Cold-Pressed Oilseed Cakes as Alternative and Sustainable Feed Ingredients: A Review
Abstract
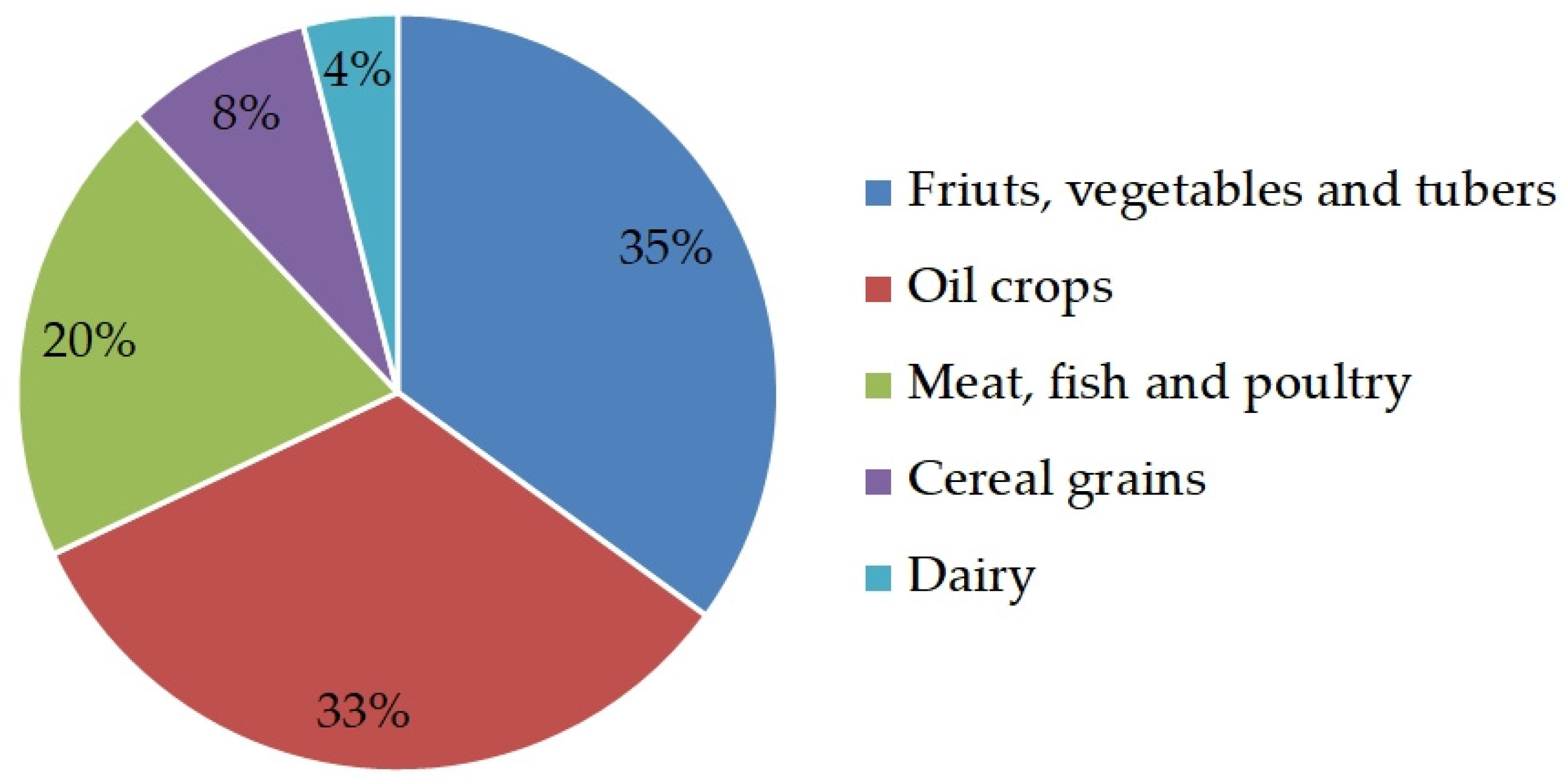
1. Introduction
2. Oilseed By-Products: General Features
3. Literature Review Methodology
4. Nutritional Composition of Cold-Pressed Oilseed Cakes
5. Cold-Pressed Oilseed Cakes in Ruminant Nutrition
5.1. Effects on In Vitro, In Vivo, and In Situ Parameters
5.2. Effects on Animal Production Performance
5.3. Effects on FA Profile of Milk and Meat
5.4. Effects on Other Parameters
6. Cold-Pressed Oilseed Cakes in Pig Nutrition
6.1. Effects on Nutrient Digestibility
6.2. Effects on Growth Performance
6.3. Effects on Other Parameters
7. Cold-Pressed Oilseed Cakes in Poultry Nutrition
7.1. Effects on Growth Performance
7.2. Effects on Blood Parameters
7.3. Effects on Quality and FA Profile of Egg and Meat
8. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Hessle, A.; Eriksson, M.; Nadeau, E.; Turner, T.; Johansson, B. Cold-pressed hempseed cake as a protein feed for growing cattle. Acta Agric. Scand. Sect. A—Anim. Sci. 2008, 58, 136–145. [Google Scholar] [CrossRef]
- Karlsson, L.; Finell, M.; Martinsson, K. Effects of increasing amounts of hempseed cake in the diet of dairy cows on the production and composition of milk. Animal 2010, 4, 1854–1860. [Google Scholar] [CrossRef] [PubMed]
- Ancuța, P.; Sonia, A. Oil Press-Cakes and Meals Valorization through Circular Economy Approaches: A Review. Appl. Sci. 2020, 10, 7432. [Google Scholar] [CrossRef]
- Rao, M.; Bast, A.; de Boer, A. Valorized Food Processing By-Products in the EU: Finding the Balance between Safety, Nutrition, and Sustainability. Sustainability 2021, 13, 4428. [Google Scholar] [CrossRef]
- Petraru, A.; Ursachi, F.; Amariei, S. Nutritional Characteristics Assessment of Sunflower Seeds, Oil and Cake. Perspective of Using Sunflower Oilcakes as a Functional Ingredient. Plants 2021, 10, 2487. [Google Scholar] [CrossRef]
- Švarc-Gajić, J.; Morais, S.; Delerue-Matos, C.; Vieira, E.F.; Spigno, G. Valorization Potential of Oilseed Cakes by Subcritical Water Extraction. Appl. Sci. 2020, 10, 8815. [Google Scholar] [CrossRef]
