Assessment of the Antimicrobial, Antioxidant, and Antiproliferative Potential of Sideritis raeseri subps. raeseri Essential Oil
Abstract
1. Introduction
2. Materials and Methods
2.1. Standard Compounds
2.2. Plant Material
2.3. Extraction of EO
2.4. Microbial Strains
2.5. Analytical Procedures
2.5.1. GC/MS
2.5.2. Antimicrobial Assays
Screening of S. raeseri subp. raeseri EO Antimicrobial Activity by the Disc Diffusion Assay
Determination of Minimum Inhibitory), Non-Inhibitory, and Minimum Bactericidal Concentrations
2.6. Assessment of Cell-Free Antioxidant Activity by DPPH and ABTS Assays
2.7. Sulforhodamine B Assay (SRB)
2.8. Single Cell Gel Electrophoresis (comet) Assay
2.9. Statistical Analysis
3. Results and Discussion
3.1. GC/MS Analysis
3.2. Antimicrobial Assays
3.3. Determination of Radical Scavenging Activity
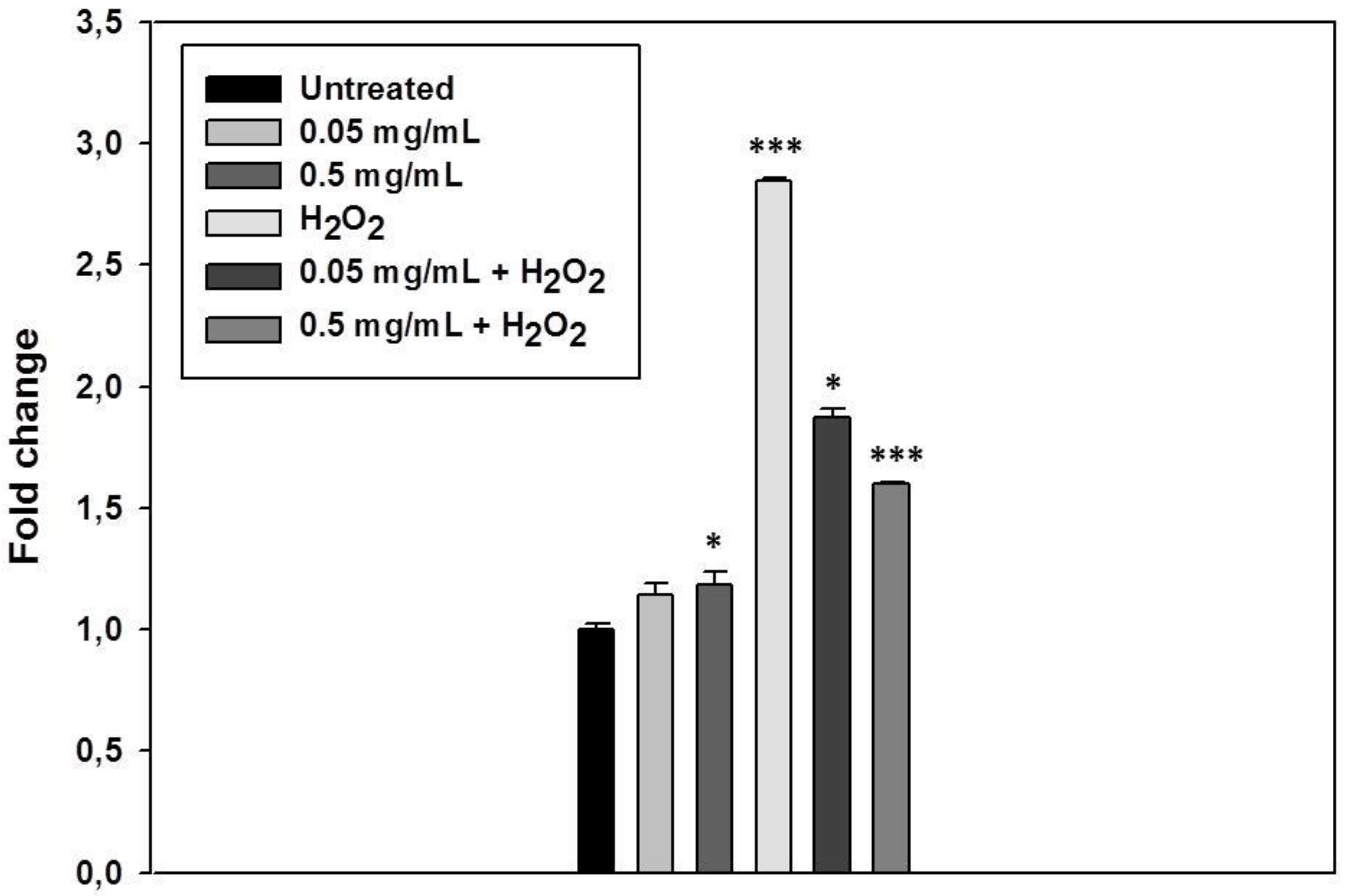
3.4. S. raeseri subsp. raeseri EO Protects Human Epidermal Keratinocytes (HaCaT) Cells from H2O2-Induced DNA Damage
3.5. Determination of Antiproliferative Activity
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Nakatsu, T.N.; Lupo, A.T.; Chinn, J.W.; Kang, R.K. Biological activity of essential oils and their constituents. Stud. Nat. Prod. Chem. 2000, 21, 571–631. [Google Scholar] [CrossRef]
- Rios, J.; Recio, M. Medicinal plants and antimicrobial activity. J. Ethnopharmacol. 2005, 100, 80–84. [Google Scholar] [CrossRef]
- Nychas, G.-J.; Skandamis, P.; Tassou, C. Antimicrobials from herbs and spices. In Natural Antimicrobials for the Minimal Processing of Foods; Elsevier BV: Amsterdam, The Netherlands, 2003; pp. 176–200. [Google Scholar]
- Burt, S. Essential oils: Their antibacterial properties and potential applications in foods—A review. Int. J. Food Microbiol. 2004, 94, 223–253. [Google Scholar] [CrossRef] [PubMed]
- Chorianopoulos, N.G.; Evergetis, E.T.; Aligiannis, N.; Mitakou, S.; Nychas, G.-J.E.; Haroutounian, S.A. Correlation between Chemical Composition of Greek Essential Oils and their Antibacterial Activity against Food-borne Pathogens. Nat. Prod. Commun. 2007, 2, 419–426. [Google Scholar] [CrossRef]
