The Physiological Effects of Amino Acids Arginine and Citrulline: Is There a Basis for Development of a Beverage to Promote Endurance Performance? A Narrative Review of Orally Administered Supplements
Abstract
1. Introduction
2. Methods
3. Background
3.1. Prevalence of Supplementation in Athletic and Recreational Populations
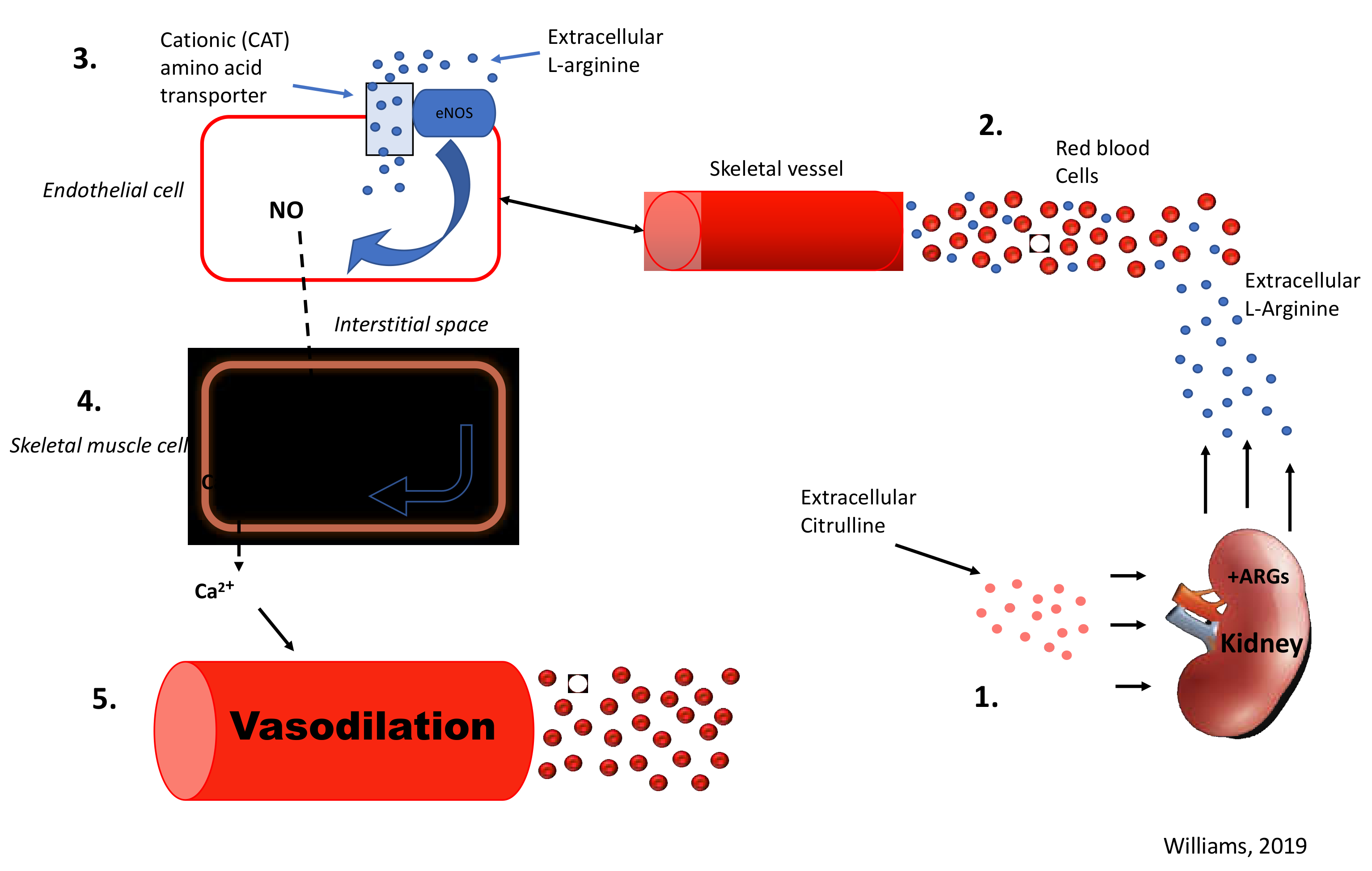
3.2. L-Arginine
3.3. L-Citrulline
3.4. The Bioavailability of L-Arg and L-Cit
4. Discussion of Results
4.1. L-Arg and L-Cit on Vasodilation and NO Biomarkers
4.2. L-Arg and L-Cit on Physiological Response and Exercise Performance Outcomes
4.3. Potential Synergistic Effects of L-Arg and L-Cit
5. Summary of Key Findings and Future Directions
6. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Applegate, E.A.; Grivetti, L.E. Search for the Competitive Edge: A History of Dietary Fads and Supplements. J. Nutr. 1997, 127, 869S–873S. [Google Scholar] [CrossRef] [PubMed]
- Council, N.R. Dietary Supplements: A Framework for Evaluating Safety; National Academies Press: Cambridge, MA, USA, 2004. [Google Scholar]
- Fairweather-Tait, S.S. Encyclopedia of Food Sciences and Nutrition, 2nd ed; Academic Press: Cambridge, MA, USA, 2003. [Google Scholar]
- Williams, M. Dietary Supplements and Sports Performance: Amino Acids. J. Int. Soc. Sports Nutr. 2005, 2, 63. [Google Scholar] [CrossRef] [PubMed]
- Hyde, R.; Taylor, P.M.; Hundal, H.S. Amino acid transporters: Roles in amino acid sensing and signalling in animal cells. Biochem. J. 2003, 373, 1. [Google Scholar] [CrossRef]
- Green, C.L.; Lamming, D.W. Regulation of metabolic health by essential dietary amino acids. Mech. Ageing Dev. 2019, 177, 186–200. [Google Scholar] [CrossRef]
- Gibala, M. Dietary protein, amino acid supplements, and recovery from exercise. Sports Sci. Exch. 2002, 15, 63–67. [Google Scholar]
- Silva, E.P.; Borges, L.S.; Mendes-da-Silva, C.; Hirabara, S.M.; Lambertucci, R.H. l-Arginine supplementation improves rats’ antioxidant system and exercise performance. Free Radic. Res. 2017, 51, 281–293. [Google Scholar] [CrossRef]
- Mor, A.; Atan, T.; Agaoglu, S.A.; Ayyildiz, M. Effect of arginine supplementation on footballers’ anaerobic performance and recovery. Prog. Nutr. 2018, 20, 104–112. [Google Scholar] [CrossRef]