- Rodríguez-Roque, M.J.; Sánchez-Vega, R.; Pérez-Leal, R.; Soto-Caballero, M.C.; Salas-Salazar, N.A.; Flores-Córdova, M.A. By-Products from Oilseed Processing and Their Potential Applications. In Oil and Oilseed Processing; Lafarga, T., Bobo, G., Aguiló-Aguayo, I., Eds.; John and Wiley and Sons: Hoboken, NJ, USA, 2021; pp. 183–201. [Google Scholar]
- Vidal, N.P.; Roman, L.; Swaraj, V.J.S.; Ragavan, K.V.; Simsek, S.; Rahimi, J.; Kroetsch, B.; Martinez, M.M. Enhancing the nutritional value of cold-pressed oilseed cakes through extrusion cooking. Innov. Food Sci. Emerg. Technol. 2022, 77, 102956. [Google Scholar] [CrossRef]
- Cakaloglu, B.; Ozyurt, V.H.; Otles, S. Cold press in oil extraction. A review. Ukr. Food J. 2018, 7, 640–654. [Google Scholar] [CrossRef]
- Woyengo, T.A.; Sánchez, J.E.; Yáñez, J.; Beltranena, E.; Cervantes, M.; Morales, A.; Zijlstra, R.T. Nutrient digestibility of canola co-products for grower pigs. Anim. Feed. Sci. Technol. 2016, 222, 7–16. [Google Scholar] [CrossRef]
- Roy, A.; Deshmukh, R. Cold-Pressed Oil Market: Global Opportunity Analysis and Industry Forecast, 2019–2026. Allied Market Research. Available online: https://www.alliedmarketresearch.com/cold-pressed-oil-market (accessed on 27 November 2022).
- Goiri, I.; Zubiria, I.; Lavín, J.L.; Benhissi, H.; Atxaerandio, R.; Ruiz, R.; Mandaluniz, N.; García-Rodríguez, A. Evaluating the Inclusion of Cold-Pressed Rapeseed Cake in the Concentrate for Dairy Cows upon Ruminal Biohydrogenation Process, Ruminal Microbial Community and Milk Production and Acceptability. Animals 2021, 11, 2553. [Google Scholar] [CrossRef]
- Pojić, M.; Mišan, A.; Tiwari, B. Eco-innovative technologies for extraction of proteins for human consumption from renewable protein sources of plant origin. Trends Food Sci. Technol. 2018, 75, 93–104. [Google Scholar] [CrossRef]
- FEDIOL. Available online: https://www.fediol.eu/web/feed/1011306087/list1187970107/f1.html (accessed on 27 November 2022).
