Smart and Mechanically Enhanced Zein–Gelatin Films Incorporating Cellulose Nanocrystals and Alizarin for Fish Spoilage Monitoring
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. pH-Sensitivities of AL
2.3. Production of Smart Films
2.4. Characterization of Smart Films
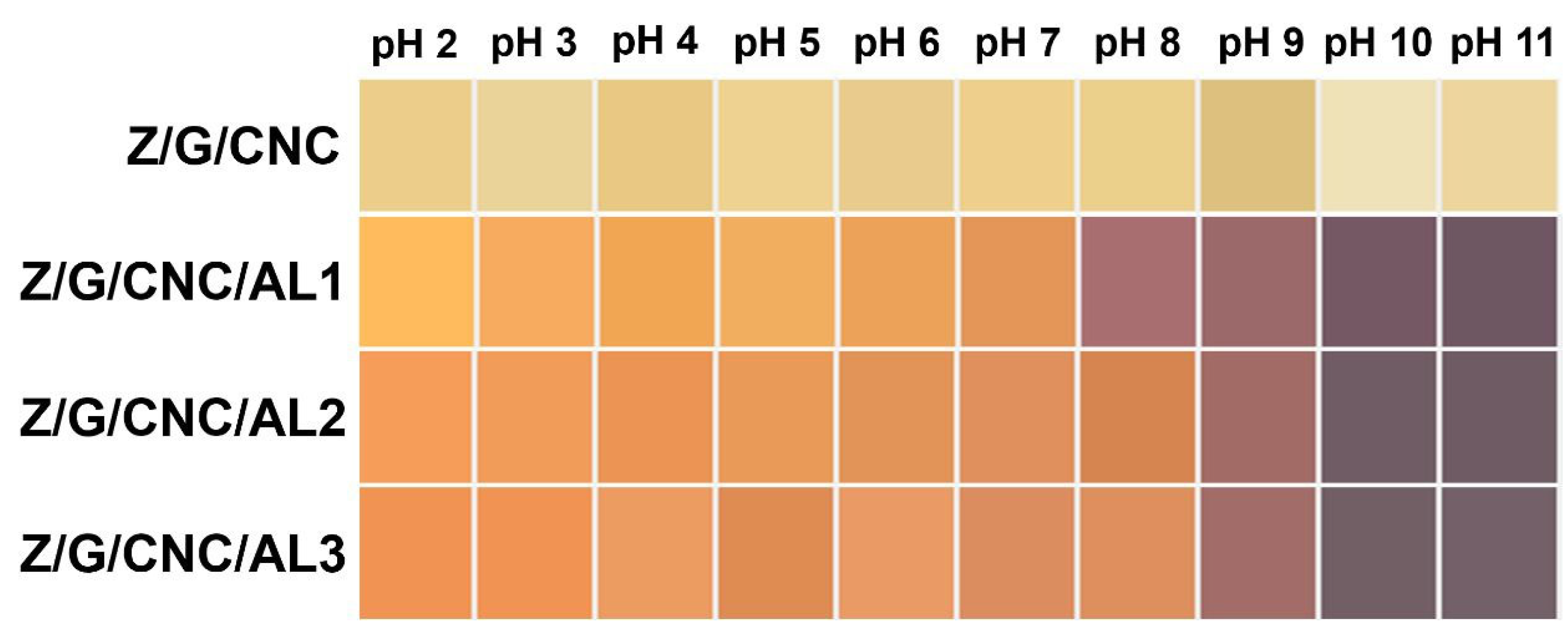
2.5. Color Response of Smart Films Under Different pH Conditions
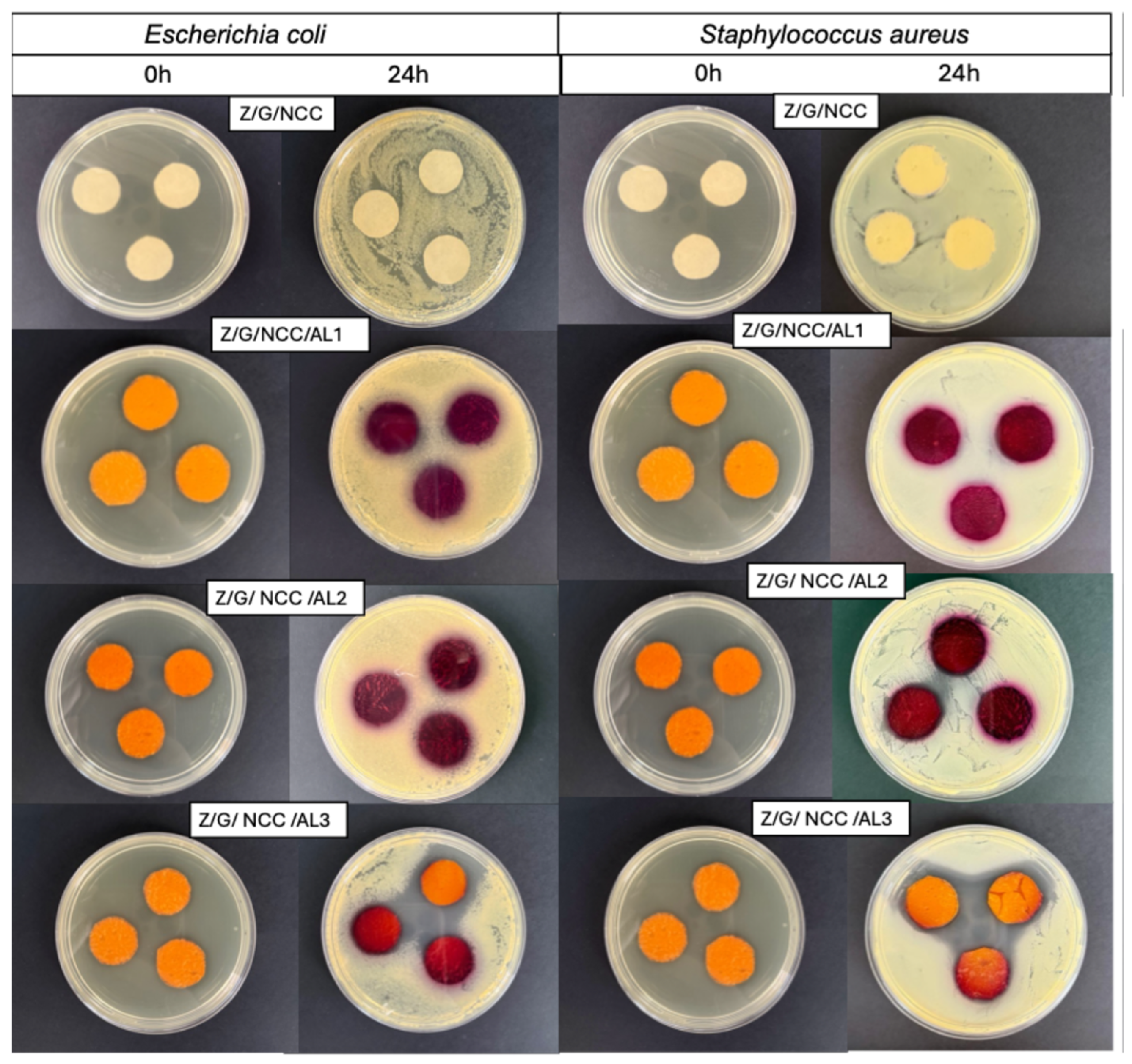
2.6. Microbial Response of Smart Films
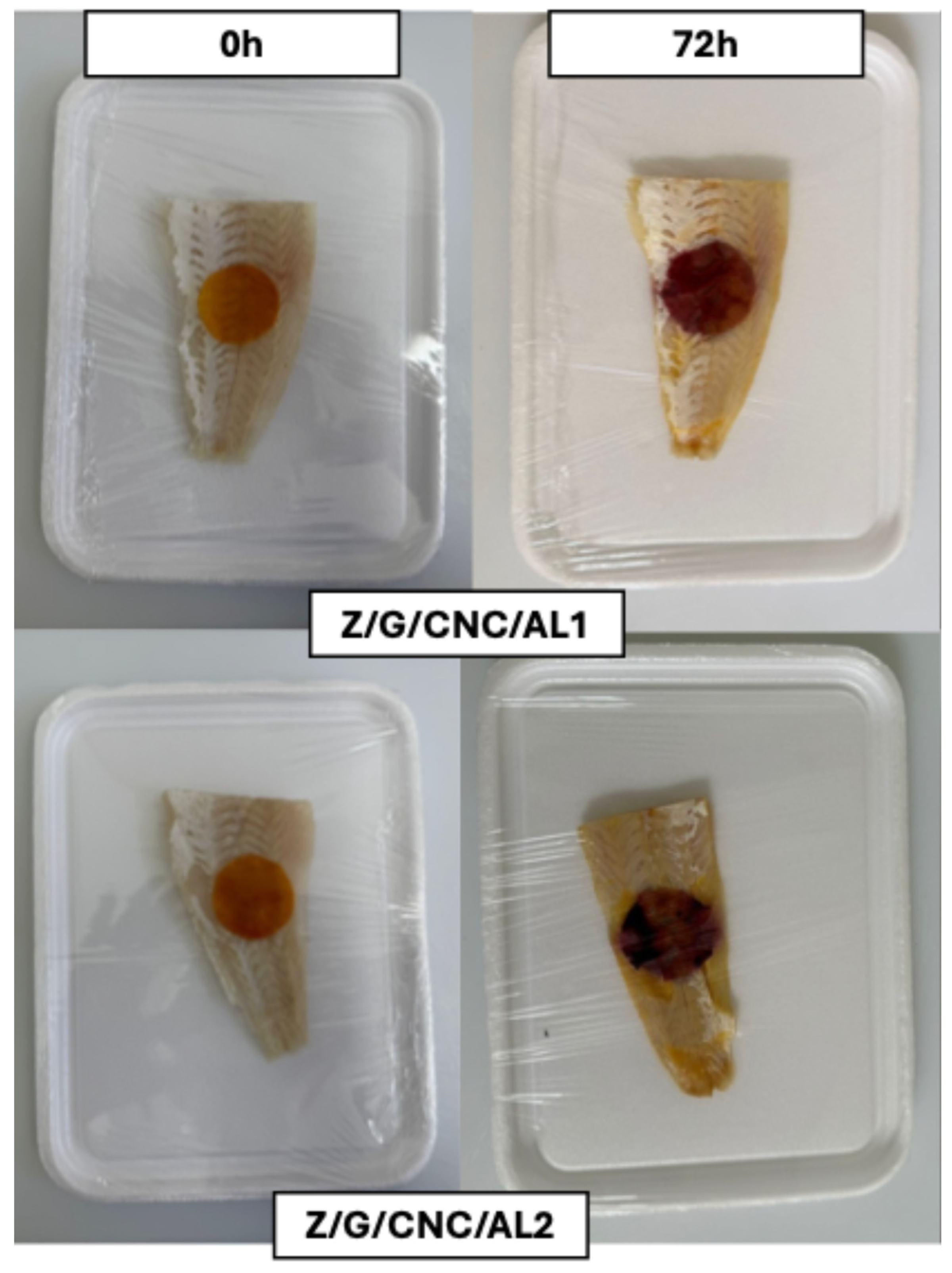
2.7. Application of Smart Films to Monitor Fish Fillet Freshness
2.8. Statistical Analysis
3. Results
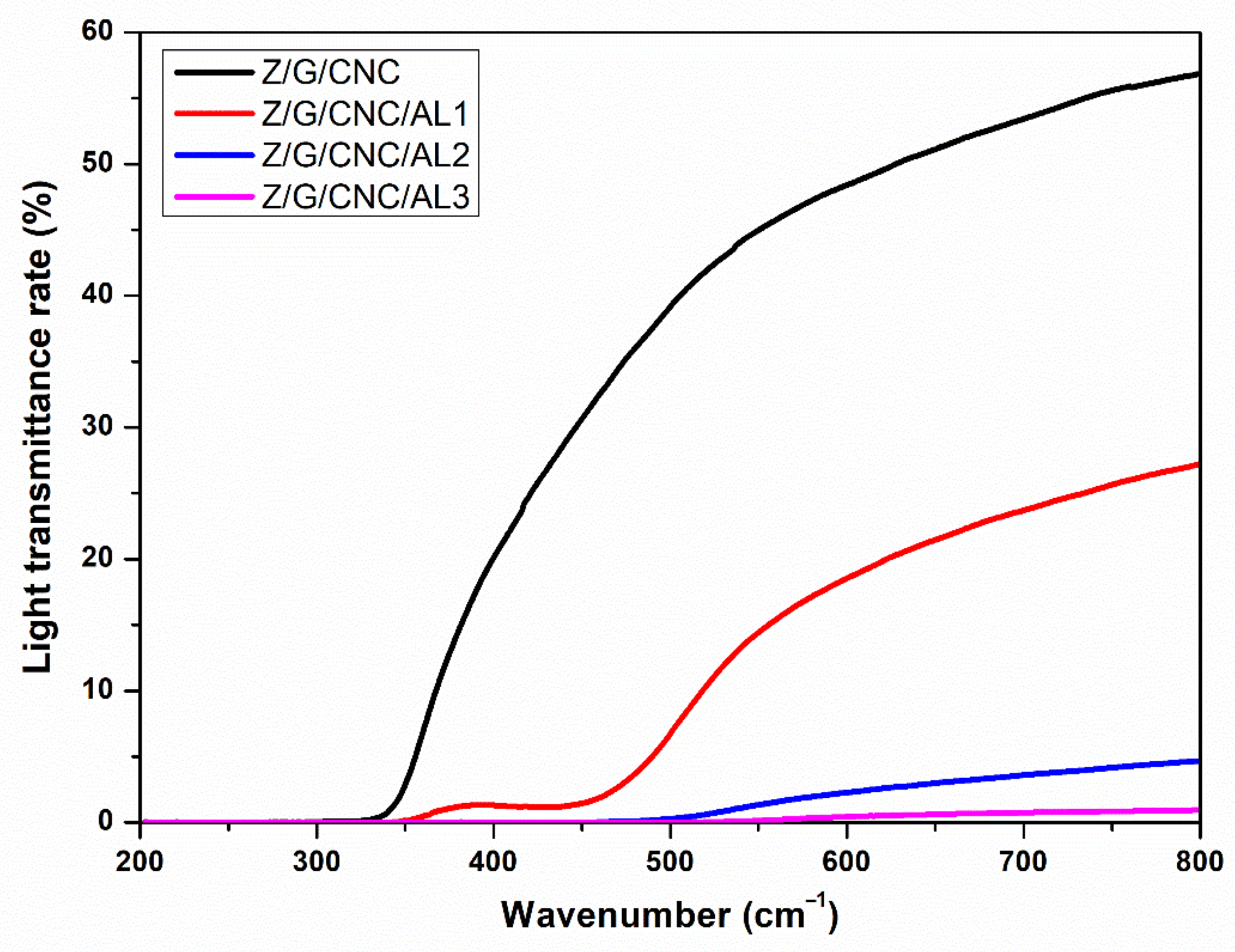
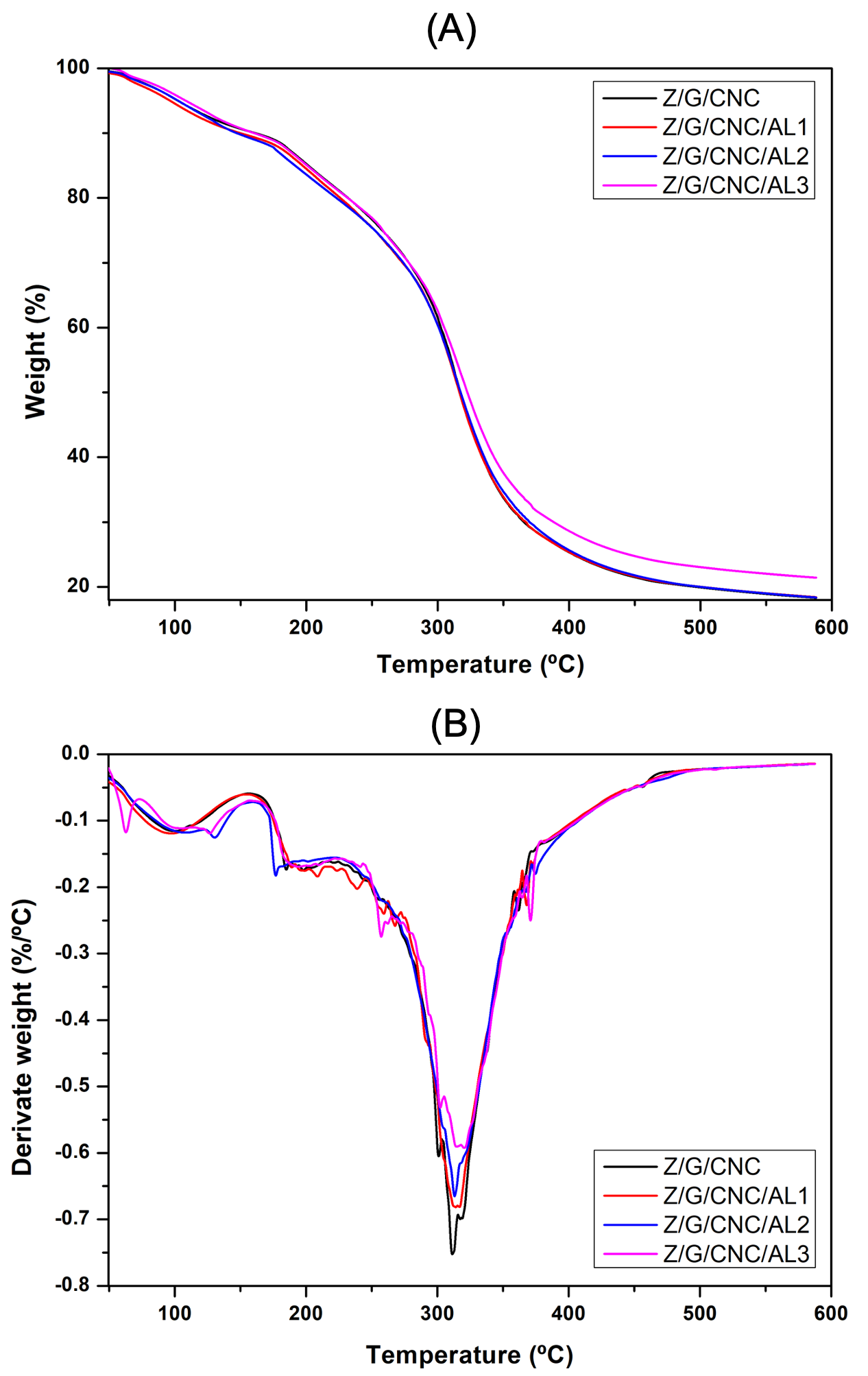
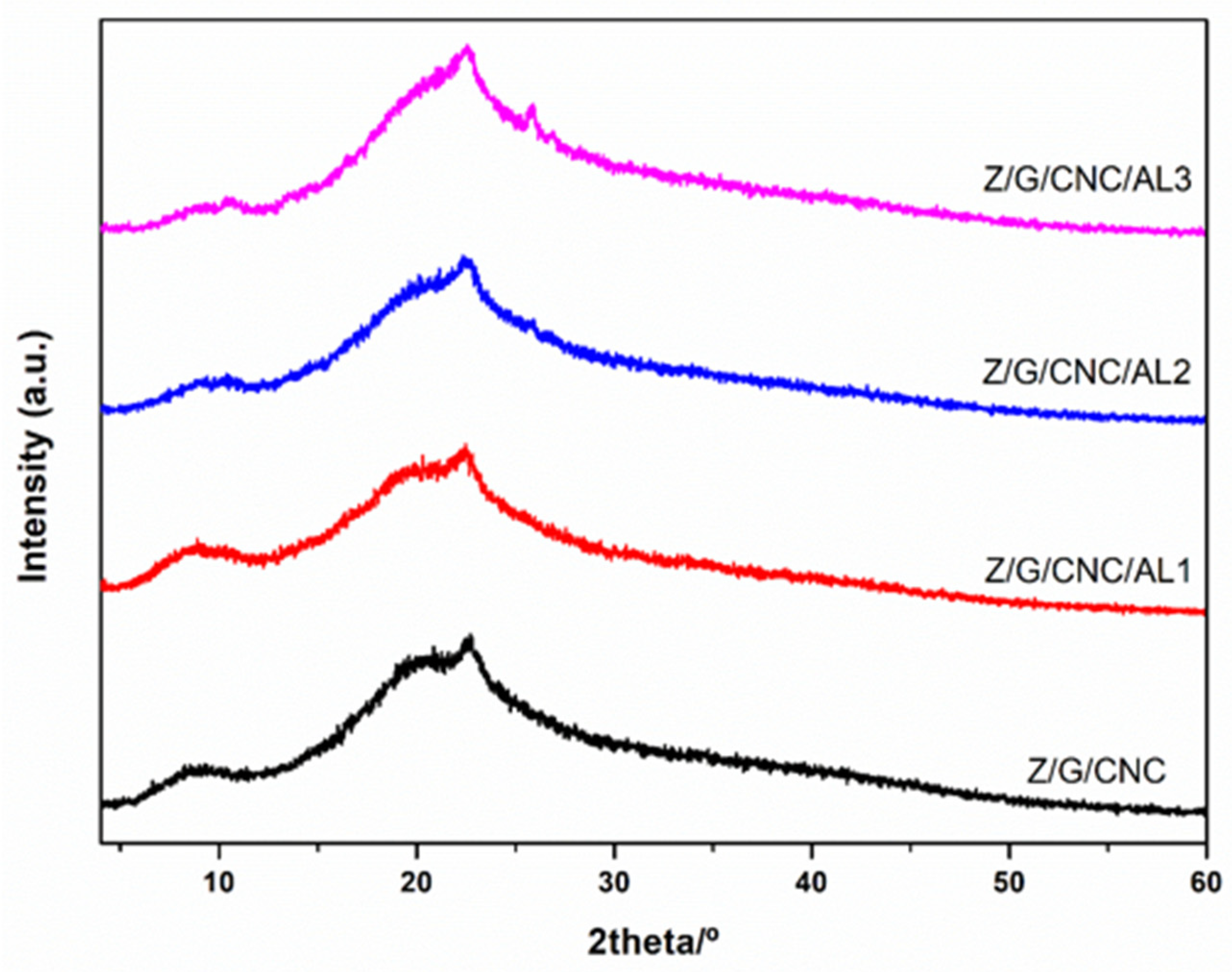
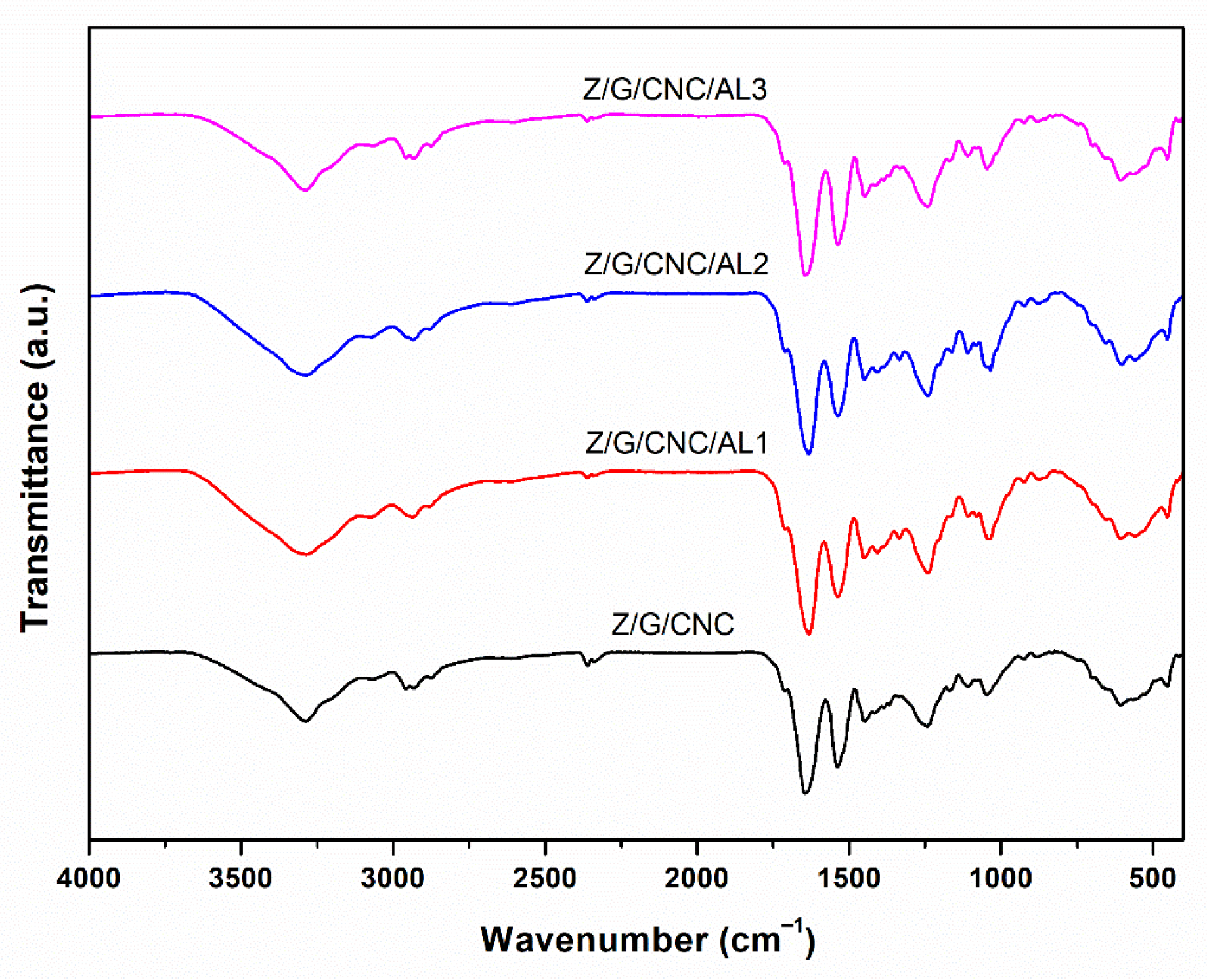
