Evolution of Phenolic Compound Profiles and Antioxidant Activity of Syrah Red and Sparkling Moscatel Wines Stored in Bottles of Different Colors
Abstract
:1. Introduction
2. Materials and Methods
2.1. Chemicals
2.2. Samples
2.3. Basic Parameters, CIE L*a*b* Color, Total Phenolic and Monomeric Anthocyanins
2.4. Antioxidant Activity In Vitro
2.5. Determination of Phenolic Compounds Profile by RP-HPLC/DAD/FD
2.6. Statistical Analysis
3. Results and Discussion
3.1. Basic Parameters of Wines
3.2. Color Evolution Based on the CIE L*a*b* System
3.3. Evolution of the Individual Phenolic Compounds
3.3.1. Sparkling Moscatel
3.3.2. Syrah Red Wine
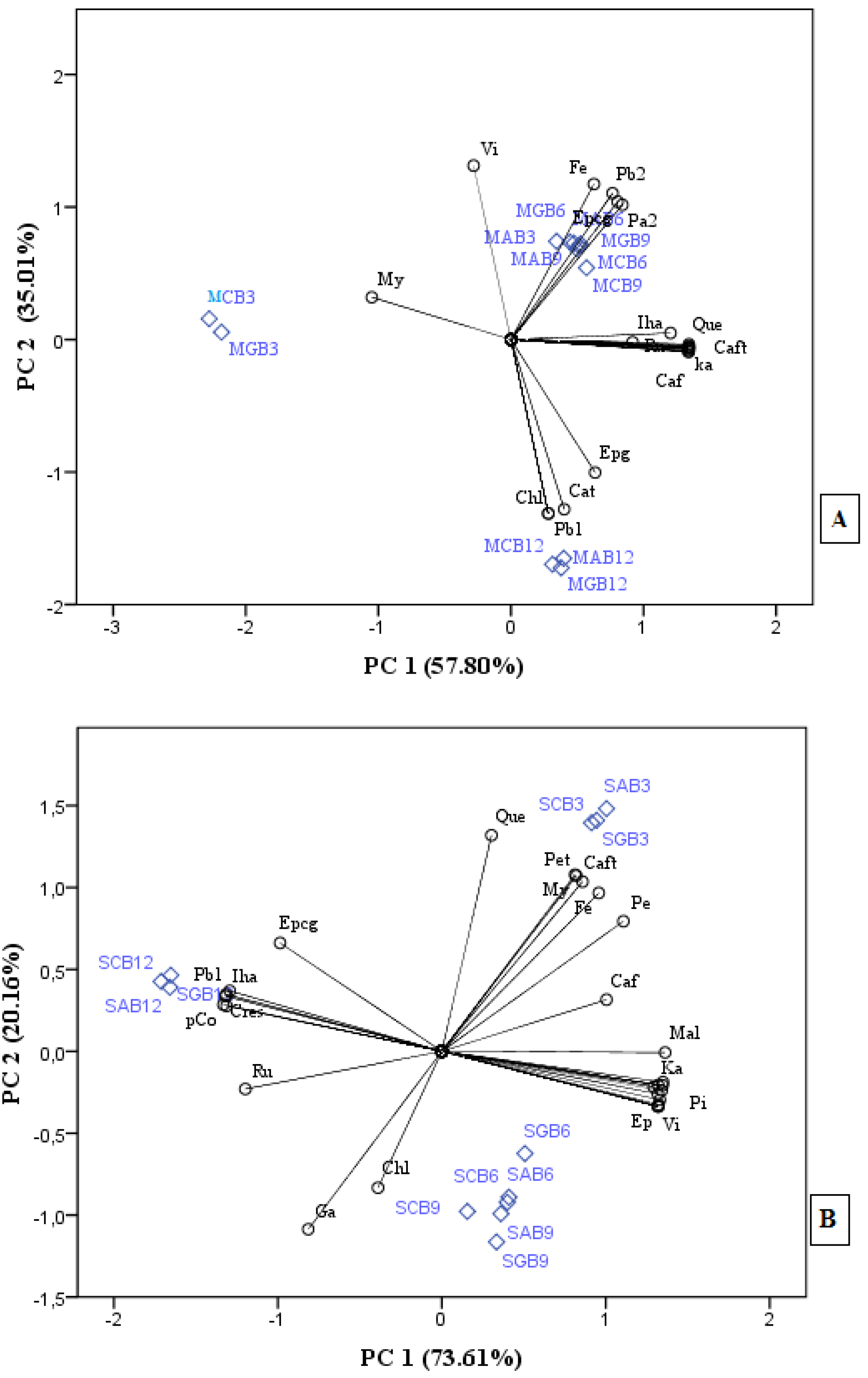
3.4. Principal Component Analysis
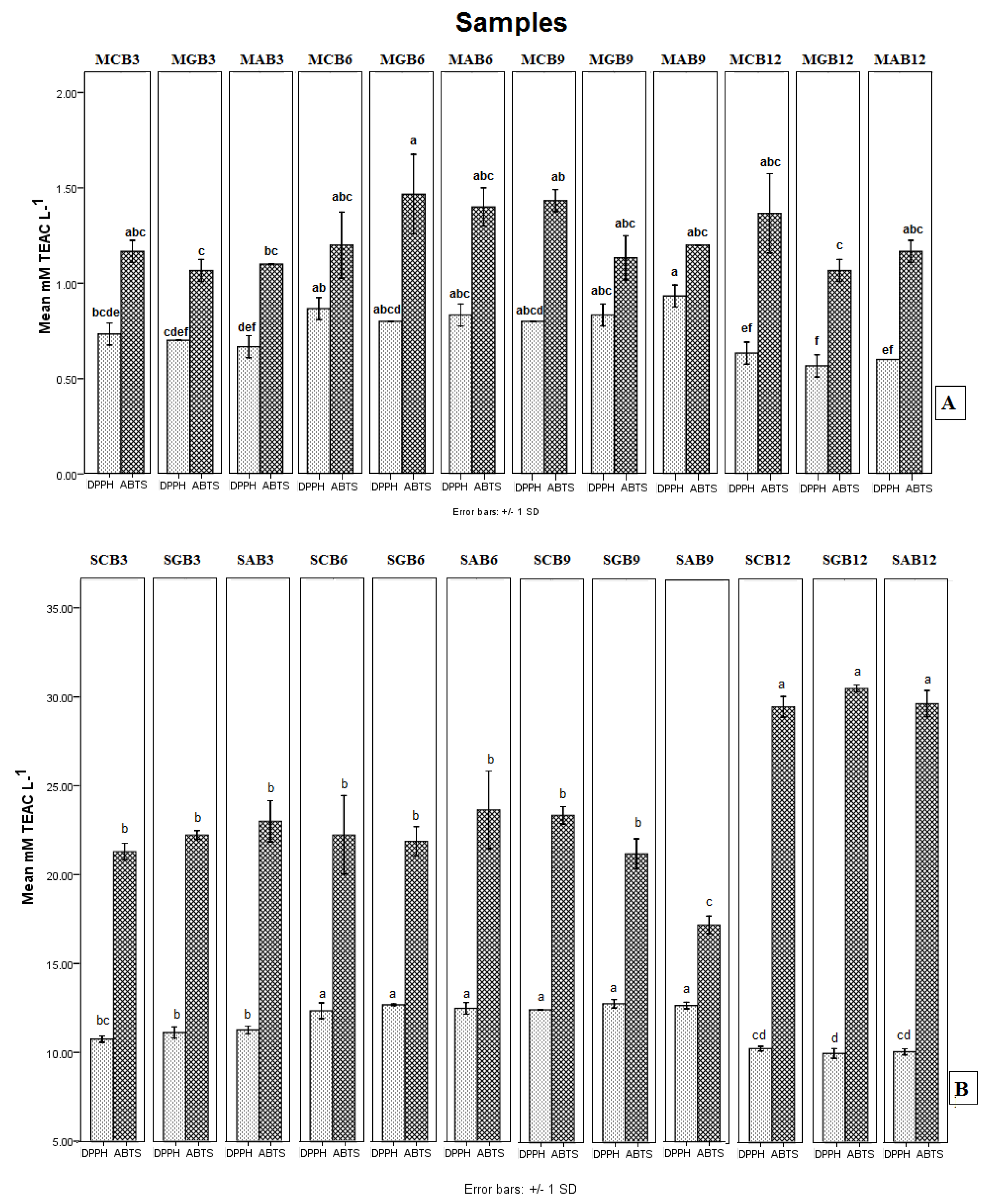
3.5. Antioxidant Activity In Vitro
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Gomes, L.S.; José-Coutinho, A.; Silva, A.G.; Ricardo-da-Silva, J.M. Sensory profile characterization and typicality assessment of “bairrada” and “beira atlântico” red wines. Ciência e Técnica Vitivinícola 2016, 31, 73–87. [Google Scholar] [CrossRef]
- Dutra, M.C.P.; De Souza, J.F.; Viana, A.C.; De Oliveira, D.; Pereira, G.E.; Dos Santos Lima, M. Rapid determination of the aromatic compounds methyl-anthranilate, 2′-aminoacetophenone and furaneol by GC-MS: Method validation and characterization of grape derivatives. Food Res. Int. 2018, 107, 613–618. [Google Scholar] [CrossRef] [PubMed]
- Pereira, G.E.; Padinha, C.; Biasoto, A.C.T.; Canuto, K.M.; Nascimento, M.A.S.; Souza, J.F. Le poids des consommateurs sur l’évolution des vins: L’exemple de la Vallée du São Francisco, Brésil. In Rencontres du Clos-VOugeot 2015: “Vin et civilisation. Les étapes de l’humanisation”; Pérard, J., Perrot, M., Eds.; Centre Georges Chevrier: Dijon, France, 2016; Volume 9, p. 301. [Google Scholar]
- Ricci, A.; Parpinello, G.P.; Versari, A. Modelling the evolution of oxidative browning during storage of white wines: Effects of packaging and closures. Int. J. Food Sci. Technol. 2017, 52, 472–479. [Google Scholar] [CrossRef]
- Agriopoulou, S.; Stamatelopoulou, E. Influence of Storage Conditions on the Quality Characteristics of Wines. EC Nutr. 2017, 8, 93–98. [Google Scholar]
- Arapitsas, P.; Speri, G.; Angeli, A.; Perenzoni, D.; Mattivi, F. The influence of storage on the “chemical age” of red wines. Metabolomics 2014, 10, 816–832. [Google Scholar] [CrossRef]
- Clark, A.C.; Dias, D.A.; Smith, T.A.; Ghiggino, K.P.; Scollary, G.R. Iron (III) Tartrate as a Potential Precursor of Light-Induced Oxidative Degradation of White Wine: Studies in a Model Wine System. J. Agric. Food Chem. 2011, 59, 3575–3581. [Google Scholar] [CrossRef] [PubMed]
- Del Caro, A.; Piombino, P.; Genovese, A.; Moio, L.; Fanara, C.; Piga, A. Effect of Bottle Storage on Colour, Phenolics and Volatile Composition of Malvasia and Moscato White Wines. S. Afr. J. Enol. Vitic. 2014, 35, 128–138. [Google Scholar] [CrossRef]
- Maury, C.; Clarkb, A.C.; Scollary, G.R. Determination of the impact of bottle colour and phenolic concentration on pigment development in white wine stored under external conditions. Anal. Chim. Acta 2010, 660, 81–86. [Google Scholar] [CrossRef] [PubMed]