- Boldea, I.M.; Dragomir, C.; Gras, M.A.; Ropota, M. Inclusion of rapeseed and pumpkin seed cakes in diets for Murciano-Granadina goats alters the fatty acid profile of milk. S. Afr. J. Anim. Sci. 2021, 51, 262–270. [Google Scholar] [CrossRef]
- Seneviratne, R.W.; Beltranena, E.; Newkirk, R.W.; Goonewardene, L.A.; Zijlstra, R.T. Processing conditions affect nutrient digestibility of cold-pressed canola cake for grower pigs. J. Anim. Sci. 2011, 89, 2452–2461. [Google Scholar] [CrossRef]
- Budžaki, S.; Strelec, I.; Krnić, M.; Alilović, K.; Tišma, M.; Zelić, B. Proximate analysis of cold-press oil cakes after biological treatment with Trametes versicolor and Humicola grisea. Eng. Life Sci. 2018, 18, 924–931. [Google Scholar] [CrossRef] [PubMed]
- Presto, M.H.; Lyberg, K.; Lindberg, J.E. Digestibility of amino acids in organically cultivated white-flowering faba bean and cake from cold-pressed rapeseed, linseed and hemp seed in growing pigs. Arch. Anim. Nutr. 2011, 65, 21–33. [Google Scholar] [CrossRef]
- Halle, I.; Schöne, F. Influence of rapeseed cake, linseed cake and hemp seed cake on laying performance of hens and fatty acid composition of egg yolk. J. Für Verbrauch. Und Lebensm. 2013, 8, 185–193. [Google Scholar] [CrossRef]
- Rinne, M.; Jaakkola, S.; Järvi, M.; Huhtanen, P. Effects of gradual replacement of rapeseed cake with linseed cake in a grass silage-based diet for dairy cows. Agric. Food Sci. 1997, 6, 161–172. [Google Scholar] [CrossRef]
- Sobczak, P.; Zawiślak, K.; Starek, A.; Żukiewicz-Sobczak, W.; Sagan, A.; Zdybel, B.; Andrejko, D. Compaction Process as a Concept of Press-Cake Production from Organic Waste. Sustainability 2020, 12, 1567. [Google Scholar] [CrossRef]
- Moloney, A.P.; Woods, V.B.; Crowley, J.G. A note on the nutritive value of camelina meal for beef cattle. Ir. J. Agric. Food Res. 1998, 37, 243–247. [Google Scholar]
- Woyengo, T.A.; Patterson, R.; Levesque, C.L. Nutritive value of multienzyme supplemented cold-pressed camelina cake for pigs. J. Anim. Sci. 2018, 96, 1119–1129. [Google Scholar] [CrossRef]
- Keller, M.; Reidy, B.; Scheurer, A.; Eggerschwiler, L.; Morel, I.; Giller, K. Soybean Meal Can Be Replaced by Faba Beans, Pumpkin Seed Cake, Spirulina or Be Completely Omitted in a Forage-Based Diet for Fattening Bulls to Achieve Comparable Performance, Carcass and Meat Quality. Animals 2021, 11, 1588. [Google Scholar] [CrossRef] [PubMed]
- Šalavardić, Ž.K.; Novoselec, J.; Đidara, M.; Steiner, Z.; Ćavar, S.; Modić Šabić, A.; Antunović, Z. Effect of dietary hempseed cake on milk performance and haemato-chemicals in lactating Alpine dairy goats. Animal 2021, 15, 100255. [Google Scholar] [CrossRef] [PubMed]
- Silversides, F.G.; LefranÇois, M.R. The effect of feeding hemp seed meal to laying hens. Br. Poult. Sci. 2005, 46, 231–235. [Google Scholar] [CrossRef] [PubMed]
- Kalmendal, R.; Wall, H. Effects of a high oil and fibre diet and supplementary roughage on performance, injurious pecking and foraging activities in two layer hybrids. Br. Poult. Sci. 2012, 53, 153–161. [Google Scholar] [CrossRef] [PubMed]
- Kalmendal, R.; Elwinger, K.; Holm, L.; Tauson, R. High-fibre sunflower cake affects small intestinal digestion and health in broiler chickens. Br. Poult. Sci. 2011, 52, 86–96. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.W.; Koo, B.; Nyachoti, C.M. Digestible, metabolizable, and net energy of camelina cake fed to growing pigs and additivity of energy in mixed diets. J. Anim. Sci. 2017, 95, 4037–4044. [Google Scholar] [CrossRef] [PubMed]
- Mierliță, D. Fatty acids profile and oxidative stability of eggs from laying hens fed diets containing hemp seed or hempseed cake. S. Afr. J. Anim. Sci. 2019, 49, 310–321. [Google Scholar] [CrossRef]
