The Oxygen Paradigm—Quantitative Impact of High Concentrations of Dissolved Oxygen on Kinetics and Large-Scale Production of Arthrospira platensis
Abstract
:1. Introduction
2. Materials and Methods
2.1. Strain and Cultivation Conditions
2.2. Cell Dry Weight Concentration () & Optical Density (OD750)
2.3. Quantification of Incident Illumination Intensity () and Calculation of Average Illumination Intensity (Iav)
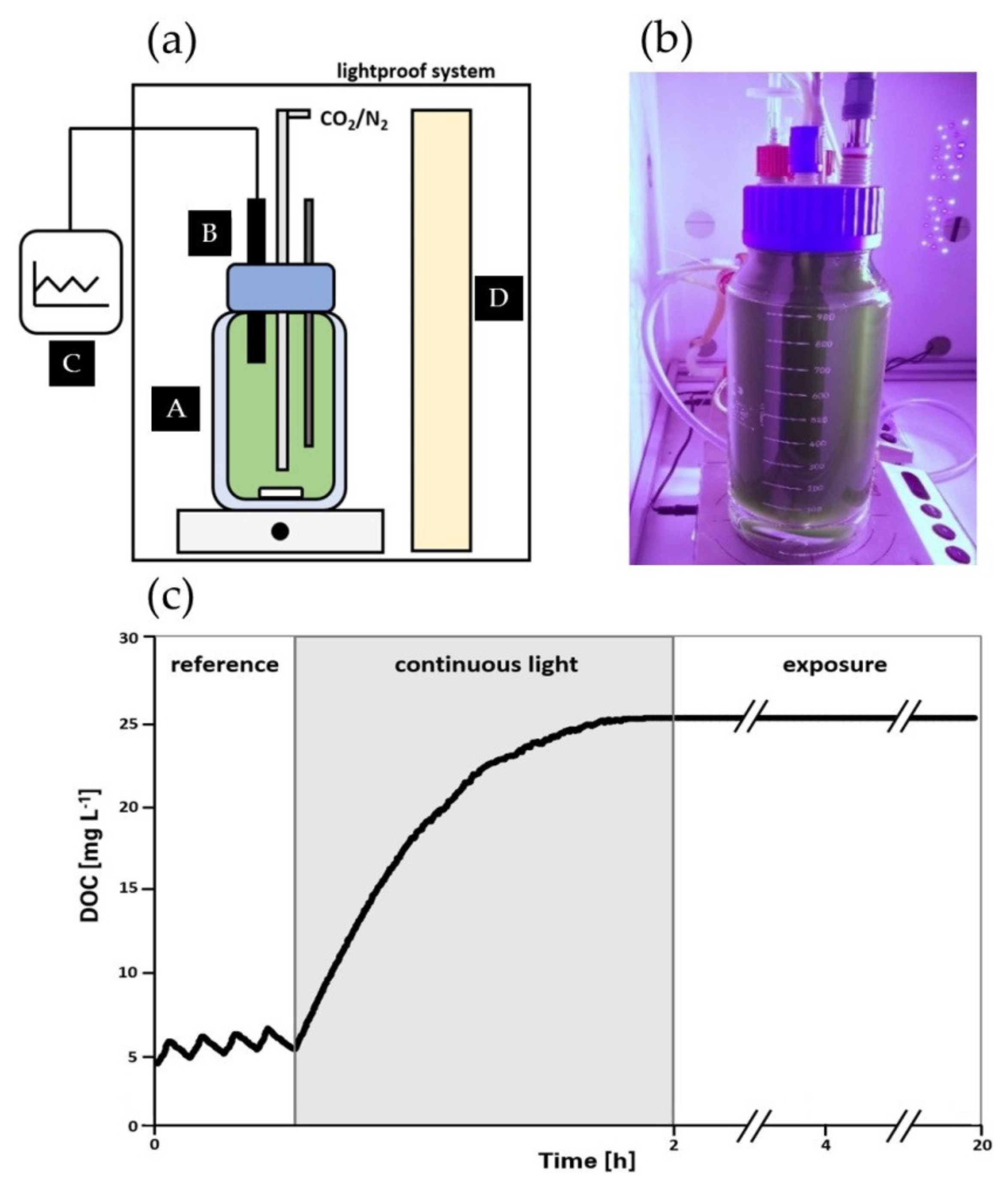
2.4. Photorespirometry Analysis
2.5. In-Vivo Phycocyanin Quantification of Suspended Cells
2.6. Modeling Approaches
2.6.1. Modeling of Respirometric Data
2.6.2. Photoautotrophic Growth Modeling
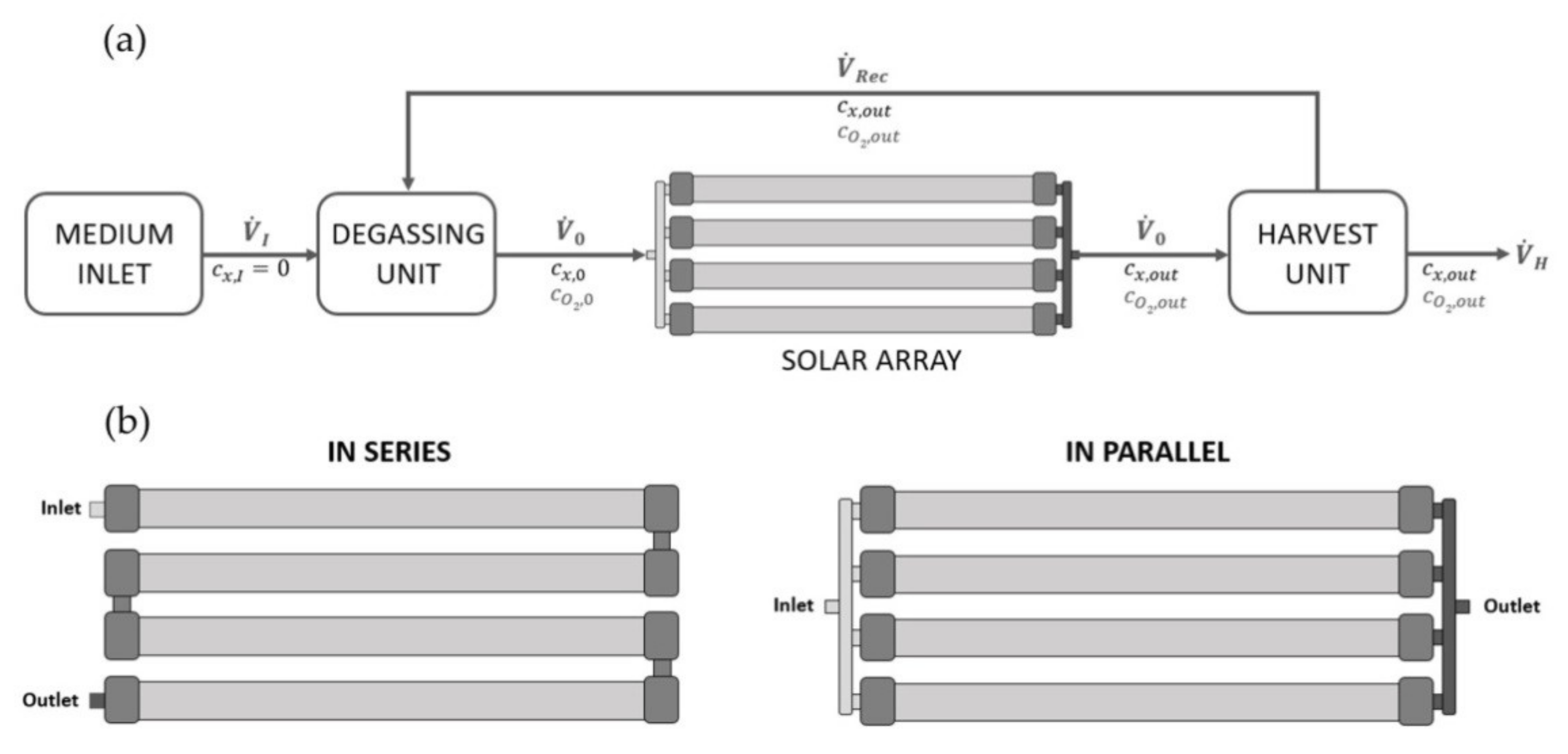
2.7. In-Silico Scale-Up Study at Industry-Scale tPBR
3. Results and Discussion
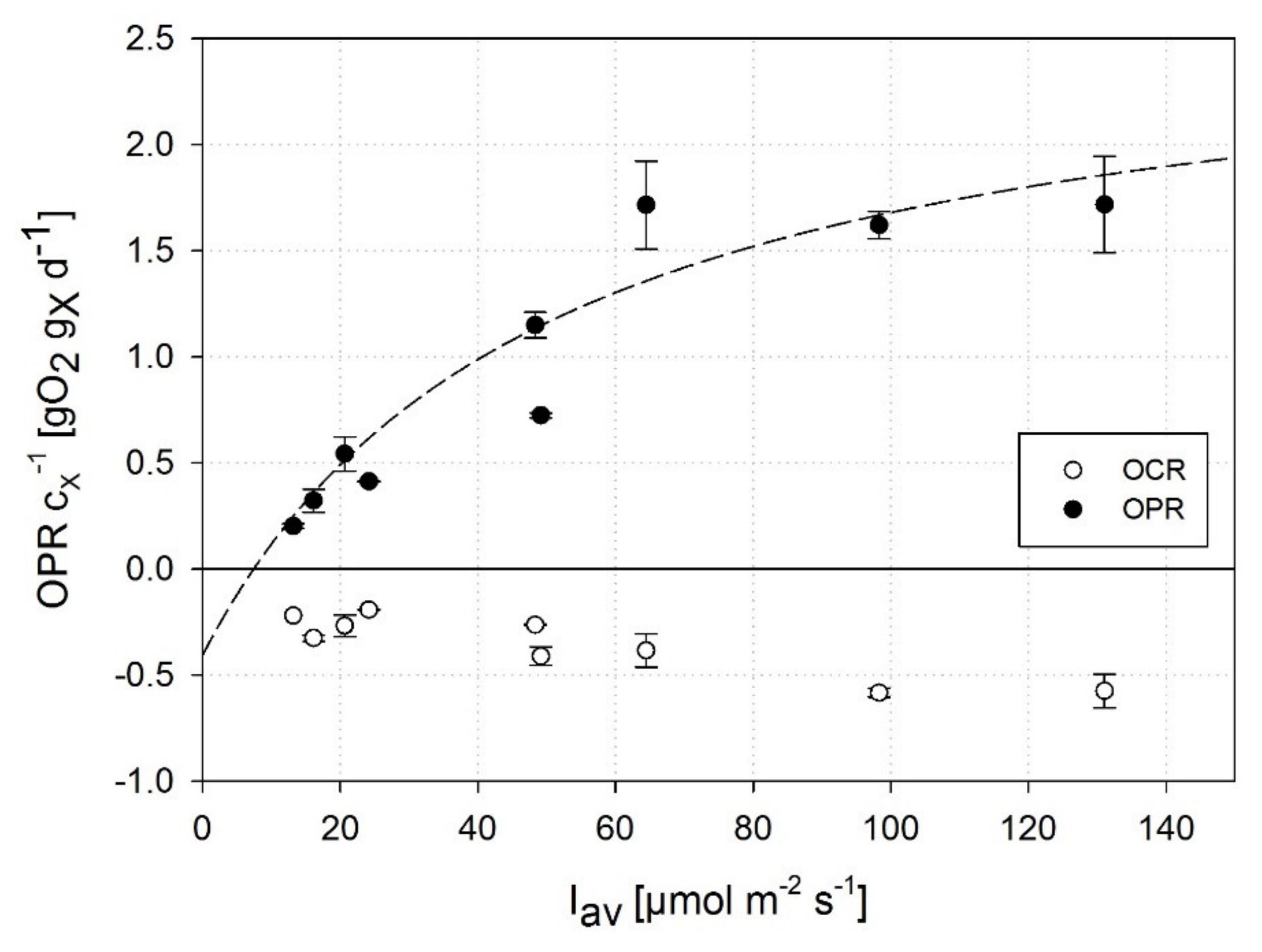
3.1. Impact of High Dissolved Oxygen Concentration (DOC) on the Oxygen Production Rate (OPR) of A. platensis PCC7345
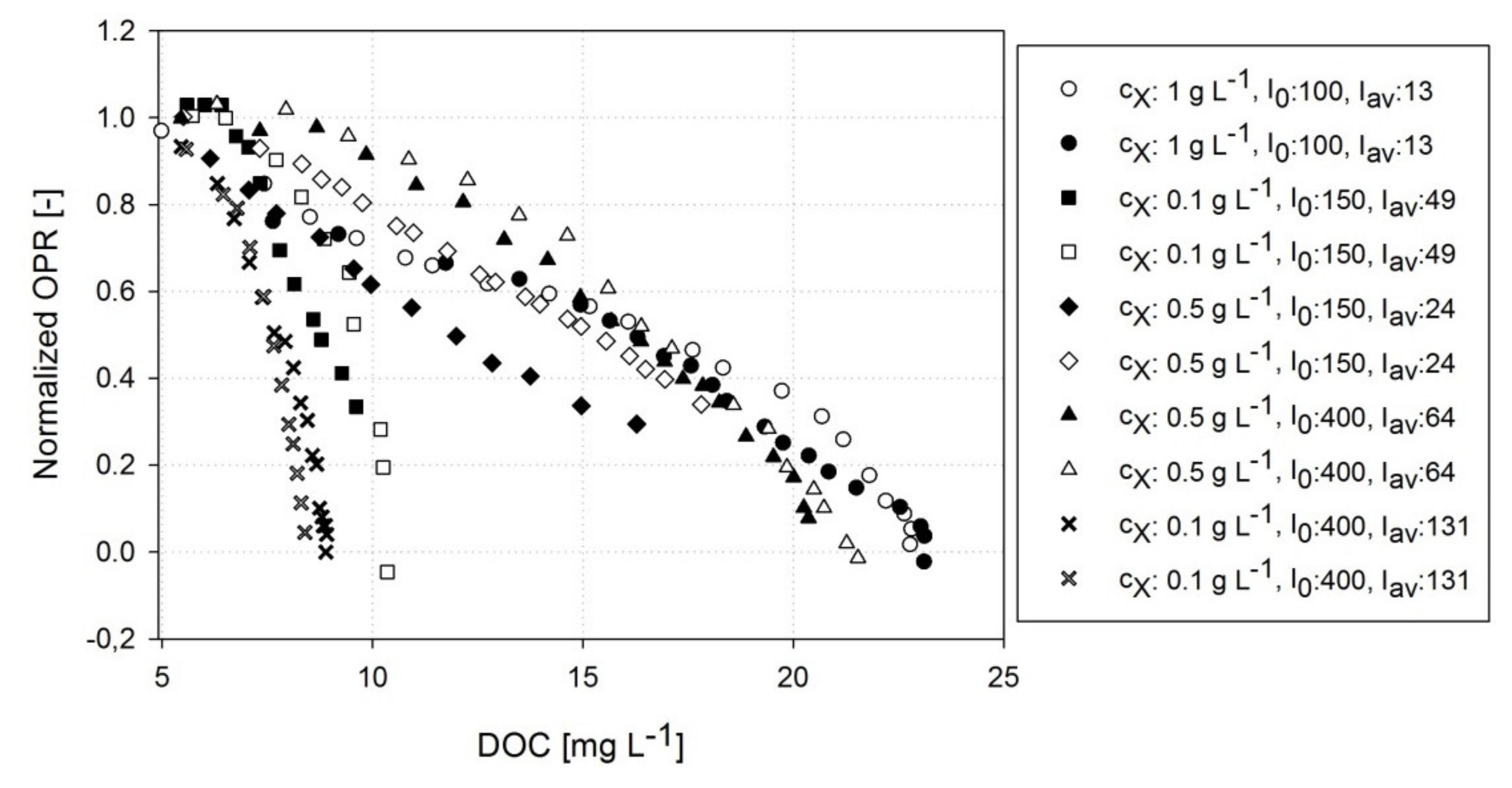
3.2. Correlation of DOC and OPR of A. platensis PCC7345
3.3. Estimating Kinetic Parameters with Photorespirometry
3.3.1. Model by Sforza et al.
3.3.2. Model by Costache et al.
3.4. Impact of DOCs on Cellular Phycocyanin Content ()
3.5. Computational Scale-Up Study and Assessment of Biomass Productivity Using Industrial tPBRs with Different Configurations
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| Symbol | Description | Unit |
| Henry’s constant (Henry’s law) | mol m−3 Pa−1 | |
| reference Henry’s constant (Henry’s law) | mol m−3 Pa−1 | |
| light intensity | µmol photons m−2 s−1 | |
| incident light intensity | µmol photons m−2 s−1 | |
| average light intensity | µmol photons m−2 s−1 | |
| maximum fluorescence intensity | ||
| half-saturation constant for CO2 (model by Sforza) | kgC m−3 | |
| light half-saturation constant | µmol photons m−2 s−1 | |
| oxygen half-saturation constant for respiration (model by Sforza) | kgO2 m−3 | |
| oxygen inhibition constant (model by Costache) | kg m−3 | |
| photorespiration constant (model by Sforza) | kgO2 m−3 | |
| tube length of the solar unit | m | |
| oxygen consumption rate | ||
| oxygen inhibition rate (model by Sforza) | kgO2 m−3 s−1 | |
| oxygen respiration rate (model by Sforza) | kgO2 m−3 s−1 | |
| oxygen production rate | ||
| biomass-related maximum rate of photosynthesis | kgO2 kgx−1 s−1 | |
| net oxygen production rate (Sforza model) | kgO2 m−3 s−1 | |
| photosynthetic oxygen production (Sforza model) | kgO2 m−3 s−1 | |
| optical density at a light wavelength of 750 nm | ||
| reference temperature (Henry’s law) | K | |
| yield of oxygen produced per biomass | kgO2 kgx−1 | |
| light path length | m | |
| concentration of dissolved CO2 | kgC m−3 | |
| oxygen concentration in the liquid phase | kgO2 m−3 | |
| oxygen concentration at the inlet of the solar unit | kgO2 m−3 | |
| oxygen concentration at the outlet of the solar unit | kgO2 m−3 | |
| saturation concentration of oxygen in the liquid phase | kgO2 m−3 | |
| phycocyanin content (biomass-related) | kgPC kgx−1 | |
| biomass concentration | kg m−3 | |
| biomass concentration at the inlet of the solar unit | kgx m−3 | |
