Effects of Calcium and Manganese on Sporulation of Bacillus Species Involved in Food Poisoning and Spoilage
Abstract
1. Introduction
2. Materials and Methods
2.1. Microorganisms
2.2. Preparation of Vegetative Cell Suspension
2.3. Preparation of Sporulation Media with Cations
2.4. Induction of Sporulation
2.5. Enumeration of Spores
2.6. Statistical Analysis
3. Results and Discussion
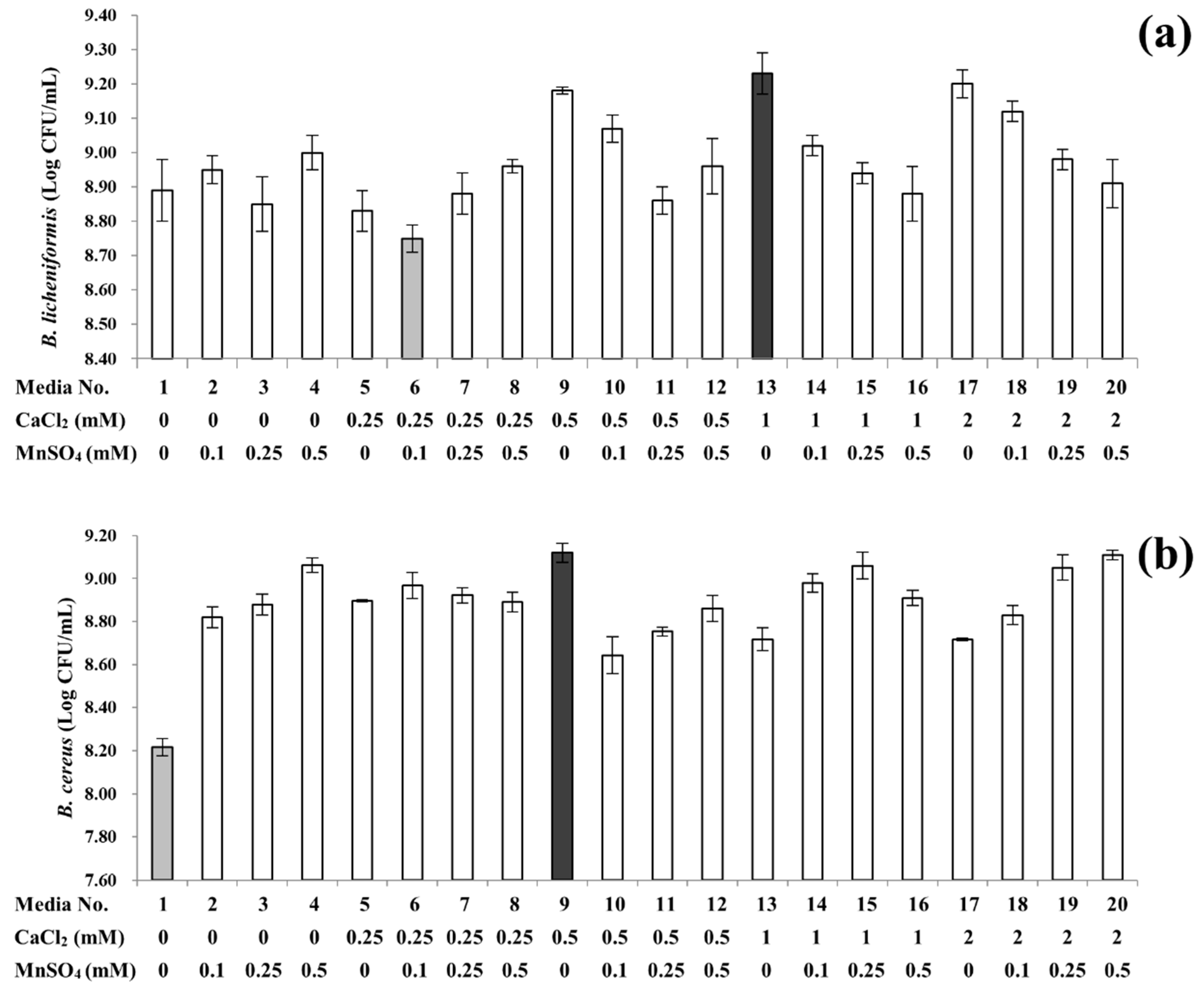
3.1. Sporulation Profiles of B. licheniformis and B. cereus
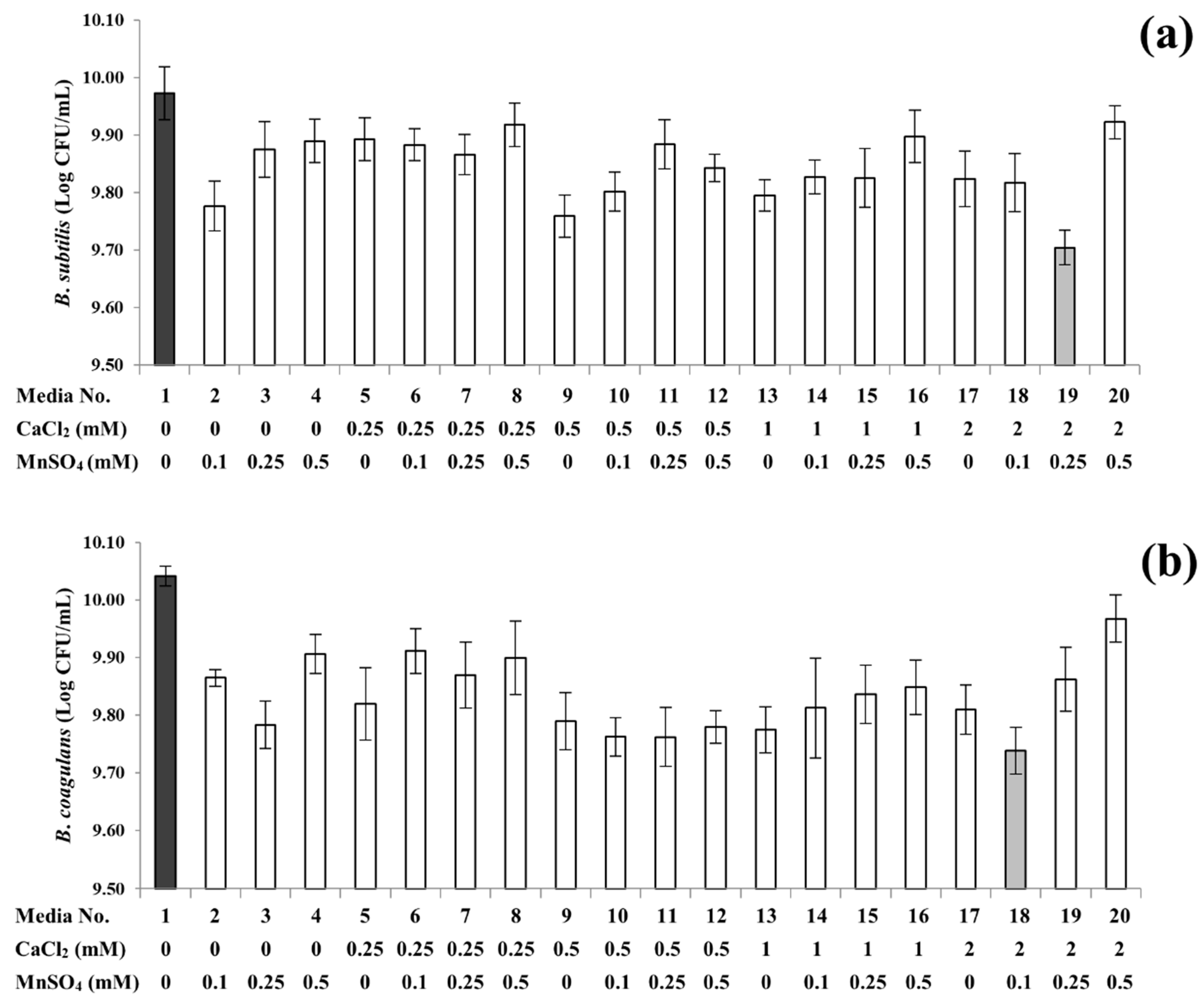
3.2. Sporulation Profiles of B. subtilis and B. coagulans
3.3. Further Implications
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Slepecky, R.A.; Hemphill, H.E. The genus Bacillus-nonmedical. In The Prokaryotes, 3rd ed.; Dworkin, M., Falkow, S., Rosenberg, E., Schleifer, K.H., Stackebrandt, E., Eds.; Springer: Berlin, Germany, 2006; Volume 4, pp. 530–562. [Google Scholar]
- Stöckel, S.; Meisel, S.; Böhme, R.; Elschner, M.; Rösch, P.; Popp, J. Effect of supplementary manganese on the sporulation of Bacillus endospores analysed by Raman spectroscopy. J. Raman Spectrosc. 2009, 40, 1469–1477. [Google Scholar] [CrossRef]
- Gopal, N.; Hill, C.; Ross, P.R.; Beresford, T.P.; Fenelon, M.A.; Cotter, P.D. The prevalence and control of Bacillus and related spore-forming bacteria in the dairy industry. Front. Microbiol. 2015, 6, 1418. [Google Scholar] [CrossRef] [PubMed]
- Salkinoja-Salonen, M.S.; Vuorio, R.; Andersson, M.A.; Kämpfer, P.; Andersson, M.C.; Honkanen-Buzalski, T.; Scoging, A.C. Toxigenic strains of Bacillus licheniformis related to food poisoning. Appl. Environ. Microbiol. 1999, 65, 4637–4645. [Google Scholar] [PubMed]
- Cazemier, A.E.; Wagenaars, S.F.M.; ter Steeg, P.F. Effect of sporulation and recovery medium on the heat resistance and amount of injury of spores from spoilage bacilli. J. Appl. Microbiol. 2001, 90, 761–770. [Google Scholar] [CrossRef] [PubMed]
