Method of Food Preparation Influences Blood Glucose Response to a High-Carbohydrate Meal: A Randomised Cross-over Trial
Abstract
1. Introduction
2. Materials and Methods
2.1. Participants
2.2. Experimental Conditions
2.3. Pasta Preparation
2.4. Blood Glucose Responses
2.5. Calculations and Data Analysis
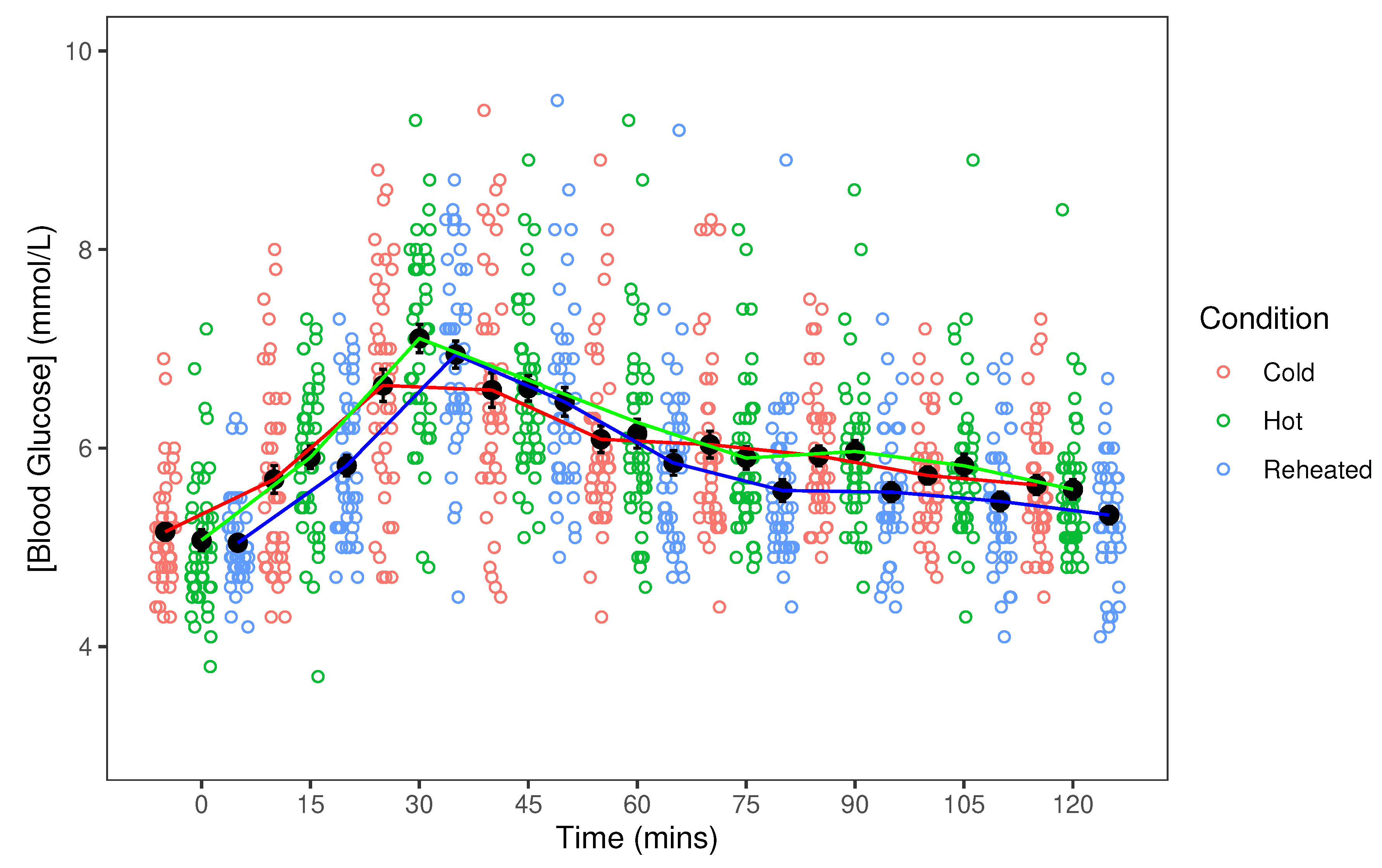
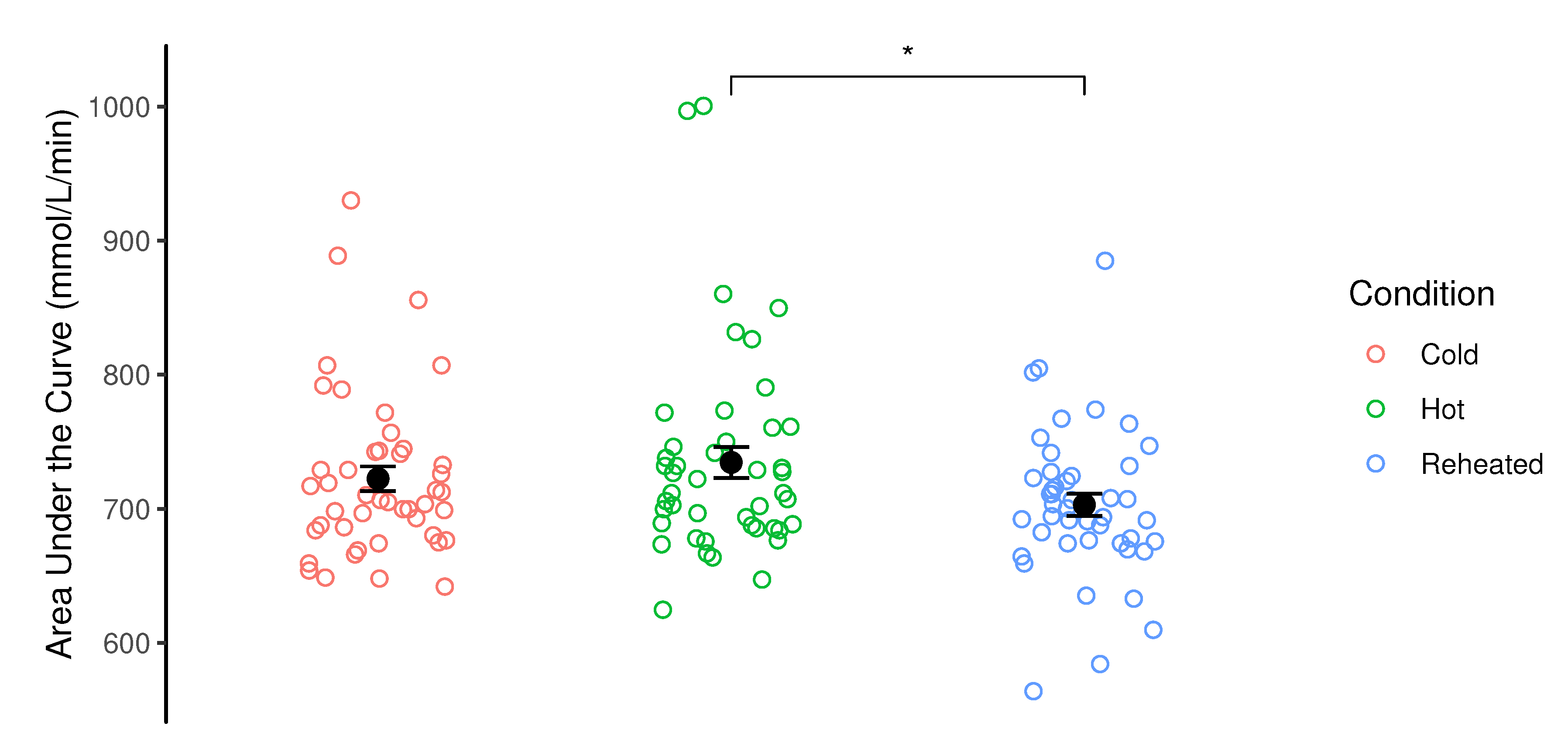
3. Results
3.1. Preparation Method
3.2. Time
3.3. Preparation Method and Time Interaction
3.4. Area Under the Curve
4. Discussion
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| ANOVA | Analysis of Variance |
| GI | Glycaemic Index |
| AUC | Area under the Curve |
| T2DM | Type-2 Diabetes Mellitus |
Appendix A
References
- Cust, A.; Skilton, M.; van Bakel, M.; Halkjaer, J.; Olsen, A.; Agnoli, C.; Psaltopoulou, T.; Buurma, E.; Sonestedt, E.; Chirlaque, M.D.; et al. Total dietary carbohydrate, sugar, starch and fibre intakes in the European Prospective Investigation into Cancer and Nutrition. Eur. J. Clin. Nutr. 2009, 63, S37–S60. [Google Scholar] [CrossRef] [PubMed]
- Heinemann, R.J.B.; Fagundes, P.L.; Pinto, E.A.; Penteado, M.V.C.; Lanfer-Marquez, U.M. Comparative study of nutrient composition of commercial brown, parboiled and milled rice from Brazil. J. Food Comp. Anal. 2005, 18, 287–296. [Google Scholar] [CrossRef]
- Huang, M.; Li, J.; Há, M.A.; Riccardi, G.; Liu, S. A systematic review on the relations between pasta consumption and cardio-metabolic risk factors. Nutr. Metab. Cardiovasc. Dis. 2017, 27, 939–948. [Google Scholar] [CrossRef] [PubMed]
- Lattimer, J.; Haub, M. Effects of Dietary Fiber and Its Components on Metabolic Health. Nutrients 2010, 2, 1266–1289. [Google Scholar] [CrossRef] [PubMed]
- Haralampu, S. Resistant starch—A review of the physical properties and biological impact of RS3. Carbohydr. Polym. 2000, 41, 285–292. [Google Scholar] [CrossRef]
- Sajilata, M.G.; Singhal, R.S.; Kulkarni, P.R. Resitant Starch—A Review. Comp. Rev. Food Sci. Food Saf. 2006, 5, 1–17. [Google Scholar] [CrossRef]
- Ojo, O.; Ojo, O.O.; Wang, X.H.; Adegboye, A.R.A. The Effect of Dietary Glycaemic Index on Glycaemia in Patients with Type 2 Diabetes: A Systematic Review and Meta-Analysis of Randomized Controlled Trials. Nutrients 2018, 10, 1584. [Google Scholar] [CrossRef]
