Mitigating Milk-Associated Bacteria through Inducing Zinc Ions Antibiofilm Activity
Abstract
1. Introduction
2. Materials and Methods
2.1. Strains and Growth Conditions
2.2. Visualizing Biofilm Bundles Using Confocal Laser Scanning Microscopy (CLSM)
2.3. Examination of Expression of Matrix Gene in B. subtilis Using CLSM and Florescence Microscope
2.4. RNA Extraction and Real-Time Reverse Transcription PCR
2.5. Visualization of Morphological Changes in Bacterial Cells Exposed to Zinc Using Scanning Electron Microscopy (SEM)
2.6. Analysis of Survival Rates Following Heat Treatment
2.7. Statistical Analysis
3. Results
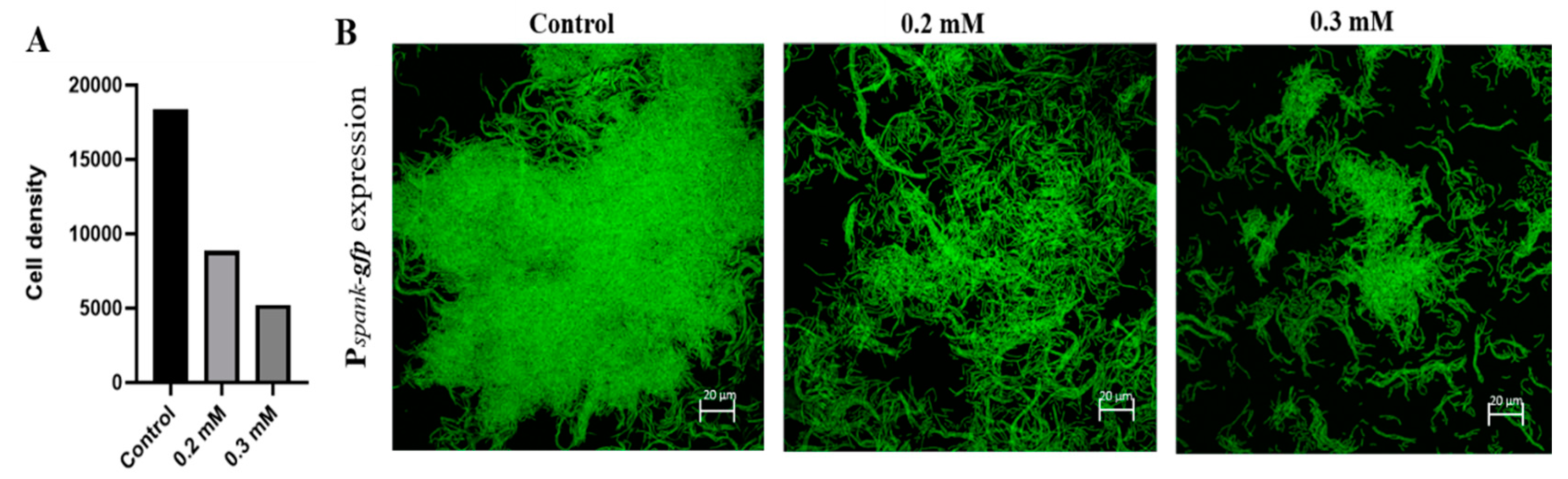
3.1. Sub-Lethal Concentarations of Zn2+ Ions Inhibit Biofilm Formation by B. subtilis
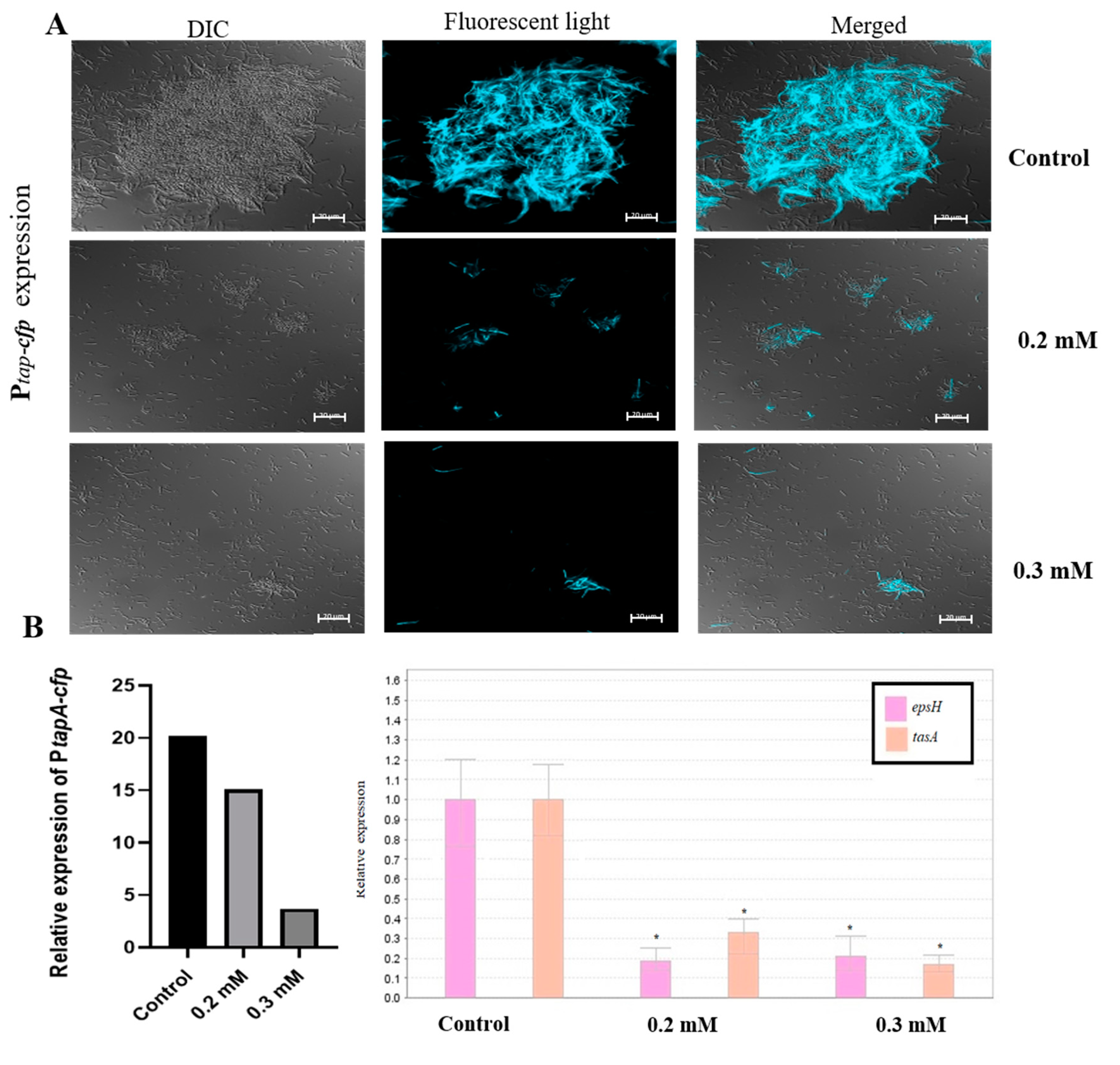
3.2. Zn2+ Ions Inhibit Biofilm Formation through Downregulation of Genes Involved in Construction of Extracellular Matrix
3.3. Zn2+ Ions Disrupt Biofilm Bundles
3.4. Morphological Changes in Bacterial Cells Exposed to Zinc
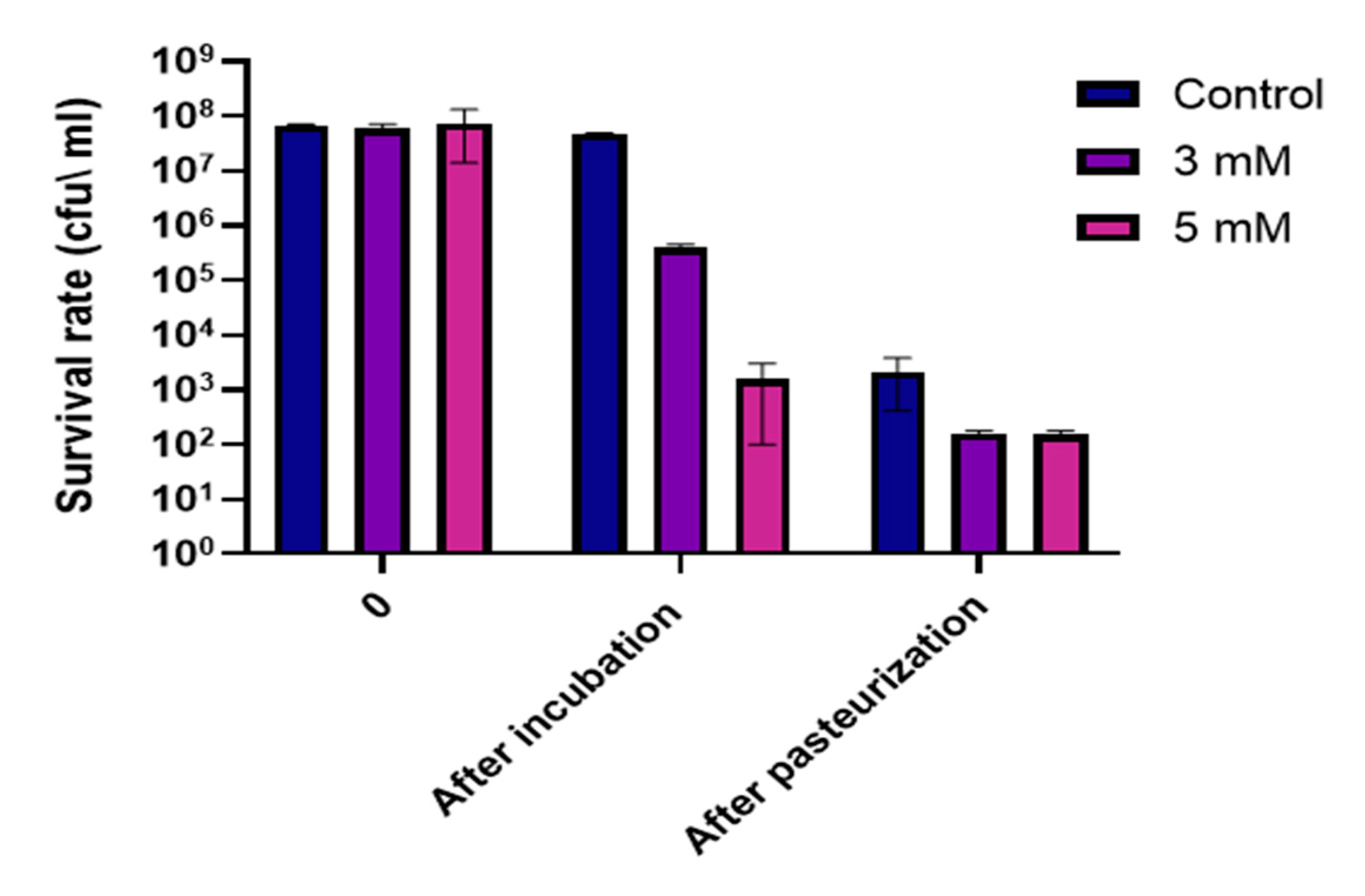
3.5. Bacterial Cells Exposed to Zinc are More Sensitive to Heat Treatment
4. Discussion
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Ranieri, M.L.; Ivy, R.; Mitchell, R.; Call, E.; Masiello, S.; Boor, M. Real-time PCR detection of Paenibacillus spp. in raw milk to predict shelf life performance of pasteurized fluid milk products. Appl. Environ. Microbiol. 2012, 78, 5855–5863. [Google Scholar] [CrossRef] [PubMed]
- Ivy, R.A.; Raniery, M.L.; Martin, N.; Den Bakker, N.; Xavier, H.; Wiedman, B.; Boor, M. Identification and characterization of psychrotolerant sporeformers associated with fluid milk production and processing. Appl. Environ. Microbiol. 2012, 78, 1853–1864. [Google Scholar] [CrossRef] [PubMed]
- Srey, S.; Jahid, I.; Ha, S.D. Biofilm formation in food industries: A food safety concern. Food Control 2013, 31, 572–585. [Google Scholar] [CrossRef]
