Mesopelagic Species and Their Potential Contribution to Food and Feed Security—A Case Study from Norway
Abstract
1. Introduction
2. Materials and Methods
2.1. Biological Material
2.2. Analytical Methods
2.3. Data Management and Presentation of Data
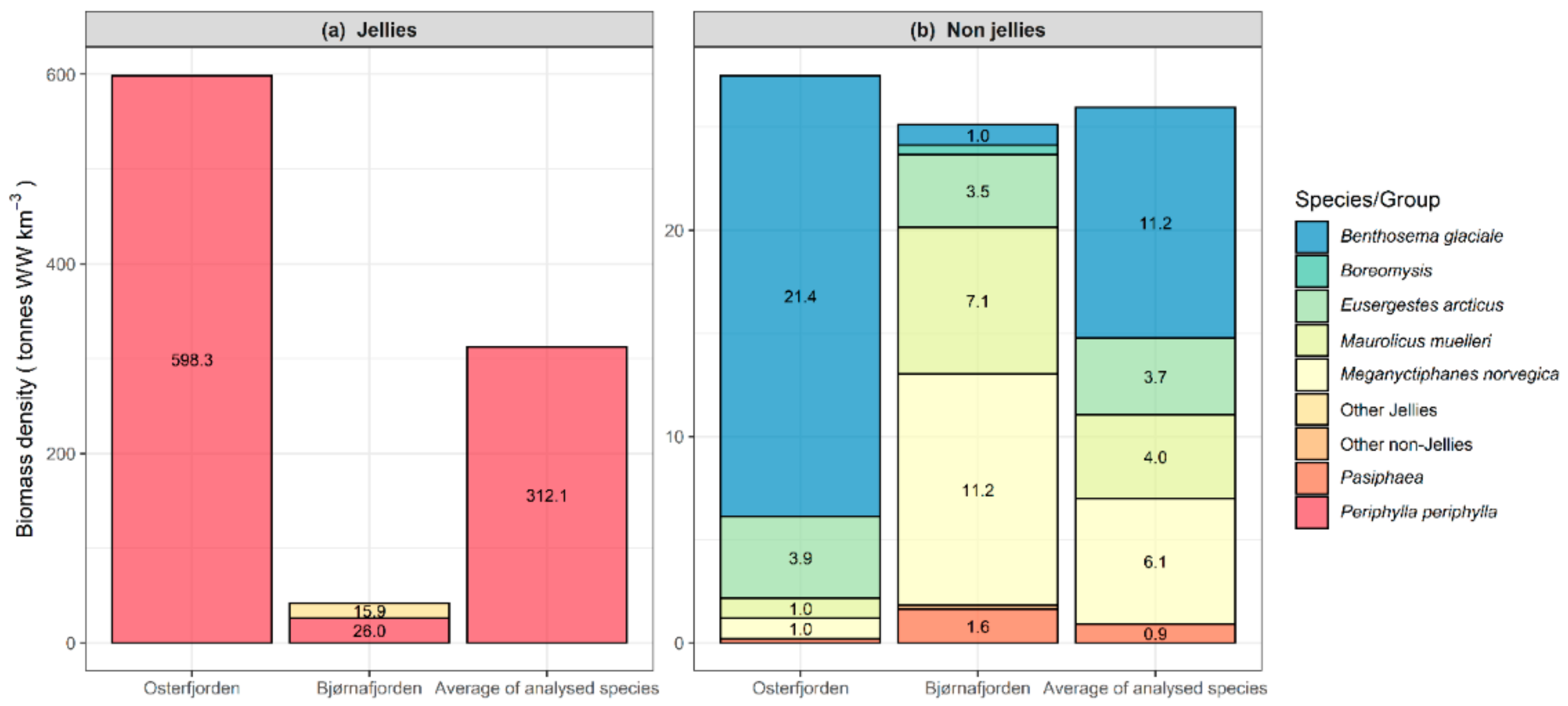
2.4. Biomass Density
Nutritional Potential
3. Results
3.1. Sample Characteristics
3.2. Nutrient Dense Mesopelagic Species
3.3. Potential Contribution to Combat Micronutrient Deficiency
3.4. Potential Contribution to Aquaculture Compared to Commercially Available Marine Feed Ingredients
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Burchi, F.; Fanzo, J.; Frison, E. The role of food and nutrition system approaches in tackling hidden hunger. Int. J. Environ. Res. Public Health 2011, 8, 358–373. [Google Scholar] [CrossRef] [PubMed]
- The Food and Agriculture Organization (FAO). The State of Food Security and Nutrition in the World; Safeguarding Against Economic Slowdowns and Downturns; FAO: Rome, Italy, 2019. [Google Scholar]
- Kawarazuka, N.; Bene, C. The potential role of small fish species in improving micronutrient deficiencies in developing countries: Building evidence. Public Health Nutr. 2011, 14, 1927–1938. [Google Scholar] [CrossRef] [PubMed]
- Ng, M.; Fleming, T.; Robinson, M.; Thomson, B.; Graetz, N.; Margono, C.; Mullany, E.C.; Biryukov, S.; Abbafati, C.; Abera, S.F.; et al. Global, regional, and national prevalence of overweight and obesity in children and adults during 1980–2013: A systematic analysis for the Global Burden of Disease Study 2013. Lancet 2014, 384, 766–781. [Google Scholar] [CrossRef]
- World Health Organization (WHO). Zero Hunger-Hunger Facts. Available online: http://www.fao.org/zhc/hunger-facts/en/ (accessed on 1 October 2019).
- Costello, C.; Cao, L.; Gelcich, S. The Future of Food from the Sea; World Resources Institute: Washington, DC, USA, 2019; Available online: https://oceanpanel.org/sites/default/files/2019-11/19_HLP_BP1%20Paper.pdf (accessed on 11 November 2019).
