Caffeine Consumption through Coffee: Content in the Beverage, Metabolism, Health Benefits and Risks
Abstract
1. Introduction
2. Chemical Aspects and Analysis of Caffeine
3. Contents of Caffeine in Green Coffee Seeds
4. Contents of Caffeine in Regular and Decaffeinated Roasted Coffee Seeds and in Brews
4.1. Caffeine in Roasted and Ground Coffee
4.2. Caffeine in Soluble Coffee
4.3. Caffeine in Coffee Brew
4.4. Caffeine Complexation with Chlorogenic Acids
4.5. Caffeine in Decaffeinated Coffee
5. Global Caffeine Consumption Through Coffee
6. Caffeine Metabolism
6.1. Absorption
6.2. Metabolism and Distribution
6.3. Excretion
7. Health Benefits of Caffeine Consumption
7.1. Caffeine, Mood, and Behavior
7.2. Caffeine and Exercise Performance
7.3. Caffeine and Antioxidant and Antiinflammatory Activities
7.4. Caffeine and Antimicrobial Activity
7.5. Caffeine and Neurodegenerative Diseases
7.6. Caffeine and Liver Diseases
8. Potential Adverse Effects of Caffeine Consumption
8.1. Caffeine Acute and Chronic Toxicity
8.2. Potential Adverse Effects of Caffeine on Mood, Behavior, and Sleep
8.3. Potential Adverse Effects of Caffeine on Cardiovascular System
8.4. Potential Adverse Effects of Caffeine on Glucose Metabolism and Insulin Resistance
8.5. Potential Adverse Effects of Caffeine on Calcium Balance
8.6. Potential Adverse Effects of Caffeine on Fertility and Reproductive and Developmental Effects
8.7. Potential Carcinogenicity of Caffeine
8.8. Caffeine Withdrawal Syndrome
9. Maximum Caffeine Intake Recommendations
10. Concluding Remarks
Funding
Acknowledgments
Conflicts of Interest
References
- Fredholm, B.B. Notes on the history of caffeine use. In Methylxanthines; Fredholm, B.B., Ed.; Springer: New York, NY, USA, 2011; pp. 1–9. [Google Scholar]
- Tarka, S.M.; Hurst, W.J. Introduction to the chemistry, isolation, and biosynthesis of methylxanthines. In Caffeine; Spiller, G.A., Ed.; CRC Press: Boca Raton, FL, USA, 1998; pp. 1–12. [Google Scholar]
- Zulak, K.G.; Liscome, D.K.; Ashihara, H.; Facchini, P.J. Alkaloids. In Plant Secondary Metabolites: Occurrence Structure, and Role in the Human Diet; Crozier, A., Clifford, M.N., Ashihara, H., Eds.; Blackwell: Oxford, UK, 2006; pp. 102–136. [Google Scholar]
- Lima, J.P.; Farah, A. Caffeine and minor methylxanthines in coffee. In Coffee: Consumption and Health Implications; Farah, A., Ed.; Royal Society of Chemistry: London, UK, 2019; ISBN 978-1782620044. [Google Scholar]
- Pauwels, R.A. Global strategy for the diagnosis, management, and prevention of chronic obstructive pulmonary disease. NHLBI/WHO Global Initiative for Chronic Obstructive Lung Disease (GOLD) Workshop summary. Am. J. Respir. Crit. Care Med. 2001, 163, 1256–1276. [Google Scholar] [CrossRef] [PubMed]
- Ashihara, H.; Crozier, A. Biosynthesis and metabolism of caffeine and related purine alkaloids in plants. Adv. Bot. Res. 1999, 30, 117–205. [Google Scholar] [CrossRef]
- Chow, T.M.; Benowitz, N.L. Caffeine and coffee: Effects on health and cardiovascular disease. Comp. Biochem. Physiol. 1994, 109, 173–189. [Google Scholar] [CrossRef]
- von Goethe, J.W.; von Biedermann, F.W. Hauswirtschaftlichen Briefen; von Goethe, J.W., von Biedermann, F.W., Eds.; Goethes Gespräche, Nachträge: Leipzig, Germany, 1896; pp. 89–96. [Google Scholar]
- Weinberg, A.; Bealer, B.K. (Eds.) The World of Caffeine: The Science and Culture of the World’s Most Popular Drug; Routledge: New York, NY, USA, 2001; p. 394. [Google Scholar]
- Meusinger, R. Solution to spectroscopy challenge 13. Caffeine. Anal Bioanal. Chem. 2009, 393, 1381–1382. [Google Scholar] [CrossRef] [PubMed]
- Fischer, E. Ueber das Caffeïn. Ber. Dtsch.Chem. Ges. 1881, 14, 1905–1915. [Google Scholar] [CrossRef]
- Fischer, E. Ueber das Caffeïn. Ber. Dtsch. Chem. Ges. 1881, 14, 637–644. [Google Scholar] [CrossRef]
- Vitzthum, O.G.; Werkhoff, P.Z. Steam volatile aroma constituents of roasted coffee: Neutral fraction. Lebensm Unters Forsch. 1976, 160, 277–291. [Google Scholar] [CrossRef]
- Arnaud, M.J. Encyclopedia of Human Nutrition’s; Caballero, B., Allen, L., Prentice, A., Eds.; Elsevier Academic Press: Cambridge, UK, 2005; p. 247. [Google Scholar]
- Belay, A.; Ture, K.; Redi, M.; Asfaw, A. Measurement of caffeine in coffee beans with UV-Vis spectrometer. Food Chem. 2008, 108, 310–315. [Google Scholar] [CrossRef]
- Holiday, E.R. The characteristic absorption of ultra-violet radiation by certain purines. Biochem. J. 1930, 24, 619–625. [Google Scholar] [CrossRef]
- Association of Official Agricultural Chemists in Official and Tentative Methods of Analysis; George Banta Publishing: Washington, DC, USA, 1945.
- De Maria, C.A.B.; Moreira, R.F.A. Cafeína: Revisão sobre métodos de análise. Quim. Nova 2007, 30, 99–105. [Google Scholar] [CrossRef]
- Hartley, W.N. Observations on chemical structure and those physical properties on which the theory of color is based. J. Chem. Soc. Trans. 1905, 87, 1822–1831. [Google Scholar] [CrossRef]
- Ishler, N.H.; Finucane, T.P.; Borker, E. Rapid spectrophotometric determination of caffeine. Anal. Chem. 1948, 20, 1162–1166. [Google Scholar] [CrossRef]
- López-Martínez, L.; López-de-Alba, P.L.; García-Campos, R.; De León-Rodríguez, L.M. Simultaneous determination of methylxanthines in coffees and teas by UV-Vis spectrophotometry and partial least squares. Anal. Chim. Acta 2003, 493, 83–94. [Google Scholar] [CrossRef]
- Brown, S.R.; Cann, P.A.; Read, N.W. Effect of coffee on distal colon function. Gut 1990, 31, 450–453. [Google Scholar] [CrossRef] [PubMed]
- Murgia, E.; Richards, P.; Walton, H.F. Liquid chromatography of xanthine’s, analgesic drugs and coffee. J. Chromatogr. 1973, 87, 523–533. [Google Scholar] [CrossRef]
- Bispo, M.S.; Veloso, M.C.C.; Pinheiro, H.L.C.; de Oliveira, R.F.S.; Reis, J.O.N.; de Andrade, J.B. Simultaneous determination of caffeine, theobromine, and theophylline by high-performance liquid chromatography. J. Chromatogr. Sci. 2002, 40, 45–48. [Google Scholar] [CrossRef] [PubMed]
- Farah, A.; de Paulis, T.; Trugo, L.C.; Martin, P.R. Chlorogenic acids and lactones in regular and water-decaffeinated arabica coffees. J. Agric. Food Chem. 2006, 54, 374–381. [Google Scholar] [CrossRef]
- Perrone, D.; Donangelo, C.M.; Farah, A. Fast simultaneous analysis of caffeine, trigonelline, nicotinic acid and sucrose in coffee by liquid chromatography-mass spectrometry. Food Chem. 2008, 110, 1030–1035. [Google Scholar] [CrossRef] [PubMed]
- Trugo, L.C.; Macrae, R.; Dick, J. Determination of purine alkaloids and trigonelline in instant coffee and other beverages using high performance liquid chromatography. J. Sci. Food Agric. 1983, 34, 300–306. [Google Scholar] [CrossRef]
- Casal, S.; Oliveira, M.B.; Ferreira, M.A. Development of an HPLC/diode-array detector method for simultaneous determination of trigonelline, nicotinic acid, and caffeine in coffee. Food Chem. 1998, 21, 3187–3195. [Google Scholar] [CrossRef]
- Casal, S.; Oliveira, M.B.; Ferreira, M.A. Discriminate analysis of roasted coffee varieties for trigonelline, nicotinic acid, and caffeine content. J. Agric. Food Chem. 2000, 48, 3420–3424. [Google Scholar] [CrossRef] [PubMed]
- Redivo, L.; Stredanský, M.; De Angelis, E.; Navarini, L.; Resmini, M.; Švorc, Ĺ. Bare carbon electrodes as simple and efficient sensors for the quantification of caffeine in commercial beverages. R. Soc. Open Sci. 2018, 5, 172146. [Google Scholar] [CrossRef] [PubMed]
- Perrone, D.; Farah, A. Caffeine: Chemistry, Analysis, Function and Effects; Preedy, V., Ed.; Royal Society of Chemistry: Cambridge, UK, 2012; p. 97. ISBN -9781849733670. [Google Scholar]
- Briandet, R.; Kemsley, E.K.; Wilson, R.H. Discrimination of arabica and robusta in instant coffees by Fourier transform infrared spectroscopy and chemometrics. J. Agric. Food Chem. 1996, 44, 170–174. [Google Scholar] [CrossRef]
- Bouhsain, Z.; Garrigues, S.; de la Guardia, M. Clean method for the simultaneous determination of propyphenazone and caffeine in pharmaceuticals by flow injection Fourier transform infrared spectrometry. Analyst 1997, 122, 441–445. [Google Scholar] [CrossRef] [PubMed]
- Harborne, B.J. Introduction to Ecological Biochemistry; Harborne, J.B., Ed.; Elsevier Academic Press: Cambridge, UK, 1993; p. 1. ISBN 9780080918587. [Google Scholar]
- Hewavitharanage, P.; Karunaratne, S.; Kumar, N.S. Effect of caffeine on shot-hole borer beetle (Xyleborusfornicatus) of tea (Camellia sinensis). Phytochemistry 1999, 51, 35–41. [Google Scholar] [CrossRef]
- Waller, G.R. Biochemical frontiers of allelopathy. Biol. Plant. 1989, 31, 418–447. [Google Scholar] [CrossRef]
- Mazzafera, P.; Carvalho, A. Breeding for low seed caffeine content of coffee (Coffea L.) by interspecific hybridization. Euphytica 1992, 59, 55–60. [Google Scholar] [CrossRef]
- Macrae, R. Coffee Chemistry; Clarke, R.J., Macrae, R., Eds.; Elsevier: New York, NY, USA, 1985; p. 115. [Google Scholar]
- Farah, A.; Monteiro, M.C.; Calado, V.; Franca, A.S.; Trugo, L.C. Correlation between cup quality and chemical attributes of Brazilian coffee. Food Chem. 2006, 98, 373–380. [Google Scholar] [CrossRef]
- Duarte, G.S.; Pereira, A.A.; Farah, A. Chlorogenic acids and other relevant compounds in Brazilian coffees processed by semi-dry and wet post-harvesting methods. Food Chem. 2010, 118, 851–855. [Google Scholar] [CrossRef]
- Alonso-Salces, R.M.; Serra, F.; Reniero, F.; Rolyheberger, A. Botanical and geographical characterization of green coffee (Coffea arabica and Coffea canephora): Chemometric evaluation of phenolic and methylxanthine contents. J. Agric. Food Chem. 2009, 57, 4224–4235. [Google Scholar] [CrossRef]
- Clifford, M.N.; Kazi, T. The influence of coffee bean maturity on the content of chlorogenic acids, caffeine and trigonelline. Food Chem. 1987, 26, 59–69. [Google Scholar] [CrossRef]
- Clifford, M.N. Coffee: Botany, Biochemistry and Production of Beans and Beverage; Clifford, M.N., Wilson, K.C., Eds.; Springer: New York, NY, USA, 1985; p. 305. [Google Scholar]
- Mazzafera, P.; Crozier, A.; Magalhães, A.C. Caffeine metabolism in Coffea arabica and other species of coffee. Phytochemistry 1991, 30, 3913–3916. [Google Scholar] [CrossRef]
- Clifford, M.N.; Ramirez-Martinez, J.R. Phenols and caffeine in wet-processed coffee beans and coffee pulp. Food Chem. 1991, 40, 35–42. [Google Scholar] [CrossRef]
- Mazzafera, P.; Carvalho, A.; Fazuoli, L.C.; Medina Filho, H.P. Variabilidade do teor de cafeína em sementes de café. Turrialba 1992, 42, 231–237. [Google Scholar]
- Ky, C.L.; Louarn, J.; Guyot, B.; Dussert, S.; Hamon, S.; Noirot, M. Caffeine, trigonelline, chlorogenic acids and sucrose diversity in wild Coffea arabica L. and Coffea canephora P. accessions. Food Chem. 2001, 75, 223–230. [Google Scholar] [CrossRef]
- Mazzafera, P.; Silvarolla, M.B. Caffeine content variation in single green Arabica coffee seeds. Seed Sci. Res. 2010, 20, 163–167. [Google Scholar] [CrossRef]
- de Souza, R.M.N.; Benassi, M.T. Discrimination of commercial roasted and ground coffees according to chemical composition. J. Braz. Chem. Soc. 2012, 23, 1347–1354. [Google Scholar] [CrossRef]
- Hečimović, I.; Belščak-Cvitanović, A.; Horžić, D.; Komes, D. Comparative study of polyphenols and caffeine in different coffee varieties affected by the degree of roasting. Food Chem. 2011, 129, 991–1000. [Google Scholar] [CrossRef]
- Mehari, B.; Redi-Abshiro, M.; Chandravanshi, B.S. Simultaneous determination of alkaloids in green coffee beans from Ethiopia: Chemometric evaluation of geographical origin. Food Anal. Methods 2016, 9, 1627–1637. [Google Scholar] [CrossRef]
- Babova, O.; Occhipinti, A.; Maffei, M.E. Chemical partitioning and antioxidant capacity of green coffee (Coffea arabica and Coffea canephora) of different geographical origin. Phytochemistry 2016, 123, 33–39. [Google Scholar] [CrossRef]
- Martín, M.J.; Pablos, F.; Gonzalez, A.G. Discrimination between arabica and robusta green coffee varieties according to their chemical composition. Talanta 1998, 46, 1259–1264. [Google Scholar] [CrossRef]
- Nebesny, E.; Budryn, G. Antioxidative activity of green and roasted coffee beans as influenced by convection and microwave roasting methods and content of certain compounds. Eur. Food Res. Technol. 2003, 217, 157–163. [Google Scholar] [CrossRef]
- Campa, C.; Doulbeau, S.; Dussert, S.; Hamon, S.; Noirot, M. Qualitative relationship between caffeine and chlorogenic acid contents among wild Coffea species. Food Chem. 2005, 93, 135–139. [Google Scholar] [CrossRef]
- Ramalakshmi, K.; Kubra, R.; Rao, L.J.M. Physicochemical characteristics of green coffee: Comparison of graded and defective beans. J. Food Sci. 2007, 72, 333–337. [Google Scholar] [CrossRef]
- Farah, A. Nutritional and health aspects of coffee. In Coffee: Emerging Health Effects and Disease Prevention; Chu, Y.-F., Ed.; IFT Press and John Wiley & Sons: New York, NY, USA, 2012; p. 21. [Google Scholar]
- Burdan, F. Coffee in Health and Disease Prevention; Preedy, V.R., Ed.; Elsevier: Cambridge, UK, 2015; p. 823. ISBN 9780124167162. [Google Scholar]
- Fujimori, N.; Ashihara, H. Biosynthesis of theobromine and caffeine in developing leaves of Coffea arabica. Phytochemistry 1994, 36, 1359–1361. [Google Scholar] [CrossRef]
- Payen, S. Undersuchung des Kaffees. Annalen 1846, 60, 286–294. [Google Scholar]
- Sondheimer, E.; Szymanski, C.D.; Corse, J.W. Coffee constituents, isolation of chlorogenic acid and its isomers from coffee. J. Agric. Food Chem. 1961, 9, 146–149. [Google Scholar] [CrossRef]
- D’Amelio, N.; Fontanive, L.; Uggeri, F.; Suggi-Liverani, F.; Navarini, L. NMR reinvestigation of the caffeine–chlorogenate complex in aqueous solution and in coffee brews. Food Biophys. 2009, 4, 321–330. [Google Scholar] [CrossRef]
- D’Amelio, N.; Papamokos, G.; Dreyer, J.; Carloni, P.; Navarini, L. NMR studies of hetero-association of caffeine with di-O-caffeoylquinic acid isomers in aqueous solution. Food Biophys. 2015, 10, 235–243. [Google Scholar] [CrossRef][Green Version]
- Horman, I.; Viani, R. The nature and complexation of the caffeine-chlorogenate complex of coffee. J. Food Sci. 1972, 37, 925–927. [Google Scholar] [CrossRef]
- Anthony, F.; Noirot, M.; Clifford, M.N. Biochemical diversity in the genus Coffea L.: Chlorogenic acids, caffeine and mozambioside contents. Genet. Resour. Crop Evol. 1993, 40, 61–70. [Google Scholar] [CrossRef]
- Teixeira, A.A.; Brando, C.H.; Thomaziello, R.A.; Teixeira, R.; Drying, R. Espresso Coffee: The Science of Quality; Illy, A., Viani, R., Eds.; Elsevier Academic Press: San Diego, CA, USA, 1995; p. 91. [Google Scholar]
- Trugo, L.C.; Macrae, R. Chlorogenic acid composition of instant coffees. Analyst 1984, 109, 263–266. [Google Scholar] [CrossRef] [PubMed]
- Balyaya, K.J.; Clifford, M.N. Chlorogenic acids and caffeine contents of monsooned Indian Arabica and Robusta coffees compared with wet and dry processed coffees from the same geographic area. In Proceedings of the 16th of the International Conference on Coffee Science, Kyoto, Japan, 1995; p. 316. [Google Scholar]
- Leloup, V.; Louvrier, A.; Liardon, R. Degradation mechanisms of chlorogenic acids during roasting. In Proceedings of the 16th of the International Conference on Coffee Science, Kyoto, Japan, 1995; p. 192. [Google Scholar]
- Franca, A.S.; Oliveira, L.S.; Mendonça, C.F.; Silva, X.A. Physical and chemical attributes of defective crude and roasted coffee beans. Food Chem. 2005, 90, 84–89. [Google Scholar] [CrossRef]
- Toci, A.T.; Farah, A. Contents of chlorogenic acids, caffeine, trigoneline and sucrose in Brazilian defective deeds. In Proceedings of the 23th of the International Conference on Coffee Science, Bali, Indonesia, 2011; p. 260. [Google Scholar]
- Bicho, N.C.; Leitão, E.R.; Ramalho, J.C.; Lidon, F.C. Identification of chemical clusters discriminators of the roast degree in Arabica and Robusta coffee beans. Eur. Food Res. Technol. 2011, 233, 303–311. [Google Scholar] [CrossRef]
- Pilipczuk, T.; Kusznierewicz, B.; Zielińska, D.; Bartoszek, A. The influence of roasting and additional processing on the content of bioactive components in special purpose coffees. J. Food Sci. Technol. 2014, 52, 5736–5744. [Google Scholar] [CrossRef] [PubMed]
- Kitzberger, C.S.G.; Scholz, M.B.; Benassi, M.T. Bioactive compounds content in roasted coffee from traditional and modern Coffea arabica cultivars grown under the same edapho-climatic conditions. Food Res. Int. 2014, 61, 61–66. [Google Scholar] [CrossRef]
- Monteiro, M.C.; Trugo, L.C. Determination of bioactive compounds in brazilian roasted coffees. Quím. Nova 2005, 28, 637–641. [Google Scholar] [CrossRef]
- Fujioka, K.; Shibamoto, T. Chlorogenic acid and caffeine contents in various commercial brewed coffees. Food Chem. 2008, 106, 217–221. [Google Scholar] [CrossRef]
- Santos, J.R.; Rangel, A.O.S.S. Development of a chromatographic low-pressure flow injection system: Application to the analysis of methylxanthines in coffee. Anal. Chim. Acta 2012, 715, 57–63. [Google Scholar] [CrossRef]
- dePaula, J.; Farah, A. Methylxanthines in stimulant foods and beverages commonly consumed in Brazil. J. Food Compos. Anal. 2019. [Google Scholar] [CrossRef]
- Pérez-Hernández, L.M.; Chávez-Quiroz, K.; Medina-Juárez, L.A.; Meza, N.G. Phenolic characterization, melanoidins, and antioxidant activity of some commercial coffees from Coffea arabica and Coffea canephora. J. Mex. Chem. Soc. 2012, 56, 430–435. [Google Scholar]
- Vignoli, J.A.; Bassoli, D.G.; Benassi, M.T. Antioxidant activity, polyphenols, caffeine and melanoidins in soluble coffee: The influence of processing conditions and raw material. Food Chem. 2011, 124, 863–868. [Google Scholar] [CrossRef]
- Nogueira, M.; Trugo, L.C. Distribuição de isômeros de ácido clorogênico e teores de cafeína e trigonelina em cafés solúveis brasileiros. Cienc. Tecnol. Aliment. 2003, 23, 296–299. [Google Scholar] [CrossRef]
- Wanyika, H.N.; Gatebe, E.G.; Gitu, L.M.; Ngumba, E.K.; Maritim, C.W. Determination of caffeine content of tea and instant coffee brands found in the Kenyan market. Afr. J. Food Sci. 2010, 4, 353–358. [Google Scholar]
- Del Campo, G.; Berregi, I.; Caracena, R.; Zuriarrain, J. Quantitative determination of caffeine, formic acid, trigonelline and 5-(hydroxymethyl)furfural in soluble coffees by 1H NMR spectrometry. Talanta 2010, 81, 367–371. [Google Scholar] [CrossRef] [PubMed]
- Farah, A. Nutritional and health effects of coffee. In Achieving Sustainable Cultivation of Coffee; Lashermes, P., Ed.; Burleigh Dodds Science Publishing: Cambridge, UK, 2017; pp. 1–31. ISBN 978-1-78676-152-1. [Google Scholar]
- McCusker, R.R.; Goldberger, B.A.; Cone, E.J. Caffeine content of specialty coffees. J. Anal. Toxicol. 2003, 27, 520–522. [Google Scholar] [CrossRef] [PubMed]
- Jeon, J.S.; Kim, H.T.; Jeong, I.H.; Hong, S.R.; Oh, M.S.; Park, K.H.; Shim, J.H.; Abd El-Aty, A.M. Determination of chlorogenic acids and caffeine in homemade brewed coffee prepared under various conditions. J. Chromatogr. B Anal. Technol. Biomed. Life Sci. 2017, 1064, 115–123. [Google Scholar] [CrossRef]
- Rodrigues, N.P.; Bragagnolo, N. Identification and quantification of bioactive compounds in coffee brews by HPLC-DAD-MS. J. Food Comp. Anal. 2013, 32, 105–115. [Google Scholar] [CrossRef]
- Burg, A.W. Effects of caffeine in the human system. Tea Coffee Trade J. 1975, 147, 40–41. [Google Scholar]
- Gilbert, R.M. Caffeine Consumption; Spiller, G.A., Ed.; A. R. Liss Incorporation: New York, NY, USA, 1984; p. 185. [Google Scholar]
- Barone, J.J.; Roberts, H.R. Caffeine consumption. Food Chem. Toxicol. 1996, 34, 119–129. [Google Scholar] [CrossRef]
- Center for Science in the Public Interest. Available online: http://www.cspinet.org/new/cafchart.htm (accessed on 1 February 2019).
