Nutrients Assimilation and Chlorophyll Contents for Different Grapevine Varieties in Calcareous Soils in the Somontano DO (Spain)
Abstract
:1. Introduction
2. Materials and Methods
2.1. Grapevine Varieties and Rootstock
2.2. Location
2.3. Climatic Conditions
2.4. Soil Analyses
2.5. Petiole Analyses
2.6. Chlorophyll Monitoring
2.7. Statistical Analyses
3. Results and Discussion
3.1. Petiole Analyses Results
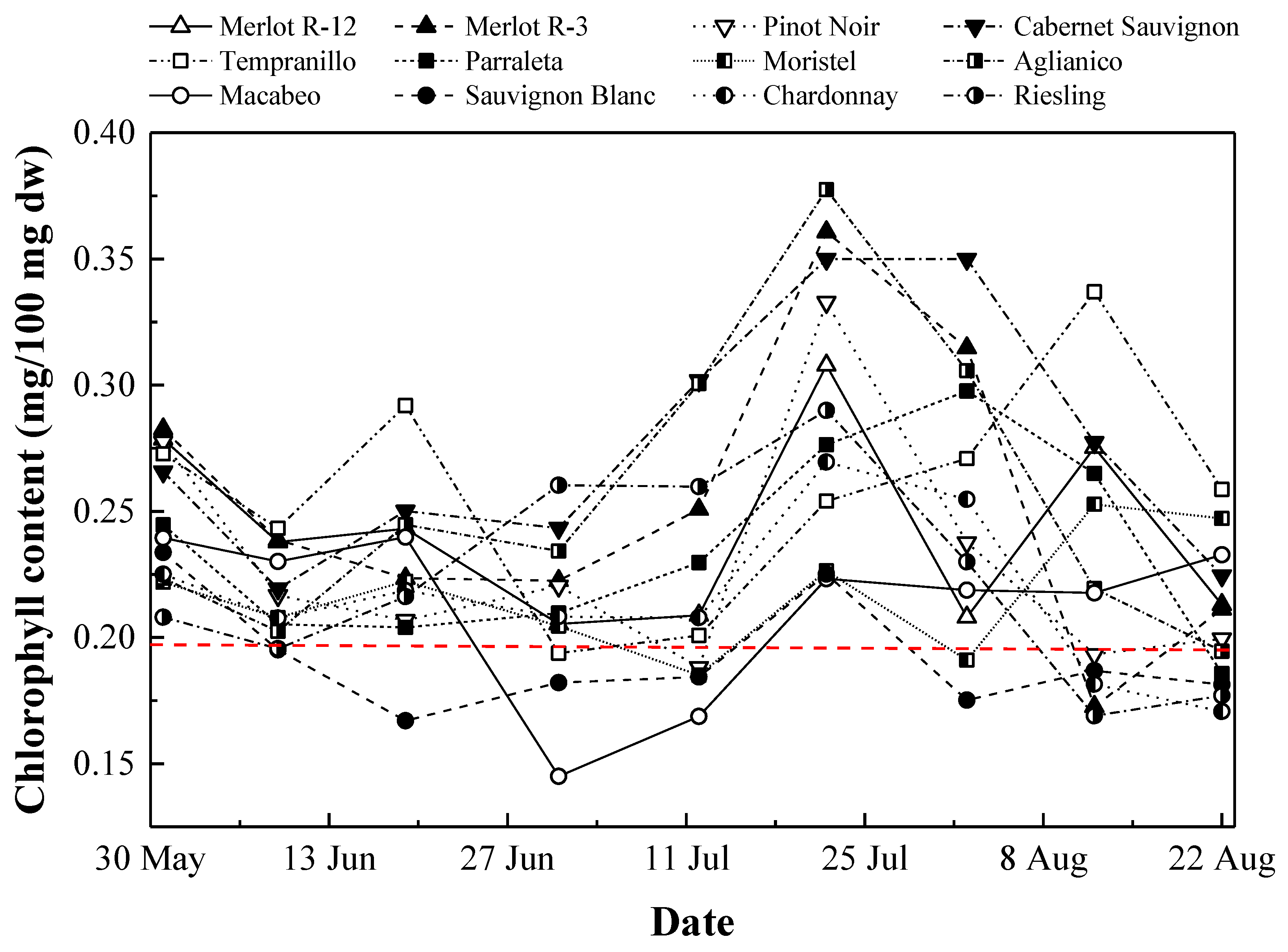
3.2. Chlorophyll Content Monitoring
3.2.1. Temporal Evolution
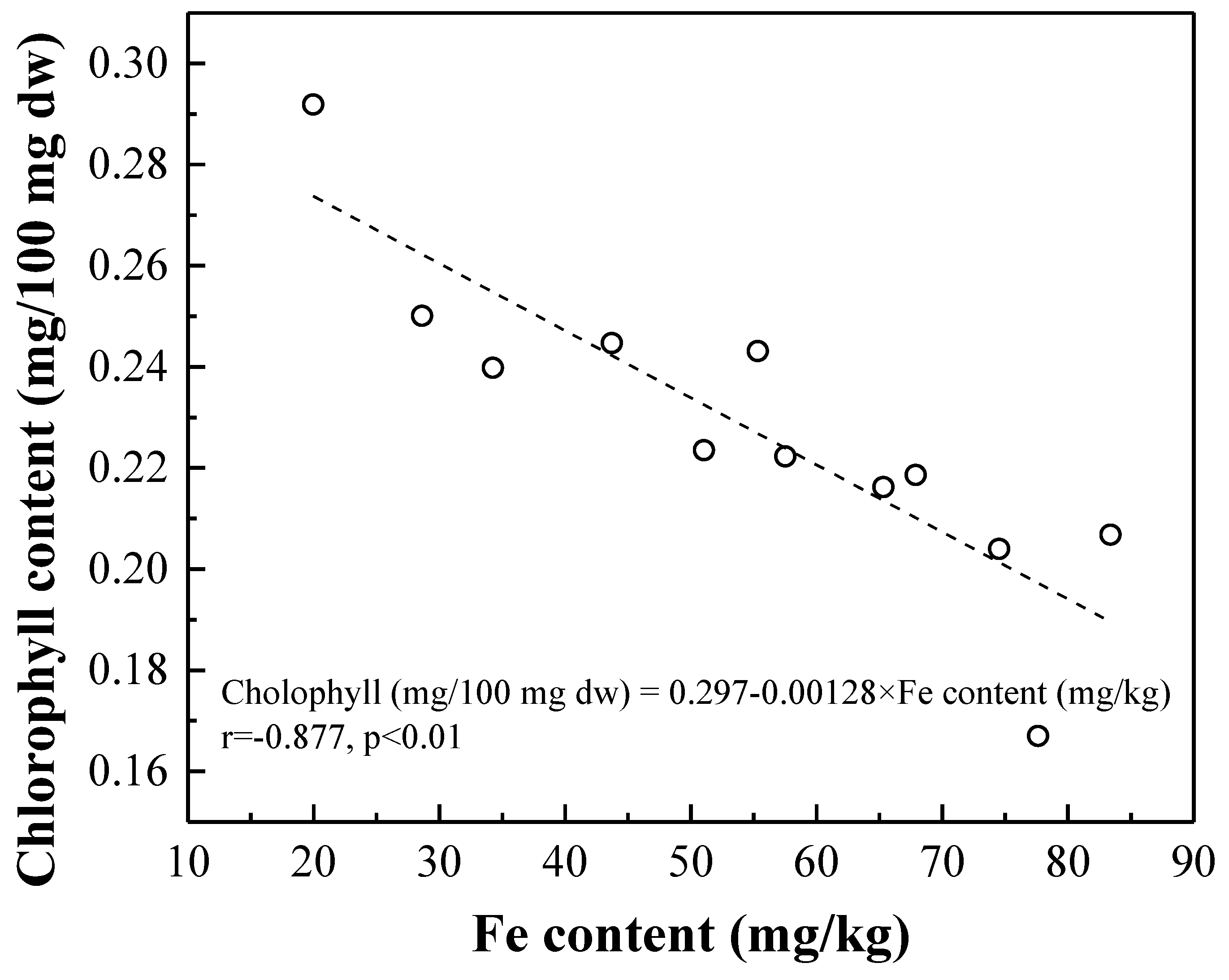
3.2.2. Correlation of Chlorophyll and Nutrient Contents
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Hyvönen, H.; Orama, M.; Saarinen, H.; Aksela, R. Studies on biodegradable chelating ligands: Complexation of iminodisuccinic acid (ISA) with Cu(II), Zn(II), Mn(II) and Fe(III) ions in aqueous solution. Green Chem. 2003, 5, 410–414. [Google Scholar] [CrossRef]
- Chen, Y.; Barak, P. Iron nutrition of plants in calcareous soils. Adv. Agron. 1982, 35, 217–240. [Google Scholar]
- Tagliavini, M.; Rombolà, A.D. Iron deficiency and chlorosis in orchard and vineyard ecosystems. Eur J. Agron. 2001, 15, 71–92. [Google Scholar] [CrossRef]
- Bavaresco, L.; Civardi, S.; Pezzutto, S.; Vezzulli, S.; Ferrari, F. Grape production, technological parameters, and stilbenic compounds as affected by lime-induced chlorosis. Vitis 2005, 44, 63–65. [Google Scholar]
- Sabir, A.; Yazar, K.; Sabir, F.; Kara, Z.; Yazici, M.A.; Goksu, N. Vine growth, yield, berry quality attributes and leaf nutrient content of grapevines as influenced by seaweed extract (Ascophyllum nodosum) and nanosize fertilizer pulverizations. Sci. Hortic. 2014, 175, 1–8. [Google Scholar] [CrossRef]
- Bavaresco, L.; Poni, S. Effect of calcareous soil on photosynthesis rate, mineral nutrition, and source-sink ratio of table grape. J. Plant Nutr. 2007, 26, 2123–2135. [Google Scholar] [CrossRef]
- Pavlousek, P. Tolerance to lime-induced chlorosis and drought in grapevine rootstocks. In Abiotic Stress. Plant Responses and Applications in Agriculture; Vahdati, K., Leslie, C., Eds.; InTech: Vienna, Austria, 2013; pp. 277–306. [Google Scholar]
- Pavlousek, P. Tolerance to Lime-Induced Chlorosis and Drought in Grapevine Rootstocks; InTech: Vienna, Austria, 2013; pp. 277–306. [Google Scholar]
- Pirie, A.J.; Mullins, M.G. Concentration of phenolics in the skin of grape berries during fruit development and ripening. Am. J. Enol. Vitic. 1980, 31, 34–36. [Google Scholar]
- Hall, A.; Lamb, D.W.; Holzapfel, B.; Louis, J. Optical remote sensing applications in viticulture—A review. Aust. J. Grape Wine Res. 2002, 8, 36–47. [Google Scholar] [CrossRef]