| biomass concentration at the outlet of the solar unit | kgx m−3 | |
| oxygen mass transfer coefficient | s−1 | |
| tube length of the solar unit (running coordinate) | m | |
| oxygen partial pressure | Pa | |
| oxygen production rate | kgO2 m−3 s−1 | |
| biomass growth rate | kgx m−3 s−1 | |
| flow velocity | m s−1 | |
| form factor (model by Costache) | ||
| cell-specific light attenuation coefficient | m2 kg−1 | |
| fractional gas hold-up | ||
| specific growth rate | s−1 | |
| maximum specific oxygen rate (model by Sforza) | s−1 | |
| maximum growth rate | s−1 | |
| oxygen inhibition exponent (model by Sforza) | m3 kg−1 | |
| residence time | s |
References
- Borowitzka, M.A.; Gershwin, M.E.; Belay, A. (Eds.) Spirulina in Human nutrition and Health; CRC Press: Boca Raton, FL, USA, 2008; 312p. [Google Scholar]
- Lupatini, A.L.; Colla, L.M.; Canan, C.; Colla, E. Potential application of microalga Spirulina platensis as a protein source. J. Sci. Food Agric. 2017, 97, 724–732. [Google Scholar] [CrossRef] [PubMed]
- Mahmoud, M.M.A.; El-Lamie, M.M.M.; Kilany, O.E.; Dessouki, A.A. Spirulina (Arthrospira platensis) supplementation improves growth performance, feed utilization, immune response, and relieves oxidative stress in Nile tilapia (Oreochromis niloticus) challenged with Pseudomonas fluorescens. Fish Shellfish. Immunol. 2018, 72, 291–300. [Google Scholar] [CrossRef] [PubMed]
- Rosas, V.T.; Poersch, L.H.; Romano, L.A.; Tesser, M.B. Feasibility of the use of Spirulina in aquaculture diets. Rev. Aquac. 2019, 11, 1367–1378. [Google Scholar] [CrossRef]
- Eriksen, N.T. Production of phycocyanin-a pigment with applications in biology, biotechnology, foods and medicine. Appl Microbiol. Biotechnol. 2008, 80, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Ramamoorthy, A.; Premakumari, S. Effect of supplementation of Spirulina on hypercholesterolemic patients. Food Sci. Technol. 1996, 33, 124–127. [Google Scholar]
- Samuels, R.; Mani, U.V.; Iyer, U.M.; Nayak, U.S. Hypocholesterolemic Effect of Spirulina in Patients with Hyperlipidemic Nephrotic Syndrome. J. Med. Food 2002, 5, 91–96. [Google Scholar] [CrossRef] [PubMed]
- Czerwonka, A.; Kaławaj, K.; Sławińska-Brych, A.; Lemieszek, M.K.; Bartnik, M.; Wojtanowski, K.; Zdzisińska, B.; Rzeski, W. Anticancer effect of the water extract of a commercial Spirulina (Arthrospira platensis) product on the human lung cancer A549 cell line. Biomed. Pharmacother. 2018, 106, 292–302. [Google Scholar] [CrossRef]
- Maryati, M.; Saifudin, A.; Wahyuni, S.; Rahmawati, J.; Arrum, A.; Priyunita, O.; Aulia, A.; Putra, F.; As'Hari, Y.; Rasyidah, U.; et al. Cytotoxic effect of Spirulina platensis extract and Ulva compressa Linn. on cancer cell lines. Food Res. 2020, 4, 1018–1023. [Google Scholar] [CrossRef]
- Joseph, J.; Thankamani, K.; Ajay, A.; Das, V.R.A.; Raj, V.S. The Use of Pseudotyped Coronaviruses for the Screening of Entry Inhibitors: Green Tea Extract Inhibits the Entry of SARS-CoV-1, MERS-CoV, and SARS-CoV-2 by Blocking Receptor-Spike Interaction. Curr. Pharm. Biotechno. 2021, 22, 1–12. [Google Scholar] [CrossRef]