- Marquez, A.; Serratosa, M.P.; Merida, J. Influence of bottle storage time on colour, phenolic composition and sensory properties of sweet red wines. Food Chem. 2014, 146, 507–514. [Google Scholar] [CrossRef] [PubMed]
- Dias, D.A.; Smith, T.A.; Ghiggino, K.P.; Scollary, G.R. The role of light, temperature and wine bottle colour on pigment enhancement in white wine. Food Chem. 2012, 135, 2934–2941. [Google Scholar] [CrossRef] [PubMed]
- Dias, D.A.; Clark, A.C.; Smith, T.A.; Ghiggino, K.P.; Scollary, G.R. Wine bottle colour and oxidative spoilage: Whole bottle light exposure experiments under controlled and uncontrolled temperature conditions. Food Chem. 2013, 138, 2451–2459. [Google Scholar] [CrossRef] [PubMed]
- Argyri, K.; Komaitis, M.; Kapsokefalou, M. Iron decreases the antioxidant capacity of red wine under conditions of in vitro digestion. Food Chem. 2006, 96, 281–289. [Google Scholar] [CrossRef]
- Organisation Internationale de la Vigne et du Vin (OIV). Vine and Wine Outlook 2012; OIV: Paris, France, 2012. [Google Scholar]
- Singleton, V.; Rossi, J.A. Colorimetry of total phenolics with phosphomolybdic phosphotungstic acid reagents. Am. J. Enol. Vitic. 1965, 27, 144–158. [Google Scholar]
- Lee, J.; Durst, R.W.; Wrolstad, R.E. Determination of total monomeric anthocyanin pigment content of fruit juices, beverages, natural colorants, and wines by the pH differential method: Collaborative study. J. AOAC Int. 2005, 88, 1269–1278. [Google Scholar] [PubMed]
- Kim, Y.K.; Guo, Q.; Packer, L. Free radical scavenging activity of red ginseng aqueous extracts. Toxicology 2002, 172, 149–156. [Google Scholar] [CrossRef]
- Re, R.; Pellegrini, N.; Proteggente, A.; Pannala, A.; Yang, M.; Rice-Evans, C. Antioxidant activity applying an improved ABTS radical cation decolorization assay. Free Radic. Biol. Med. 1999, 26, 1231–1237. [Google Scholar] [CrossRef]
- Silva, J.K.; Cazarin, C.B.B.; Correa, L.C.; Batista, Â.G.; Furlan, C.P.B.; Biasoto, A.C.T.; Maróstica, M.R., Jr. Bioactive compounds of juices from two Brazilian grape cultivars. J. Sci. Food Agric. 2015, 96, 1990–1996. [Google Scholar] [CrossRef] [PubMed]
- Hongyu, K.; Sandanielo, V.L.M.; Oliveira-Junior, G.J. Principal Component Analysis: Theory, interpretations and applications. Eng. Sci. 2015, 5, 1–8. [Google Scholar] [CrossRef]
- Organisation Internationale de la Vigne et du Vin (OIV). Recueil des methods internationals d’analyse des vins et des mouts. In Proceedings of the 8th Assemblée Générale, Paris, France, 21 June 2010. [Google Scholar]
- Sant’Anna, V.; Gurak, P.D.; Marczak, L.D.F.; Tessaro, I.C. Tracking bioactive compounds with colour changes in foods—A review. Dyes Pigment. 2013, 98, 601–608. [Google Scholar] [CrossRef]
- Garrido, J.; Borges, F. Wine and grape polyphenols—A chemical perspective. Food Res. Int. 2013, 54, 1844–1858. [Google Scholar] [CrossRef]
- Xing, R.R.; Li, L.Z.D.; Yuan, T.Y.; Zhang, X.X.; Li, J.M.; Pan, Q.H. Impact of different types of stoppers on sensorial and Phenolic characteristics evolution during a bottle storage time of a white wine from Chardonnay grape variety. J. Food Sci. Technol. 2016, 53, 4043–4055. [Google Scholar] [CrossRef] [PubMed]
- Ribéreau-Gayon, P.; Glories, Y.; Maujean, A.; Dubourdieu, D. Tratado de Enologia 2: Quimica Del Vino, Estabilizacion y Tratamientos; Hemisferio Sur: Buenos Aires, Argentina, 2003. [Google Scholar]