- Martín, P.; Zarco-Tejada, P.; González, M.; Berjón, A. Using hyperspectral remote sensing to map grape quality in ’Tempranillo’ vineyards affected by iron deficiency chlorosis. Vitis 2007, 46, 7–14. [Google Scholar]
- Bavaresco, L.; Giachino, E.; Pezzutto, S. Grapevine rootstock effects on lime-induced chlorosis, nutrient uptake, and source–sink relationships. J. Plant Nutr. 2003, 26, 1451–1465. [Google Scholar] [CrossRef]
- Castino, M.; Ubigli, M.; Corino, L.; Luzzati, A.; Siragusa, N.; Nappi, P. Oenological effects of nutrients deficiencies on the grape variety Barbera cultivated in Piedmont vineyards. Vignevini Bol. 1987, 14, 37–54. [Google Scholar]
- Veliksar, S.; Toma, S.; Kreidman, J. Effect of Fe-containing compounds on the chlorosis manifestation and grape quality. In Proceedings of the International Workshop on Advances in Grapevine and Wine Research, Venosa, Italy, 15–17 September 2005. [Google Scholar]
- Tavares, S.; Vesentini, D.; Fernandes, J.C.; Ferreira, R.B.; Laureano, O.; Ricardo-Da-Silva, J.M.; Amâncio, S. Vitis vinifera secondary metabolism as affected by sulfate depletion: Diagnosis through phenylpropanoid pathway genes and metabolites. Plant Physiol. Biochem. 2013, 66, 118–126. [Google Scholar] [CrossRef] [PubMed]
- López-Millán, A.-F.; Grusak, M.A.; Abadía, A.; Abadía, J. Iron deficiency in plants: An insight from proteomic approaches. Front. Plant Sci. 2013, 4. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Covarrubias, J.I.; Pisi, A.; Rombola, A.D. Evaluation of sustainable management techniques for preventing iron chlorosis in the grapevine. Aust. J. Grape Wine Res. 2014, 20, 149–159. [Google Scholar] [CrossRef]
- Díaz de la Torre, I.; Del Campillo, M.D.C.; Barrón, V.; Torrent, J. Predicting the occurrence of iron chlorosis in grapevine with tests based on soil iron forms. OENO One 2010, 44. [Google Scholar] [CrossRef]
- Pestana, M.; de Varennes, A.; Araújo, E. Diagnosis and correction of iron chlorosis in fruit trees: A review. Food Agric. Environ. 2003, 1, 46–51. [Google Scholar]
- Bertoni, G.; Morard, P. Blade or petiole analysis as a guide for grape nutrition. Commun. Soil Sci. Plant Anal. 2008, 13, 593–605. [Google Scholar] [CrossRef]
- Benito, A.; Romero, I.; Domínguez, N.; García-Escudero, E.; Martín, I. Leaf blade and petiole analysis for nutrient diagnosis in Vitis vinifera L. cv. Garnacha tinta. Aust. J. Grape Wine Res. 2013, 19, 285–298. [Google Scholar] [CrossRef]
- Süß, A.; Danner, M.; Obster, C.; Locherer, M.; Hank, T.; Richter, K. Measuring Leaf Chlorophyll Content with the Konica Minolta SPAD-502Plus—Theory, Measurement, Problems, Interpretation; EnMAP: Potsdam, Germany, 2015; p. 18. [Google Scholar] [CrossRef]