- Ojo, O.; Ojo, O.O.; Wang, X.H.; Adegboye, A.R.A. The Effects of a Low GI Diet on Cardiometabolic and Inflammatory Parameters in Patients with Type 2 and Gestational Diabetes: A Systematic Review and Meta-Analysis of Randomised Controlled Trials. Nutrients 2019, 11, 1584. [Google Scholar] [CrossRef]
- Robertson, M.D. Could resistant starch be the redeemer of dietary carbohydrate? Pract. Diab. 2014, 31, 375. [Google Scholar] [CrossRef]
- Cocci, E.; Sacchetti, G.; Vallicelli, M.; Angioloni, A.; Dalla Rossa, M. Spaghetti cooking by microwave oven: Cooking kineticsand product quality. J. Food Eng. 2008, 85, 537–546. [Google Scholar] [CrossRef]
- Brouns, F.; Bjorck, I.; Frayn, K.; Gibbs, A.L.; Lang, V.; Slama, G.; Wolever, T.M.S. Glycaemic index methodology. Nutr. Res. Rev. 2005, 18, 145–171. [Google Scholar] [CrossRef] [PubMed]
- The Jamovi Project (2019). Available online: https://www.jamovi.org (accessed on 9 October 2019).
- Akerberg, A.K.; Liljeberg, H.G.; Granfeldt, Y.E.; Drews, A.W.; Björck, I.M. An in vitro method, based on chewing, to predict resistant starch content in foods allows parallel determination of potentially available starch and dietary fiber. J. Nutr. 1998, 128, 651–660. [Google Scholar] [CrossRef] [PubMed]
- Wang, N.; Hatcher, D.W.; Tyler, R.T.; Toews, T.; Gawalko, E.J. Influence of cooking and dehulling on nutritional composition of several varieties of lentils (lens culinaris). LWT Food Sci. Technol. 2009, 42, 842–848. [Google Scholar] [CrossRef]
- Garcia-Alonso, A.; Jimenez-Escrig, A.; Martin-Carron, N.; Bravo, L.; Saura-Calixto, F. Assessment of some parameters involved in the gelatinization and retrogradation of starch. Food Chem. 1999, 66, 181–187. [Google Scholar] [CrossRef]
- Monro, J.; Mishra, S.; Blandford, E.; Anderson, J.; Genet, R. Potato genotype differences in nutritionally distinct starch fractions after cooking, and cooking plus storing cool. J. Food Compost. Anal. 2009, 22, 539–545. [Google Scholar] [CrossRef]
- Raatz, S.K.; Idso, L.; Johnson, L.K.; Jackso, M.I.; Combs, G.F. Resistant starch analysis of commonly consumed potatoes: Content varies by cooking method and service temperature but not by variety. Food Chem. 2016, 208, 297–300. [Google Scholar] [CrossRef]