- Nicholson, W.L.; Munakata, N.; Horneck, G.; Melosh, H.J.; Setlow, P. Resistance of Bacillus endospores to extreme terrestrial and extraterrestrial environments. Microbiol. Mol. Biol. Rev. 2000, 64, 548–572. [Google Scholar] [CrossRef] [PubMed]
- Ryu, J.-H.; Kim, H.; Beuchat, L.R. Spore formation by Bacillus cereus in broth as affected by temperature, nutrient availability, and manganese. J. Food Prot. 2005, 68, 1734–1738. [Google Scholar] [CrossRef] [PubMed]
- Cronin, U.P.; Wilkinson, M.G. Bacillus cereus endospores exhibit a heterogeneous response to heat treatment and low-temperature storage. Food Microbiol. 2008, 25, 235–243. [Google Scholar] [CrossRef] [PubMed]
- Leggett, M.J.; McDonnell, G.; Denyer, S.P.; Setlow, P.; Maillard, J.-Y. Bacterial spore structures and their protective role in biocide resistance. J. Appl. Microbiol. 2012, 113, 485–498. [Google Scholar] [CrossRef] [PubMed]
- Bassi, D.; Colla, F.; Gazzola, S.; Puglisi, E.; Delledonne, M.; Cocconcelli, P.S. Transcriptome analysis of Bacillus thuringiensis spore life, germination and cell outgrowth in a vegetable-based food model. Food Microbiol. 2016, 55, 73–85. [Google Scholar] [CrossRef] [PubMed]
- Slepecky, R.; Foster, J.W. Alterations in metal content of spores of Bacillus megaterium and the effect of some spore properties. J. Bacteriol. 1959, 78, 117–123. [Google Scholar] [PubMed]
- Sella, S.R.B.R.; Vandenberghe, L.P.S.; Soccol, C.R. Life cycle and spore resistance of spore-forming Bacillus atrophaeus. Microbiol. Res. 2014, 169, 931–939. [Google Scholar] [CrossRef]
- Charney, J.; Fisher, W.P.; Hegarty, C.P. Manganese as an essential element for sporulation in the genus Bacillus. J. Bacteriol. 1951, 62, 145–148. [Google Scholar]
- Kolodziej, B.J.; Slepecky, R.A. Trace metal requirements for sporulation of Bacillus megaterium. J. Bacteriol. 1964, 88, 821–830. [Google Scholar]