- Patelou, E.; Chatzopoulou, P.; Polidoros, A.N.; Mylona, P.V. Genetic diversity and structure of Sideritis raeseri Boiss. & Heldr. (Lamiaceae) wild populations from Balkan Peninsula. J. Appl. Res. Med. Aromat. Plants 2020, 16, 100241. [Google Scholar] [CrossRef]
- Papageorgiou, V.P.; Kokkini, S.; Argyriadou, N. Chemotaxonomy of the Greek Species of Sideritis I. Components of the volatile fraction of Sideritis raeseri ssp. raeseri. In Aromatic Plants; Springer Science and Business Media LLC: Berlin/Heidelberg, Germany, 1982; pp. 211–220. [Google Scholar]
- Garzoli, S.; Navarra, A.; Garzoli, S.; Pepi, F.; Ragno, R. Esential oils extraction: A 24-hour steam distillation systematic methodology. Nat. Prod. Res. 2017, 31, 2387–2396. [Google Scholar] [CrossRef]
- Aligiannis, N.; Kalpoutzakis, E.; Chinou, I.B.; Mitakou, S.; Gikas, E.; Tsarbopoulos, A. Composition and Antimicrobial Activity of the Essential Oils of Five Taxa of Sideritis from Greece. J. Agric. Food Chem. 2001, 49, 811–815. [Google Scholar] [CrossRef] [PubMed]
- Koedam, A. Volatile oil composition of greek mountain tea (Sideritis spp.). J. Sci. Food Agric. 1986, 37, 681–684. [Google Scholar] [CrossRef]
- González-Burgos, E.; Carretero, M.; Gómez-Serranillos, M.P. Sideritis spp.: Uses, chemical composition and pharmacological activities—A review. J. Ethnopharmacol. 2011, 135, 209–225. [Google Scholar] [CrossRef]
- Stagos, D.; Portesis, N.; Spanou, C.; Mossialos, D.; Aligiannis, N.; Chaita, E.; Panagoulis, C.; Reri, E.; Skaltsounis, L.; Tsatsakis, A.; et al. Correlation of total polyphenolic content with antioxidant and antibacterial activity of 24 extracts from Greek domestic Lamiaceae species. Food Chem. Toxicol. 2012, 50, 4115–4124. [Google Scholar] [CrossRef]
- Romanucci, V.; Di Fabio, G.; D’Alonzo, D.; Guaragna, A.; Scapagnini, G.; Zarrelli, A. Traditional uses, chemical composition and biological activities of Sideritis raeseri Boiss. & Heldr. J. Sci. Food Agric. 2016, 97, 373–383. [Google Scholar] [CrossRef]