- Gil, A.; Gil, F. Fish, a Mediterranean source of n-3 PUFA: Benefits do not justify limiting consumption. Brit. J. Nutr. 2015, 113, S58–S67. [Google Scholar] [CrossRef]
- Lund, E.K. Health benefits of seafood; Is it just the fatty acids? Food Chem. 2013, 140, 413–420. [Google Scholar] [CrossRef]
- Roos, N.; Islam, M.M.; Thilsted, S.H. Small indigenous fish species in Bangladesh: Contribution to vitamin A, calcium and iron intakes. J. Nutr. 2003, 133, 4021–4026. [Google Scholar] [CrossRef]
- Bjorn-Rasmussen, E.; Hallberg, L. Effect of animal proteins on the absorption of food iron in man. Nutr. Metab. 1979, 23, 192–202. [Google Scholar] [CrossRef]
- Cook, J.D.; Monsen, E.R. Food iron absorption in human subjects. III. Comparison of the effect of animal proteins on nonheme iron absorption. Am. J. Clin. Nutr. 1976, 29, 859–867. [Google Scholar] [CrossRef]
- Layrisse, M.; Martinez, C.; Roche, M. Effect of Interaction of Various Foods on Iron Absorption. Am. J. Clin. Nutr. 1968, 21, 1175–1183. [Google Scholar] [CrossRef]
- Navas-Carretero, S.; Perez-Granados, A.M.; Sarria, B.; Carbajal, A.; Pedrosa, M.M.; Roe, M.A.; Fairweather-Tait, S.J.; Vaquero, M.P. Oily fish increases iron bioavailability of a phytate rich meal in young iron deficient women. J. Am. Coll. Nutr. 2008, 27, 96–101. [Google Scholar] [CrossRef]
- Pikitch, E.K.; Santora, C.; Babcock, E.A.; Bakun, A.; Bonfil, R.; Conover, D.O.; Dayton, P.; Doukakis, P.; Fluharty, D.; Heneman, B.; et al. Ecosystem-based fishery management. Science 2004, 305, 346–347. [Google Scholar] [CrossRef] [PubMed]
- Henriksson, P.; Pelletier, P.; Troell, M.; Tyedmers, P. Encyclopedia of Sustainability Science and Technology; Springer-Verlag New York Inc.: New York, NY, USA, 2012. [Google Scholar]
- Hoeg-Guldberg, O.C.K.; Chopin, T.; Gaines, S.; Haugan, P.; Hemer, M.; Howard, J.K.M.; Krause-Jensen, D.; Lindstad, E.; Lovelock, C.E.; Michelin, M.; et al. The Ocean as a Solution to Climate Change: Five Opportunities for Action; World Resources Institute: Washington, DC, USA, 2019. [Google Scholar]
- The Food and Agriculture Organization (FAO). The State of World Fisheries and Aquaculture 2018-Meeting the Sustainable Development Goals; FAO: Rome, Italy, 2018. [Google Scholar]
- The Food and Agriculture Organization (FAO). State of World Fisheries and Aquaculture 2016. Contributing to Food Security and Nutrition for all; FAO: Rome, Italy, 2016. [Google Scholar]
- EU. Food from the Oceans-How Can More Food and Biomass be Obtained form the Oceans in a Way that Does Not Deprive Future Generations of Theirs Benefits? High Level Group of Scientific Advisors: Brussels, Belgium, 2017. [Google Scholar]
- Gjøsæter, J.; Kawaguchi, K. A Review of the World Resources of Mesopelagic Fish; FAO: Rome, Italy, 1980. [Google Scholar]
- Irigoien, X.; Klevjer, T.A.; Røstad, A.; Martinez, U.; Boyra, G.; Acuna, J.L.; Bode, A.; Echevarria, F.; Gonzalez-Gordillo, J.I.; Hernandez-Leon, S.; et al. Large mesopelagic fishes biomass and trophic efficiency in the open ocean. Nat. Commun. 2014, 5. [Google Scholar] [CrossRef] [PubMed]
- Kaartvedt, S.; Staby, A.; Aksnes, D.L. Efficient trawl avoidance by mesopelagic fishes causes large underestimation of their biomass. Mar. Ecol. Prog. Ser. 2012, 456, 1–6. [Google Scholar] [CrossRef]
- Everson, I. Krill: Biology, Ecology and Fisheries; Wiley-Blackwell: Hoboken, NJ, USA, 2008. [Google Scholar]
- Vereshchaka, A.L.; Olesen, J.; Lunina, A.A. Global diversity and phylogeny of pelagic shrimps of the former genera Sergestes and Sergia (Crustacea, Dendrobranchiata, Sergestidae), with definition of eight new genera. PLoS ONE 2014, 9, e112057. [Google Scholar] [CrossRef] [PubMed]
- Heino, M.; Porteiro, F.M.; Sutton, T.T.; Falkenhaug, T.; Godø, O.R.; Piatkowsk, U. Catchability of pelagic trawls for sampling deep-living nekton in the mid-North Atlantic. ICES J. Mar. Sci. 2011, 68, 377–389. [Google Scholar] [CrossRef]
- Krafft, B.A.; Melle, W.; Knutsen, T.; Bagøien, E.; Broms, C.; Ellertsen, B.; Siegel, V. Distribution and demography of Antarctic krill in the Southeast Atlantic sector of the Southern Ocean during the austral summer 2008. Polar. Biol. 2010, 33, 957–968. [Google Scholar] [CrossRef]
- Association of Official Analytical Chemists (AOAC). Crude Protein in Meat and Meat Products, Combustion Method, 16th ed.; Method 992.15; AOAC: Arlington, VA, USA, 1995. [Google Scholar]
- Wasta, Z.; Mjøs, S.A. A database of chromatographic properties and mass spectra of fatty acid methyl esters from omega-3 products. J. Chromatogr. A 2013, 1299, 94–102. [Google Scholar] [CrossRef]
- Meier, S.; Mjos, S.A.; Joensen, H.; Grahl-Nielsen, O. Validation of a one-step extraction/methylation method for determination of fatty acids and cholesterol in marine tissues. J. Chromatogr. A 2006, 1104, 291–298. [Google Scholar] [CrossRef]
- Comité Européen de Normalisation. Foodstuffs–Determination of Vitamin a by High Preformance Liquid Chromatography- Part 1: Measurement of All-Trans-Retinol and 13-Cis-Retinol; Comité Européen de Normalisation: Brussels, Belgium, 2000. [Google Scholar]
- Comité Européen de Normalisation. Foodstuffs–Determination of Vitamin D by High Performance Liquid Chromatography -Measurement of Cholecalciferol (D3) or Ergocalciferol (D2); Comité Européen de Normalisation: Brussels, Belgium, 2009. [Google Scholar]
- Julshamn, K.; Maage, A.; Norli, H.S.; Grobecker, H.K.; Jorhem, L.; Fecher, P. Determination of arsenic, cadmium, mercury, and lead by inductively coupled plasma/mass spectrometry in foods after pressure digestion: NMKL1 interlaboratory study. J. AOAC Int. 2007, 90, 844–856. [Google Scholar] [CrossRef]
- Julshamn, K.; Dahl, L.; Eckhoff, K. Determination of iodine in seafood by inductively coupled plasma/mass spectrometry. J. AOAC Int. 2001, 84, 1976–1983. [Google Scholar] [CrossRef]
- Greenfield, H.; Southgate, D. Food Composition Data-Production, Management and Use, 2nd ed.; FAO: Rome, Italy, 2003. [Google Scholar]
- Helsel, D.R. Fabricating data: How substituting values for nondetects can ruin results, and what can be done about it. Chemosphere 2006, 65, 2434–2439. [Google Scholar] [CrossRef]
- Shantz, E.M.; Brinkman, J.H. Biological Activity of Pure Vitamin-A2. J. Biol. Chem. 1950, 183, 467–471. [Google Scholar]
- Lock, E.J.; Waagbo, R.; Bonga, S.W.; Flik, G. The significance of vitamin D for fish: A review. Aquac. Nutr. 2010, 16, 100–116. [Google Scholar] [CrossRef]
- Klevjer, T.A.; Irigoien, X.; Røstad, A.; Fraile-Nuez, E.; Benítez-Barrios, V.M.; Kaartvedt, S. Large scale patterns in vertical distribution and behaviour of mesopelagic scattering layers. Sci. Rep. 2016, 6. [Google Scholar] [CrossRef] [PubMed]
- Nordic Nutrition Recommendations 2012. Nordic Nutrition Recommendations. Integrating Nutrition and Physical Activity. 2012. Available online: http://norden.diva-portal.org/smash/get/diva2:704251/FULLTEXT01.pdf (accessed on 1 October 2019).
