Phytochemical Profile and Biological Properties of Colchicum triphyllum (Meadow Saffron)
Abstract
1. Introduction
2. Materials and Methods
2.1. Plant Material
2.2. Extraction Methods
2.3. UHPLC-QTOF Profiling of Polyphenols and Alkaloids
2.4. In Vitro Antioxidant Capacity and Inhibitory Potential
2.5. Statistical Analysis and Chemometrics
3. Results and Discussion
3.1. Phytochemical Profiling of the Different Extracts
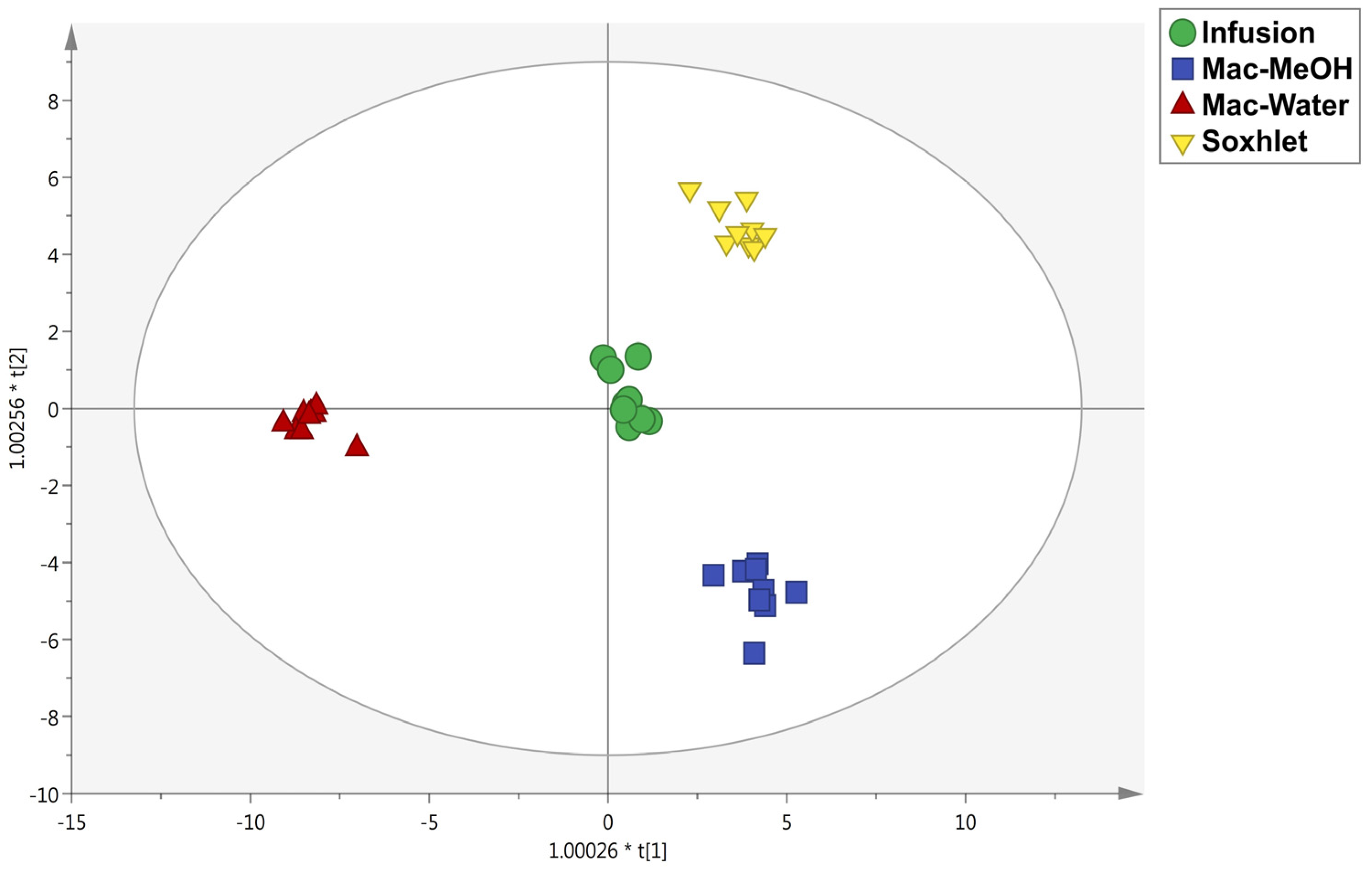
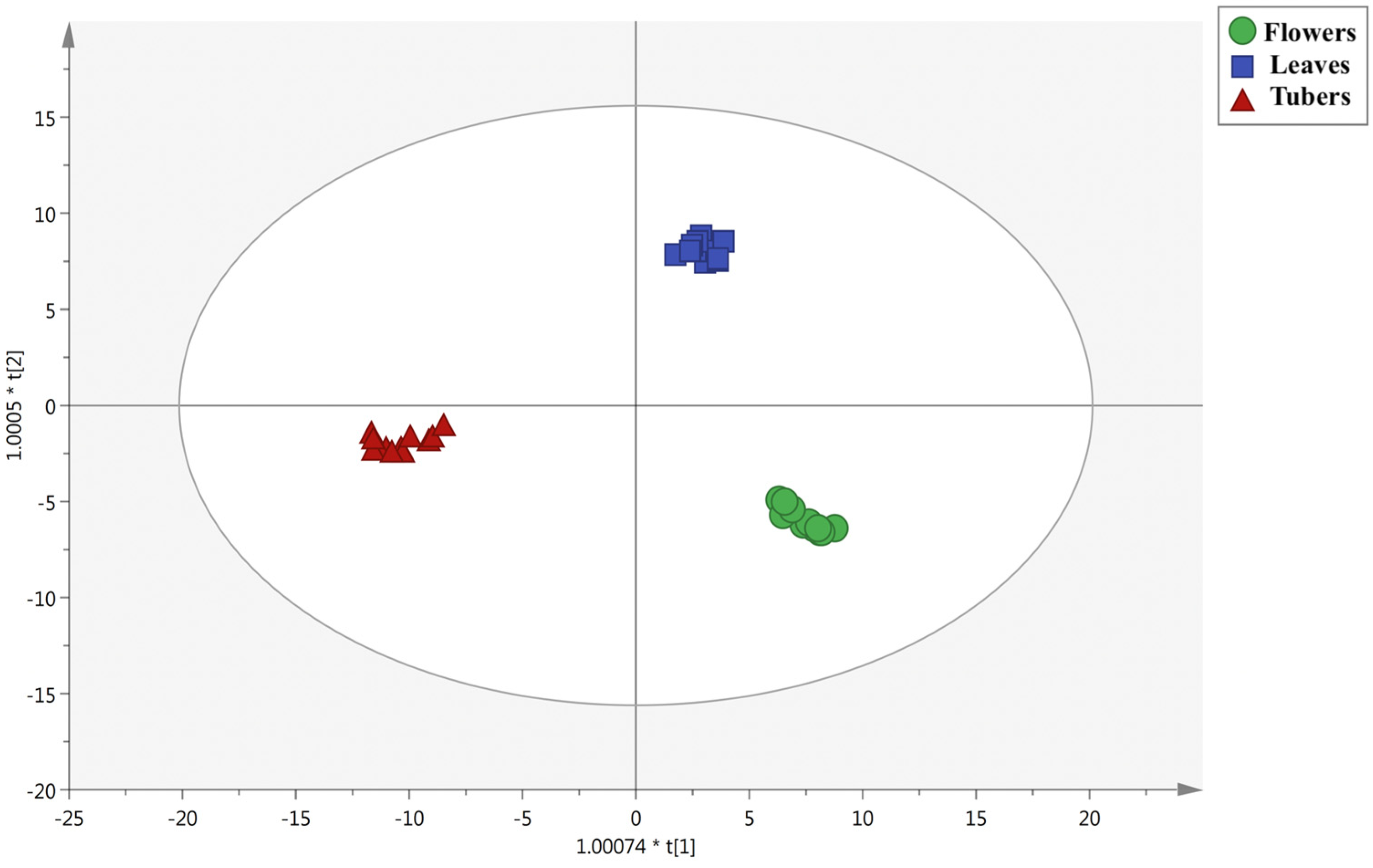
3.2. Multivariate Statistical Discrimination of the Different Extraction Methods
3.3. In Vitro Antioxidant Capacity of the Tested Extracts
3.4. Enzyme Inhibitory Activity of the Tested Extracts
3.5. Correlations
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Toplan, G.G.; Gurer, C.; Mat, A. Importance of Colchicum species in modern therapy and its significance in Turkey. J. Fac. Pharm. 2016, 46, 129–144. [Google Scholar]
- Alali, F.Q.; Tawaha, K.; El-Elimat, T. Determination of (–)-demecolcine and (–)-colchicine content in selected Jordanian Colchicum species. Int. J. Pharm. Sci. Res. 2007, 62, 739–742. [Google Scholar]
- Terkeltaub, R.A.; Furst, D.E.; Bennett, K.; Kook, K.A.; Crockett, R.S.; Davis, M.W. High versus low dosing of oral colchicine for early acute gout flare: Twenty-four-hour outcome of the first multicenter, randomized, double-blind, placebo-controlled, parallel-group, dose-comparison colchicine study. Arthritis Rheum. 2010, 62, 1060–1068. [Google Scholar] [CrossRef] [PubMed]
- Sakane, T.; Takeno, M. Novel approaches to Behcet’s disease. Expert Opin. Investig. Drugs 2000, 9, 1993–2005. [Google Scholar] [CrossRef]
- Cifuentes, M.; Schilling, B.; Ravindra, R.; Winter, J.; Janik, M.E. Synthesis and biological evaluation of B-ring modi ed colchicine and iso colchicine analogs. Bioorg. Med. Chem. Lett. 2006, 16, 2761–2764. [Google Scholar] [CrossRef]
- Cocco, G.; Chu, D.C.; Pandolfi, S. Colchicine in Clinical Medicine. A Guide for Internists. Eur. J. Intern. Med. 2010, 21, 503–508. [Google Scholar] [CrossRef]
- Ondra, P.; Válka, I.; Vičar, J.; Sütlüpinar, N.; Šimánek, V. Chromatographic determination of constituents of the genus Colchicum (Liliaceae). J. Chromatogr. A 1995, 704, 351–356. [Google Scholar] [CrossRef]
