The Effect of Juicing Methods on the Phytochemical and Antioxidant Characteristics of the Purple Prickly Pear (Opuntia ficus indica)—Preliminary Findings on Juice and Pomace
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design
2.2. Chemicals
2.3. Sample Preparation
2.4. Juicing Methods and Pomace Separation
2.5. Titratable Acidity (TA), Total Soluble Solids (TSS) and pH
2.6. Extraction of Phytochemicals from Pomace
2.7. Bioactive Content (Total Betalain Content (TBC), Total Polyphenolic Content (TPC) and Total Flavonoid Content (TFC))
2.7.1. Total Betalain Content
2.7.2. Total Phenolic (TPC) and Flavonoid (TFC) Content
2.7.3. Antioxidant Characteristics (DPPH and FRAP)
2.7.4. Vitamin C Content
2.8. Statistical Analysis
3. Results and Discussion
3.1. Physiochemical Characteristics
3.1.1. Juice Yield
3.1.2. Titratable Acidity
3.1.3. Total Soluble Solids (TSS)
3.1.4. pH
3.2. The Phytochemical Content of Prickly Pear Fruit Juice and Methanolic Pomace Extracts
3.2.1. Total Polyphenol Content (TPC) of Prickly Pear Fruit Juice and Methanolic Pomace Extract
3.2.2. The Total Flavonoid Content (TFC) of Prickly Pear Fruit Juice and Methanolic Pomace Extracts
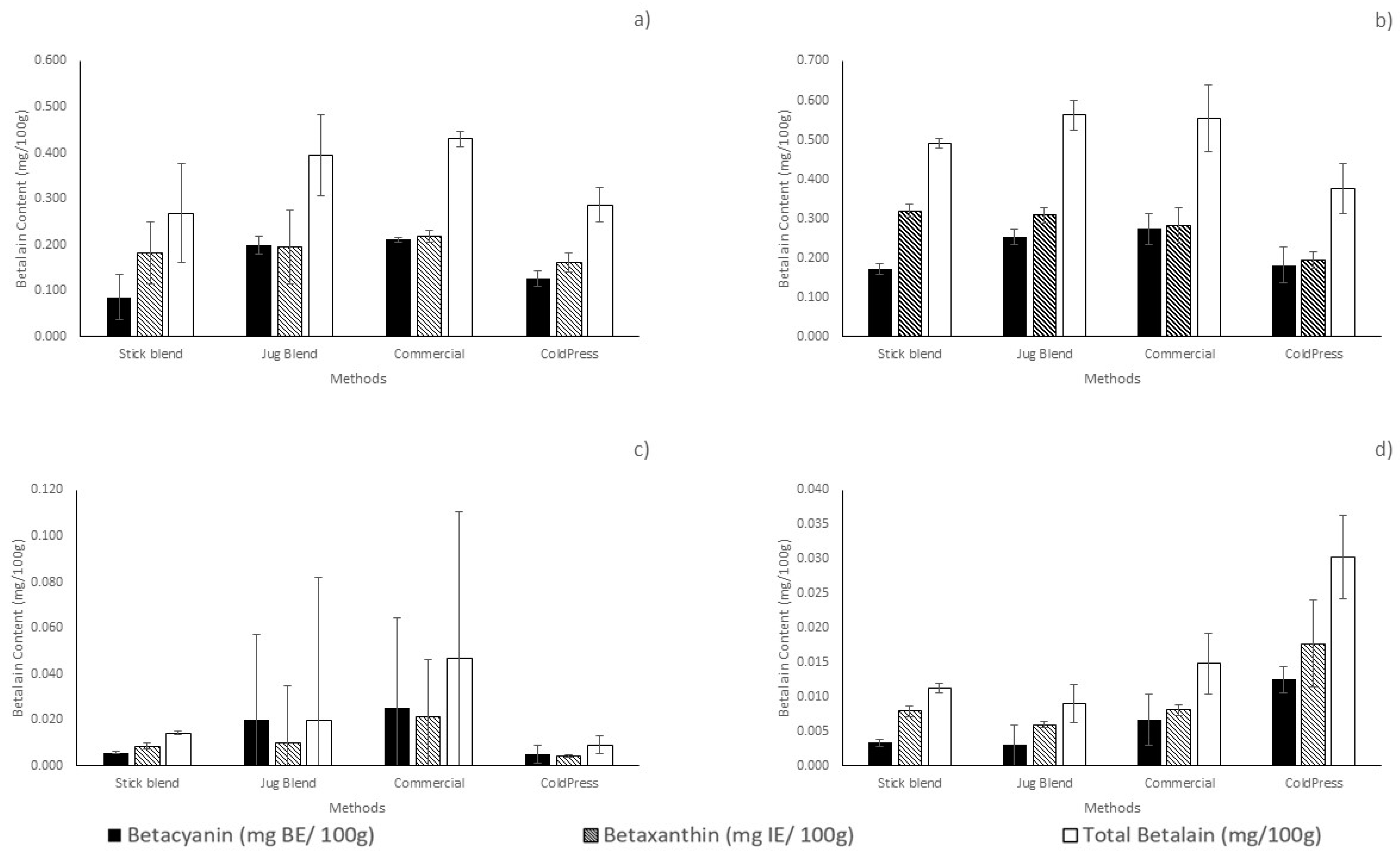
3.2.3. Total Betalain, Betacyanin and Betaxanthin Content of Prickly Pear Fruit Juice and Methanolic Pomace Extracts
Total Betalain, Betacyanin and Betaxanthin Content of Prickly Pear Fruit Juice
Total Betalain, Betacyanin and Betaxanthin Content of Prickly Pear Methanolic Pomace Extracts
3.3. Antioxidant Characteristics (DPPH, FRAP and Vitamin C)
3.3.1. Free Radical Capacity (DPPH) of Prickly Pear Fruit Juice and Methanolic Pomace Extracts
3.3.2. Antioxidant Capacity by Ferric Reducing Antioxidant Power (FRAP) of Prickly Pear Fruit Juice and Methanolic Pomace Extracts
3.3.3. Vitamin C Content of Prickly Pear Fruit Juice and Methanolic Pomace Extracts