- Wepner, B.; Berghofer, E.; Miesenberger, E.; Tiefenbacher, K. Citrate starch: Application as resistant starch in different food systems. Starch/Stärke 1999, 51, 354–361. [Google Scholar] [CrossRef]
- Garcia-Alonso, A.; Goni, I. Effect of processing on potato starch: In vitro availability and glycaemic index. Nahrung 2000, 44, 19–22. [Google Scholar] [CrossRef]
- Leeman, A.M.; Barstro¨m, L.M.; Bjo¨rck, I.M.E. In vitro availability of starch in heat-treated potatoes as related to genotype, weight and storage time. J. Sci. Food Agric. 2005, 85, 751–756. [Google Scholar] [CrossRef]
- Nayak, B.; De, J.; Berrios, J.; Tang, J. Impact of food processing on the glycemic index (GI) of potato products. Food Res. Int. 2014, 56, 35–46. [Google Scholar] [CrossRef]
- Sharma, A.; Yadav, B.S.; Yadav, R.B. Resistant starch: Physiological roles and food applications. Food Rev. Int. 2008, 24, 193–234. [Google Scholar] [CrossRef]
- Englyst, H.; Wiggins, H.S.; Cummings, J.H. Determination of the non-starch polysaccharides in plant foods by gas-liquid chromatography of constituent sugars as alditol acetates. Analyst 1982, 107, 307–318. [Google Scholar] [CrossRef] [PubMed]
- Granito, M.; Perez, S.; Valero, Y. Quality of cooking acceptability and glycemic index of enriched pasta with legumes. Rev. Chilena Nutr. 2014, 41, 425–432. [Google Scholar] [CrossRef]
- Sonia, S.; Witjaksono, F.; Ridwan, R. Effect of cooling of cooked white rice on resistant starch content and glycemic response. Asia Pac. J. Clin. Nutr. 2015, 24, 620–625. [Google Scholar] [PubMed]
- Lu, L.; Venn, B.; Lu, J.; Monro, J.; Rush, E. Effect of Cold Storage and Reheating of Parboiled Rice on Postprandial Glycaemic Response, Satiety, Palatability and Chewed Particle Size Distribution. Nutrients 2017, 9, 475. [Google Scholar] [CrossRef] [PubMed]
- Lu, L.; Monro, J.; Lu, J.; Rush, E. Effect of Cold Storage, Reheating, and Particle Sizes on In Vitro Glucose Release and Starch Digestibility among Five Rice Products in Auckland, New Zealand. J. Rice Res. 2016, 4, 171. [Google Scholar]
- Boers, H.M.; ten Hoorn, J.S.; Mela, D.J. A systematic review of the influence of rice characteristics and processing methods on post-prandial glycaemic and insulinaemic responses. Br. J. Nutr. 2015, 114, 1035–1045. [Google Scholar] [CrossRef]