- Brvar, M.; Ploj, T.; Kozelj, G.; Mozina, M.; Noc, M.; Bunc, M. Case report: Fatal poisoning with Colchicum autumnale. Crit. Care 2004, 8, R56–R59. [Google Scholar] [CrossRef]
- Rocchetti, G.; Senizza, B.; Zengin, G.; Okur, M.A.; Montesano, D.; Yildiztugay, E.; Lobine, D.; Mahomoodally, M.F.; Lucini, L. Chemical Profiling and Biological Properties of Extracts from Different Parts of Colchicum Szovitsii Subsp. Szovitsii. Antioxidants 2019, 8, 632. [Google Scholar] [CrossRef]
- Senizza, B.; Rocchetti, G.; Ghisoni, S.; Busconi, M.; De Los Mozos Pascual, J.; Fernandez, A.; Lucini, L.; Trevisan, M. Identification of phenolic markers for saffron authenticity and origin: An untargeted metabolomics approach. Food Res. Int. 2019, 126, 108584. [Google Scholar] [CrossRef]
- Mohamed, M.B.; Rocchetti, G.; Montesano, D.; Ali, S.B.; Guasmi, F.; Grati-Kamoun, N.; Lucini, L. Discrimination of Tunisian and Italian extra-virgin olive oils according to their phenolic and sterolic fingerprints. Food Res. Int. 2018, 106, 920–927. [Google Scholar] [CrossRef]
- Fellah, B.; Rocchetti, G.; Senizza, B.; Giuberti, G.; Bannour, M.; Ferchichi, A.; Lucini, L. Untargeted metabolomics reveals changes in phenolic profile following in vitro large intestine fermentation of non-edible parts of Punica granatum L. Food Res. Int. 2020, 128, 108807. [Google Scholar] [CrossRef] [PubMed]
- Salek, R.M.; Neumann, S.; Schober, D.; Hummel, J.; Billiau, K.; Kopka, J.; Correa, E.; Reijmers, T.; Rosato, A.; Tenori, L.; et al. COordination of Standards in MetabOlomicS (COSMOS): Facilitating integrated metabolomics data access. Metabolomics 2015, 11, 1587–1597. [Google Scholar] [CrossRef]
- Schrimpe-Rutledge, A.C.; Codreanu, S.G.; Sherrod, S.D.; McLean, J.A. Untargeted metabolomics strategies-Challenges and emerging directions. J. Am. Soc. Mass Spectrom. 2016, 27, 1897–1905. [Google Scholar] [CrossRef]
- Zengin, G. A study on in vitro enzyme inhibitory properties of Asphodeline anatolica: New sources of natural inhibitors for public health problems. Ind. Crops Prod. 2016, 83, 39–43. [Google Scholar] [CrossRef]
