).
- Cocquyt, G.; Driessen, B.; Simoens, P. Variability in the eruption of the permanent incisor teeth in sheep. Vet. Rec. 2005, 157, 619–623. [Google Scholar] [CrossRef] [PubMed]
- Olatunji-akioye, O.K.; Adeyemoa, O. Liveweight and Chest Girth Correlation in Commercial Sheep and Goat Herds in Southwestern Nigeria. Int. J. Morphol. 2009, 27, 49–52. [Google Scholar] [CrossRef]
- Zrunek, M.; Happak, W.; Hermann, M.; Streinzer, W. Comparative Anatomy of Human and Sheep Laryngeal Skeleton. Acta Otolaryngol. 1988, 105, 155–162. [Google Scholar] [CrossRef] [PubMed]
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2016. [Google Scholar]
- Bluefaced Leicester Sheep Breeder’s Association; Breed Description. Available online: http://www.blueleicester.co.uk/breed/descriptionnew.html (accessed on 8 January 2019).
- Harvey, C. Inherited and congenital airway conditions. J. Small Anim. Pract. 1989, 30, 184–187. [Google Scholar] [CrossRef]
- Liu, N.-C.; Troconis, E.L.; Kalmar, L.; Price, D.J.; Wright, H.E.; Adams, V.J.; Sargan, D.R.; Ladlow, J.F. Conformational risk factors of brachycephalic obstructive airway syndrome (BOAS) in pugs, French bulldogs, and bulldogs. PLoS ONE 2017, 12, e0181928. [Google Scholar] [CrossRef] [PubMed]
- Coyne, B.; Fingland, R. Hypoplasia of the trachea in dogs: 103 cases (1974–1990). J. Am. Vet. Med. Assoc. 1992, 201, 768–772. [Google Scholar] [PubMed]
- Stadler, K.; Hartman, S.; Matheson, J.; O’Brien, R. Computed Tomographic Imaging of Dogs with Primary Laryngeal or Tracheal Airway Obstruction. Vet. Radiol. Ultrasound 2011, 52, 377–384. [Google Scholar] [CrossRef] [PubMed]
