The Combination of Hot Air and Chitosan Treatments on Phytochemical Changes during Postharvest Storage of ‘Sanhua’ Plum Fruits
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials and Treatments
2.2. Determination of Fruit Firmness and the Total Soluble Solids Content
2.3. Phytochemical Extraction
2.4. Determination of Phenolic Content
2.5. Determination of Flavonoid Content
2.6. Determination of Phytochemical Profiles
2.7. Determination of Total Antioxidant Activity
2.8. Statistical Analysis
3. Results
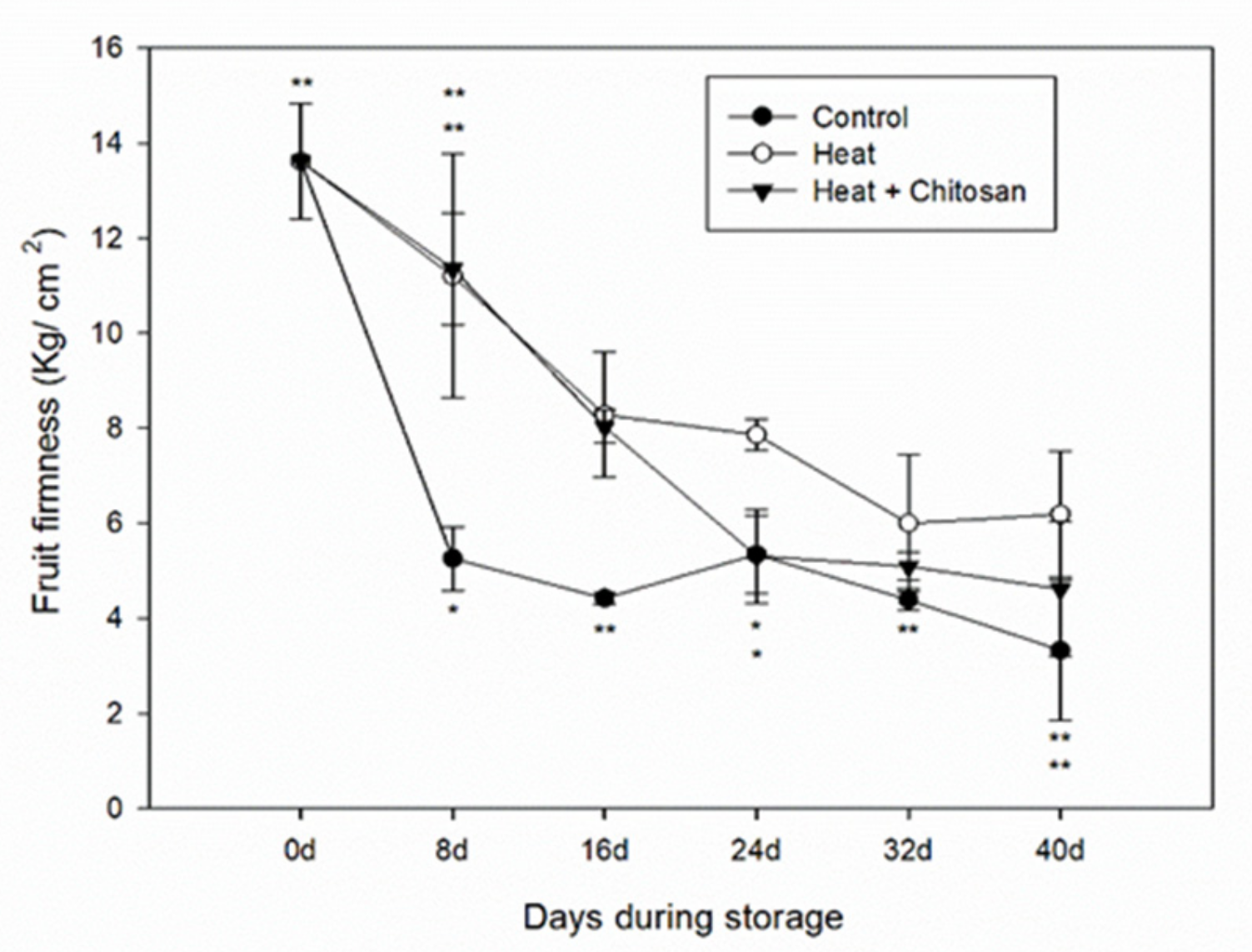
3.1. Fruit Firmness of ‘Sanhua’ Plum Fruit during Storage
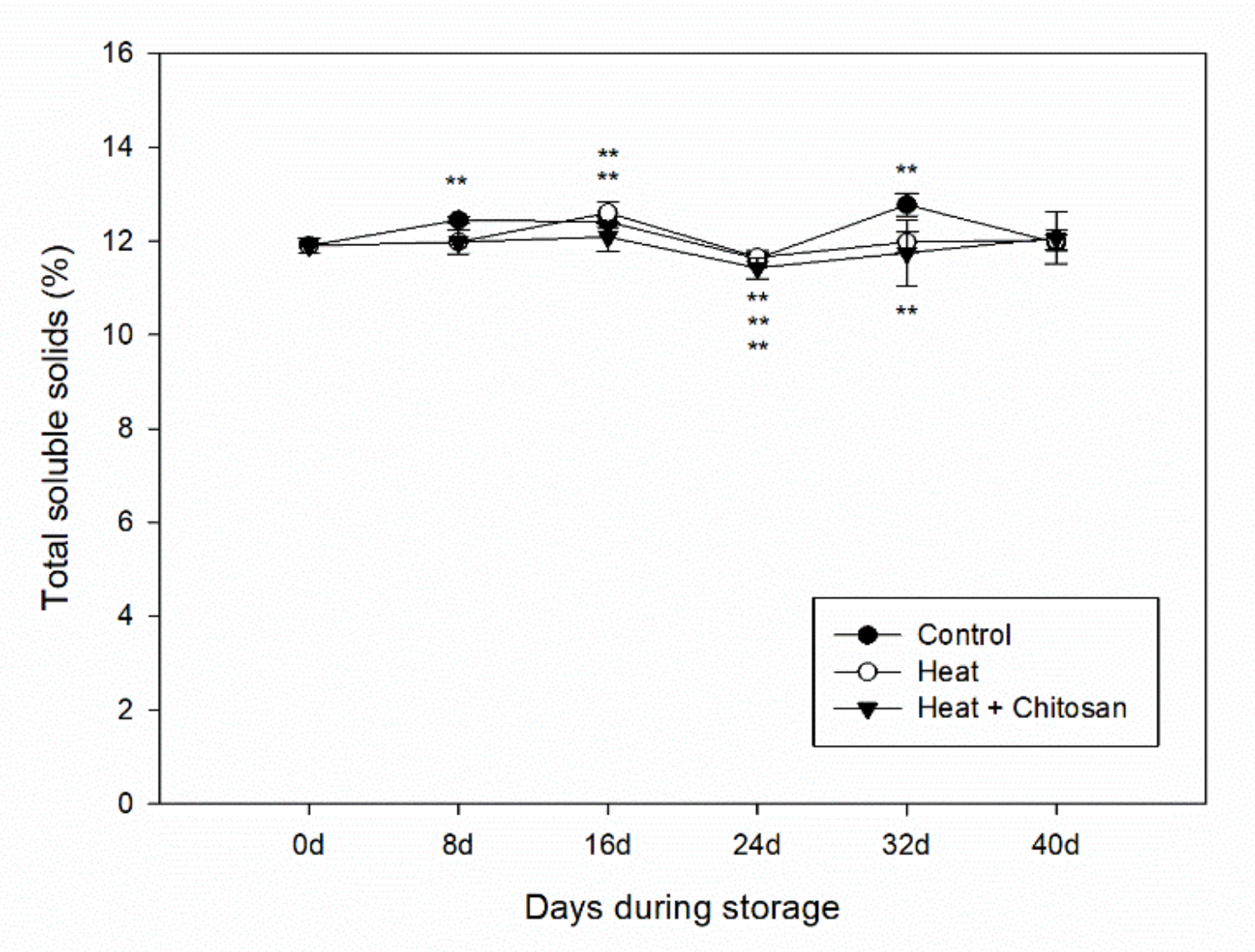
3.2. Total Soluble Solids of ‘Sanhua’ Plum Fruit during Storage
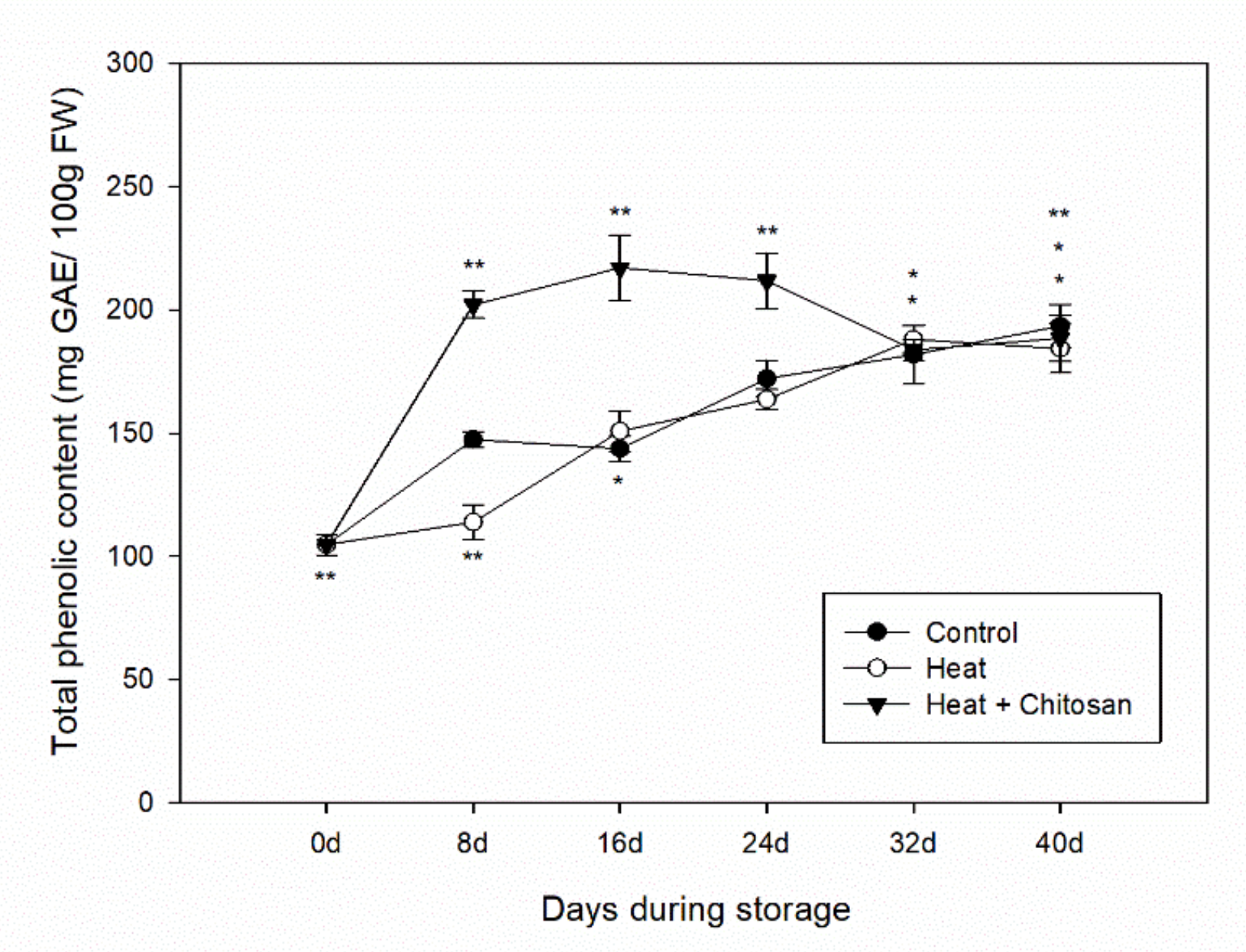
3.3. Total Phenolic Content of ‘Sanhua’ Plum Fruit during Storage
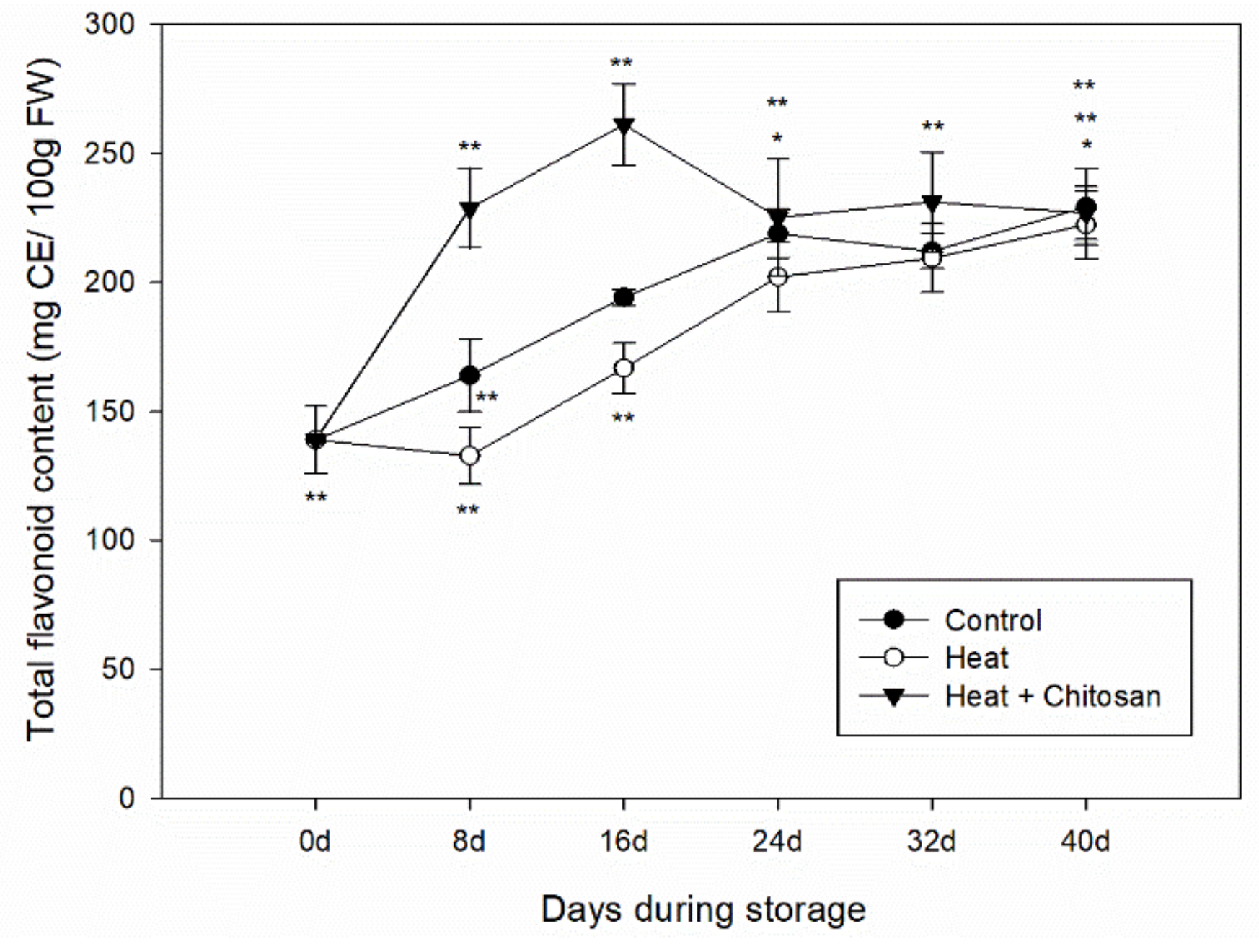
3.4. Total Flavonoid Content of ‘Sanhua’ Plum Fruit during Storage
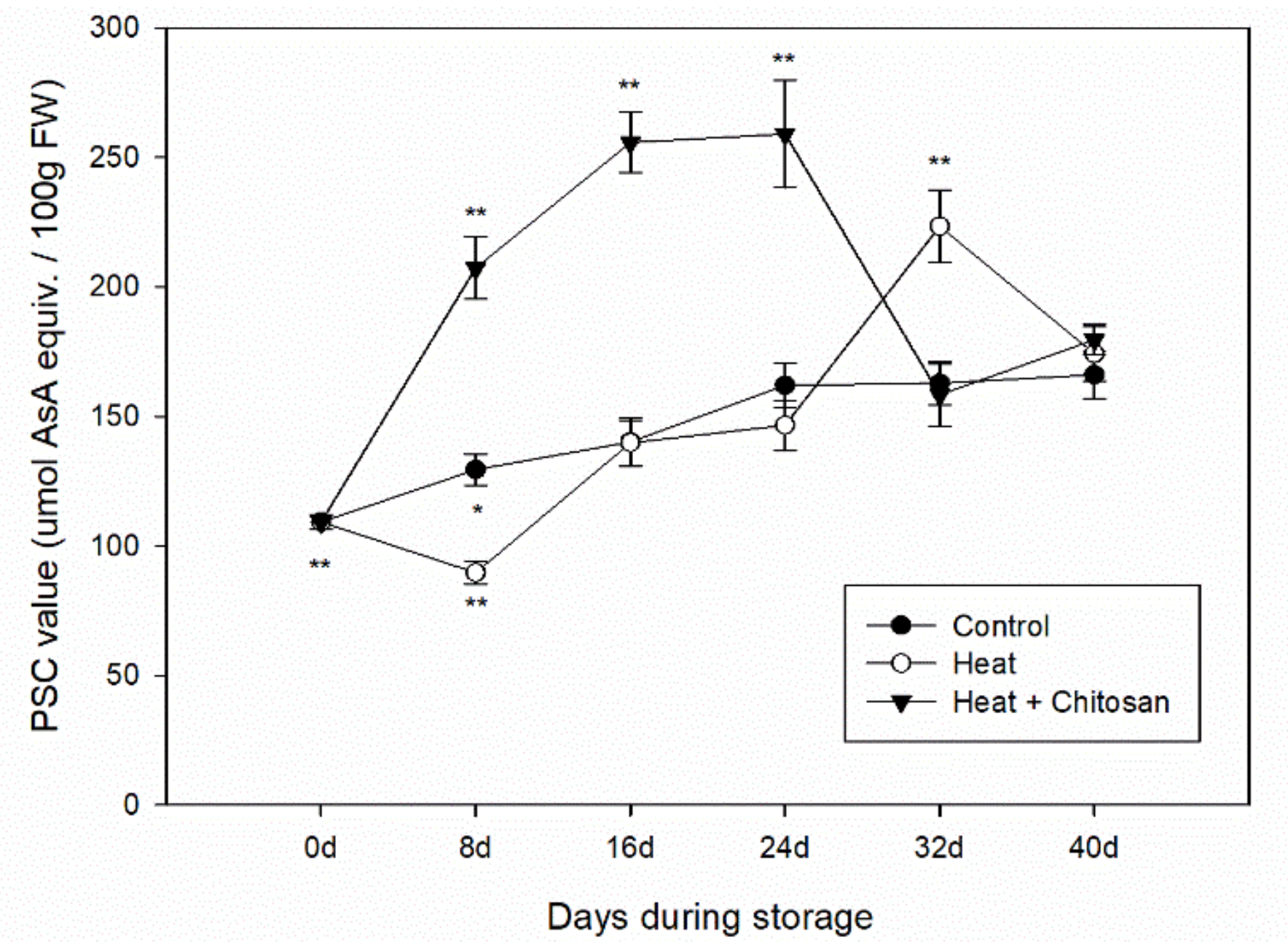
3.5. Total Antioxidant Activity of ‘Sanhua’ Plum Fruit during Storage
3.6. Phytochemical Profiles of ‘Sanhua’ Plum Fruit during Storage
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- González-Vallinas, M.; González-Castejón, M.; Rodríguez-Casado, A.; Ramírez de Molina, A. Dietary phytochemicals in cancer prevention and therapy: A complementary approach with promising perspectives. Nutr. Rev. 2013, 71, 585–599. [Google Scholar] [CrossRef] [PubMed]
- Liu, R.H. Potential Synergy of Phytochemicals in Cancer Prevention: Mechanism of Action. J. Nutr. 2004, 134, 3479S–3485S. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Williams, D.J.; Edwards, D.; Hamernig, I.; Jian, L.; James, A.P.; Johnson, S.K.; Tapsell, L.C. Vegetables containing phytochemicals with potential anti-obesity properties: A review. Food Res. Int. 2013, 52, 323–333. [Google Scholar] [CrossRef] [Green Version]
- Willett, W.C. Balancing Life-Style and Genomics Research for Disease Prevention. Science 2002, 296, 695–698. [Google Scholar] [CrossRef] [PubMed]