- Iqbal, M.N.; Anjum, A.A.; Ali, M.A.; Hussain, F.; Ali, S.; Muhammad, A.; Ahmad, A.; Irfan, M.; Shabbir, A. Assessment of Microbial Load of Un-pasteurized Fruit Juices and in vitro Antibacterial Potential of Honey Against Bacterial Isolates. Open Microbiol. J. 2015, 9, 26–32. [Google Scholar] [CrossRef]
- Jay, J.M.; Loessner, M.J.; Golden, D.A. Modern Food Microbiology, 7th ed.; Springer Science Business Media, Inc.: New York, NJ, USA, 2005. [Google Scholar]
- Juvonen, R.; Virkajarvi, V.; Priha, O.; Laitila, A. Microbiological Spoilage and Safety Risks in Non-Beer Beverages Produced in a Brewery Environment; VTT Tiedotteita-Research: Espoo, Finland, 2011. [Google Scholar]
- Aneja, K.R.; Dhiman, R.; Aggarwal, N.K.; Kumar, V.; Kaur, M. Microbes Associated with Freshly Prepared Juices of Citrus and Carrots. Int. J. Food Sci. 2014, 2014, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Mitropoulou, G.; Fitsiou, E.; Spyridopoulou, K.; Tiptiri-Kourpeti, A.; Bardouki, H.; Vamvakias, M.; Panas, P.; Chlichlia, K.; Pappa, A.; Kourkoutas, Y. Citrus medica essential oil exhibits significant antimicrobial and antiproliferative activity. LWT 2017, 84, 344–352. [Google Scholar] [CrossRef]
- Chorianopoulos, N.; Lambert, R.; Skandamis, P.; Evergetis, E.; Haroutounian, S.; Nychas, G.-J.E. A newly developed assay to study the minimum inhibitory concentration of Satureja spinosa essential oil. J. Appl. Microbiol. 2006, 100, 778–786. [Google Scholar] [CrossRef] [PubMed]
- Lambert, R.; Lambert, R. A model for the efficacy of combined inhibitors. J. Appl. Microbiol. 2003, 95, 734–743. [Google Scholar] [CrossRef] [PubMed]
- Arendrup, M.C.; Meletiadis, J.; Mouton, J.W.; Lagrou, K.; Hamal, P.; Guinea, J.; Subcommittee on Antifungal Susceptibility Testing (AFST) of the ESCMID European Committee for Antimicrobial Susceptibility Testing. EUCAST Definitive Document EDEF 9.3.2 Method for the Determination of Broth Dilution Minimum Inhibitory Concentrations of Antifungal Agents for Conidia Forming Moulds. 2020. Available online: https://www.eucast.org/fileadmin/src/media/PDFs/EUCAST_files/AFST/Files/EUCAST_E_Def_9.3.2_Mould_testing_definitive_revised_2020 (accessed on 7 April 2020).