- Ruiz-Espinoza, F.H.; Murillo-Amador, B.; García-Hernández, J.L.; Fenech-Larios, L.; Rueda-Puente, E.O.; Troyo-Diéguez, E.; Kaya, C.; Beltrán-Morales, A. Field evaluation of the relationship between chlorophyll content in basil leaves and a portable chlorophyll meter (SPAD-502) readings. J. Plant Nutr. 2010, 33, 423–438. [Google Scholar] [CrossRef]
- Brunetto, G.; Trentin, G.; Ceretta, C.A.; Girotto, E.; Lorensini, F.; Miotto, A.; Moser, G.R.Z.; Melo, G.W.D. Use of the SPAD-502 in estimating nitrogen content in leaves and grape yield in grapevines in soils with different texture. Am. J. Plant Sci. 2012, 03, 1546–1561. [Google Scholar] [CrossRef]
- Munoz-Huerta, R.F.; Guevara-Gonzalez, R.G.; Contreras-Medina, L.M.; Torres-Pacheco, I.; Prado-Olivarez, J.; Ocampo-Velazquez, R.V. A review of methods for sensing the nitrogen status in plants: Advantages, disadvantages and recent advances. Sensors 2013, 13, 10823–10843. [Google Scholar] [CrossRef] [PubMed]
- Gonzalez-Flor, C.; Gorchs, G.; Serrano, L. Assessing petiole iron content in Vitis vinifera ‘Chardonnay’ using reflectance based hyperspectral indices. Acta Hortic. 2013, 101–105. [Google Scholar] [CrossRef]
- Retzlaff, R.; Molitor, D.; Behr, M.; Bossung, C.; Rock, G.; Hoffmann, L.; Evers, D.; Udelhoven, T. UAS-based multi-angular remote sensing of the effects of soil management strategies on grapevine. J. Int. Sci. Vigne Vin 2015, 49, 85–102. [Google Scholar] [CrossRef]
- Huglin, P.; Schneider, C. Biologie et Écologie de la Vigne, 2nd ed.; Tec & Doc Lavoisier: Paris, France, 1998; p. 370. [Google Scholar]
- Badía, D.; Cuchí, J.; Martí, C.; Casanova, J. Los Suelos de los viñedos en la Denominación de Origen Somontano, Ed.; Prensas Universitarias de Zaragoza: Zaragoza, Spain, 2006; Volume 8, p. 205. [Google Scholar]
- Tonietto, J.; Carbonneau, A. A multicriteria climatic classification system for grape-growing regions worldwide. Agric. For. Meteorol. 2004, 124, 81–97. [Google Scholar] [CrossRef]
- Catania, C.D.; Martín Uliarte, E.; Monte, R.F.D.; Avagnina de del Monte, S.; Antelo Bruno, L.; Molina, J.; Mendoza, O.; Flores, N.; Kohlberg, E.J.; Tonietto, J.; et al. Clima, Zonificación y Tipicidad del vino en Regiones Vitivinícolas Iberoamericanas; CYTED: Madrid, Spain, 2012; p. 411. [Google Scholar]
- MAPA. Métodos Oficiales de Análisis. Tomo III; Ministerio de Agricultura, pesca y alimentación: Madrid, Spain, 1994; p. 662. [Google Scholar]
- McLean, E.O. Soil pH and lime requirement. In Methods of Soil Analysis. Part 2. Chemical and Microbiological Properties, 2nd ed.; Page, A.L., Miller, R.H., Keeney, D.R., Eds.; ASA-SSSA: Madison, WI, USA, 1982; pp. 199–224. [Google Scholar]