- Chen, I.F.; Wu, H.-J.; Chen, C.-Y.; Chou, K.-M.; Chang, C.-K. Branched-chain amino acids, arginine, citrulline alleviate central fatigue after 3 simulated matches in taekwondo athletes: A randomized controlled trial. J. Int. Soc. Sports Nutr. 2016, 13, 28. [Google Scholar] [CrossRef]
- Maiorana, A.; O’Driscoll, G.; Taylor, R.; Green, D. Exercise and the Nitric Oxide Vasodilator System. Sports Med. 2003, 33, 1013–1035. [Google Scholar] [CrossRef] [PubMed]
- Khalaf, D.; Krüger, M.; Wehland, M.; Infanger, M.; Grimm, D. The Effects of Oral l-Arginine and l-Citrulline Supplementation on Blood Pressure. Nutrients 2019, 11, 1679. [Google Scholar] [CrossRef] [PubMed]
- Morita, M.; Hayashi, T.; Ochiai, M.; Maeda, M.; Yamaguchi, T.; Ina, K.; Kuzuya, M. Oral supplementation with a combination of l-citrulline and l-arginine rapidly increases plasma l-arginine concentration and enhances NO bioavailability. Biochem. Biophys. Res. Commun. 2014, 454, 53–57. [Google Scholar] [CrossRef] [PubMed]
- Wu, G.; Morris, S.M., Jr. Arginine metabolism: Nitric oxide and beyond. Biochem. J. 1998, 336, 1–17. [Google Scholar] [CrossRef] [PubMed]
- Rajapakse, N.W.; Mattson, D.L. Role of L-Arginine in nitric oxide production in health and hypertension. Clin. Exp. Pharmacol. Physiol. 2009, 36, 249–255. [Google Scholar] [CrossRef] [PubMed]
- Chin-Dusting, J.P.F.; Willems, L.; Kaye, D.M. l-Arginine transporters in cardiovascular disease: A novel therapeutic target. Pharmacol. Ther. 2007, 116, 428–436. [Google Scholar] [CrossRef] [PubMed]
- Szefel, J.; Danielak, A.; Kruszewski, W.J. Metabolic pathways of L-arginine and therapeutic consequences in tumors. Adv. Med. Sci. 2019, 64, 104–110. [Google Scholar] [CrossRef]
- Bahri, S.; Zerrouk, N.; Aussel, C.; Moinard, C.; Crenn, P.; Curis, E.; Chaumeil, J.-C.; Cynober, L.; Sfar, S. Citrulline: From metabolism to therapeutic use. Nutrition 2013, 29, 479–484. [Google Scholar] [CrossRef]
- Watford, M. The urea cycle: Teaching intermediary metabolism in a physiological setting. Biochem. Mol. Biol. Educ. 2003, 31, 289–297. [Google Scholar] [CrossRef]
- Song, W.; Sun, X.; Chen, X.; Liu, D.; Liu, L. Enzymatic production of l-citrulline by hydrolysis of the guanidinium group of l-arginine with recombinant arginine deiminase. J. Biotechnol. 2015, 208, 37–43. [Google Scholar] [CrossRef]