- Kempe, R.; Saastamoinen, M. Effect of linseed cake supplementation on digestibility and faecal and haematological parameters in dogs. J. Anim. Physiol. Anim. Nutr. 2007, 91, 319–325. [Google Scholar] [CrossRef]
- Ahmad, S.; Kamran, Z.; Koutoulis, K.C. Supplemental Linseed on Egg Production. In Egg Innovations and Strategies for Improvements; Hester, P.Y., Ed.; Academic Press: San Diego, CA, USA, 2017; pp. 349–363. [Google Scholar]
- Ryhänen, E.-L.; Perttilä, S.; Tupasela, T.; Valaja, J.; Eriksson, C.; Larkka, K. Effect of Camelina sativa expeller cake on performance and meat quality of broilers. J. Sci. Food Agric. 2007, 87, 1489–1494. [Google Scholar] [CrossRef]
- Pekel, A.Y.; Kim, J.I.; Chapple, C.; Adeola, O. Nutritional characteristics of camelina meal for 3-week-old broiler chickens. Poult. Sci. 2015, 94, 371–378. [Google Scholar] [CrossRef]
- Święch, E.; Raj, S.; Pastuszewska, B.; Taciak, M.; Bartkowiak-Broda, I.; Smulikowska, S. Nutritional value of yellow-seeded winter rapeseed cakes for growing pigs. Agric. Food Sci. 2016, 25, 99–110. [Google Scholar] [CrossRef]
- He, M.L.; Gibb, D.; McKinnon, J.J.; McAllister, T.A. Effect of high dietary levels of canola meal on growth performance, carcass quality and meat fatty acid profiles of feedlot cattle. Can. J. Anim. Sci. 2013, 93, 269–280. [Google Scholar] [CrossRef]
- Almeida, F.N.; Htoo, J.K.; Thomson, J.; Stein, H.H. Amino acid digestibility in camelina products fed to growing pigs. Can. J. Anim. Sci. 2013, 93, 335–343. [Google Scholar] [CrossRef]
- Lawrence, R.D.; Anderson, J.L.; Clapper, J.A. Evaluation of camelina meal as a feedstuff for growing dairy heifers. J. Dairy Sci. 2016, 99, 6215–6228. [Google Scholar] [CrossRef] [PubMed]
- Kaczmarek, P.; Korniewicz, D.; Lipiński, K.; Mazur-Kuśnirek, M. The effect of hydrothermally processed soybean-and rapeseed-based diets on performance, meat and carcass quality characteristics in growing-finishing pigs. Ann. Anim. Sci. 2019, 19, 1083–1097. [Google Scholar] [CrossRef]
- Serrapica, F.; Masucci, F.; Raffrenato, E.; Sannino, M.; Vastolo, A.; Barone, C.M.A.; Di Francia, A. High Fiber Cakes from Mediterranean Multipurpose Oilseeds as Protein Sources for Ruminants. Animals 2019, 9, 918. [Google Scholar] [CrossRef]
- Meadus, W.J.; Duff, P.; McDonald, T.; Caine, W.R. Pigs fed camelina meal increase hepatic gene expression of cytochrome 8b1, aldehyde dehydrogenase, and thiosulfate transferase. J. Anim. Sci. Biotechnol. 2014, 5, 1. [Google Scholar] [CrossRef]
- Wu, G.; Bazer, F.W.; Satterfield, M.C.; Gilbreath, K.R.; Posey, E.A.; Sun, Y. L-Arginine nutrition and metabolism in ruminants. In Recent Advances in Animal Nutrition and Metabolism; Wu, G., Ed.; Springer: Berlin/Heidelberg, Germany, 2022; pp. 177–206. [Google Scholar]
- Hou, Y.; Wu, G. L-Glutamate nutrition and metabolism in swine. Amino Acids 2018, 50, 1497–1510. [Google Scholar] [CrossRef]
- Upadhaya, S.D.; Lee, S.S.; Kim, Y.H.; Wu, Z.; Kim, I.H. Glutamic acid supplementation recovers the reduced performance of weanling pigs fed reduced crude protein diets. Anim. Nutr. 2022, 8, 249–255. [Google Scholar] [CrossRef]
- House, J.D.; Neufeld, J.; Leson, G. Evaluating the Quality of Protein from Hemp Seed (Cannabis sativa L.) Products Through the use of the Protein Digestibility-Corrected Amino Acid Score Method. J. Agric. Food Chem. 2010, 58, 11801–11807. [Google Scholar] [CrossRef]
- Nega, T.; Woldes, Y. Review on Nutritional Limitations and Opportunities of using Rapeseed Meal and other Rape Seed by-Products in Animal Feeding. J. Nutr. Health Food Eng. 2018, 8, 00254. [Google Scholar] [CrossRef]
- Kahindi, R.K.; Woyengo, T.A.; Thacker, P.A.; Nyachoti, C.M. Energy and amino acid digestibility of camelina cake fed to growing pigs. Anim. Feed. Sci. Technol. 2014, 193, 93–101. [Google Scholar] [CrossRef]