- Shemesh, M.; Kolter, R.; Losick, R. The biocide chlorine dioxide stimulates biofilm formation in Bacillus subtilis by activation of the histidine kinase KinC. J. Bacteriol. 2010, 192, 6352–6356. [Google Scholar] [CrossRef] [PubMed]
- Allison, D.G. The biofilm matrix. Biofouling 2003, 19, 139–150. [Google Scholar] [CrossRef]
- Shaheen, R.; Svensson, B.; Andersson, M.A.; Christiansson, A.; Salkinoja-Salonen, M. Persistence strategies of Bacillus cereus spores isolated from dairy silo tanks. Food Microbiol. 2010, 27, 347–355. [Google Scholar] [CrossRef]
- Checinska, A.; Paszczynski, A.; Burbank, M. Bacillus and other spore-forming genera: Variations in responses and mechanisms for survival. Annu. Rev. Food Sci. Technol. 2015, 6, 351–369. [Google Scholar] [CrossRef]
- Mah, T.F.; O’Toole, G.A. Mechanisms of biofilm resistance to antimicrobial agents. Trends Microbiol. 2001, 9, 34–39. [Google Scholar] [CrossRef]
- Simões, M.; Simões, L.C.; Machado, I.; Pereira, M.O.; Vieira, M.J. Control of flow-generated biofilms with surfactants: Evidence of resistance and recovery. Food Bioprod. Process. 2006, 84, 338–345. [Google Scholar] [CrossRef]
- Simões, M.; Simões, L.C.; Vieira, M.J. A review of current and emergent biofilm control strategies. LWT Food Sci. Technol. 2010, 43, 573–583. [Google Scholar] [CrossRef]
- Shemesh, M.; Ostrov, I. Role of Bacillus species in biofilm persistence and emerging antibiofilm strategies in the dairy industry. J. Sci. Food Agric. 2020, 100, 2327–2336. [Google Scholar] [CrossRef] [PubMed]
- Marchand, S.; Block, J.; De Jonghe, V.; Coorevits, A.; Heyndrickx, M.; Herman, L. Biofilm Formation in Milk Production and Processing Environments; Influence on Milk Quality and Safety. Compr. Rev. Food Sci. Food Saf. 2012, 11, 133–147. [Google Scholar] [CrossRef]
- Pasvolsky, R.; Zakin, V.; Ostrova, I.; Shemesh, M. Butyric acid released during milk lipolysis triggers biofilm formation of Bacillus species. Int. J. Food Microbiol. 2014, 181, 19–27. [Google Scholar] [CrossRef] [PubMed]