- Morata, A.; Loira, I.; Heras, J.M.; Callejo, M.J.; Tesfaye, W.; González, C.; Suárez-Lepe, J.Á. Yeast influence on the formation of stable pigments in red winemaking. Food Chem. 2016, 197, 686–691. [Google Scholar] [CrossRef] [PubMed]
- Scrimgeour, N.; Nordestgaard, S.; Lloyd, N.D.R.; Wilkes, E.N. Exploring the effect of elevated storage temperature on wine composition. Aust. J. Grape Wine Res. 2015, 21, 713–722. [Google Scholar] [CrossRef]
- Vincenzi, S.; Tomasi, D.; Gaiotti, F.; Lovat, L.; Giacosa, S.; Torchio, F.; Segade, S.R.; Rolle, L. Comparative Study of the Resveratrol Content of Twenty-one Italian Red Grape Varieties. S. Afr. J. Enol. Vitic. 2012, 34, 30–35. [Google Scholar] [CrossRef]
- Guerrero, R.F.; Puertas, B.; Jiménez, M.J.; Cacho, J.; Cantos-Villar, E. Monitoring the process to obtain red wine enriched in resveratrol and piceatannol without quality loss. Food Chem. 2010, 122, 195–202. [Google Scholar] [CrossRef]
- Gambuti, A.; Siani, T.; Picariello, L.; Rinaldi, A.; Lisanti, M.T.; Ugliano, M.; Dieval, J.B.; Moio, L. Oxygen exposure of tannins-rich red wines during bottle aging. Influence on phenolics and color, astringency markers and sensory attributes. Eur. Food Res. Technol. 2016, 243, 669–680. [Google Scholar] [CrossRef]
- Kallithraka, S.; Salacha, M.I.; Tzourou, I. Changes in phenolic composition and antioxidant activity of white wine during bottle storage: Accelerated browning test versus bottle storage. Food Chem. 2009, 113, 500–505. [Google Scholar] [CrossRef]
- Muselík, J.; García-Alonso, M.; Martín-López, M.P.; Žemlička, M.; Rivas-Gonzalo, J.C. Measurement of antioxidant activity of wine catechins, procyanidins, anthocyanins and pyranoanthocyanins. Int. J. Mol. Sci. 2007, 8, 797–809. [Google Scholar] [CrossRef]
- Padilha, C.V.S.; Biasoto, A.C.T.; Corrêa, L.C.; Lima, M.S.; Pereira, G.E. Phenolic compounds profile and antioxidant activity of commercial tropical red wines (Vitis vinifera L.) from São Francisco Valley, Brazil. J. Food Biochem. 2017, 41, e12346. [Google Scholar] [CrossRef]
- Cetó, A.X.; Apetreib, C.; Valle, M.; Rodríguez-Méndez, M.L. Evaluation of red wines antioxidant capacity by meansof a voltammetric e-tongue with an optimized sensor array. Electrochim. Acta 2014, 120, 180–186. [Google Scholar] [CrossRef]
| Basic Parameters | Sparkling Moscatel | Syrah |
|---|---|---|
| pH | 3.40 ± 0.02 | 3.90 ± 0.02 |
| Titratable acidity (g L−1) | 7.6 ± 0.1 | 5.2 ± 0.2 |
| Alcohol content (% v/v) | 9.1 ± 0.1 | 13.1 ± 0.1 |
| Volatile acidity (g L−1) | 0.5 ± 0.1 | 0.6 ± 0.1 |
| Free SO2 (mg L−1) | 47 ± 2 | 52 ± 2 |
| Total SO2 (mg L−1) | 159 ± 1 | 96 ± 1 |
| Color | Reference (0 Months) | Clear Bottle | Green Bottle | Amber Bottle | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 3 Months | 6 Months | 9 Months | 12 Months | 3 Months | 6 Months | 9 Months | 12 Months | 3 Months | 6 Months | 9 Months | 12 Months | ||
| Sparkling Moscatel | |||||||||||||
| L* | 54.03 ± 0.40 | 53.69 ± 0.55 a | 53.75 ± 0.34 a | 53.89 ± 0.42 a | 54.21 ± 0.06 a | 53.36 ± 0.08 ab | 53.58 ± 0.31 a | 53.63 ± 0.48 a | 54.08 ± 0.26 a | 54.08 ± 0.12 a | 52.47 ± 0.09 b | 53.53 ± 0.15 a | 53.53 ± 0.45 a |