- Schwedhelm, E.; Maas, R.; Freese, R.; Jung, D.; Lukacs, Z.; Jambrecina, A.; Spickler, W.; Schulze, F.; Böger, R.H. Pharmacokinetic and pharmacodynamic properties of oral L-citrulline and L-arginine: Impact on nitric oxide metabolism. Br. J. Clin. Pharm. 2008, 65, 51–59. [Google Scholar] [CrossRef] [PubMed]
- Kaore, S.N.; Kaore, N.M. Chapter 53—Citrulline: Pharmacological perspectives and role as a biomarker in diseases and toxicities. In Biomarkers in Toxicology; Gupta, R.C., Ed.; Academic Press: Cambridge, MA, USA, 2014; pp. 883–905. [Google Scholar] [CrossRef]
- Pahlavani, N.; Entezari, M.H.; Nasiri, M.; Miri, A.; Rezaie, M.; Bagheri-Bidakhavidi, M.; Sadeghi, O. The effect of l-arginine supplementation on body composition and performance in male athletes: A double-blinded randomized clinical trial. Eur. J. Clin. Nutr. 2017, 71, 544. [Google Scholar] [CrossRef] [PubMed]
- Bassett, D.R.; Howley, E.T. Limiting factors for maximum oxygen uptake and determinants of endurance performance. Med. Sci. Sports Exerc. 2000, 32, 70–84. [Google Scholar] [CrossRef] [PubMed]
- Conley, D.L.; Krahenbuhl, G.S.; Burkett, L.N. Training for Aerobic Capacity and Running Economy. Physician Sportsmed. 1981, 9, 107–146. [Google Scholar] [CrossRef] [PubMed]
- Cheng, I.S.; Wang, Y.-W.; Chen, I.F.; Hsu, G.-S.; Hsueh, C.-F.; Chang, C.-K. The Supplementation of Branched-Chain Amino Acids, Arginine, and Citrulline Improves Endurance Exercise Performance in Two Consecutive Days. J. Sports Sci. Med. 2016, 15, 509–515. [Google Scholar] [PubMed]
- Hsueh, C.-F.; Wu, H.-J.; Tsai, T.-S.; Wu, C.-L.; Chang, C.-K. The Effect of Branched-Chain Amino Acids, Citrulline, and Arginine on High-Intensity Interval Performance in Young Swimmers. Nutrients 2018, 10. [Google Scholar] [CrossRef] [PubMed]
- McKinley-Barnard, S.; Andre, T.; Morita, M.; Willoughby, D.S. Combined L-citrulline and glutathione supplementation increases the concentration of markers indicative of nitric oxide synthesis. J. Int. Soc. Sports Nutr. 2015, 12, 27. [Google Scholar] [CrossRef] [PubMed]
- Fragkos, K.C.; Forbes, A. Citrulline as a marker of intestinal function and absorption in clinical settings: A systematic review and meta-analysis. United Eur. Gastroenterol. J. 2018, 6, 181–191. [Google Scholar] [CrossRef] [PubMed]
- Gregory, S.; Gary, S.; Louise, M.B. Changes in the Supplementation Practices of Elite Australian Swimmers Over 11 Years. Int. J. Sport Nutr. Exerc. Metab. 2016, 26, 565–571. [Google Scholar] [CrossRef]