- Asp, N.-G.; van Amelsvoort, J.M.M.; Hautvast, J.G.A.J. Nutritional implications of resistant starch. Nutr. Res. Rev. 1996, 9, 1–31. [Google Scholar] [CrossRef]
- Larsen, H.N.; Rasmussen, O.W.; Rasumussen, P.H.; Alstrup, K.K.; Biswas, S.K.; Tetens, I.; Thilsted, S.H.; Hermansen, K. Glycaemic index of parboiled rice depends on the severity of processing: Study in type 2 diabetic subjects. Eur. J. Clin. Nutr. 2000, 54, 380–385. [Google Scholar] [CrossRef]
- Fernandes, G.; Velangi, M.; Wolever, T. Glycemic index of potatoes commonly consumed in North America. J. Am. Diet. Assoc. 2005, 105, 557–562. [Google Scholar] [CrossRef]
- Darman-Djoulde, R.; Oldewage-Theron, W.; Egal, A.A.; Samuel, F. Influence of room-temperatures storage-reheating cycles on nutritional properties of maize meal porridge (PAP) used in the Vaal region, South Africa. J. Food Process. Preserv. 2010, 34, 181–191. [Google Scholar] [CrossRef]
- World Health Organization, Food and Agriculture Organization of the United Nations. Carbohydrates in Human Nutrition Report of a Joint FAO/WHO Expert Consultation; FAO: Rome, Italy, 1998. [Google Scholar]
- Wolever, T.M.S.; Jenkins, A.L.; Prudence, K.; Johnson, J.; Duss, R.; Chu, Y.; Steinert, R.E. Effect of adding oat bran to instant oatmeal on glycaemic response in humans-a study to establish the minimum effective dose of oat β-glucan. Food Funct. 2018, 9, 1692–1700. [Google Scholar] [CrossRef] [PubMed]
- Schisano, B.; Tripathi, G.; McGee, K.; McTernan, P.G.; Ceriello, A. Glucose oscillations, more than constant high glucose, induce p53 activation and a metabolic memory in human endothelial cells. Diabetologia 2011, 54, 1219–1226. [Google Scholar] [CrossRef] [PubMed]
- Dickinson, S.; Hancock, D.P.; Petocz, P.; Ceriello, A.; Brand-Miller, J. High-glycemic index carbohydrate increases nuclear factor-kappaB activation in mononuclear cells of young, lean healthy subjects. Am. J. Clin. Nutr. 2008, 87, 1188–1193. [Google Scholar] [PubMed]
- Benalia, M.; Zeller, M.; Mouhat, B.; Guenancia, C. Glycaemic variability is associated with severity of coronary artery disease in patients with poorly controlled type 2 diabetes and acute myocardial infarction. Diabet. Metab. 2019, 45, 446–452. [Google Scholar] [CrossRef]
