The Antibacterial Effect of Humulus lupulus (Hops) against Mycobacterium bovis BCG: A Promising Alternative in the Fight against Bovine Tuberculosis?
Abstract
:1. Introduction
2. Materials and Methods
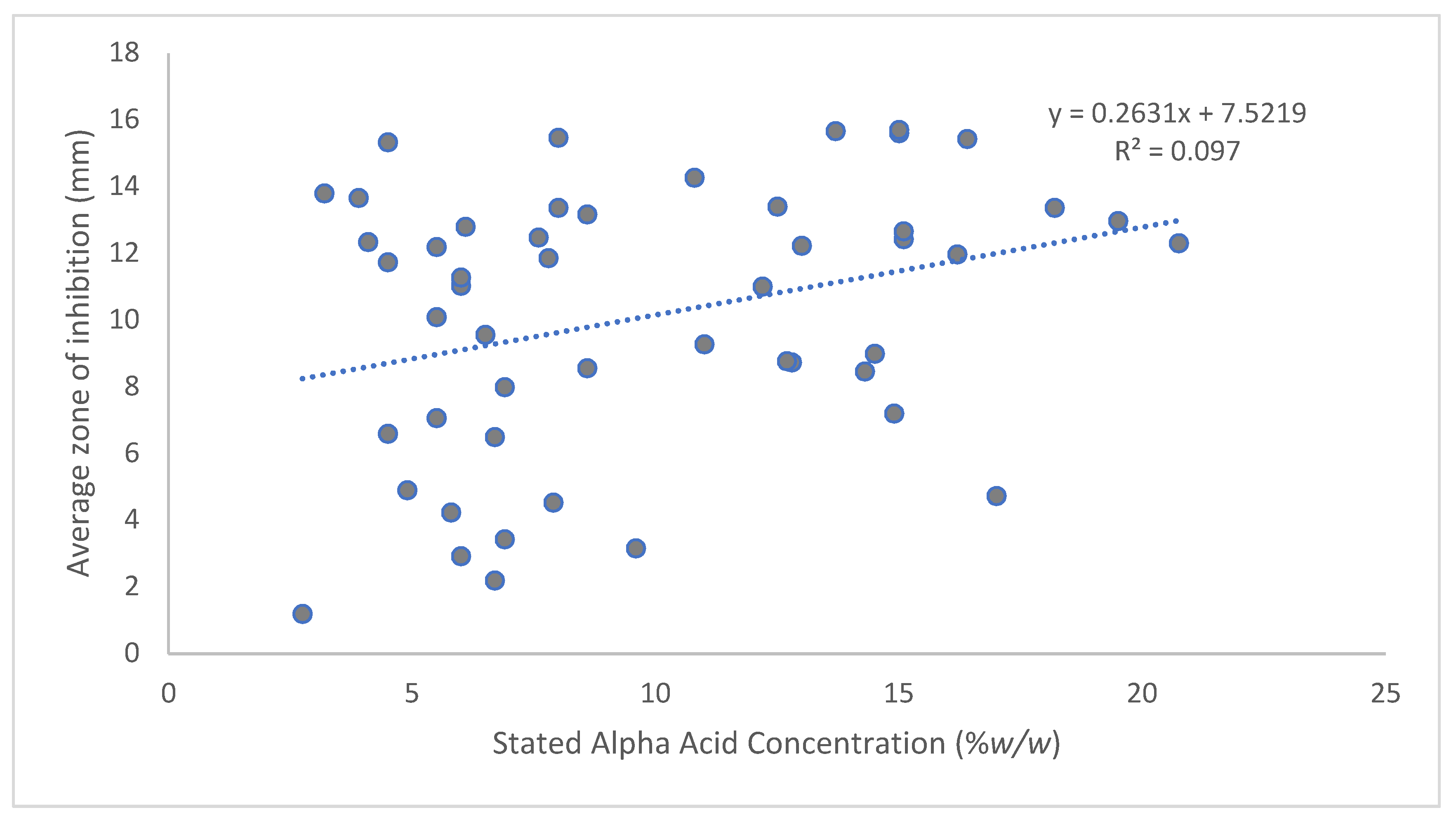
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Conflicts of Interest
References
- Hieronymus, S. For the Love of Hops: The Practical Guide to Aroma, Bitterness, and the Culture of Hops; Brewing elements series; Brewers Publisher, bp: Boulder, CO, USA, 2012; ISBN 978-1-938469-01-5. [Google Scholar]
- Kramer, B.; Thielmann, J.; Hickisch, A.; Muranyi, P.; Wunderlich, J.; Hauser, C. Antimicrobial Activity of Hop Extracts against Foodborne Pathogens for Meat Applications. J. Appl. Microbiol. 2015, 118, 648–657. [Google Scholar] [CrossRef] [PubMed]
- Filmer, R. Hops and Hop Picking; History in camera; Shire Publications Ltd.: Oxford, UK, 2011; ISBN 978-0-85263-617-6. [Google Scholar]
- Rettberg, N.; Biendl, M.; Garbe, L.-A. Hop Aroma and Hoppy Beer Flavor: Chemical Backgrounds and Analytical Tools—A Review. J. Am. Soc. Brew. Chem. 2018, 76, 1–20. [Google Scholar] [CrossRef]
- Karabín, M.; Hanko, V.; Nešpor, J.; Jelínek, L.; Dostálek, P. Hop Tannin Extract: A Promising Tool for Acceleration of Lautering. J. Inst. Brew. 2018, 124, 374–380. [Google Scholar] [CrossRef] [Green Version]
- Blaxland, J.A.; Watkins, A.J.; Baillie, L.W.J. The Ability of Hop Extracts to Reduce the Methane Production of Methanobrevibacter Ruminantium. Archaea 2021, 2021, 5510063. [Google Scholar] [CrossRef] [PubMed]
- Rattan, A.; Kalia, A.; Ahmad, N. Multidrug-Resistant Mycobacterium Tuberculosis: Molecular Perspectives. Emerg. Infect. Dis. 1998, 4, 195–209. [Google Scholar] [CrossRef] [Green Version]