- Ingman, J.; Naslund, V.; Hansson, K. Comparison between tracheal ratio methods used by three observers at three occasions in English Bulldogs. Acta Vet. Scand. 2014, 56, 79. [Google Scholar] [CrossRef] [PubMed]
- SRUC (Scotland’s Rural College) CT Scanning Service. Available online: https://www.sruc.ac.uk/info/120274/ct_scanning_service (accessed on 8 January 2019).
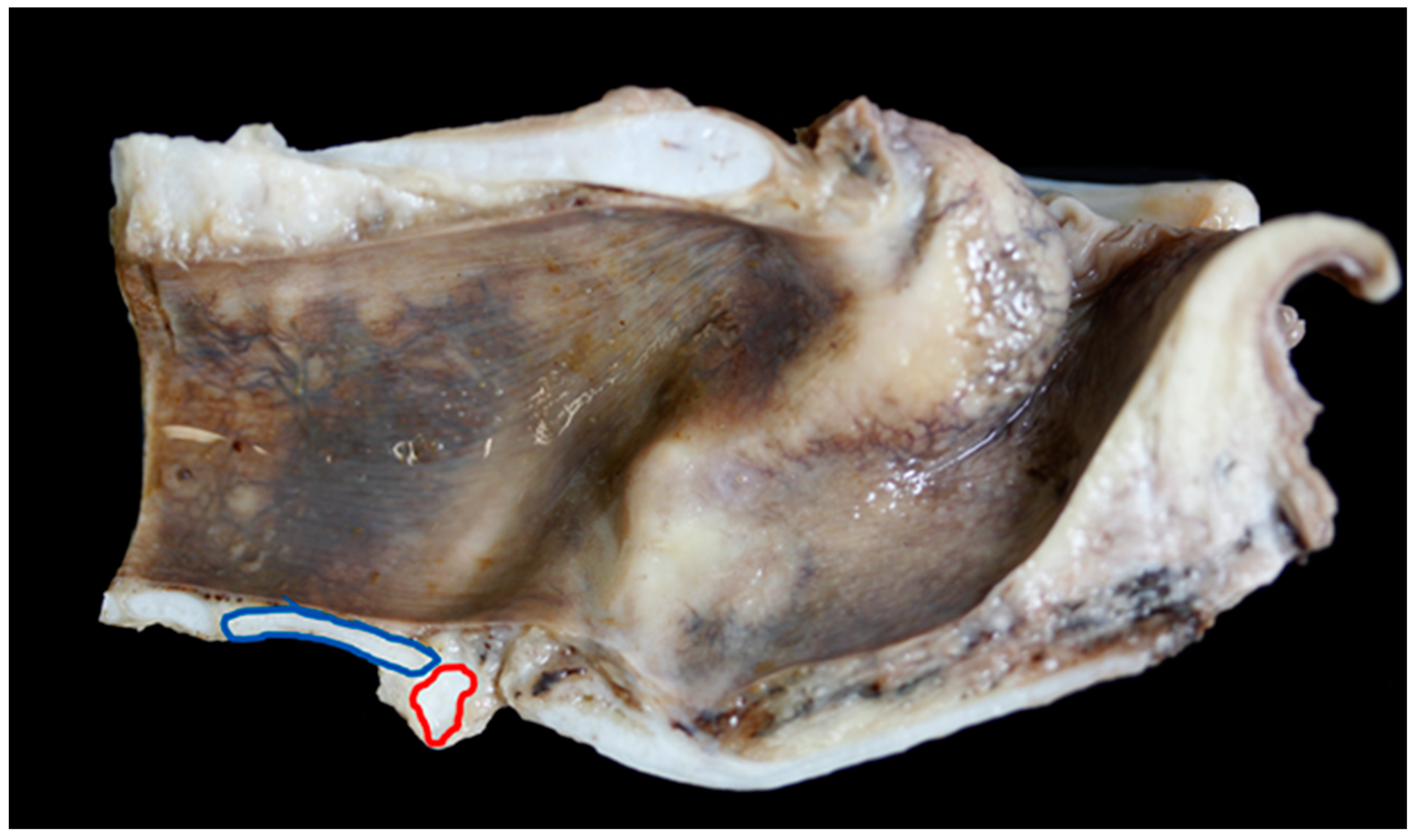
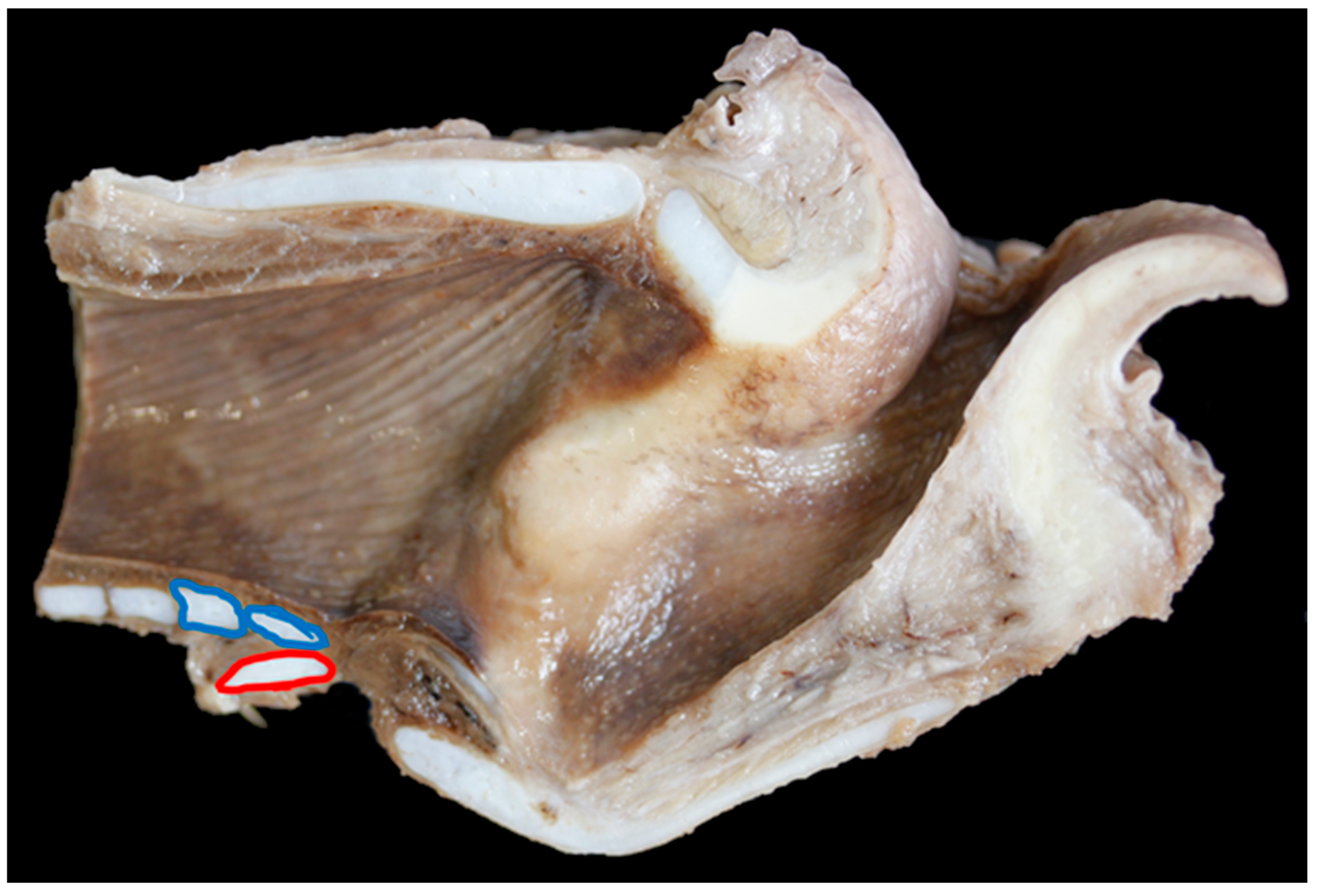
| Carcase Measurement | Points of Measurement |
|---|---|
| Head length | Caudal mandibular edge to cranial mandibular point of lower jaw |
| Neck circumference | Circumference of cranial neck, measured immediately caudal to ear base |
| Neck length | Cranial point of sternum to caudal mandible, measured along ventral neck |
| Height | Caudal ventral heel to withers |
| Girth | Circumference of girth |
| Anatomical Structure | Measurement | Texel | Blue-Faced Leicester | p Value | ||
|---|---|---|---|---|---|---|
| n | Mean (mm) | n | Mean (mm) | |||
| Head and neck | Head length | 17 | 240 | 14 | 238 | 0.7 |
| Neck Circumference | 17 | 495 | 14 | 404 | <0.001 | |
| Neck length | 14 | 402 | 14 | 486 | <0.001 | |
| Height | 17 | 742 | 14 | 843 | <0.001 | |
| Girth | 16 | 1037 | 13 | 1034 | <0.001 | |
| Larynx length | Anterior thyroid length | 23 | 39.5 | 20 | 45.1 | <0.001 |
| Dorsal thyroid length | 23 | 55.8 | 20 | 65.1 | <0.001 | |
| Length of thyroid laminae | 23 | 26.8 | 20 | 30.9 | <0.001 | |
| Main length of larynx | 23 | 47.8 | 20 | 51.4 | <0.001 | |
| Split thyroid length | 22 | 38.6 | 18 | 43.9 | <0.001 | |
