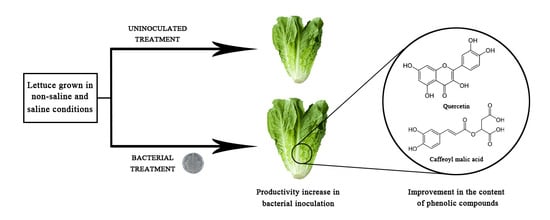
Rhizobium laguerreae Improves Productivity and Phenolic Compound Content of Lettuce (Lactuca sativa L.) under Saline Stress Conditions
Abstract
1. Introduction
2. Materials and Methods
2.1. Bacterial Strain and Green Fluorescent Protein (GFP) Labeling
2.2. Phylogenetic Analysis
2.3. HUTR05 Genome Sequencing and Annotation
2.4. Analysis of In Vitro PGPR Mechanisms
2.5. Colonization Assays and In Vitro Effect on Plant Growth
2.6. Growth Promotion and Saline Tolerance Assays in Plants
2.7. Phenolic Analysis of Lactuca sativa L. Leaves
2.8. Statistics
3. Results and Discussion
3.1. Isolation and Phylogenetic Classification of HUTR05 Strain
3.2. Analysis of In Vitro PGPR Mechanisms
3.3. HUTR05 Draft Genome Analysis
3.4. Roots Colonization Assays and In Vitro Growth Promotion of Lettuce
3.5. Effects on Lettuce Growth Promotion and Tolerance to Salinity Conditions
3.6. Analysis of Phenolic Compounds of Lactuca sativa L. Leaves
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Llorach, R.; Martínez-Sánchez, A.; Tomás-Barberán, F.A.; Gil, M.I.; Ferreres, F. Characterisation of polyphenols and antioxidant properties of five lettuce varieties and escarole. Food Chem. 2008, 108, 1028–1038. [Google Scholar] [CrossRef] [PubMed]
- Baslam, M.; Garmendia, I.; Goicoechea, N. The arbuscular mycorrhizal symbiosis can overcome reductions inyield and nutritional quality in greenhouse-lettuces cultivated atinappropriate growing seasons. Sci. Hortic. 2013, 164, 145–154. [Google Scholar] [CrossRef]
- Avio, L.; Sbrana, C.; Giovannetti, M.; Frassinetti, S. Arbuscular mycorrhizal fungi affect total phenolics content and antioxidant activity in leaves of oak leaf lettuce varieties. Sci. Hortic. 2017, 224, 265–271. [Google Scholar] [CrossRef]
- Shatilov, M.V.; Razin, A.F.; Ivanova, M.I. Analysis of the world lettuce market. IOP Conf. Ser. Earth Environ. Sci. 2019, 395, 012053. [Google Scholar] [CrossRef]
- Ahmed, S.; Ahmed, S.; Roy, S.K.; Woo, S.H.; Sonawane, K.D.; Shohael, A.M. Effect of salinity on the morphological, physiological and biochemical properties of lettuce (Lactuca sativa L.) in Bangladesh. Open Agric. 2019, 4, 361–373. [Google Scholar] [CrossRef]
- Waśkiewicz, A.; Muzolf-Panek, M.; Goliński, P. Phenolic content changes in plants under salt stress. In Ecophysiology and Responses of Plants under Salt Stress; Ahmad, P., Azooz, M., Prasad, M., Eds.; Springer: New York, NY, USA, 2013; pp. 283–314. [Google Scholar]
- Lim, J.H.; Park, K.J.; Kim, B.K.; Jeong, J.W.; Kim, H.J. Effect of salinity stress on phenolic compounds and carotenoids in buckwheat (Fagopyrum esculentum M.) sprout. Food Chem. 2012, 135, 1065–1070. [Google Scholar] [CrossRef]
- Dueñas, M.; Martínez-Villaluenga, C.; Limón, R.I.; Peñas, E.; Frias, J. Effect of germination and elicitation on phenolic composition and bioactivity of kidney beans. Food Res. Int. 2015, 70, 55–63. [Google Scholar] [CrossRef]
- Kaushal, M.; Wani, S.P. Rhizobacterial-plant interactions: Strategies ensuring plant growth promotion under drought and salinity stress. Agric. Ecosyst. Environ. 2016, 231, 68–78. [Google Scholar] [CrossRef]
- Benidire, L.; Lahrouni, M.; Daoui, K.; Fatemi, Z.A.; Carmona, R.G.; Goöttfert, M.; Oufdou, K. Phenotypic and genetic diversity of Moroccan rhizobia isolated from Vicia faba and study of genes that are likely to be involved in their osmotolerance. Syst. Appl. Microbiol. 2017, 41, 51–61. [Google Scholar] [CrossRef]