- Müller, B.; Borrell, S.; Rose, G.; Gagneux, S. The Heterogeneous Evolution of Multidrug-Resistant Mycobacterium Tuberculosis. Trends Genet. 2013, 29, 160–169. [Google Scholar] [CrossRef] [Green Version]
- Perdigão, J.; Silva, C.; Maltez, F.; Machado, D.; Miranda, A.; Couto, I.; Rabna, P.; Florez de Sessions, P.; Phelan, J.; Pain, A.; et al. Emergence of Multidrug-Resistant Mycobacterium Tuberculosis of the Beijing Lineage in Portugal and Guinea-Bissau: A Snapshot of Moving Clones by Whole-Genome Sequencing. Emerg. Microbes Infect. 2020, 9, 1342–1353. [Google Scholar] [CrossRef]
- Rosa, P.S.; D’Espindula, H.R.S.; Melo, A.C.L.; Fontes, A.N.B.; Finardi, A.J.; Belone, A.F.F.; Sartori, B.G.C.; Pires, C.A.A.; Soares, C.T.; Marques, F.B.; et al. Emergence and Transmission of Drug-/Multidrug-Resistant Mycobacterium Leprae in a Former Leprosy Colony in the Brazilian Amazon. Clin. Infect. Dis. 2020, 70, 2054–2061. [Google Scholar] [CrossRef]
- Stavri, M.; Schneider, R.; O’Donnell, G.; Lechner, D.; Bucar, F.; Gibbons, S. The Antimycobacterial Components of Hops (Humulus Lupulus) and Their Dereplication. Phytother. Res. PTR 2004, 18, 774–776. [Google Scholar] [CrossRef]
- Serkani, J.E.; Isfahani, B.N.; Safaei, H.G.; Kermanshahi, R.K.; Asghari, G. Evaluation of the Effect of Humulus Lupulus Alcoholic Extract on Rifampin-Sensitive and Resistant Isolates of Mycobacterium Tuberculosis. Res. Pharm. Sci. 2012, 7, 235–242. [Google Scholar]
- Blaxland, J. The Antibacterial Activity of Humulus Lupulus against Mycobacteria. Ph.D. Thesis, Cardiff University, Cardiff, UK, 2015. [Google Scholar]
- The Tuberculosis (Wales) (Amendment) Order 2017. Available online: https://www.legislation.gov.uk/wsi/2017/711/contents/made (accessed on 20 July 2022).