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- El-Mostafa, K.; El Kharrassi, Y.; Badreddine, A.; Andreoletti, P.; Vamecq, J.; El Kebbaj, M.; Latruffe, N.; Lizard, G.; Nasser, B.; Cherkaoui-Malki, M. Nopal cactus (Opuntia ficus-indica) as a source of bioactive compounds for nutrition, health and disease. Molecules 2014, 19, 14879–14901. [Google Scholar] [CrossRef] [PubMed]
- Russell, C.E.; Felker, P. The prickly-pears (Opuntia spp., Cactaceae): A source of human and animal food in semiarid regions. Econ. Bot. 1987, 41, 433–445. [Google Scholar] [CrossRef]
- Teles, F.F.F. Nutrient Analysis of Prickly Pear (Opuntia Ficus Indica, Linn); ProQuest Dissertations Publishing; University of Arizona: Tucson, AZ, USA, 1977; Volume 1, pp. 1–23. [Google Scholar]
- Department of Environment. Opuntia spp.; Australian Government: Canberra, Australia, 2015.
- Butera, D.; Tesoriere, L.; Di Gaudio, F.; Bongiorno, A.; Allegra, M.; Pintaudi, A.M.; Kohen, R.; Livrea, M.A. Antioxidant activities of sicilian prickly pear (Opuntia Ficus Indica) fruit extracts and reducing properties of its betalains: Betanin and indicaxanthin. J. Agric. Food Chem. 2002, 50, 6895–6901. [Google Scholar] [CrossRef] [PubMed]
- El Kossori, R.L.; Villaume, C.; El Boustani, E.; Sauvaire, Y.; Méjean, L. Composition of pulp, skin and seeds of prickly pears fruit (Opuntia ficus indica sp.). Plant Foods Hum. Nutr. 1998, 52, 263–270. [Google Scholar] [CrossRef] [PubMed]
- Wills, R.B.H.; Lim, J.S.K.; Greenfield, H. Composition of Australian foods. 31. Tropical and sub-tropical fruit. Food Technol. Aust. 1986. Available online: http://agris.fao.org/agris-search/search.do;jsessionid=EDCCC9840C3EAA0E883A20661AF2206F?request_locale=ar&recordID=US201301456640&query=&sourceQuery=&sortField=&sortOrder=&agrovocString=&advQuery=¢erString=&enableField= (accessed on 18 March 2019).