- Tufa, T.; Damianakos, H.; Zengin, G.; Graikou, K.; Chinou, I. Antioxidant and enzyme inhibitory activities of disodium rabdosiin isolated from Alkanna sfikasiana Tan, Vold and Strid. S. Afr. J. Bot. 2019, 120, 157–162. [Google Scholar] [CrossRef]
- Mykhailenko, O.; Kovalyov, V.; Goryacha, O.; Ivanauskas, L.; Georgiyants, V. Biologically active compounds and pharmacological activities of species of the genus Crocus: A review. Phytochemistry 2019, 162, 56–89. [Google Scholar] [CrossRef]
- Moratalla-López, N.; Bagur, M.J.; Lorenzo, C.; Martínez-Navarro, M.E.; Salinas, M.R.; Alonso, G.L. Bioactivity and Bioavailability of the Major Metabolites of Crocus sativus L. Flower. Molecules 2019, 24, 2827. [Google Scholar] [CrossRef] [PubMed]
- Amin Mir, M.; Parihar, K.; Tabasum, U.; Kumari, E. Estimation of alkaloid, saponin and flavonoid, content in various extracts of Crocus sativa. J. Med. Plant Res. 2016, 4, 171–174. [Google Scholar]
- Hosseinzadeh, H.; Younesi, H.M. Antinociceptive and anti-inflammatory effects of Crocus sativus L. stigma and petal extracts in mice. BMC Pharmacol. 2002, 2, 7. [Google Scholar]
- Jadouali, S.M.; Atifi, H.; Mamouni, R.; Majourhat, K.; Bouzoubaâ, Z.; Laknifli, A.; Faouzi, A. Chemical characterization and antioxidant compounds of flower parts of Moroccan crocus sativus L. J. Saudi Soc. Agric. Sci. 2019, 18, 476–480. [Google Scholar] [CrossRef]
- Suica-bunghez, I.; Ion, R.; Teodorescu, I.; Sorescu, A.; Stirbescu, R.; Stirbescu, N. Fitochemical and antioxidant characterization of autumn crocus (Colchicum autumnale) flowers and tubers plant extracts. J. Sci. Arts 2017, 3, 539–546. [Google Scholar]
- Lapornik, B.; Prošek, M.; Golc Wondra, A. Comparison of extracts prepared from plant by-products using different solvents and extraction time. J. Food Eng. 2005, 71, 214–222. [Google Scholar] [CrossRef]
- Larsson, S.; Rønsted, N. Reviewing Colchicaceae Alkaloids – Perspectives of Evolution on Medicinal Chemistry. Curr. Top. Med. Chem. 2014, 14, 274–289. [Google Scholar] [CrossRef]
- Borrelli, F.; Borbone, N.; Capasso, R.; Montesano, D.; De Marino, S.; Aviello, G.; Aprea, G.; Masone, S.; Izzo, A.A. Potent relaxant effect of a Celastrus paniculatus extract in the rat and human ileum. J. Ethnopharmacol. 2009, 122, 434–438. [Google Scholar] [CrossRef] [PubMed]