- Nelson, R.E. Carbonate and gypsum. In Methods of Soil Analysis. Part 2: Chemical and Microbiological Properties, 2nd ed.; Page, A.L., Miller, R.H., Keeney, D.R., Eds.; ASA-SSSA: Madison, WI, USA, 1982; pp. 181–198. [Google Scholar]
- Nelson, D.R.; Sommers, L.E. Total carbon and organic matter. In Methods of Soil Analysis. Part 2: Chemical and Microbiological Properties, 2nd ed.; Page, A.L., Miller, R.H., Keeney, D.R., Eds.; ASA-SSSA: Madison, WI, USA, 1982; pp. 539–557. [Google Scholar]
- Rhoades, J.D. Cation exchange capacity. In Methods of Soil Analysis. Part 2: Chemical and Microbiological Properties, 2nd ed.; Page, A.L., Miller, R.H., Keeney, D.R., Eds.; ASA-SSSA: Madison, WI, USA, 1982; pp. 149–158. [Google Scholar]
- Rhoades, J.D. Soluble salts. In Methods of Soil Analysis. Part 2: Chemical and Microbiological Properties, 2nd ed.; Page, A.L., Miller, R.H., Keeney, D.R., Eds.; ASA-SSSA: Madison, WI, USA, 1982; pp. 167–180. [Google Scholar]
- O.I.V. Organización Internacional de la Viña y el Vino. Resolution Viti 4/95. Diagnostic foliaire: Une méthode harmonisée. Bull. de L’O.I.V 1996, 68, 35–40. [Google Scholar]
- Hidalgo Fernández-Cano, L.; Hidalgo Togores, J. Tratado de Viticultura; Mundi-Prensa: Madrid, Spain, 2011; Volume 2. [Google Scholar]
- González, M.R.; Martín, P. Niveles de referencia para el diagnóstico nutricional de la vid. Vida Rural 2006, 1, 44–48. [Google Scholar]
- Reuter, D.J.; Robinson, J.B. Plant Analysis: An Interpretation Manual; CSIRO Publishing: Collingwood, Australia, 1997. [Google Scholar]
- Bavaresco, L. Utilization of a non-destructive chlorophyll meter to assess chlorophyll concentration in gravepine leaves. Bulletin de l’OIV 1995, 68, 404–414. [Google Scholar]
- Callejas, R.; Kania, E.; Contreras, A.; Peppi, C.; Morales, L. Evaluación de un método no destructivo para estimar las concentraciones de clorofila en hojas de variedades de uva de mesa. Idesia (Arica) 2013, 31, 19–26. [Google Scholar] [CrossRef] [Green Version]
- Chadha, K.; Shikhamany, S. The Grape: Improvement, Production and Post-Harvest Management; Malhotra Publishing House: New Delhi, India, 1999; p. 579. [Google Scholar]
- Brunetto, G.; Melo, G.W.B.D.; Toselli, M.; Quartieri, M.; Tagliavini, M. The role of mineral nutrition on yields and fruit quality in grapevine, pear and apple. Revista Brasileira de Fruticultura 2015, 37, 1089–1104. [Google Scholar] [CrossRef]
- Cooperative Research Centre for Viticulture. Grapevine Nutrition—Literature Review; Cooperative Research Centre for Viticulture: Mitcham, Australia, 2006; p. 50.