- Zou, D.; Brewer, M.; Garcia, F.; Feugang, J.M.; Wang, J.; Zang, R.; Liu, H.; Zou, C. Cactus pear: A natural product in cancer chemoprevention. Nutr. J. 2005, 4, 25. [Google Scholar] [CrossRef] [PubMed]
- Chavez-Santoscoy, R.A.; Gutierrez-Uribe, J.A.; Serna-Saldivar, S.O. Phenolic composition, antioxidant capacity and in vitro cancer cell cytotoxicity of nine prickly pear (Opuntia spp.) juices. Plant Foods Hum. Nutr. 2009, 64, 146–152. [Google Scholar] [CrossRef]
- Hahm, S.; Park, J.; Son, Y.-S. Opuntia humifusa partitioned extracts inhibit the growth of U87MG human glioblastoma cells. Plant Foods Hum. Nut. 2010, 65, 247–252. [Google Scholar] [CrossRef] [PubMed]
- Chang, S.; Hsieh, C.-L.; Yen, G.-C. The protective effect of Opuntia dillenii Haw fruit against low-density lipoprotein peroxidation and its active compounds. Food Chem. 2008, 106, 569–575. [Google Scholar] [CrossRef]
- Budinsky, A.; Wolfram, R.; Oguogho, A.; Efthimiou, Y.; Stamatopoulos, Y.; Sinzinger, H. Regular ingestion of opuntia robusta lowers oxidation injury. Prostaglandins Leukot. Essent. Fatty Acids 2001, 65, 45–50. [Google Scholar] [CrossRef]
- Galati, E.M.; Mondello, M.R.; Giuffrida, D.; Dugo, G.; Miceli, N.; Pergolizzi, S.; Taviano, M.F. Chemical characterization and biological effects of Sicilian Opuntia ficus indica (L.) mill. Fruit juice: Antioxidant and antiulcerogenic activity. J Agric. Food Chem. 2003, 51, 4903. [Google Scholar] [CrossRef]
- Chao, J.C.; Hung, H.; Chen, S.; Fang, C. Effects of Ginkgo biloba extract on cytoprotective factors in rats with duodenal ulcer. World J. Gastroenterol. 2004, 10, 560–566. [Google Scholar] [CrossRef]
- Tadićnja, V.M.; Jeremic, I.; Dobric, S.; Isakovic, A.; Markovic, I.; Trajkovic, V.; Bojovic, D.; Arsic, I. Anti-inflammatory, gastroprotective, and cytotoxic effects of sideritis scardica extracts. Planta Med. 2012, 78, 415–427. [Google Scholar] [CrossRef]
- Beil, W.; Birkholz, C.; Sewing, K.-F. Effects of flavonoids on parietal cell acid secretion, gastric mucosal prostaglandin production and Helicobacter pylori growth. Arzneimittelforschung 1995, 45, 697–700. [Google Scholar]
- Ito, S.; Lacy, E.; Rutten, M.; Critchlow, J.; Silen, W. Rapid repair of injured gastric mucosa. Scand. J Gastroenterol. 1983, 101, 87–95. [Google Scholar]
- Trejo-Gonzalez, A.; Gabriel-Ortiz, G.; Puebla-Perez, A.M.; Huizar-Contreras, M.D.; Munguia-Mazariegos, M.R.; Mejia-Arreguin, S.; Calva, E. A purified extract from prickly pear cactus (Opuntia fuliginosa) controls experimentally induced diabetes in rats. J. Ethnopharmacol. 1996, 55, 27–33. [Google Scholar] [CrossRef]
- Frati, A.C.; Gordillo, B.E.; Altamirano, P.; Ariza, C.R.; Cortes-Franco, R.; Chavez-Negrete, A. Acute hypoglycemic effect of Opuntia streptacantha Lemaire in NIDDM. Diabetes Care 1990, 13, 455–456. [Google Scholar] [CrossRef] [PubMed]
- Frati Munari, A.C.; Vera Lastra, O.; Ariza Andraca, C.R. Evaluation of nopal capsules in diabetes mellitus. Gaceta Médica de México 1992, 128, 431–436. [Google Scholar]
- Ramierz, G.; Aguilar, C. The effect of Opuntia in lowering serum glucose among NIDDM patients. JPACD 1995. Available online: https://s3.amazonaws.com/academia.edu.documents/5989478/012proc71-78.pdf?AWSAccessKeyId=AKIAIWOWYYGZ2Y53UL3A&Expires=1552894687&Signature=uuE%2BPkWwK%2FVCoDipE289jB%2BobJg%3D&response-content-disposition=inline%3B%20filename%3DThe_effect_of_Opuntia_in_lowering_serum.pdf (accessed on 18 March 2019).
- Godard, M.P.; Ewing, B.A.; Pischel, I.; Ziegler, A.; Benedek, B.; Feistel, B. Acute blood glucose lowering effects and long-term safety of OpunDia™ supplementation in pre-diabetic males and females. J. Ethnopharmacol. 2010, 130, 631–634. [Google Scholar] [CrossRef] [PubMed]