- Ostrov, I.; Sela, N.; Belausov, E.; Steinberg, D.; Shemesh, M. Adaptation of Bacillus species to dairy associated environment facilitates their bio fi lm forming ability. J. Food Microbiol. 2019, 82, 316–324. [Google Scholar] [CrossRef] [PubMed]
- Sharma, M.; Anand, S.K. Biofilms evaluation as an essential component of HACCP for food/dairy processing industry—A case. Food Control 2002, 13, 469–477. [Google Scholar] [CrossRef]
- Sharma, M.; Anand, S.K. Characterization of constitutive microflora of biofilms in dairy processing lines. Food Microbiol. 2002, 19, 627–636. [Google Scholar] [CrossRef]
- Lücking, G.; Stoeckel, M.; Atamer, Z.; Hinrichs, J.; Ehling-Schulz, M. Characterization of aerobic spore-forming bacteria associated with industrial dairy processing environments and product spoilage. Int. J. Food Microbiol. 2013, 166, 270–279. [Google Scholar] [CrossRef]
- Kumari, S.; Sarkar, P.K. Bacillus cereus hazard and control in industrial dairy processing environment. Food Control 2016, 69, 20–29. [Google Scholar] [CrossRef]
- Salkinoja-Salonen, M.S.; Vuorio, R.; Andersson, M.A.; Kämpfer, P.; Andersson, M.C.; Honkanen-Buzalski, T.; Scoging, A.C. Toxigenic strains of Bacillus licheniformis related to food poisoning. Appl. Environ. Microbiol. 1999, 65, 4637–4645. [Google Scholar] [CrossRef]
- Nieminen, T.; Rintaluoma, N.; Andersson, M.; Taimisto, A.M.; Ali-Vehmas, T.; Seppälä, A.; Priha, O.; Salkinoja-Salonen, M.S. Toxinogenic Bacillus pumilus and Bacillus licheniformis from mastitic milk. Vet. Microbiol. 2007, 124, 329–339. [Google Scholar] [CrossRef]
- Logan, N.A. Bacillus and relatives in foodborne illness. J. Appl. Microbiol. 2012, 112, 417–429. [Google Scholar] [CrossRef] [PubMed]
- Kearns, D.B.; Chu, F.; Branda, S.S.; Kolter, R.; Losick, R. A master regulator for biofilm formation by Bacillus subtilis. Mol. Microbiol. 2005, 55, 739–749. [Google Scholar]
- Nagórska, K.; Ostrowski, A.; Hinc, K.; Holland, I.B.; Obuchowski, M. Importance of eps genes from Bacillus subtilis in biofilm formation and swarming. J. Appl. Genet. 2010, 51, 369–381. [Google Scholar] [CrossRef]