- Montesano, D.; Rocchetti, G.; Putnik, P.; Lucini, L. Bioactive profile of pumpkin: An overview on terpenoids and their health-promoting properties. Curr. Opin. Food Sci. 2018, 22, 81–87. [Google Scholar] [CrossRef]
- Rocchetti, G.; Pellizzoni, M.; Montesano, D.; Lucini, L. Italian Opuntia ficus-indica cladodes as rich source of bioactive compounds with health-promoting properties. Foods 2018, 7, 24. [Google Scholar] [CrossRef]
- Pandey, K.B.; Rizvi, S.I. Plant polyphenols as dietary antioxidants in human health and disease. Oxid. Med. Cell. Logev. 2009, 2, 897484. [Google Scholar] [CrossRef]
- Saucedo-Pompa, S.; Torres-Castillo, J.A.; Castro-López, C.; Rojas, R.; Sánchez-Alejo, E.J.; Ngangyo-Heya, M.; Martínez-Ávila, G.C.G. Moringa plants: Bioactive compounds and promising applications in food products. Food Res. Int. 2018, 111, 438–450. [Google Scholar] [CrossRef]
- de Morais Cardoso, L.; Pinheiro, S.S.; Martino, H.S.D.; Pinheiro-Sant’Ana, H.M. Sorghum (Sorghum bicolor L.): Nutrients, bioactive compounds, and potential impact on human health. Crit. Rev. Food Sci. Nutr. 2017, 57, 372–390. [Google Scholar] [CrossRef]
- Borbone, N.; Borrelli, F.; Montesano, D.; Izzo, A.A.; De Marino, S.; Capasso, R.; Zollo, F. Identification of a new sesquiterpene polyol ester from Celastrus paniculatus. Planta Med. 2007, 73, 792–794. [Google Scholar] [CrossRef]
- Granato, D.; Shahidi, F.; Wrolstad, R.; Kilmartin, P.; Melton, L.D.; Hidalgo, F.J.; Miyashita, K.; Camp, J.V.; Alasalvar, C.; Ismail, A.B.; et al. Antioxidant activity, total phenolics and flavonoids contents: Should we ban in vitro screening methods? Food Chem. 2018, 264, 471–475. [Google Scholar] [CrossRef]
- Ahmadinejad, F.; Geir Møller, S.; Hashemzadeh-Chaleshtori, M.; Bidkhori, G.; Jami, M.-S. Molecular mechanisms behind free radical scavengers function against oxidative stress. Antioxidants 2017, 6, 51. [Google Scholar] [CrossRef]
- Silman, I.; Sussman, J.L. Acetylcholinesterase: ‘classical’ and ‘non-classical’ functions and pharmacology. Curr. Opin. Pharmacol. 2005, 5, 293–302. [Google Scholar] [CrossRef]
- Zolghadri, S.; Bahrami, A.; Hassan Khan, M.T.; Munoz-Munoz, J.; Garcia-Molina, F.; Garcia-Canovas, F.; Saboury, A.A. A comprehensive review on tyrosinase inhibitors. J. Enzym. Inhib. Med. Chem. 2019, 34, 279–309. [Google Scholar] [CrossRef] [PubMed]