- Bravo, J.; Juániz, I.; Monente, C.; Caemmererb, B.; Krohb, L.W.; Paz De Peña, M.; Cid, C. Evaluation of spent coffee obtained from the most common coffeemakers as a source of hydrophilic bioactive compounds. J. Agric. Food Chem. 2012, 60, 12565–12573. [Google Scholar] [CrossRef] [PubMed]
- Lopez-Galilea, I.; de Peña, M.P.; Cid, C. Correlation of selected constituents with the total antioxidant capacity of coffee beverages: influence of the brewing procedure. J. Agric. Food Chem. 2007, 55, 6110–6117. [Google Scholar] [CrossRef]
- Angeloni, G.; Guerrini, L.; Masella, P.; Bellumori, M.; Daluiso, S.; Parenti, A.; Innocenti, M. What kind of coffee do you drink? An investigation on effects of eight different extraction methods. Food Res. Int. 2018. [Google Scholar] [CrossRef]
- Maeztu, L.; Andueza, S.; Ibañez, C.; de Peña, M.P.; Bello, J.; Cid, C. Multivariate methods for characterization and classification of espresso coffees from different botanical varieties and types of roast by foam, taste, and mouthfeel. J. Agric. Food Chem. 2001, 49, 4743–4747. [Google Scholar] [CrossRef] [PubMed]
- Crozier, T.W.M.; Stalmach, A.; Lean, M.E.J.; Crozier, A. Espresso coffees, caffeine and chlorogenic acid intake: Potential health implications. Food Funct. 2012, 3, 30–33. [Google Scholar] [CrossRef]
- Niseteo, T.; Komes, D.; Belščak-Cvitanović, A.; Horžić, D.; Budeč, M. Bioactive composition and antioxidant potential of different commonly consumed coffee brews affected by their preparation technique and milk addition. Food Chem. 2012, 134, 1870–1877. [Google Scholar] [CrossRef] [PubMed]
- Desbrow, B.; Hughes, R.; Leveritt, M.; Scheelings, P. An examination of consumer exposure to caffeine from retail coffee outlets. Food Chem. Toxicol. 2007, 45, 1588–1592. [Google Scholar] [CrossRef] [PubMed]
- Fuller, M.; Rao, N.Z. The effect of time, roasting temperature, and grind size on caffeine and chlorogenic acid concentrations in cold brew coffee. Sci. Rep. 2017, 7, 17979. [Google Scholar] [CrossRef] [PubMed]
- Tfouni, S.A.V.; Carreiro, L.B.; Teles, C.R.A.; Furlani, R.P.Z.; Cipolli, K.M.V.A.B.; Camargo, M.C.R. Caffeine and chlorogenic acids intake from coffee brew: Influence of roasting degree and brewing procedure. Int. J. Food Sci. Technol. 2014, 49, 747–752. [Google Scholar] [CrossRef]
- Aragão, N.M.D.; Veloso, M.C.C.; Bispo, M.S.; Ferreira, S.L.C.; Andrade, J.B. Multivariate optimization of the experimental conditions for determination of three methylxanthines by reversed-phase high-performance liquid chromatography. Talanta 2005, 67, 1007–1013. [Google Scholar] [CrossRef]
- Sereshti, H.; Samadi, S. Rapid and simple determination of caffeine in teas, coffees and eight beverages. Food Chem. 2014, 158, 8–13. [Google Scholar] [CrossRef] [PubMed]
- Farah, A.; Lima, J.P. Consumption of chlorogenic acids through coffee and health implications. Beverages 2019, 5, 11. [Google Scholar] [CrossRef]
- National Coffee Association. National Coffee Drinking Trends. 2009. Available online: http://www.ncausa.org (accessed on 5 February 2019).
- Scientific American. How Is Caffeine Remove to Produce Decaffeinated Coffee? Available online: https://www.scientificamerican.com/article/how-is-caffeine-removed-t/ (accessed on 9 February 2019).
- International Trade Centre. Available online: http://www.intracen.org/coffee-guide/the-markets-for-coffee/demand---Decaffeinated-coffee (accessed on 9 February 2019).
- European Coffee Federation. Available online: http://www.ecf-coffee.org (accessed on 1 February 2019).
- United States Department of Agriculture. Available online: https://ndb.nal.usda.gov/ndb (accessed on 1 February 2019).
- Agência Nacional de Vigilância Sanitária. Available online: http://www.anvisa.gov.br (accessed on 1 February 2019).
- McCusker, R.R.; Goldberger, B.A.; Cone, E.J. Caffeine content of energy drinks, carbonated sodas, and other beverages. J. Anal. Toxicol. 2006, 30, 112–114. [Google Scholar] [CrossRef] [PubMed]
- Ogawa, H.; Ueki, N. Clinical importance of caffeine dependence and abuse. Psychiatry Clin. Neurosci. 2007, 61, 263–268. [Google Scholar] [CrossRef] [PubMed]
- Fredholm, B.B.; Battig, K.; Holmén, J.; Nehlig, A.; Zvartau, E.E. Actions of caffeine in the brain with special reference to factors that contribute to its widespread use. Pharmacol. Rev. 1999, 51, 83–133. [Google Scholar] [PubMed]
- Smit, H.J.; Rogers, P.J. Effects of ‘energy’ drinks on mood and mental performance: Critical methodology. Food Qual. Pref. 2002, 13, 317–326. [Google Scholar] [CrossRef]
- Arnaud, M.J. Pharmacokinetics and metabolism of natural methylxanthines in animal and man. Handb. Exp. Pharmacol. 2011, 200, 33–91. [Google Scholar] [CrossRef]
- Borota, D.; Murray, E.; Keceli, G.; Chang, A.; Watabe, J.M.; Ly, M.; Toscano, J.P.; Yassa, M.A. Post-study caffeine administration enhances memory consolidation in humans. Nat. Neurosci. 2014, 17, 201–203. [Google Scholar] [CrossRef]
- Ludwig, I.A.; Clifford, M.N.; Lean, M.E.; Ashihara, H.; Crozier, A. Coffee: Biochemistry and potential impact on health. Food Funct. 2014, 5, 1695–1717. [Google Scholar] [CrossRef] [PubMed]
- International Coffee Organization. World Coffee Consumption. Available online: http://www.ico.org (accessed on 1 February 2019).
- World Atlas. Top 10 Coffee Consuming Nations. Available online: https://www.worldatlas.com/articles/top-10-coffee-consuming-nations.html (accessed on 1 February 2019).
- Clifford, M.N.; Ramirez-Martinez, J.R. Chlorogenic acids and purine alkaloids contents of mate (Ilex paraguariensis) leaf and beverage. Food Chem. 1990, 35, 13–21. [Google Scholar] [CrossRef]
- Pelozo, M.I.G.; Cardoso, M.L.C.; de Mello, J.C.P. Spectrophotometric determination of tannins and caffeine in preparations from Paullinia cupana var. sorbilis. Braz. Arch. Biol. Technol. 2008, 51, 447–451. [Google Scholar] [CrossRef]
- Vester, J.C.; Koenig, J. Caffeine intake and its sources: A review of national representative studies. Crit. Rev. Food Sci. Nutr. 2018, 58, 1250–1259. [Google Scholar] [CrossRef] [PubMed]
- Lima, J.P.; Farah, A. Caffeine Consumption. In Coffee: Consumption and Health Implications; Farah, A., Ed.; Royal Society of Chemistry: London, UK, 2018; ISBN 978-1782620044. [Google Scholar]
- Food and Agriculture Organization of the United Nations. Food Balance Sheets; FAO: Rome, Italy, 1995. [Google Scholar]
- Mitchell, D.C.; Knight, C.A.; Hockenberry, J.; Teplansky, R.; Hartman, T.J. Beverage caffeine intakes in the U.S. Food Chem. Toxicol. 2014, 63, 136–142. [Google Scholar] [CrossRef] [PubMed]
- Mitchell, D.C.; Hockenberry, J.; Teplansky, R.; Hartman, T.J. Assessing dietary exposure to caffeine from beverages in the U.S. population using brand-specific versus category-specific caffeine values. Food Chem. Toxicol. 2015, 80, 247–252. [Google Scholar] [CrossRef] [PubMed]
- European Food Safety Authority—(EFSA Panel on Dietetic Products, Nutrition and Allergies). Scientific Opinion on the Safety of Caffeine; European Food Safety Authority: Parma, Italy, 2015.
- Heckman, M.A.; Weil, J.; De Mejia, E.G. Caffeine (1, 3, 7-trimethylxanthine) in foods: A comprehensive review on consumption, functionality, safety, and regulatory matters. J. Food Sci. 2010, 75, 77–87. [Google Scholar] [CrossRef] [PubMed]
- Lima, J.P. Contribuição de alimentos fonte para a ingestão dietética habitual estimada de metilxantinas no Brasil e no município do Rio de Janeiro. Master’s Thesis, Instituto de Nutrição, Universidade Federal do Rio de Janeiro, Rio de Janeiro, Brasil, 2014. [Google Scholar]
- Knight, C.A.; Knight, I.; Mitchell, D.C.; Zepp, J.E. Beverage caffeine intake in US consumers and subpopulations of interest: Estimates from the Share of Intake Panel survey. Food Chem. Toxic. 2004, 42, 1923–1930. [Google Scholar] [CrossRef]
- Olmos, V.; Bardoni, N.; Ridolfi, A.S.; Villaamil, E.C. Caffeine levels in beverages from Argentina’s market: Application to caffeine dietary intake assessment. Food Addit. Contam. Part A 2009, 26, 275–281. [Google Scholar] [CrossRef]
- Bizzo, M.L.G.; Farah, A.; Kemp, J.A.; Scancetti, L.B. Highlights in the History of Coffee Science Related to Health. In Coffee and Health Disease Prevention; Preedy, V.R., Ed.; Elsevier: Cambridge, UK, 2015; pp. 11–17, 812. ISBN 9780124167162. [Google Scholar]
- Lima, J.P.; Farah, A. Caffeine Metabolism and Health Effects. In Coffee: Consumption and Health Implications; Farah, A., Ed.; Royal Society of Chemistry: London, UK, 2018; ISBN 978-1782620044. [Google Scholar]
- Cornish, H.H.; Christman, A.A. A study of the metabolism of theobromine, theophylline, and caffeine in man. J. Biol. Chem. 1957, 228, 315–323. [Google Scholar] [PubMed]
- Otomo, T. Changes of caffeine in the animal body. Nichidai Igaku Zasshi 1959, 18, 77–86. [Google Scholar]
- Schmidt, G.; Schoyerer, R. Determination of caffeine and its metabolites in urine. Deut. Z. Gesamte Gerichtl. Med. 1966, 57, 402–409. [Google Scholar]
- Czok, G.; Schmidt, B.; Lang, K. Influence of nutrition and habituation on the distribution of 8-C14-caffeine in the rat. Klin. Wochenschr 1968, 46, 1055–1059. [Google Scholar] [CrossRef] [PubMed]
- Bertoli, M.A.; Dragoni, G.; Rodari, A. Tissue distribution of labeled caffeine in mice. Med. Nucl. Radiobiol. Lat. 1968, 11, 231. [Google Scholar]
- Blanchard, J.; Sawers, S.J. The absolute bioavailability of caffeine in man. Eur. J. Clin. Pharmacol. 1983, 24, 93–98. [Google Scholar] [CrossRef] [PubMed]
- Lelo, A.; Birkett, D.J.; Robson, R.A.; Miners, J.O. Comparative pharmacokinetics of caffeine and its primary demethylated metabolites paraxanthine, theobromine and theophylline in man. Br. J. Clin. Pharmacol. 1986, 22, 1771–1782. [Google Scholar] [CrossRef] [PubMed]
- Mumford, G.K.; Benowitz, N.L.; Evans, S.M.; Kaminski, B.J.; Preston, K.L.; Sannerud, C.A.; Silverman, K.; Griffiths, R.R. Absorption rate of methylxanthines following capsules, cola and chocolate. Eur. J. Clin. Pharmacol. 1996, 51, 319–325. [Google Scholar] [CrossRef] [PubMed]
- Kuwazawa, T.; Seno, H.; Lee, X.-P.; Ishii, A.; Watanabe-Suzuki, K.; Sato, K.; Suzuki, O. Extraction of methylxanthines from human body fluids by solid-phase microextraction. Anal. Quim. Acta 1999, 387, 53–60. [Google Scholar] [CrossRef]
- Bonati, M.; Latini, R.; Galletti, F.; Young, J.F.; Tognoni, G.; Garattini, S. Caffeine disposition after oral doses. Clin. Pharmacol. Ther. 1982, 32, 98–106. [Google Scholar] [CrossRef] [PubMed]
- Krul, C.; Hageman, G. Analysis of urinary caffeine metabolites to assess biotransformation enzyme activities by reversed-phase high-performance liquid chromatography. J. Chromatogr. B Biomed. Sci. Appl. 1998, 709, 27–34. [Google Scholar] [CrossRef]
- Parke, D.V. The Biochemistry of Foreign Compounds; Parke, D.V., Ed.; Pergamon Press: London, UK, 1968; p. 139. [Google Scholar]
- Perera, V.; Gross, A.S.; McLachlan, A.J. Caffeine and paraxanthine HPLC assay for CYP1A2 phenotype assessment using saliva and plasma. Biom. Chromatogr. 2010, 24, 1136–1144. [Google Scholar] [CrossRef]
- Teekachunhatean, S.; Tosri, N.; Rojanasthien, M.; Srichairatanakool, S.; Sangdee, C. Pharmacokinetics of caffeine following a single administration of coffee enema versus oral coffee consumption in healthy male subjects. ISRN Pharmacol. 2013, 4. [Google Scholar] [CrossRef]
- Lang, R.; Dieminger, N.; Beusch, A.; Lee, Y.; Dunkel, A.; Suess, B.; Skurk, T.; Wahl, A.; Hauner, H.; Hofman, T. Bioappearance and pharmacokinetics of bioactives upon coffee consumption. Anal. Bioanal. Chem. 2013, 405, 8487–8503. [Google Scholar] [CrossRef] [PubMed]
- Martinez-Lopéz, S.; Sarriá, B.; Baeza, G.; Mateos, R.; Bravo-Clemente, L. Pharmacokinetics of caffeine and its metabolites in plasma and urine after consuming a soluble green/roasted coffee blend by healthy subjects. Food Res. Int. 2014, 64, 125–133. [Google Scholar] [CrossRef] [PubMed]
- Scheneider, H.; Ma, L.; Glatt, H. Extraction less method for the determination of urinary caffeine metabolites using high-performance liquid chromatography coupled with tandem mass spectrometry. J. Chromatogr. B Analyt. Technol. Biomed. Life Sci. 2003, 789, 227–237. [Google Scholar] [CrossRef]
- Rodopoulos, N.; Norman, A. Assessment of dimethylxanthine formation from caffeine in healthy adults: Comparison between plasma and saliva concentrations and urinary excretion of metabolites. Scand. J. Clin. Lab. Investig. 1996, 56, 259–268. [Google Scholar] [CrossRef] [PubMed]
- Rodopoulos, N.; Winsen, O.; Norman, A. Caffeine metabolism in patients with chronic liver disease. Scand. J. Clin. Lab. Investig. 1995, 55, 229–242. [Google Scholar] [CrossRef]
- Gilbert, S.G.; Stavric, B.; Klassen, R.D.; Rice, D.C. The fate of chronically consumed caffeine in the monkey (Macaca fascicularis). Fundam. Appl. Toxicol. 1985, 5, 578–587. [Google Scholar] [CrossRef]
- Kamimori, G.H.; Karyekar, C.S.; Otterstetter, R.; Cox, D.S.; Balkin, T.J.; Belenky, G.L.; Eddington, N.D. The rate of absorption and relative bioavailability of caffeine administered in chewing gum versus capsules to normal healthy volunteers. Int. J. Pharm. 2002, 234, 159–167. [Google Scholar] [CrossRef]
- Cohen, S.; Booth, G.H., Jr. Gastric acid secretion and lower-esophageal-sphincter pressure in response to coffee and caffeine. N. Engl. J. Med. 1975, 293, 897–899. [Google Scholar] [CrossRef] [PubMed]
- Kaplan, G.B.; Greenblatt, D.J.; Ehrenberg, B.L.; Goddard, J.E.; Cotreau, M.M.; Harmatz, J.S.; Shader, R.I. Dose-dependent pharmacokinetics and psychomotor effects of caffeine in humans. J. Clin. Pharmacol. 1997, 37, 693–703. [Google Scholar] [CrossRef] [PubMed]
- Perera, V.; Grossa, A.S.; Xu, H.; McLachlan, A.J. Pharmacokinetics of caffeine in plasma and saliva, and the influence of caffeine abstinence on CYP1A2 metrics. J. Pharm. Pharmacol. 2011, 63, 1161–1168. [Google Scholar] [CrossRef]
- Benowitz, N.L. Clinical pharmacology of caffeine. Annu. Rev. Med. 1990, 41, 277–288. [Google Scholar] [CrossRef] [PubMed]
- Martínez-López, S.; Sarriá, B.; Gómez-Juaristi, M.; Goya, L.; Mateos, R.; Bravo-Clemente, L. Theobromine, caffeine, and theophylline metabolites in human plasma and urine after consumption of soluble cocoa products with different methylxanthine contents. Food Res. Int. 2014, 63, 446–455. [Google Scholar] [CrossRef]
- Marks, V.; Kelly, J.F. Absorption of caffeine from tea, coffee, and coca cola. Lancet 1973, 1, 827. [Google Scholar] [CrossRef]
- Biederbick, W.; Joseph, G.; Rump, A.; Theisohn, M.; Klaus, W. Caffeine in saliva after peroral intake: Early sample collection as a possible source of error. Ther. Drug Monit. 1997, 19, 521–524. [Google Scholar] [CrossRef] [PubMed]
- Khanna, N.N.; Bada, H.S.; Somani, S.M. Use of salivary concentrations in the prediction of serum caffeine and theophylline concentrations in premature infants. J. Pediatr. 1980, 96, 494–499. [Google Scholar] [CrossRef]
- Newton, R.; Broughton, L.J.; Lind, M.J.; Morrisson, P.J.; Rogers, H.J.; Bradbrook, I.D. Plasma and salivary pharmacokinetics of caffeine in man. Eur. J. Clin. Pharmacol. 1981, 21, 45–52. [Google Scholar] [CrossRef] [PubMed]
- Soto, J.; Sacristan, A.; Alsar, M.J. Cerebrospinal fluid concentrations of caffeine following oral drug administration: Correlation with salivary and plasma concentrations. Ther. Drug Monit. 1994, 16, 108–110. [Google Scholar] [CrossRef] [PubMed]
- Caraco, Y.; Zylber-Katz, E.; Berry, E.M.; Levy, M. Caffeine pharmacokinetics in obesity and following significant weight reduction. Int. J. Obes. Relat. Metab. Disord. 1995, 19, 234–239. [Google Scholar] [PubMed]
- Abernethy, D.R.; Todd, E.L.; Schwartz, J.B. Caffeine disposition in obesity. Br. J. Clin. Pharmacol. 1985, 20, 61–66. [Google Scholar] [CrossRef]
- Kamimori, G.H.; Somani, S.M.; Knowlton, R.G.; Perkins, R.M. The effects of obesity and exercise on the pharmacokinetics of caffeine in lean and obese volunteers. Eur. J. Clin. Pharmacol. 1987, 31, 595–600. [Google Scholar] [CrossRef]