- Onakpoya, I.J.; O‘Sullivan, J.; Heneghan, C.J. The effect of cactus pear (Opuntia ficus-indica) on body weight and cardiovascular risk factors: A systematic review and meta-analysis of randomized clinical trials. Nutrition 2015, 31, 640–646. [Google Scholar] [CrossRef] [PubMed]
- Odden, M.C.; McPherson, S.; Shlipak, M.G.; Wiese, J. Effect of Opuntia ficus indica on Symptoms of the Alcohol Hangover. Arch. Intern. Med. 2004, 164, 1334–1340. [Google Scholar] [CrossRef]
- Galati, E.M.; Mondello, M.R.; Lauriano, E.R.; Taviano, M.F.; Galluzzo, M.; Miceli, N. Opuntia ficus indica (L.) Mill. fruit juice protects liver from carbon tetrachloride-induced injury. Phytother. Res. 2005, 19, 796–800. [Google Scholar] [CrossRef] [PubMed]
- Medina, E.M.D.; Rodríguez, E.M.R.; Romero, C.D. Chemical characterization of Opuntia dillenii and Opuntia ficus indica fruits. Food Chem. 2007, 103, 38–45. [Google Scholar] [CrossRef]
- Kevers, C.; Falkowski, M.; Tabart, J.; Defraigne, J.-O.; Dommes, J.; Pincemail, J. Evolution of antioxidant capacity during storage of selected fruits and vegetables. J. Agric. Food Chem. 2007, 55, 8596–8603. [Google Scholar] [CrossRef] [PubMed]
- Piga, A. Cactus pear: A fruit of nutraceutical and functional importance. J. Prof. Assoc. Cactus 2004, 6, 9–22. [Google Scholar]
- Abdel-Hameed, S.; Nagaty, M.; Salman, M.; Bazaid, S. Phytochemicals, nutritionals and antioxidant properties of two prickly pear cactus cultivars (Opuntia ficus indica Mill.) growing in Taif, KSA. Food Chem. 2014, 160, 31–38. [Google Scholar] [CrossRef] [PubMed]
- Gouws, C.A.; D’Cunha, N.; Georgousopoulou, E.; Thomas, J.; Mellor, D.D.; Roach, P.D.; Naumovski, N. The effect of drying methods on bioactive properties in three varieties of prickly pear (Opuntia ficus indica). JNIM 2017, 8, 107. [Google Scholar] [CrossRef]
- Fellers, P. Shelf Life and Quality of Freshly Squeezed, Unpasteurized, Polyethylene-Bottled Citrus Juice. J. Food Sci. 1988, 53, 1699–1702. [Google Scholar] [CrossRef]
- Lee, H.S.; Coates, G.A. Vitamin C in frozen, fresh squeezed, unpasteurized, polyethylene-bottled orange juice: A storage study. Food Chem. 1999, 65, 165–168. [Google Scholar] [CrossRef]
- Cansino, N.; Carrera, G.; Rojas, Q.; Olivares, L.; García, E.; Moreno, E. Ultrasound processing on green cactus pear (Opuntia ficus indica) juice: Physical, microbiological and antioxidant properties. Int. J. Food Process. Technol. 2013, 4, 2. [Google Scholar] [CrossRef]
- Alimi, H.; Hfaeidh, N.; Bouoni, Z.; Sakly, M.; Rhouma, K.B. Protective effect of Opuntia ficus indica f. inermis prickly pear juice upon ethanol-induced damages in rat erythrocytes. J. Alcohol. 2012, 46, 235–243. [Google Scholar] [CrossRef]
- Pyo, Y.; Jin, Y.; Hwang, J. Comparison of the Effects of Blending and Juicing on the Phytochemicals Contents and Antioxidant Capacity of Typical Korean Kernel Fruit Juices. Prev. Nutr. Food Sci. 2014, 19, 108–114. [Google Scholar] [CrossRef]
- Ochoa-Velasco, C.E.; Guerrero-Beltrán, J.Á. Postharvest quality of peeled prickly pear fruit treated with acetic acid and chitosan. Postharvest Biol. Technol. 2014, 92, 139–145. [Google Scholar] [CrossRef]
- Gurrieri, S.; Miceli, L.; Lanza, C.M.; Tomaselli, F.; Bonomo, R.P.; Rizzarelli, E. Chemical characterization of sicilian prickly pear (Opuntia ficus indica) and perspectives for the storage of its juice. J. Agric. Food Chem. 2000, 48, 5424–5431. [Google Scholar] [CrossRef] [PubMed]
- Duru, B.; Turker, N. Changes in physical properties and chemical composition of cactus pear (Opuntia ficus-indica) during maturation. J. Prof. Assoc. Cactus Dev. 2005, 24–33. [Google Scholar]