- Abu-Taweel, G.M.; Mohsen, G.A.-M.; Antonisamy, P.; Arokiyaraj, S.; Kim, H.-J.; Kim, S.-J.; Park, K.H.; Kim, Y.O. Spirulina consumption effectively reduces anti-inflammatory and pain related infectious diseases. J. Infect. Public Health 2019, 12, 777–782. [Google Scholar] [CrossRef]
- Jensen, G.S.; Attridge, V.L.; Beaman, J.L.; Guthrie, J.; Ehmann, A.; Benson, K.F. Antioxidant and Anti-Inflammatory Properties of an Aqueous Cyanophyta Extract Derived from Arthrospira Platensis: Contribution to Bioactivities by the Non-Phycocyanin Aqueous Fraction. J. Med. Food 2015, 18, 535–541. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Taufikurahman, T.; Ilhamsyah, D.P.A.; Rosanti, S.; Ardiansyah, M.A. Preliminary Design of Phycocyanin Production from Spirulina platensis Using Anaerobically Digested Dairy Manure Wastewater. In IOP Conference Series: Earth and Environmental Science; IOP Publishing: Bristol, UK, 2020; p. 012007. [Google Scholar]
- Jassby, A. Spirulina: A model for microalgae as human food. In Algae and Human Affairs; Lembi, C.A., Waaland, J.R., Eds.; Cambridge University Press: Cambridge, UK, 1988; pp. 149–179. [Google Scholar]
- Dawczynski, J.; Jentsch, S.; Schweitzer, D.; Hammer, M.; Lang, G.E.; Strobel, J. Long term effects of lutein, zeaxanthin and omega-3-LCPUFAs supplementation on optical density of macular pigment in AMD patients: The LUTEGA study. Graefes Arch. Clin. Exp. Ophthalmol. 2013, 251, 2711–2723. [Google Scholar] [CrossRef] [PubMed]
- Seddon, J.M.; Ajani, U.A.; Sperduto, R.D.; Hiller, R.; Blair, N.; Burton, T.C.; Farber, M.D.; Gragoudas, E.S.; Haller, J.; Miller, D.T.; et al. Dietary carotenoids, vitamins A, C, and E, and advanced age-related macular degeneration. Eye Disease Case-Control Study Group. JAMA 1994, 272, 1413–1420. [Google Scholar] [CrossRef]
- Cohen, Z. The chemicals of Spirulina. In Spirulina Platensis (Arthrospira): Physiology, Cell-Biology and Biotechnology; Vonshak, A., Ed.; CRC Press: London, UK, 1997; pp. 175–204. [Google Scholar]
- Fan, Y.Y.; Ramos, K.S.; Chapkin, R.S. Dietary gamma-linolenic acid suppresses aortic smooth muscle cell proliferation and modifies atherosclerotic lesions in apolipoprotein E knockout mice. J. Nutr. 2001, 131, 1675–1681. [Google Scholar] [CrossRef] [Green Version]
- Van Hoorn, R.; Kapoor, R.; Kamphuis, J. A short review on sources and health benefits of GLA, The GOOD omega-6. Oléagineux Corps Gras Lipides 2008, 15, 262–264. [Google Scholar] [CrossRef] [Green Version]
- Posten, C. Design principles of photo-bioreactors for cultivation of microalgae. Eng. Life Sci. 2009, 9, 165–177. [Google Scholar] [CrossRef]
- Carvalho, A.P.; Meireles, L.A.; Malcata, F.X. Microalgal Reactors: A Review of Enclosed System Designs and Performances. Biotechnol. Prog. 2006, 22, 1490–1506. [Google Scholar] [CrossRef] [PubMed]
- Lee, Y.-K. Microalgal mass culture systems and methods: Their limitation and potential. J. Appl. Phycol. 2001, 13, 307–315. [Google Scholar] [CrossRef]
- Weissman, J.C.; Goebel, R.P.; Benemann, J.R. Photobioreactor design: Mixing, carbon utilization, and oxygen accumulation. Biotechnol. Bioeng. 1988, 31, 336–344. [Google Scholar] [CrossRef]