- Skinner, P.W.; Matthews, M.A. Reproductive development in grape (Vitis vinifera L.) under phosphorus-limited conditions. Sci. Hortic. 1989, 38, 49–60. [Google Scholar] [CrossRef]
- Catalina, A.; Matei, P.; González, R.; González, M.; Martín, P. Relaciones Entre Niveles de Asimilación de Nutrientes y Calidad de la uva en Viñedos cv. Tempranillo Afectados por Clorosis Férrica; IV Jornadas Fertilización SECH. Actas de Horticultura 61: Castelldefels, Spain, 2011; pp. 96–101. [Google Scholar]
- Mpelasoka, B.S.; Schachtman, D.P.; Treeby, M.T.; Thomas, M.R. A review of potassium nutrition in grapevines with special emphasis on berry accumulation. Aust. J. Grape Wine Res. 2003, 9, 154–168. [Google Scholar] [CrossRef]
- Walker, R.R.; Blackmore, D.H. Potassium concentration and pH inter-relationships in grape juice and wine of Chardonnay and Shiraz from a range of rootstocks in different environments. Aust. J. Grape Wine Res. 2012, 18, 183–193. [Google Scholar] [CrossRef]
- Ahlawat, V.; Yamdagni, R. Effects of various levels of nitrogen and potassium application on growth yield and petiole composition on grapes cv. Perlette. Progress. Hortic. 1988, 20, 190–196. [Google Scholar]
- Morris, J.R.; Cawthon, D.L.; Fleming, J.W. Effects of high rates of potassium fertilization on raw product quality and changes in pH and acidity during storage of Concord grape juice. Am. J. Enol. Vitic. 1980, 31, 323–328. [Google Scholar]
- Delgado, R.; Martín, P.; delÁlamo, M.; González, M.-R. Changes in the phenolic composition of grape berries during ripening in relation to vineyard nitrogen and potassium fertilisation rates. J. Sci. Food Agric. 2004, 84, 623–630. [Google Scholar] [CrossRef]
- Ksouri, R.; Debez, A.; Mahmoudi, H.; Ouerghi, Z.; Gharsalli, M.; Lachaâl, M. Genotypic variability within Tunisian grapevine varieties (Vitis vinifera L.) facing bicarbonate-induced iron deficiency. Plant Physiol. Biochem. 2007, 45, 315–322. [Google Scholar] [CrossRef] [PubMed]
- Romero, I. Análisis de limbo y peciolo para el diagnóstico nutricional de la vid (Vitis vinifera L.), cv. Tempranillo; Univeridad de La Rioja: Logroño, Spain, 2015. [Google Scholar]
- Bavaresco, L. Investigations on some physiological parameters involved in chlorosis occurrence in different grapevine rootstocks and a Vitis vinifera cultivar. Vitis 1990, 29, 305–317. [Google Scholar]
- Rustioni, L.; Grossi, D.; Brancadoro, L.; Failla, O. Characterization of iron deficiency symptoms in grapevine (Vitis spp.) leaves by reflectance spectroscopy. Plant Physiol. Biochem. 2017, 118, 342–347. [Google Scholar] [CrossRef] [PubMed]
- Romero, I.; García-Escudero, E.; Martín, I. Effects of leaf position on blade and petiole mineral nutrient concentration of Tempranillo grapevine (Vitis vinifera L.). Am. J. Enol. Vitic. 2010, 61, 544–550. [Google Scholar] [CrossRef] [Green Version]
- Garcia, M.; Daverede, C.; Gallego, P.; Toumi, M. Effect of various potassium-calcium ratios on cation nutrition of grape grown hydroponically. J. Plant Nutr. 2008, 22, 417–425. [Google Scholar] [CrossRef]