- Geromichalos, G.D.; Lamari, F.N.; Papandreou, M.A.; Trafalis, D.T.; Margarity, M.; Papageorgiou, A.; Sinakos, Z. Saffron as a source of novel acetylcholinesterase inhibitors: Molecular docking and in vitro enzymatic studies. J. Agric. Food Chem. 2012, 60, 6131–6138. [Google Scholar] [CrossRef] [PubMed]
- Menghini, L.; Leporini, L.; Vecchiotti, G.; Locatelli, M.; Carradori, S.; Ferrante, C.; Zengin, G.; Recinella, L.; Chiavaroli, A.; Leone, S.; et al. Crocus sativus L. stigmas and byproducts: Qualitative fingerprint, antioxidant potentials and enzyme inhibitory activities. Food Res. Int. 2018, 109, 91–98. [Google Scholar] [CrossRef] [PubMed]
- Rocchetti, G.; Giuberti, G.; Busconi, M.; Marocco, A.; Trevisan, M.; Lucini, L. Pigmented sorghum polyphenols as potential inhibitors of starch digestibility: An in vitro study combining starch digestion and untargeted metabolomics. Food Chem. 2020, 312, 126077. [Google Scholar] [CrossRef]
- Rocchetti, G.; Giuberti, G.; Gallo, A.; Bernardi, J.; Marocco, A.; Lucini, L. Effect of dietary polyphenols on the in vitro starch digestibility of pigmented maize varieties under cooking conditions. Food Res. Int. 2018, 108, 183–191. [Google Scholar] [CrossRef] [PubMed]
- Tan, Y.; Chang, S.K.C. Digestive enzyme inhibition activity of the phenolic substances in selected fruits, vegetables and tea as compared to black legumes. J. Funct. Foods 2017, 38, 644–655. [Google Scholar] [CrossRef]
- Martinez-Gonzalez, A.I.; Díaz-Sánchez, Á.G.; de la Rosa, L.A.; Bustos-Jaimes, I.; Alvarez-Parrilla, E. Inhibition of α-amylase by flavonoids: Structure activity relationship (SAR). Spectrochim. Acta A 2019, 206, 437–447. [Google Scholar] [CrossRef] [PubMed]
- Xiao, J.; Kai, G.; Yamamoto, K.; Chen, X. Advance in Dietary Polyphenols as α-Glucosidases Inhibitors: A Review on Structure-Activity Relationship Aspect. Crit. Rev. Food Sci. Nutr. 2013, 53, 818–836. [Google Scholar] [CrossRef] [PubMed]
- Takahama, U.; Hirota, S. Interactions of flavonoids with α-amylase and starch slowing down its digestion. Food Funct. 2018, 9, 677–687. [Google Scholar] [CrossRef] [PubMed]