| a* | −1.12 ± 0.21 | −1.10 ± 0.20 a | −1.45 ± 0.23 a | −1.65 ± 0.16 ab | −2.04 ± 0.04 bc | −1.58 ± 0.06 ab | −1.64 ± 0.17 ab | −1.67 ± 0.15 ab | −2.15 ± 0.03 c | −1.62 ± 0.10 ab | 1.51 ± 0.25 a | −1.72 ± 0.18 abc | −2.05 ± 0.03 bc |
| b* | 6.90 ± 0.47 | 8.05 ± 0.50 de | 8.94 ± 1.11 bcde | 9.44 ± 0.73 bcd | 11.87 ± 0.23 a | 8.35 ± 0.10 cde | 9.50 ± 0.26 bc | 10.20 ± 0.16 b | 12.70 ± 0.31 a | 7.74 ± 0.24 e | 9.71 ± 0.58 bc | 10.20 ± 0.24 b | 12.45 ± 0.19 a |
| C* | 7.00 ± 0.44 | 8.20 ± 0.54 d | 9.39 ± 0.68 bc | 9.59 ± 0.74 bc | 12.04 ± 0.23 a | 8.49 ± 0.11 cd | 9.64 ± 0.29 b | 10.33 ± 0.14 b | 12.88 ± 0.31 a | 7.91 ± 0.22 d | 10.16 ± 0.03 b | 10.35 ± 0.23 b | 12.62 ± 0.19 a |
| Syrah red wine | |||||||||||||
| L* | 17.55 ± 1.11 | 13.48 ± 0.34 cd | 14.24 ± 0.37 bc | 15.82 ± 0.27 a | 13.97 ± 0.22 bcd | 13.62 ± 0.49 bcd | 14.10 ± 0.03 bc | 13.57 ± 0.1 bcd | 13.66 ± 0.09 bcd | 14.36 ± 0.31 b | 13.24 ± 0.09 d | 15.91 ± 0.26 a | 13.75 ± 0.38 bcd |
| a* | 7.99 ± 0.57 | 4.47 ± 0.38 bcd | 4.34 ± 0.37 cde | 3.52 ± 0.05 de | 3.48 ± 0.44 e | 4.33 ± 0.23 cde | 4.39 ± 0.21 cde | 5.49 ± 0.46 a | 4.49 ± 0.35 e | 5.39 ± 0.33 ab | 4.59 ± 0.21 abc | 4.56 ± 0.39 abc | 3.69 ± 0.26 cde |
| b* | 6.74 ± 0.84 | 6.51 ± 0.27 a | 6.60 ± 0.28 a | 5.02 ± 0.04 c | 6.08 ± 0.43 ab | 6.27 ± 0.24 ab | 6.56 ± 0.09 a | 6.59 ± 0.19 a | 6.29 ± 0.24 a | 6.27 ± 0.40 ab | 6.67 ± 0.13 a | 5.49 ± 0.38 bc | 6.37 ± 0.21 a |
| C* | 10.46 ± 0.90 | 7.39 ± 0.27 cd | 8.46 ± 0.21 ab | 6.39 ± 0.20 d | 6.39 ± 0.35 d | 8.45 ± 0.37 ab | 8.44 ± 0.44 ab | 7.78 ± 0.21 bc | 7.54 ± 0.34 bc | 7.69 ± 0.41 bc | 8.45 ± 0.18 ab | 8.84 ± 0.56 a | 7.36 ± 0.31 cd |
| h* | 40.07 ± 2.54 | 57.74 ± 0.26 bc | 53.99 ± 0.90 cde | 54.72 ± 0.46 cde | 64.36 ± 4.31 a | 55.14 ± 0.68 bcde | 52.40 ± 1.26 de | 58.01 ± 0.27 bc | 56.52 ± 0.82 bcd | 51.26 ± 0.15 e | 54.47 ± 2.67 cde | 53.15 ± 2.17 cde | 59.92 ± 1.16 ab |
| Compounds | Reference (0 Months) | Clear Bottle | Green Bottle | Amber Bottle | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 3 Months | 6 Months | 9 Months | 12 Months | 3 Months | 6 Months | 9 Months | 12 Months | 3 Months | 6 months | 9 Months | 12 Months | ||
| Flavonols | |||||||||||||
| Kaempferol | 0.2 ± 0.0 | 0.1 ± 0.0 b | 0.2 ± 0.0 a | 0.2 ± 0.0 a | 0.2 ± 0.0 a | 0.1 ± 0.0 b | 0.2 ± 0.0 a | 0.2 ± 0.0 a | 0.2 ± 0.0 a | 0.2 ± 0.0 a | 0.2 ± 0.0 a | 0.2 ± 0.0 a | 0.2 ± 0.0 a |
| Quercetin | 1.4 ± 0.2 | 0.4 ± 0.0 d | 1.0 ± 0.2 abc | 0.9 ± 0.1 c | 1.1 ± 0.0 ab | 0.3 ± 0.0 d | 1.0 ± 0.7 bc | 0.9 ± 0.1 c | 1.2 ± 0.1 a | 1.0 ± 0.0 bc | 0.9 ± 0.1 bc | 0.9 ± 0.1 c | 1.1 ± 0.1 ab |
| Isorhamnetin | 0.1 ± 0.0 | 0.12 ± 0.0 b | 0.1 ± 0.0 a | 0.1 ± 0.0 ab | 0.1 ± 0.0 a | 0.1 ± 0.0 a | 0.1 ± 0.0 a | 0.1 ± 0.0 a | 0.1 ± 0.0 ab | 0.1 ± 0.0 a | 0.1 ± 0.0 a | 0.1 ± 0.0 ab | 0.1 ± 0.0 a |
| Myricetin | 0.2 ± 0.0 | 0.2 ± 0.0 b | 0.2 ± 0.0 a | 0.2 ± 0.0 a | 0.2 ± 0.0 a | 0.2 ± 0.0 b | 0.2 ± 0.0 a | 0.2 ± 0.0 a | 0.2 ± 0.0 a | 0.2 ± 0.0 a | 0.2 ± 0.0 a | 0.2 ± 0.0 a | 0.2 ± 0.0 a |
| Rutin | 0.2 ± 0.0 | 0.1 ± 0.0 e | 0.2 ± 0.0 bcd | 0.2 ± 0.0 cd | 0.2 ± 0.0 cd | 0.1 ± 0.0 e | 0.2 ± 0.0 bc | 0.2 ± 0.0 ab | 0.2 ± 0.0 e | 0.2 ± 0.0 bc | 0.2 ± 0.0 cd | 0.2 ± 0.0 cd | 0.3 ± 0.0 a |
| Ʃ Flavonols | 2.1 | 0.9 | 1.7 | 1.6 | 1.8 | 0.9 | 1.7 | 1.7 | 1.8 | 1.7 | 1.7 | 1.6 | 1.9 |