- Nerhus, I.; Markhus, M.W.; Nilsen, B.M.; Øyen, J.; Maage, A.; Ødegård, E.R.; Midtbø, L.K.; Frantzen, S.; Kögel, T.; Graff, I.E.; et al. Iodine content of six fish species, Norwegian dairy products and hen’s egg. Food Nutr. Res. 2018, 62, 1291. [Google Scholar] [CrossRef] [PubMed]
- Måge, A.; Julshamn, K.; Espe, M.; Lunestad, B. Årsrapport 2008 og 2009. In Overvakningsprogram for Fôrvarer til Fisk og Andre Akvatiske Dyr; Norwegian Food Safety Authority: Oslo, Norway, 2010. [Google Scholar]
- Lam, V.; Pauly, D. Sea Around Us Project Newsletter. 2005. Sea Around Us. Available online: http://www.seaaroundus.org/doc/Researcher+Publications/dpauly/PDF/2005/OtherItems/MappingGlobalBiomassMesopelagicFishes.pdf (accessed on 1 October 2019).
- Nelson, J.; Grande, T.; Wilson, M. Fishes of the World; Wiley-Blackwell: Hoboken, NJ, USA, 2016. [Google Scholar]
- St John, M.A.; Borja, A.; Guillem, C.; Heath, M.; Grigorov, I.; Mariani, P.; Martin, A.P.; Santos, R.S. A Dark Hole in Our Understanding of Marine Ecosystems and Their Services: Perspectives from the Mesopelagic Community. Front. Mar. Sci. 2016, 3. [Google Scholar] [CrossRef]
- Webb, T.J.; Vanden Berghe, E.; O’Dor, R. Biodiversity’s Big Wet Secret: The Global Distribution of Marine Biological Records Reveals Chronic Under-Exploration of the Deep Pelagic Ocean. PLoS ONE 2010, 5. [Google Scholar] [CrossRef]
- Flock, M.E.; Hopkins, T.L. Species Composition, Vertical-Distribution, and Food-Habits of the Sergestid Shrimp Assemblage in the Eastern Gulf-of-Mexico. J. Crustacean Biol. 1992, 12, 210–223. [Google Scholar] [CrossRef]
- Hoving, H.J.T.; Perez, J.A.A.; Bolstad, K.S.R.; Braid, H.E.; Evans, A.B.; Fuchs, D.; Judkins, H.; Kelly, J.T.; Marian, J.E.A.R.; Nakajima, R.; et al. The Study of Deep-Sea Cephalopods. Adv. Cephalop. Sci. Biol. Ecol. Cultiv. Fish. 2014, 67, 235–359. [Google Scholar] [CrossRef]
- Kaartvedt, S. Diel Vertical Migration Behaviour of the Northern Krill (Meganyctiphanes Norvegica Sars). Adv. Mar. Biol. 2010, 57, 255–275. [Google Scholar] [CrossRef]
- Kennedy, E.; Mannar, V.; Iyengar, V. Alleviating hidden hunger. Approaches that work. IAEA Bull. 2003, 45, 54–61. [Google Scholar]
- Hallstrom, E.; Bergmana, K.; Mifflin, K.; Parker, R.; Tyedmers, P.; Troell, M.; Ziegler, F. Combined climate and nutritional performance of seafoods. J. Clean Prod. 2019, 230, 402–411. [Google Scholar] [CrossRef]
- Roos, N.; Leth, T.; Jakobsen, J.; Thilsted, S.H. High vitamin A content in some small indigenous fish species in Bangladesh: Perspectives for food-based strategies to reduce vitamin A deficiency. Int. J. Food Sci. Nutr. 2002, 53, 425–437. [Google Scholar] [CrossRef]
- Roos, N.; Wahab, M.A.; Chamnan, C.; Thilsted, S.H. The role of fish in food-based strategies to combat vitamin A and mineral deficiencies in developing countries. J. Nutr. 2007, 137, 1106–1109. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization (WHO). Micronutrient Deficiencies-Vitamin a Deficiency. Available online: https://www.who.int/nutrition/topics/vad/en/ (accessed on 1 October 2019).
- Catul, V.; Gauns, M.; Karuppasamy, P.K. A review on mesopelagic fishes belonging to family Myctophidae. Rev. Fish Biol. Fisher. 2011, 21, 339–354. [Google Scholar] [CrossRef]
- El-Mowafi, A.; Nanton, D.; Berntssen, M. Evaluation of lantern fish (Benthosema pterotum) as marine source in fish feeds: Nutrient composition and contaminants assessment. In Proceedings of the 3rd Global Fisheries Aquacult. Res. Conf. Foreign Agricultural Relations (FAR), Cairo, Egypt, 29 November–1 December 2010; Volume 29, pp. 12–23. [Google Scholar]
- Tou, J.C.; Jaczynski, J.; Chen, Y.C. Krill for human consumption: Nutritional value and potential health benefits. Nutr. Rev. 2007, 65, 63–77. [Google Scholar] [CrossRef]
- Roos, N.; Thorseng, H.; Chamnan, C.; Larsen, T.; Gondolf, U.H.; Bukhave, K.; Thilsted, S.H. Iron content in common Cambodian fish species: Perspectives for dietary iron intake in poor, rural households. Food Chem. 2007, 104, 1226–1235. [Google Scholar] [CrossRef]
- World Health Organization (WHO). Micronutrient Deficiency-Iodine Deficiency Disorder. Available online: https://www.who.int/nutrition/topics/idd/en/ (accessed on 1 October 2019).