- Arendrup, M.C.; Meletiadis, J.; Mouton, J.W.; Lagrou, K.; Hamal, P.; Guinea, J.; Subcommittee on Antifungal Susceptibility Testing (AFST) of the ESCMID European Committee for Antimicrobial Susceptibility Testing. EUCAST Definitive Document EDEF 7.3.2 Method for the Determination of Broth Dilution Minimum Inhibitory Concentrations of Antifungal Agents for Yeasts. 2020. Available online: https://www.eucast.org/fileadmin/src/media/PDFs/EUCAST_files/AFST/Files/EUCAST_E_Def_7.3.2_Yeast_testing_definitive_revised_2020 (accessed on 8 April 2020).
- Balkan, C.; Ercan, I.; Isik, E.; Akdeniz, E.S.; Balcioglu, O.; Kodedová, M.; Zimmermannová, O.; Dundar, M.; Sychrová, H.; Koc, A. Genomewide Elucidation of Drug Resistance Mechanisms for Systemically Used Antifungal Drugs Amphotericin B, Caspofungin, and Voriconazole in the Budding Yeast. Antimicrob. Agents Chemother. 2019, 63. [Google Scholar] [CrossRef]
- Mandras, N.; Roana, J.; Scalas, D.; Fucale, G.; Allizond, V.; Banche, G.; Barbui, A.; Vigni, N.L.; Newell, V.A.; Cuffini, A.M.; et al. In vitro antifungal activity of fluconazole and voriconazole against non-Candida yeasts and yeast-like fungi clinical isolates. New Microbiol. 2015, 38, 583–587. [Google Scholar]
- Lee, S.K.; Mbwambo, Z.H.; Chung, H.; Luyengi, L.; Gamez, E.J.; Mehta, R.G.; Kinghorn, A.D.; Pezzuto, J.M. Evaluation of the antioxidant potential of natural products. Comb. Chem. High Throughput Screen. 1998, 1, 35. [Google Scholar]
- Re, R.; Pellegrini, N.; Proteggente, A.; Pannala, A.; Yang, M.; Rice-Evans, C. Antioxidant activity applying an improved ABTS radical cation decolorization assay. Free. Radic. Boil. Med. 1999, 26, 1231–1237. [Google Scholar] [CrossRef]
- Fitsiou, E.; Mitropoulou, G.; Spyridopoulou, K.; Tiptiri-Kourpeti, A.; Vamvakias, M.; Bardouki, H.; Panayiotidis, M.; Galanis, A.; Kourkoutas, Y.; Chlichlia, K.; et al. Phytochemical Profile and Evaluation of the Biological Activities of Essential Oils Derived from the Greek Aromatic Plant Species Ocimum basilicum, Mentha spicata, Pimpinella anisum and Fortunella margarita. Molecules 2016, 21, 1069. [Google Scholar] [CrossRef]
- Olive, P.L.; Banáth, J.P. The comet assay: A method to measure DNA damage in individual cells. Nat. Protoc. 2006, 1, 23–29. [Google Scholar] [CrossRef]