- The Tuberculosis in Animals (England) Order 2021. Available online: https://www.legislation.gov.uk/uksi/2021/1001/article/1/made (accessed on 20 July 2022).
- Incidence of Tuberculosis (TB) in Cattle in Great Britain: March 2021. Available online: https://gov.wales/incidence-tuberculosis-tb-cattle-great-britain-march-2021 (accessed on 1 February 2022).
- EU 2019/6; Veterinary Medicinal Products and Repealing Directive 2001/82/EC (Text with EEA Relevance). European Parliament and of the Council: Brussels, Belgium, 2018; Volume 004.
- Altaf, M.; Miller, C.H.; Bellows, D.S.; O’Toole, R. Evaluation of the Mycobacterium Smegmatis and BCG Models for the Discovery of Mycobacterium Tuberculosis Inhibitors. Tuberc. Edinb. Scotl. 2010, 90, 333–337. [Google Scholar] [CrossRef] [PubMed]
- Fitzgerald, S.D.; Schooley, A.M.; Berry, D.E.; Kaneene, J.B. Antimicrobial Susceptibility Testing of Mycobacterium Bovis Isolates from Michigan White-Tailed Deer during the 2009 Hunting Season. Vet. Med. Int. 2010, 2011, 903683. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ritz, N.; Tebruegge, M.; Connell, T.G.; Sievers, A.; Robins-Browne, R.; Curtis, N. Susceptibility of Mycobacterium Bovis BCG Vaccine Strains to Antituberculous Antibiotics. Antimicrob. Agents Chemother. 2009, 53, 316–318. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gregory, E.R.; Bakhaider, R.F.; Gomez, G.F.; Huang, R.; Moser, E.A.S.; Gregory, R.L. Evaluating Hop Extract Concentrations Found in Commercial Beer to Inhibit Streptococcus Mutans Biofilm Formation. J. Appl. Microbiol. 2022. [Google Scholar] [CrossRef] [PubMed]
- Bland, J.S.; Minich, D.; Lerman, R.; Darland, G.; Lamb, J.; Tripp, M.; Grayson, N. Isohumulones from Hops (Humulus lupulus) and Their Potential Role in Medical Nutrition Therapy. PharmaNutrition 2015, 3, 46–52. [Google Scholar] [CrossRef] [Green Version]
- Bartmańska, A.; Wałecka-Zacharska, E.; Tronina, T.; Popłoński, J.; Sordon, S.; Brzezowska, E.; Bania, J.; Huszcza, E. Antimicrobial Properties of Spent Hops Extracts, Flavonoids Isolated Therefrom, and Their Derivatives. Mol. J. Synth. Chem. Nat. Prod. Chem. 2018, 23, 2059. [Google Scholar] [CrossRef] [Green Version]
- Bogdanova, K.; Röderova, M.; Kolar, M.; Langova, K.; Dusek, M.; Jost, P.; Kubelkova, K.; Bostik, P.; Olsovska, J. Antibiofilm Activity of Bioactive Hop Compounds Humulone, Lupulone and Xanthohumol toward Susceptible and Resistant Staphylococci. Res. Microbiol. 2018, 169, 127–134. [Google Scholar] [CrossRef]
- Lou, H.; Zhang, F.; Lu, L.; Ding, Y.; Hao, X. Xanthohumol from Humulus lupulus L. Potentiates the Killing of Mycobacterium Tuberculosis and Mitigates Liver Toxicity by the Combination of Isoniazid in Mouse Tuberculosis Models. RSC Adv. 2020, 10, 13223–13231. [Google Scholar] [CrossRef] [Green Version]
- Bocquet, L.; Rivière, C.; Dermont, C.; Samaillie, J.; Hilbert, J.-L.; Halama, P.; Siah, A.; Sahpaz, S. Antifungal Activity of Hop Extracts and Compounds against the Wheat Pathogen Zymoseptoria Tritici. Ind. Crops Prod. 2018, 122, 290–297. [Google Scholar] [CrossRef]