- Brantsaeter, A.L.; Abel, M.H.; Haugen, M.; Meltzer, H.M. Risk of suboptimal iodine intake in pregnant Norwegian women. Nutrients 2013, 5, 424–440. [Google Scholar] [CrossRef]
- Dahl, L.; Markhus, M.W.; Sanchez, P.V.R.; Moe, V.; Smith, L.; Meltzer, H.M.; Kjellevold, M. Iodine Deficiency in a Study Population of Norwegian Pregnant Women-Results from the Little in Norway Study (LiN). Nutrients 2018, 10, 513. [Google Scholar] [CrossRef]
- Zimmermann, M.B.; Gizak, M.; Abbott, K.; Andersson, M.; Lazarus, J.H. Iodine deficiency in pregnant women in Europe. Lancet Diabetes Endocrinol. 2015, 3, 672–674. [Google Scholar] [CrossRef]
- Abel, M.H.; Caspersen, I.H.; Meltzer, H.M.; Haugen, M.; Brandlistuen, R.E.; Aase, H.; Alexander, J.; Torheim, L.E.; Brantsæter, A.L. Suboptimal Maternal Iodine Intake Is Associated with Impaired Child Neurodevelopment at 3 Years of Age in the Norwegian Mother and Child Cohort Study. J. Nutr. 2017, 147, 1314–1324. [Google Scholar] [CrossRef] [PubMed]
- Bath, S.C. The effect of iodine deficiency during pregnancy on child development. Proc. Nutr. Soc. 2019, 78, 150–160. [Google Scholar] [CrossRef] [PubMed]
- Markhus, M.W.; Dahl, L.; Moe, V.; Abel, M.H.; Brantsæter, A.L.; Øyen, J.; Meltzer, H.M.; Stormark, K.M.; Graff, I.E.; Smith, L.; et al. Maternal Iodine Status is Associated with Offspring Language Skills in Infancy and Toddlerhood. Nutrients 2018, 10, 1270. [Google Scholar] [CrossRef] [PubMed]
- Brantsaeter, A.L.; Knutsen, H.K.; Johansen, J.C.; Nyheim, K.A.; Erlund, I.; Meltzer, H.M.; Henjum, S. Inadequate Iodine Intake in Population Groups Defined by Age, Life Stage and Vegetarian Dietary Practice in a Norwegian Convenience Sample. Nutrients 2018, 10, 230. [Google Scholar] [CrossRef]
- Medin, A.; Carlsen, M.; Andersen, L. Iodine intake among childrenand adolescents in Norway: Estimates from the national dietary survey Ungkost 3 (2015–2016). J. Trace Elem. Med. Biol. 2020, 58, 126427. [Google Scholar] [CrossRef]
- Hansen, M.; Thilsted, S.H.; Sandström, B.; Kongsbak, K.; Larsen, T.; Jensen, M.; Sørensen, S.S. Calcium absorption from small soft-boned fish. J. Trace Elem. Med Biol. 1998, 12, 148–154. [Google Scholar] [CrossRef]
- Larsen, T.; Thilsted, S.H.; Kongsbak, K.; Hansen, M. Whole small fish as a rich calcium source. Brit. J. Nutr. 2000, 83, 191–196. [Google Scholar] [CrossRef]
- World Health Organization (WHO). Micronutrient Deficiency-Iron Deficiency Anaemia. Available online: https://www.who.int/nutrition/topics/ida/en/ (accessed on 1 October 2019).
- Francois, C.A.; Connor, S.L.; Wander, R.C.; Connor, W.E. Acute effects of dietary fatty acids on the fatty acids of human milk. Am. J. Clin. Nutr. 1998, 67, 301–308. [Google Scholar] [CrossRef]
- Lindqvist, H.; Sandberg, A.S.; Undeland, I.; Stener-Victorin, E.; Larsson, B.M.; Sannaveerappa, T.; Lonn, M.; Holmang, A. Influence of herring (Clupea harengus) and herring fractions on metabolic status in rats fed a high energy diet. Acta Physiol. 2009, 196, 303–314. [Google Scholar] [CrossRef]
- Ruyter, B.; Østbye, T.K.; Romarheim, O.H.; Nilsson, A.; Kristiansen, I.Ø.; Bjerke, M.; Bergum, S.K.; Bou, M.; Berge, G. Vil ketolinsyre (22:1n-11) fra lodde-, sild- og tobisolje føre til bedre utnyttelse av omega-3 fettsyrer? Nofima 2016, 2016, 24. [Google Scholar]
- Yang, Z.; Amar, M.; Sorokin, A.; Troendle, J.; Bourville, A.B.; Sampson, M.; Playford, M.P.; Yang, S.; Stagliane, M.; Ling, C.; et al. Supplementation with saury oil, a fish oil high in omega-11 monounsaturated fatty acids, improves plasma lipids in healthy subjects. J. Clin. Lipidol. 2019, in press. [Google Scholar] [CrossRef] [PubMed]
- Place, A.R. Comparative Aspects of Lipid Digestion and Absorption-Physiological Correlates of Wax Ester Digestion. Am. J. Physiol. 1992, 263, R464–R471. [Google Scholar] [CrossRef]
- Bogevik, A.S.; Tocher, D.R.; Langmyhr, E.; Waagbo, R.; Olsen, R.E. Atlantic salmon (Salmo salar) postsmolts adapt lipid digestion according to elevated dietary wax esters from Calanus finmarchicus. Aquac. Nutr. 2009, 15, 94–103. [Google Scholar] [CrossRef]
- Stetten, D.; Schoenheimer, R. The biological relations of the higher aliphatic aclohols to fatty acids. J. Biol. Chem. 1940, 133, 347–357. [Google Scholar]