- Bano, A.; Fatima, M. Salt tolerance in Zea mays (L.) following inoculation with Rhizobium and Pseudomonas. Biol. Fertil. Soil 2009, 45, 405–413. [Google Scholar] [CrossRef]
- Roubtsova, T.V.; Bostock, R.M. Episodic abiotic stress as a potential contributing factor to onset and severity of disease caused by Phytophthora ramorum in Rhododendron and Viburnum. Plant Dis. 2009, 93, 912–918. [Google Scholar] [CrossRef] [PubMed]
- Egamberdieva, D.; Lugtenberg, B. Use of plant growth-promoting rhizobacteria to alleviate salinity stress in plants. In Use of Microbes for the Alleviation of Soil Stresses; Miransari, M., Ed.; Springer: New York, NY, USA, 2014; pp. 73–96. [Google Scholar]
- Radhakrishnan, R.; Baek, K.H. Physiological and biochemical perspectives of non-salt tolerant plants during bacterial interaction against soil salinity. Plant Physiol. Biochem. 2017, 116, 116–126. [Google Scholar] [CrossRef] [PubMed]
- Niu, G.; Davis, T.D.; Masabni, J. A review of salinity tolerance research in horticultural crops. J. Arid Land Stud. 2019, 29, 53–59. [Google Scholar]
- Santander, C.; Ruiz, A.; García, S.; Aroca, R.; Cumming, J.; Cornejo, P. Efficiency of two arbuscular mycorrhizal fungal inocula to improve saline stress tolerance in lettuce plants by changes of antioxidant defense mechanisms. J. Sci. Food Agric. 2020, 100, 1577–1587. [Google Scholar] [CrossRef] [PubMed]
- Flores-Félix, J.D.; Velázquez, E.; García-Fraile, P.; González-Andrés, F.; Silva, L.R.; Rivas, R. Rhizobium and Phyllobacterium bacterial inoculants increase bioactive compounds and quality of strawberries cultivated in field conditions. Food Res. Int. 2018, 111, 416–422. [Google Scholar] [CrossRef] [PubMed]
- Jiménez-Gómez, A.; Flores-Félix, J.D.; García-Fraile, P.; Mateos, P.F.; Menéndez, E.; Velázquez, E.; Rivas, R. Probiotic activities of Rhizobium laguerreae on growth and quality of spinach. Sci. Rep. 2018, 8, e00295. [Google Scholar] [CrossRef]
- Maurya, R.; Verma, S.; Bahadur, I. Advances in the application of plant growth-promoting rhizobacteria in horticulture. In Plant Growth Promoting Rhizobacteria for Agricultural Sustainability; Kumar, A., Meena, V.S., Eds.; Springer: Singapore, 2019; pp. 67–76. [Google Scholar]
- Jiménez-Gómez, A.; García-Estévez, I.; García-Fraile, P.; Escribano-Bailón, M.T.; Rivas, R. Increase in phenolics compounds of Coriandrum sativum L. after the application of a Bacillus halotolerans biofertilizer. J. Sci. Food Agric. 2020, 100, 2742–2749. [Google Scholar] [CrossRef]
- Seshadri, R.; Reeve, W.G.; Ardley, J.K.; Tennessen, K.; Woyke, T.; Kyrpides, N.C.; Ivanova, N.N. Discovery of novel plant interaction determinants from the genomes of 163 root nodule bacteria. Sci. Rep. 2015, 5, e16825. [Google Scholar] [CrossRef]
- García-Fraile, P.; Carro, L.; Robledo, M.; Ramírez-Bahena, M.H.; Flores-Félix, J.D.; Fernández, M.T.; Mateos, P.F.; Rivas, R.; Igual, J.M.; Martínez-Molina, E.; et al. Rhizobium promotes non-legumes growth and quality in several production steps: Towards a biofertilization of edible raw vegetables healthy for humans. PLoS ONE 2012, 7, e38122. [Google Scholar] [CrossRef]