- Weinberg, E.D. Manganese requirement for sporulation and other secondary biosynthetic processes of Bacillus. Appl. Environ. Microbiol. 1964, 12, 436–441. [Google Scholar]
- Bender, G.R.; Marquis, R.E. Spore heat resistance and specific mineralization. Appl. Environ. Microbiol. 1985, 50, 1414–1421. [Google Scholar] [PubMed]
- Amaha, M.; Ordal, Z.J. Effect of divalent cations in the sporulation medium on the thermal death rate of Bacillus coagulans var. thermoacidurans. J. Bacteriol. 1957, 74, 596–604. [Google Scholar]
- Levinson, H.S.; Hyatt, M.T. Effect of sporulation medium on heat resistance, chemical composition, and germination of Bacillus megaterium spores. J. Bacteriol. 1964, 87, 876–886. [Google Scholar]
- Bai, X. Development of Spore Starter Technology to Control Food-Borne Pathogenic Bacteria in Fermented Soybean Products. Master’s Thesis, Korea University, Sejong, Korea, 2013. [Google Scholar]
- Vasantha, N.; Freese, E. The role of manganese in growth and sporulation of Bacillus subtilis. J. Gen. Microbiol. 1979, 112, 329–336. [Google Scholar] [CrossRef]
- Amaha, M.; Ordal, Z.J.; Touba, A. Sporulation requirements of Bacillus coagulans var. thermoacodurans in complex media. J. Bacteriol. 1956, 72, 34–41. [Google Scholar]
- Oomes, S.J.C.M.; Jonker, M.J.; Wittink, F.R.A.; Hehenkamp, J.O.; Breit, T.M.; Brul, S. The effect of calcium on the transcriptome of sporulating B. subtilis cells. Int. J. Food Microbiol. 2009, 133, 234–242. [Google Scholar] [CrossRef] [PubMed]
- Rey, M.W.; Ramaiya, P.; Nelson, B.A.; Brody-Karpin, S.D.; Zaretsky, E.J.; Tang, M.; Lopez de Leon, A.; Xiang, H.; Gusti, V.; Clausen, I.G.; et al. Complete genome sequence of the industrial bacterium Bacillus licheniformis and comparisons with closely related Bacillus species. Genome Biol. 2004, 5, R77. [Google Scholar] [CrossRef]