- Kimmel, C.A.; Kimmel, G.L.; White, C.G.; Grafton, T.F.; Young, J.F.; Nelson, C.J. Blood flow changes and conceptal development in pregnant rats in response to caffeine. Fundam. Appl. Toxicol. 1984, 4, 240–247. [Google Scholar] [CrossRef]
- Dorrbecker, S.H.; Kramer, P.A.; Dorrbecker, B.R.; Raye, J.R. Caffeine disposition in the pregnant rabbit. I. Pharmacokinetics following administration by intravenous bolus and continuous zero-order infusion. Dev. Pharmacol. Ther. 1988, 11, 109–117. [Google Scholar] [PubMed]
- Djordjevic, N.; Ghotbi, R.; Bertilsson, L.; Jankovic, S.; Aklillu, E. Induction of CYP1A2 by heavy coffee consumption in Serbs and Swedes. Eur. J. Clin. Pharmacol. 2008, 64, 381–385. [Google Scholar] [CrossRef]
- Gu, L.; Gonzalez, F.J.; Kalow, W.; Tang, B.K. Biotransformation of caffeine, paraxanthine, theobromine and theophylline by cDNA-expressed human CYP1A2 and CYP2E1. Pharmacogenetics 1992, 2, 73–77. [Google Scholar] [CrossRef] [PubMed]
- Miners, O.; Birkett, D.J. The use of caffeine as a metabolic probe for human drug metabolizing enzymes. Gen. Pharmacol. 1996, 27, 245–249. [Google Scholar] [CrossRef]
- Moura-Nunes, N.; Farah, A. Caffeine Consumption and Health; Romano, F.D., Russo, P.F., Eds.; Nova Science Publisher: New York, NY, USA, 2012; p. 1. [Google Scholar]
- Grant, D.M.; Campbell, M.E.; Tang, B.K.; Kalow, W. Biotransformation of caffeine by microsomes from human liver. Kinetics and inhibition studies. Biochem. Pharmacol. 1987, 36, 1251–1260. [Google Scholar] [CrossRef]
- Jodynis-Liebert, J.; Matuszewska, A. Effect of toluidines and dinitrotoluenes on caffeine metabolic ratio in rat. Toxicol. Lett. 1999, 104, 159–165. [Google Scholar] [CrossRef]
- Hakooz, N.M. Caffeine metabolic ratios for the in vivo evaluation of CYP1A2, N-acetyltransferase 2, xanthine oxidase and CYP2A6 enzymatic activities. Curr. Drug. Metab. 2009, 10, 329–338. [Google Scholar] [CrossRef] [PubMed]
- Bozikas, V.P.; Papakosta, M.; Niopas, I.; Karavatos, A.; Mirtsou-Fidani, V. Smoking impact on CYP1A2 activity in a group of patients with schizophrenia. Eur. Neuropsychopharmacol. 2004, 14, 39–44. [Google Scholar] [CrossRef]
- Ghotbi, R.; Christensen, M.; Roh, H.-K.; Ingelman-Sundberg, M.; Aklillu, E.; Bertilsson, L. Comparisons of CYP1A2 genetic polymorphisms, enzyme activity and the genotype-phenotype relationship in Swedes and Koreans. Eur. J. Clin. Pharmacol. 2007, 63, 537–546. [Google Scholar] [CrossRef]
- Aranda, J.V.; Louridas, A.T.; Vitullo, B.B.; Thom, P.; Aldridge, A.; Haber, R. Metabolism of theophylline to caffeine in human fetal liver. Science 1979, 206, 1319–1321. [Google Scholar] [CrossRef] [PubMed]
- Parsons, W.D.; Neims, A.H. Prolonged half-life of caffeine in healthy newborn infants. J. Pediatr. 1981, 98, 640–641. [Google Scholar] [CrossRef]
- Stravic, B.; Gilbert, S.G. Caffeine metabolism: a problem in extrapolating results from animal studies to humans. Acta Pharma Iugosl. 1990, 40, 475–489. [Google Scholar]
- Murphy, T.L.; McIvor, C.; Yap, A.; Cooksley, W.G.; Halliday, J.W.; Powell, L.W. The effect of smoking on caffeine elimination: Implications for its use as a semiquantitative test of liver function. Clin. Exp. Pharmacol. Physiol. 1988, 15, 9–13. [Google Scholar] [CrossRef] [PubMed]
- Kalow, W.; Tang, B.K. Caffeine as a metabolic probe: Exploration of the enzyme-inducing effect of cigarette smoking. Clin. Pharmacol. Ther. 1991, 49, 44–48. [Google Scholar] [CrossRef]
- Patwardhan, R.; Desmond, P.; Johnson, R.; Schenker, S. Impaired elimination of caffeine by oral contraceptive steroids. J. Lab. Clin. Med. 1980, 95, 603–608. [Google Scholar]
- Brazier, J.L.; Ritter, J.; Berland, M.; Khenfer, D.; Faucon, G. Pharmacokinetics of caffeine during and after pregnancy. Dev. Pharmacol. Ther. 1983, 6, 315–322. [Google Scholar] [CrossRef]
- Lamba, J.K.; Lin, Y.S.; Schuetz, E.G.; Thummel, K.E. Genetic contribution to variable human CYP3A-mediated metabolism. Adv. Drug Deliv. Rev. 2002, 54, 1271–1294. [Google Scholar] [CrossRef]
- Rasmussen, B.B.; Brix, T.H.; Kyvik, K.O.; Brøsen, K. The interindividual differences in the 3-demthylation of caffeine alias CYP1A2 is determined by both genetic and environmental factors. Pharmacogenetics 2002, 12, 473–478. [Google Scholar] [CrossRef]
- Guengerich, F.P. Cytochrome Pwhat have we learned and what are the future issues? Drug Metab. Rev. 2004, 36, 159–197. [Google Scholar] [CrossRef]
- Landi, M.T.; Sinha, R.; Lang, N.P.; Kadlubar, F.F. Human cytochrome P4501A2. IARC Sci. Publ. 1999, 148, 173–195. [Google Scholar]
- Chung, W.G.; Kang, J.H.; Park, C.S.; Cho, M.H.; Cha, Y.N. Effect of age and smoking on in vivo CYP1A2, flavin-containing monooxygenase, and xanthine oxidase activities in Koreans: Determination by caffeine metabolism. Clin. Pharmacol. Ther. 2000, 67, 258–266. [Google Scholar] [CrossRef] [PubMed]
- Begas, E.; Kouvaras, E.; Tsakalof, A.; Papakosta, S.; Asprodini, E.K. In vivo evaluation of CYP1A2, CYP2A6, NAT-2 and xanthine oxidase activities in a Greek population sample by the RP-HPLC monitoring of caffeine metabolic ratios. Biomed. Chromatogr. 2007, 21, 190–200. [Google Scholar] [CrossRef] [PubMed]
- Relling, M.V.; Lin, J.S.; Avers, G.D.; Evans, W.E. Racial and gender differences in N-acetyltransferase, xanthine oxidase, and CYP1A2 activities. Clin. Pharmacol. Ther. 1992, 52, 643–658. [Google Scholar] [CrossRef] [PubMed]
- Sachse, C.; Brockmoller, J.; Bauer, S.; Roots, I. Functional significance of a C-->A polymorphism in intron 1 of the cytochrome P450 CYP1A2 gene tested with caffeine. Br. J. Clin. Pharmacol. 1999, 47, 445–449. [Google Scholar] [CrossRef] [PubMed]
- Ueng, Y.F.; Jan, W.C.; Lin, L.C.; Chen, T.L.; Guengerich, F.P.; Chen, C.F. The alkaloid rutaecarpine is a selective inhibitor of cytochrome P450 1A in mouse and human liver microsomes. Drug Metab. Dispos. 2002, 30, 349–353. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Tu, J.H.; He, Y.J.; Zhang, W.; Wang, G.; Tan, Z.R.; Zhou, G.; Fan, L.; Zhou, H.H. Effect of sodium tanshinone II A sulfonate on the activity of CYP1A2 in healthy volunteers. Xenobiotica 2009, 39, 508–513. [Google Scholar] [CrossRef]
- Wang, X.; Lee, W.Y.; Or, P.M.; Yeung, J.H. Effects of major tanshinones isolated from Danshen (Salvia miltiorrhiza) on rat CYP1A2 expression and metabolism of model CYP1A2 probe substrates. Phytomedicine 2009, 16, 712–725. [Google Scholar] [CrossRef]
- Wang, X.; Cheung, C.M.; Lee, M.Y.; Or, P.M.; Yeung, J.H. Major tanshinones of Danshen (Salvia miltiorrhiza) exhibit different modes of inhibition on human CYP1A2, CYP2C9, CYP2E1 and CYP3A4 activities in vitro. Phytomedicine 2010, 17, 868–875. [Google Scholar] [CrossRef]
- Scott, N.R.; Chakraborty, J.; Marks, V. Urinary metabolites of caffeine in pregnant women. Br. J. Clin. Pharmacol. 1986, 22, 475–478. [Google Scholar] [CrossRef]
- Bologa, M.; Tang, B.; Klein, J.; Tesoro, A.; Koren, G. Pregnancy-induced changes in drug metabolism in epileptic women. J. Pharmacol. Exp. Ther. 1991, 257, 735–740. [Google Scholar] [PubMed]
- Cornelis, M.C.; El-Sohemy, A.; Kabagambe, E.K.; Campos, H. Coffee, CYP1A2 genotype, and risk of myocardial infarction. JAMA 2006, 295, 1135–1141. [Google Scholar] [CrossRef] [PubMed]
- Djordjevic, N.; Carrillo, J.A.; Gervasini, G.; Jankovic, S.; Aklillu, E. In vivo evaluation of CYP2A6 and xanthine oxidase enzyme activities in the Serbian population. Eur. J. Clin. Pharmacol. 2010, 66, 571–578. [Google Scholar] [CrossRef] [PubMed]
- Palatini, P.; Ceolotto, G.; Ragazzo, F.; Dorigatti, F.; Saladini, F.; Papparella, I.; Mos, L.; Zanata, G.; Santonastaso, M. CYP1A2 genotype modifies the association between coffee intake and the risk of hypertension. J. Hypertens. 2009, 27, 1594–1601. [Google Scholar] [CrossRef] [PubMed]
- Rodenburg, E.M.; Eijgelsheim, M.; Geleijnse, J.M.; Amin, N.; van Duijn, C.M.; Hofman, A.; Uitterlinden, A.G.; Stricker, B.H.; Visser, L.E. CYP1A2 and coffee intake and the modifying effect of sex, age, and smoking. Am. J. Clin. Nutr. 2012, 96, 182–187. [Google Scholar] [CrossRef]
- Cornelis, M.C.; Monda, K.L.; Yu, K.; Paynter, N.; Azzato, E.M.; Bennett, S.N.; Berndt, S.I.; Boerwinkle, E.; Chanock, S.; Chatterjee, N. Genome-wide meta-analysis identifies regions on 7p21 (AHR) and 15q24 (CYP1A2) as determinants of habitual caffeine consumption. PLoS Genet. 2011, 7, e1002033. [Google Scholar] [CrossRef] [PubMed]
- Tantcheva-Poór, I.; Zaigler, M.; Rietbrock, S.; Fuhr, U. Estimation of cytochrome P-450 CYP1A2 activity in 863 healthy Caucasians using a saliva-based caffeine test. Pharmacogenetics 1999, 9, 131–144. [Google Scholar]
- Carrillo, J.A.; Benitez, J. CYP1A2 activity, gender and smoking, as variables influencing the toxicity of caffeine. Br. J. Clin. Pharmacol. 1996, 41, 605–608. [Google Scholar] [CrossRef]
- Denden, S.; Bouden, B.; Haj Khelil, A.; Ben Chibani, J.; Hamdaoui, M.H. Gender and ethnicity modify the association between the CYP1A2 rs762551 polymorphism and habitual coffee intake: Evidence from a meta-analysis. Genet. Mol. Res. 2016, 15. [Google Scholar] [CrossRef]
- Arnaud, M.J. Identification, kinetic and quantitative study of [2-14C] and [1-Me-14C] caffeine metabolites in rat’s urine by chromatographic separations. Biochem. Med. 1976, 16, 67–76. [Google Scholar] [CrossRef]
- Tang-Liu, D.D.S.; Williams, R.L.; Reigelman, S. Disposition of caffeine and its metabolites in man. J. Pharmacol. Exp. Ther. 1983, 224, 180–185. [Google Scholar] [PubMed]
- Callahan, H.M.; Robertson, R.S.; Arnaud, M.J.; Branfman, A.R.; McComish, M.F.; Yesair, D.H. Human metabolism of [l-methyl-14C]-and [2-14C] caffeine after oral administration. Drug Metab. Dispos. 1982, 10, 417–423. [Google Scholar] [PubMed]
- Freedman, N.; Park, Y.; Abnet, C.C.; Hollenbeck, A.R.; Sinha, R. Association of coffee drinking with total and cause-specific mortality. N. Engl. J. Med. 2012, 366, 1891–1894. [Google Scholar] [CrossRef] [PubMed]
- O’Keefe, J.H.; Bhatti, S.K.; Patil, H.R.; DiNicolantonio, J.J.; Lucan, S.C.; Lavie, C.J. Effects of habitual coffee consumption on cardiometabolic disease, cardiovascular health, and all-cause mortality. J. Am. Coll. Cardiol. 2013, 62, 1043–1051. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Sui, X.; Lavie, C.J.; Hebert, J.R.; Earnest, C.P.; Zhang, J.; Blair, S.N. Association of coffee consumption with all-cause and cardiovascular disease mortality. Mayo Clin. Proc. 2013, 88, 1066–1074. [Google Scholar] [CrossRef] [PubMed]
- Crippa, A.; Discacciati, A.; Larsson, S.C.; Wolk, A.; Orsini, N. Coffee consumption and mortality from all causes, cardiovascular disease, and cancer: A dose-response meta-analysis. Am. J. Epidemiol. 2014, 180, 763–765. [Google Scholar] [CrossRef]
- Loftfield, E.; Freedman, N.D.; Graubard, B.I.; Guertin, K.A.; Black, A.; Huang, W.-Y.; Shebl, F.M.; Mayne, S.T.; Sinha, R. Association of coffee consumption with overall and cause-specific mortality in a large US prospective cohort study. Am. J. Epidemiol. 2015, 182, 1010–1022. [Google Scholar] [CrossRef]
- Ding, M.; Satija, A.; Bhupathiraju, S.N.; Hu, Y.; Sun, Q.; Han, J.; Lopez-Garcia, E.; Willet, W.; van Dam, R.M.; Hu, F.A. Association of coffee consumption with total and cause-specific mortality in 3 large prospective cohorts. Circulation 2015, 132, 2305–2315. [Google Scholar] [CrossRef] [PubMed]
- Grosso, G.; Micek, A.; Godos, J.; Sciacca, S.; Pajak, A.; Martínez-González, M.A.; Giovannucci, E.L.; Galvano, F. Coffee consumption and risk of all-cause, cardiovascular, and cancer mortality in smokers and non-smokers: A dose-response meta-analysis. Eur. J. Epidemiol. 2016, 31, 1191–1205. [Google Scholar] [CrossRef]
- Park, S.Y.; Freedman, N.D.; Haiman, C.A.; Marchand, L.L.; Wilkens, L.R.; Setiawan, V.W. Association of coffee consumption with total and cause-specific mortality among nonwhite populations. Ann. Intern. Med. 2017, 167, 228–235. [Google Scholar] [CrossRef]
- Poole, R.; Kennedy, O.J.; Roderick, P.; Fallowfield, J.A.; Hayes, P.C.; Parkes, J. Coffee consumption and health: Umbrella review of meta-analyses of multiple health outcomes. BMJ 2017, 359, j5024. [Google Scholar] [CrossRef] [PubMed]
- Grosso, G.; Godos, J.; Galvano, F.; Giovannucci, E.L. Coffee, caffeine, and health outcomes: An umbrella review. Annu. Rev. Nutr. 2017, 37, 131–156. [Google Scholar] [CrossRef] [PubMed]
- McPherson, P.S.; Kim, Y.K.; Valdivia, H.; Knudson, C.M.; Takekura, H.; Franzini-Armstrong, C.; Coronado, R.; Campbell, K.P. The brain ryanodine receptor: A caffeine-sensitive calcium release channel. Neuron 1991, 7, 17–25. [Google Scholar] [CrossRef]
- McLellan, T.M.; Caldwell, J.A.; Lieberman, H.R. A review of caffeine’s effects on cognitive, physical and occupational performance. Neurosci. Biobehav. Rev. 2016, 71, 294–312. [Google Scholar] [CrossRef] [PubMed]
- Gliottoni, R.C.; Meyers, J.R.; Arrigrimsson, S.A.; Boglio, S.P.; Motl, R.W. Effect of caffeine on quadriceps muscle pain during acute cycling exercise in low versus high caffeine consumers. Int. J. Sports Nutr. Exerc. Metab. 2009, 19, 150–161. [Google Scholar] [CrossRef]
- Watson, J. Encyclopedia of Food Sciences and Nutrition; Caballero, B., Finglas, P.M., Toldra, F., Eds.; Academic Press: Cambridge, UK, 2003; p. 745. [Google Scholar]
- Fredholm, B.B. Are methylxanthine effects due to antagonism of endogenous adenosine? Trends Pharmacol. Sci. 1980, 1, 129–132. [Google Scholar] [CrossRef]
- Fredholm, B.B.; Svenningsson, P. Adenosine-dopamine interactions: Development of a concept and some comments on therapeutic possibilities. Neurology 2003, 61, 5–9. [Google Scholar] [CrossRef]
- Nehlig, A.; Daval, J.L.; Debry, G. Caffeine and the central nervous system: Mechanisms of action, biochemical, metabolic and psychostimulant effects. Brain Res. Rev. 1992, 17, 139–170. [Google Scholar] [CrossRef]
- Olson, C.A.; Thornton, J.A.; Adam, G.E.; Lieberman, H.R. Effects of 2 adenosine antagonists, quercetin and caffeine, on vigilance and mood. J. Clin. Psychopharmacol. 2010, 30, 573–578. [Google Scholar] [CrossRef]
- Fisone, G.; Borgvist, A.; Usiello, A. Caffeine as a psychomotor stimulant: Mechanism of action. Cell. Mol. Life Sci. 2004, 61, 857–872. [Google Scholar] [CrossRef]
- Einöther, S.J.L.; Giesbrecht, T. Caffeine as an attention enhancer: Reviewing existing assumptions. Psychopharmaco 2013, 225, 251–274. [Google Scholar] [CrossRef] [PubMed]
- Nehlig, A. Is caffeine a cognitive enhancer? J. Alzheimers Dis. 2010, 20, 85–94. [Google Scholar] [CrossRef] [PubMed]
- Nehlig, A. Effects of coffee/caffeine on brain health and disease: What should I tell my patients? Pract. Neurol. 2016, 16, 89–95. [Google Scholar] [CrossRef] [PubMed]
- Nawrot, P.; Jordan, S.; Eastwood, J.; Rotstein, J.; Hugenholtz, A.; Feeley, M. Effects of caffeine on human health. Food Addit. Contam. 2003, 20, 1–30. [Google Scholar] [CrossRef] [PubMed]
- Goldstein, E.R.; Ziegenfuss, T.; Kalman, D.; Kreider, R.; Campbell, B.; Wilborn, C.; Taylor, L.; Willoughby, D.; Stout, J.; Graves, B.S.; et al. International society of sports nutrition position stand: Caffeine and performance. J. Int. Soc. Sports Nutr. 2010, 7, 5. [Google Scholar] [CrossRef] [PubMed]
- Sinclair, C.J.; Geiger, J.D. Caffeine use in sports. A pharmacological review. J. Sports. Med. Phys. Fitness 2000, 40, 71–79. [Google Scholar] [PubMed]
- Van Soeren, M.H.; Sathasivam, P.; Spriet, L.L.; Graham, T.E. Caffeine metabolism and epinephrine responses during exercise in users and nonusers. J. Appl. Physiol. 1985, 75, 805–812. [Google Scholar] [CrossRef]
- European Food Safety Authority—(EFSA Panel on Dietetic Products, Nutrition and Allergies). Scientific Opinion on the Substantiation of Health Claims Related to Caffeine and Increase in Physical Performance during Short-Term High-Intensity Exercise, Increase in Endurance Performance, Increase in Endurance Capacity and Reduction in the Rated Perceived Exertion/Effort during Exercise; European Food Safety Authority: Parma, Italy, 2011.