- Torzillo, G.; Bernardini, P.; Masojídek, J. On-line monitoring of chlorophyll fluorescence to assess the extent of photoinhibition of photosynthesis induced by high oxygen concentration and low temperature and its effect on the productivity of outdoor cultures of Spirulina platensis (Cyanobacteria). J. Phycol. 1998, 34, 504–510. [Google Scholar] [CrossRef]
- Torzillo, G.; Chini Zittelli, G. Tubular Photobioreactors. In Algal Biorefineries; Prokop, A., Bajpai, R.K., Zappi, M.E., Eds.; Springer International Publishing: Cham, Switzerland, 2015; pp. 187–212. [Google Scholar]
- Kazbar, A.; Cogne, G.; Urbain, B.; Marec, H.; Le-Gouic, B.; Tallec, J.; Takache, H.; Ismail, A.; Pruvost, J. Effect of dissolved oxygen concentration on microalgal culture in photobioreactors. Algal Res. 2019, 39, 101432. [Google Scholar] [CrossRef] [Green Version]
- Rubio, F.C.; Camacho, F.G.; Grima, E.M. Prediction of dissolved oxygen and carbon dioxide concentration profiles in tubular photobioreactors for microalgal culture. Biotechnol. Bioeng. 1999, 62, 16. [Google Scholar] [CrossRef]
- Babcock, R.W.; Malda, J.; Radway, J.C. Hydrodynamics and mass transfer in a tubular airlift photobioreactor. J. Appl. Phycol. 2002, 14, 169–184. [Google Scholar] [CrossRef]
- Chai, X.; Zhao, X. Enhanced removal of carbon dioxide and alleviation of dissolved oxygen accumulation in photobioreactor with bubble tank. Bioresour. Technol. 2012, 116, 360–365. [Google Scholar] [CrossRef]
- Ojanen, S.; Tyystjärvi, E.; Holmberg, H.; Ahtila, P. Porous membrane as a means of gas and nutrient exchange in a tubular photobioreactor. J. Appl. Phycol. 2015, 27, 1169–1175. [Google Scholar] [CrossRef]
- Ferreira, B.S.; Fernandes, H.L.; Reis, A.; Mateus, M. Microporous hollow fibres for carbon dioxide absorption: Mass transfer model fitting and the supplying of carbon dioxide to microalgal cultures. J. Chem. Technol. Biotechnol. 1998, 71, 61–70. [Google Scholar] [CrossRef]
- Pires, J.C.M.; Alvim-Ferraz, M.C.M.; Martins, F.G. Photobioreactor design for microalgae production through computational fluid dynamics: A review. Renew. Sustain. Energy Rev. 2017, 79, 248–254. [Google Scholar] [CrossRef]
- Massart, A.; Mirisola, A.; Lupant, D.; Thomas, D.; Hantson, A.-L. Experimental characterization and numerical simulation of the hydrodynamics in an airlift photobioreactor for microalgae cultures. Algal Res. 2014, 6, 210–217. [Google Scholar] [CrossRef]
- Trentin, G.; Barbera, E.; Bertucco, A.; Sforza, E. Role of oxygen in tubular photobioreactors: Model-Based design and operating conditions to minimize productivity losses. Chem. Eng. Processing-Process 2020, 157, 108151. [Google Scholar] [CrossRef]
- Costache, T.A.; Acién Fernández, F.G.; Morales, M.M.; Fernández-Sevilla, J.M.; Stamatin, I.; Molina, E. Comprehensive model of microalgae photosynthesis rate as a function of culture conditions in photobioreactors. Appl. Microbiol. Biotechnol. 2013, 97, 7627–7637. [Google Scholar] [CrossRef]
- Sforza, E.; Pastore, M.; Franke, S.M.; Barbera, E. Modeling the oxygen inhibition in microalgae: An experimental approach based on photorespirometry. New Biotechnol. 2020, 59, 26–32. [Google Scholar] [CrossRef] [PubMed]