- Mihalache Arion, C.; Tabart, J.; Kevers, C.; Niculaua, M.; Filimon, R.; Beceanu, D.; Dommes, J. Antioxidant potential of different plum cultivars during storage. Food Chem. 2014, 146, 485–491. [Google Scholar] [CrossRef] [PubMed]
- Rupasinghe, H.P.V.; Jayasankar, S.; Lay, W. Variation in total phenolics and antioxidant capacity among European plum genotypes. Sci. Hortic. 2006, 108, 243–246. [Google Scholar] [CrossRef]
- Puerta-Gomez, A.F.; Cisneros-Zevallos, L. Postharvest studies beyond fresh market eating quality: Phytochemical antioxidant changes in peach and plum fruit during ripening and advanced senescence. Postharvest Biol. Technol. 2011, 60, 220–224. [Google Scholar] [CrossRef]
- Díaz-Mula, H.M.; Zapata, P.J.; Guillén, F.; Valverde, J.M.; Valero, D.; Serrano, M. Modified atmosphere packaging of yellow and purple plum cultivars. 2. Effect on bioactive compounds and antioxidant activity. Postharvest Biol. Technol. 2011, 61, 110–116. [Google Scholar] [CrossRef]
- Valero, D.; Díaz-Mula, H.M.; Zapata, P.J.; Guillén, F.; Martínez-Romero, D.; Castillo, S.; Serrano, M. Effects of alginate edible coating on preserving fruit quality in four plum cultivars during postharvest storage. Postharvest Biol. Technol. 2013, 77, 1–6. [Google Scholar] [CrossRef]
- Abu-Kpawoh, J.C.; Xi, Y.F.; Zhang, Y.Z.; Jin, Y.F. Polyamine Accumulation Following Hot-Water Dips Influences Chilling Injury and Decay in ‘Friar’ Plum Fruit. J. Food Sci. 2002, 67, 2649–2653. [Google Scholar] [CrossRef]
- Cristina, B.; Alejandra, S.; Lucía, A.; Jose María, M.-J. Hot Water Treatment for Chilling Injury Reduction of Astringent ‘Rojo Brillante’ Persimmon at Different Maturity Stages. HortScience 2008, 43, 2120–2123. [Google Scholar]
- Lara, M.V.; Borsani, J.; Budde, C.O.; Lauxmann, M.A.; Lombardo, V.A.; Murray, R.; Andreo, C.S.; Drincovich, M.F. Biochemical and proteomic analysis of ‘Dixiland’ peach fruit (Prunus persica) upon heat treatment. J. Exp. Bot. 2009, 60, 4315–4333. [Google Scholar] [CrossRef] [PubMed]