- Veitha, B.; Herzberga, C.; Steckel, S.; Feesche, J.; Maurer, K.H.; Ehrenreich, P.; Bäumer, S.; Hennea, A.; Liesegang, H.; Merkl, R.; et al. The complete genome sequence of Bacillus licheniformis DSM13, an organism with great industrial potential. J. Mol. Microbiol. Biotechnol. 2004, 7, 204–211. [Google Scholar] [CrossRef] [PubMed]
- Kung, H.-F.; Tsai, Y.-H.; Wei, C.-I. Histamine and other biogenic amines and histamine-forming bacteria in miso products. Food Chem. 2007, 101, 351–356. [Google Scholar] [CrossRef]
- Troller, J.A. Influence of water activity on microorganisms in foods. Food Technol. 1980, 34, 76–82. [Google Scholar]
- Sofos, J.N. Antimicrobial effects of sodium and other ions in foods: a review. J. Food Saf. 1983, 6, 45–78. [Google Scholar] [CrossRef]
- Nagler, K.; Setlow, P.; Li, Y.-Q.; Moeller, R. High salinity alters the germination behavior of Bacillus subtilis spores with nutrient and nonnutrient germinants. Appl. Environ. Microbiol. 2014, 80, 1314–1321. [Google Scholar] [CrossRef]
- Setlow, P. Mechanisms for the prevention of damage to DNA in spores of Bacillus species. Annu. Rev. Microbiol. 1995, 49, 29–54. [Google Scholar] [CrossRef] [PubMed]
- Setlow, P. Spores of Bacillus subtilis: their resistance to and killing by radiation, heat and chemicals. J. Appl. Microbiol. 2006, 101, 514–525. [Google Scholar] [CrossRef]
- Lee, M.Y.; Park, S.-Y.; Jung, K.-O.; Park, K.-Y.; Kim, S.D. Quality and functional characteristics of chungkukjang prepared with various Bacillus sp. isolated from traditional chungkukjang. J. Food Sci. 2005, 70, M191–M196. [Google Scholar] [CrossRef]
- Kubo, Y.; Rooney, A.P.; Tsukakoshi, Y.; Nakagawa, R.; Hasegawa, H.; Kimura, K. Phylogenetic analysis of Bacillus subtilis strains applicable to natto (fermented soybean) production. Appl. Environ. Microbiol. 2011, 77, 6463–6469. [Google Scholar] [CrossRef]
- Mah, J.-H.; Kang, D.-H.; Tang, J. Morphological study of heat-sensitive and heat-resistant spores of Clostridium sporogenes, using transmission electron microscopy. J. Food Prot. 2008, 71, 953–958. [Google Scholar] [CrossRef]