3.1. Film Characterization
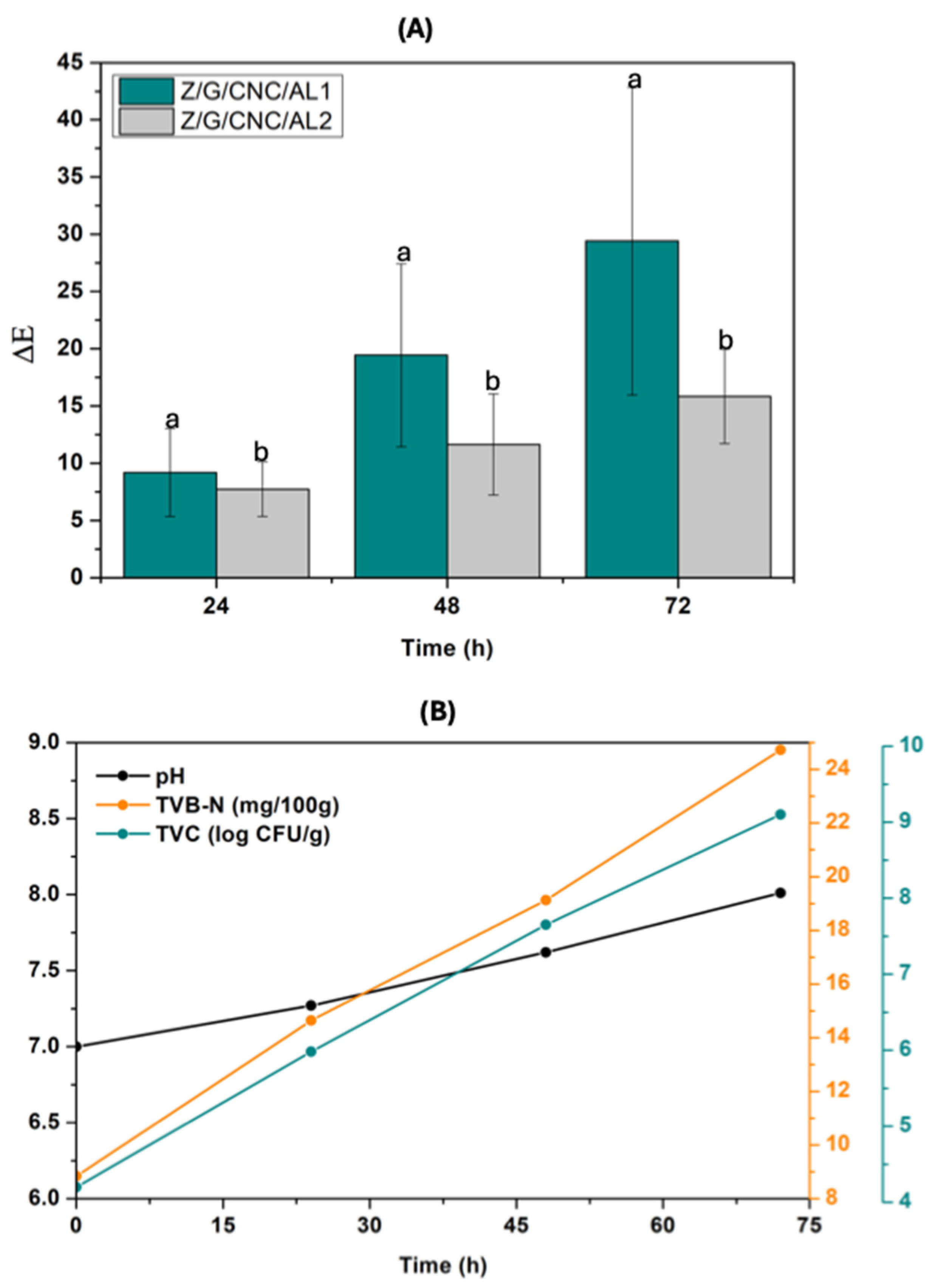
3.2. Application of Smart Films for Monitoring Fish Fillet Quality
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- de Oliveira Filho, J.G.; de Oliveira Silva, C.; Egea, M.B.; Tonon, R.V.; Paschoalin, R.T.; de Oliveira Rios, A.; de Azeredo, H.M.C.; Mattoso, L.H.C. Efficient and sustainable production of intelligent nonwovens as indicators of food spoilage through solution blow spinning of proteins and natural pigments from agri-food waste. Food Control 2024, 166, 110707. [Google Scholar] [CrossRef]
- Albu, C.; Chira, A.; Radu, G.-L.; Eremia, S.A. Advances in Cost-Effective Chemosensors for Sustainable Monitoring in Food Safety and Processing. Chemosensors 2025, 13, 113. [Google Scholar] [CrossRef]