- Johansson, B.; Nadeau, E. Performance of dairy cows fed an entirely organic diet containing cold-pressed rapeseed cake. Acta Agric. Scand. Sect. A—Anim. Sci. 2006, 56, 128–136. [Google Scholar] [CrossRef]
- Mierliță, D. Effects of diets containing hemp seeds or hemp cake on fatty acid composition and oxidative stability of sheep milk. S. Afr. J. Anim. Sci. 2018, 48, 504–515. [Google Scholar] [CrossRef]
- Taranu, I.; Gras, M.; Pistol, G.C.; Motiu, M.; Marin, D.E.; Lefter, N.; Ropota, M.; Habeanu, M. ω-3 PUFA rich Camelina oil by-products improve the systemic metabolism and spleen cell functions in fattening pigs. PLoS ONE 2014, 9, e110186. [Google Scholar] [CrossRef] [PubMed]
- Anca, G.; Hăbeanu, M.; Lefter, N.; Ropotă, M. Performance parameters, plasma lipid status, and lymphoid tissue fatty acid profile of broiler chicks fed camelina cake. Braz. J. Poult. Sci. 2019, 21, 1–8. [Google Scholar] [CrossRef]
- Grings, E.E.; Sackey, A.; Brake, D.W.; Perry, G.A. Comparison of Camelina Meal and Distiller’s Dried Grains with Solubles in Diet of Beef Replacement Heifers, South Dakota Beef Report; South Dakota State University: Brookings, SD, USA, 2015; p. 5. [Google Scholar]
- Dragojlović, D.; Rakita, S.; Stojkov, V.; Vidosavljević, S.; Đuragić, O.; Marjanović-Jeromela, A.; Cvejić, S. Cold-pressed camelina cake as a source of essential fatty acids in animal nutrition. In Proceedings of the 18th Euro Fed Lipid Congress and Expo, Leipzig, Germany, 18–21 October 2021; p. 293. [Google Scholar]
- Benhissi, H.; García-Rodríguez, A.; Beltrán de Heredia, I. Effect of type and inclusion level of cold-pressed oilseed cakes on in vitro rumen fermentation. Anim. Prod. Sci. 2014, 54, 1709–1713. [Google Scholar] [CrossRef]
- Benhissi, H.; Beltrán de Heredia, I.; García-Rodríguez, A. Effect of replacing palm fat with high-linoleic cold-pressed rapeseed or sunflower cakes on fatty acid biohydrogenation in an artificial rumen (Rusitec). Anim. Prod. Sci. 2016, 58, 499–506. [Google Scholar] [CrossRef]
- McKinnon, J.J.; Walker, A.M. Comparison of canola and mustard presscake from biodiesel production as protein sources for growing cattle. Can. J. Anim. Sci. 2009, 89, 401–408. [Google Scholar] [CrossRef]
- Karlsson, L.; Martinsson, K. Growth performance of lambs fed different protein supplements in barley-based diets. Livest. Sci. 2011, 138, 125–131. [Google Scholar] [CrossRef]
- Lawrence, R.D.; Anderson, J.L. Ruminal degradation and intestinal digestibility of camelina meal and carinata meal compared with other protein sources. Prof. Anim. Sci. 2018, 34, 10–18. [Google Scholar] [CrossRef]
- Mihhejev, K.; Henno, M.; Ots, M.; Rihma, E.; Elias, P.; Kuusik, S.; Kärt, O. Effects of fat-rich oil cakes on cheese fatty acid composition, and on cheese quality. Vet. Ir Zootech. 2007, 40, 55–61. [Google Scholar]
- Moate, P.J.; Williams, S.R.O.; Grainger, C.; Hannah, M.C.; Ponnampalam, E.N.; Eckard, R.J. Influence of cold-pressed canola, brewers grains and hominy meal as dietary supplements suitable for reducing enteric methane emissions from lactating dairy cows. Anim. Feed. Sci. Technol. 2011, 166–167, 254–264. [Google Scholar] [CrossRef]
- Lerch, S.; Ferlay, A.; Pomiès, D.; Martin, B.; Pires, J.A.A.; Chilliard, Y. Rapeseed or linseed supplements in grass-based diets: Effects on dairy performance of Holstein cows over 2 consecutive lactations. J. Dairy Sci. 2012, 95, 1956–1970. [Google Scholar] [CrossRef] [PubMed]
- Johansson, B.; Kumm, K.-I.; Åkerlind, M.; Nadeau, E. Cold-pressed rapeseed cake or full fat rapeseed to organic dairy cows—Milk production and profitability. Org. Agric. 2015, 5, 29–38. [Google Scholar] [CrossRef]