- Maughan, R.J.; Burke, L.M.; Dvorak, J.; Larson-Meyer, D.E.; Peeling, P.; Phillips, S.M.; Rawson, E.S.; Walsh, N.P.; Garthe, I.; Geyer, H.; et al. IOC consensus statement: Dietary supplements and the high-performance athlete. Br. J. Sports Med. 2018, 52, 439. [Google Scholar] [CrossRef]
- The AIS Sports Supplement Framework. Available online: https://ais.gov.au/__data/assets/pdf_file/0004/698557/AIS-Sports-Supplement-Framework-2019.pdf (accessed on 11 August 2019).
- Botchlett, R.; Lawler, J.M.; Wu, G. Chapter 45 - L-Arginine and L-Citrulline in Sports Nutrition and Health. In Nutrition and Enhanced Sports Performance; Bagchi, D., Nair, S., Sen, C.K., Eds.; Academic Press: Cambridge, MA, USA, 2013; pp. 439–446. [Google Scholar] [CrossRef]
- Botchlett, R.; Lawler, J.M.; Wu, G. Chapter 55—L-Arginine and l-Citrulline in Sports Nutrition and Health. In Nutrition and Enhanced Sports Performance, 2nd ed.; Bagchi, D., Nair, S., Sen, C.K., Eds.; Academic Press: Cambridge, MA, USA, 2019; pp. 645–652. [Google Scholar] [CrossRef]
- Bentley, R.F.; Walsh, J.J.; Drouin, P.J.; Velickovic, A.; Kitner, S.J.; Fenuta, A.M.; Tschakovsky, M.E. Dietary nitrate restores compensatory vasodilation and exercise capacity in response to a compromise in oxygen delivery in the noncompensator phenotype. J. Appl. Physiol. 2017, 123, 594–605. [Google Scholar] [CrossRef]
- Goret, L.; Tanguy, S.; Guiraud, I.; Dauzat, M.; Obert, P. Acute administration of l-arginine restores nitric oxide-mediated relaxation in isolated pulmonary arteries from pulmonary hypertensive exercise trained rats. Eur. J. Pharmacol. 2008, 581, 148–156. [Google Scholar] [CrossRef]
- Gonzales, J.U.; Raymond, A.; Ashley, J.; Kim, Y. Does l-citrulline supplementation improve exercise blood flow in older adults? Exp. Physiol. 2017, 102, 1661–1671. [Google Scholar] [CrossRef]
- Tian, H.; Zheng, N.; Li, S.; Zhang, Y.; Zhao, S.; Wen, F.; Wang, J. Characterization of chiral amino acids from different milk origins using ultra-performance liquid chromatography coupled to ion-mobility mass spectrometry. Sci. Rep. 2017, 7, 46289. [Google Scholar] [CrossRef]
- Available online: https://www.nature.com/articles/srep46289#supplementary-information (accessed on 18 August 2019).