- Rivas, R.; García-Fraile, P.; Mateos, P.F.; Martínez-Molina, E.; Velázquez, E. Characterization of xylanolytic bacteria present in the bract phyllosphere of the date palm Phoenix dactylifera. Lett. Appl. Microbiol. 2007, 44, 181–187. [Google Scholar] [CrossRef]
- Flores-Félix, J.D.; Sánchez-Juanes, F.; García-Fraile, P.; Valverde, A.; Mateos, P.F.; Gónzalez-Buitrago, J.M.; Velázquez, E.; Rivas, R. Phaseolus vulgaris is nodulated by the symbiovar viciae of several genospecies of Rhizobium laguerreae complex in a Spanish region where Lens culinaris is the traditionally cultivated legume. Syst. Appl. Microbiol. 2019, 42, 240–247. [Google Scholar] [CrossRef]
- Zerbino, D.R.; Birney, E. Velvet: Algorithms for de novo short read assembly using de Bruijn graphs. Genome Res. 2008, 18, 821–829. [Google Scholar] [CrossRef] [PubMed]
- Overbeek, R.; Olson, R.; Pusch, G.D.; Olsen, G.J.; Davis, J.J.; Disz, T.; Edwars, R.A.; Gerdes, S.; Parrello, B.; Shukla, M.; et al. The SEED and the rapid annotation of microbial genomes using subsystems technology (RAST). Nucleic Acids Res. 2014, 42, 206–214. [Google Scholar] [CrossRef] [PubMed]
- Pikovskaya, R.I. Mobilization of phosphorus in soil in connection with vital activity of some microbial species. Mikrobiologiya 1948, 17, 362–370. [Google Scholar]
- Nautiyal, C.S. An efficient microbiological growth medium for screening phosphate solubilizing microorganisms. FEMS Microbiol. Lett. 1999, 170, 265–270. [Google Scholar] [CrossRef]
- Alexander, D.B.; Zuberer, D.A. Use of chrome azurol S reagents to evaluate siderophore production by rhizosphere bacteria. Biol. Fertil. Soils 1991, 12, 39–45. [Google Scholar] [CrossRef]
- Flores-Félix, J.D.; Menéndez, E.; Rivera, L.P.; Marcos-García, M.; Martínez-Hidalgo, P.; Mateos, P.F.; Martínez-Molina, E.; Velázquez, M.D.L.E.; García-Fraile, P.; Rivas, R. Use of Rhizobium leguminosarum as a potential biofertilizer for Lactuca sativa and Daucus carota crops. J. Plant Nutr. Soil Sci. 2013, 176, 876–882. [Google Scholar] [CrossRef]
- Saïdi, S.; Ramírez-Bahena, M.H.; Santillana, N.; Zuniga, D.; Álvarez-Martínez, E.; Peix, A.; Mhamdi, R.; Velázquez, E. Rhizobium laguerreae sp. nov. nodulates Vicia faba on several continents. Int. J. Syst. Evolut. Microbiol. 2014, 64, 242–247. [Google Scholar] [CrossRef]
- Wang, D.; Couderc, F.; Tian, C.F.; Gu, W.; Liu, L.X.; Poinsot, V. Conserved composition of nod factors and exopolysaccharides produced by different phylogenetic lineage Sinorhizobium strains nodulating soybean. Front. Microbiol. 2018, 9, e02852. [Google Scholar] [CrossRef]
- Menéndez, E.; Robledo, M.; Jiménez-Zurdo, J.I.; Velázquez, E.; Rivas, R.; Murray, J.D.; Mateos, P.F. Legumes display common and host-specific responses to the rhizobial cellulase CelC2 during primary symbiotic infection. Sci. Rep. 2019, 9, 1–10. [Google Scholar] [CrossRef]
- Petridis, A.; Therios, I.; Samouris, G.; Tananaki, C. Salinity-induced changes in phenolic compounds in leaves and roots of four olive cultivars (Olea europaea L.) and their relationship to antioxidant activity. Environ. Exp. Bot. 2012, 79, 37–43. [Google Scholar] [CrossRef]
- Bistgani, Z.E.; Hashemi, M.; DaCosta, M.; Craker, L.; Maggi, F.; Morshedloo, M.R. Effect of salinity stress on the physiological characteristics, phenolic compounds and antioxidant activity of Thymus vulgaris L. and Thymus daenensis Celak. Ind. Crops Prod. 2019, 135, 311–320. [Google Scholar] [CrossRef]