- Jóźwik, A.; Strzałkowska, N.; Markiewicz-Kęszycka, M.; Krzyżewski, J.; Lipińska, P.; Rutkowska, J.; Wróblewska, B.; Klusek, J.; Cooper, R.G. Effects of replacing rapeseed cake with linseed cake in a corn-grass silage-based diet for milking cows. Anim. Sci. Pap. Rep. 2016, 34, 129–142. [Google Scholar]
- Goiri, I.; Zubiria, I.; Benhissi, H.; Atxaerandio, R.; Ruiz, R.; Mandaluniz, N.; Garcia-Rodriguez, A. Use of Cold-Pressed Sunflower Cake in the Concentrate as a Low-Input Local Strategy to Modify the Milk Fatty Acid Profile of Dairy Cows. Animals 2019, 9, 803. [Google Scholar] [CrossRef]
- Toma, S.; Dragomir, C.; Habeanu, M.; Ropota, M.; Cismileanu, A.; Grosu, H. Effects of partial or total replacement of sunflower meal with camelina meal on dairy cows’ milk fatty acids profile. Arch. Zootech. 2015, 18, 85–94. [Google Scholar]
- Amores, G.; Virto, M.; Nájera, A.I.; Mandaluniz, N.; Arranz, J.; Bustamante, M.A.; Valdivielso, I.; Ruiz de Gordoa, J.C.; García-Rodríguez, A.; Barron, L.J.R.; et al. Rapeseed and sunflower oilcake as supplements for dairy sheep: Animal performance and milk fatty acid concentrations. J. Dairy Res. 2014, 81, 410–416. [Google Scholar] [CrossRef]
- Nájera, A.I.; Bustamante, M.A.; Albisu, M.; Valdivielso, I.; Amores, G.; Mandaluniz, N.; Arranz, J.; Barron, L.J.R.; de Renobales, M. Fatty acids, vitamins and cholesterol content, and sensory properties of cheese made with milk from sheep fed rapeseed oilcake. J. Dairy Sci. 2017, 100, 6962–6971. [Google Scholar] [CrossRef]
- Pascual, A.; Pineda-Quiroga, C.; Goiri, I.; Atxaerandio, R.; Ruiz, R.; García-Rodríguez, A. Effects of feeding UFA-rich cold-pressed oilseed cakes and sainfoin on dairy ewes’ milk fatty acid profile and curd sensory properties. Small Rumin. Res. 2019, 175, 96–103. [Google Scholar] [CrossRef]
- Jozwik, A.; Strzalkowska, N.; Bagnicka, E.; Lagodzinski, Z.; Pyzel, B.; Chylinski, W.; Czajkowska, A.; Grzybek, W.; Sloniewska, D.; Krzyzewski, J.; et al. The effect of feeding linseed cake on milk yield and milk fatty acid profile in goats. Anim. Sci. Pap. Rep. 2010, 28, 245–251. [Google Scholar]
- Markiewicz-Kęszycka, M.; Czyżak-Runowska, G.; Lipińska, P.; Wójtowski, J. Fatty acid profile of milk—A review. Bull. Vet. Inst. Pulawy 2013, 57, 135–139. [Google Scholar] [CrossRef]
- Drackley, J.K. Lipid metabolism. In Farm Animal Metabolism and Nutrition; D’Mello, J.P.F., Ed.; CABI Publishing: Wallingford, UK, 2000; Volume 1, pp. 97–119. [Google Scholar]
- Fox, P.F.; McSweeney, P.L.H. Advanced Dairy Chemistry Volume 2: Lipids; Springer Science & Business Media: New York, NY, USA, 2006; Volume 2. [Google Scholar]
- Christie, W.W. Lipid Metabolism in Ruminant Animals; Elsevier: London, UK, 2014. [Google Scholar]
- Enjalbert, F.; Combes, S.; Zened, A.; Meynadier, A. Rumen microbiota and dietary fat: A mutual shaping. J. Appl. Microbiol. 2017, 123, 782–797. [Google Scholar] [CrossRef] [PubMed]
- Doreau, M.; Bauchart, D.; Chilliard, Y. Enhancing fatty acid composition of milk and meat through animal feeding. Anim. Prod. Sci. 2011, 51, 19–29. [Google Scholar] [CrossRef]
- Ferlay, A.; Bernard, L.; Meynadier, A.; Malpuech-Brugère, C. Production of trans and conjugated fatty acids in dairy ruminants and their putative effects on human health: A review. Biochimie 2017, 141, 107–120. [Google Scholar] [CrossRef] [PubMed]
- Fredeen, A.H. Considerations in the nutritional modification of milk composition. Anim. Feed. Sci. Technol. 1996, 59, 185–197. [Google Scholar] [CrossRef]
- Dewanckele, L.; Toral, P.G.; Vlaeminck, B.; Fievez, V. Invited review: Role of rumen biohydrogenation intermediates and rumen microbes in diet-induced milk fat depression: An update. J. Dairy Sci. 2020, 103, 7655–7681. [Google Scholar] [CrossRef]
- Bauman, D.E.; McGuire, M.A.; Harvatine, K.J. Mammary gland, milk biosynthesis and secretion: Milk fat. In Encyclopedia of Dairy Sciences, 2nd ed.; Elsevier: New York, NY, USA, 2011; pp. 352–358. [Google Scholar]