- Mocan, A.; Zengin, G.; Crisan, G.; Mollica, A. Enzymatic assays and molecular modeling studies of Schisandra chinensis lignans and phenolics from fruit and leaf extracts. J. Enzym. Inhib. Med. Chem. 2016, 31, 200–210. [Google Scholar] [CrossRef] [PubMed]
| Parts | Methods | Extraction Yield (%) | Total Alkaloids (mg Eq./g) | Anthocyanins (mg Eq./g) | Flavones (mg Eq./g) | Flavonols (mg Eq./g) | Phenolic Acids (mg Eq./g) | Lignans (mg Eq./g) | Tyrosols (mg Eq./g) |
|---|---|---|---|---|---|---|---|---|---|
| Flowers | Infusion | 35.12 | 1.49 ± 0.49 ab | 0.57 ± 0.05 d | 1.98 ± 0.2 c | 1.48 ± 0.21 c | 0.11 ± 0.02 a | 2.27 ± 0.29 ab | 0.72 ± 0.35 ab |
| Maceration-MeOH | 42.6 | 1.77 ± 0.44 abc | 0.78 ± 0.01 f | 1.99 ± 0.94 c | 1.71 ± 0.20 d | 0.20 ± 0.00 c | 2.67 ± 1.36 abc | 1.09 ± 0.24 bc | |
| Maceration-Water | 26.65 | 2.08 ± 0.02 bcd | 0.06 ± 0.00 b | 0.47 ± 0.08 a | 0.14 ± 0.00 a | 0.19 ± 0.01 bc | 3.01 ± 0.28 abc | 0.44 ± 0.03 a | |
| Soxhlet-MeOH | 51.25 | 1.39 ± 0.01 ab | 0.67 ± 0.01 e | 2.52 ± 0.14 c | 1.67 ± 0.01 d | 0.15 ± 0.03 abc | 1.28 ± 1.30 a | 0.42 ± 0.01 a | |
| Average value | 1.68 | 2.08 | 1.74 | 1.25 | 0.16 | 2.31 | 0.67 | ||
| Tubers | Infusion | 13.44 | 1.86 ± 1.00 abc | 0.005 ± 0.00 a | 0.15 ± 0.00 a | nd a | 0.50 ± 0.02 g | 4.57 ± 0.03 bc | 1.22 ± 0.05 cd |
| Maceration-MeOH | 28.41 | 3.19 ± 0.03 cd | nd a | 1.06 ± 0.39 b | nd a | 0.45 ± 0.05 fg | 10.37 ± 0.72 d | 1.84 ± 0.05 e | |
| Maceration-Water | 44.02 | 3.46 ± 0.74 d | nd a | 0.39 ± 0.24 a | nd a | 0.41 ± 0.01 e | 4.84 ± 1.41 bc | 1.60 ± 0.01 de | |
| Soxhlet-MeOH | 12.68 | 3.08 ± 1.14 cd | nd a | 0.45 ± 0.02d a | nd a | 0.35 ± 0.10 de | 10.60 ± 2.50 d | 2.85 ± 0.72 f | |
| Average value | 2.89 | <0.01 | 0.51 | nd | 0.43 | 7.59 | 1.88 | ||
| Leaves | Infusion | 25.58 | 2.26 ± 0.49 abcd | 0.06 ± 0.00 b | 0.33 ± 0.01 a | 0.07 ± 0.01 a | 0.21 ± 0.01 c | 4.77 ± 1.25 d | 3.43 ± 0.07 g |
| Maceration-MeOH | 31.48 | 1.10 ± 0.54 a | 0.05 ± 0.01 b | 0.22 ± 0.04 a | 0.11 ± 0.00 a | 0.30 ± 0.04 d | 1.65 ± 0.57 a | 0.59 ± 0.09 a | |
| Maceration-Water | 22.88 | 2.28 ± 0.04 abcd | 0.13 ± 0.00 c | 1.10 ± 0.01 b | 0.43 ± 0.00 b | 0.49 ± 0.06 fg | 4.99 ± 2.75 c | 2.48 ± 0.01 f | |
| Soxhlet-MeOH | 33.61 | 2.79 ± 1.70 bcd | 0.07 ± 0.01 b | 0.56 ± 0.00 ab | 0.09 ± 0.01 a | 0.12 ± 0.00 ab | 3.11 ± 0.15 abc | 1.45 ± 0.04 cde | |