- Kim, H.J.; Fonseca, J.M.; Choi, J.H.; Kubota, C.; Kwon, D.Y. Salt in irrigation water affects the nutritional and visual properties of romaine lettuce (Lactuca sativa L.). J. Agric. Food Chem. 2008, 56, 3772–3776. [Google Scholar] [CrossRef] [PubMed]
- Mahmoudi, H.; Huang, J.; Gruber, M.Y.; Kaddour, R.; Lachaâl, M.; Ouerghi, Z.; Hannoufa, A. The impact of genotype and salinity on physiological function, secondary metabolite accumulation, and antioxidative responses in lettuce. J. Agric. Food Chem. 2010, 58, 5122–5130. [Google Scholar] [CrossRef] [PubMed]
- Ismail, H.; Maksimovic, J.D.; Maksimovic, V.; Shabala, L.; Živanovic, B.D.; Tian, Y.; Jacobsen, S.E.; Shabala, S. Rutin, a flavonoid with antioxidant activity, improves plant salinity tolerance by regulating K+ retention and Na+ exclusion from leaf mesophyll in quinoa and broad beans. Funct. Plant Biol. 2016, 43, 75–86. [Google Scholar] [CrossRef]
- Nautiyal, C.S.; Govindarajan, R.; Lavania, M.; Pushpangadan, P. Novel mechanism of modulating natural antioxidants in functional foods: Involvement of plant growth promoting rhizobacteria NRRL B-30488. J. Agric. Food Chem. 2008, 56, 4474–4481. [Google Scholar] [CrossRef] [PubMed]

| In Vitro Growth Promotion Assays | ||||||||
| Treatment | Shoot Length (±S.E.) (mm) | Root Length (±S.E.) (mm) | Number of Secondary Roots (±S.E.) | Number of Leaves (±S.E.) | Shoot Length (±S.E.) (mm) | Root Length (±S.E.) (mm) | Number of Secondary Roots (±S.E.) | Number of Leaves (±S.E.) |
| 7 dpi | 14 dpi | |||||||
| Control | 19.91 (±0.85) | 24.61 (±0.83) | 2.75 (±0.17) | 2.73 (±0.12) | 26.94 (±0.88) | 59.68 (±2.64) | 3.32 (±0.21) | 3.67 (±0.11) |
| HUTR05 | 21.25 (±0.48) | 28.77 (±1.58) * | 2.62 (±0.21) | 3.00 (±0.00) * | 26.68 (±1.25) | 56.50 (±3.02) | 4.13 (±0.36) * | 3.90 (±0.07) |
| Greenhouse Assays | ||||||||
| Treatment | Chlorophyll (±S.E.) (SPAD Units) | Number of leaves (±S.E.) | SFW (±S.E.) (g) | SDW (±S.E.) (g) | Chlorophyll (±S.E.) (SPAD Units) | Number of leaves (±S.E.) | SFW (±S.E.) (g) | SDW (±S.E.) (g) |
| Non-saline conditions | 100 mM NaCl | |||||||
| Control | 25.34 (±0.72) | 5.53 (±0.23) | 2.39 (±0.26) | 0.14 (±0.01) | 25.54 (±0.54) | 5.29 (±0.17) | 2.95 (±0.18) | 0.12 (±0.01) |
| HUTR05 | 26.00 (±0.53) | 6.56 (±0.25) * | 4.54 (±0.28) * | 0.17 (±0.01) | 29.32 (±0.69) * | 5.53 (±0.19) | 3.46 (±0.16) | 0.16 (±0.01) * |
| Normal Conditions | 100 mM NaCl | |||
|---|---|---|---|---|
| Control (±S.E.) | HUTR05 (±S.E.) | Control (±S.E.) | HUTR05 (±S.E.) | |
| N (%) | 5.68 (±0.15) | 6.09 (±0.06) * | 5.31 (±0.05) # | 5.22 (±0.02) |
| Ca (g/plant kg) | 13.38 (±1.04) | 14.28 (±0.25) | 11.30 (±0.21) # | 10.48 (±0.56) |
| Fe (mg/plant kg) | 225.39 (±139.42) | 132.80 (±4.17) | 109.89 (±15.94) | 137.07 (±16.53) |
| K (g/plant kg) | 92.69 (±1.19) | 110.85 (±4.15) | 99.08 (±7.34) | 91.89 (±3.64) |
| Mg (g/plant kg) | 6.21 (±0.89) | 7.34 (±0.29) | 5.21 (±0.09) | 4.89 (±0.29) |
| Mn (mg/plant kg) | 46.36 (±9.46) | 49.45 (±2.19) | 47.217 (±1.91) | 45.88 (±2.18) |
| Na (g/plant kg) | 3.23 (±0.32) | 3.58 (±0.11) | 22.75 (±0.41) # | 22.68 (±0.83) |
| P (g/plant kg) | 8.10 (±0.27) | 10.09 (±0.12) * | 8.75 (±0.24) | 7.95 (±0.22) |
| Phenolics Acids (g kg−1) | Normal Conditions | 100 mM NaCl | ||