- Buckwold, V.E.; Wilson, R.J.H.; Nalca, A.; Beer, B.B.; Voss, T.G.; Turpin, J.A.; Buckheit, R.W.; Wei, J.; Wenzel-Mathers, M.; Walton, E.M.; et al. Antiviral Activity of Hop Constituents against a Series of DNA and RNA Viruses. Antivir. Res. 2004, 61, 57–62. [Google Scholar] [CrossRef]
- Wang, Q.; Ding, Z.-H.; Liu, J.-K.; Zheng, Y.-T. Xanthohumol, a Novel Anti-HIV-1 Agent Purified from Hops Humulus Lupulus. Antivir. Res. 2004, 64, 189–194. [Google Scholar] [CrossRef]
- Di Lodovico, S.; Menghini, L.; Ferrante, C.; Recchia, E.; Castro-Amorim, J.; Gameiro, P.; Cellini, L.; Bessa, L.J. Hop Extract: An Efficacious Antimicrobial and Anti-Biofilm Agent Against Multidrug-Resistant Staphylococci Strains and Cutibacterium Acnes. Front. Microbiol. 2020, 11, 1852. [Google Scholar] [CrossRef] [PubMed]
- Rozalski, M.; Micota, B.; Sadowska, B.; Stochmal, A.; Jedrejek, D.; Wieckowska-Szakiel, M.; Rozalska, B. Antiadherent and Antibiofilm Activity of Humulus lupulus L. Derived Products: New Pharmacological Properties. BioMed Res. Int. 2013, 2013, e101089. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Narvaez, N.; Wang, Y.; Xu, Z.; Alexander, T.; Garden, S.; McAllister, T. Effects of Hop Varieties on Ruminal Fermentation and Bacterial Community in an Artificial Rumen (Rusitec). J. Sci. Food Agric. 2013, 93, 45–52. [Google Scholar] [CrossRef]
- Team, B.T.S. Badger Trust Welcomes End of Selective Badger Cull in Wales and Rethink of BTB Policy. Available online: https://www.badgertrust.org.uk/post/badger-trust-welcomes-end-of-selective-badger-cull-in-wales-and-rethink-of-btb-policy (accessed on 20 July 2022).
- Holder, I.A.; Boyce, S.T. Agar Well Diffusion Assay Testing of Bacterial Susceptibility to Various Antimicrobials in Concentrations Non-Toxic for Human Cells in Culture. Burns 1994, 20, 426–429. [Google Scholar] [CrossRef]
- Make Hops Pellets for Brewery/Hops Pellet Machine. Available online: https://www.pelletmachines.net/biopellet-plant/hops-pellet-production-line.html (accessed on 1 February 2022).
- How Are Hop Pellets Made? Available online: https://www.canuckhomebrewsupply.com/blogs/all/how-are-hop-pellets-made (accessed on 1 February 2022).
- Gerhäuser, C. Broad Spectrum Antiinfective Potential of Xanthohumol from Hop (Humulus lupulus L.) in Comparison with Activities of Other Hop Constituents and Xanthohumol Metabolites. Mol. Nutr. Food Res. 2005, 49, 827–831. [Google Scholar] [CrossRef] [PubMed]
- Simpson, W.J.; Smith, A.R.W. Factors Affecting Antibacterial Activity of Hop Compounds and Their Derivatives. J. Appl. Bacteriol. 1992, 72, 327–334. [Google Scholar] [CrossRef]
- Development of a New High-Performance Liquid Chromatography Method with Diode Array and Electrospray Ionization-Mass Spectrometry Detection for the Metabolite Fingerprinting of Bioactive Compounds in Humulus lupulus L.—ScienceDirect. Available online: https://www.sciencedirect.com/science/article/pii/S0021967314007195 (accessed on 20 July 2022).