- Krujatz, F.; Illing, R.; Krautwer, T.; Liao, J.; Helbig, K.; Goy, K.; Opitz, J.; Cuniberti, G.; Bley, T.; Weber, J. Light-field-characterization in a continuous hydrogen-producing photobioreactor by optical simulation and computational fluid dynamics. Biotechnol. Bioeng. 2015, 112, 2439–2449. [Google Scholar] [CrossRef] [PubMed]
- Rossi, S.; Bellucci, M.; Marazzi, F.; Mezzanotte, V.; Ficara, E. Activity assessment of microalgal-bacterial consortia based on respirometric tests. Water Sci. Technol. 2018, 78, 207–215. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sforza, E.; Pastore, M.; Barbera, E.; Bertucco, A. Respirometry as a tool to quantify kinetic parameters of microalgal mixotrophic growth. Bioprocess Biosyst. Eng. 2019, 42, 839–851. [Google Scholar] [CrossRef]
- Takors, R.; Weuster-Botz, D. Prozessmodelle. In Bioprozesstechnik, 4th ed.; Chmiel, H., Takors, R., Weuster-Botz, D., Eds.; Springer Spektrum: Berlin/Heidelberg, Germany, 2018; pp. 71–105. [Google Scholar]
- ASCE. Measurement of Oxygen Transfer in Clean Water, 2nd ed.; American Society of Civil Engineers: New York, NY, USA, 1993. [Google Scholar]
- Vonshak, A. Spirulina: Growth, Physiology and Biochemistry. In Spirulina Platensis Arthrospira: Physiology, Cell-Biology and Biotechnology; Vonshak, A., Ed.; CRC Press: London, UK, 1997; pp. 43–66. [Google Scholar]
- Bennett, A.; Bogorad, L. Complementary chromatic adaptation in a filamentous blue-green alga. J. Cell Biol. 1973, 58, 419–435. [Google Scholar] [CrossRef]
- Vanrolleghem, P.; Borchardt, D.; Henze, M.; Rauch, W.; Reichert, P.; Shanahan, P.; Somlyódy, L. River Water Quality Model no. 1 (RWQM1): III. Biochemical submodel selection. Water Sci. Technol. 2001, 43, 31–40. [Google Scholar] [CrossRef]
- Schuurmans, R.M.; van Alphen, P.; Schuurmans, J.M.; Matthijs, H.C.P.; Hellingwerf, K.J. Comparison of the photosynthetic yield of cyanobacteria and green algae: Different methods give different answers. PLoS ONE 2015, 10, e0139061. [Google Scholar] [CrossRef] [Green Version]
- Grima, E.M.; Fernandez, F.G.A.; Camacho, F.G.; Chisti, Y. Photobioreactors: Light regime, mass transfer, and scaleup. J. Biotechnol. 1999, 70, 231–247. [Google Scholar] [CrossRef]
- Geider, R.J.; Osborne, B.A. Respiration and microalgal growth: A review of the quantitative relationship between dark respiration and growth. New Phytol. 1989, 112, 327–341. [Google Scholar] [CrossRef]
- Laws, E.A.; Bannister, T.T. Nutrient- and light-limited growth of Thalassiosira fluviatilis in continuous culture, with implications for phytoplankton growth in the ocean1. Limnol. Oceanogr. 1980, 25, 457–473. [Google Scholar] [CrossRef]
- Laws, E.; Caperon, J. Carbon and nitrogen metabolism by Monochrysis lutheri: Measurement of growth-rate-dependent respiration rates. Mar. Biol. 1976, 36, 85–97. [Google Scholar] [CrossRef]
- Ogbonna, J.; Tanaka, H. Light requirement and photosynthetic cell cultivation-Development of processes for efficient light utilization in photobioreactors. J. Appl. Phycol. 2000, 12, 207–218. [Google Scholar] [CrossRef]