- Folmer, B.; Farah, A.; Jones, L.; Fogliano, V. Human Wellbeing—Sociability, Performance, and Health. In The Craft and Science of Coffee; Folmer, B., Ed.; Elsevier: Amsterdam, The Netherlands, 2017; pp. 493–520. [Google Scholar]
- Hogervost, E.; Bandelow, S.; Schmitt, J.; Jentjens, R.; Oliveira, M.; Allgrove, J.; Carter, T.; Gleeson, M. Caffeine improves physical and cognitive performance during exhaustive exercise. Med. Sci. Sports Exerc. 2008, 40, 1841–1851. [Google Scholar] [CrossRef]
- Ballard, R.A.; Truog, W.E.; Cnaan, A.; Martin, R.J.; Ballard, P.L.; Merrill, J.D. Inhaled nitric oxide in preterm infants undergoing mechanical ventilation. N. Engl. J. Med. 2006, 355, 343–353. [Google Scholar] [CrossRef]
- Dallas, C.; Gerbi, A.; Tenca, G.; Juchaux, F.; Bernard, F.X. Lipolytic effect of a polyphenolic citrus dry extract of red orange, grapefruit, orange (SINETROL) in human body fat adipocytes. Mechanism of action by inhibition of cAMP-phosphodiesterase (PDE). Phytomedicine 2008, 15, 783–792. [Google Scholar] [CrossRef]
- Catlin, D.H.; Kammerer, R.C.; Hatton, C.K.; Severa, M.H.; Merdink, J.L. Analytical chemistry at the Games of the XXIIIrd Olympiad in Los Angeles, 1984. Clin. Chem. 1984, 33, 319–327. [Google Scholar]
- World Anti-Doping Agency—WADA. Available online: http://www.wada-ama.org (accessed on 1 February 2019).
- World Anti-Doping Agency (WADA). The 2009 Monitoring Program. Available online: www.wada-ama.org (accessed on 1 February 2019).
- Monteiro, J.P.; Alves, G.M.; Oliveira, P.F.; Silva, B.M. Structure-bioactivity relationships of methylxanthines: Trying to make sense of all the promises and the drawbacks. Molecules 2016, 21, 974. [Google Scholar] [CrossRef] [PubMed]
- Cappelletti, S.; Daria, P.; Sani, G.; Aromatario, M. Caffeine: Cognitive and physical performance enhancer or psychoactive drug? Curr. Neuropharmacol. 2015, 13, 71–88. [Google Scholar] [CrossRef] [PubMed]
- Costa, M.S.; Botton, P.H.; Mioranzza, S.; Souza, D.O.; Porciúncula, L.O. Caffeine prevents age-associated recognition memory decline and changes brain-derived neurotrophic factor and tyrosine kinase receptor (TrkB) content in mice. Neuroscience 2008, 153, 1071–1078. [Google Scholar] [CrossRef] [PubMed]
- Costenla, A.R.; Cunha, R.; de Mendonça, A. Caffeine, adenosine receptors, and synaptic plasticity. J. Alzheimers. 2010, 20, 25–34. [Google Scholar] [CrossRef]
- Stefanello, N.; Spanevello, R.M.; Passamontic, S.; Porciúncula, L.; Bonane, C.D.; Olabiyif, A.A.; Rocha, J.B.; Assmanna, C.E.; Morscha, V.M.; Schetingera, M.R.C. Coffee, caffeine, chlorogenic acid, and the purinergic system. Food Chem. Toxicol. 2019, 123, 298–313. [Google Scholar] [CrossRef] [PubMed]
- Shi, X.; Dalal, N.S.; Jain, A.C. Antioxidant behaviour of caffeine: Efficient scavenging of hydroxyl radicals. Food Chem. Toxicol. 1991, 29, 1–6. [Google Scholar] [CrossRef]
- Varma, S.D.; Hegde, K.R.; Kovtun, S. Oxidative stress in lens in vivo: Inhibitory effect of caffeine. A preliminary report. Mol. Vis. 2010, 23, 501–505. [Google Scholar]
- Lee, C. Antioxidant ability of caffeine and its metabolites based on the study of oxygen radical absorbing capacity and inhibition of LDL peroxidation. Clin. Chim. Acta 2000, 295, 141–154. [Google Scholar] [CrossRef]
- Devasagayam, P.; Kesavan, P.C. Radioprotective and antioxidant action of caffeine: Mechanistic considerations. Indian J. Exp. Biol. 1996, 34, 291–297. [Google Scholar]
- Gomez-Ruiz, J.A.; Leake, D.S.; Ames, J.M. In vitro antioxidant activity of coffee compounds and their metabolites. J. Agric. Food Chem. 2007, 55, 6962–6969. [Google Scholar] [CrossRef] [PubMed]
- Moura-Nunes, N.; Perrone, D.; Farah, A.; Donangelo, C.M. The increase in human plasma antioxidant capacity after acute coffee intake is not associated with endogenous non-enzymatic antioxidant components. Int. Food Sci. Nutr. 2009, 60, 173–181. [Google Scholar] [CrossRef] [PubMed]
- Buscemi, S.; Batsis, J.A.; Arcoleo, G.; Verga, S. Coffee and endothelial function: A battle between caffeine and antioxidants? Eur. J. Clin. Nutr. 2010, 64, 1242–1243. [Google Scholar] [CrossRef] [PubMed]
- Vicente, S.J.V.; Queiroz, Y.S.; Gotlieb, S.L.D.; Torres, A.F.S. Stability of phenolic compounds and antioxidant capacity of regular and decaffeinated coffees. Braz. Arch. Biol. Technol. 2014, 57, 110. [Google Scholar] [CrossRef]
- Frost-Meyer, N.J.; Logomarsino, J.V. Impact of coffee components on inflammatory markers: A review. J. Func. Foods 2012, 4, 819–830. [Google Scholar] [CrossRef]
- Lee, I.A.; Kamba, A.; Low, D.; Mizoguchi, E. Novel methylxanthine derivative-mediated anti-inflammatory effects in inflammatory bowel disease. World J. Gastroenterol. 2014, 20, 1127–1138. [Google Scholar] [CrossRef] [PubMed]
- Daly, J.W. Caffeine analogs: Biomedical impact. Cell. Mol. Life Sci. 2007, 64, 2153–2169. [Google Scholar] [CrossRef] [PubMed]
- Tauler, P.; Martinez, S.; Martinez, P.; Lozano, L. Effects of caffeine supplementation on plasma and blood mononuclear cell interleukin-10 levels after exercise. Hum. Kinet. J. 2015, 26, 8–16. [Google Scholar] [CrossRef]
- Ouyang, W.; Rutz, S.; Crellin, N.K.; Valdez, P.A.; Hymowitz, S.G. Regulation and functions of the IL-10 family of cytokines in inflammation and disease. Annu. Rev. Immunol. 2010, 29, 71–109. [Google Scholar] [CrossRef]
- Rittera, M.; Hohenberger, K.; Altera, P.; Herzuma, M.; Tebbe, J.; Maisch, M. Caffeine inhibits cytokine expression in lymphocytes. Cytokine 2005, 30, 177–181. [Google Scholar] [CrossRef]
- Horrigan, L.A.; Kelly, J.P.; Connor, T.J. Caffeine suppresses TNF-α production via activation of the cyclic AMP/protein kinase A pathway. Int. Immunopharmacol. 2004, 4, 1409–1417. [Google Scholar] [CrossRef] [PubMed]
- Antonio, A.G.; Iorio, N.L.P.; Pierro, V.S.S.; Candreva, M.S.; Farah, A.; dos Santos, K.R.N.; Maia, L.C. Inhibitory properties of Coffea canephora extract against oral bacteria and its effect on demineralisation of deciduous teeth. Arch. Oral Biol. 2011, 56, 556–564. [Google Scholar] [CrossRef] [PubMed]
- Van Loreven, C.; Broukal, Z.; Oganessian, E. Functional foods/ingredients and dental caries. Eur. J. Nutr. 2012, 51, 15–25. [Google Scholar] [CrossRef] [PubMed]
- Sharma, R.; Reddy, V.K.L.; Prashant, G.M.; Ojha, V.; Kumar, N.P.G. Antimicrobial and anti-adherence activity of various combinations of coffee-chicory solutions on Streptococcus mutans: An in-vitro study. J. Oral. Maxillofac. Pathol. 2014, 18, 201–206. [Google Scholar] [CrossRef] [PubMed]
- Ibrahim, S.A.; Salameh, M.M.; Phetsomphou, H.; Yang, H.; Seo, C.W. Application of caffeine, 1,3,7-trimethylxanthine, to control Escherichia coli O157:H7. Food Chem. 2006, 99, 645–650. [Google Scholar] [CrossRef]
- Maia, L.; de Mendonça, A. Does caffeine intake protect from Alzheimer’s disease? Eur. J. Neurol. 2002, 9, 377–382. [Google Scholar] [CrossRef] [PubMed]
- Eskelinen, M.H.; Ngandu, T.; Tuomilehto, J.; Soininena, H.; Kivipelto, M.J. Midlife coffee and tea drinking and the risk of late-life dementia: A population-based CAIDE study. J. Alzheimers Dis. 2009, 16, 85–91. [Google Scholar] [CrossRef] [PubMed]
- Arendash, G.W.; Cao, C. Caffeine and coffee as therapeutics against Alzheimer’s disease. J. Alzheimers Dis. 2010, 20, 117–126. [Google Scholar] [CrossRef]
- Costa, J.; Lunet, N.; Santos, C.; Santos, J.; Vaz-Cameiro, A. Caffeine exposure and the risk of Parkinson’s disease: A systemic review and meta-analysis of observational studies. J. Alzheimers Dis. 2010, 20, 221–238. [Google Scholar] [CrossRef] [PubMed]
- Postuma, R.B.; Lang, A.E.; Munhoz, R.P.; Charland, K.; Pelletier, A.; Moscovich, M.; Filla, L.; Zanatta, D.; Rios Romenets, S.; Altman, R. Caffeine for treatment of Parkinson disease: A randomized controlled trial. Neurology 2012, 79, 651–658. [Google Scholar] [CrossRef] [PubMed]
- Hashimoto, M.; Rockenstein, E.; Crews, L.; Masliah, E. Role of protein aggregation in mitochondrial dysfunction and neurodegeneration in Alzheimer’s and Parkinson’s diseases. Neuromol. Med. 2003, 4, 21–36. [Google Scholar] [CrossRef]
- Eskelinen, M.H.; Kivipelto, M. Caffeine as a protective factor in dementia and Alzheimer’s disease. J. Alzheimers. Dis. 2010, 20, 167–174. [Google Scholar] [CrossRef] [PubMed]
- Hardy, J.; Gwinn-Hardy, K. Genetic classification of primary neurodegenerative disease. Science 1998, 282, 1075–1079. [Google Scholar] [CrossRef] [PubMed]
- Dauer, W.; Przedborski, S. Parkinson’s disease: Mechanisms and models. Neuron 2003, 39, 889–909. [Google Scholar] [CrossRef]
- Chen, J.-F. Caffeine and Parkinson’s disease: From molecular targets to epidemiology to clinic trials. In Coffee: Consumption and Health Implications; Farah, A., Ed.; Royal Society of Chemistry: London, UK, 2019; ISBN 978-1782620044. [Google Scholar]
- Ascherio, A.; Zhang, S.M.; Hernan, M.A.; Kawachi, I.; Colditz, G.A.; Speizer, F.E.; Willett, W.C. Prospective study of caffeine consumption and risk of Parkinson’s disease in men and women. Ann. Neurol. 2001, 50, 56–63. [Google Scholar] [CrossRef] [PubMed]
- Ascherio, A.; Weisskopf, M.G.; O’Reilly, E.J.; McCullough, M.L.; Calle, E.E.; Rodriguez, C.; Thun, M.J. Coffee consumption, gender, and Parkinson’s disease mortality in the cancer prevention study II cohort: The modifying effects of estrogen. Am. J. Epidemiol. 2004, 160, 977–984. [Google Scholar] [CrossRef] [PubMed]
- Ascherio, A.; Chen, H.; Schwarzschild, M.A.; Zhang, S.M.; Colditz, G.A.; Speizer, F.E. Caffeine, postmenopausal estrogen, and risk of Parkinson’s disease. Neurology 2003, 60, 790–795. [Google Scholar] [CrossRef] [PubMed]
- Blum, D.; Farah, A.; Lopes, L.V. Coffee and Alzheimer’s Disease. In Coffee: Consumption and Health Implications; Farah, A., Ed.; Royal Society of Chemistry: London, UK, 2019; ISBN 978-1782620044. [Google Scholar]
- Checler, F. Processing of the beta-amyloid precursor protein and its regulation in Alzheimer’s disease. J. Neurochem. 1995, 65, 1431–1444. [Google Scholar] [CrossRef] [PubMed]
- Ittner, L.M.; Ke, Y.D.; Delerue, F.; Bi, N.; Gladbach, A.; van Eersel, J.; Wolfing, H.; Chieng, B.C.; Christie, M.J.; Napier, I.A. Dendritic function of tau mediates amyloid-beta toxicity in Alzheimer’s disease mouse models. Cell 2010, 142, 387–397. [Google Scholar] [CrossRef]
- Arendash, G.W.; Schleif, W.; Rezai Zadeh, K.; Jackson, E.K.; Zacharia, L.C.; Cracchiolo, J.R.; Shippy, D.; Tan, J. Caffeine protects Alzheimer’s mice against cognitive impairment and reduces brain β-amyloid production. Neuroscience 2006, 142, 941–952. [Google Scholar] [CrossRef]
- Waite, M. Treatment for Alzheimer’s disease: Has anything changed? Aust. Prescr. 2015, 38, 60–63. [Google Scholar] [CrossRef] [PubMed]
- Liu, Q.P.; Wu, Y.F.; Cheng, H.Y.; Xia, T.; Ding, H.; Wang, H.; Wang, Z.M.; Xu, Y. Habitual coffee consumption and risk of cognitive decline/dementia: A systematic review and meta-analysis of prospective cohort studies. Nutrition 2016, 32, 628–636. [Google Scholar] [CrossRef] [PubMed]
- Arendash, G.W.; Mori, T.; Cao, C.; Mamcarz, M.; Runfeldt, M.; Dickson, A.; Rezai-Zadeh, K.; Tane, J.; Citron, B.A.; Lin, X.; et al. Caffeine reverses cognitive impairment and decreases brain amyloid-beta levels in aged Alzheimer’s disease mice. J. Alzheimers Dis. 2009, 17, 661–680. [Google Scholar] [CrossRef] [PubMed]
- Gelber, R.P.; Petrovitch, H.; Masaki, K.H.; Ross, G.W.; White, L.R. Coffee intake in midlife and risk of dementia and its neuropathologic correlates. J. Alzheimer’s Dis. 2011, 23, 607–615. [Google Scholar] [CrossRef] [PubMed]
- Ruhl, C.E.; Everhart, J.E. Coffee and tea consumption are associated with a lower incidence of chronic liver disease in the United States. Gastroenterology 2005, 129, 1928–1936. [Google Scholar] [CrossRef] [PubMed]
- Casiglia, E.; Spolaore, P.; Inocchio, G. Unexpected effects of coffee consumption on liver enzymes. Eur. J. Epidemiol. 1993, 9, 293–297. [Google Scholar] [CrossRef]
- Jaruvongvanich, V.; Sanguankeo, A.; Klomjit, N.; Upala, S. Effects of caffeine consumption in patients with chronic hepatitis C: A systematic review and meta-analysis. Clin. Res. Hepatol. Gastroenterol. 2017, 41, 46–55. [Google Scholar] [CrossRef] [PubMed]
- Shen, H.; Rodriguez, A.C.; Shiani, A.; Lipka, S.; Shahzad, G.; Kumar, A.; Mustacchia, P. Association between caffeine consumption and nonalcoholic fatty liver disease: A systemic review and meta-analysis. Ther. Adv. Gastroenterol. 2016, 9, 113–120. [Google Scholar] [CrossRef]
- Machado, S.R.; Parise, E.R.; Carvalho, L. Coffee has hepatoprotective benefits in Brazilian patients with chronic hepatitis C even in lower daily consumption than in American and European populations. Braz. J. Infect. Dis. 2014, 18, 170–176. [Google Scholar] [CrossRef]
- Loomis, D.; Guyton, K.Z.; Grosse, Y.; Lauby-Secretan, B.; El Ghissassi, F.; Bouvard, V.; Benbrahim-Tallaa, L.; Guha, N.; Mattock, H.; Straif, K. Carcinogenicity of drinking coffee, mate, and very hot beverages. Lancet Oncol. 2016, 17, 877–878. [Google Scholar] [CrossRef]
- La Vecchia, C. Coffee, liver enzymes, cirrhosis and liver cancer. J. Hepatol. 2005, 42, 444–446. [Google Scholar] [CrossRef] [PubMed]
- Klatsky, A.L.; Morton, C.; Udaltsova, N.; Friedman, G.D. Coffee, cirrhosis, and transaminase enzymes. Arch. Intern. Med. 2006, 166, 1190–1195. [Google Scholar] [CrossRef] [PubMed]