- Toral, P.G.; Hervás, G.; Frutos, P. Effect of lipid supplementation on the endogenous synthesis of milk cis-9,trans-11 conjugated linoleic acid in dairy sheep and goats: A tracer assay with 13C-vaccenic acid. J. Dairy Sci. 2022, 105, 255–268. [Google Scholar] [CrossRef]
- Hanuš, O.; Samková, E.; Křížová, L.; Hasoňová, L.; Kala, R. Role of Fatty Acids in Milk Fat and the Influence of Selected Factors on Their Variability—A Review. Molecules 2018, 23, 1636. [Google Scholar] [CrossRef]
- Lerch, S.; Ferlay, A.; Shingfield, K.J.; Martin, B.; Pomiès, D.; Chilliard, Y. Rapeseed or linseed supplements in grass-based diets: Effects on milk fatty acid composition of Holstein cows over two consecutive lactations. J. Dairy Sci. 2012, 95, 5221–5241. [Google Scholar] [CrossRef]
- Turner, T.; Hessle, A.; Lundström, K.; Pickova, J. Influence of hempseed cake and soybean meal on lipid fractions in bovine M. longissimus dorsi. Acta Agric. Scand. Sect. A—Anim. Sci. 2008, 58, 152–160. [Google Scholar] [CrossRef]
- Turner, T.D.; Karlsson, L.; Mapiye, C.; Rolland, D.C.; Martinsson, K.; Dugan, M.E.R. Dietary influence on the m. longissimus dorsi fatty acid composition of lambs in relation to protein source. Meat Sci. 2012, 91, 472–477. [Google Scholar] [CrossRef] [PubMed]
- Orczewska-Dudek, S.; Pietras, M. The Effect of Dietary Camelina sativa Oil or Cake in the Diets of Broiler Chickens on Growth Performance, Fatty Acid Profile, and Sensory Quality of Meat. Animals 2019, 9, 734. [Google Scholar] [CrossRef] [PubMed]
- Untea, A.E.; Panaite, T.D.; Dragomir, C.; Ropota, M.; Olteanu, M.; Varzaru, I. Effect of dietary chromium supplementation on meat nutritional quality and antioxidant status from broilers fed with Camelina-meal-supplemented diets. Animal 2019, 13, 2939–2947. [Google Scholar] [CrossRef] [PubMed]
- Orczewska-Dudek, S.; Pietras, M.; Puchała, M.; Nowak, J. Camelina sativa oil and camelina cake as sources of polyunsaturated fatty acids in the diets of laying hens: Effect on hen performance, fatty acid profile of yolk lipids, and egg sensory quality. Ann. Anim. Sci. 2020, 20, 1365–1377. [Google Scholar] [CrossRef]
- Pekel, A.Y.; Adedokun, S.A.; Adeola, O. True phosphorus digestibility of camelina meal in broiler chickens. Can. J. Anim. Sci. 2018, 98, 194–203. [Google Scholar] [CrossRef]
- Orczewska-Dudek, S.; Pietras, M.; Mika, M. Effect of Camelina sativa oil and expeller cake on plasma level of iodothyronines and lipid profile of broiler chickens. Rocz. Nauk. Zootech. 2019, 46, 105–115. [Google Scholar]

| Nutrient | RSC | HSC | LSC | SFC | CSC | PSC |
|---|---|---|---|---|---|---|
| Protein (%) | 19.4 [15]–45.0 [16] | 24.8 [17]–36.1 [18] | 32.2 [19]–35.9 [20] | 21.6 [5]–37.7 [21] | 31.3 [22]–42.0 [23] | 38.3 [17]–62.3 [24] |
| Fat (%) | 9.6 [16]–31.3 [10] | 8.9 [25]–16.4 [26] | 11.5 [21]–21.4 [17] | 11.7 [27]–31.4 [21] | 10.5 [23]–26.0 [22] | 9.0 [24]–36.2 [17] |
| Ash (%) | 4.2 [15]–8.1 [16] | 6.3 [25]–7.5 [17] | 4.9 [18]–5.9 [20] | 4.3 [28]–6.3 [8] | 4.5 [29]–6.5 [23] | 7.5 [15]–8.1 [21] |
| Fiber (%) | 6.5 [16]–19.5 [15] | 25.1 [30]–26.1 [25] | 8.6 [31]–9.5 [32] | 12.6 [5]–37.0 [28] | 11.3 [33]–17.0 [34] | 23.1 [15] |
| NDF (%) | 17.0 [35]–33.7 [36] | 37.7 [18]–53.4 [17] | 17.6 [18]–21.3 [20] | n.d | 23.7 [37]–43.4 [29] | 11.8 [17]–13.9 [24] |
| ADF (%) | 9.0 [35]–21.0 [36] | 29.7 [30]–39.2 [17] | 13.0 [19]–14.2 [17] | n.d. | 11.1 [38]–21.6 [29] | 5.1 [17]–11.1 [24] |
| Glucosinolates, (µmol/g) | 8.8 [35]–16.9 [39] | n.d. | n.d. | n.d. | 22.9 [33]–46.1 [37] | n.d. |
| Essential AA (%) | RSC | HSC | LSC | SFC | CSC | PSC |
|---|---|---|---|---|---|---|
| Arginine | 1.4 [10]–1.9 [18] | 4.1 [18] | 3.0 [18] | n.d. | 2.5 [37]–3.5 [23] | n.d. |