- McDonald, R.E.; McCollum, T.G.; Baldwin, E.A. Temperature of water heat treatments influences tomato fruit quality following low-temperature storage. Postharvest Biol. Technol. 1999, 16, 147–155. [Google Scholar] [CrossRef]
- Pesis, E.; Faure, M.; Arie, M.B. Induction of chilling tolerance in mango by temperature conditioning, heat, low O2 and ethanol vapours. Acta Hortic. 1997, 455, 629–634. [Google Scholar] [CrossRef]
- Sun, J.-H.; Chen, J.-Y.; Kuang, J.-F.; Chen, W.-X.; Lu, W.-J. Expression of sHSP genes as affected by heat shock and cold acclimation in relation to chilling tolerance in plum fruit. Postharvest Biol. Technol. 2010, 55, 91–96. [Google Scholar] [CrossRef]
- Zhang, J.; Huang, W.; Pan, Q.; Liu, Y. Improvement of chilling tolerance and accumulation of heat shock proteins in grape berries (Vitis vinifera cv. Jingxiu) by heat pretreatment. Postharvest Biol. Technol. 2005, 38, 80–90. [Google Scholar] [CrossRef]
- García, J.F.; Olmo, M.; García, J.M. Decay incidence and quality of different citrus varieties after postharvest heat treatment at laboratory and industrial scale. Postharvest Biol. Technol. 2016, 118, 96–102. [Google Scholar] [CrossRef]
- Jemric, T.; Ivic, D.; Fruk, G.; Matijas, H.S.; Cvjetkovic, B.; Bupic, M.; Pavkovic, B. Reduction of Postharvest Decay of Peach and Nectarine Caused by Monilinia laxa Using Hot Water Dipping. Food Bioprocess Technol. 2011, 4, 149–154. [Google Scholar] [CrossRef]
- Spadoni, A.; Guidarelli, M.; Sanzani, S.M.; Ippolito, A.; Mari, M. Influence of hot water treatment on brown rot of peach and rapid fruit response to heat stress. Postharvest Biol. Technol. 2014, 94, 66–73. [Google Scholar] [CrossRef]
- Sui, Y.; Wisniewski, M.; Droby, S.; Norelli, J.; Liu, J. Recent advances and current status of the use of heat treatments in postharvest disease management systems: Is it time to turn up the heat? Trends Food Sci. Technol. 2016, 51, 34–40. [Google Scholar] [CrossRef]