- Pedersen, A.M.; Salma, W.; Hoper, A.C.; Larsen, T.S.; Olsen, R.L. Lipid profile of mice fed a high-fat diet supplemented with a wax ester-rich marine oil. Eur. J. Lipid Sci. Tech. 2014, 116, 1718–1726. [Google Scholar] [CrossRef]
- Biancarosa, I.; Sele, V.; Belghit, I.; Ornsrud, R.; Lock, E.J.; Amlund, H. Replacing fish meal with insect meal in the diet of Atlantic salmon (Salmo salar) does not impact the amount of contaminants in the feed and it lowers accumulation of arsenic in the fillet. Food Addit. Contam. A 2019, 36, 1191–1205. [Google Scholar] [CrossRef]
- Sarker, P.K.; Kapuscinski, A.R.; Bae, A.Y.; Donaldson, E.; Sitek, A.J.; Fitzgerald, D.S.; Edelson, O.F. Towards sustainable aquafeeds: Evaluating substitution of fishmeal with lipid-extracted microalgal co-product (Nannochloropsis oculata) in diets of juvenile Nile tilapia (Oreochromis niloticus). PLoS ONE 2018, 13. [Google Scholar] [CrossRef]
- Ytrestoyl, T.; Aas, T.S.; Asgard, T. Utilisation of feed resources in production of Atlantic salmon (Salmo salar) in Norway. Aquaculture 2015, 448, 365–374. [Google Scholar] [CrossRef]
- Shepherd, C.J.; Monroig, O.; Tocher, D.R. Future availability of raw materials for salmon feeds and supply chain implications: The case of Scottish farmed salmon. Aquaculture 2017, 467, 49–62. [Google Scholar] [CrossRef]
- Sanden, M.; Liland, N.S.; Saele, O.; Rosenlund, G.; Du, S.S.; Torstensen, B.E.; Stubhaug, I.; Ruyter, B.; Sissener, N.H. Minor lipid metabolic perturbations in the liver of Atlantic salmon (Salmo salar L.) caused by suboptimal dietary content of nutrients from fish oil. Fish Physiol. Biochem. 2016, 42, 1463–1480. [Google Scholar] [CrossRef]
- Hamre, K.; Sissener, N.H.; Lock, E.J.; Olsvik, P.A.; Espe, M.; Torstensen, B.E.; Silva, J.; Johansen, J.; Waagbo, R.; Hemre, G.I. Antioxidant nutrition in Atlantic salmon (Salmo salar) parr and post-smolt, fed diets with high inclusion of plant ingredients and graded levels of micronutrients and selected amino acids. PeerJ 2016. [Google Scholar] [CrossRef] [PubMed]
- Prabhu, P.A.J.; Lock, E.J.; Hemre, G.I.; Hamre, K.; Espe, M.; Olsvik, P.A.; Silva, J.; Hansen, A.C.; Johansen, J.; Sissener, N.H.; et al. Recommendations for dietary level of micro-minerals and vitamin D-3 to Atlantic salmon (Salmo salar) parr and post-smolt when fed low fish meal diets. PeerJ 2019, 7. [Google Scholar] [CrossRef]
- Hemre, G.I.; Lock, E.J.; Olsvik, P.A.; Hamre, K.; Espe, M.; Torstensen, B.E.; Silva, J.; Hansen, A.C.; Waagbo, R.; Johansen, J.S.; et al. Atlantic salmon (Salmo salar) require increased dietary levels of B-vitamins when fed diets with high inclusion of plant based ingredients. PeerJ 2016, 4. [Google Scholar] [CrossRef] [PubMed]
- Wiech, M.; Duinker, A.; Sanden, M. Kartlegging av Fremmedstoff i Mesopelagiske Arter Fra Norske Farvann; Institute of Marine Research: Bergen, Norway, 2018. [Google Scholar]
- Kolding, J.; van Zwieten, P.A.; Marttin, F.; Funge-Smith, S.; Poulain, F. Freshwater Small Pelagic Fish and Fisheries in the Main African Great Lakes and Reservoirs in Relation to Food Security and Nutrition; Food and Agriculture Organization of the United Nations: Rome, Italy, 2019. [Google Scholar]
- Longley, C.; Thilsted, S.H.; Beveridge, M.; Cole, S.; Nyirenda, D.B.; Heck, S.; Hother, A.-L. The Role of Fish in the First 1000 Days in Zambia; WorldFish: Penang, Malaysia, 2014; pp. 27–37. [Google Scholar]
- Golden, C.; Allison, E.H.; Cheung, W.W.L.; Dey, M.M.; Halpern, B.S.; McCauley, D.J.; Smith, M.; Vaitla, B.; Zeller, D.; Myers, S.S. Fall in fish catch threatens human health. Nature 2016, 534, 317–320. [Google Scholar] [CrossRef]
- Wood, A.; Gordon, L.J.; Röös, E.; Karlsson, J.O.; Häyhä, T.; Bignet, V.; Rydenstam, T.; Segerstad, L.H.; Bruckner, M. Nordic Food Systems for Improved Health and Sustainability. Stockholm Resilience Center. Available online: https://www.stockholmresilience.org/download/18.8620dc61698d96b1904a2/1554132043883/SRC_Report%20Nordic%20Food%20Systems.pdf (accessed on 1 October 2019).
- The Food and Agriculture Organization (FAO). Food-Based Dietary Guidelines. Available online: http://www.fao.org/nutrition/education/food-dietary-guidelines/home/en/ (accessed on 11 November 2019).