- Béchet, Q.; Shilton, A.; Guieysse, B. Modeling the effects of light and temperature on algae growth: State of the art and critical assessment for productivity prediction during outdoor cultivation. Biotechnol. Adv. 2013, 31, 1648–1663. [Google Scholar] [CrossRef] [PubMed]
- Pruvost, J.; Cornet, J.F.; Goetz, V.; Legrand, J. Theoretical investigation of biomass productivities achievable in solar rectangular photobioreactors for the cyanobacterium Arthrospira platensis. Biotechnol. Prog. 2012, 28, 699–714. [Google Scholar] [CrossRef] [PubMed]
- Phélippé, M.; Gonçalves, O.; Thouand, G.; Cogne, G.; Laroche, C. Characterization of the polysaccharides chemical diversity of the cyanobacteria Arthrospira platensis. Algal Res. 2019, 38, 101426. [Google Scholar] [CrossRef]
- Krause, G. The Role of Oxygen in Photoinhibition of Photosynthesis. In Causes of Photooxidative Stress and Amelioration of Defense Systems in Plants, 2nd ed.; Foyer, C.H., Mullineaux, C.W., Eds.; CRC Press: New York, NY, USA, 1994; pp. 43–76. [Google Scholar]
- Latifi, A.; Ruiz, M.; Zhang, C.-C. Oxidative stress in cyanobacteria. FEMS Microbiol. Rev. 2009, 33, 258–278. [Google Scholar] [CrossRef] [Green Version]
- Badger, M.R.; von Caemmerer, S.; Ruuska, S.; Nakano, H. Electron flow to oxygen in higher plants and algae: Rates and control of direct photoreduction (Mehler reaction) and rubisco oxygenase. Phil. Trans. R. Soc. Lond. B 2000, 355, 1433–1446. [Google Scholar] [CrossRef] [Green Version]
- Hsieh, P.; Pedersen, J. Reactive oxygen species in cyanobacteria. In Cyanobacteria: Ecological Importance, Biotechnological Uses and Risk Management, 1st ed.; Davison, D., Ed.; Nova Science Publishers: New York, NY, USA, 2014; pp. 1–41. [Google Scholar]
- Allahverdiyeva, Y.; Isojärvi, J.; Zhang, P.; Aro, E.-M. Cyanobacterial Oxygenic Photosynthesis is Protected by Flavodiiron Proteins. Life 2015, 5, 716–743. [Google Scholar] [CrossRef]
- Raven, J.A.; Beardall, J. Respiration in aquatic photolithotrophs. In Respiration in Aquatic Ecosystem; Del Giorgio, P.E., Williams, P.J.L.B., Eds.; Oxford University Press: Oxford, UK; New York, NY, USA, 2005; pp. 36–46. [Google Scholar]
- Sousa, C.; Compadre, A.; Vermuë, M.H.; Wijffels, R.H. Effect of oxygen at low and high light intensities on the growth of Neochloris oleoabundans. Algal Res. 2013, 2, 122–126. [Google Scholar] [CrossRef]
- Marquez, F.J.; Sasaki, K.; Nishio, N.; Nagai, S. Inhibitory effect of oxygen accumulation on the growth of Spirulina platensis. Biotechnol. Lett. 1995, 17, 225–228. [Google Scholar] [CrossRef]
- Torzillo, G.; Giovannetti, L.; Bocci, F.; Materassi, R. Effect of oxygen concentration on the protein content of Spirulina biomass. Biotechnol. Bioeng. 1984, 26, 1134–1135. [Google Scholar] [CrossRef] [PubMed]
- Torzillo, G. Tubular bioreactors. In Spirulina platensis (Arthrospira): Physiology, Cell-Biology and Biotechnology, 1st ed.; CRC Press: London, UK, 1997; pp. 101–115. [Google Scholar]
- Vonshak, A.; Torzillo, G.; Accolla, P.; Tomaselli, L. Light and oxygen stress in Spirulina platensis (cyanobacteria) grown outdoors in tubular reactors. Physiol. Plant 1996, 97, 175–179. [Google Scholar] [CrossRef]