- Wei, Y.; Zhou, D.; Peng, J.; Pan, L.; Tu, K. Hot Air Treatment Induces Disease Resistance through Activating the Phenylpropanoid Metabolism in Cherry Tomato Fruit. J. Agric. Food Chem. 2017, 65, 8003–8010. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Y.; Li, Y.; Yin, J. Effects of hot air treatment in combination with Pichia guilliermondii on postharvest preservation of peach fruit. J. Sci. Food Agric. 2019, 99, 647–655. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.; Cheng, Z.; Wisniewski, M.; Liu, Y.; Liu, J. Ecofriendly hot water treatment reduces postharvest decay and elicits defense response in kiwifruit. Environ. Sci. Pollut. Res. 2015, 22, 15037–15045. [Google Scholar] [CrossRef] [PubMed]
- Yuan, L.; Bi, Y.; Ge, Y.; Wang, Y.; Liu, Y.; Li, G. Postharvest hot water dipping reduces decay by inducing disease resistance and maintaining firmness in muskmelon (Cucumis melo L.) fruit. Sci. Hortic. 2013, 161, 101–110. [Google Scholar] [CrossRef]
- Zhou, D.; Sun, Y.; Li, M.; Zhu, T.; Tu, K. Postharvest hot air and UV-C treatments enhance aroma-related volatiles by simulating the lipoxygenase pathway in peaches during cold storage. Food Chem. 2019, 292, 294–303. [Google Scholar] [CrossRef] [PubMed]
- Dhall, R.K. Advances in Edible Coatings for Fresh Fruits and Vegetables: A Review. Crit. Rev. Food Sci. Nutr. 2013, 53, 435–450. [Google Scholar] [CrossRef] [PubMed]
- Kerch, G. Chitosan films and coatings prevent losses of fresh fruit nutritional quality: A review. Trends Food Sci. Technol. 2015, 46, 159–166. [Google Scholar] [CrossRef]
- Cissé, M.; Polidori, J.; Montet, D.; Loiseau, G.; Ducamp-Collin, M.N. Preservation of mango quality by using functional chitosan-lactoperoxidase systems coatings. Postharvest Biol. Technol. 2015, 101, 10–14. [Google Scholar] [CrossRef]