- Larsson, S.C.; Wolk, A. Coffee consumption and risk of liver cancer: A meta-analysis. Gastroenterology 2007, 132, 1740–1745. [Google Scholar] [CrossRef] [PubMed]
- Bravi, F.; Bosetti, C.; Tavani, A.; Bagnardi, V.; Gallus, S.; Negri, E. Coffee drinking and hepatocellular carcinoma risk: A meta-analysis. Hepatology 2007, 46, 430–435. [Google Scholar] [CrossRef] [PubMed]
- Gressner, O.A.; Lahme, B.; Rehbein, K.; Siluschek, M.; Weiskirchen, R.; Gressner, A.M. Pharmacological application of caffeine inhibits TGF-β-stimulated connective tissue growth factor expression in hepatocytes via PPARγ and SMAD2/3-dependent pathways. J. Hepatol. 2008, 49, 758–767. [Google Scholar] [CrossRef] [PubMed]
- Shim, S.G.; Jun, D.W.; Kim, E.K.; Saeed, W.K.; Lee, K.N.; Lee, H.L.; Lee, O.Y.; Choi, H.S.; Yoon, B.C. Caffeine attenuates liver fibrosis via defective adhesion of hepatic stellate cells in cirrhotic model. JGH 2013, 28, 1877–1884. [Google Scholar] [CrossRef]
- Saaksjarvi, K.; Knekt, P.; Rissanen, H.; Laaksonen, M.A.; Reunanen, A.; Mannisto, S. Prospective study of coffee consumption and risk of Parkinson’s disease. Eur. J. Clin. Nutr. 2008, 62, 908–915. [Google Scholar] [CrossRef] [PubMed]
- Kyrozis, A.; Ghika, A.; Stathopoulos, P.; Vassilopoulos, D.; Trichopoulos, D.; Trichopoulou, A. Dietary and lifestyle variables in relation to incidence of Parkinson’s disease in Greece. Eur. J. Epidemiol. 2013, 28, 67–77. [Google Scholar] [CrossRef]
- Kenborg, L.; Lassen, C.F.; Ritz, B.; Andersen, K.K.; Christensen, J.; Schernhammer, E.S.; Hansen, J.; Wermuth, L.; Rod, N.H.; Olsen, J.H. Lifestyle, family history, and risk of idiopathic Parkinson disease: A large Danish case-control study. Am. J. Epidemiol. 2015, 181, 808–816. [Google Scholar] [CrossRef]
- Qi, H.; Li, S. Dose-response meta-analysis on coffee, tea and caffeine consumption with risk of Parkinson’s disease. Geriatr. Gerontol. Int. 2014, 14, 430–439. [Google Scholar] [CrossRef]
- Lindsay, J.; Laurin, D.; Verreault, R.; Hebert, R.; Helliwell, B.; Hill, G.B.; McDowell, I. Risk factors for Alzheimer’s disease: A prospective analysis from the Canadian Study of Health and Aging. Am. J. Epidemiol. 2002, 156, 445–453. [Google Scholar] [CrossRef] [PubMed]
- Panza, F.; Solfrizzi, V.; Barulli, M.R.; Bonfiglio, C.; Guerra, V.; Osella, A.; Seripa, D.; Sabbà, C.; Pilotto, A.; Logroscino, G. Coffee, tea, and caffeine consumption and prevention of late-life cognitive decline and dementia: A systematic review. J. Nutr. Health Aging 2015, 19, 313–328. [Google Scholar] [CrossRef] [PubMed]
- Kenned, O.J.; Roderick, P.; Buchanan, R.; Fallowfield, J.A.; Hayes, P.C.; Parkes, J. Coffee, including caffeinated and decaffeinated coffee, and the risk of hepatocellular carcinoma: A systematic review and dose-response meta-analysis. BMJ Open 2017, 7, e013739. [Google Scholar] [CrossRef] [PubMed]
- Wijarnpreecha, K.; Thongprayoon, C.; Ungprasert, P. Impact of caffeine in hepatitis C virus infection: A systematic review and meta-analysis. Eur. J. Gastroenterol. Hepatol. 2017, 29, 17–22. [Google Scholar] [CrossRef] [PubMed]
- Ruhl, C.E.; Everhart, J.E. Coffee and caffeine consumption reduce the risk of elevated serum alanine aminotransferase activity in the United States. Gastroenterology 2005, 128, 24–32. [Google Scholar] [CrossRef] [PubMed]
- Corrao, G.; Zambon, A.; Bagnardi, V.; D’Amicis, A.; Klatsky, A. Coffee, caffeine, and the risk of liver cirrhosis. Ann. Epidemiol. 2001, 11, 458–465. [Google Scholar] [CrossRef]
- Food and Drug Administration—FDA. Select Committee on GRAS substances (SCOGS); FDA: Silver Spring, MD, USA, 1959.
- Scientific Committee for Food—SCF. Report of the Scientific Committee for Food on Caffeine; SCF: Oslo, Norway, 1983. [Google Scholar]
- Mulder, C.J. Chemische Untersuchung des chinesischen und des javanischen. Thees. Arch. Pharm. 1838, 65, 68. [Google Scholar] [CrossRef]
- Lehmann, C.G. Lehrbuch der physiologischen Chemie. Leipizig 1850, 2, 367. [Google Scholar]
- James, J.E. Caffeine and Health; James, J.E., Ed.; London Academic Press: London, UK, 1991; p. 63. [Google Scholar]
- Lima, J.P.; Farah, A. Potential negative effects of caffeine consumption on health. In Coffee: Consumption and Health Implications; Farah, A., Ed.; Royal Society of Chemistry: London, UK, 2019; ISBN 978-1782620044. [Google Scholar]
- Frerichs, F.T. Die Verdauung. Handbuch Phys. 1846, 3, 721. [Google Scholar]
- Tarka, S.M.; Cornish, H.H. The toxicology of cocoa and methylxanthines: A review of the literature. Crit. Rev. Toxicol. 1982, 9, 275–312. [Google Scholar] [CrossRef]
- Kerrigan, S.; Lindsey, T. Fatal caffeine overdose: Two case reports. Forensic. Sci. Int. 2005, 153, 67–69. [Google Scholar] [CrossRef] [PubMed]
- Stavric, B.; Klassen, R.; Watkinson, B.; Karpinski, K.; Stapley, R.; Fried, P. Variability in caffeine consumption from coffee and tea: Possible significance for epidemiological studies. Food Chem. Toxicol. 1988, 26, 111–118. [Google Scholar] [CrossRef]
- Arya, L.A.; Myers, D.L.; Jackson, N.D. Dietary caffeine intake and the risk for detrusor instability: A case-control study. Obstet. Gynecol. 2000, 96, 85–89. [Google Scholar] [CrossRef] [PubMed]
- Tajima, Y. Coffee-induced hypokalaemia. Clin. Med. Insights Case Rep. 2010, 3, 9–13. [Google Scholar]
- Food Standards Australia New Zealand. Report from the Expert Working Group on the Safety Aspects of Dietary Caffeine; Food Standards Australia New Zealand: Canberra, Australia, 2000.
- Nordic Council of Ministers. Risk Assessment of Caffeine among Children and Adolescents in the Nordic Countries; Nordic Council of Ministers: Copenhagen, Denmark, 2008. [Google Scholar]
- Belgium Superior Health Council. The Use of Caffeine in Foodstuffs; Belgium Superior Health Council: Bruxelles, Belgium, 2012. [Google Scholar]
- Rogers, P.J.; Hohoff, C.; Heatherley, S.V.; Mullings, E.L.; Maxfield, P.J.; Evershed, R.P.; Deckert, J.; Nutt, D.J. Association of the anxiogenic and alerting effects of caffeine with ADORA2A and ADORA1 polymorphisms and habitual level of caffeine consumption. Neuropsychopharmacology 2010, 35, 1973–1983. [Google Scholar] [CrossRef] [PubMed]
- Childs, E.; Hohoff, C.; Deckert, J.; Xu, K.; Badner, J.; de Wit, H. Association between ADORA2A and DRD2 polymorphisms and caffeine-induced anxiety. Neuropsychopharmacology 2008, 33, 2791–2800. [Google Scholar] [CrossRef] [PubMed]
- Ammon, H.P. Biochemical mechanism of caffeine tolerance. Arch. Pharm. 1991, 324, 261–267. [Google Scholar] [CrossRef]
- Dorfman, L.J.; Jarvik, M.E. Comparative stimulant and diuretic actions of caffeine and theobromine in man. Clin. Pharmacol. Ther. 1970, 11, 869–872. [Google Scholar] [CrossRef]
- Zwyghuizen-Doorenbos, A.; Roehrs, T.A.; Lipschutz, L.; Timms, V.; Roth, T. Effects of caffeine on alertness. Psychopharmacology 1990, 100, 36–39. [Google Scholar] [CrossRef]
- Elkins, R.N.; Rapoport, J.L.; Zahn, T.P.; Buchsbaum, M.S.; Weingartner, H.; Kopin, I.J.; Langer, D.; Johnson, C. Acute effects of caffeine in normal prepubertal boys. Am. J. Psychiatry 1981, 138, 178–183. [Google Scholar] [CrossRef]
- Temple, J.L.; Ziegler, A.M.; Graczyk, A.M.; Crandall, A. Effects of acute and chronic caffeine on risk-taking behavior in children and adolescents. J. Psychopharmacol. 2017, 31, 561–568. [Google Scholar] [CrossRef] [PubMed]
- Rapoport, J.L.; Elkins, R.; Neims, A.; Zahn, T.; Berg, C.J. Behavioral and autonomic effects of caffeine in normal boys. Dev. Pharmacol. Ther. 1981, 3, 74–82. [Google Scholar] [CrossRef] [PubMed]
- Rapoport, J.L.; Berg, C.J.; Ismond, D.R.; Zahn, T.P.; Neims, A. Behavioral effects of caffeine in children. Relationship between dietary choice and effects of caffeine challenge. Arch. Gen. Psychiatry 1984, 41, 1073–1079. [Google Scholar] [CrossRef] [PubMed]
- Leviton, A. Behavioral correlates of caffeine consumption by children. Clin. Pediatr. 1992, 31, 742. [Google Scholar] [CrossRef] [PubMed]
- Baer, R.A. Effects of caffeine on classroom behavior, sustained attention, and a memory task in preschool children. J. App. Behav. Anal. 1987, 20, 225–234. [Google Scholar] [CrossRef]
- Bernstein, G.A.; Carroll, M.E.; Crosby, R.D.; Perwien, A.R.; Go, F.S.; Benowitz, N.L. Caffeine effects on learning, performance, and anxiety in normal school-age children. J. Am. Acad. Child Adolesc. Psychiatry 1994, 33, 407–415. [Google Scholar] [CrossRef] [PubMed]
- Hale, K.L.; Hughes, J.R.; Oliveto, A.H.; Higgins, S.T. Caffeine self-administration and subjective effects in adolescents. Exp. Clin. Pharmacol. 1995, 3, 364–370. [Google Scholar] [CrossRef]
- Davis, R.E.; Osorio, I. Childhood caffeine tic syndrome. Pediatrics 1998, 101, e4. [Google Scholar] [CrossRef]
- Temple, J.L.; Bulkley, A.M.; Briatico, L.; Dewey, A.M. Sex differences in reinforcing value of caffeinated beverages in adolescents. Behav. Pharmacol. 2009, 20, 731–741. [Google Scholar] [CrossRef]
- Temple, J.L.; Ziegler, A.M. Gender differences in subjective and physiological responses to caffeine and the role of steroid hormones. J. Caffeine Res. 2011, 1, 41–48. [Google Scholar] [CrossRef]
- Stein, M.A.; Krasowski, M.; Leventhal, B.L.; Phillips, W.; Bender, B.G. Behavioral and cognitive effects of methylxanthines. A meta-analysis of theophylline and caffeine. Arch. Pediatr. Adolesc. Med. 1996, 150, 284–288. [Google Scholar] [CrossRef] [PubMed]
- Robertson, D.; Frolich, J.C.; Carr, R.K.; Watson, J.T.; Hollifield, J.W.; Shand, D.G.; Oates, J.A. Effects of caffeine on plasma renin activity, catecholamines and blood pressure. N. Engl. J. Med. 1978, 298, 1811–1886. [Google Scholar] [CrossRef] [PubMed]
- Dobmeyer, D.J.; Stine, R.A.; Leier, C.V.; Greenberg, R.; Schaal, S.F. The arrhythmogenic effects of caffeine in human beings. N. Engl. J. Med. 1983, 308, 814–816. [Google Scholar] [CrossRef] [PubMed]
- Nurminen, M.L.; Niittynen, L.; Korpela, R.; Vapaatalo, H. Coffee, caffeine and blood pressure: A critical review. Eur. J. Clin. Nutr. 1999, 53, 831–839. [Google Scholar] [CrossRef] [PubMed]
- Watanabe, T.; Arai, Y.; Mitsui, Y.; Kusaura, T.; Okawaand, W.; Kajihara, Y. The blood pressure-lowering effect and safety of chlorogenic acid from green coffee bean extract in essential hypertension. Clin. Exp. Hypertens. 2006, 28, 439–449. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, A.; Kagawa, D.; Fujii, A.; Ochiai, R.; Tokimitsu, I.; Saito, I. Short- and long-term effects of ferulic acid on blood pressure in spontaneously hypertensive rats. Am. J. Hypertens. 2002, 15, 351–357. [Google Scholar] [CrossRef]
- Farag, N.H.; Whitsett, T.L.; McKey, B.S.; Wilson, M.F.; Vincent, A.S.; Everson-Rose, S.A.; Lovallo, W.R. Caffeine and blood pressure response: Sex, age, and hormonal status. J. Wom. Health 2010, 19, 1171–1176. [Google Scholar] [CrossRef] [PubMed]
- Worthley, M.I.; Prabhu, A.; De Sciscio, P.; Schultz, C.; Sanders, P.; Willoughby, S.R. Detrimental effects of energy drink consumption on platelet and endothelial function. Am. J. Med. 2010, 123, 1841–1887. [Google Scholar] [CrossRef] [PubMed]
- Buscemi, S.; Mattina, A.; Tranchina, M.R.; Verga, S. Acute effects of coffee on QT interval in healthy subjects. Nutr. J. 2011, 10, 15. [Google Scholar] [CrossRef] [PubMed]
- Shi, J.; Benowitz, N.L.; Denaro, C.P.; Sheiner, L.B. Pharmacokinetic-pharmacodynamic modeling of caffeine: Tolerance to pressor effects. Clin. Pharmacol. Ther. 1993, 53, 6–14. [Google Scholar] [CrossRef]
- Green, P.J.; Kirby, R.; Suls, J. The effects of caffeine on blood pressure and heart rate: A review. Ann. Behav. Med. 1996, 18, 201–216. [Google Scholar] [CrossRef] [PubMed]
- Greenland, S. A meta-analysis of coffee, myocardial infarction, and coronary death. Epidemiology 1993, 4, 366–374. [Google Scholar] [CrossRef] [PubMed]
- Wikoff, D.; Welsh, B.T.; Henderson, R.; Brorby, G.P.; Britt, J.; Myers, E.; Goldberger, J.; Lieberman, H.R.; O’Brien, C.; Peck, J.; et al. Systematic review of the potential adverse effects of caffeine consumption in healthy adults, pregnant women, adolescents, and children. Food Chem. Toxicol. 2017, 109, 585–648. [Google Scholar] [CrossRef] [PubMed]
- Ding, M.; Bhupathiraju, S.N.; Satija, A.; van Dam, R.M.; Hu, F.B. Long-term coffee consumption and risk of cardiovascular disease: A systematic review and a dose-response meta-analysis of prospective cohort studies. Circulation 2014, 129, 643–659. [Google Scholar] [CrossRef] [PubMed]
- Caldeira, D.; Martins, C.; Alves, L.B.; Pereira, H.; Ferreira, J.J.; Costa, J. Caffeine does not increase the risk of atrial fibrillation: A systematic review and meta-analysis of observational studies. Heart 2013, 99, 1383–1389. [Google Scholar] [CrossRef] [PubMed]
- Kim, B.; Nam, Y.; Kim, J.; Choi, H.; Won, C. Coffee consumption and stroke risk: A meta-analysis of epidemiologic studies. Korean J. Fam. Med. 2012, 33, 356–365. [Google Scholar] [CrossRef] [PubMed]
- Salazar-Martinez, E.; Willett, W.C.; Ascherio, A.; Manson, J.E.; Leitzmann, M.F.; Stampfer, M.J.; Hu, F.B. Coffee consumption and risk for type 2 diabetes mellitus. Ann. Intern. Med. 2004, 140, 1–8. [Google Scholar] [CrossRef] [PubMed]
- van Dam, R.M.; Willett, W.C.; Manson, J.E.; Hu, F.B. Coffee, caffeine, and risk of type 2 diabetes: A prospective cohort study in younger and middle-aged U.S. women. Diabetes Care 2006, 29, 398–403. [Google Scholar] [CrossRef]