- Mah, J.-H.; Kang, D.-H.; Tang, J. Effects of minerals on sporulation and heat resistance of Clostridium sporogenes. Int. J. Food Microbiol. 2008, 128, 385–389. [Google Scholar] [CrossRef]
- Mah, J.-H.; Kang, D.-H.; Tang, J. Comparison of viability and heat resistance of Clostridium sporogenes stored at different temperatures. J. Food Sci. 2009, 74, M23–M27. [Google Scholar] [CrossRef]
- Byun, B.Y.; Liu, Y.; Tang, J.; Kang, D.-H.; Cho, H.-Y.; Hwang, H.-J.; Mah, J.-H. Optimization and evaluation of heat-shock condition for spore enumeration being used in thermal-process verification: Differential responses of spores and vegetative cells of Clostridium sporogenes to heat shock. Food Sci. Biotechnol. 2011, 20, 751–757. [Google Scholar] [CrossRef]
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sinnelä, M.T.; Park, Y.K.; Lee, J.H.; Jeong, K.C.; Kim, Y.-W.; Hwang, H.-J.; Mah, J.-H. Effects of Calcium and Manganese on Sporulation of Bacillus Species Involved in Food Poisoning and Spoilage. Foods 2019, 8, 119. https://doi.org/10.3390/foods8040119
Sinnelä MT, Park YK, Lee JH, Jeong KC, Kim Y-W, Hwang H-J, Mah J-H. Effects of Calcium and Manganese on Sporulation of Bacillus Species Involved in Food Poisoning and Spoilage. Foods. 2019; 8(4):119. https://doi.org/10.3390/foods8040119
Chicago/Turabian StyleSinnelä, Martti Tapani, Young Kyoung Park, Jae Hoan Lee, KwangCheol Casey Jeong, Young-Wan Kim, Han-Joon Hwang, and Jae-Hyung Mah. 2019. "Effects of Calcium and Manganese on Sporulation of Bacillus Species Involved in Food Poisoning and Spoilage" Foods 8, no. 4: 119. https://doi.org/10.3390/foods8040119
APA StyleSinnelä, M. T., Park, Y. K., Lee, J. H., Jeong, K. C., Kim, Y.-W., Hwang, H.-J., & Mah, J.-H. (2019). Effects of Calcium and Manganese on Sporulation of Bacillus Species Involved in Food Poisoning and Spoilage. Foods, 8(4), 119. https://doi.org/10.3390/foods8040119