| Flavanols | |||||||||||||
| Catechin | 1.5 ± 0.5 | 0.5 ± 0.0 c | 1.8 ± 0.1 b | 1.7 ± 0.1 b | 6.0 ± 0.0 a | 0.5 ± 0.0 c | 1.8 ± 0.0 b | 1.7 ± 0.1 b | 5.8 ± 0.0 a | 1.7 ± 0.1 b | 1.7 ± 0.1 b | 1.6 ± 0.2 b | 5.8 ± 0.0 a |
| Epicatechin | 0.9 ± 0.2 | 0.3 ± 0.0 e | 1.0 ± 0.0 abc | 0.9 ± 0.1 bc | 0.5 ± 0.1 d | 0.3 ± 0.0 e | 1.0 ± 0.0 a | 0.8 ± 0.0 bc | 0.9 ± 0.0 abc | 0.9 ± 0.0 abc | 0.9 ± 0.1 abc | 0.8 ± 0.0 c | 0.8 ± 0.1 c |
| Epigallocatechin | 0.7 ± 0.1 | 0.4 ± 0.0 fg | 0.7 ± 0.0 c | 1.1 ± 0.0 b | 1.2 ± 0.0 b | 0.3 ± 0.0 g | 0.4 ± 0.0 efg | 0.5 ± 0.0 cdef | 1.4 ± 0.1 a | 0.6 ± 0.0 cd | 0.7 ± 0.2 c | 0.5 ± 0.0 cde | 1.2 ± 0.0 b |
| Epicatechin gallate | 0.7 ± 0.0 | 0.5 ± 0.0 fg | 0.8 ± 0.0 cd | 0.9 ± 0.0 a | 0.5 ± 0.1 fg | 0.5 ± 0.0 fg | 0.6 ± 0.0 e | 0.8 ± 0.0 b | 0.5 ± 0.0 g | 0.7 ± 0.0 d | 0.7 ± 0.0 d | 0.8 ± 0.0 bc | 0.6 ± 0.0 f |
| Ʃ Flavanols | 3.8 | 1.7 | 4.3 | 4.6 | 8.2 | 1.6 | 3.8 | 3.8 | 8.6 | 3.9 | 4.0 | 3.7 | 8.4 |
| Proanthocyanidins | |||||||||||||
| Procyanidin A2 | 0.4 ± 0.0 | 0.3 ± 0.0 fg | 0.4 ± 0.0 de | 0.5 ± 0.0 a | 0.3 ± 0.0 f | 0.2 ± 0.0 g | 0.4 ± 0.0 bcd | 0.4 ± 0.0 bc | 0.3 ± 0.0 f | 0.4 ± 0.0 e | 0.4 ± 0.0 cde | 0.5 ± 0.0 ab | 0.3 ± 0.0 f |
| Procyanidin B1 | 1.0 ± 0.3 | 0.3 ± 0.0 de | 0.7 ± 0.0 bc | 0.5 ± 0.1 de | 3.7 ± 0.1 a | 0.3 ± 0.0 e | 0.6 ± 0.0 c | 0.5 ± 0.0 d | 3.6 ± 0.1 a | 0.8 ± 0.0 b | 0.6 ± 0.0 c | 0.4 ± 0.1 de | 3.7 ± 0.1 a |
| Procyanidin B2 | 4.1 ± 0.6 | 1.4 ± 0.0 c | 6.1 ± 0.1 a | 6.0 ± 0.3 a | 2.4 ± 0.0 b | 1.4 ± 0.0 c | 6.1 ± 0.2 a | 5.8 ± 0.3 a | 2.3 ± 0.1 b | 5.6 ± 0.2 a | 5.8 ± 0.3 a | 6.0 ± 0.1 a | 2.4 ± 0.1 b |
| Ʃ Proanthocyanidins | 5.6 | 2.0 | 7.2 | 7.0 | 6.4 | 1.9 | 7.1 | 6.7 | 6.2 | 6.6 | 6.8 | 6.9 | 6.4 |
| Phenolic acids | |||||||||||||
| Gallic acid | 1.1 ± 0.4 | 0.4 ± 0.0 g | 1.8 ± 0.0 abc | 1.8 ± 0.0 ab | 1.5 ± 0.0 cde | 0.4 ± 0.0 g | 1.7 ± 0.0 bcd | 1.9 ± 0.0 a | 1.4 ± 0.0 f | 1.6 ± 0.0 def | 1.8 ± 0.1 abc | 1.5 ± 0.0 ef | 1.4 ± 0.1 ef |
| Caffeic acid | 1.6 ± 0.0 | 0.5 ± 0.0 d | 1.8 ± 0.1 abc | 1.9 ± 0.0 ab | 2.0 ± 0.0 ab | 0.5 ± 0.0 d | 1.9 ± 0.0 ab | 2.0 ± 0.1 ab | 2.1 ± 0.1 a | 1.6 ± 0.0 c | 1.8 ± 0.1 bc | 1.9 ± 0.2 ab | 2.1 ± 0.1 a |
| Caftaric acid | 57.5 ± 1.7 | 18.1 ± 0.5 d | 71.8 ± 0.6 ab | 66.8 ± 1.6 abc | 64.8 ± 1.5 c | 17.6 ± 0.1 d | 73.2 ± 1.5 a | 66.4 ± 1.4 abc | 66.3 ± 1.3 bc | 71.8 ± 2.5 ab | 71.7 ± 3.7 ab | 65.3 ± 5.7 bc | 67.4 ± 0.7 abc |
| Chlorogenic acid | 1.4 ± 0.2 | 0.4 ± 0.0 e | 1.1 ± 0.1 cd | 0.8 ± 0.0 d | 3.9 ± 0.1 a | 0.4 ± 0.0 e | 1.1 ± 0.0 bc | 0.9 ± 0.1 d | 3.8 ± 0.2 a | 1.3 ± 0.1 b | 1.0 ± 0.0 cd | 0.8 ± 0.1 d | 4.0 ± 0.0 a |
| p-Coumaric acid | 0.3 ± 0.0 | 0.2 ± 0.0 e | 0.3 ± 0.0 d | 0.3 ± 0.0 bcd | 0.4 ± 0.0 a | 0.1 ± 0.0 e | 0.4 ± 0.0 abcd | 0.4 ± 0.0 abcd | 0.4 ± 0.0 abc | 0.3 ± 0.1 d | 0.3 ± 0.0 cd | 0.4 ± 0.0 abcd | 0.4 ± 0.0 ab |
| Ferulic acid | 0.3 ± 0.0 | 0.2 ± 0.0 b | 0.4 ± 0.0 a | 0.4 ± 0.0 a | 0.1 ± 0.0 c | 0.2 ± 0.0 c | 0.4 ± 0.0 a | 0.4 ± 0.0 a | 0.1 ± 0.0 c | 0.4 ± 0.0 a | 0.4 ± 0.0 a | 0.4 ± 0.1 a | 0.1 ± 0.0 c |
| Ʃ Phenolic acids | 62.2 | 19.7 | 77.2 | 72.1 | 72.8 | 19.7 | 78.7 | 71.9 | 74.2 | 77.0 | 77.0 | 70.3 | 75.4 |
| Stilbenes | |||||||||||||
| trans Resveratrol | Nd | Nd | Nd | Nd | Nd | Nd | Nd | Nd | Nd | Nd | Nd | Nd | Nd |