- Hjellvik, V.; Tverdal, A.; Strom, H. Boiled coffee intake and subsequent risk for type 2 diabetes. Epidemiology 2011, 22, 418–421. [Google Scholar] [CrossRef]
- Floegel, A.; Pischon, T.; Bergmann, M.M.; Teucher, B.; Kaaks, R.; Boeing, H. Coffee consumption and risk of chronic disease in the European Prospective Investigation into Cancer and Nutrition (EPIC)-Germany study. Am. J. Clin. Nutr. 2012, 95, 901–908. [Google Scholar] [CrossRef]
- Doo, T.; Morimoto, Y.; Steinbrecher, A.; Kolonel, L.N.; Maskarinec, G. Coffee intake and risk of type 2 diabetes: The multiethnic cohort. Public Health Nutr. 2014, 17, 1328–1336. [Google Scholar] [CrossRef] [PubMed]
- Carlström, M.; Larsson, S.C. Coffee consumption and reduced risk of developing type 2 diabetes: A systematic review with meta-analysis. Nutr. Rev. 2018, 76, 395–417. [Google Scholar] [CrossRef] [PubMed]
- Ding, M.; Bhupathiraju, S.N.; Chen, M.; van Dam, R.M.; Hu, F.B. Caffeinated and decaffeinated coffee consumption and risk of type 2 diabetes: A systematic review and a dose-response meta-analysis. Diabetes Care 2014, 37, 569–586. [Google Scholar] [CrossRef] [PubMed]
- Shearer, J.; Sellars, E.; Farah, A.; Graham, T.E.; Wasserman, D.H. Effects of chronic coffee consumption on glucose kinetics in the conscious rat. Can. J. Physiol. Pharmacol. 2007, 85, 823–830. [Google Scholar] [CrossRef] [PubMed]
- Lane, J.D.; Feinglos, M.N.; Surwit, R.S. Caffeine increases ambulatory glucose and postprandial response in coffee drinkers with type 2 diabetes. Diabetes Care 2008, 31, 221–222. [Google Scholar] [CrossRef] [PubMed]
- Moisey, L.L.; Kacker, S.; Bickerton, A.C.; Robinson, L.E.; Graham, T.E. Caffeinated coffee consumption impairs blood glucose homeostasis in response to high and low glycemic index meals in healthy men. Am. J. Clin. Nutr. 2008, 87, 1254–1261. [Google Scholar] [CrossRef] [PubMed]
- Robinson, L.E.; Spafford, C.; Graham, T.E.; Smith, G.N. Acute caffeine ingestion and glucose tolerance in women with or without gestational diabetes mellitus. J. Obstet. Gynaecol. Can. 2009, 31, 304–312. [Google Scholar] [CrossRef]
- Lane, J.D. Caffeine, glucose metabolism, and type 2 diabetes. J. Caffeine Res. 2011, 1, 23–28. [Google Scholar] [CrossRef]
- Han, D.H.; Hansen, P.A.; Nolte, L.A.; Holloszy, J.O. Removal of adenosine decreases the responsiveness of muscle glucose transport to insulin and contractions. Diabetes 1998, 47, 1671–1675. [Google Scholar] [CrossRef]
- Keijzers, G.B.; De Galan, B.E.; Tack, C.J.; Smits, P. Caffeine can decrease insulin sensitivity in humans. Diabetes Care 2002, 25, 364–369. [Google Scholar] [CrossRef]
- Wu, T.; Willett, W.C.; Hankinson, S.E.; Giovannucci, E. Caffeinated coffee, decaffeinated coffee, and caffeine in relation to plasma C-peptide levels, a marker of insulin secretion, in U.S. women. Diabetes Care 2005, 28, 1390–1396. [Google Scholar] [CrossRef] [PubMed]
- Greenberg, J.A.; Geliebter, A.; Boozer, C.N. Coffee, diabetes, and weight control. Am. J. Clin. Nutr. 2006, 84, 682–693. [Google Scholar] [CrossRef] [PubMed]
- Pereira, M.A.; Parker, E.D.; Folsom, A.R. Coffee consumption and risk of type 2 diabetes mellitus: An 11-year prospective study of 28 812 postmenopausal women. Arch. Intern. Med. 2006, 166, 1311–1316. [Google Scholar] [CrossRef] [PubMed]
- Hamer, M.; Witte, D.R.; Mosdøl, A.; Marmot, M.G.; Brunner, E.J. Prospective study of coffee and tea consumption in relation to risk of type 2 diabetes mellitus among men and women: The Whitehall II study. Br. J. Nutr. 2008, 100, 1046–1053. [Google Scholar] [CrossRef] [PubMed]
- Sartorelli, D.S.; Fagherazzi, G.; Balkau, B.; Touillaud, B.M.S.; Boutron-Ruault, M.C.; Lauzon-Guillain, B.; Clavel-Chapelon, F. Differential effects of coffee on the risk of type 2 diabetes according to meal consumption in a French cohort of women: The E3N/EPIC cohort study. Am. J. Clin. Nutr. 2010, 91, 1002–1012. [Google Scholar] [CrossRef]
- Bhupathiraju, S.N.; Pan, A.; Malik, V.S.; Manson, J.E.; Willett, W.C.; van Dam, R.M.; Hu, F.B. Caffeinated and caffeine-free beverages and risk of type 2 diabetes. Am. J. Clin. Nutr. 2013, 97, 155–166. [Google Scholar] [CrossRef] [PubMed]
- Battram, D.S.; Arthur, R.; Weekes, A.; Graham, T.E. The glucose intolerance induced by caffeinated coffee ingestion is less pronounced than that due to alkaloid caffeine in men. J. Nutr. 2006, 136, 1276–1280. [Google Scholar] [CrossRef] [PubMed]
- Johnston, K.L.; Clifford, M.N.; Morgan, L.M. Coffee acutely modifies gastrointestinal hormone secretion and glucose tolerance in humans: Glycemic effects of chlorogenic acid and caffeine. Am. J. Clin. Nutr. 2003, 78, 728–733. [Google Scholar] [CrossRef] [PubMed]
- Porto, A.C.V.; Farah, A. Potential effects of trigonelline derivatives on health. In Coffee: Consumption and Health Implications; Farah, A., Ed.; Royal Society of Chemistry: London, UK, 2019; ISBN 978-1782620044. [Google Scholar]
- van Dijk, A.E.; Olthof, M.R.; Meeuse, J.C.; Seebus, E.; Heine, R.J.; van Dam, R.M. Acute effects of decaffeinated coffee and the major coffee components chlorogenic acid and trigonelline on glucose tolerance. Diabetes Care 2009, 32, 1023–1025. [Google Scholar] [CrossRef]
- van Dieren, S.; Uiterwaal, C.S.; van der Schouw, Y.T.; van der, A.D.I.; Boer, J.M.; Spijkerman, A.; Grobbee, D.E.; Beulens, J.W. Coffee and tea consumption and risk of type 2 diabetes. Diabetologia 2009, 52, 2561–2569. [Google Scholar] [CrossRef]
- Albrand, G.; Munoz, F.; Sornay-Rendu, E.; DuBoeuf, F.; Delmas, P.D. Independent predictors of all osteoporosis-related fractures in healthy postmenopausal women: The OFELY study. Bone 2003, 32, 78–85. [Google Scholar] [CrossRef]
- Fung, T.T.; Arasaratnam, M.H.; Grodstein, F.; Katz, J.N.; Rosner, B.; Willett, W.C.; Feskanich, D. Soda consumption and risk of hip fractures in postmenopausal women in the Nurses’ Health Study. Am. J. Clin. Nutr. 2014, 100, 953–958. [Google Scholar] [CrossRef] [PubMed]
- Hallstrom, H.; Byberg, L.; Glynn, A.; Lemming, E.W.; Wolk, A.; Michaelsson, K. Long-term coffee consumption in relation to fracture risk and bone mineral density in women. Am. J. Epidemiol. 2013, 178, 898–909. [Google Scholar] [CrossRef] [PubMed]
- Jha, R.M.; Mithal, A.; Malhotra, N.; Brown, E.M. Pilot case-control investigation of risk factors for hip fractures in the urban Indian population. BMC Musculoskelet. Disord. 2010, 11, 48. [Google Scholar] [CrossRef] [PubMed]
- Lee, D.R.; Lee, J.; Rota, M.; Lee, J.; Ahn, H.S.; Park, S.M.; Shin, D. Coffee consumption and risk of fractures: A systematic review and dose-response meta-analysis. Bone 2014, 63, 20–28. [Google Scholar] [CrossRef] [PubMed]
- Conlisk, A.J.; Galuska, D.A. Is caffeine associated with bone mineral density in young adult women? Prev. Med. 2000, 31, 562–568. [Google Scholar] [CrossRef] [PubMed]
- Hannan, M.T.; Felson, D.T.; Dawson-Hughes, N.; Tucker, K.L.; Cupples, L.A.; Wilson, P.W.; Kiel, D.P. Risk factors for longitudinal bone loss in elderly men and women: The Framingham Osteoporosis Study. J. Bone Miner. Res. 2000, 15, 710–720. [Google Scholar] [CrossRef] [PubMed]
- El Maghraoui, A.; Ghazi, M.; Gassim, S.; Ghozlani, I.; Mounach, A.; Rezqi, A.; Dehhaoui, M. Risk factors of osteoporosis in healthy Moroccan men. BMC Musculoskelet. Disord. 2010, 11, 148. [Google Scholar] [CrossRef] [PubMed]
- Hallström, H.; Melhus, H.; Glynn, A.; Lind, L.; Syvänen, A.C.; Michaëlsson, K. Coffee consumption and CYP1A2 genotype in relation to bone mineral density of the proximal femur in elderly men and women: A cohort study. Nutr. Metab. 2010, 22, 7–12. [Google Scholar] [CrossRef] [PubMed]
- Harter, D.L.; Busnello, F.M.; Dibi, R.P.; Stein, A.T.; Kato, S.K.; Vanin, C.M.D.N. Association between low bone mass and calcium and caffeine intake among perimenopausal women in Southern Brazil: Cross-sectional study. São Paulo Med. J. 2013, 131, 315–322. [Google Scholar] [CrossRef] [PubMed]
- Rapuri, P.B.; Gallagher, J.C.; Kinyamu, H.K.; Ryschon, K.L. Caffeine intake increases the rate of bone loss in elderly women and interacts with vitamin D receptor genotypes. Am. J. Clin. Nutr. 2001, 74, 694–700. [Google Scholar] [CrossRef] [PubMed]
- Heaney, R.P.; Rafferty, K. Carbonated beverages and urinary calcium excretion. Am. J. Clin. Nutr. 2001, 74, 343–347. [Google Scholar] [CrossRef] [PubMed]
- Ribeiro-Alves, M.; Trugo, L.C.; Donangelo, C.M. Use of oral contraceptives blunts the calciuric effect of caffeine in young adult women. J. Nutr. 2003, 133, 393–398. [Google Scholar] [CrossRef] [PubMed]
- Hernández-Avila, M.; Colditz, G.A.; Stampfer, M.J.; Rosner, B.; Speizer, F.E.; Willet, W.C. Caffeine, moderate alcohol intake, and risk of fractures of the hip and forearm in middle-aged women. Am. J. Clin. Nutr. 1991, 54, 157–163. [Google Scholar] [CrossRef] [PubMed]
- Hallstrom, H.; Wolk, A.; Glynn, A.; Michaëlsson, K. Coffee, tea and caffeine consumption in relation to osteoporotic fracture risk in a cohort of Swedish women. Osteoporos. Int. 2006, 17, 1055–1064. [Google Scholar] [CrossRef] [PubMed]
- Heaney, R.P.; Recker, R.R. Effects of nitrogen, phosphorus, and caffeine on calcium balance in women. J. Lab. Clin. Med. 1982, 99, 46–55. [Google Scholar] [PubMed]
- Barger-Lux, M.J.; Heaney, R.P.; Stegman, M.R. Effects of moderate caffeine intake on the calcium economy of premenopausal women. Am. J. Clin. Nutr. 1990, 52, 722–725. [Google Scholar] [CrossRef] [PubMed]
- Hernández-Avila, M.; Stampfer, M.J.; Ravnikar, V.A.; Willet, W.C.; Schiff, I.; Francis, M.; Longscope, C.; McKinley, S.M. Caffeine and other predictors of bone density among pre- and perimenopausal women. Epidemiology 1993, 4, 128–134. [Google Scholar] [CrossRef] [PubMed]
- Barger-Lux, M.J.; Heaney, R.P. Vitamin D receptor gene polymorphism, bone mass, body size, and vitamin D receptor density. Calcif. Tissue Int. 1995, 57, 161–162. [Google Scholar] [CrossRef]
- Barbour, K.E.; Zmuda, J.M.; Strotmeyer, E.S.; Horwitz, M.J.; Boudreau, R.; Evans, R.W.; Ensrud, K.E.; Petit, M.A.; Gordon, C.L.; Cauley, J.A. Correlates of trabecular and cortical volumetric bone mineral density of the radius and tibia in older men: The Osteoporotic Fractures in Men Study. Bone Miner. Res. 2010, 25, 1017–1028. [Google Scholar] [CrossRef]
- Hasling, C.; Søndergaard, K.; Charles, P.; Mosekilde, L. Calcium metabolism in postmenopausal osteoporotic women is determined by dietary calcium and coffee intake. J. Nutr. 1992, 122, 1119–1126. [Google Scholar] [CrossRef] [PubMed]
- Gressner, O.A.; Lahme, B.; Siluschek, M.; Gressner, A.M. Identification of paraxanthine as the most potent caffeine-derived inhibitor of connective tissue growth factor expression in liver parenchymal cells. Liver Int. 2009, 29, 886–897. [Google Scholar] [CrossRef] [PubMed]
- Fromigue, O.; Modrowski, D.; Marie, P.J. Growth factors and bone formation in osteoporosis: Roles for fibroblast growth factor and transforming growth factor beta. Curr. Pharma. Des. 2004, 10, 2593–2603. [Google Scholar] [CrossRef]
- Evans, B.A.; Elford, C.; Pexa, A.; Francis, K.; Hughes, A.C.; Deussen, A.; Ham, J. Human osteoblast precursors produce extracellular adenosine, which modulates their secretion of IL-6 and osteoprotegerin. J. Bone Miner. Res. 2006, 21, 228–236. [Google Scholar] [CrossRef] [PubMed]
- Dlugosz, L.; Bracken, M.B. Reproductive effects of caffeine: A review and theoretical analysis. Epidemiol. Rev. 1992, 14, 83–100. [Google Scholar] [CrossRef]
- Golding, J. Reproduction and caffeine consumption-a literature review. Early Hum. Dev. 1995, 43, 1–14. [Google Scholar] [CrossRef]
- Leviton, A. Does coffee consumption increase the risk of reproductive adversities? J. Am. Med. Womens Assoc. 1995, 50, 20–22. [Google Scholar]
- Northstone, K.; Golding, J. Caffeine and Activation Theory Effects on Health and Behavior; Smith, B.D., Gupta, U., Gupta, B.S., Eds.; CRC Press: Boca Raton, FL, USA, 2007; p. 199. [Google Scholar]
- Morgan, S.; Koren, G.; Bozzo, P. Is caffeine consumption safe during pregnancy? Can. Fam. Physician 2013, 59, 361–362. [Google Scholar]
- Peck, J.D.; Leviton, A.; Cowan, L.D. A review of the epidemiologic evidence concerning the reproductive health effects of caffeine consumption: A 2000–2009 update. Food Chem. Toxicol. 2010, 48, 2549–2576. [Google Scholar] [CrossRef]
- Rondo, P.H.C.; Rodrigues, L.C.; Tomkins, A.M. Coffee consumption and intrauterine growth retardation in Brazil. Eur. J. Clin. Nutr. 1996, 50, 705–709. [Google Scholar]
- Karen, G. Caffeine during pregnancy? In moderation. Can. Fam. Physician 2000, 46, 801–803. [Google Scholar]
- Rosenberg, L.; Mitchell, A.A.; Shapiro, S.; Slone, D. Selected birth defects in relation to caffeine-containing beverages. J. Am. Med. Assoc. 1982, 247, 1429–1432. [Google Scholar] [CrossRef]
- Olsen, J.; Overvad, K.; Frische, G. Coffee consumption, birthweight, and reproductive failures. Epidemiology 1991, 2, 370–374. [Google Scholar] [CrossRef] [PubMed]
- Christian, M.S.; Brent, R.L. Teratogen update: Evaluation of the reproductive and developmental risks of caffeine. Teratology 2001, 64, 51–78. [Google Scholar] [CrossRef] [PubMed]
- CARE Study Group. Maternal caffeine intake during pregnancy and risk of fetal growth restriction: A large prospective observational study. BMJ 2008, 337, 2332. [Google Scholar] [CrossRef] [PubMed]