- Skinner, P.W.; Matthews, M.A. A novel interaction of magnesium translocation with the supply of phosphorus to roots of grapevine (Vitis vinifera L.). Plant Cell Environ. 1990, 13, 821–826. [Google Scholar] [CrossRef]
- Römheld, V. The chlorosis paradox: Fe inactivation as a secondary event in chlorotic leaves of grapevine. J. Plant Nutr. 2000, 23, 1629–1643. [Google Scholar] [CrossRef]
- Hailemichael, G.; Catalina, A.; Gonzalez, M.R.; Martin, P. Relationships between water status, leaf chlorophyll content and photosynthetic performance in Tempranillo vineyards. S. Afr. J. Enol. Vitic. 2016, 37, 149–156. [Google Scholar] [CrossRef]
- Montañés García, L.; Sanz Encinas, M. Diagnóstico visual de la clorosis férrica [tr. Visual diagnosis of iron deficiency]. ITEA 1997, 93, 7–22. [Google Scholar]
- Bavaresco, L.; Lovisolo, C. Effect of grafting on grapevine chlorosis and hydraulic conductivity. VITIS-J. Grapevine Res. 2015, 39, 89. [Google Scholar]
- Zulini, L.; Rubinigg, M.; Zorer, R.; Bertamini, M. Effects of drought stress on chlorophyll fluorescence and photosynthetic pigments in grapevine leaves (Vitis vinifera cv. ’White Riesling’). Acta Hortic. 2007, 289–294. [Google Scholar] [CrossRef]
- Almanza-Merchán, P.; González-Almanza, S.; Balaguera-Lópe, H. La posición de la hoja y su efecto sobre la calidad y producción de frutos de vid (Vitis vinifera L.) var. Riesling x Silvaner Leaf position and its effect on quality and yield of the grapevine fruit (Vitis vinifera L.)‘Riesling x Silvaner’. Rev. Colomb. Cinecias Hortíc. 2012, 6, 9–18. [Google Scholar] [CrossRef]
- Gezgin, S.; Er, F. Relationship between total and active iron contents of leaves and observed chlorosis in vineyards in Konya-Hadmalada region of Turkey. Commun. Soil Sci. Plant Anal. 2007, 32, 1513–1521. [Google Scholar] [CrossRef]
- Jones, J.B.; Wallace, A. Sample preparation and determination of iron in plant tissue samples. J. Plant Nutr. 2008, 15, 2085–2108. [Google Scholar] [CrossRef]
- Hernández, I.; Munné-Bosch, S. Linking phosphorus availability with photo-oxidative stress in plants. J. Exp. Bot. 2015, 66, 2889–2900. [Google Scholar] [CrossRef] [PubMed] [Green Version]
| Climatic conditions | June | July | August |
|---|---|---|---|
| Rainfall (mm) | 30.6 | 77.6 | 39.8 |
| Mean maximum temperature (°C) | 39.5 | 42.0 | 36.7 |
| Mean minimum temperature (°C) | 10.7 | 11.4 | 10.2 |
| Average daily temperature (°C) | 22.9 | 26.3 | 23.7 |
| Parameter | Depth | |
|---|---|---|
| 0–30 cm | 30–60 cm | |
| Coarse fraction (%) | 2.15 | 0.88 |
| pH (H2O) | 8.7 | 8.8 |
| pH (KCl) | 7.9 | 8.0 |
| Total carbonates (% CaCO3 eq.) | 32.4 | 31.7 |
| Active limestone (% CaCO3) | 21.9 | 22.1 |
| Bicarbonate (meq/L, saturated paste extract) | 3.15 | 2.86 |
| Organic matter (%) | 1.12 | 1.19 |
| Total N (%) | 0.089 | 0.079 |
| C/N ratio | 7.3 | 8.7 |
| ECe (dS/m, 25 °C) | 1.01 | 1.05 |
| K (ammonium acetate, mg/kg) | 457.79 | 378.35 |
| P (Olsen-Watanabe, mg/kg) | 7.47 | 7.16 |
| Mg (ammonium acetate, mg/kg) | 112.11 | 148.62 |
| Total Fe (mg/kg) | 10,289.54 | 14,576 |
| Fe-EDTA (mg/kg) | 42.64 | 51.76 |
| Fe (ammonium oxalate, mg/kg) | 68.43 | 74.99 |