| Average value | 2.11 | 0.08 | 0.55 | 0.17 | 0.28 | 3.63 | 1.87 |
| Parts | Methods | Phosphomolybdenum (mmol TE/g) | DPPH (mg TE/g) | ABTS (mg TE/g) | CUPRAC (mg TE/g) | FRAP (mg TE/g) | Metal Chelating (mg EDTAE/g) |
|---|---|---|---|---|---|---|---|
| Flowers | Infusion | 0.51 ± 0.04 b | 31.69 ± 0.45 c | 27.47 ± 1.57 c | 35.00 ± 0.60 a | 25.02 ± 0.34 a | 20.90 ± 3.06 e |
| Maceration-MeOH | 0.46 ± 0.07 ab | 33.99 ± 0.79 cd | 20.53 ± 1.91 a | 41.52 ± 1.13 c | 25.71 ± 1.78 a | 14.40 ± 0.55 c | |
| Maceration-water | 1.10 ± 0.15 d | 24.34 ± 0.60 b | 19.26 ± 0.64 a | 37.00 ± 0.22 ab | 38.88 ± 0.67 d | 21.53 ± 1.41 ef | |
| Soxhlet-MeOH | 0.74 ± 0.09 c | 34.63 ± 0.27 cd | 24.48 ± 1.15 b | 58.31 ± 0.89 f | 30.72 ± 0.64 b | 16.04 ± 0.41 cd | |
| Average value | 0.70 | 31.16 | 22.93 | 42.96 | 30.08 | 18.22 | |
| Tubers | Infusion | 0.33 ± 0.02 a | 12.14 ± 10.18 a | 30.38 ± 0.38 d | 38.38 ± 0.63 b | 28.92 ± 0.74 b | 33.88 ± 0.39 g |
| Maceration-MeOH | 0.72 ± 0.02 c | 46.17 ± 1.15 f | 50.93 ± 0.90 h | 63.12 ± 1.52 g | 46.16 ± 0.52 e | 7.48 ± 0.27 b | |
| Maceration-water | 0.77 ± 0.05 c | 29.12 ± 1.05 bc | 45.27 ± 0.60 f | 44.75 ± 0.32 d | 35.34 ± 0.71 c | 23.99 ± 1.14 f | |
| Soxhlet-MeOH | 0.85 ± 0.07 c | 38.19 ± 0.91 de | 38.79 ± 0.31 e | 50.65 ± 2.06 e | 35.85 ± 0.06 c | 1.89 ± 0.25 a | |
| Average value | 0.67 | 31.40 | 41.34 | 49.22 | 36.57 | 16.81 | |
| Leaves | Infusion | 1.25 ± 0.09 e | 44.91 ± 0.35 f | 47.90 ± 0.62 g | 68.13 ± 0.45 g | 60.66 ± 0.66 f | 32.89 ± 2.20 g |
| Maceration-MeOH | 1.38 ± 0.05 e | 45.49 ± 0.74 f | 52.55 ± 1.65 h | 109.63 ± 2.06 l | 70.04 ± 3.09 h | 22.26 ± 0.45 ef | |
| Maceration-water | 1.52 ± 0.02 f | 42.85 ± 1.09 ef | 43.95 ± 1.34 f | 84.71 ± 1.74 i | 65.92 ± 1.39 g | 15.74 ± 2.33 cd | |
| Soxhlet-MeOH | 1.35 ± 0.10 e | 47.65 ± 0.38 f | 55.73 ± 0.36 i | 123.34 ± 2.24 m | 70.80 ± 0.88 h | 18.25 ± 0.29 d | |
| Average value | 1.37 | 45.22 | 50.03 | 96.45 | 66.67 | 22.28 |
| Parts | Methods | AChE Inhibition (mg GALAE/g) | BChE Inhibition (mg GALAE/g) | Tyrosinase Inhibition (mg KAE/g) | α-amylase Inhibition (mmol ACAE/g) | α-glucosidase Inhibition (mmol ACAE/g) |
|---|---|---|---|---|---|---|
| Flowers | Infusion | nd a | nd a | nd a | 0.14 ± 0.01 ab | nd a |