- Sengpiel, V.; Elind, E.; Bacelis, J.; Nilsson, S.; Grove, J.; Myhre, R.; Haugen, M.; Meltzer, H.M.; Alexander, J.; Jacobsson, B.; et al. Maternal caffeine intake during pregnancy is associated with birth weight but not with gestational length: Results from a large prospective observational cohort study. BMC Med. 2013, 11, 42. [Google Scholar] [CrossRef] [PubMed]
- Bross, I.D.J.; Tidings, J. Another look at coffee drinking and cancer of the urinary bladder. Prev. Med. 1973, 2, 445–451. [Google Scholar] [CrossRef]
- Bravo, P.; deI Rey, J.; Sánchez, J.; Conde, M. Coffee and analgesics as risk factors for cancer of the bladder. Arch. Esp. Urol. 1986, 39, 337–341. [Google Scholar]
- Ciccne, G.; Vineis, P. Coffee drinking and bladder cancer. Cancer Lett. 1988, 41, 45–52. [Google Scholar] [CrossRef]
- International Agency for Research on Cancer—IARC Working Group. IARC Monographs on the Evaluation of Carcinogenic Risks to Humans: Coffee, Tea, Maté, Methylxanthines and Methylglyoxal; IARC Working Group: Lyon, France, 1991. [Google Scholar]
- Botelho, F.; Lunet, N.; Barros, H. Coffee and gastric cancer: Systematic review and meta-analysis. Cad. Saúde Públ. 2006, 22, 889–900. [Google Scholar] [CrossRef]
- Lee, J.E.; Hunter, D.J.; Spiegelman, D.; Adami, H.O.; Bernstein, L.; van den Brandt, P.A.; Buring, J.E.; Cho, E.; English, D.; Folsom, A.R.; et al. Intakes of coffee, tea, milk, soda and juice and renal cell cancer in a pooled analysis of 13 prospective studies. Int. J. Cancer 2007, 121, 2246–2253. [Google Scholar] [CrossRef] [PubMed]
- Boggs, D.A.; Palmer, J.R.; Stampfer, M.J.; Spiegelman, D.; Adams-Campbell, L.L.; Rosenberg, L. Tea and coffee intake in relation to risk of breast cancer in the Black Women’s Health Study. Cancer Causes Control 2010, 21, 1941–1948. [Google Scholar] [CrossRef] [PubMed]
- Fagherazzi, G.; Touillaud, M.S.; Boutron-Ruault, M.C.; Clavel-Chapelon, F.; Romieu, I. No association between coffee, tea or caffeine consumption and breast cancer risk in a prospective cohort study. Public Health Nutr. 2011, 5, 1315–1320. [Google Scholar] [CrossRef] [PubMed]
- Giovannucci, E. Meta-analysis of coffee consumption and risk of colorectal cancer. Am. J. Epidemiol. 1998, 147, 1043–1052. [Google Scholar] [CrossRef]
- Naganuma, T.; Kuriyama, S.; Akhter, M.; Kakizaki, M.; Nakaya, N.; Matsuda-Ohmori, K. Coffee consumption and the risk of colorectal cancer: A prospective cohort study in Japan. Int. J. Cancer 2007, 120, 1542–1547. [Google Scholar] [CrossRef] [PubMed]
- Yu, X.; Zou, Z.J.; Dong, J. Coffee consumption and risk of cancers: A meta-analysis of cohort studies. BMC Cancer 2011, 11, 96. [Google Scholar] [CrossRef] [PubMed]
- Wang, A.; Wang, S.; Zhu, C.; Huang, H.; Wu, L.; Wan, X.; Yang, X.; Zhang, H.; Miao, R.; He, L.; et al. Coffee and cancer risk: A meta-analysis of prospective observational studies. Sci. Rep. 2016, 6, 33711. [Google Scholar] [CrossRef] [PubMed]
- Strain, E.; Mumford, G.; Silverman, K.; Griffiths, R.R. Caffeine dependence syndrome. Evidence from case histories and experimental evaluations. JAMA 1994, 272, 1043–1048. [Google Scholar] [CrossRef] [PubMed]
- Silverman, K.; Evans, S.M.; Strain, E.C.; Griffiths, R.R. Withdrawal syndrome after the double-blind cessation of caffeine consumption. N. Engl. J. Med. 1992, 327, 1109–1114. [Google Scholar] [CrossRef] [PubMed]
- American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders; American Psychiatric Association: Washington, DC, USA, 1994. [Google Scholar]
- Evans, S.M.; Griffiths, R.R. Caffeine withdrawal: A parametric analysis of caffeine dosing conditions. J. Pharm. Exp. Ther. 1999, 289, 285–294. [Google Scholar]
- Phillips-Bute, B.G.; Lane, J.D. Caffeine withdrawal symptoms following brief caffeine deprivation. Physiol. Behav. 1998, 63, 35–39. [Google Scholar] [CrossRef]
- Höfer, I.F.; Bättig, K. Psychophysiological effects of switching to caffeine tablets or decaffeinated coffee under field conditions. Pharmacopsychoecologia 1994, 7, 169–177. [Google Scholar]
- Griffiths, R.R.; Bigelow, G.E.; Liebson, I.A. Human coffee drinking: Reinforcing and physical dependence producing effects of caffeine. J. Pharmacol. Exp. Ther. 1986, 239, 416–425. [Google Scholar] [PubMed]
- Lader, M.; Cardwell, C.; Shine, P.; Scott, N. Caffeine withdrawal symptoms and rate of metabolism. J. Psychopharmacol. 1996, 10, 110–118. [Google Scholar] [CrossRef] [PubMed]
- McGowan, J.D.; Altman, R.E.; Kanto, W.P., Jr. Neonatal withdrawal symptoms after chronic maternal ingestion of caffeine. South. Med. J. 1988, 81, 1092–1094. [Google Scholar] [CrossRef]
- Goldstein, A.; Wallace, M.E. Caffeine dependence in schoolchildren? Exp. Clin. Psychopharmacol. 1997, 5, 388–392. [Google Scholar] [CrossRef]
- Pallarés, J.G.; Fernández-Elías, V.E.; Ortega, J.F.; Muñoz, G.; Muñoz-Guerra, J.; Mora Rodríguez, R. Neuromuscular responses to incremental caffeine doses: Performance and side effects. Med. Sci. Sports Exerc. 2013, 45, 2184–2192. [Google Scholar] [CrossRef]
- Nardi, A.E.; Lopes, F.L.; Freire, R.C.; Veras, A.B.; Nascimento, I.; Valença, A.M.; de-Melo Neto, V.L.; Soares-Filho, G.L.; King, A.L.; Araújo, D.M.; et al. Panic disorder and social anxiety disorder subtypes in a caffeine challenge test. Psychiatry Res. 2009, 169, 149–153. [Google Scholar] [CrossRef] [PubMed]
- Mesas, A.E.; Leon-Munoz, L.M.; Rodriguez-Artalejoand, F.; Lopez-Garcia, E. The effect of coffee on blood pressure and cardiovascular disease in hypertensive individuals: A systematic review and meta-analysis. Am. J. Clin. Nutr. 2011, 94, 1113–1126. [Google Scholar] [CrossRef]
- Noordzij, M.; Uiterwaal, C.; Arends, L.; Kok, F.; Grobbee, D.; Geleijnse, J. Blood pressure response to chronic intake of coffee and caffeine: A meta-analysis of randomized controlled trials. J. Hypertens. 2005, 23, 921–992. [Google Scholar] [CrossRef]
- Shi, X.; Xue, W.; Liang, S.; Zhao, J. Acute caffeine ingestion reduces insulin sensitivity in healthy subjects: A systematic review and meta-analysis. Nutr. J. 2016, 15, 103. [Google Scholar] [CrossRef] [PubMed]
- Whitehead, N.; White, H. Systematic review of randomized controlled trials of the effects of caffeine or caffeinated drinks on blood glucose concentrations and insulin sensitivity in people with diabetes mellitus. J. Hum. Nutr. Diet. 2013, 26, 111–125. [Google Scholar] [CrossRef]
- Harris, S.S.; Dawson-Hughes, B. Caffeine and bone loss in healthy postmenopausal women. Am. J. Clin. Nutr. 1994, 60, 573–578. [Google Scholar] [CrossRef] [PubMed]
- Lyngsø, J.; Ramlau-Hansen, C.H.; Bay, B.; Ingerslev, H.J.; Hulman, A.; Kesmodel, U.S. Association between coffee or caffeine consumption and fecundity and fertility: A systematic review and dose–response meta-analysis. Clin. Epidemiol. 2017, 9, 699–719. [Google Scholar] [CrossRef]
- Vaseghi, G.; Haghjoo-Javanmard, S.; Naderi, J.; Eshraghi, A.; Mahdavi, M.; Mansourian, M. Coffee consumption and risk of nonmelanoma skin cancer: A dose-response meta-analysis. Eur. J. Cancer Prev. 2018, 27, 164–170. [Google Scholar] [CrossRef]
- Liu, J.; Shen, B.; Shi, M.; Cai, J. Higher caffeinated coffee intake is associated with reduced malignant melanoma risk: A meta-analysis study. PLoS ONE 2016, 11, e0147056. [Google Scholar] [CrossRef]
- New Zealand Ministry of Health. Nutrient Reference Values for Australia and New Zealand Including Recommended Dietary Intakes; New Zealand Ministry of Health: Wellington, New Zealand, 2006.
- Higdon, J.V.; Frei, B. Coffee and health: A review of recent human research. Crit. Rev. Food Sci. Nutr. 2006, 46, 101–123. [Google Scholar] [CrossRef]
- Yang, A.; Palmer, A.A.; de Wit, H. Genetics of caffeine consumption and responses to caffeine. Psychopharmacology 2010, 211, 245–257. [Google Scholar] [CrossRef] [PubMed]
- Caffeine Informer. Available online: https://www.caffeineinformer.com (accessed on 9 February 2019).
- Aepli, A.; Kurth, S.; Tesler, N.; Jenni, O.G.; Huber, R. Caffeine consuming children and adolescents show altered sleep behavior and deep sleep. Brain Sci. 2015, 5, 441–455. [Google Scholar] [CrossRef] [PubMed]
- Cunha, R.A.; Ferré, S.; Vaugeois, J.M.; Chen, J.-F. Potential therapeutic interest of adenosine A2A receptors in psychiatric disorders. Curr. Pharm. Des. 2008, 14, 1512–1524. [Google Scholar] [CrossRef] [PubMed]
- Garfinkel, B.D.; Webster, C.D.; Sloman, L. Responses to methylphenidate and varied doses of caffeine in children with attention deficit disorder. Can. J. Psychiatry 1981, 26, 395–401. [Google Scholar] [CrossRef] [PubMed]
- Castellanos, F.X.; Rapoport, J.L. Effects of caffeine on development and behavior in infancy and childhood: A review of the published literature. Food Chem. Toxicol. 2002, 40, 1235–1242. [Google Scholar] [CrossRef]
- ABIC—Associação Brasileira das Indústrias de Café. Available online: http://www.abic.com.br (accessed on 9 February 2019).
- American Academy of Pediatrics; Adolescent Sleep Working Group; Committee on Adolescence; Council on School Health. School start times for adolescents. Pediatrics 2014, 134, 642–649. [Google Scholar] [CrossRef] [PubMed]
- Health Canada. It’s Your Health-Caffeine. Available online: http://www.hc-sc.gc.ca/hlvs/iyh-vsv/food-aliment/caffeine-eng.php (accessed on 9 February 2019).
- Korea Food and Drug Administration. Korea Nutrition Society; Korea Food and Drug Administration: Seoul, Korea, 2006.
- International Life Sciences Institute—ILSI. Food Safety – Caffeine; ILSI: Washington, DC, USA, 2010. [Google Scholar]
- United Kingdom Food Stand Agency. The Safety Aspects of Dietary Caffeine; Ministry of Heath: London, UK.
- Knight, C.A.; Knight, I.; Mitchell, D.C. Beverage caffeine intakes in young children in Canada and the US. J. Diet. Prac. Res. 2006, 67, 96–99. [Google Scholar] [CrossRef] [PubMed]
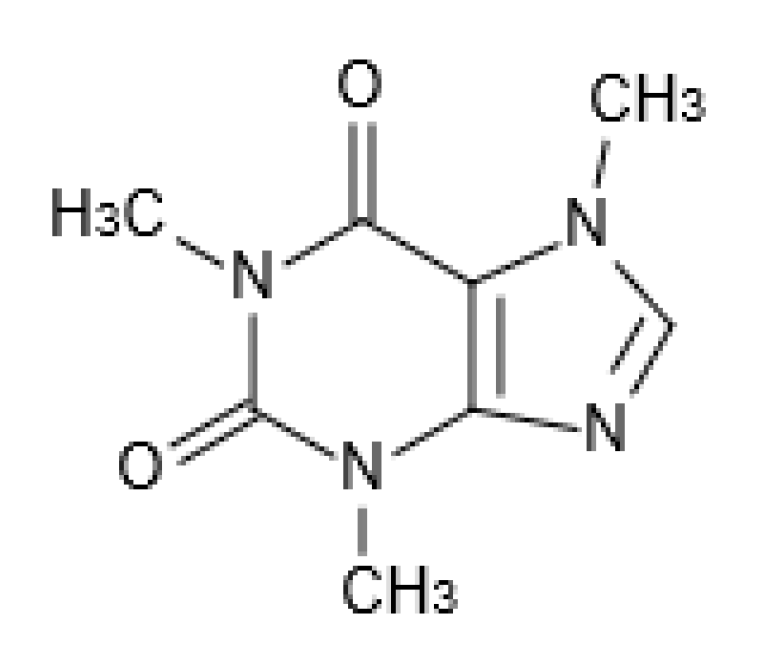
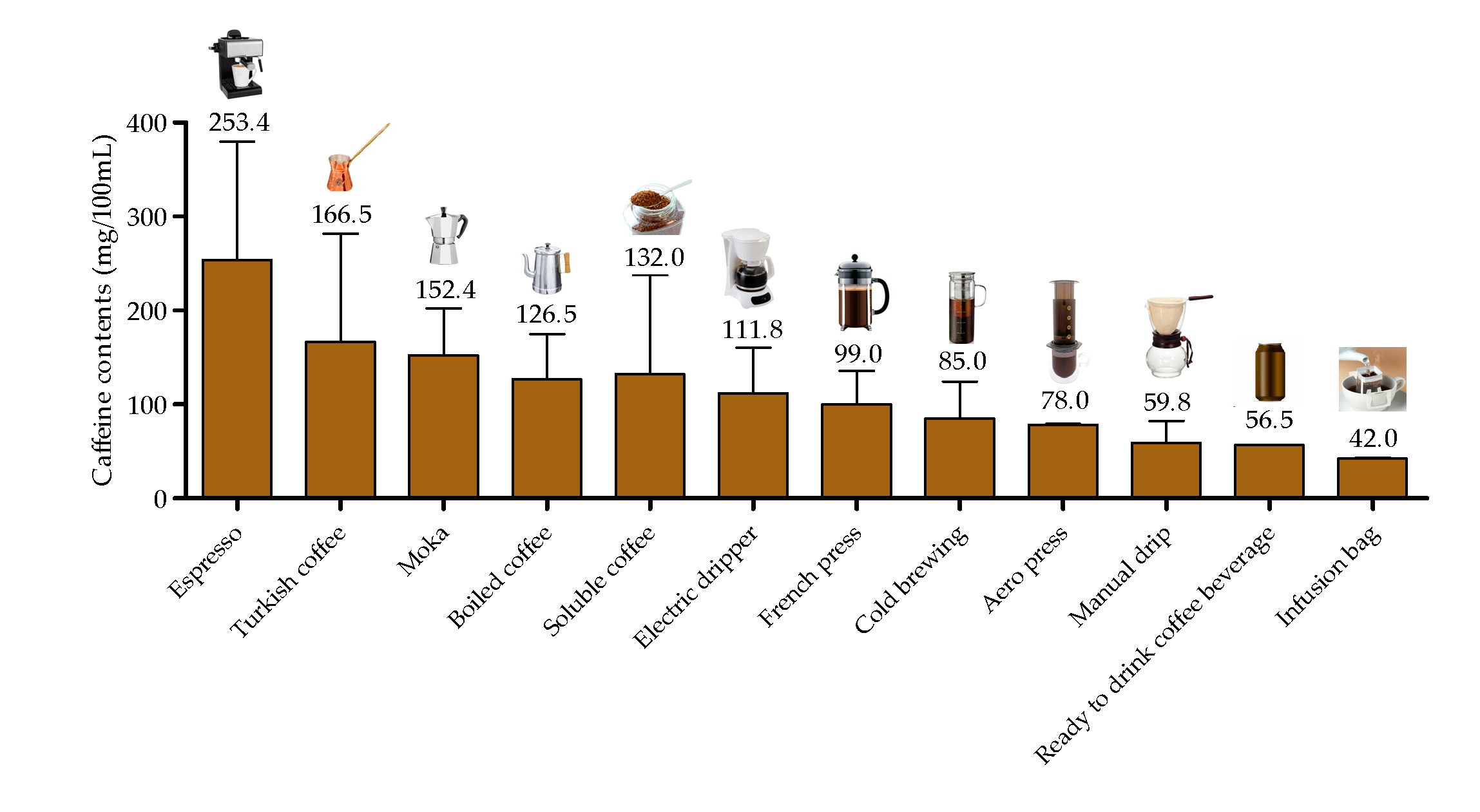
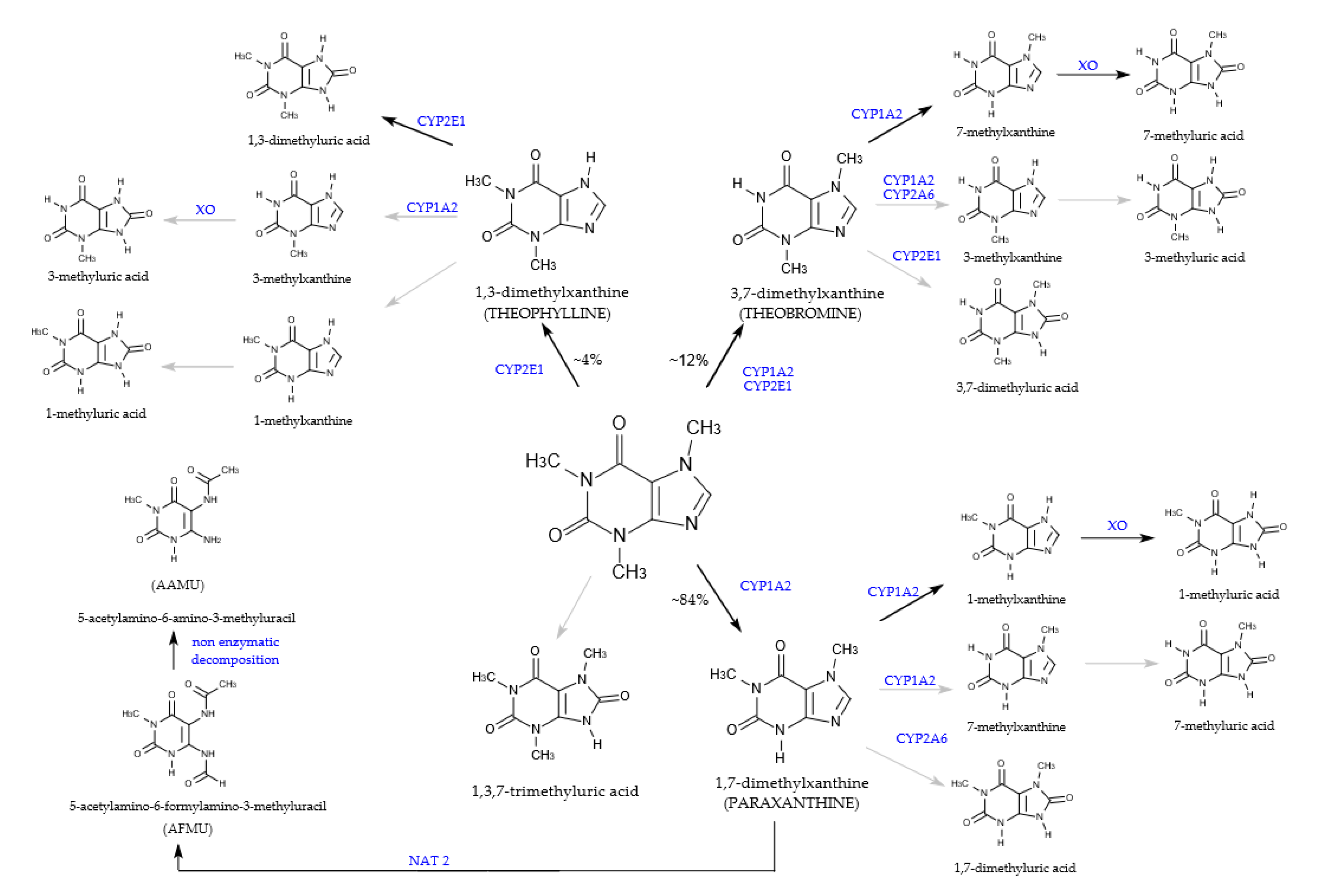
| Coffee Species/Cultivar/ Type | Country | N | Range of Caffeine Content (g/100 g) | Mean Caffeine Content (g/100 g) | Reference |
|---|---|---|---|---|---|
| Coffea arabica | |||||
| cv. Minas | USA | 2 | 0.9–1.2 | 1.1 | [4] |
| Nr | Ethiopia | 4 | 0.9–1.1 | 1.0 | [15] * |
| Nr | Hawaii, Colombia, Brazil, Africa | 6 | 0.8–0.9 | 0.8 | [26] |
| Nr | Brazil, Mexico, Colombia, Guatemala | 9 | 1.2–1.6 | 1.3 | [29] |
| Nr | Croatia | 6 | 0.8–1.4 | 1.3 | [50] |
| Nr | Brazil | 4 | 0.7–1.1 | 0.9 | [70] |
| Nr | Brazil | 3 | 1.2–1.3 | 1.3 | [72] |
| Nr | Poland | 2 | 1.0–1.1 | 1.0 | [73] |
| cv. Bourbon, Catuaí, Icatu | Brazil | 3 | 1.0–1.3 | 1.2 | [74] |
| Coffea canephora | |||||
| cv. Robusta | India, Honduras, Vietnam, Angola, Caneron | 20 | 2.0–2.6 | 2.3 | [29] |
| cv. Robusta | Croatia | 2 | 1.8–2.5 | 2.2 | [50] |
| cv. Robusta | Ivory Coast | 1 | - | 2.0 | [54] |
| - | Brazil | 3 | 2.2–2.3 | 2.2 | [72] |
| Commercial blends of C. arabica or C. arabica and C. canephora (cv Robusta or cv. Conilon) | |||||
| Arabica blends | USA | 36 | 0.8–1.4 | 1.2 | [4] |
| Blend | Africa | 1 | - | 1.4 | [26] |
| Blends | Brazil | 10 | 0.8–1.4 | Nr | [75] |
| Blends | USA | 6 | 1.0–1.6 | 1.3 | [76] |
| Nr | Nr | 3 | 1.4–1.9 | 1.7 | [77] |
| Blends | Brazil | 40 | 0.8–1.6 | 1.2 | [78] |
| - | Mexico | 3 | 1.5–1.7 | 1.5 | [79] |
| Soluble coffee | |||||
| Nr | Brazil | 2 | 2.0–2.2 | 2.1 | [26] |
| Nr | Nr | 2 | 4.5–4.8 | Nr | [77] |
| Blends | Brazil | 10 | 1.8–3.1 | 2.5 | [78] |
| Arabica | Brazil | 3 | 2.8–4.1 | 3.4 | [80] |
| cv. Conilon | Brazil | 3 | 3.9–5.8 | 5.0 | [80] |
| Nr | Brazil | 9 | 1.6–3.2 | 2.5 | [81] |
| Nr | Kenya | 2 | 1.6–3.4 | 2.7 | [82] |
| Nr | Nr | 5 | 2.2–3.9 | 2.8 | [83] |
| Coffee Species/ Type | Country | N | Amount of Powder (%) to Water/100 mL | Water Temperature | Brewing Time | Range of Caffeine Contents (mg/100 mL) | Mean Caffeine Content (mg/100 mL) | Reference |
|---|---|---|---|---|---|---|---|---|
| Manual drip | ||||||||
| Arabica | Colombia | 2 | 10 | 100 °C | 2 min | 48.1–53.0 | 50.8 | [86] |
| Arabica | Indonesia | 2 | 10 | 100 °C | 2 min | 48.3–57.9 | 53.1 | [86] |
| Arabica | Kenya | 2 | 10 | 100 °C | 2 min | 54.2–57.3 | 55.8 | [86] |
| Arabica | Costa Rica | 2 | 10 | 100 °C | 2 min | 50.7–59.0 | 57.1 | [86] |
| Arabica | USA | 1 | 7 | 95 °C | ~3 min | - | 60.2 | [4] |
| Blend | Brazil | 40 | 10 | 90 °C | 4 min | 71.8–44.1 | 59.9 | [85] |
| Blend | Brazil | 7 | 10 | 92.3 °C | Nr | 39.4–51.8 | 45.6 | [87] |
| Blend | UK | 1 | Nr | Nr | Nr | - | 55.0 | [88] |
| Blend | USA | 26 | Nr | Nr | Nr | Nr | 51.1 | [89] |
| Nr | USA | 1 | Nr | Nr | Nr | - | 52.0 | [90] |
| Nr | USA | 14 | Nr | Nr | Nr | 30.3–54.8 | 39.7 | [85] |
| Nr | USA | 7 | 10 | 100 °C | 2 min | Nr | 131.0 | [91] |
| Electric dripper | ||||||||
| Arabica | Guatemala | 1 | 6 | 90 °C | 6 min | - | 100.8 | [92] |
| Arabica | USA | 1 | 7 | 95 °C | 4 min | - | 76.5 | [4] |
| Robusta | Vietnam | 1 | 6 | 90 °C | 6 min | - | 200.8 | [92] |
| Robusta | Spain | 1 | 6 | 90 °C | 7 min | - | 110.0 | [93] |
| Blend | Brazil | 40 | 10 | 90 °C | 4 min | 100.9–138.3 | 119.6 | [85] |
| Blend | Spain | 2 | 6 | 90 °C | 7 min | 22.0–105.0 | 63.5 | [93] |
| Espresso | ||||||||
| Arabica | USA | 1 | 7 | 90 °C | 28 s | - | 72.2 | [4] |
| Arabica | Ethiopia | 1 | 14 | 92 °C | 30 s | - | 410.0 | [94] |
| Arabica | Ethiopia | 1 | 18 | 92 °C | 25 s | - | 420.0 | [94] |