- van Gestel, J.; Vlamakis, H.; Kolter, R. From Cell Differentiation to Cell Collectives: Bacillus subtilis Uses Division of Labor to Migrate. PLoS Biol. 2015, 13, e1002141. [Google Scholar] [CrossRef] [PubMed]
- Gram, L.; Bagge-Ravn, D.; Ng, Y.Y.; Gymoese, P.; Vogel, B.F. Influence of food soiling matrix on cleaning and disinfection efficiency on surface attached Listeria monocytogenes. Food Control 2007, 18, 1165–1171. [Google Scholar] [CrossRef]
- Harrison, J.J.; Ceri, H.; Stremick, C.A.; Turner, R.J. Biofilm susceptibility to metal toxicity. Environ. Microbiol. 2004, 6, 1220–1227. [Google Scholar] [CrossRef]
- Nies, D.H. Microbial heavy-metal resistance. Appl. Microbiol. Biotechnol. 1999, 51, 730–750. [Google Scholar] [CrossRef]
- Winslow, C.E.; Haywood, E.T. The Specific Potency of Certain Cations with Reference to Their Effect on Bacterial Viability. J. Bacteriol. 1931, 22, 49–69. [Google Scholar] [CrossRef]
- Ou, J.T. Effect of Zn2+ on bacterial conjugation: Increase in ability of F- cells to form mating pairs. J. Bacteriol. 1973, 115, 648–654. [Google Scholar] [CrossRef]
- Wu, C.; Labrie, J.; Tremblay, Y.; Haine, D.; Mourez, M.; Jacques, M. Zinc as an agent for the prevention of biofilm formation by pathogenic bacteria. J. Appl. Microbiol. 2013, 115, 30–40. [Google Scholar] [CrossRef]
- Little, D.J.; Poloczek, J.; Whitney, J.C.; Robinson, H.; Nitz, M.; Howell, P.L. The structure- and metal-dependent activity of Escherichia coli PgaB provides insight into the partial de-N-acetylation of poly-beta-1,6-N-acetyl-D-glucosamine. J. Biol. Chem. 2012, 31126–31137. [Google Scholar] [CrossRef] [PubMed]
- Klemm, P.; Vejborg, R.M.; Hancock, V. Prevention of bacterial adhesion. Appl. Microbiol. Biotechnol. 2010, 88, 451–459. [Google Scholar] [CrossRef] [PubMed]
- Jribi, S.; Molnàr, H.; Antal, T.; Adànyi, N.; Kheriji, O.; Debbabi, H. Zinc fortification as a tool for improving sprout hygienic and nutritional quality: A factorial design approach. J. Sci. Food. Agric. 2019, 99, 5187–5194. [Google Scholar] [CrossRef] [PubMed]