- Monnier, L.; Colette, C.; Mas, E.; Michel, F.; Cristol, J.P.; Boegner, C.; Owens, D.R. Regulation of oxidative stress by glycaemic control: Evidence for an independent inhibitory effect of insulin therapy. Diabetologia 2010, 53, 562–571. [Google Scholar] [CrossRef]
- Esposito, K.; Ciotola, M.; Carlen, D.; Schisano, B.; Sardelli, L.; Di Tommaso, D.; Misso, L.; Saccomanno, F.; Ceriello, A.; Guigliano, D. Post-meal glucose peaks at home associate with carotid intima-media thickness in type 2 diabetes. J. Clin. Endocrinol. 2008, 93, 1345–1350. [Google Scholar] [CrossRef]
| Time (mins) | 0 | 15 | 30 | 45 | 60 | 75 | 90 | 105 | 120 |
|---|---|---|---|---|---|---|---|---|---|
| Cold | 5.16 | 5.68 | 6.63 | 6.58 | 6.09 | 6.04 | 5.72 | 5.72 | 5.63 |
| [0.57] | [0.94] | [1.08] | [1.17] | [0.91] | [0.91] | [0.66] | [0.58] | [0.65] | |
| Hot | 5.07 | 5.91 | 7.10 | 6.68 | 6.26 | 5.90 | 5.97 | 5.82 | 5.58 |
| [0.70] | [0.76] | [0.95] | [1.00] | [1.23] | [0.77] | [0.78] | [0.78] | [0.67] | |
| Reheated | 5.05 | 5.82 | 6.94 | 6.47 | 5.85 | 5.57 | 5.56 | 5.46 | 5.33 |
| [0.44] | [0.71] | [0.92] | [0.97] | [0.93] | [0.74] | [0.62] | [0.63] | [0.59] |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hodges, C.; Archer, F.; Chowdhury, M.; Evans, B.L.; Ghelani, D.J.; Mortoglou, M.; Guppy, F.M. Method of Food Preparation Influences Blood Glucose Response to a High-Carbohydrate Meal: A Randomised Cross-over Trial. Foods 2020, 9, 23. https://doi.org/10.3390/foods9010023
Hodges C, Archer F, Chowdhury M, Evans BL, Ghelani DJ, Mortoglou M, Guppy FM. Method of Food Preparation Influences Blood Glucose Response to a High-Carbohydrate Meal: A Randomised Cross-over Trial. Foods. 2020; 9(1):23. https://doi.org/10.3390/foods9010023
Chicago/Turabian StyleHodges, Caroline, Fay Archer, Mardiyyah Chowdhury, Bethany L. Evans, Disha J. Ghelani, Maria Mortoglou, and Fergus M. Guppy. 2020. "Method of Food Preparation Influences Blood Glucose Response to a High-Carbohydrate Meal: A Randomised Cross-over Trial" Foods 9, no. 1: 23. https://doi.org/10.3390/foods9010023
APA StyleHodges, C., Archer, F., Chowdhury, M., Evans, B. L., Ghelani, D. J., Mortoglou, M., & Guppy, F. M. (2020). Method of Food Preparation Influences Blood Glucose Response to a High-Carbohydrate Meal: A Randomised Cross-over Trial. Foods, 9(1), 23. https://doi.org/10.3390/foods9010023