| Mn-DTPA (mg/kg) | 1.55 | 2.17 |
| K/Mg ratio | 1.27 | 0.78 |
| Chlorotic power index | 46.83 | 40.63 |
| Variety | N (%) | K (%) | P (%) | Mg (%) | Ca (%) | Fe (mg/kg) | Mn (mg/kg) |
|---|---|---|---|---|---|---|---|
| Merlot R-12 | 1.25 ± 0.09 a | 3.76 ± 0.72 a | 0.27 ± 0.005 ** c | 1.29 ± 0.08 a | 1.21 ± 0.06 e | 55.33 ± 2.83 ef | 34.55 ± 18.17 bcd |
| Merlot R-3 | 1.37 ± 0.01 * abc | 3.84 ± 0.32 a | 0.19 ± 0.020 ** b | 1.48 ± 0.09 ab | 1.05 ± 0.11 de | 51.05 ± 1.16 de | 52.74 ± 10.33 d |
| Pinot Noir | 1.32 ± 0.02 * ab | 2.85 ± 0.12 a | 0.07 ± 0.010 a | 1.21 ± 0.11 a | 1.03 ± 0.16 cde | 83.38 ± 4.00 i | 40.01 ± 9.58 cd |
| Cabernet S. | 1.82 ± 0.09 * de | 2.73 ± 0.12 a | 0.14 ± 0.005 b | 1.76 ± 0.03 b | 0.83 ± 0.03 abcd | 28.63 ± 7.04 ab | 37.28 ± 4.16 cd |
| Tempranillo | 1.55 ± 0.05 * bcd | 3.47 ± 0.88 a | 0.09 ± 0.020 ** a | 1.06 ± 0.25 a | 0.76 ± 0.14 ab | 19.96 ± 2.89 a | 7.95 ± 1.03 a |
| Parraleta | 2.74 ± 0.01 * h | 3.34 ± 0.87 a | 0.38 ± 0.000 ** fg | 1.49 ± 0.08 ab | 0.81 ± 0.01 abcd | 74.53 ± 1.86 hi | 12.61 ± 1.44 ab |
| Moristel | 1.79 ± 0.07 * de | 5.81 ± 0.47 b | 0.24 ± 0.005 ** c | 1.30 ± 0.16 a | 0.80 ± 0.01 abcd | 57.51 ± 4.78 efg | 16.82 ± 1.04 abc |
| Aglianico | 1.90 ± 0.15 * ef | 4.00 ± 0.35 a | 0.32 ± 0.005 ** de | 1.51 ± 0.08 ab | 0.78 ± 0.02 abc | 43.72 ± 2.40 cd | 35.46 ± 7.21 bcd |
| Macabeo | 2.14 ± 0.10 * fg | 3.47 ± 0.12 a | 0.36 ± 0.005 ** ef | 1.75 ± 0.05 b | 0.91 ± 0 06 bcd | 34.25 ± 3.00 bc | 12.95 ± 0.68 ab |
| Sauvignon | 2.32 ± 0.07 * g | 3.27 ± 0.28 a | 0.45 ± 0.005 ** h | 1.77 ± 0.27 b | 0.71 ± 0.06 ab | 77.62 ± 3.12 hi | 17.73 ± 2.97 abc |
| Chardonnay | 1.62 ± 0.12 * cde | 2.69 ± 0.06 a | 0.28 ± 0.040 ** cd | 1.26 ± 0.17 a | 0.63 ± 0.02 a | 67.89 ± 2.57 gh | 19.77 ± 3.12 abc |
| Riesling | 1.76 ± 0.22 * de | 3.10 ± 1.04 a | 0.43 ± 0.007 ** gh | 1.15 ± 0.18 a | 0.91 ± 0.02 abcd | 65.32 ± 5.52 fgh | 7.50 ± 0.96 a |
| Variety | N (%) | K (%) | P (%) | Mg (%) | Ca (%) | Fe (mg/kg) | Mn (mg/kg) |
|---|---|---|---|---|---|---|---|
| Merlot R-12 | 0.57 ± 0.02 a | 3.80 ± 0.25 bc | 0.10 ± 0.01 ** abc | 2.91 ± 0.18 a | 1.42 ± 0.07 a | 57.61 ± 3.87 d | 93.67 ± 11.36 a |
| Merlot R-3 | 0.70 ± 0.03 ab | 3.27 ± 0.72 ab | 0.15 ± 0.005 de | 3.22 ± 0.17 a | 1.41 ± 0.07 a | 55.10 ± 1.94 d | 452.93 ± 109.24 c |
| Pinot Noir | 0.76 ± 0.03 ab | 2.37 ± 0.75 ab | 0.15 ± 0.01 de | 2.73 ± 0.34 a | 1.59 ± 0.11 a | 92.65 ± 1.23 g | 242.83 ± 69.61 abc |
| Cabernet S. | 0.82 ± 0.17 ab | 2.73 ± 0.32 ab | 0.12 ± 0.00 ** bcd | 3.63 ± 0.78 ab | 1.40 ± 0.13 a | 31.52 ± 1.63 ab | 398.35 ± 87.44 bc |
| Tempranillo | 1.02 ± 0.06 * bc | 3.31 ± 0.25 abc | 0.08 ± 0.03 ** ab | 3.08 ± 0.55 a | 1.00 ± 0.03 ** a | 23.61 ± 1.96 ** a | 14.09 ± 6.86 ** a |
| Parraleta | 1.69 ± 0.45 * d | 2.43 ± 0.26 ab | 0.27 ± 0.000 f | 5.55 ± 2.31 b | 2.79 ± 0.78 b | 83.73 ± 3.66 fg | 271.48 ± 198.72 abc |
| Moristel | 1.04 ± 0.07 * bc | 2.86 ± 1.24 ab | 0.06 ± 0.03 ** a | 2.77 ± 0.52 a | 1.13 ± 0.23 ** a | 72.02 ± 0.40 e | 102.77 ± 15.98 a |
| Aglianico | 1.21 ± 0.10 * c | 5.04 ± 0.91 c | 0.18 ± 0.005 e | 2.91 ± 0.05 a | 1.05 ± 0.10 ** a | 36.83 ± 5.14 bc | 351.97 ± 89.83 bc |