- Cosme Silva, G.M.; Silva, W.B.; Medeiros, D.B.; Salvador, A.R.; Cordeiro, M.H.M.; Da Silva, N.M.; Santana, D.B.; Mizobutsi, G.P. The chitosan affects severely the carbon metabolism in mango (Mangifera indica L. cv. Palmer) fruit during storage. Food Chem. 2017, 237, 372–378. [Google Scholar] [CrossRef]
- Zhang, W.; Zhao, H.; Zhang, J.; Sheng, Z.; Cao, J.; Jiang, W. Different molecular weights chitosan coatings delay the senescence of postharvest nectarine fruit in relation to changes of redox state and respiratory pathway metabolism. Food Chem. 2019, 289, 160–168. [Google Scholar] [CrossRef]
- Eberhardt, M.V.; Lee, C.Y.; Liu, R.H. Antioxidant activity of fresh apples. Nature 2000, 405, 903–904. [Google Scholar] [CrossRef] [PubMed]
- Guo, X.; Li, T.; Tang, K.; Liu, R.H. Effect of Germination on Phytochemical Profiles and Antioxidant Activity of Mung Bean Sprouts (Vigna radiata). J. Agric. Food Chem. 2012, 60, 11050–11055. [Google Scholar] [CrossRef] [PubMed]
- He, X.; Liu, D.; Liu, R.H. Sodium Borohydride/Chloranil-Based Assay for Quantifying Total Flavonoids. J. Agric. Food Chem. 2008, 56, 9337–9344. [Google Scholar] [CrossRef] [PubMed]
- Guo, R.; Guo, X.; Li, T.; Fu, X.; Liu, R.H. Comparative assessment of phytochemical profiles, antioxidant and antiproliferative activities of Sea buckthorn (Hippophaë rhamnoides L.) berries. Food Chem. 2017, 221, 997–1003. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Guo, X.; Hu, X.; Li, T.; Fu, X.; Liu, R.H. Comparison of phytochemical profiles, antioxidant and cellular antioxidant activities of different varieties of blueberry (Vaccinium spp.). Food Chem. 2017, 217, 773–781. [Google Scholar] [CrossRef]
- Adom, K.K.; Liu, R.H. Rapid peroxyl radical scavenging capacity (PSC) assay for assessing both hydrophilic and lipophilic antioxidants. J. Agric. Food Chem. 2005, 53, 6572–6580. [Google Scholar] [CrossRef] [PubMed]