- Mkhari, T.; Adeyemi, J.O.; Fawole, O.A. Recent advances in the fabrication of intelligent packaging for food preservation: A review. Processes 2025, 13, 539. [Google Scholar] [CrossRef]
- Liu, D.; Zhang, C.; Pu, Y.; Chen, S.; Liu, L.; Cui, Z.; Zhong, Y. Recent advances in pH-responsive freshness indicators using natural food colorants to monitor food freshness. Foods 2022, 11, 1884. [Google Scholar] [CrossRef]
- Hosseini, S.F.; Gómez-Guillén, M.C. A state-of-the-art review on the elaboration of fish gelatin as bioactive packaging: Special emphasis on nanotechnology-based approaches. Trends Food Sci. Technol. 2018, 79, 125–135. [Google Scholar] [CrossRef]
- Jiang, L.; Ye, R.; Xie, C.; Wang, F.; Zhang, R.; Tang, H.; He, Z.; Han, J.; Liu, Y. Development of zein edible films containing different catechin/cyclodextrin metal-organic frameworks: Physicochemical characterization, antioxidant stability and release behavior. LWT 2023, 173, 114306. [Google Scholar] [CrossRef]
- Li, L.; Wang, H.; Chen, M.; Jiang, S.; Cheng, J.; Li, X.; Zhang, M.; Jiang, S. Gelatin/zein fiber mats encapsulated with resveratrol: Kinetics, antibacterial activity and application for pork preservation. Food Hydrocoll. 2020, 101, 105577. [Google Scholar] [CrossRef]
- Dogaru, B.-I.; Stoleru, V.; Mihalache, G.; Yonsel, S.; Popescu, M.-C. Gelatin reinforced with CNCs as nanocomposite matrix for Trichoderma harzianum KUEN 1585 spores in seed coatings. Molecules 2021, 26, 5755. [Google Scholar] [CrossRef]