- Holt, B.A.; Gregory, S.A.; Sulchek, T.; Yee, S.; Losego, M.D. Aqueous Zinc Compounds as Residual Antimicrobial Agents for Textiles. ACS. Appl. Mater. Interfaces. 2018, 10, 7709–7716. [Google Scholar] [CrossRef] [PubMed]
- Branda, S.S.; Gonzalez-Pastor, J.E.; Ben-Yehuda, S.; Losick, R.; Kolter, R. Fruiting body formation by Bacillus subtilis. Proc. Natl. Acad. Sci. USA 2001, 98, 11621–11626. [Google Scholar] [CrossRef]
- Chai, Y.; Chu, F.; Kolter, R.; Losick, R. Bistability and biofilm formation in Bacillus subtilis. Mol. Microbiol. 2008, 67, 254–263. [Google Scholar] [CrossRef]
- Chai, Y.; Norman, T.; Kolter, R.; Losick, R. Evidence that metabolism and chromosome copy number control mutually exclusive cell fates in Bacillus subtilis. EMBO J. 2011, 30, 1402–1413. [Google Scholar] [CrossRef]
- Duanis-Assaf, D.; Steinberg, D.; Chai, Y.; Shemesh, M. The LuxS based quorum sensing governs lactose induced biofilm formation by Bacillus subtilis. Front. Microbiol. 2016, 6, 1517. [Google Scholar] [CrossRef]
- Ostrov, I.; Paz, T.; Shemesh, M. Robust biofilm-forming Bacillus isolates from the dairy environment demonstrate an enhanced resistance to cleaning-in-place procedures. Foods 2019, 8, 134. [Google Scholar] [CrossRef]
- Flint, S.H.; Bremer, P.J.; Brooks, J.D. Biofilms in dairy manufacturing plant-description, current concerns and methods of control. Biofouling 1997, 11, 81–97. [Google Scholar] [CrossRef]
- Hsueh, Y.H.; Ke, W.J.; Te Hsieh, C.; Lin, K.S.; Tzou, D.Y.; Chiang, C.L. ZnO nanoparticles affect Bacillus subtilis cell growth and biofilm formation. PLoS ONE 2015, 10, e0128457. [Google Scholar] [CrossRef] [PubMed]
- Huang, Z.; Wu, L.; Li, X.; Ma, L.; Borriss, R.; Gao, X. Zn(II) suppresses biofilm formation in Bacillus amyloliquefaciens by inactivation of the Mn(II) uptake. Environ. Microbiol. 2020, 22, 1547–1558. [Google Scholar] [CrossRef] [PubMed]