|---|---|---|---|---|
| Control (±S.E.) | HUTR05 (±S.E.) | Control (±S.E.) | HUTR05 (±S.E.) | |
| Caffeoyl malic acid | 1.690 (±0.032) | 1.914 (±0.013) * | 1.741 (±0.087) | 2.013 (±0.002) * |
| Caffeoyl quinic acids | 3.566 (±0.018) | 5.080 (±0.023) * | 4.490 (±0.245) # | 4.691 (±0.018) |
| Caffeoyl tartaric acid | 1.868 (±0.013) | 2.417 (±0.010) * | 3.697 (±0.163) # | 4.172 (±0.043) * |
| Cichoric acid (dicaffeoyltartaric acid) | 10.293 (±0.056) | 16.893 (±0.177) * | 12.682 (±0.734) # | 12.452 (±0.109) |
| Coumaric acid | 0.390 (±0.009) | 0.520 (±0.013) * | 0.506 (±0.035) # | 0.588 (±0.007) * |
| Dicaffeoyl quinic acid | 0.765 (±0.016) | 1.610 (±0.014) * | 0.724 (±0.038) | 0.797 (±0.015) |
| Protocatechuic acid glucoside | 0.370 (±0.012) | 0.664 (±0.021) * | 0.522 (±0.054) # | 0.830 (±0.010) * |
| Total phenolics acids | 18.942 (±0.079) | 29.097 (±0.134) * | 24.361 (±1.352) # | 25.542 (±0.156) |
| Flavonoids (g kg−1) | Normal Conditions | 100 mM NaCl | ||
|---|---|---|---|---|
| Control (±S.E.) | HUTR05 (±S.E.) | Control (±S.E.) | HUTR05 (±S.E.) | |
| Apigenin derivative | 0.137 (±0.001) | 0.449 (±0.018) * | 0.229 (±0.014) # | 0.717 (±0.009) * |
| Luteolin 7-O-glucoside | 0.231 (±0.003) | 0.341 (±0.010) * | 0.324 (±0.021) # | 0.314 (±0.006) |
| Quercetin 3-O-glucuronide | 2.370 (±0.014) | 3.758 (±0.056) * | 2.862 (±0.145) # | 2.436 (±0.007) |
| Quercetin 3-O-malonyl glucoside | 3.185 (±0.025) | 4.597 (±0.055) * | 4.115 (±0.093) # | 3.745 (±0.021) |
| Quercetin 3-O-glucoside | 0.810 (±0.010) | 2.732 (±0.042) * | 1.437 (±0.082) # | 1.417 (±0.001) |
| Total flavonoids | 6.733 (±0.046) | 11.877 (±0.076) * | 8.967 (±0.339) # | 8.629 (±0.039) |
| Total Phenolic Content | Total Flavonoid Content | |||
|---|---|---|---|---|
| F-Value | p-Value | F-Value | p-Value | |
| Salinity | 5.555 | 0.0462 | 557.3 | 0.0011 |
| Inoculation | 205.5 | <0.0001 | 24.81 | <0.0001 |
| Interaction salinity × inoculation | 128.8 | <0.0001 | 725.1 | <0.0001 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ayuso-Calles, M.; García-Estévez, I.; Jiménez-Gómez, A.; Flores-Félix, J.D.; Escribano-Bailón, M.T.; Rivas, R. Rhizobium laguerreae Improves Productivity and Phenolic Compound Content of Lettuce (Lactuca sativa L.) under Saline Stress Conditions. Foods 2020, 9, 1166. https://doi.org/10.3390/foods9091166
Ayuso-Calles M, García-Estévez I, Jiménez-Gómez A, Flores-Félix JD, Escribano-Bailón MT, Rivas R. Rhizobium laguerreae Improves Productivity and Phenolic Compound Content of Lettuce (Lactuca sativa L.) under Saline Stress Conditions. Foods. 2020; 9(9):1166. https://doi.org/10.3390/foods9091166
Chicago/Turabian StyleAyuso-Calles, Miguel, Ignacio García-Estévez, Alejandro Jiménez-Gómez, José D. Flores-Félix, M. Teresa Escribano-Bailón, and Raúl Rivas. 2020. "Rhizobium laguerreae Improves Productivity and Phenolic Compound Content of Lettuce (Lactuca sativa L.) under Saline Stress Conditions" Foods 9, no. 9: 1166. https://doi.org/10.3390/foods9091166
APA StyleAyuso-Calles, M., García-Estévez, I., Jiménez-Gómez, A., Flores-Félix, J. D., Escribano-Bailón, M. T., & Rivas, R. (2020). Rhizobium laguerreae Improves Productivity and Phenolic Compound Content of Lettuce (Lactuca sativa L.) under Saline Stress Conditions. Foods, 9(9), 1166. https://doi.org/10.3390/foods9091166