| Macabeo | 1.25 ± 0.04 * c | 1.83 ± 0.39 a | 0.14 ± 0.02 ** cd | 3.35 ± 0.64 a | 1.49 ± 0.42 a | 45.55 ± 3.04 c | 199.17 ± 71.56 ab |
| Sauvignon | 1.05 ± 0.01 * bc | 3.92 ± 0.07 bc | 0.09 ± 0.005 ** ab | 3.64 ± 0.21 ab | 1.05 ± 0.03 ** a | 75.94 ± 4.97 ef | 231.01 ± 13.99 abc |
| Chardonnay | 1.05 ± 0.03 * bc | 2.74 ± 0.12 ab | 0.24 ± 0.005 f | 3.80 ± 0.13 ab | 1.45 ± 0.05 a | 86.36 ± 2.58 g | 346.51 ± 94.40 bc |
| Riesling | 1.23 ± 0.03 * c | 2.24 ± 0.18 ab | 0.11 ± 0.007 ** bcd | 2.98 ± 0.37 a | 1.45 ± 0.14 a | 68.76 ± 0 63 e | 72.30 ± 21.22 a |
| Kf | Kv | Caf | Cav | Mgf | Mgv | Nf | Nv | Pf | Pv | Fef | Fev | Mnf | Mnv | Chlf | Chlv | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Kf | 1 | |||||||||||||||
| Kv | 0.25 | 1 | ||||||||||||||
| Caf | 0.06 | −0.07 | 1 | |||||||||||||
| Cav | −0.26 | −0.48 | 0.12 | 1 | ||||||||||||
| Mgf | −0.10 | 0.08 | −0.14 | 0.07 | 1 | |||||||||||
| Mgv | −0.31 | −0.24 | −0.34 | 0.81 | 0.33 | 1 | ||||||||||
| Nf | −0.02 | −0.12 | −0.52 | 0.49 | 0.55 | 0.75 | 1 | |||||||||
| Nv | 0.00 | −0.22 | −0.55 | 0.52 | 0.14 | 0.65 | 0.86 | 1 | ||||||||
| Pf | 0.03 | 0.05 | −0.23 | 0.20 | 0.35 | 0.36 | 0.65 | 0.59 | 1 | |||||||
| Pv | −0.35 | −0.07 | −0.29 | 0.68 | 0.15 | 0.75 | 0.44 | 0.55 | 0.29 | 1 | ||||||
| Fef | 0.03 | −0.07 | 0.24 | 0.39 | −0.03 | 0.16 | 0.21 | 0.12 | 0.34 | 0.01 | 1 | |||||
| Fev | −0.08 | −0.33 | −0.01 | 0.45 | −0.18 | 0.27 | 0.13 | 0.15 | 0.27 | 0.27 | 0.80 | 1 | ||||
| Mnf | −0.04 | 0.34 | 0.52 | −0.11 | 0.19 | −0.27 | −0.52 | −0.67 | −0.47 | −0.01 | 0.06 | −0.07 | 1 | |||
| Mnv | −0.28 | 0.15 | −0.11 | 0.18 | 0.54 | 0.30 | 0.09 | −0.08 | −0.08 | 0.54 | −0.04 | 0.04 | 0.67 | 1 | ||
| Chlf | 0.19 | 0.11 | −0.02 | 0.02 | −0.14 | 0.12 | 0.03 | 0.01 | −0.41 | −0.19 | −0.47 | −0.55 | −0.22 | −0.41 | 1 | |
| Chlv | −0.02 | 0.00 | 0.22 | −0.25 | −0.41 | −0.41 | −0.53 | −0.30 | −0.74 | −0.24 | −0.66 | −0.65 | 0.12 | −0.23 | 0.61 | 1 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Casanova-Gascón, J.; Martín-Ramos, P.; Martí-Dalmau, C.; Badía-Villas, D. Nutrients Assimilation and Chlorophyll Contents for Different Grapevine Varieties in Calcareous Soils in the Somontano DO (Spain). Beverages 2018, 4, 90. https://doi.org/10.3390/beverages4040090
Casanova-Gascón J, Martín-Ramos P, Martí-Dalmau C, Badía-Villas D. Nutrients Assimilation and Chlorophyll Contents for Different Grapevine Varieties in Calcareous Soils in the Somontano DO (Spain). Beverages. 2018; 4(4):90. https://doi.org/10.3390/beverages4040090
Chicago/Turabian StyleCasanova-Gascón, José, Pablo Martín-Ramos, Clara Martí-Dalmau, and David Badía-Villas. 2018. "Nutrients Assimilation and Chlorophyll Contents for Different Grapevine Varieties in Calcareous Soils in the Somontano DO (Spain)" Beverages 4, no. 4: 90. https://doi.org/10.3390/beverages4040090
APA StyleCasanova-Gascón, J., Martín-Ramos, P., Martí-Dalmau, C., & Badía-Villas, D. (2018). Nutrients Assimilation and Chlorophyll Contents for Different Grapevine Varieties in Calcareous Soils in the Somontano DO (Spain). Beverages, 4(4), 90. https://doi.org/10.3390/beverages4040090