| Length of cricoid cartilage | 23 | 40.7 | 19 | 44.8 | 0.02 | |
| Cricoid split length | 22 | 34.4 | 18 | 38.8 | 0.02 | |
| Anterior cricoid length | 22 | 7.8 | 20 | 8.3 | 0.4 | |
| Larynx width | Maximum thyroid breadth | 21 | 52.3 | 20 | 50.2 | 0.06 |
| Lower thyroid breadth | 22 | 38.4 | 20 | 38.9 | 0.7 | |
| Upper thyroid breadth | 21 | 48.2 | 20 | 43.1 | <0.001 | |
| Larynx height | Anteroposterior dimension | 23 | 50.6 | 20 | 58 | 0.004 |
| Cricothyroid distance | 20 | 43.3 | 18 | 51.5 | <0.001 | |
| Larynx angle | Angle of thyroid cartilage | 22 | 58.1° | 20 | 50.4° | <0.001 |
| Airway | Tracheal width | 23 | 15.6 | 20 | 19 | <0.001 |
| Tracheal height | 23 | 19.2 | 19 | 23.9 | <0.001 | |
| Tracheal area | 23 | 239 mm2 | 19 | 372 mm2 | <0.01 | |
| Arytenoid cartilage | Corniculate process distance | 21 | 15.7 | 19 | 16.8 | 0.23 |
| Depth of corniculate process | 21 | 18.6 | 19 | 19.3 | 0.5 | |
| Height of arytenoid cartilage | 18 | 39.7 | 18 | 42.2 | 0.02 | |
| Epiglottis | Length of epiglottis | 22 | 32.1 | 19 | 35.8 | 0.04 |
| Larynx | Gross Lesion | Vocal Fold Contact |
|---|---|---|
| TX1 | Moderate unilateral (right side) vocal fold mucosal hyperplasia | Yes |
| TX4 | Moderate haemorrhage in right dorsal cricoarytenoid muscle, mild bilateral vocal fold mucosal erosion and hyperplasia | Yes |
| TX6 | Mild bilateral vocal fold mucosal hyperplasia | No |
| TX7 | Mild unilateral vocal fold mucosal hyperplasia | No |
| TX8 | Mild vocal fold mucosal erosion | No |
| TX9 | Unilateral (left side) mild vocal fold mucosal hyperplasia, right-sided mild vocal fold mucosa flattening, bilateral severe suppurative cricoarytenoid osteoarthritis | Yes |