| Arabica | Guatemala | 1 | 17.5 | 90 °C | 24 s | - | 100.7 | [92] |
| Arabica | Ethiopia | 1 | 18 | 92 °C | 25 s | - | 420.0 | [94] |
| Arabica | Guatemala | 1 | 17.5 | 90 °C | 24 s | - | 100.7 | [92] |
| Arabica | Spain | 1 | 18.7 | 96 °C | 21 s | - | 209.0 | [95] |
| Robusta | Vietnam | 1 | 17.5 | 90 °C | 24 s | - | 177.3 | [92] |
| Robusta | Spain | 1 | 18.7 | 96 °C | 21 s | - | 296.0 | [95] |
| Robusta | Spain | 1 | 17.5 | 90 °C | 30 s | - | 375.0 | [93] |
| Blend | Scotland | 20 | Nr | Nr | Nr | 160.4–650.0 | 378.0 | [96] |
| Blend | Croatia | 4 | 14 | 95–97 °C | Nr | 97.7–190.0 | 142.0 | [97] |
| Blend | Australia | 97 | Nr | Nr | Nr | 58.0–700.0 | 247.3 | [98] |
| Blend | Brazil | 40 | 10 | 90 °C | 28 s | 159.5–233.2 | 196.4 | [85] |
| Blend | Spain | 2 | 17.5 | 90 °C | 30 s | 63.0–241.0 | 152.0 | [93] |
| Blend | Spain | 1 | 18.7 | 96 °C | 21 s | - | 288.0 | [95] |
| Nr | USA | 1 | Nr | Nr | Nr | - | 123.0 | [90] |
| Nr | USA | 6 | Nr | Nr | Nr | 138.3–290.2 | 197.1 | [92] |
| Nr | USA | 27 | Nr | Nr | Nr | - | 250.0 | [91] |
| Regular/Nr | Italy | 1 | Nr | Nr | Nr | - | 317.1 | [30] |
| Lungo/Nr | Italy | 1 | Nr | Nr | Nr | - | 171.3 | [30] |
| Ristretto/Nr | Italy | 1 | Nr | Nr | Nr | - | 533.4 | [30] |
| Mocha | ||||||||
| Arabica | USA | 1 | 7 | 95 °C | 10 min | - | 92.4 | [4] |
| Arabica | Guatemala | 1 | 8 | 93 °C | 10 min | - | 156.4 | [92] |
| Arabica | Ethiopia | 1 | 10 | 90 °C | Nr | - | 128.0 | [94] |
| Robusta | Spain | 1 | 8 | 93 °C | 10 min | - | 192.0 | [93] |
| Robusta | Vietnam | 1 | 8 | 93 °C | 10 min | - | 226.8 | [92] |
| Blend | Spain | 2 | 8 | 93 °C | 10 min | 28.0–159.0 | 93.5 | [93] |
| Infusion bag | ||||||||
| Arabica | Japan | 4 | 2.5 | 95 °C | 5 min | - | 43.2 | [4] |
| French press | ||||||||
| Arabica | Guatemala | 1 | 8 | 98 °C | 5 min | - | 118.3 | [92] |
| Arabica | Hawaii | 4 | 10 | 98 °C | 6 min | 97.0–104.9 | 100.5 | [99] |
| Arabica | USA | 1 | 7 | 90 °C | 5 min | - | 75.0 | [4] |
| Arabica | Portugal | 1 | 13 | 100 °C | 2.5 min | 84.0–106.0 | 95.0 | [99] |
| Arabica | Ethiopia | 1 | 10 | 95 °C | 5 min | - | 52.0 | [94] |
| Robusta | Vietnam | 1 | 8 | 98 °C | 5 min | - | 156.4 | [92] |
| Robusta | Spain | 1 | 8 | 98 °C | 5 min | - | 136.0 | [93] |
| Blend | Spain | 2 | 8 | 98 °C | 5 min | 20.0–112.0 | 66.0 | [93] |
| Aero press | ||||||||
| Arabica | Ethiopia | 1 | 6.6 | 93 °C | 1 min | - | 78.0 | [94] |
| Cold brewing | ||||||||
| Arabica | USA | 1 | 7 | 95 °C followed by 10 °C | 12 h | - | 52.3 | [4] |
| Arabica | USA | 1 | 7 | 10 °C | 12 h | - | 51.2 | [4] |
| Arabica | Hawaii | 4 | 10 | 25 °C | 24 h | 99.0–123.0 | 112.0 | [99] |
| Arabica | Ethiopia | 1 | 10 | 25 °C | 6 h | - | 125.0 | [94] |
| Turkish coffee | ||||||||
| Arabica | USA | 1 | 7 | 90 °C | 5 min | - | 85.3 | [4] |
| Blend | Croatia | 3 | 8 | 98 °C | 5 min | 190.0–260.0 | 233.0 | [94] |
| Boiled coffee | ||||||||
| Arabica | USA | 1 | 7 | 95 °C | 5 min | - | 85.0 | [4] |
| Arabica | Brazil | 3 | 10 | 100 °C | Nr | 110.8–122.5 | 114.7 | [100] |
| Conilon | Brazil | 3 | 10 | 100 °C | Nr | 171.3–192.0 | 179.8 | [100] |
| Soluble or instant coffee beverage | ||||||||
| - | Brazil | 10 | 2 | 90 °C | - | 257.3–280.7 | 269.0 | [85] |
| - | Nr | 2 | 1 | hot water | - | Nr | 212 | [21] |
| - | Nr | 2 | 2.2 | hot water | - | Nr | 74.5 | [101] |
| - | Nr | 2 | 1 | hot water | - | Nr | 246.8 | [102] |
| - | UK | Nr | Nr | hot water | - | - | 39.0 | [93] |
| - | USA | 26 | Nr | hot water | - | - | 44.0 | [97] |
| - | USA | 20 | Nr | hot water | - | - | 39.0 | [90] |
| Read to drink coffee beverage | ||||||||
| Nr | Japan | 4 | - | - | - | - | 57.1 | [4] |
| Subjects’ Origin | Sample | Study Type | Conclusions | Reference |
|---|---|---|---|---|
| Neurodegenerative diseases | ||||
| Finland | n = 6.710 | Large prospective study (follow-up for 22 years) | Results support the hypothesis that coffee consumption reduces the risk of Parkinson’s disease. | [301] |
| Greece | n = 26.173 | Population-based prospective cohort (follow-up for 3–5 years) | Results support the hypothesis that coffee consumption reduces the risk of Parkinson’s disease. | [302] |
| Denmark | n = 1.876 | Large case-control study | Moderate coffee intake (3.1–5 cups/day) was associated with a lower odds ratio for Parkinson’s disease. | [303] |
| Mostly from USA, Europe, and Asia | - | Dose-response meta-analysis | Non-linear relationship between coffee intake and the risk of Parkinson’s disease was found, with maximum protection effect at approximately 3 cups/day and no improvement after that. | [304] |
| Canada | n = 6.434 | Prospective analysis of risk factors (follow-up for 5 years) | Daily coffee consumption decreased the risk of Alzheimer’s disease by 31% during the follow-up. | [305] |
| Finland | n = 1.409 | Population-based cohort study (follow-up for 21 years) | Coffee drinkers at midlife had a lower risk of dementia and Alzheimer’s disease later in life compared to those who drank no or little coffee, with lower risk (65% decreased) in those who drank 3–5 cups per day. | [269] |
| Mostly from USA, Europe, and Asia | - | Systematic review | Findings indicate that moderate coffee/caffeine intake decreases the risk of cognitive impairment/decline and dementia/Alzheimer’s disease later in life. | [306] |
| Liver diseases | ||||
| Mostly from USA, Europe, and Asia | - | Systematic review and dose-response meta-analysis | Regular intake of three cups of caffeinated and decaffeinated coffees was associated with reductions of 27% and 14% in the risk of hepatocellular carcinoma, respectively. | [307] |
| USA, France and Brazil | - | Systematic review and meta-analysis | Decreased risk of advanced liver fibrosis and liver inflammation among hepatitis C virus-infected patients who consumed caffeine on a regular basis. | [308] |
| USA | n = 5.944 | Population-based cohort study | Dose–effect relationship between coffee and caffeine consumption and decrease in aminotransferase (ALT) levels, reducing the prevalence of above-normal ALT value by 50% for two cups/day, and by 66% for three cups/day. | [309] |
| Italy | n = 732 | Population-based study | Coffee caffeine may inhibit the onset of alcoholic and nonalcoholic liver cirrhosis. | [310] |
| Sample | Type of Study | Coffee/Caffeine Doses | Conclusions | Reference |
|---|---|---|---|---|
| Anxiety | ||||
| Healthy subjects (n = 13) | Clinical study | 9 mg of caffeine/kg body weight (bw) | High caffeine doses (> 400 mg/day) significantly increased some anxiety aspects. | [442] |
| Subjects with panic disorders (n = 28), performance social anxiety disorder (n= 19), and healthy subjects (n = 26) | Clinical study | 480 mg of caffeine | Patients with panic disorders and performance social anxiety disorders had a higher number of induced panic attacks, some specific anxiety symptoms, and a more severe anxiety response than healthy volunteers. | [443] |
| Cardiovascular system | ||||
| Normotensive subjects | Critical review and meta-analysis | Single doses 200–250 mg of caffeine | Increase in systolic blood pressure by 3–14 mm Hg and diastolic blood pressure by 4–13 mm Hg. | [345] |
| Hypertensive subjects | Systematic review and meta-analysis | Single doses 200–300 mg of caffeine | Induced average increases of 8.1 mm Hg in systolic and 5.7 mm Hg diastolic blood pressure, observed within the first 60 min after intake and persisting for 180 min, on average. | [444] |
| Hypertensive subjects | Systematic review and meta-analysis | Doses ≥ 410 mg of caffeine/day for at least 7 days | No effect on heart rate associated with consumption as compared to those who consumed < 410 mg/day. | [445] |
| Glucose metabolism and insulin resistance | ||||
| Healthy subjects | Systematic review and meta-analysis | 3–6 mg of caffeine/kg bw | Acute caffeine ingestion reduces insulin sensitivity. | [446] |
| People with type II diabetes | Systematic review of randomized controlled trials | ~200–500 mg of caffeine | Caffeine intake increased blood glucose concentrations by 16–28% of the area under the curve (AUC) and insulin concentrations by 19–48% of the AUC. | [447] |
| Bone and calcium balance | ||||
| Healthy postmenopausal women (n = 205) | Randomized clinical trial | 280–420 mg of caffeine/day | Daily consumption of 280–420 mg caffeine/day may accelerates bone loss from the spine and total body in women with calcium intakes below the recommended dietary allowance of 800 mg. | [448] |
| Healthy women (n = 61.433) | Longitudinal population-based | ≥ 560 mg of caffeine/day | Not associated with higher rate of fractures and bone mineral density | [385] |
| Healthy women | Systematic review and meta-analysis | 2–8 cups of coffee/day | The fracture risk was 14% higher in women and 24% lower in men with the highest level of coffee consumption. | [387] |
| Fertility andreproductive and developmental effects | ||||
| Healthy women | Systematic review and dose-response meta-analysis | ≥600 mg of caffeine/day | Coffee/caffeine consumption is associated with a significantly increased risk of spontaneous abortion. | [449] |
| Healthy pregnant women | Systematic review | up to 300 mg of caffeine/day | Lack of birth defects following consumption of caffeine in healthy pregnant women. | [354] |
| Carcinogenesis | ||||
| Healthy subjects | Dose-response meta-analysis | 1–3 cups of coffee/day | Caffeinated but not decaffeinated coffee consumption was negatively associated with basal cell carcinoma risk. | [450] |
| Healthy subjects | Meta-analysis | 5 cups of coffee/day | Caffeinated but not decaffeinated coffee consumtion might have chemo-preventive effects against malignant melanoma risk, coffee. | [451] |
| Agency or Study | Safe Limit of Caffeine Consumption (mg/day or mg/kg Body Weight – bw/day) | Equivalent to (Approximately) |
|---|---|---|
| Adults | ||
| USA: Nawrot et al. [232] | 1cup of 100 mL espresso coffee + 2 cups of 100 mL manual dripped coffee or 6 cups of 100 mL manual dripped coffee or 3 cups of 100 mL French press coffee + 1 cup 100 mL cold brewing coffee | |
| Canada: Health Canada [462] | ||
| South Korea: Korean Food and Drug Administration [463] | 400 mg/day | |
| Belgium: Belgium Superior Health Council [325] | ||
| Europe: European Food Safety Authority [126] | ||
| Europe: European Food Safety Authority [126] | Single doses of up to 200 mg | 3 cups of 100 mL manual dripped coffee or 1 cup of 100 mL soluble coffee + 1 cup 100 mL cold brewing coffee or 2 cups of 100 mL aero press coffee + 1 cup of 100 mL infusion coffee bag |
| Reproductive-aged women | ||
| International Life Science Institute (ILSI) [464] | Less than 5 to 6 mg/kg bw/day | (amounts estimated for a 70 kg bw woman) 1cup of 100mL espresso coffee + 1 cup of 100 mL French press coffee or 2 cups of 100 mL mocha coffee |
| Pregnant and lactating woman | ||
| USA: Nawrot et al. [232] | 1 cup of 100 mL espresso coffee + 1 cup of 100 mL dripped coffee or 2 cups of 100 mL mocha coffee or 2 cups of 100 mL electric dripper coffee + 1 cup 100 mL ready to drink coffee beverage | |
| New Zealand: New Zealand Ministry of Health [452] | ||
| South Korea: Korean Food and Drug Administration [463] | 300 mg/day | |
| Belgium: Belgium Superior Health Council [325] | ||
| United Kingdom: UK Food Standard Agency [465] Europe: European Food Safety Authority [126] | 200 mg/day | 3 cups of 100 mL manual dripped coffee or 1 cup of 100 mL soluble coffee + 1 cup 100mL cold brewing coffee or 2 cups of 100 mL aero press coffee + 1 cup of 100mL infusion coffee bag |
| Children | ||
| USA: Nawrot et al. [232] Canada: Health Canada [462] | (amounts estimated for a child a 5-8-year-old, with 22 kg bw) 1 cup of 100 mL manual dripped coffee or 1 cup of 100 mL ready to drink coffee beverage | |
| South Korea: Korean Food and Drug Administration [463] | < 2.5 mg/kg bw/day | |
| Belgium: Belgium Superior Health Council [325] | ||
| UK: Knight et al. [466] | 45 mg/day (up to 4 years) | 1 cup of 50 mL cold brewing coffee or 1 cup of 75 mL of manual dripped coffee |
| New Zealand: New Zealand Ministry of Health [452] | 95 mg/day (aged 5-12 years) | 1 cup of 100mL manual dripped coffee + 1 cup of 50mL ready to drink coffee beverage |
| Europe: European Food Safety Authority [126] | < 3.0 mg/kg bw/day | (amounts estimated for a 5-8-year-old child, with 22 kg bw) 1 cup of 100 mL manual dripped coffee or 1 cup of 100 mL ready to drink coffee beverage |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
dePaula, J.; Farah, A. Caffeine Consumption through Coffee: Content in the Beverage, Metabolism, Health Benefits and Risks. Beverages 2019, 5, 37. https://doi.org/10.3390/beverages5020037
dePaula J, Farah A. Caffeine Consumption through Coffee: Content in the Beverage, Metabolism, Health Benefits and Risks. Beverages. 2019; 5(2):37. https://doi.org/10.3390/beverages5020037
Chicago/Turabian StyledePaula, Juliana, and Adriana Farah. 2019. "Caffeine Consumption through Coffee: Content in the Beverage, Metabolism, Health Benefits and Risks" Beverages 5, no. 2: 37. https://doi.org/10.3390/beverages5020037
APA StyledePaula, J., & Farah, A. (2019). Caffeine Consumption through Coffee: Content in the Beverage, Metabolism, Health Benefits and Risks. Beverages, 5(2), 37. https://doi.org/10.3390/beverages5020037