| Maceration-MeOH | 3.41 ± 0.12 d | nd a | 104.09 ± 3.66 c | 0.66 ± 0.01 ef | 2.69 ± 0.02 b | |
| Maceration-Water | nd a | nd a | nd a | 0.27 ± 0.00 c | nd a | |
| Soxhlet-MeOH | 4.09 ± 0.13 f | nd a | 102.52 ± 0.56c | 0.61 ± 0.06 d | 2.73 ± 0.01c | |
| Average value | 1.87 | nd | 51.65 | 0.42 | 1.35 | |
| Tubers | Infusion | nd a | nd a | nd a | 0.13 ± 0.00 a | nd a |
| Maceration-MeOH | 4.80 ± 0.02 h | 3.91 ± 0.30b | 118.70 ± 0.85 d | 0.73 ± 0.01 g | nd a | |
| Maceration-Water | nd a | nd a | nd a | 0.18 ± 0.01 b | nd a | |
| Soxhlet-MeOH | 5.11 ± 0.05 i | 7.42 ± 1.48 c | 118.61 ± 1.41 d | 0.64 ± 0.01 de | nd a | |
| Average value | 2.48 | 2.83 | 59.32 | 0.42 | nd | |
| Leaves | Infusion | 2.45 ± 0.16 b | nd a | nda | 0.14 ± 0.002 ab | nd a |
| Maceration-MeOH | 3.62 ± 0.16 e | nd a | 125.78 ± 0.13 f | 0.71 ± 0.05 fg | 2.75 ± 0.01 d | |
| Maceration-Water | 2.95 ± 0.08 c | nd a | 4.10 ± 0.34 b | 0.24 ± 0.00 c | nd a | |
| Soxhlet-MeOH | 4.56 ± 0.09 g | nd a | 121.78 ± 0.57 e | 0.73 ± 0.02 g | 2.76 ± 0.01 d | |
| Average value | 3.39 | nd | 62.91 | 0.45 | 1.37 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Senizza, B.; Rocchetti, G.; Okur, M.A.; Zengin, G.; Yıldıztugay, E.; Ak, G.; Montesano, D.; Lucini, L. Phytochemical Profile and Biological Properties of Colchicum triphyllum (Meadow Saffron). Foods 2020, 9, 457. https://doi.org/10.3390/foods9040457
Senizza B, Rocchetti G, Okur MA, Zengin G, Yıldıztugay E, Ak G, Montesano D, Lucini L. Phytochemical Profile and Biological Properties of Colchicum triphyllum (Meadow Saffron). Foods. 2020; 9(4):457. https://doi.org/10.3390/foods9040457
Chicago/Turabian StyleSenizza, Biancamaria, Gabriele Rocchetti, Murat Ali Okur, Gokhan Zengin, Evren Yıldıztugay, Gunes Ak, Domenico Montesano, and Luigi Lucini. 2020. "Phytochemical Profile and Biological Properties of Colchicum triphyllum (Meadow Saffron)" Foods 9, no. 4: 457. https://doi.org/10.3390/foods9040457
APA StyleSenizza, B., Rocchetti, G., Okur, M. A., Zengin, G., Yıldıztugay, E., Ak, G., Montesano, D., & Lucini, L. (2020). Phytochemical Profile and Biological Properties of Colchicum triphyllum (Meadow Saffron). Foods, 9(4), 457. https://doi.org/10.3390/foods9040457