- Knez Hrnčič, M.; Španinger, E.; Košir, I.J.; Knez, Ž.; Bren, U. Hop Compounds: Extraction Techniques, Chemical Analyses, Antioxidative, Antimicrobial, and Anticarcinogenic Effects. Nutrients 2019, 11, 257. [Google Scholar] [CrossRef] [Green Version]
- Probasco, E.G.; Perrault, J. Hop Plant Named ‘HBC 394’ USA. US PP21,289 P3, 14 September 2010. [Google Scholar]
- MacKinnon, D.; Pavlovič, V.; Čeh, B.; Naglič, B.; Pavlovič, M. The Impact of Weather Conditions on Alpha-Acid Content in Hop (Humulus lupulus L.) Cv. Aurora. Plant Soil Environ. 2020, 66, 519–525. [Google Scholar] [CrossRef]
- Park, K.-Y.; Ree, D.-W.; Min, I.-K. Influence of Days after Flowering and Cultural Environments on Changes of Alpha-Acid Content in Hops (Humulus lupulus L.). Korean J. Crop Sci. 1987, 32, 97–102. [Google Scholar]
- Chen, G.-H.; Yang, C.-Y.; Lee, S.-J.; Wu, C.-C.; Tzen, J.T.C. Catechin Content and the Degree of Its Galloylation in Oolong Tea Are Inversely Correlated with Cultivation Altitude. J. Food Drug Anal. 2014, 22, 303–309. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Vodnar, D.C. Inhibition of Listeria Monocytogenes ATCC 19115 on Ham Steak by Tea Bioactive Compounds Incorporated into Chitosan-Coated Plastic Films. Chem. Cent. J. 2012, 6, 74. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pérez, M.J.; Falqué, E.; Domínguez, H. Antimicrobial Action of Compounds from Marine Seaweed. Mar. Drugs 2016, 14, 52. [Google Scholar] [CrossRef] [Green Version]
- Kenny, O.; Brunton, N.P.; Walsh, D.; Hewage, C.M.; McLoughlin, P.; Smyth, T.J. Characterisation of Antimicrobial Extracts from Dandelion Root (Taraxacum officinale) Using LC-SPE-NMR. Phytother. Res. PTR 2015, 29, 526–532. [Google Scholar] [CrossRef] [PubMed]
- Murray, C.J.; Ikuta, K.S.; Sharara, F.; Swetschinski, L.; Aguilar, G.R.; Gray, A.; Han, C.; Bisignano, C.; Rao, P.; Wool, E.; et al. Global Burden of Bacterial Antimicrobial Resistance in 2019: A Systematic Analysis. Lancet 2022, 399, 629–655. [Google Scholar] [CrossRef]
- Natarajan, P.; Katta, S.; Andrei, I.; Babu Rao Ambati, V.; Leonida, M.; Haas, G.J. Positive Antibacterial Co-Action between Hop (Humulus lupulus) Constituents and Selected Antibiotics. Phytomedicine 2008, 15, 194–201. [Google Scholar] [CrossRef]
- Fahle, A.; Bereswill, S.; Heimesaat, M.M. Antibacterial Effects of Biologically Active Ingredients in Hop Provide Promising Options to Fight Infections by Pathogens Including Multi-Drug Resistant Bacteria. Eur. J. Microbiol. Immunol. 2022, 12, 22–30. [Google Scholar] [CrossRef]

| Hop Name/Type | Pellet or Whole Hop | Stated Alpha Acid Content (% w/w) |
|---|---|---|
| Amarillo | Whole | 6.9 |
| Amarillo | Pellet | 6.9 |
| Apollo | Whole | 19.5 |
| Aurora | Whole | 7.8 |
| Boadicea | Whole | 6 |
| Bobek | Whole | 3.9 |
| Brambling Cross | Whole | 6 |