- Díaz-Mula, H.M.; Zapata, P.J.; Guillén, F.; Martínez-Romero, D.; Castillo, S.; Serrano, M.; Valero, D. Changes in hydrophilic and lipophilic antioxidant activity and related bioactive compounds during postharvest storage of yellow and purple plum cultivars. Postharvest Biol. Technol. 2009, 51, 354–363. [Google Scholar] [CrossRef]
- Kalinowska, M.; Bielawska, A.; Lewandowska-Siwkiewicz, H.; Priebe, W.; Lewandowski, W. Apples: Content of phenolic compounds vs. variety, part of apple and cultivation model, extraction of phenolic compounds, biological properties. Plant Physiol. Biochem. 2014, 84, 169–188. [Google Scholar] [CrossRef]
- Ma, T.; Sun, X.; Zhao, J.; You, Y.; Lei, Y.; Gao, G.; Zhan, J. Nutrient compositions and antioxidant capacity of kiwifruit (Actinidia) and their relationship with flesh color and commercial value. Food Chem. 2017, 218, 294–304. [Google Scholar] [CrossRef]
- Serrano, M.A.; Martínez-Romero, D.; Castillo, S.; Guillén, F.; Valero, D. Role of calcium and heat treatments in alleviating physiological changes induced by mechanical damage in plum. Postharvest Biol. Technol. 2004, 34, 155–167. [Google Scholar] [CrossRef]
- Luo, Z.; Zhang, L.; Xu, T.; Xie, J.; Xi, Y. Effect of hot-air treatment on the ripening of ‘Qingnai’ plum (Prunus salicina Lindl.). J. Hortic. Sci. Biotechnol. 2010, 85, 12–16. [Google Scholar] [CrossRef]
- Zhang, L.; Yu, Z.; Jiang, L.; Jiang, J.; Luo, H.; Fu, L. Effect of post-harvest heat treatment on proteome change of peach fruit during ripening. J. Proteom. 2011, 74, 1135–1149. [Google Scholar] [CrossRef] [PubMed]
- Kumar, P.; Sethi, S.; Sharma, R.R.; Srivastav, M.; Varghese, E. Effect of chitosan coating on postharvest life and quality of plum during storage at low temperature. Sci. Hortic. 2017, 226, 104–109. [Google Scholar] [CrossRef]
- Petriccione, M.; De Sanctis, F.; Pasquariello, M.S.; Mastrobuoni, F.; Rega, P.; Scortichini, M.; Mencarelli, F. The Effect of Chitosan Coating on the Quality and Nutraceutical Traits of Sweet Cherry During Postharvest Life. Food Bioprocess Technol. 2015, 8, 394–408. [Google Scholar] [CrossRef]