- Biancarosa, I.; Espe, M.; Bruckner, C.; Heesch, S.; Liland, N.; Waagbø, R.; Torstensen, B.; Lock, E. Amino acid composition, protein content, and nitrogen-to-protein conversion factors of 21 seaweed species from Norwegian waters. J. Appl. Phycol. 2017, 29, 1001–1009. [Google Scholar] [CrossRef]
- Mariotti, F.; Tomé, D.; Mirand, P.P. Converting nitrogen into protein-Beyond 6.25 and Jones’ factors. Crit. Rev. Food Sci. Nutr. 2008, 48, 177–184. [Google Scholar] [CrossRef]
- Lea, M.A.; Nichols, P.D.; Wilson, G. Fatty acid composition of lipid-rich myctophids and mackerel icefish (Champsocephalus gunnari)-Southern ocean food-web implications. Polar Biol. 2020, 25, 843–854. [Google Scholar] [CrossRef]
- Olsen, R.E.; Strand, E.; Melle, W.; Nørstebø, J.T.; Lalld, S.P.; Ringøe, E.; Tocher, D.R.; Sprague, M. Can mesopelagic mixed layers be used as feed sources for salmon aquaculture? Deep Sea Res. Part II Top. Stud. Oceanogr. 2020, in press. [Google Scholar] [CrossRef]

| Location | Species | Classification | Composite Samples (n) | Specimens per Composite Sample | Average Length (mm) |
|---|---|---|---|---|---|
| Osterfjorden | Benthosema glaciale | Pisces | 4 | 27 | 62.6 ± 2.3 |
| 135 | 50 ± 2.8 | ||||
| 135 | 48.8 ± 2.9 | ||||
| >50 | 23.9 ± 5.6 | ||||
| Maurolicus muelleri | Pisces | 2 | >50 | 23.6 ± 2.6 | |
| >50 | 44.5 ± 3.9 | ||||
| Meganyctiphanes norvegica | Crustacea | 2 | >50 | 17.2 ± 2.1 | |
| >50 | 33.2 ± 2.3 | ||||
| Pasiphaea sp. | Crustacea | 1 | >50 | 70.1 ± 10.6 | |
| Eusergestes arcticus | Crustacea | 2 | >50 | 32.2 ± 5.2 | |
| >50 | 26.4 ± 4.8 | ||||
| Boknafjorden | Benthosema glaciale | Pisces | 1 | >50 | 52.7 ± 6 |
| Maurolicus muelleri | Pisces | 1 | >50 | 48.5 ± 5.9 | |
| M. norvegica (Northern krill) | Crustacea | 1 | >50 | 33.4 ± 2.4 | |
| Pasiphaea sp. | Crustacea | 1 | >50 | 82 ± 8 | |
| Eusergestes arcticus | Crustacea | 1 | >50 | 50.3 ± 10.3 | |
| Bjørnafjorden | Benthosema glaciale | Pisces | 1 | 83 | 41.4 ± 10.5 |
| Maurolicus muelleri | Pisces | 1 | >50 | 36.6 ± 9.4 | |
| M. norvegica (Northern krill) | Crustacea | 1 | >50 | 30.3 ± 5.1 | |
| Pasiphaea sp. | Crustacea | 1 | >50 | 49.3 ± 19.6 | |
| Eusergestes arcticus | Crustacea | 1 | >50 | 43.6 ± 7.2 | |
| Weight (g) | |||||
| Osterfjorden | Periphylla periphylla (Helmet jellyfish) | Cnidaria | 1 | 12 | 575 ± 446 |
| Bjørnafjorden | Periphylla periphylla (Helmet jellyfish) | Cnidaria | 1 | 10 | 952 ± 293 |
| Species | n | Protein g/100g (min–max) | Total Fat g/100g (min–max) | Dry Matter % (min–max) |
|---|---|---|---|---|
| Benthosema glaciale (Glacier lantern fish) | 7 | 14.0 ± 0.5 (13.5–14.6) | 13.7 ± 3.7 (6.1–16.0) | 30.8 ± 3.9 (22.0–33.7) |
| Maurolicus muelleri (Silvery lightfish) | 4 | 12.3 ± 0.4 (11.9–12.7) | 17.8 ± 8.1 (7.1–24.7) | 33.3 ± 8.1 (23.0–41.2) |
| Meganyctyphanes norvegica (Northern krill) | 4 | 15.5 ± 0.9 (14.8–16.8) | 5.5 ± 0.6 (4.9–5.9) | 24.0 ± 1.9 (21.3–25.3) |
| Pasiphaea sp. | 3 | 14.1 ± 4.6 (42–50) | 5.4 ± 2.7 (3.3–8.4) | 21.7 ± 5.1 (15.9–24.1) |
| Eusergestes arcticus | 4 | 15.5 ± 0.5 (14.9–15.9) | 9.4 ± 3.1 (4.9–12.1) | 27.5 ± 3.6 (22.3–30.7) |
| Periphylla periphylla (Helmet jellyfish) | 2 | 0.95 (0.90–1.00) | 0.45 (0.34–0.56) | 4.82 (4.76–4.87) |
| Micromesistius poutassou * (Blue whiting) | 10 | 16.1 (15.5–17.1) | 3.9 (2.9–5.8) | 20.8 (18.4–22.9) |
| Species | n | Vitamin A1 µg/100g | Iodine µg/100g (min–max) | Calcium mg/100g (min–max) | Potassium mg/100g (min–max) | Magnesium mg/100g (min–max) | Phosphorus mg/100g (min–max) | Sodium mg/100g (min–max) | Selenium µg/100g (min–max) | Zinc mg/100g (min–max) | Iron mg/100g (min–max) |
|---|---|---|---|---|---|---|---|---|---|---|---|
| B. glaciale | 7 | 1633 ± 356 (1300–2300) | 43 ± 6 (30–49) | 500 ± 47 (420–550) | 258 ± 51 (160–300) | 67 ± 12 (52–89) | 383 ± 60 (280–440) | 385 ± 108 (300–600) | 61 ± 9 (47–72) | 0.8 ± 0.1 (0.7–1.0) | 1.08 ± 0.44 (0.61–1.83) |