| cis Resveratrol | Nd | Nd | Nd | Nd | Nd | Nd | Nd | Nd | Nd | Nd | Nd | Nd | Nd |
| Piceatannol | 0.1 ± 0.0 | 0.2 ± 0.0 c | 0.3 ± 0.0 b | 0.3 ± 0.0 b | 0.5 ± 0.0 a | 0.2 ± 0.0 c | 0.3 ± 0.0 b | 0.3 ± 0.0 b | 0.5 ± 0.0 a | 0.3 ± 0.0 b | 0.3 ± 0.0 b | 0.3 ± 0.0 b | 0.5 ± 0.0 a |
| Viniferin | 0.2 ± 0.0 | 0.2 ± 0.0 b | 0.2 ± 0.0 a | 0.2 ± 0.0 a | 0.0 ± 0.0 c | 0.2 ± 0.0 b | 0.2 ± 0.0 a | 0.2 ± 0.0 a | 0.0 ± 0.0 c | 0.2 ± 0.0 a | 0.2 ± 0.0 a | 0.2 ± 0.0 a | 0.0 ± 0.0 c |
| Ʃ Stilbenes | 0.3 | 0.4 | 0.5 | 0.5 | 0.5 | 0.4 | 0.5 | 0.5 | 0.5 | 0.5 | 0.5 | 0.5 | 0.5 |
| Total Phenolics ¥ | 280.0 ± 8.1 | 245.0 ± 18.1 abc | 252.9 ± 12.7 ab | 231.4 ± 5.4 bcd | 214.7 ± 8.3 cd | 237.9 ± 9.8 abcd | 229.9 ± 6.7 bcd | 248.2 ± 5.6 ab | 150.0 ± 1.1 e | 212.5 ± 20.5 d | 212.3 ± 6.7 d | 267.0 ± 1.9 a | 225.2 ± 11.2 bcd |
| Compounds | Reference (0 Months) | Clear | Green | Amber | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 3 Months | 6 Months | 9 Months | 12 Months | 3 Months | 6 Months | 9 Months | 12 Months | 3 Months | 6 Months | 9 Months | 12 Months | ||
| Flavonols | |||||||||||||
| Kaempferol | 1.2 ± 0.1 | 1.1 ± 0.0 a | 1.0 ± 0.0 abc | 1.1 ± 0.0 a | 0.9 ± 0.1 cd | 1.0 ± 0.1 abc | 1.0 ± 0.0 abc | 1.1 ± 0.0 a | 0.9 ± 0.0 bcd | 1.1 ± 0.0 ab | 1.0 ± 0.0 abc | 1.0 ± 0.1 abcd | 0.9 ± 0.1 d |
| Quercetin | 32.5 ± 1.4 | 23.2 ± 0.1 ab | 20.0 ± 0.4 c | 20.3 ± 0.3 c | 21.3 ± 1.2 bc | 24.4 ± 1.0 a | 20.8 ± 0.3 bc | 19.6 ± 0.7 c | 21.2 ± 1.1 bc | 24.3 ± 1.0 a | 20.6 ± 0.8 bc | 19.1 ± 0.6 c | 21.0 ± 1.7 bc |
| Isorhamnetin | 4.1 ± 0.3 | 3.3 ± 0.2 a | 2.8 ± 0.1 b | 3.1 ± 0.1 b | 4.9 ± 0.3 b | 3.4 ± 0.1 a | 3.0 ± 0.0 b | 3.0 ± 0.0 b | 5.0 ± 0.3 b | 3.3 ± 0.0 a | 3.0 ± 0.1 b | 3.0 ± 0.1 b | 4.9 ± 0.4 b |
| Myricetin | 1.0 ± 0.0 | 1.0 ± 0.0 a | 0.9 ± 0.0 b | 0.8 ± 0.0 c | 0.9 ± 0.0 bc | 1.0 ± 0.0 a | 0.9 ± 0.0 b | 0.8 ± 0.0 c | 0.9 ± 0.0 bc | 1.0 ± 0.0 a | 0.9 ± 0.0 b | 0.9 ± 0.0 bc | 0.9 ± 0.0 bc |
| Rutin | 1.7 ± 0.2 | 1.3 ± 0.3 c | 1.7 ± 0.1 abc | 2.6 ± 0.2 ab | 2.9 ± 0.7 a | 1.6 ± 0.2 bc | 1.9 ± 0.0 abc | 2.2 ± 0.5 abc | 2.5 ± 0.6 ab | 1.8 ± 0.1 abc | 2.0 ± 0.0 abc | 1.9 ± 0.1 abc | 2.6 ± 0.8 ab |
| Ʃ Flavonols | 40.4 | 29.7 | 26.5 | 27.9 | 30.9 | 31.4 | 27.6 | 26.7 | 30.5 | 31.3 | 27.5 | 25.8 | 30.2 |
| Flavanols | |||||||||||||
| Catechin | 26.1 ± 1.9 | 19.8 ± 0.5 ab | 17.5 ± 0.9 c | 17.3 ± 0.7 c | 3.5 ± 0.1 e | 21.0 ± 0.7 a | 18.7 ± 0.0 bc | 17.4 ± 0.2 c | 3.4 ± 0.1 e | 20.5 ± 0.3 a | 18.4 ± 0.8 bc | 15.6 ± 0.7 d | 3.3 ± 0.1 e |
| Epicatechin | 19.5 ± 1.3 | 13.4 ± 1.0 ab | 11.3 ± 0.7 cd | 10.7 ± 0.3 de | 1.7 ± 0.1 f | 13.9 ± 0.4 a | 12.2 ± 0.4 bc | 10.6 ± 0.0 de | 1.6 ± 0.1 f | 13.6 ± 0.2 ab | 11.7 ± 0.7 cd | 9.8 ± 0.6 e | 1.5 ± 0.1 f |
| Epigallocatechin | 4.6 ± 0.5 | 2.2 ± 0.0 def | 3.9 ± 0.1 a | 3.7 ± 0.3 a | 1.4 ± 0.0 f | 3.1 ± 0.0 abcd | 3.5 ± 0.2 ab | 2.6 ± 0.9 bcde | 1.7 ± 0.1 ef | 3.4 ± 0.1 ab | 3.3 ± 0.8 abc | 2.3 ± 0.1 cdef | 1.7 ± 0.2 f |
| Epicatechin gallate | 3.2 ± 0.8 | 2.8 ± 0.2 b | 2.6 ± 0.1 b | 2.7 ± 0.1 b | 3.4 ± 0.3 a | 2.9 ± 0.1 b | 2.7 ± 0.1 b | 2.6 ± 0.1 b | 2.9 ± 0.2 b | 2.7 ± 0.1 b | 2.6 ± 0.2 b | 2.5 ± 0.1 b | 2.9 ± 0.1 b |
| Ʃ Flavanols | 53.3 | 40,2 | 35.4 | 36.3 | 9.6 | 41 | 37 | 33.1 | 9.7 | 40.2 | 36 | 30.1 | 9.4 |
| Proanthocyanidins | |||||||||||||