- Leite, L.S.; Ferreira, C.M.; Corrêa, A.C.; Moreira, F.K.; Mattoso, L.H. Scaled-up production of gelatin-cellulose nanocrystal bionanocomposite films by continuous casting. Carbohydr. Polym. 2020, 238, 116198. [Google Scholar] [CrossRef]
- Leite, L.S.; Le Gars, M.; Azeredo, H.M.; Moreira, F.K.; Mattoso, L.H.; Bras, J. Insights into the effect of carboxylated cellulose nanocrystals on mechanical and barrier properties of gelatin films for flexible packaging applications. Int. J. Biol. Macromol. 2024, 280, 135726. [Google Scholar] [CrossRef] [PubMed]
- Jafarzadeh, S.; Yildiz, Z.; Yildiz, P.; Strachowski, P.; Forough, M.; Esmaeili, Y.; Naebe, M.; Abdollahi, M. Advanced technologies in biodegradable packaging using intelligent sensing to fight food waste. Int. J. Biol. Macromol. 2024, 261, 129647. [Google Scholar] [CrossRef]
- Ezati, P.; Rhim, J.-W. pH-responsive chitosan-based film incorporated with alizarin for intelligent packaging applications. Food Hydrocoll. 2020, 102, 105629. [Google Scholar] [CrossRef]
- Kavoosi, G.; Rahmatollahi, A.; Dadfar, S.M.M.; Purfard, A.M. Effects of essential oil on the water binding capacity, physico-mechanical properties, antioxidant and antibacterial activity of gelatin films. LWT-Food Sci. Technol. 2014, 57, 556–561. [Google Scholar] [CrossRef]
- ASTM, A. E96/E96M-16; Standard Test Methods for Water Vapor Transmission of Materials. Annual Book of American Standard Testing Methods. American Society for Testing and Materials: West Conshohocken, PA, USA, 2016.