- Saeeduddin, M.; Abid, M.; Jabbar, S.; Wu, T.; Yuan, Q.; Riaz, A.; Hu, B.; Zhou, L.; Zeng, X. Nutritional, microbial and physicochemical changes in pear juice under ultrasound and commercial pasteurization during storage. J. Food Process. Preserv. 2017, e13237. [Google Scholar] [CrossRef]
- Graciela Ruiz-Gutierrez, M.; Abel Amaya-Guerra, C.; Quintero-Ramos, A.; de Jesus Ruiz-Anchondo, T.; Alejandra Gutierrez-Uribe, J.; Gabriel Baez-Gonzalez, J.; Lardizabal-Gutierrez, D.; Campos-Venegas, K. Effect of Soluble Fiber on the Physicochemical Properties of Cactus Pear (Opuntia Ficus Indica) Encapsulated Using Spray Drying. FSB 2014, 23, 755–763. [Google Scholar] [CrossRef]
- Schabort, C.; Otto, A.; Bothma, M.; van der Gryp, P.; Marx, S. Recovery of water from cacti for use in small farming communities. AJB 2013, 12. [Google Scholar] [CrossRef]
- Chaudhary, P.R.; Jayaprakasha, G.K.; Patil, B.S. Ethylene degreening modulates health promoting phytochemicals in Rio Red grapefruit. Food Chem. 2015, 188, 77–83. [Google Scholar] [CrossRef] [PubMed]
- Horwitz, W.; Latimer, G.W.; International, A. Official Methods of Analysis of AOAC International; AOAC International: Gaithersburg, MD, USA, 2010; Volume 18. [Google Scholar]
- Moussa-Ayoub, T.E.; El-Hady, E.A.; Omran, H.T.; El-Samahy, S.K.; Kroh, L.W.; Rohn, S. Influence of cultivator and origin on the flavonol profle of fruits and cladodes from cactus Opuntia ficus-indica. Food Res. Int. 2014, 64, 863–872. [Google Scholar] [CrossRef] [PubMed]
- Prakash Maran, J.; Manikandan, S.; Mekala, V. Modeling and optimization of betalain extraction from Opuntia ficus-indica using Box–Behnken design with desirability function. Ind. Crops Prod. 2013, 49, 304–311. [Google Scholar] [CrossRef]
- Moßhammer, M.R.; Maier, C.; Stintzing, F.C.; Carle, R. Impact of Thermal Treatment and Storage on Color of Yellow-Orange Cactus Pear (Opuntia ficus-indica [L.] Mill. cv. ‘Gialla’) Juices. J. Food Sci. 2006, 71, C400–C406. [Google Scholar] [CrossRef]
- Kähkönen, M.P.; Hopia, A.I.; Vuorela, H.J.; Rauha, J.-P.; Pihlaja, K.; Kujala, T.S.; Heinonen, M. Antioxidant activity of plant extracts containing phenolic compounds. J. Agric. Food Chem. 1999, 47, 3954–3962. [Google Scholar] [CrossRef]
- Singleton, V.; Rossi, J.A. Colorimetry of total phenolics with phosphomolybdic-phosphotungstic acid reagents. AJEV 1965, 16, 144–158. [Google Scholar]
- Wu, S.; Ng, L. Antioxidant and free radical scavenging activities of wild bitter melon (Momordica charantia Linn. var. abbreviata Ser.) in Taiwan. LWT Food Sci. Technol. 2008, 41, 323–330. [Google Scholar] [CrossRef]
- Thaipong, K.; Boonprakob, U.; Crosby, K.; Cisneros-Zevallos, L.; Hawkins Byrne, D. Comparison of ABTS, DPPH, FRAP, and ORAC assays for estimating antioxidant activity from guava fruit extracts. J. Food Comp. Anal. 2006, 19, 669–675. [Google Scholar] [CrossRef]
- Benzie, I.F.; Strain, J. The ferric reducing ability of plasma (FRAP) as a measure of “antioxidant power”: The FRAP assay. Anal. Biochem. 1996, 239, 70–76. [Google Scholar] [CrossRef] [PubMed]
- Ragupathi Raja Kannan, R.; Arumugam, R.; Thangaradjou, T.; Anantharaman, P. Phytochemical constituents, antioxidant properties and p-coumaric acid analysis in some seagrasses. Food Res. Int. 2013, 54, 1229–1236. [Google Scholar] [CrossRef]
- Papoutsis, K.; Pristijono, P.; Golding, J.B.; Stathopoulos, C.E.; Scarlett, C.J.; Bowyer, M.C.; Vuong, Q.V. Impact of different solvents on the recovery of bioactive compounds and antioxidant properties from lemon (Citrus limon L.) pomace waste. Food Sci. Biotechnol. 2016, 25, 971–977. [Google Scholar] [CrossRef] [PubMed]