- Mhatre, E.; Troszok, A.; Gallegos-Monterrosa, R.; Lindstädt, S.; Hölscher, T.; Kuipers, O.P.; Kovács, Á.T. The impact of manganese on biofilm development of Bacillus subtilis. Microbiol 2016, 162, 1468–1478. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; He, L.; Mustapha, A.; Li, H.; Hu, Z.Q.; Lin, M. Antibacterial activities of zinc oxide nanoparticles against Escherichia coli O157:H7. J. Appl. Microbiol. 2009, 107, 1193–1201. [Google Scholar] [CrossRef]
- Brayner, R.; Ferrari-Iliou, R.; Brivois, N.; Djediat, S.; Benedetti, M.F.; Fievet, F. Toxicological impact studies based on Escherichia coli bacteria in ultrafine ZnO nanoparticles colloidal medium. Nano Lett. 2006, 6, 866–870. [Google Scholar] [CrossRef]
- Stoimenov, P.K.; Klinger, R.L.; Marchin, G.L.; Klabunde, K.J. Metal Oxide Nanoparticles as Bactericidal Agents. Langmuir 2002, 18, 6679–6686. [Google Scholar] [CrossRef]
- Matai, I.; Sachdev, A.; Dubey, P.; Kumar, S.U.; Bhushan, B.; Gopinath, P. Colloids and Surfaces B: Biointerfaces Antibacterial activity and mechanism of Ag – ZnO nanocomposite on S. aureus and GFP-expressing antibiotic resistant E coli. Colloids Surf. B Biointerfaces 2014, 115, 359–367. [Google Scholar] [CrossRef]
- Ghosh, S.; Goudar, V.S.; Padmalekha, K.G.; Bhat, S.V.; Indi, S.S.; Vasan, H.N. ZnO/Ag nanohybrid: Synthesis, characterization, synergistic antibacterial activity and its mechanism. RSC Adv. 2012, 2, 930–940. [Google Scholar] [CrossRef]
- Sinha, R.; Karan, R.; Sinha, A.; Khare, S.K. Interaction and nanotoxic effect of ZnO and Ag nanoparticles on mesophilic and halophilic bacterial cells. Bioresour. Technol. 2011, 102, 1516–1520. [Google Scholar] [CrossRef]
- Chandrangsu, P.; Rensing, C.; Helmann, J.D. Metal homeostasis and resistance in bacteria. Nat. Rev. Microbiol. 2017, 15, 338–350. [Google Scholar] [CrossRef]
- Chandrangsu, P.; Helmann, J.D. Intracellular Zn(II) Intoxication Leads to Dysregulation of the PerR Regulon Resulting in Heme Toxicity in Bacillus subtilis. PLoS Genet. 2016, 12, e1006515. [Google Scholar] [CrossRef] [PubMed]