| Cascade | Whole | 5.5 |
| Cascade | Pellet | 5.5 |
| Centennial | Whole | 8.6 |
| Centennial | Pellet | 8.6 |
| Challenger | Whole | 7.6 |
| Chinnok | Whole | 12.7 |
| Chinook | Pellet | 11 |
| Citra | Whole | 15 |
| Citra | Pellet | 16.5 |
| Colombus | Whole | 16.2 |
| Delta | Whole | 6.5 |
| First Gold | Whole | 7.9 |
| Fuggles | Whole | 4.9 |
| Fuggles | Pellet | 4.9 |
| Galaxy | Whole | 15.1 |
| Galena | Whole | 13 |
| Glacier | Whole | 6.7 |
| Goldings | Whole | 5.8 |
| Green Bullet | Pellet | 13.7 |
| Hallertauer | Whole | 3.2 |
| Liberty | Whole | 4.5 |
| Magnum | Whole | 14.5 |
| Motueka | Pellet | 6.7 |
| Nelson Sauvin | Whole | 12.8 |
| New Zealand Rakau | Whole | 10.8 |
| Northdown | Whole | 8 |
| Northern Brewer | Whole | 8 |
| NZ Pacifica | Whole | 6.1 |
| Pacific Gem | Whole | 17 |
| Pacific Jade | Whole | 15.1 |
| Perle | Whole | 9.6 |
| Progress | Whole | 5.5 |
| Savinjski Goldings | Whole | 20.75 |
| Simcoe | Whole | 12.2 |
| Sonnet | Whole | 4.1 |
| Sorachi Ace | Whole | 14.9 |
| Summer | Pellet | 6 |
| Summit | Whole | 14.3 |
| Syrian Goldings | Whole | 2.75 |
| Target | Whole | 12.5 |
| Tettnang | Whole | 4.5 |
| Warrier | Whole | 18.2 |
| Willamette | Pellet | 4.5 |
| Well Number | SDW Amount (μL) | Agar Amount (μL) | Hop Amount (μL) | % Volume of Hop Extract |
|---|---|---|---|---|
| A1 | 0.00 | 2000 | 1000 | 33.00 |
| B1 | 100 | 2000 | 900 | 30.00 |
| C1 | 200 | 2000 | 800 | 26.00 |
| D1 | 300 | 2000 | 700 | 23.00 |
| A2 | 350 | 2000 | 650 | 21.60 |
| B2 | 400 | 2000 | 600 | 20.00 |
| C2 | 450 | 2000 | 550 | 18.30 |
| D2 | 500 | 2000 | 500 | 16.00 |
| A3 | 550 | 2000 | 450 | 15.00 |
| B3 | 600 | 2000 | 400 | 13.00 |
| C3 | 650 | 2000 | 350 | 11.60 |
| D3 | 700 | 2000 | 300 | 10.00 |
| A4 | 750 | 2000 | 250 | 8.00 |
| B4 | 800 | 2000 | 200 | 6.60 |
| C4 | 820 | 2000 | 180 | 6.00 |
| D4 | 840 | 2000 | 160 | 5.30 |
| A5 | 850 | 2000 | 150 | 5.00 |
| B5 | 860 | 2000 | 140 | 4.60 |
| C5 | 880 | 2000 | 120 | 4.00 |
| D5 | 900 | 2000 | 100 | 3.30 |
| A6 | 910 | 2000 | 90 | 3.00 |
| B6 | 920 | 2000 | 80 | 2.66 |
| C6 | 945 | 2000 | 55 | 0.83 |
| D6 | 0.00 | 3000 | 0 | 0.00 |
| Hop Variant | Average Zone of Inhibition (mm) | MIC (%v/v) | MBC (%v/v) |
|---|---|---|---|
| Pacific Gem | 12.8 | >33% | >33% |
| Apollo | 12.9 | 26% | >33% |
| Centennial (Whole) | 13.1 | 26% | >33% |
| Northern Brewer | 13.3 | 26% | 26% |
| Willamette (Whole) | 13.3 | 26% | 26% |
| Tettnang | 13.4 | 26% | 26% |
| Bobek | 13.6 | 26% | 26% |
| Liberty | 13.8 | 26% | 26% |
| Northdown | 14.2 | 23% | 26% |
| Magnum | 15.3 | 23% | 26% |
| Columbus | 15.4 | 23% | 26% |
| NZ Pacifica | 15.4 | 16% | 26% |
| Citra (Whole) | 15.6 | 16% | 26% |
| Hallertauer | 15.6 | 16% | 26% |
| Citra (Pellet) | 15.7 | 16% | 16% |