| Procyanidin A2 | 4.2 ± 0.1 | 3.5 ± 0.0 abc | 3.1 ± 0.0 de | 2.9 ± 0.2 e | 1.0 ± 0.0 f | 3.6 ± 0.1 a | 3.3 ± 0.0 bcd | 3.1 ± 0.3 de | 1.0 ± 0.0 f | 3.6 ± 0.1 ab | 3.2 ± 0.1 cd | 2.9 ± 0.1 e | 1.0 ± 0.0 f |
| Procyanidin B1 | 18.8 ± 0.4 | 21.1 ± 0.3 a | 21.3 ± 0.5 a | 21.5 ± 0.7 a | 36.8 ± 0.8 b | 22.4 ± 0.5 a | 22.6 ± 0.4 a | 21.6 ± 0.2 a | 39.0 ± 0.2 a | 21.6 ± 0.2 a | 21.4 ± 1.5 a | 18.7 ± 0.3 d | 37.3 ± 0.2 ab |
| Procyanidin B2 | 21.7 ± 0.5 | 22.0 ± 1.1 de | 19.5 ± 0.4 f | 25.4 ± 0.9 c | 30.8 ± 1.0 b | 22.4 ± 0.5 de | 21.9 ± 0.6 def | 24.3 ± 1.5 cd | 33.4 ± 0.5 a | 21.9 ± 0.5 def | 21.4 ± 0.5 ef | 20.7 ± 1.4 ef | 31.9 ± 0.1 ab |
| Ʃ Proanthocyanidins | 44.7 | 44.6 | 43.9 | 47.8 | 68.6 | 48.4 | 47.8 | 49 | 73.4 | 47.1 | 46 | 42.3 | 70.2 |
| Phenolic acids | |||||||||||||
| Gallic acid | 25.0 ± 1.4 | 29.1 ± 0.8 e | 30.9 ± 0.9 cde | 33.9 ± 0.7 a | 33.5 ± 0.6 ab | 29.7 ± 1.0 de | 31.57.6 ± 0.2 bcd | 33.8 ± 0.4 a | 33.6 ± 0.4 a | 28.9 ± 0.6 e | 31.4 ± 0.8 cd | 32.1 ± 0.1 abc | 31.8 ± 1.9 abc |
| Caffeic acid | 9.6 ± 0.3 | 9.1 ± 0.0 bc | 9.1 ± 0.4 bc | 8.5 ± 0.1 cd | 7.9 ± 0.1 d | 10.6 ± 0.2 a | 9.4 ± 0.3 bc | 9.5 ± 0.2 abc | 8.6 ± 0.6 cd | 9.9 ± 0.5 ab | 9.4 ± 0.3 bc | 9.4 ± 0.2 bc | 8.6 ± 0.8 cd |
| Caftaric acid | 91.2 ± 0.7 | 84.6 ± 1.2 ab | 77.3 ± 2.6 cde | 76.4 ± 0.3 de | 75.4 ± 1.1 e | 85.6 ± 1.0 a | 80.8 ± 1.6 bc | 77.4 ± 0.5 cde | 76.6 ± 1.1 de | 86.8 ± 2.3 a | 79.6 ± 1.6 cd | 77.6 ± 1.1 cde | 76.1 ± 0.6 de |
| Chlorogenic acid | 4.4 ± 0.3 | 4.4 ± 0.1 b | 5.7 ± 0.2 a | 5.6 ± 0.3 a | 5.5 ± 0.2 a | 5.8 ± 0.1 a | 5.6 ± 0.1 a | 6.1 ± 0.2 a | 5.7 ± 0.6 a | 5.3 ± 0.5 a | 5.8 ± 0.3 ª | 5.6 ± 0.2 a | 5.9 ± 0.5 a |
| p-Coumaric acid | 1.4 ± 0.0 | 2.4 ± 0.0 de | 2.3 ± 0.0 de | 3.2 ± 0.0 b | 3.8 ± 0.0 a | 2.3 ± 0.0 e | 2.4 ± 0.0 de | 3.1 ± 0.1 bc | 3.7 ± 0.2 a | 1.9 ± 0.1 f | 2.5 ± 0.0 d | 3.0 ± 0.0 c | 3.8 ± 0.1 a |
| Ferulic acid | 3.1 ± 0.1 | 2.8 ± 0.0 a | 2.5 ± 0.1 b | 2.5 ± 0.1 b | 2.3 ± 0.1 c | 2.8 ± 0.1 a | 2.6 ± 0.0 b | 2.6 ± 0.0 b | 2.3 ± 0.1 c | 2.8 ± 0.1 a | 2.6 ± 0.1 b | 2.6 ± 0.1 b | 2.2 ± 0.1 c |
| Ʃ Phenolic acids | 134.6 | 132.5 | 127.8 | 130.0 | 128.4 | 136.9 | 132.4 | 132.5 | 130.5 | 135.7 | 131.2 | 130.2 | 128.4 |
| Stilbenes | |||||||||||||
| trans Resveratrol | 0.6 ± 0.0 | 0.6 ± 0.0 ab | 0.6 ± 0.0 ab | 0.6 ± 0.0 bc | 0.5 ± 0.0 c | 0.6 ± 0.0 a | 0.6 ± 0.0 ab | 0.6 ± 0.1 ab | 0.5 ± 0.0 c | 0.6 ± 0.0 ab | 0.6 ± 0.0 ab | 0.6 ± 0.0 ab | 0.5 ± 0.0 c |
| cis Resveratrol | 0.8 ± 0.1 | 0.4 ± 0.0 cd | 0.5 ± 0.1 ab | 0.4 ± 0.0 cd | 0.6 ± 0.0 a | 0.4 ± 0.0 cd | 0.4 ± 0.0 bcd | 0.5 ± 0.0 bc | 0.6 ± 0.0 a | 0.4 ± 0.0 cd | 0.4 ± 0.0 d | 0.4 ± 0.0 cd | 0.5 ± 0.0 a |
| Piceatannol | 2.0 ± 0.1 | 1.6 ± 0.0 bc | 1.4 ± 0.0 de | 1.4 ± 0.1 d | 0.8 ± 0.0 f | 1.7 ± 0.1 a | 1.5 ± 0.0 cd | 1.4 ± 0.1 d | 0.8 ± 0.0 f | 1.7 ± 0.0 ab | 1.5 ± 0.1 cd | 1.3 ± 0.0 e | 0.8 ± 0.0 f |
| Viniferin | 0.8 ± 0.0 | 0.8 ± 0.0 ab | 0.8 ± 0.0 c | 0.8 ± 0.0 c | 0.7 ± 0.0 d | 0.8 ± 0.0 a | 0.8 ± 0.0 cd | 0.8 ± 0.0 cd | 0.7 ± 0.0 d | 0.8 ± 0.0 cd | 0.8 ± 0.0 cd | 0.8 ± 0.0 c | 0.7 ± 0.0 d |
| Ʃ Stilbenes | 4.2 | 3.4 | 3.3 | 3.2 | 2.6 | 3.5 | 3.3 | 3.3 | 2.6 | 3.5 | 3.3 | 3.1 | 2.6 |
| Anthocyanins | |||||||||||||
| Pelargonidin 3-glucoside | 15.8 ± 0.4 | 9.1 ± 0.4 a | 5.3 ± 0.5 b | 4.1 ± 0.2 c | 2.9 ± 0.2 d | 9.8 ± 0.3 a | 6.1 ± 0.2 b | 4.0 ± 0.1 cd | 3.4 ± 0.1 cd | 9.2 ± 0.6 ª | 5.7 ± 0.6 b | 3.7 ± 0.7 cd | 3.2 ± 0.1 cd |