- Fitsiou, E.; Mitropoulou, G.; Spyridopoulou, K.; Vamvakias, M.; Bardouki, H.; Galanis, A.; Chlichlia, K.; Kourkoutas, Y.; Panayiotidis, M.; Pappa, A. Chemical Composition and Evaluation of the Biological Properties of the Essential Oil of the Dietary Phytochemical Lippia citriodora. Molecules 2018, 23, 123. [Google Scholar] [CrossRef] [PubMed]
- Panagiotidis, M.; Tsolas, O.; Galaris, D. Glucose oxidase-produced H2O2 induces Ca2+-dependent DNA damage in human peripheral blood lymphocytes. Free Radic. Boil. Med. 1999, 26, 548–556. [Google Scholar] [CrossRef]
- Kırmızıbekmez, H.; Karaca, N.; Demirci, F. Characterization of Sideritis trojana Bornm. essential oil and its antimicrobial activity. Marmara Pharm. J. 2017, 21, 860–865. [Google Scholar] [CrossRef]
- Kirimer, N.; Baser, K.H.C.; Demirci, B.; Duman, H. Essential Oils of Sideritis Species of Turkey Belonging to the Section Empedoclia. Chem. Nat. Compd. 2004, 40, 19–23. [Google Scholar] [CrossRef]
- Chorianopoulos, N.; Kalpoutzakis, E.; Aligiannis, N.; Mitaku, S.; Nychas, G.-J.E.; Haroutounian, S.A. Essential Oils of Satureja, Origanum, and Thymus Species: Chemical Composition and Antibacterial Activities Against Foodborne Pathogens. J. Agric. Food Chem. 2004, 52, 8261–8267. [Google Scholar] [CrossRef]
- Battey, A.S.; Duffy, S.; Schaffner, D. Modeling Yeast Spoilage in Cold-Filled Ready-To-Drink Beverages with Saccharomyces cerevisiae, Zygosaccharomyces bailii, and Candida lipolytica. Appl. Environ. Microbiol. 2002, 68, 1901–1906. [Google Scholar] [CrossRef]
- Garcia, D.; Ramos, A.J.; Sanchis, V.; Marín, S. Predicting mycotoxins in foods: A review. Food Microbiol. 2009, 26, 757–769. [Google Scholar] [CrossRef]
- Deveci, E.; Tel-Çayan, G.; Usluer, Ö.; Duru, M.E. Chemical Composition, Antioxidant, Anticholinesterase and Anti-Tyrosinase Activities of Essential Oils of Two Sideritis Species from Turkey. Iran J. Pharm. Res. 2019, 18, 903–913. [Google Scholar]
- Deveci, E.; Tel-Çayan, G.; Duru, M.E.; Öztürk, M. Phytochemical contents, antioxidant effects, and inhibitory activities of key enzymes associated with Alzheimer’s disease, ulcer, and skin disorders of Sideritis albiflora and Sideritis leptoclada. J. Food Biochem. 2019, 43, e13078. [Google Scholar] [CrossRef]