- Luchese, C.L.; Sperotto, N.; Spada, J.C.; Tessaro, I.C. Effect of blueberry agro-industrial waste addition to corn starch-based films for the production of a pH-indicator film. Int. J. Biol. Macromol. 2017, 104, 11–18. [Google Scholar] [CrossRef]
- de Oliveira Filho, J.G.; de Souza, B.B.; Robles, J.R.; de Azeredo, H.M.C.; Tonon, R.V.; Abiade, J.; Mattoso, L.H.C.; Yarin, A.L. Fast production of highly sensitive nanotextured nonwovens for detection of volatile amines, bacterial growth, and pH monitoring: New tools for real-time food quality monitoring. Food Chem. 2025, 464, 141896. [Google Scholar] [CrossRef]
- Gasti, T.; Dixit, S.; D’souza, O.J.; Hiremani, V.D.; Vootla, S.K.; Masti, S.P.; Chougale, R.B.; Malabadi, R.B. Smart biodegradable films based on chitosan/methylcellulose containing Phyllanthus reticulatus anthocyanin for monitoring the freshness of fish fillet. Int. J. Biol. Macromol. 2021, 187, 451–461. [Google Scholar] [CrossRef]
- Zhai, X.; Shi, J.; Zou, X.; Wang, S.; Jiang, C.; Zhang, J.; Huang, X.; Zhang, W.; Holmes, M. Novel colorimetric films based on starch/polyvinyl alcohol incorporated with roselle anthocyanins for fish freshness monitoring. Food Hydrocoll. 2017, 69, 308–317. [Google Scholar] [CrossRef]
- Khan, A.; Ezati, P.; Rhim, J.-W. Alizarin: Prospects and sustainability for food safety and quality monitoring applications. Colloids Surf. B Biointerfaces 2023, 223, 113169. [Google Scholar] [CrossRef] [PubMed]
- Xia, G.; Ma, Y.; Ma, Q.; Yao, X.; Xu, Z.; Ji, X.; Zhang, F. Anti-ultraviolet, antioxidant and pH-responsive cellulose-based composite film incorporated with alizarin for intelligent packaging applications. Food Packag. Shelf Life 2024, 46, 101413. [Google Scholar] [CrossRef]
- Zhang, D.; Shu, Q.; Liu, Y. The use of novel colorimetric films to monitor the freshness of pork, utilizing konjac glucomannan with curcumin/alizarin. J. Food Prot. 2024, 87, 100339. [Google Scholar] [CrossRef] [PubMed]
- Lin, W.; Zhang, Y.; Huang, J.; Li, Z. pH-responsive double-layer film based on chitosan/curcumin-β-cyclodextrin complex/cinnamaldehyde and zein/alizarin for pork freshness monitoring and maintaining. Food Res. Int. 2023, 173, 113460. [Google Scholar] [CrossRef]
- Ruzi, M.; Celik, N.; Onses, M.S. Superhydrophobic coatings for food packaging applications: A review. Food Packag. Shelf Life 2022, 32, 100823. [Google Scholar] [CrossRef]
- Zou, Y.; Chen, S.; Dou, H.; Zhu, W.; Zhao, D.; Wang, Y.; Wang, H.; Xia, X. Development of a colorimetric nanofibrous film sensor based on polycaprolactone (PCL) incorporated with alizarin for tracking shrimp freshness. Food Packag. Shelf Life 2024, 42, 101249. [Google Scholar] [CrossRef]
- Durand, E.; Laguerre, M.; Bourlieu-Lacanal, C.; Lecomte, J.; Villeneuve, P. Navigating the complexity of lipid oxidation and antioxidation: A review of evaluation methods and emerging approaches. Prog. Lipid Res. 2025, 97, 101317. [Google Scholar] [CrossRef]
- Sheraz, M.A.; Kazi, S.H.; Ahmed, S.; Anwar, Z.; Ahmad, I. Photo, thermal and chemical degradation of riboflavin. Beilstein J. Org. Chem. 2014, 10, 1999–2012. [Google Scholar] [CrossRef] [PubMed]
- Ahammed, S.; Liu, F.; Khin, M.N.; Yokoyama, W.H.; Zhong, F. Improvement of the water resistance and ductility of gelatin film by zein. Food Hydrocoll. 2020, 105, 105804. [Google Scholar] [CrossRef]
- de Oliveira Filho, J.G.; de Sousa, T.L.; Bertolo, M.R.V.; Junior, S.B.; Mattoso, L.H.C.; Pimentel, T.C.; Egea, M.B. Next-generation food packaging: Edible bioactive films with alginate, mangaba pulp (Hancornia speciosa), and Saccharomyces boulardii. Food Biosci. 2023, 54, 102799. [Google Scholar] [CrossRef]
- El-Nahass, M.; Zeyada, H.; El-Ghamaz, N.; Awed, A. Structural investigation, thermal analysis and AC conduction mechanism of thermally evaporated alizarin red S thin films. Optik 2018, 170, 304–313. [Google Scholar] [CrossRef]
- Liu, Z.; Upadhyay, P.; Ullah, A. Enhanced properties of novel canola meal nanocomposite packaging films reinforced with cellulose nanocrystals and glycidyl methacrylate. Food Packag. Shelf Life 2025, 49, 101511. [Google Scholar] [CrossRef]
- Alizadeh-Sani, M.; Tavassoli, M.; Mohammadian, E.; Ehsani, A.; Khaniki, G.J.; Priyadarshi, R.; Rhim, J.-W. pH-responsive color indicator films based on methylcellulose/chitosan nanofiber and barberry anthocyanins for real-time monitoring of meat freshness. Int. J. Biol. Macromol. 2021, 166, 741–750. [Google Scholar] [CrossRef]
- Xiao, J.; Gu, C.; Zhu, D.; Huang, Y.; Luo, Y.; Zhou, Q. Development and characterization of an edible chitosan/zein-cinnamaldehyde nano-cellulose composite film and its effects on mango quality during storage. LWT 2021, 140, 110809. [Google Scholar] [CrossRef]