- Sousa, C.; de Winter, L.; Janssen, M.; Vermuë, M.; Wijffels, R.H. Growth of the microalgae Neochloris oleoabundans at high partial oxygen pressures and sub-saturating light intensity. Bioresour. Technol. 2012, 104, 565–570. [Google Scholar] [CrossRef] [PubMed]
- Raso, S.; van Genugten, B.; Vermuë, M.; Wijffels, R.H. Effect of oxygen concentration on the growth of Nannochloropsis sp. at low light intensity. J. Appl. Phycol. 2012, 24, 863–871. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Beaumont, J.A.G. Impact of Oxygen on Cyanobacterium Arthrospira sp. PCC 8005, Master End Project; EHU-UPV: Leioa, Spain, 2015. [Google Scholar]
- Molina, E.; Fernández, J.; Acién, F.G.; Chisti, Y. Tubular photobioreactor design for algal cultures. J. Biotechnol. 2001, 92, 113–131. [Google Scholar] [CrossRef]
- Warneck, P.; Williams, J. Aqueous Phase Chemistry. In The Atmospheric Chemist’s Companion: Numerical Data for Use in the Atmospheric Sciences, 1st ed.; Warneck, P., Williams, J., Eds.; Springer: Dodrecht, The Netherlands, 2012; pp. 271–314. [Google Scholar]
- Converti, A.; Lodi, A.; Del Borghi, A.; Solisio, C. Cultivation of Spirulina platensis in a combined airlift-tubular reactor system. Biochem. Eng. J. 2006, 32, 13–18. [Google Scholar] [CrossRef]
- Torzillo, G.; Sacchi, A.; Materassi, R.; Richmond, A. Effect of temperature on yield and night biomass loss in Spirulina platensis grown outdoors in tubular photobioreactors. J. Appl. Phycol. 1991, 3, 103–109. [Google Scholar] [CrossRef]
- Travieso, L.; Hall, D.O.; Rao, K.K.; Benítez, F.; Sánchez, E.; Borja, R. A helical tubular photobioreactor producing Spirulina in a semicontinuous mode. Int. Biodeterior. Biodegrad. 2001, 47, 151–155. [Google Scholar] [CrossRef]
- Acién Fernández, F.G.; Fernández Sevilla, J.M.; Sánchez Pérez, J.A.; Grima, E.M.; Chisti, Y. Airlift-driven external-loop tubular photobioreactors for outdoor production of microalgae: Assessment of design and performance. Chem. Eng. Sci. 2001, 56, 2721–2732. [Google Scholar] [CrossRef] [Green Version]
| Microorganism | Culture Conditions | DOC (mg L−1) | Effects on Pigment Composition | Source | |
|---|---|---|---|---|---|
| Neochloris oleoabundans | 200 μE (SS); 25 °C | ≈26 ≈35 | decrease in μ by: <2% ≈23% | [65] | |
| 500 μE (NS); 25 °C | ≈17 ≈26 ≈35 | ≈15% ≈32% ≈50% | no impact on and | [60] | |
| Nannochloropsis sp. | 100 μE (SS); 25 °C | ≥6 ≈21 | linear decrease in μ ≈38% of μ left | [66] | |
| Chlorella vulgaris | 250 μE; 25 °C | ≤25 31 | no impacts 30% decrease in Px | no impact on pigments | [26] |
| Scenedesmus almeriensis | 200 μE (SS); 25 °C | ≤9 9–23 >23 32 | maximum OPR decrease in OPR ≤ 20% exponential decline OPR = 0 | [35] | |
| Chlorella protothecoides | 150 μE (SS); 24 °C | ≤16 >16 ≈21–26 | constant OPR exponential decline OPR = 0 | [36] | |
| 1000 μE(NS) | ≈27 | OPR = 0 | |||
| 1500 μE (IL) | ≈30 | OPR = 0 | |||
| Arthrospira maxima | ≈125 μE, 30 °C | ≈16 | no significant effects | reduction of (50%); (70%), (20%) | [62] |
| Arthrospira platensis | ≈140 μE (SS); 30 °C | ≈10 ≈20 ≈30 ≈40 | decrease in μ by: 10% 20% 33% 46% | reduction of ; , (all up to approx. 80%) | [61] |
| 32 μE (SS); 30 °C | ≈40 ≈60 ≥80 | decrease in μ by: ≈13% ≈25% no growth | [67] | ||