| Histidine | 0.6 [10]–0.8 [18] | 1.0 [18] | 0.8 [18] | n.d. | 0.8 [37]–1.1 [47] | n.d. |
| Isoleucine | 1.0 [10]–2.6 [16] | 1.4 [18] | 1.5 [18] | n.d. | 1.1 [29]–1.6 [23] | n.d. |
| Leucine | 1.7 [10]–2.0 [18] | 2.3 [18] | 2.0 [18] | n.d. | 2.1 [37]–2.7 [23] | n.d. |
| Lysine | 1.4 [10]–1.6 [18] | 1.3 [18] | 1.3 [18] | n.d. | 1.4 [37]–2.1 [23] | n.d. |
| Methionine | 0.5 [10]–0.6 [16] | 0.8 [18] | 0.6 [18] | n.d. | 0.3 [29]–0.7 [23] | n.d. |
| Phenilalanine | 1.0 [10]–1.5 [16] | 1.6 [18] | 1.5 [18] | n.d. | 1.3 [29]–1.7 [23] | n.d. |
| Threonine | 1.0 [10]–1.3 [18] | 1.9 [18] | 1.2 [18] | n.d. | 1.3 [37]–1.6 [23] | n.d. |
| Nonessential AA (%) | n.d. | |||||
| Valine | 1.3 [10]–2.3 [16] | 1.8 [18] | 1.7 [18] | n.d. | 1.6 [37]–2.2 [23] | n.d. |
| Alanine | 1.1 [10]–1.5 [16] | 1.5 [18] | 1.5 [18] | n.d. | 1.4 [33]–1.9 [23] | n.d. |
| Aspartic acid | 1.7 [10]–2.8 [16] | 3.5 [18] | 3.1 [18] | n.d. | 2.5 [33]–3.4 [23] | n.d. |
| Cysteine | 0.6 [10]–1.2 [16] | 0.6 [18] | 0.7 [18] | n.d. | 0.3 [47]–0.9 [23] | n.d. |
| Glutamic acid | 3.7 [10]–7.8 [16] | 5.8 [18] | 6.2 [18] | n.d. | 5.3 [37]–6.8 [23] | n.d. |
| Glycine | 1.2 [10]–2.1 [16] | 1.5 [18] | 1.9 [18] | n.d. | 1.6 [37]–2.1 [23] | n.d. |
| Proline | 1.5 [10]–1.6 [18] | 1.4 [18] | 1.2 [18] | n.d. | 1.5 [29]–2.1 [47] | n.d. |
| Serine | 0.8 [16]–1.3 [18] | 1.7 [18] | 1.5 [18] | n.d. | 1.3 [47]–1.8 [29] | n.d. |
| Tyrosine | 0.7 [10]–1.0 [18] | 1.3 [18] | 0.9 [18] | n.d. | 0.9 [29]–1.1 [23] | n.d. |
| Fatty Acids | RSC | HSC | LSC | SFC | CSC | PSC |
|---|---|---|---|---|---|---|
| Palmitic acid | 4.4 [19]–4.8 [48] | 6.0 [19]–9.3 [49] | 5.9 [19] | 5.5 [5] | 7.1 [50]–7.4 [51] | 12.3 [15]–13.2 [24] |
| Stearic acid | 0.6 [19]–1.6 [48] | 1.6 [19]–3.8 [49] | 2.9 [19] | 1.1 [5] | 2.0 [51] | 4.9 [24]–5.2 [15] |
| Oleic acid | 51.6 [48]–59.6 [19] | 11.4 [19]–13.1 [49] | 22.1 [19] | 10.3 [5] | 15.6 [50]–18.8 [52] | 28.8 [15]–29.6 [24] |
| Linoleic acid (LA) | 20.2 [36]–23.5 [19] | 52.5 [49]–56.0 [25] | 17.1 [19] | 32.8 [5] | 19.4 [53]–25.0 [50] | 49.0 [24]–51.2 [15] |
| α-linolenic acid (ALA) | 9.4 [36]–10.6 [19] | 14.4 [25]–24.7 [19] | 51.5 [19] | 0.2 [5] | 22.0 [52]–35.0 [53] | 0.5 [15]–0.6 [24] |
| PUFA | 29.6 [36]–34.1 [19] | 70.4 [25]–80.5 [19] | 68.6 [19] | 34.2 [5] | 44.7 [52]–60.5 [50] | 47.0 [24]–52.9 [15] |
| n-3 | 9.4 [36]–10.6 [19] | 14.4 [25]–24.7 [19] | 51.5 [19] | 0.2 [5] | 22.0 [52]–32.9 [50] | 1.3 [15]–1.7 [24] |
| n-6 | 20.2 [36]–23.5 [19] | 53.5 [30]–56.0 [25] | 17.1 [19] | 34.0 [5] | 21.1 [51]–27.7 [50] | 45.1 [24]–51.7 [15] |
| n-6/n-3 | 2.2 [36]–2.4 [48] | 2.3 [19]–3.9 [25] | 0.3 [19] | 170 [5] | 0.7 [51]–1.0 [52] | 26.5 [24]–39.2 [15] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rakita, S.; Kokić, B.; Manoni, M.; Mazzoleni, S.; Lin, P.; Luciano, A.; Ottoboni, M.; Cheli, F.; Pinotti, L. Cold-Pressed Oilseed Cakes as Alternative and Sustainable Feed Ingredients: A Review. Foods 2023, 12, 432. https://doi.org/10.3390/foods12030432
Rakita S, Kokić B, Manoni M, Mazzoleni S, Lin P, Luciano A, Ottoboni M, Cheli F, Pinotti L. Cold-Pressed Oilseed Cakes as Alternative and Sustainable Feed Ingredients: A Review. Foods. 2023; 12(3):432. https://doi.org/10.3390/foods12030432
Chicago/Turabian StyleRakita, Slađana, Bojana Kokić, Michele Manoni, Sharon Mazzoleni, Peng Lin, Alice Luciano, Matteo Ottoboni, Federica Cheli, and Luciano Pinotti. 2023. "Cold-Pressed Oilseed Cakes as Alternative and Sustainable Feed Ingredients: A Review" Foods 12, no. 3: 432. https://doi.org/10.3390/foods12030432
APA StyleRakita, S., Kokić, B., Manoni, M., Mazzoleni, S., Lin, P., Luciano, A., Ottoboni, M., Cheli, F., & Pinotti, L. (2023). Cold-Pressed Oilseed Cakes as Alternative and Sustainable Feed Ingredients: A Review. Foods, 12(3), 432. https://doi.org/10.3390/foods12030432