- Vuong, Q.V.; Hirun, S.; Chuen, T.L.; Goldsmith, C.D.; Bowyer, M.C.; Chalmers, A.C.; Phillips, P.A.; Scarlett, C.J. Physicochemical composition, antioxidant and anti-proliferative capacity of a lilly pilly (Syzygium paniculatum) extract. J. Herb. Med. 2014, 4, 134–140. [Google Scholar] [CrossRef]
- Cole, T.J. Too many digits: The presentation of numerical data. Arch. Dis. Child. 2015, 100, 608–609. [Google Scholar] [CrossRef]
- Sadler, G.D.; Murphy, P.A. pH and titratable acidity. In Food Analysis; Springer: Berlin, Germany, 2010; pp. 219–238. [Google Scholar]
- Sawaya, W.; Khatchadourian, H.; Safi, W.; Al-Muhammad, H. Chemical characterization of prickly pear pulp, Opuntia ficus-indica, and the manufacturing of prickly pear jam. IJFST 1983, 18, 183–193. [Google Scholar] [CrossRef]
- Peng, Y.; Lu, R. Analysis of spatially resolved hyperspectral scattering images for assessing apple fruit firmness and soluble solids content. Postharvest Biol. Technol. 2008, 48, 52–62. [Google Scholar] [CrossRef]
- McGlone, V.A.; Kawano, S. Firmness, dry-matter and soluble-solids assessment of postharvest kiwifruit by NIR spectroscopy. Postharvest Biol. Technol. 1998, 13, 131–141. [Google Scholar] [CrossRef]
- Amerine, M.A.; Roessler, E.B.; Ough, C.S. Acids and the Acid Taste. I. The Effect of pH and Titratable Acidity. AJEV 1965, 16, 29–37. [Google Scholar]
- Naumovski, N. Bioactive composition of plants and plant foods. In Plant Bioactive Compounds for Pancreatic Cancer; Scarlett, C.J., Vuong, Q.V., Eds.; Nova Science Pub Inc.: Hauppauge, NY, USA, 2014; pp. 81–115. [Google Scholar]
- Gengatharan, A.; Dykes, G.A.; Choo, W.S. Betalains: Natural plant pigments with potential application in functional foods. LWT Food Sci. Technol. 2015, 64, 645–649. [Google Scholar] [CrossRef]
- Sakihama, Y.; Maeda, M.; Hashimoto, M.; Tahara, S.; Hashidoko, Y. Beetroot betalain inhibits peroxynitrite-mediated tyrosine nitration and DNA strand cleavage. Free Rad. Res. 2012, 46, 93–99. [Google Scholar] [CrossRef]
- Esatbeyoglu, T.; Wagner, A.E.; Motafakkerazad, R.; Nakajima, Y.; Matsugo, S.; Rimbach, G. Free radical scavenging and antioxidant activity of betanin: Electron spin resonance spectroscopy studies and studies in cultured cells. Food Chem. Toxicol. 2014, 73, 119–126. [Google Scholar] [CrossRef]
- Shelby, M.D.; Newbold, R.R.; Tully, D.B.; Chae, K.; Davis, V.L. Assessing environmental chemicals for estrogenicity using a combination of in vitro and in vivo assays. Environ. Health Perspect. 1996, 104, 1296. [Google Scholar] [CrossRef] [PubMed]
- Netzel, M.; Stintzing, F.C.; Quaas, D.; Straß, G.; Carle, R.; Bitsch, R.; Bitsch, I.; Frank, T. Renal excretion of antioxidative constituents from red beet in humans. Food Res. Int. 2005, 38, 1051–1058. [Google Scholar] [CrossRef]
- Allegra, M.; Tesoriere, L.; Livrea, M.A. Betanin inhibits the myeloperoxidase/nitrite-induced oxidation of human low-density lipoproteins. Free Rad. Res. 2007, 41, 335–341. [Google Scholar] [CrossRef] [PubMed]
- Tesoriere, L.; Allegra, M.; Butera, D.; Livrea, M.A. Absorption, excretion, and distribution of dietary antioxidant betalains in LDLs: Potential health effects of betalains in humans. Am. J. Clin. Nutr. 2004, 80, 941–945. [Google Scholar] [CrossRef] [PubMed]
- Clemente, A.; Desai, P. Evaluation of the hematological, hypoglycemic, hypolipidemic and antioxidant properties of Amaranthus tricolor leaf extract in rat. TJPR 2011, 10, 595–602. [Google Scholar] [CrossRef]