| 350 μE (NS-IL); 35 °C | 36 | max. 18% lower ; chlorotic after long exposure (>32 h) | reduction of (30%) | [64] |
| Costache et al. | Sforza et al. | |||||||
|---|---|---|---|---|---|---|---|---|
(μmol m−2 s−1) | (μmol m−2 s−1) | (g L−1) | (mgO2 L−1) | (-) | R2 (-) | (d−1) | (L mgO2−1) | R2 [-] |
| 100 | 16 | 0.5 | 16.13 | 2.35 | 0.9835 | 1.24 × 10−2 | 0.22 | 0.9463 |
| 16.45 | 2.30 | 0.9882 | 1.42 × 10−2 | 0.18 | 0.9947 | |||
| 13 | 1 | 24.51 | 1.61 | 0.9761 | 2.58 × 10−2 | 0.09 | 0.9805 | |
| 23.76 | 1.66 | 0.9831 | 2.67 × 10−2 | 0.08 | 0.9914 | |||
| 150 | 49 | 0.1 | 10.16 | 5.49 | 0.9606 | 1.52 × 10−4 | 0.61 | 0.9103 |
| 10.44 | 8.69 | 0.9710 | 1.25 × 10−4 | 0.93 | 0.9378 | |||
| 24 | 0.5 | 19.14 | 1.62 | 0.9610 | 3.86 × 10−2 | 0.13 | 0.9126 | |
| 21.40 | 2.07 | 0.9844 | 1.05 × 10−2 | 0.18 | 0.9413 | |||
| 21 | 0.8 | 22.80 | 1.78 | 0.9847 | 5.24 × 10−2 | 0.11 | 0.9442 | |
| 27.36 | 1.71 | 0.9870 | 4.49 × 10−2 | 0.09 | 0.9466 | |||
| 400 | 131 | 0.1 | 8.97 | 5.08 | 0.9886 | 6.47 × 10−3 | 0.64 | 0.9896 |
| 8.45 | 6.70 | 0.9990 | 1.16 × 10−3 | 0.85 | 0.9971 | |||
| 64 | 0.5 | 21.09 | 2.80 | 0.9937 | 3.68 × 10−2 | 0.17 | 0.9763 | |
| 21.37 | 3.19 | 0.9928 | 3.82 × 10−2 | 0.17 | 0.9831 | |||
| Parameter | Value | Remarks/Source of Supply |
|---|---|---|
| 6 cm | Assumption; tube diameter equal to the light path length Z | |
| 1800 m 450 m | Serial configurationParallel configuration | |
| variable | ||
| variable | ||
| 4 h−1 | Assumption, inspired by [28] | |
| 0.02 | Assumption, inspired by [28] | |
| 7.4 mgO2 L−1 | Solubility concentration of oxygen at 30 °C | |
| 0.35 m s−1 | Based on [68] | |
| 30 °C | Assuming constant temperature conditions | |
| 0.21 atm | [39] | |
| 1.2 × 10−5 mol m−3 Pa−1 | [69] | |
| Kinetic parameters | ||
| 49.4 μmol m−2 s−1 | This work | |
| 1.97 gO2 gx−1 | [25] | |
| [L cm−1 g−1] = | This work | |
| = 80.489(−0.476) | This work | |
| 1.85 | This work | |
| 0.032 h−1 | Experimentally determined | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Franke, S.; Steingröwer, J.; Walther, T.; Krujatz, F. The Oxygen Paradigm—Quantitative Impact of High Concentrations of Dissolved Oxygen on Kinetics and Large-Scale Production of Arthrospira platensis. ChemEngineering 2022, 6, 14. https://doi.org/10.3390/chemengineering6010014
Franke S, Steingröwer J, Walther T, Krujatz F. The Oxygen Paradigm—Quantitative Impact of High Concentrations of Dissolved Oxygen on Kinetics and Large-Scale Production of Arthrospira platensis. ChemEngineering. 2022; 6(1):14. https://doi.org/10.3390/chemengineering6010014
Chicago/Turabian StyleFranke, Sabine, Juliane Steingröwer, Thomas Walther, and Felix Krujatz. 2022. "The Oxygen Paradigm—Quantitative Impact of High Concentrations of Dissolved Oxygen on Kinetics and Large-Scale Production of Arthrospira platensis" ChemEngineering 6, no. 1: 14. https://doi.org/10.3390/chemengineering6010014
APA StyleFranke, S., Steingröwer, J., Walther, T., & Krujatz, F. (2022). The Oxygen Paradigm—Quantitative Impact of High Concentrations of Dissolved Oxygen on Kinetics and Large-Scale Production of Arthrospira platensis. ChemEngineering, 6(1), 14. https://doi.org/10.3390/chemengineering6010014