- Petriccione, M.; Pasquariello, M.S.; Mastrobuoni, F.; Zampella, L.; Di Patre, D.; Scortichini, M. Influence of a chitosan coating on the quality and nutraceutical traits of loquat fruit during postharvest life. Sci. Hortic. 2015, 197, 287–296. [Google Scholar] [CrossRef]
- Jongsri, P.; Wangsomboondee, T.; Rojsitthisak, P.; Seraypheap, K. Effect of molecular weights of chitosan coating on postharvest quality and physicochemical characteristics of mango fruit. LWT 2016, 73, 28–36. [Google Scholar] [CrossRef]
- Kou, X.; Li, Y.; Wu, J.; Chen, Q.; Xue, Z. Effects of Edible Coatings on Quality and Antioxidant Activity of Zizyphus Jujuba Miller cv. Dongzao During Storage. Trans. Tianjin Univ. 2017, 23, 51–61. [Google Scholar] [CrossRef]
- Nair, M.S.; Saxena, A.; Kaur, C. Effect of chitosan and alginate based coatings enriched with pomegranate peel extract to extend the postharvest quality of guava (Psidium guajava L.). Food Chem. 2018, 240, 245–252. [Google Scholar] [CrossRef]
| Phytochemicals | Treatments | Storage Time | |||||
|---|---|---|---|---|---|---|---|
| 0 d | 8 d | 16 d | 24 d | 32 d | 40 d | ||
| Epicatechin | Control | 4.76 ± 0.12 a | 13.50 ± 0.99 b | 9.93 ± 0.43 b | 11.31 ± 0.96 b | 10.46 ± 1.13 a | 11.00 ± 0.83 a |
| Heat | 4.76 ± 0.12 a | 11.35 ± 1.06 c | 9.90 ± 1.17 b | 5.08 ± 0.48 c | 5.42 ± 0.47 b | 7.16 ± 0.09 b | |
| Heat and chitosan | 4.76 ± 0.12 a | 42.17 ± 1.02 a | 38.54 ± 3.63 a | 36.79 ± 2.55 a | 9.00 ± 1.01 a | 2.91 ± 0.78 c | |
| Myricetin | Control | 0.53 ± 0.04 a | 0.84 ± 0.08 b | 0.73 ± 0.08 b | 0.96 ± 0.09 a | 0.85 ± 0.07 a | 0.66 ± 0.03 b |
| Heat | 0.53 ± 0.04 a | 0.31 ± 0.02 c | 0.62 ± 0.05 b | 0.72 ± 0.02 b | 1.00 ± 0.11 a | 1.03 ± 0.14 a | |
| Heat and chitosan | 0.53 ± 0.04 a | 1.06 ± 0.04 a | 0.88 ± 0.04 a | 0.78 ± 0.03 b | 0.54 ± 0.06 b | 0.59 ± 0.04 b | |