| Cyanidin 3-glucoside | 1.2 ± 0.0 | 1.1 ± 0.0 a | 0.8 ± 0.0 de | 0.7 ± 0.0 efg | 0.6 ± 0.0 g | 1.0 ± 0.1 ab | 0.8 ± 0.1 cd | 0.7 ± 0.0 defg | 0.6 ± 0.0 g | 0.9 ± 0.1 bc | 0.8 ± 0.0 def | 0.7 ± 0.0 fg | 0.6 ± 0.0 g |
| Delphinidin 3-glucoside | 9.9 ± 0.2 | 6.3 ± 0.2 a | 3.8 ± 0.4 cd | 3.26 ± 0.07 de | 2.1 ± 0.1 g | 6.7 ± 0.2 a | 4.5 ± 0.2 b | 3.1 ± 0.0 e | 2.4 ± 0.1 fg | 6.3 ± 0.2 ª | 4.2 ± 0.4 bc | 2.9 ± 0.4 ef | 2.3 ± 0.1 fg |
| Malvidin 3-glucoside | 88.5 ± 1.6 | 52.4 ± 0.8 a | 29.7 ± 3.0 c | 22.7 ± 0.8 d | 13.6 ± 0.0 f | 55.7 ± 1.0 a | 34.6 ± 1.4 b | 22.0 ± 0.1 d | 16.6 ± 0.5 ef | 55.0 ± 1.8 ª | 32.3 ± 2.5 bc | 17.9 ± 0.5 e | 15.6 ± 0.4 ef |
| Peonidin 3-glucoside | 14.2 ± 0.7 | 8.8 ± 0.3 a | 4.8 ± 0.6 b | 3.6 ± 0.1 c | 2.5 ± 0.2 d | 8.8 ± 0.2 a | 5.7 ± 0.4 b | 3.5 ± 0.1 cd | 2.8 ± 0.1 cd | 8.7 ± 0.3 a | 5.2 ± 0.5 b | 3.2 ± 0.5 cd | 2.7 ± 0.1 cd |
| Petunidin 3-glucoside | 4.3 ± 0.8 | 2.3 ± 0.1 a | 1.4 ± 0.1 c | 1.32 ± 0.01 c | 1.0 ± 0.0 d | 2.5 ± 0.1 a | 1.6 ± 0.0 b | 1.3 ± 0.0 c | 1.1 ± 0.0 d | 2.4 ± 0.1 a | 1.5 ± 0.0 bc | 1.1 ± 0.1 d | 1.1 ± 0.0 d |
| Ʃ Anthocyanins | 134.4 | 80.5 | 45.7 | 35.6 | 22.7 | 84.5 | 53.3 | 34.6 | 26.9 | 82.5 | 49.7 | 29.3 | 26.4 |
| Total Monomeric Anthocyanins Ψ | 243.6 ± 1.0 | 179.0 ± 3.0 a | 129.0 ± 6.2 b | 94.2 ± 1.0 c | 38.2 ± 1.0 e | 182.6 ± 5.0 a | 135.1 ± 5.4 b | 105.8 ± 5.4 c | 65.8 ± 2.0 d | 186.3 ± 4.9 a | 126.6 ± 3.0 b | 97.7 ± 6.6 c | 73.8 ± 1.0 d |
| Total Phenolics ¥ | 2439.8 ± 22.0 | 1945.6 ± 86.3 d | 1955.3 ± 62.3 cd | 2125.2 ± 27.9 abc | 1933.2 ± 37.6 d | 2023.9 ± 8.0 bcd | 2050.9 ± 9.3 bcd | 2236.7 ± 18.6 a | 2029.2 ± 93.1 bcd | 1954.6 ± 58.5 cd | 2071.8 ± 24.1 abcd | 2193.4 ± 51.2 ab | 2094.3 ± 115.4 abcd |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
De Souza, J.F.; De Souza Nascimento, A.M.; Linhares, M.d.S.S.; Dutra, M.d.C.P.; Dos Santos Lima, M.; Pereira, G.E. Evolution of Phenolic Compound Profiles and Antioxidant Activity of Syrah Red and Sparkling Moscatel Wines Stored in Bottles of Different Colors. Beverages 2018, 4, 89. https://doi.org/10.3390/beverages4040089
De Souza JF, De Souza Nascimento AM, Linhares MdSS, Dutra MdCP, Dos Santos Lima M, Pereira GE. Evolution of Phenolic Compound Profiles and Antioxidant Activity of Syrah Red and Sparkling Moscatel Wines Stored in Bottles of Different Colors. Beverages. 2018; 4(4):89. https://doi.org/10.3390/beverages4040089
Chicago/Turabian StyleDe Souza, Joyce Fagundes, Antonio Mendes De Souza Nascimento, Maria do Socorro Silva Linhares, Maria da Conceição Prudêncio Dutra, Marcos Dos Santos Lima, and Giuliano Elias Pereira. 2018. "Evolution of Phenolic Compound Profiles and Antioxidant Activity of Syrah Red and Sparkling Moscatel Wines Stored in Bottles of Different Colors" Beverages 4, no. 4: 89. https://doi.org/10.3390/beverages4040089
APA StyleDe Souza, J. F., De Souza Nascimento, A. M., Linhares, M. d. S. S., Dutra, M. d. C. P., Dos Santos Lima, M., & Pereira, G. E. (2018). Evolution of Phenolic Compound Profiles and Antioxidant Activity of Syrah Red and Sparkling Moscatel Wines Stored in Bottles of Different Colors. Beverages, 4(4), 89. https://doi.org/10.3390/beverages4040089