- Adibi, S.A.; Gray, S.J. Intestinal Absorption of Essential Amino Acids in Man. Gastroenterology 1967, 52, 837–845. [Google Scholar] [CrossRef]
- Cynober, L. Pharmacokinetics of Arginine and Related Amino Acids. J. Nutr. 2007, 137, 1646S–1649S. [Google Scholar] [CrossRef] [PubMed]
- Wang, W. Advanced protein formulations. Protein Sci. 2015, 24, 1031–1039. [Google Scholar] [CrossRef] [PubMed]
- Kotsonis, F.N.; Mackey, M.A. Nutritional Toxicology; CRC Press: Boca Raton, FL, USA, 2002. [Google Scholar]
- Sarwar Gilani, G.; Wu Xiao, C.; Cockell, K.A. Impact of Antinutritional Factors in Food Proteins on the Digestibility of Protein and the Bioavailability of Amino Acids and on Protein Quality. Br. J. Nutr. 2012, 108, S315–S332. [Google Scholar] [CrossRef] [PubMed]
- Ermolao, A.; Zanotto, T.; Carraro, N.; Fornasier, T.; Zaccaria, M.; Neunhaeuserer, D.; Bergamin, M. Repeated sprint ability is not enhanced by caffeine, arginine, and branched-chain amino acids in moderately trained soccer players. J. Exerc. Rehabil. 2017, 13, 55–61. [Google Scholar] [CrossRef]
- Doyle, M.P.; Duling, B.R. Acetylcholine induces conducted vasodilation by nitric oxide-dependent and -independent mechanisms. Am. J. Physiol. Heart Circ. Physiol. 1997, 272, H1364–H1371. [Google Scholar] [CrossRef]
- Bailey, S.J.; Blackwell, J.R.; Williams, E.; Vanhatalo, A.; Wylie, L.J.; Winyard, P.G.; Jones, A.M. Two weeks of watermelon juice supplementation improves nitric oxide bioavailability but not endurance exercise performance in humans. Nitric Oxide 2016, 59, 10–20. [Google Scholar] [CrossRef]
- Chappell, A.J.; Allwood, D.M.; Johns, R.; Brown, S.; Sultana, K.; Anand, A.; Simper, T. Citrulline malate supplementation does not improve German Volume Training performance or reduce muscle soreness in moderately trained males and females. J. Int. Soc. Sports Nutr. 2018, 15, 42. [Google Scholar] [CrossRef]
- Cutrufello, P.T.; Gadomski, S.J.; Zavorsky, G.S. The effect of l-citrulline and watermelon juice supplementation on anaerobic and aerobic exercise performance. J. Sports Sci. 2015, 33, 1459–1466. [Google Scholar] [CrossRef] [PubMed]
- Sarabia, J.M.; Moya-Ramón, M.; Hernández-Davó, J.L.; Fernandez-Fernandez, J.; Sabido, R. The effects of training with loads that maximise power output and individualised repetitions vs. traditional power training. PLoS ONE 2017, 12, e0186601. [Google Scholar] [CrossRef] [PubMed]
- Brooke, J.D. Assessment of performance capacity and potential. Br. J. Sports Med. 1973, 7, 344–348. [Google Scholar] [CrossRef]
- Sjodin, B.; Svedenhag, J. Applied Physiology of Marathon Running. Sports Med. 1985, 2, 83–99. [Google Scholar] [CrossRef] [PubMed]
- Bailey, S.J.; Blackwell, J.R.; Lord, T.; Vanhatalo, A.; Winyard, P.G.; Jones, A.M. l-Citrulline supplementation improves O2 uptake kinetics and high-intensity exercise performance in humans. J. Appl. Physiol. 2015, 119, 385–395. [Google Scholar] [CrossRef] [PubMed]
| Author, Year | Participants (n) | Objective | Outcomes | Concentration of Supplement | Strengths | Limitations |
|---|---|---|---|---|---|---|
| Bailey et al., 2015 [53] | n = 10 | To compare the effects of L-Cit and L-Arg supplementation on NO biomarkers, V̇O2 kinetics, and exercise performance. | Plasma NO concentration was increased with L-Arg supplementation (p < 0.05) and tended to increase with L-Cit supplementation (p = 0.08) compared with PL. V̇O2 during moderate-intensity cycle exercise was not significantly different (p > 0.05). | 6 g (of each) | Due to the short study design, a familiarization and baseline testing session was conducted to discount a ‘learning effect’ in exercise testing. | Short study duration (seven days) and small sample size (n = 10). |