| Hesperetin | Control | 0.027 ± 0.002 a | 0.036 ± 0.004 b | 0.030 ± 0.001 c | 0.031 ± 0.002 b | 0.030 ± 0.003 c | 0.030 ± 0.006 b |
| Heat | 0.027 ± 0.002 a | 0.022 ± 0.002 c | 0.022 ± 0.001 b | 0.034 ± 0.002 b | 0.055 ± 0.001 a | 0.039 ± 0.001 a | |
| Heat and chitosan | 0.027 ± 0.002 a | 0.053 ± 0.001a | 0.046 ± 0.001 a | 0.044 ± 0.002 a | 0.046 ± 0.005 b | 0.039 ± 0.002 a | |
| Cyanidin | Control | 0.74 ± 0.06 a | 0.70 ± 0.08 c | 1.11 ± 0.07 b | 1.55 ± 0.19 b | 1.65 ± 0.18 a | 1.28 ± 0.06 a |
| Heat | 0.74 ± 0.06 a | 0.93 ± 0.10 b | 0.92 ± 0.09 b | 0.75 ± 0.06 c | 0.74 ± 0.05 c | 0.73 ± 0.06 c | |
| Heat and chitosan | 0.74 ± 0.06 a | 1.38 ± 0.11 a | 1.88 ± 0.19 a | 2.39 ± 0.10 a | 1.00 ± 0.10 b | 1.06 ± 0.06 b | |
| Pelargonidin | Control | 0.105 ± 0.002 a | 0.089 ± 0.000 b | 0.106 ± 0.017 a | 0.116 ± 0.014 b | 0.123 ± 0.020 a | 0.111 ± 0.014 a |
| Heat | 0.105 ± 0.002 a | 0.108 ± 0.010 ab | 0.097 ± 0.004 a | 0.109 ± 0.003 b | 0.138 ± 0.011 a | 0.096 ± 0.004 a | |
| Heat and chitosan | 0.105 ± 0.002 a | 0.119 ± 0.020 a | 0.124 ± 0.021 a | 0.144 ± 0.014 a | 0.108 ± 0.010 a | 0.108 ± 0.014 a | |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chang, X.; Lu, Y.; Li, Q.; Lin, Z.; Qiu, J.; Peng, C.; Brennan, C.S.; Guo, X. The Combination of Hot Air and Chitosan Treatments on Phytochemical Changes during Postharvest Storage of ‘Sanhua’ Plum Fruits. Foods 2019, 8, 338. https://doi.org/10.3390/foods8080338
Chang X, Lu Y, Li Q, Lin Z, Qiu J, Peng C, Brennan CS, Guo X. The Combination of Hot Air and Chitosan Treatments on Phytochemical Changes during Postharvest Storage of ‘Sanhua’ Plum Fruits. Foods. 2019; 8(8):338. https://doi.org/10.3390/foods8080338
Chicago/Turabian StyleChang, Xiaoxiao, Yusheng Lu, Quan Li, Zhixiong Lin, Jishui Qiu, Cheng Peng, Charles Stephen Brennan, and Xinbo Guo. 2019. "The Combination of Hot Air and Chitosan Treatments on Phytochemical Changes during Postharvest Storage of ‘Sanhua’ Plum Fruits" Foods 8, no. 8: 338. https://doi.org/10.3390/foods8080338