| Maurolicus muelleri | 4 | 1020 ± 395 (480–1400) | 27 ± 14 (16–47) | 543 ± 60 (480–600) | 227 ± 6 (220–230) | 61 ± 8 (54–70) | 400 ± 10 (390–410) | 380 ± 69 (340–460) | 44 ± 8 (34–52) | 1.1 ± 0.1 (1.1–1.2) | 1.56 ± 0.05 (1.50–1.60) |
| M. norvegica | 4 | 63.3 ± 15.3 (50.0–80.0) | 119 ± 42 (85–180) | 658 ± 57 (590–730) | 358 ± 33 (320–390) | 163 ± 13 (150–180) | 368 ± 22 (340–390) | 495 ± 124 (360–660) | 101 ± 41 (71–160) | 1.0 ± 0.1 (0.9–1.1) | 2.15–1.39 (0.98–4.00) |
| Pasiphaea sp. | 3 | 11.0 ± 1.0 (10.0–12.0) | 46 ± 4 (42–50) | 633 ± 211 (410–830) | 283 ± 110 (160–370) | 83 ± 29 (53–110) | 333 ± 119 (200–430) | 337 ± 107 (220–430) | 43 ± 21 (23–65) | 0.9 ± 0.3 (0.6–1.1) | 0.39 ± 0.30 (0.19–0.74) |
| Eusergestes arcticus | 4 | 34.5 ± 29.6 (6.0–60.0) | 117 ± 6 (110–120) | 532 ± 88 (460 -660) | 358 ± 22 (300–420) | 378 ± 22 (350–400) | 377 ± 22 (350–400) | 363 ± 51 (300–420) | 52 ± 17 (38–76) | 1.8 ± 0.9 (1.0–3.1) | 0.32 ± 0.11 (0.23–0.45) |
| Periphylla periphylla | 2 | 0.3 (0.15–0.45) | 2.3 (2–2.5) | 43 (42–44) | 83 (80–86) | 105 (110–110) | 12.3 (9.6–15.0) | 1000 (1000–1000) | 3.9 (3.4–4.4) | 0.1 (0.1–0.1) | 0.04 (0.04–0.05) |
| M. * poutassou | 10 | 2370 (1000–4500) | 23 (19–34) | 429 (198–785) | 264 (233–282) | 64 (55–72) | 309 (222–517) | 425 (373–466) | 62 (60–64) | 1.1 (1.0–1.2) | 1.75 (1.50–2.00) |
| B. glaciale g/100 g ww min–max % (n = 8) | Maurolicus muelleri g/100 g ww min–max % (n = 4) | M. norvegica g/100 g ww min–max % (n = 4) | Pasiphaea sp. g/100 g ww min–max % (n = 3) | E. arcticus g/100 g ww min–max % (n = 4) | P. periphylla g/100 g ww min–max % (n = 2) | M. poutassou * g/100g ww min–max (n = 10) | Fish oil ** % (n=10) | |
|---|---|---|---|---|---|---|---|---|
| Amount FA (g/100 g sample weight) | 6.8 ± 1.7 | 14.5 ± 7.9 | 3.4 ± 1.7 | 3.7 ± 0.8 | 5.3 ± 2.1 | 0.2 | ||
| Amount FAOH (g/100 g sample weight) | 4.2 ± 1.2 | 0.03 ± 0.01 | 0.07 ± 0.02 | 0.02 ± 0.01 | 2.4 ± 1.0 | 0.04 ± 0.05 | ||
| 14:0 | 0.34 ± 0.10 0.13–0.42 5.0 ± 0.5 | 1.05 ± 0.62 0.34–1.65 7.1 ± 0.6 | 0.19 ± 0.11 0.04–0.29 5.1 ± 1.0 | 0.10 ± 0.06 0.04–0.16 2.4 ± 0.8 | 0.17 ± 0.04 0.11–0.20 3.3 ±0.8 | 0.006 0.004–0.008 3.0 | 0.14 0.09–0.21 | 7.3 ± 1.3 |
| 16:0 | 0.39 ± 0.09 0.21–0.47 5.9 ± 0.5 | 2.29 ± 1.23 1.00–3.39 16.1 ± 2.0 | 0.52 ± 0.25 0.16–0.72 15.2 ± 0.4 | 0.59 ± 0.29 0.38–0.92 15.9 ± 0.1 | 0.47 ± 0.16 0.27–0.66 9.0 ± 0.8 | 0.018 0.013–0.023 9.6 | 0.50 0.37–0.79 | 15.8 ±2.6 |
| Sum SFA | 0.90 ± 0.21 0.73–1.10 13.0 ± 1.5 | 3.78 ± 2.04 1.58–5.52 26.5 ± 2.4 | 0.85 ± 0.43 1.10–4.88 24.7 ± 1.1 | 0.86 ± 0.41 0.53–1.31 23.0 ± 0.8 | 0.74 ± 0.22 0.45–0.99 14.5 ± 1.8 | 0.035 0.03–0.04 18.5 | 26.9 ± 4.8 | |
| 18:1n-9 | 1.35± 0.43 0.62–1.82 19.8 ± 3.1 | 1.35 ± 0.79 0.57–2.207 9.4 ± 1.8 | 0.43 ± 0.19 0.16–0.509 13.4 ± 3.5 | 0.80 ± 0.36 0.57–1.22 21.8 ± 3.5 | 0.80 ± 0.43 0.50–1.43 15.1 ± 4.0 | 0.029 0.025–0.033 16.6 | 0.44 0.29–0.76 | 10.0 ± 3.3 |
| 20:1n-9 | 0.53 ± 0.17 0.17–0.73 7.6 ± 1.1 | 1.52 ± 0.89 0.36–2.216 10.0 ± 2.2 | 0.24 ± 0.18 0.01–0.43 5.9 ± 3.2 | 0.19 ± 0.13 0.09–0.34 4.7 ± 1.2 | 0.55 ± 0.26 0.18–0.77 9.9 ± 2.3 | 0.018 ± 0.005 0.015–0.022 9.7 | 0.21 0.10–0.42 | 5.4 ± 4.8 |
| 20:1n-11 | 0.13 ± 0.4 0.04 -0.17 3.0 ± 0.8 | 0.15 ± 0.08 0.13–0.23 1.9 ± 0.3 | 0.03 ± 0.02 0.00–0.04 1.0 ±0.2 | 0.04 ± 0.03 0.02–0.07 0.7 ± 0.2 | 0.17 ± 0.09 0.05–0.25 1.0 ± 0.2 | 0.00 0.002–0.003 1.4 | 0.045 0.02–0.09 | 0.5 ± 0.4 |
| 22:1n-11 | 0.78 ± 0.24 0.26–1.07 11.3 ± 1.5 | 3.08 ± 1.77 0.74–4.28 20.4 ± 4.8 | 0.26 ± 0.22 0.01–0.54 6.1 ± 4.15 | 0.20 ± 0.16 0.09–0.38 4.9 ± 1.5 | 0.52 ± 0.23 0.19–0.69 9.5 ±2.1 | 0.027 0.019–0.035 14.4 | 0.28 0.11–0.67 | 7.5 ± 7.0 |