| Chen et al., 2016 [10] | n = 12 | To investigate the effect of the combination L-Arg and L-Cit on central fatigue. | Significantly better response time measured in AA group compared with PL. | 0.05 g/kg of body weight (of each) | Previously tested the point after supplementation when plasma L-Arg would peak, and began testing protocol 1-h after supplementation. | Small sample size (n = 12). |
| Cheng et al., 2016 [26] | n = 13 | To investigate the combination of L-Arg and L-Cit on endurance performance. | AA group significantly better in two endurance running distances on both days. | 0.05 g/kg of body weight (of each) | Testing protocol was repeated a day later to validate outcomes. | Assessed acute effect of supplementation with one day washout period—potential accumulation of plasma AA not discussed? |
| Hsueh et al., 2018 [27] | n = 16 | To examine the effect of co-ingestion of L-Arg and L-Cit on high-intensity interval swim performance in trained young swimmers. | Average swim time was shorter in treatment group when compared with PL. RPE was similar between the two groups. | 0.05 g/kg of body weight (of each) | Testing protocol began one hour after supplementation to allow for peak plasma levels. | Relatively small sample size (n = 16). |
| Morita et al., 2014 [13] | [animal] n = 24 | To investigate the acute effects of a combination of oral L-Cit and L-Arg on plasma L-Arg and NO levels, as well as on blood circulation. | L-Arg and L-Cit combined caused a more rapid increase and enhancement of NO bioavailability compared with single individual AA. | 2.85 mmol·kg−1 (of each) animal body weight, and 1.43 mmol/kg of combined L-Arg/L-Cit | Strong study design, highlights future direction for human studies. | Animal trial—not directly transferrable? |
| Silva et al., 2017 [8] | [animal] n = 40 | To evaluate the effects of supplementing with L-Arg and L-Cit on performance and oxidative stress in trained and untrained rats | Supplementation improved physical performance in both control and trained groups. | 300 mg/kg animal body weight | Strong study design, highlights future direction for human studies. | Animal trial—not directly transferrable? |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Speer, H.; D’Cunha, N.M.; Davies, M.J.; McKune, A.J.; Naumovski, N. The Physiological Effects of Amino Acids Arginine and Citrulline: Is There a Basis for Development of a Beverage to Promote Endurance Performance? A Narrative Review of Orally Administered Supplements. Beverages 2020, 6, 11. https://doi.org/10.3390/beverages6010011
Speer H, D’Cunha NM, Davies MJ, McKune AJ, Naumovski N. The Physiological Effects of Amino Acids Arginine and Citrulline: Is There a Basis for Development of a Beverage to Promote Endurance Performance? A Narrative Review of Orally Administered Supplements. Beverages. 2020; 6(1):11. https://doi.org/10.3390/beverages6010011
Chicago/Turabian StyleSpeer, Hollie, Nathan M. D’Cunha, Michael J. Davies, Andrew J. McKune, and Nenad Naumovski. 2020. "The Physiological Effects of Amino Acids Arginine and Citrulline: Is There a Basis for Development of a Beverage to Promote Endurance Performance? A Narrative Review of Orally Administered Supplements" Beverages 6, no. 1: 11. https://doi.org/10.3390/beverages6010011
APA StyleSpeer, H., D’Cunha, N. M., Davies, M. J., McKune, A. J., & Naumovski, N. (2020). The Physiological Effects of Amino Acids Arginine and Citrulline: Is There a Basis for Development of a Beverage to Promote Endurance Performance? A Narrative Review of Orally Administered Supplements. Beverages, 6(1), 11. https://doi.org/10.3390/beverages6010011