- Gabrieli, C.; Kefalas, P.; Kokkalou, E. Antioxidant activity of flavonoids from Sideritis raeseri. J. Ethnopharmacol. 2005, 96, 423–428. [Google Scholar] [CrossRef] [PubMed]
- Da Silva, L.C.N.; Filho, C.M.B.; De Paula, R.A.; Silva, C.S.S.E.; De Souza, L.I.O.; Da Silva, L.C.N.; Correia, M.T.D.S.; De Figueiredo, R.C.B.Q. In vitro cell-based assays for evaluation of antioxidant potential of plant-derived products. Free. Radic. Res. 2016, 50, 801–812. [Google Scholar] [CrossRef] [PubMed]
- González-Burgos, E.; Duarte, A.I.; Carretero, M.E.; Moreira, P.; Gómez-Serranillos, M.P. Mitochondrial-Targeted Protective Properties of Isolated Diterpenoids from Sideritis spp. in Response to the Deleterious Changes Induced by H2O2. J. Nat. Prod. 2013, 76, 933–938. [Google Scholar] [CrossRef] [PubMed]
- Tóth, B.; Kúsz, N.; Forgo, P.; Bózsity, N.; Zupkó, I.; Pinke, G.; Hohmann, J.; Vasas, A. Abietane diterpenoids from Sideritis montana L. and their antiproliferative activity. Fitoterapia 2017, 122, 90–94. [Google Scholar] [CrossRef]
- Tadić, V.; Jeremic, I.; Dobrić, S.; Isakovic, A.; Marković, I.; Trajkovic, V.; Bojović, D.; Arsić, I. Anti-inflammatory, Gastroprotective, and Cytotoxic Effects of Sideritis scardica Extracts. Planta Med. 2012, 78, 415–427. [Google Scholar] [CrossRef]
- Jorge, L.; Meniqueti, A.; Silva, R.; Santos, K.; Da Silva, E.A.; Gonçalves, J.E.; De Rezende, C.; Colauto, N.B.; Gazim, Z.; Linde, G. Antioxidant activity and chemical composition of oleoresin from leaves and flowers of Brunfelsia uniflora. Genet. Mol. Res. 2017, 16. [Google Scholar] [CrossRef]
- Tian, M.; Liu, T.; Wu, X.; Hong, Y.; Liu, X.; Lin, B.; Zhou, Y. Chemical composition, antioxidant, antimicrobial and anticancer activities of the essential oil from the rhizomes of Zingiber striolatum Diels. Nat. Prod. Res. 2019, 25, 1–5. [Google Scholar] [CrossRef] [PubMed]
- Demiray, H.; Tabanca, N.; Estep, A.S.; Becnel, J.J.; Demirci, B. Chemical composition of the essential oil and n-hexane extract of Stachys tmolea subsp. Tmolea Boiss., an endemic species of Turkey, and their mosquitocidal activity against dengue vector Aesdes aegypti. Saudi Pharm. J. 2019, 27, 877–881. [Google Scholar] [CrossRef] [PubMed]
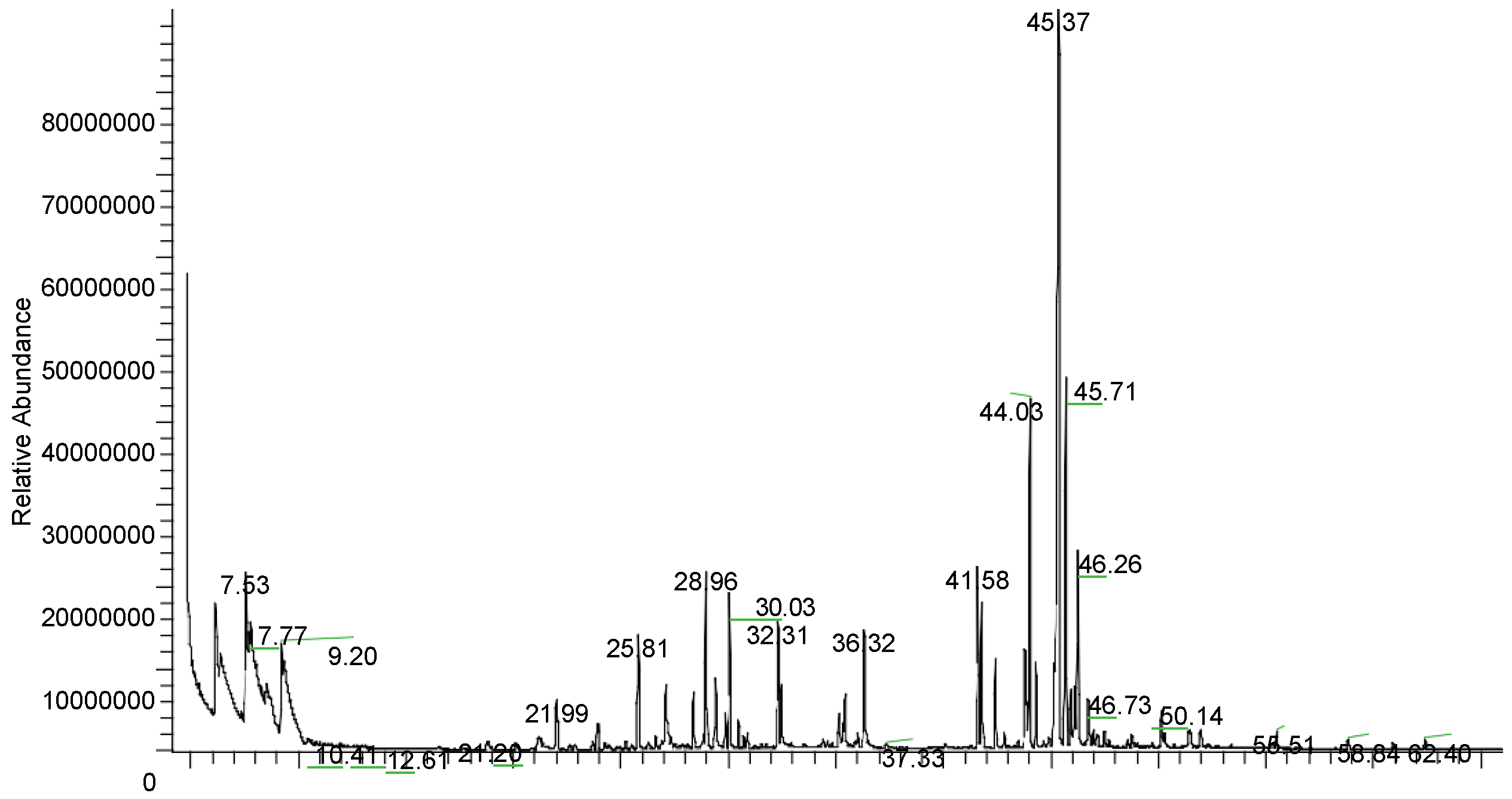
| RT (min) | Component Name | MS Fragments | Area (%) |
|---|---|---|---|
| 6.11 | α-Pinene | 136 (MW), 121, 105, 93, 92, 91, 77, 41, 39, 27 | 2.13 |