- Sani, H.A.; Baharoom, A.; Ahmad, M.A.; Ismail, I.I. Effectiveness of Hylocereus polyrhizus extract in decreasing serum lipids and liver MDA-TBAR level in hypercholesterolemic rats. Sains Malaysiana 2009, 38, 271–279. [Google Scholar]
- Herbach, K.M.; Stintzing, F.C.; Carle, R. Betalain stability and degradation—Structural and chromatic aspects. J. Food Sci. 2006, 71, R41–R50. [Google Scholar] [CrossRef]
- Polydera, A.C.; Stoforos, N.G.; Taoukis, P.S. Comparative shelf life study and vitamin C loss kinetics in pasteurised and high pressure processed reconstituted orange juice. J. Food Eng. 2003, 60, 21–29. [Google Scholar] [CrossRef]
| Fruit Component | Stick Blend | Jug Blend | Commercial Juice | Cold Press Juicer | ||||
|---|---|---|---|---|---|---|---|---|
| WF | FF | WF | FF | WF | FF | WF | FF | |
| Juice yield (%) | 69.72 | 85.32 | 68.01 | 82.29 | 65.83 | 67.13 | 84.53 | 75.76 |
| TA (%) | 0.19 ± 0.03 | 0.16 ± 0.04 | 0.18 ± 0.03 | 0.2 ± 0.05 | 0.18 ± 0.02 | 0.24 ± 0.09 | 0.23 ± 0.01 | 0.14 ± 0.01 |
| TSS (Brix°) | 13.8 | 13.0 | 11.0 | 10.7 | 10.3 | 10.8 | 10.8 | 10.4 |
| pH | 5.65 | 5.92 | 5.72 | 5.70 | 5.83 | 5.50 | 5.66 | 5.97 |
| pH Temp (°C) | 13.5 | 15.5 | 15.8 | 14.9 | 16.2 | 15.0 | 16.4 | 17.2 |
| STICK BLEND | Fruit Preparation | Jug Blend | Fruit Preparation | Commercial Juicer | Fruit Preparation | Cold Press | Fruit Preparation | KRUSKAL-WALLIS | Style of Processing | ||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| WF | FF | p | WF | FF | p | WF | FF | p | WF | FF | p | WF | FF | p | |
| Prickly Pear fruit Juice | |||||||||||||||
| TPC (GAE) | 2301 ± 89.8 (2316) | 1516 ± 162 (1560) | 0.100 | 3031 ± 73.9 (3060) | 2432 ± 27.4 (2419) | 0.100 | 1048 ± 0.001 (1048) | 2015 ± 142 (1987) | 0.100 | 1001 ± 288 (1079) | 1524 ± 125 (1503) | 0.100 | 0.022 * | 0.025 * | 0.001 * |
| TFC (CE) | 396 ± 51.6 (409) | 346 ± 8.58 (347) | 0.400 | 396 ± 45.6 (422) | 273 ± 24.8 (269) | 0.100 | 428 ± 68.6 (409) | 266 ± 6.14 (264) | 0.100 | 291 ± 35.4 (277) | 382 ± 54.4 (370) | 0.100 | 0.091 | 0.038 * | 0.590 |
| TBC | 0.269 ± 0.107 (0.210) | 0.491 ± 0.013 (0.492) | 0.100 | 0.395 ± 0.087 (0.372) | 0.709 ± 0.105 (0.560) | 0.100 | 0.431 ± 0.017 (0.434) | 0.555 ± 0.85 (0.596) | 0.100 | 0.287 ± 0.038 (0.292) | 0.377 ± 0.064 (0.387) | 0.200 | 0.086 | 0.053 | 0.514 |
| Betacyanin (BE) | 0.086 ± 0.049 (0.081) | 0.172 ± 0.013 (0.165) | 0.100 | 0.199 ± 0.019 (0.205) | 0.563 ± 0.038 (0.236) | 0.100 | 0.212 ± 0.006 (0.215) | 0.273 ± 0.040 (0.293) | 0.100 | 0.126 ± 0.017 (0.132) | 0.182 ± 0.045 (0.196) | 0.200 | 0.029 * | 0.036 * | 0.551 |
| Betaxanthin (IE) | 0.182 ± 0.067 (0.169) | 0.319 ± 0.017 (0.313) | 0.100 | 0.196 ± 0.080 (0.194) | 0.309 ± 0.020 (0.314) | 0.100 | 0.219 ± 0.012 (0.219) | 0.282 ± 0.045 (0.303) | 0.200 | 0.161 ± 0.021 (0.160) | 0.195 ± 0.021 (0.191) | 0.200 | 0.459 | 0.053 | 0.178 |
| Methanolic (70%) Pomace extract | |||||||||||||||
| TPC (GAE) | 44.5 ± 0.072 (44.4) | 25.8 ± 0.444 (25.7) | 0.100 | 23.0 ± 17.1 (32.7) | 28.3 ± 0.494 (28.5) | 0.700 | 41.96 ± 0.106 (42.0) | 37.2 ± 0.327 (37.3) | 0.100 | 41.6 ± 0.049 (41.6) | 40.2 ± 0.024 (40.2) | 0.100 | 0.015 * | 0.015 * | 0.039 * |
| TFC (CE) | 12.78 ± 1.34 (13.3) | 9.86 ± 0.048 (9.58) | 0.100 | 7.92 ± 0.417 (7.92) | 9.86 ± 0.636 (10.0) | 0.100 | 9.19 ± 0.868 (7.92) | 8.75 ± 1.10 (8.33) | 0.400 | 8.75 ± 1.10 (8.33) | 10.3 ± 2.30 (9.17) | 0.400 | 0.066 | 0.476 | 0.101 |
| TBC | 0.014 ± 0.001 (0.014) | 0.011 ± 0.001 (0.011) | 0.100 | 0.040 ± 0.049 (0.012) | 0.009 ± 0.003 (0.008) | 0.200 | 0.047 ± 0.064 (0.100) | 0.015 ± 0.005 (0.012) | 0.700 | 0.898 ± 2.29 (0.008) | 0.030 ± 0.006 (0.029) | 0.100 | 0.430 | 0.034 * | 0.551 |