| TX11 | Bilateral, moderate vocal fold mucosal hyperplasia | No |
| TX12 | Bilateral, mild, vocal fold mucosal ulceration | Yes |
| TX13 | Massive bilateral necrosis of the corniculate process of the arytenoid cartilages with haemorrhage | No |
| TX16 | Bilateral moderate vocal fold mucosal hyperplasia | Yes |
| TX17 | Bilateral moderate vocal fold mucosal hyperplasia | Yes |
| TX21 | Severe bilateral (left sided) vocal fold mucosal hyperplasia | Yes |
| BL5 | Mild unilateral vocal fold mucosal hyperplasia | No |
| BL20 | Mild unilateral vocal fold mucosal hyperplasia | No |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Waine, K.; Strugnell, B.; Remnant, J.; Lovatt, F.; Green, M.; Rideout, H.; Genever, E.; Baiker, K. Anatomy and Pathology of the Texel Sheep Larynx. Vet. Sci. 2019, 6, 21. https://doi.org/10.3390/vetsci6010021
Waine K, Strugnell B, Remnant J, Lovatt F, Green M, Rideout H, Genever E, Baiker K. Anatomy and Pathology of the Texel Sheep Larynx. Veterinary Sciences. 2019; 6(1):21. https://doi.org/10.3390/vetsci6010021
Chicago/Turabian StyleWaine, Katie, Ben Strugnell, John Remnant, Fiona Lovatt, Martin Green, Hannah Rideout, Elizabeth Genever, and Kerstin Baiker. 2019. "Anatomy and Pathology of the Texel Sheep Larynx" Veterinary Sciences 6, no. 1: 21. https://doi.org/10.3390/vetsci6010021
APA StyleWaine, K., Strugnell, B., Remnant, J., Lovatt, F., Green, M., Rideout, H., Genever, E., & Baiker, K. (2019). Anatomy and Pathology of the Texel Sheep Larynx. Veterinary Sciences, 6(1), 21. https://doi.org/10.3390/vetsci6010021