| 7.53 | β-Pinene | 136 (MW), 121, 93, 91, 79, 77, 69, 41, 39, 27 | 2.31 |
| 9.20 | 1,3,8-p-Menthatriene | 134 (MW), 119, 91 | 2.67 |
| 18.78 | 1,1,6-Trimethyl-tetralin | 174 (MW), 159, 131, 115 | 0.43 |
| 21.20 | Carvacrol | 150 (MW), 135, 91 | 0.93 |
| 23.91 | β-Copaene | 204 (MW), 161, 119, 105, 93 | 0.72 |
| 25.81 | caryophyllene | 204 (MW), 189, 175, 161, 147, 133, 120, 105, 93, 91, 79, 69, 41 | 2.88 |
| 28.96 | γ-Elemene | 204(MW), 161, 121, 107, 93 | 5.73 |
| 29.36 | α-Bisabolene | 204 (MW), 121, 119, 109, 93 | 1.56 |
| 29.80 | (+)-δ-Cadinene | 204 (MW), 189, 161, 134, 119, 105, 81, 41 | 0.60 |
| 30.03 | Cadina-1,3,5-triene | 202 (MW), 187, 159, 144, 129, 115, 105 | 3.25 |
| 32.31 | (-)-Spathulenol | 220 (MW), 205, 187, 159, 105, 91 | 2.82 |
| 32.49 | Caryophyllene oxide | 220 (MW), 205, 177, 161, 149, 135, 121, 109, 93, 79, 43, 41 | 1.24 |
| 36.32 | α-Bisabolol | 204 (MW), 189, 161, 139, 119, 109, 93, 69, 43, 41 | 2.90 |
| 41.58 | (2E,6Ε)-Farnesyl acetate | 264 (MW), 204, 161, 138, 123, 107, 93, 69, 43, 41 | 3.59 |
| 44.03 | Geranyl-linalool | 290 (MW), 272, 203, 161, 147, 135, 119, 107, 93, 81, 69, 41 | 14.04 |
| 44.32 | (6E,10E)-7,11,15-Trimethyl-3-methylene-1,6,10,14- hexadecatetrene | 272 (MW), 148, 132, 109, 93, 69, 41 | 1.54 |
| 45.37 | Geranyl-p-cymene | 242 (MW), 134, 119, 91 | 25.08 |
| 45.71 | Geranyl-γ-terpinene | 272 (MW), 136, 121, 93, 91, 77 | 15.17 |
| Microbial Species | S. raeseri subsp. raeseri EO | Ciproxin (Data Reproduced by Fitsiou et al. (2016) [27] | Gentamycin (Data Reproduced by Mitropoulou et al. (2017) [18] | Voriconazole | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| MIC * | NIC * | MLC ** | MIC * | NIC * | MLC ** | MIC * | NIC * | MLC ** | MIC * | NIC * | MLC ** | |
| Staphylococcus aureus | 7116 ± 26 | 5887 ± 35 | 31630 | 0.982 ± 0.002 | 0.963 ± 0.003 | 4 | 3.332 ± 0.003 | 3.021 ± 0.001 | 16 | - | - | - |
| Staphylococcus epidermidis | 7732 ± 35 | 5535 ± 35 | 30751 | 0.979 + 0.002 | 0.957 + 0.002 | 4 | 3.421 ± 0.001 | 3.127 ± 0.001 | 16 | - | - | - |
| Escherichia coli | 6414 ± 26 | 5974 ± 26 | 26358 | 0.984 + 0.001 | 0.956 ± 0.002 | 4 | 3.952 ± 0.001 | 3.253 ± 0.002 | 16 | - | - | - |
| Listeria monocytogenes | 6853 ± 35 | 5799 ± 44 | 28115 | 0.979 ± 0.001 | 0.968 + 0.001 | 4 | 3.121 ± 0.002 | 3.001 ± 0.002 | 16 | - | - | - |