| Type of Analysis | Reference | Microorganisms Analyzed | Hop Varieties /Components Employed |
|---|---|---|---|
| Antimicrobial (Gram-positive and Gram-negative) | [23] [2] [24] | S. aureus S. epidermidis, L. monocytogenes, S. typhimurium L. monocytogenes, S. aureus, salmonella enterica, E. coli. Vancomycin and methicillin resistant Gram-positive bacteria | Hop extracts and spent hop extracts Hop extracts—alpha/beta acids, xanthohumol Purified hop components α-bitter acids (humulones), β-bitter acids (lupulones) and xanthohumol, and a commercial CO2 hop extract of bitter acids |
| Antimycobacterial | [13] [11] [12,25] | Mycobacterium abscessus Mycobacterium fortuitum Mycobacterium tuberculosis | Whole Hops and Pellets (Inflorescences) Hexane Extracts Alcohol (polar extracts) and Xanthohumol extracts |
| Antifungal | [26] [23] | Zymoseptoria tritici Botrytis cinereal, Fusarium oxysporum, F. culmorum,andF. semitectum M. hiemalis and P. purpurogenum | Hydro-alcoholic crude extracts from leaves, stems, rhizomes, and female cones Solvent extracts; hop flavonoids |
| Antiviral | [27] [28] | BVDV, HIV, FLU-A, FLU-B, Rhino, RSV, YFV, CMV, HBV and HSV-1, HSV-2. HIV-1 | CO2 extrctas, isomerized kettle extract, xanthohumol enriched extract, iso-α-Acids, β-Acids, Hop oil, xanthohumol and iso-xanthohumol Xanthohumol |
| Antibiofilm | [29] [21] [30] | S. aureus, S. epidermidis and C. acnes S. mutans S. aureus | Purified Hop Extracts Purified Hop Extracts Purified hop extracts containing 51% xanthohumol |
| Rumen Fermentation and Methane Reduction | [31] [6] | Fibrobacter succinogenes, Ruminococcus albus Methanobrevibacter ruminantium | Artificial Rumen System Aqueous (polar hop extracts); in vitro |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Blaxland, J.; Thomas, R.; Baillie, L. The Antibacterial Effect of Humulus lupulus (Hops) against Mycobacterium bovis BCG: A Promising Alternative in the Fight against Bovine Tuberculosis? Beverages 2022, 8, 43. https://doi.org/10.3390/beverages8030043
Blaxland J, Thomas R, Baillie L. The Antibacterial Effect of Humulus lupulus (Hops) against Mycobacterium bovis BCG: A Promising Alternative in the Fight against Bovine Tuberculosis? Beverages. 2022; 8(3):43. https://doi.org/10.3390/beverages8030043
Chicago/Turabian StyleBlaxland, James, Richard Thomas, and Leslie Baillie. 2022. "The Antibacterial Effect of Humulus lupulus (Hops) against Mycobacterium bovis BCG: A Promising Alternative in the Fight against Bovine Tuberculosis?" Beverages 8, no. 3: 43. https://doi.org/10.3390/beverages8030043
APA StyleBlaxland, J., Thomas, R., & Baillie, L. (2022). The Antibacterial Effect of Humulus lupulus (Hops) against Mycobacterium bovis BCG: A Promising Alternative in the Fight against Bovine Tuberculosis? Beverages, 8(3), 43. https://doi.org/10.3390/beverages8030043