| Betacyanin (BE) | 0.006 ± 0.001 (0.006) | 0.004 ± 0.001 (0.004) | 0.100 | 0.023 ± 0.0307 (0.008) | 0.003 ± 0.003 (0.002) | 0.200 | 0.026 ± 0.039 (0.003) | 0.007 ± 0.004 (0.005) | 0.700 | 0.029 ± 0.485 (0.004) | 0.013 ± 0.003 (0.012) | 0.100 | 0.730 | 0.047 * | 0.319 |
| Betaxanthin (IE) | 0.009 ± 0.002 (0.009) | 0.008 ± 0.001 (0.008) | 0.400 | 0.016 ± 0.018 (0.008) | 0.006 ± 0.001 (0.006) | 0.700 | 0.021 ± 0.025 (0.007) | 0.008 ± 0.001 (0.008) | 0.700 | 0.009 ± 0.004 (0.004) | 0.018 ± 0.006 (0.014) | 0.100 | 0.148 | 0.017 * | 0.630 |
| Stick Blend | Fruit Preparation | Jug Blend | Fruit Preparation | Commercial Juicer | Fruit Preparation | Cold Press | Fruit Preparation | KRUSKAL, WALLIS | Style of Processing | ||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| WF | FF | p | WF | FF | p | WF | FF | p | WF | FF | p | WF | FF | p | |
| Prickly Pear fruit Juice | |||||||||||||||
| DPPH (TE) | 639 ± 198 (693) | 667 ± 126 (660) | 1.000 | 458 ± 74.2 (444) | 709 ± 105 (790) | 0.100 | 571 ± 69.3 (562) | 753 ± 15.1 (855) | 0.100 | 707 ± 408 (484) | 851 ± 437 (1044) | 1.00 | 0.468 | 0.826 | 0.590 |
| FRAP (TE) | 13500 ± 1300 (12800) | 4800 ± 265 (4700) | 0.100 | 7833 ± 416 (7700) | 4767 ± 208 (365) | 0.100 | 9900 ± 1044 (9400) | 29733 ± 153 (297) | 0.100 | 5967 ± 351 (277) | 9133 ± 611 (9000) | 0.100 | 0.016 * | 0.024 * | 0.039 * |
| Vitamin C(AAE) | 0.158 ± 0.002 (0.158) | 0.112 (0.112) | 0.100 | 0.135 ± 0.001 (0.135) | 0.093 ± 0.002 (2.89) | 0.100 | 0.121 ± 0.001 (0.121) | 0.097 ± 0.001 (2.48) | 0.100 | 0.167 ± 0.001 (0.166) | 0.120 ± 0.002 (0.120) | 0.100 | 0.015 * | 0.016 * | 0.630 |
| Methanolic (70%) Pomace extract | |||||||||||||||
| DPPH (TE) | 883 ± 1.39 (883) | 804 ± 3.28 (803) | 0.100 | 802 ± 1.89 (803) | 791 ± 2.78 (790) | 0.100 | 810 ± 12.9 (803) | 855 ± 0.910 (855) | 0.100 | 898 ± 2.29 (898) | 824 ± 2.73 (923) | 0.100 | 0.024 * | 0.016 * | 0.028 * |
| FRAP (TE) | 344 ± 4.04 (342) | 383 ± 2.082 (382) | 0.100 | 210 ± 3.06 (209) | 365 ± 2.52 (365) | 0.100 | 211 ± 3.00 (211) | 297 ± 0.578 (297) | 0.100 | 229 ± 3.06 (230) | 333 ± 1.53 (333) | 0.100 | 0.023 * | 0.015 * | 0.024 * |
| Vitamin C(AAE) | 4.28 ± 0.159 (4.35) | 1.94 ± 0.131 (0.011) | 0.100 | 4.52 ± 0.061 (4.53) | 2.9 ± 0.04 (2.89) | 0.100 | 3.53 ± 0.144 (3.60) | 2.52 ± 0.111 (2.48) | 0.100 | 2.78 ± 0.064 (2.78) | 2.98 ± 0.633 (2.95) | 0.100 | 0.016 * | 0.022 * | 0.266 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gouws, C.A.; Georgouopoulou, E.; Mellor, D.D.; Naumovski, N. The Effect of Juicing Methods on the Phytochemical and Antioxidant Characteristics of the Purple Prickly Pear (Opuntia ficus indica)—Preliminary Findings on Juice and Pomace. Beverages 2019, 5, 28. https://doi.org/10.3390/beverages5020028
Gouws CA, Georgouopoulou E, Mellor DD, Naumovski N. The Effect of Juicing Methods on the Phytochemical and Antioxidant Characteristics of the Purple Prickly Pear (Opuntia ficus indica)—Preliminary Findings on Juice and Pomace. Beverages. 2019; 5(2):28. https://doi.org/10.3390/beverages5020028
Chicago/Turabian StyleGouws, Caroline A., Ekavi Georgouopoulou, Duane D. Mellor, and Nenad Naumovski. 2019. "The Effect of Juicing Methods on the Phytochemical and Antioxidant Characteristics of the Purple Prickly Pear (Opuntia ficus indica)—Preliminary Findings on Juice and Pomace" Beverages 5, no. 2: 28. https://doi.org/10.3390/beverages5020028
APA StyleGouws, C. A., Georgouopoulou, E., Mellor, D. D., & Naumovski, N. (2019). The Effect of Juicing Methods on the Phytochemical and Antioxidant Characteristics of the Purple Prickly Pear (Opuntia ficus indica)—Preliminary Findings on Juice and Pomace. Beverages, 5(2), 28. https://doi.org/10.3390/beverages5020028