| Salmonella Enteritidis | 6326 ± 44 | 5799 ± 26 | 26358 | 0.976 ± 0.001 | 0.957 ± 0.001 | 8 | 4.942 ± 0.001 | 4.011 ± 0.001 | 18 | - | - | - |
| Salmonella Typhimurium | 5974 ± 26 | 5447 ± 53 | 26358 | 0.979 ± 0.001 | 0.964 ± 0.001 | 8 | 4.211 ± 0.002 | 4.026 ± 0.001 | 18 | - | - | - |
| Pseudomonas fragi | 5184 ± 35 | 4305 ± 26 | 21965 | 0.955 ± 0.001 | 0.940 ± 0.002 | 8 | 4.134 ± 0.002 | 4.009 ± 0.002 | 18 | - | - | - |
| S. cerevisiae | 7029 | - | 28115 | - | - | - | - | - | - | 0.25 | - | 1.00 |
| A. niger | 8786 | - | 35144 | - | - | - | - | - | - | 0.50 | - | 2.00 |
| IC50 (mg/mL) * | ||
|---|---|---|
| DPPH Assay | ABTS Assay | |
| S. raesaeri | 24.77 ± 4.21 | 1.27 ± 0.59 |
| Ascorbic acid ** | 0.012 ± 0.004 | 0.0045 ± 0.0002 |
| Cell Line | EC50 (mg/mL) * |
|---|---|
| A375 | 0.151 ± 0.008 |
| HaCaT | 0.114 ± 0.015 |
| Caco2 | 0.175 ± 0.080 |
| PC3 | 0.216 ± 0.090 |
| DU145 | 0.188 ± 0.060 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mitropoulou, G.; Sidira, M.; Skitsa, M.; Tsochantaridis, I.; Pappa, A.; Dimtsoudis, C.; Proestos, C.; Kourkoutas, Y. Assessment of the Antimicrobial, Antioxidant, and Antiproliferative Potential of Sideritis raeseri subps. raeseri Essential Oil. Foods 2020, 9, 860. https://doi.org/10.3390/foods9070860
Mitropoulou G, Sidira M, Skitsa M, Tsochantaridis I, Pappa A, Dimtsoudis C, Proestos C, Kourkoutas Y. Assessment of the Antimicrobial, Antioxidant, and Antiproliferative Potential of Sideritis raeseri subps. raeseri Essential Oil. Foods. 2020; 9(7):860. https://doi.org/10.3390/foods9070860
Chicago/Turabian StyleMitropoulou, Gregoria, Marianthi Sidira, Myria Skitsa, Ilias Tsochantaridis, Aglaia Pappa, Christos Dimtsoudis, Charalampos Proestos, and Yiannis Kourkoutas. 2020. "Assessment of the Antimicrobial, Antioxidant, and Antiproliferative Potential of Sideritis raeseri subps. raeseri Essential Oil" Foods 9, no. 7: 860. https://doi.org/10.3390/foods9070860
APA StyleMitropoulou, G., Sidira, M., Skitsa, M., Tsochantaridis, I., Pappa, A., Dimtsoudis, C., Proestos, C., & Kourkoutas, Y. (2020). Assessment of the Antimicrobial, Antioxidant, and Antiproliferative Potential of Sideritis raeseri subps. raeseri Essential Oil. Foods, 9(7), 860. https://doi.org/10.3390/foods9070860