| Sum MUFA | 4.03 ± 1.14 1.60–5.05 57.1 ± 5.8 | 7.82 ± 4.41 2.40–11.84 52.7 ±5.8 | 1.42 ± 0.79 0.34–2.21 39.6 ± 6.6 | 1.75 ± 0.95 1.12–2.84 46.1 ± 4.1 | 3.05 ± 1.33 1.33–4.58 56.0 ± 4.1 | 0.104 0.09–0.11 56.7 | 35.5 ±12.8 | |
| 18:2n-6 | 0.12 ± 0.03 0.06–0.15 1.8 ± 0.2 | 0.19 ± 0.10 0.07–0.28 1.2 ± 0.1 | 0.08 ± 0.03 0.04–0.11 1.7 ± 0.4 | 0.06 ± 0.02 0.04–0.07 1.7 ± 0.4 | 0.11 ± 0.05 0.07–0.18 2.2 ± 0.4 | 0.002 0.002–0.003 1.2 | 0.04 0.03–0.07 | 1.6 ±0.4 |
| 20:4n-6 | 0.04 ± 0.01 0.02–0.05 0.6 ± 0.1 | 0.05 ± 0.02 0.03–0.06 0.4 ± 0.1 | 0.03 ± 0.01 0.03–0.04 1.2 ± 0.7 | 0.04 ± 0.01 0.03–0.04 1.1 ± 0.4 | 0.04 ± 0.01 0.03–0.05 0.7 ± 0.2 | 0.002 0.002–0.002 1.0 | 0.03 0.01–0.04 | 0.9 ±0.4 |
| SUM n-6 | 0.217 ± 0.050 0.12–0.26 3.2 ± 0.3 | 0.31 ± 0.15 0.14–0.46 2.3 ± 0.2 | 0.15 ± 0.05 0.08–0.19 5.0 ± 1.9 | 0.13 ± 0.03 0.10–0.16 3.7 ± 0.8 | 0.20 ± 0.08 0.13–0.30 3.8 ± 0.7 | 0.01 0.01–0.01 6.6 | 2.8 ± 0.5 | |
| 20:5n-3 | 0.414 ± 0.104 0.21–0.51 6.2 ± 0.6 | 0.61 ± 0.35 0.28–0.97 4.3 ± 0.8 | 0.32 ± 0.14 0.15–0.44 10.2 ± 2.7 | 0.37 ± 0.14 0.25–0.52 10.3 ± 1.1 | 0.46 ± 0.16 0.27 - 0.65 8.9 ± 0.8 | 0.013 0.008–0.017 2.6 | 0.23 0.15–0.36 | 11.8 ± 4.1 |
| 22:6n-3 | 0.68 ± 0.13 0.42–0.79 10.4 ± 1.6 | 1.11 ± 0.57 0.61–1.79 8.2 ± 2. 5 | 0.45 ± 0.19 0.21–0.61 14.2 ± 3.1 | 0.44 ± 0.16 0.30–0.62 12.1 ± 1.2 | 0.47 ± 0.16 0.29–0.68 9.1 ± 1.2 | 0.05 0.04–0.06 6.6 | 0.53 0.44–0.64 | 10.2 ± 2.2 |
| Sum n-3 | 1.54 ± 0.35 0.84–1.84 22.5 ± 3.1 | 2.44 ± 1.31 1.16–3.97 17.5 ± 3.5 | 0.96 ± 0.44 0.41–1.33 29.7 ± 5.7 | 0.96 ± 0.37 0.61–1.36 26.4 ± 3.0 | 1.27 ± 0.47 0.72–1.86 24.3 ± 1.9 | 0.039 0.03–0.05 3.3 | 0.92 0.71–1.25 | 30.1 ± 6.9 |
| Sum PUFA | 1.85 ± 0.41 1.00–2.22 27.1 ± 3.3 | 2.89 ± 1.52 1.35–4.62 20.8 ± 3.7 | 1.14 ± 0.49 0.50–1.55 35.6 ± 7.2 | 1.12 ± 0.42 0.73–1.56 30.9 ± 3.6 | 1.54 ± 0.57 0.88–2.26 29.5 ± 2.5 | 0.047 0.03–0.06 24.9 | 33.7 ± 7.6 |
| Potential contribution to RI (%) | Iodine | Calcium | Iron | Zinc | Selenium | Vitamin A1 | Vitamin D3 |
|---|---|---|---|---|---|---|---|
| RI a | 150 µg | 800 mg | 15 mg | 7 mg | 50 µg | 700 RE | 10 µg |
| Benthosema glaciale | 14 | 31 | 4 | 6 | 61 | 117 | <LOQ |
| Maurolicus muelleri | 9 | 34 | 5 | 8 | 44 | 73 | <LOQ |
| M. norvegica | 40 | 41 | 7 | 7 | 101 | 5 | <LOQ |
| Pasiphaea sp. | 15 | 40 | 1 | 6 | 43 | 1 | <LOQ |
| Eusergestes arcticus | 39 | 33 | 1 | 13 | 52 | 3 | <LOQ |
| Periphylla periphylla | 1 | 3 | 0 | 8 | 4 | 0 | <LOQ |
| Salmon filet (Salmo salar) | 1 | 0 | 1 | 3 | 17 | - | 43 |
| Cod filet (Gadus morhua) | 63 * | - | 0 | 3 | 25 | 1 | - |
| Sprat (Sprattus sprattus) | 2 | - | 6 | 15 | 36 | - | - |
| Pork | 0 | 0 | 3 | 13 | 6 | 0 | 0 |
| Chicken b | 0 | 1 | 2 | 11 | 12 | 1 | 0 |
| Beef | 1 | 0 | 8 | 29 | 6 | 0 | 0 |
| No of daily doses of RI from mesopelagic species/ km3 fjord c | 169,000 | 353,000 | 31,800 | 87,300 | 591,000 | 348,000 | - |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Alvheim, A.R.; Kjellevold, M.; Strand, E.; Sanden, M.; Wiech, M. Mesopelagic Species and Their Potential Contribution to Food and Feed Security—A Case Study from Norway. Foods 2020, 9, 344. https://doi.org/10.3390/foods9030344
Alvheim AR, Kjellevold M, Strand E, Sanden M, Wiech M. Mesopelagic Species and Their Potential Contribution to Food and Feed Security—A Case Study from Norway. Foods. 2020; 9(3):344. https://doi.org/10.3390/foods9030344
Chicago/Turabian StyleAlvheim, Anita R., Marian Kjellevold, Espen Strand, Monica Sanden, and Martin Wiech. 2020. "Mesopelagic Species and Their Potential Contribution to Food and Feed Security—A Case Study from Norway" Foods 9, no. 3: 344. https://doi.org/10.3390/foods9030344
APA StyleAlvheim, A. R., Kjellevold, M., Strand, E., Sanden, M., & Wiech, M. (2020). Mesopelagic Species and Their Potential Contribution to Food and Feed Security—A Case Study from Norway. Foods, 9(3), 344. https://doi.org/10.3390/foods9030344