- Wakeman, C.A.; Hammer, N.D.; Stauff, D.L.; Attia, A.S.; Anzaldi, L.L.; Dikalov, S.I.; Calcutt, M.W.; Skaar, E.P. Menaquinone biosynthesis potentiates haem toxicity in Staphylococcus aureus. Mol. Microbiol. 2012, 86, 1376–1392. [Google Scholar] [CrossRef] [PubMed]
- Bansal, V. Minimally Processed Foods: Overview, Washing, Peeling and Cutting of Fresh-Cut Fruits and Vegetables, 1st ed.; Siddiqui, M.W., Rahman, M.S., Eds.; Springer: New York, NY, USA, 2015; pp. 1–15. [Google Scholar]
- Fellows, P. Food Processing Technology: Principles and Practice, 4th ed.; Woodhead Publishing: London, UK, 2017; pp. 431–512. [Google Scholar]
- Vallee, B.L.; Falchuk, K.H. The biochemical basis of zinc physiology. Physiol. Rev. 1993, 73, 79–118. [Google Scholar] [CrossRef] [PubMed]
- Plum, L.M.; Rink, L.; Haase, H. The essential toxin: Impact of zinc on human health. Int. J. Environ. Res. Public Health 2010, 7, 1342–1365. [Google Scholar] [CrossRef] [PubMed]
- Ackland, M.L.; Michalczyk, A. Zinc deficiency and its inherited disorders—A review. Genes Nutr. 2006, 1, 41–49. [Google Scholar] [CrossRef] [PubMed]


© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hutchings, C.; Rajasekharan, S.K.; Reifen, R.; Shemesh, M. Mitigating Milk-Associated Bacteria through Inducing Zinc Ions Antibiofilm Activity. Foods 2020, 9, 1094. https://doi.org/10.3390/foods9081094
Hutchings C, Rajasekharan SK, Reifen R, Shemesh M. Mitigating Milk-Associated Bacteria through Inducing Zinc Ions Antibiofilm Activity. Foods. 2020; 9(8):1094. https://doi.org/10.3390/foods9081094
Chicago/Turabian StyleHutchings, Carmel, Satish Kumar Rajasekharan, Ram Reifen, and Moshe Shemesh. 2020. "Mitigating Milk-Associated Bacteria through Inducing Zinc Ions Antibiofilm Activity" Foods 9, no. 8: 1094. https://doi.org/10.3390/foods9081094
APA StyleHutchings, C., Rajasekharan, S. K., Reifen, R., & Shemesh, M. (2020). Mitigating Milk-Associated Bacteria through Inducing Zinc Ions Antibiofilm Activity. Foods, 9(8), 1094. https://doi.org/10.3390/foods9081094