- Shen, C.; Cao, Y.; Rao, J.; Zou, Y.; Zhang, H.; Wu, D.; Chen, K. Application of solution blow spinning to rapidly fabricate natamycin-loaded gelatin/zein/polyurethane antimicrobial nanofibers for food packaging. Food Packag. Shelf Life 2021, 29, 100721. [Google Scholar] [CrossRef]
- Ki, C.S.; Baek, D.H.; Gang, K.D.; Lee, K.H.; Um, I.C.; Park, Y.H. Characterization of gelatin nanofiber prepared from gelatin–formic acid solution. Polymer 2005, 46, 5094–5102. [Google Scholar] [CrossRef]
- Gao, Y.; Chen, G.; Wu, C.; Zhou, J.; Jin, G.; Yu, H.-Y. Thioctic acid driven chemical cross-linking of cellulose chains for fabricating high-performance cellulose films as food packaging. Int. J. Biol. Macromol. 2025, 318, 145076. [Google Scholar] [CrossRef] [PubMed]
- Chen, Z.; Wen, J.; Huang, S.; Sun, Y.; Liu, X.; Chen, L.; Li, H.; Zhao, P. Highly TVB-N sensitive film with CMS as the ‘bridge’via electrostatic interaction and hydrogen bond self-assembly for monitoring food freshness in intelligent packaging. Talanta 2023, 252, 123881. [Google Scholar] [CrossRef]
- Xia, G.; Ji, X.; Peng, J.; Ji, X. Cellulose/poly (meta-phenylene isophthalamide) light-management films with high antiultraviolet and tunable haze performances. ACS Appl. Polym. Mater. 2022, 4, 8407–8417. [Google Scholar] [CrossRef]
- He, J.; Peng, Q.; Wang, J.; Deng, J.; Li, W.; Lin, Q.; Zhong, F.; Xia, X. An intelligent thymol/alizarin-loaded polycaprolactone/gelatin/zein nanofibrous film with pH-responsive and antibacterial properties for shrimp freshness monitoring and preservation. Food Chem. 2025, 471, 142812. [Google Scholar] [CrossRef]
- Zhang, X.; Chen, X.; Dai, J.; Cui, H.; Lin, L. A pH indicator film based on dragon fruit peel pectin/cassava starch and cyanidin/alizarin for monitoring the freshness of pork. Food Packag. Shelf Life 2023, 40, 101215. [Google Scholar] [CrossRef]
- Balakrishnan, N.K.; Ostheller, M.-E.; Aldeghi, N.; Schmitz, C.; Groten, R.; Seide, G. Pilot-scale electrospinning of PLA using biobased dyes as multifunctional additives. Polymers 2022, 14, 2989. [Google Scholar] [CrossRef]
- Frank, D.; Hughes, J.; Piyasiri, U.; Zhang, Y.; Kaur, M.; Li, Y.; Mellor, G.; Stark, J. Volatile and non-volatile metabolite changes in 140-day stored vacuum packaged chilled beef and potential shelf life markers. Meat Sci. 2020, 161, 108016. [Google Scholar] [CrossRef]


| Treatments | WS (%) | WVP (g·mm/kPa-h-m2) | WCA (°) |
|---|---|---|---|
| Z/G/CNC | 80.25 ± 5.36 a | 2.76 ± 1.21 b | 67.39 ± 4.58 c |
| Z/G/CNC/AL1 | 63.09 ± 2.40 b | 3.97 ± 1.13 a | 97.84 ± 8.56 a |
| Z/G/CNC/AL2 | 64.09 ± 3.20 b | 3.05 ± 1.63 ab | 81.58 ± 8.04 b |
| Z/G/CNC/AL3 | 60.73 ± 1.14 c | 3.15 ± 2.44 a | 72.48 ± 5.77 c |
| Treatments | L | h (°) | C (*) | ΔE |
|---|---|---|---|---|
| Z/G/CNC | 87.39 ± 1.80 a | 93.91 ± 0.87 a | 19.87 ± 5.60 d | - |
| Z/G/CNC/AL1 | 71.30 ± 4.16 b | 76.31 ± 4.87 b | 76.45 ± 2.25 a | 86.09 ± 2.71 a |
| Z/G/CNC/AL2 | 61.12 ± 1.32 c | 60.28 ± 1.99 c | 69.92 ± 3.11 b | 81.58 ± 2.80 b |
| Z/G/CNC/AL3 | 57.03 ± 2.12 d | 55.01 ± 2.27 d | 66.48 ± 3.21 c | 79.58 ± 2.37 c |
| Treatments | Thickness (mm) | Tensile Strength (MPa) | Elongation at Break (%) | Young’s Modulus (%) | Tonset (°C) | Tmax (°C) |
|---|---|---|---|---|---|---|
| Z/G/CNC | 0.19 ± 0.02 c | 61.5 ± 13.72 a | 9.75 ± 2.01 c | 52.61 ± 12.02 a | 195.54 | 316.12 |
| Z/G/CNC/AL1 | 0.18 ± 0.04 c | 36.78 ± 5.45 c | 20.17 ± 2.75 b | 19.81 ± 6.71 c | 193.31 | 317.24 |
| Z/G/CNC/AL2 | 0.22 ± 0.04 b | 39.81 ± 5.23 b | 18.64 ± 6.88 b | 32.16 ± 5.90 b | 185.05 | 317.24 |
| Z/G/CNC/AL3 | 0.26 ± 0.04 a | 24.56 ± 12.72 d | 23.23 ± 3.37 a | 18.04 ± 3.96 c | 194.43 | 324.23 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sentanin, L.; Oliveira Filho, J.G.d.; Egea, M.B.; Mattoso, L.H.C. Smart and Mechanically Enhanced Zein–Gelatin Films Incorporating Cellulose Nanocrystals and Alizarin for Fish Spoilage Monitoring. Foods 2025, 14, 3015. https://doi.org/10.3390/foods14173015
Sentanin L, Oliveira Filho JGd, Egea MB, Mattoso LHC. Smart and Mechanically Enhanced Zein–Gelatin Films Incorporating Cellulose Nanocrystals and Alizarin for Fish Spoilage Monitoring. Foods. 2025; 14(17):3015. https://doi.org/10.3390/foods14173015
Chicago/Turabian StyleSentanin, Leonardo, Josemar Gonçalves de Oliveira Filho, Mariana Buranelo Egea, and Luiz Henrique Capparelli Mattoso. 2025. "Smart and Mechanically Enhanced Zein–Gelatin Films Incorporating Cellulose Nanocrystals and Alizarin for Fish Spoilage Monitoring" Foods 14, no. 17: 3015. https://doi.org/10.3390/foods14173015
APA StyleSentanin, L., Oliveira Filho, J. G. d., Egea, M. B., & Mattoso, L. H. C. (2025). Smart and Mechanically Enhanced Zein–Gelatin Films Incorporating Cellulose Nanocrystals and Alizarin for Fish Spoilage Monitoring. Foods, 14(17), 3015. https://doi.org/10.3390/foods14173015










