Functional Perspective of Leeks: Active Components, Health Benefits and Action Mechanisms
Abstract
:1. Introduction
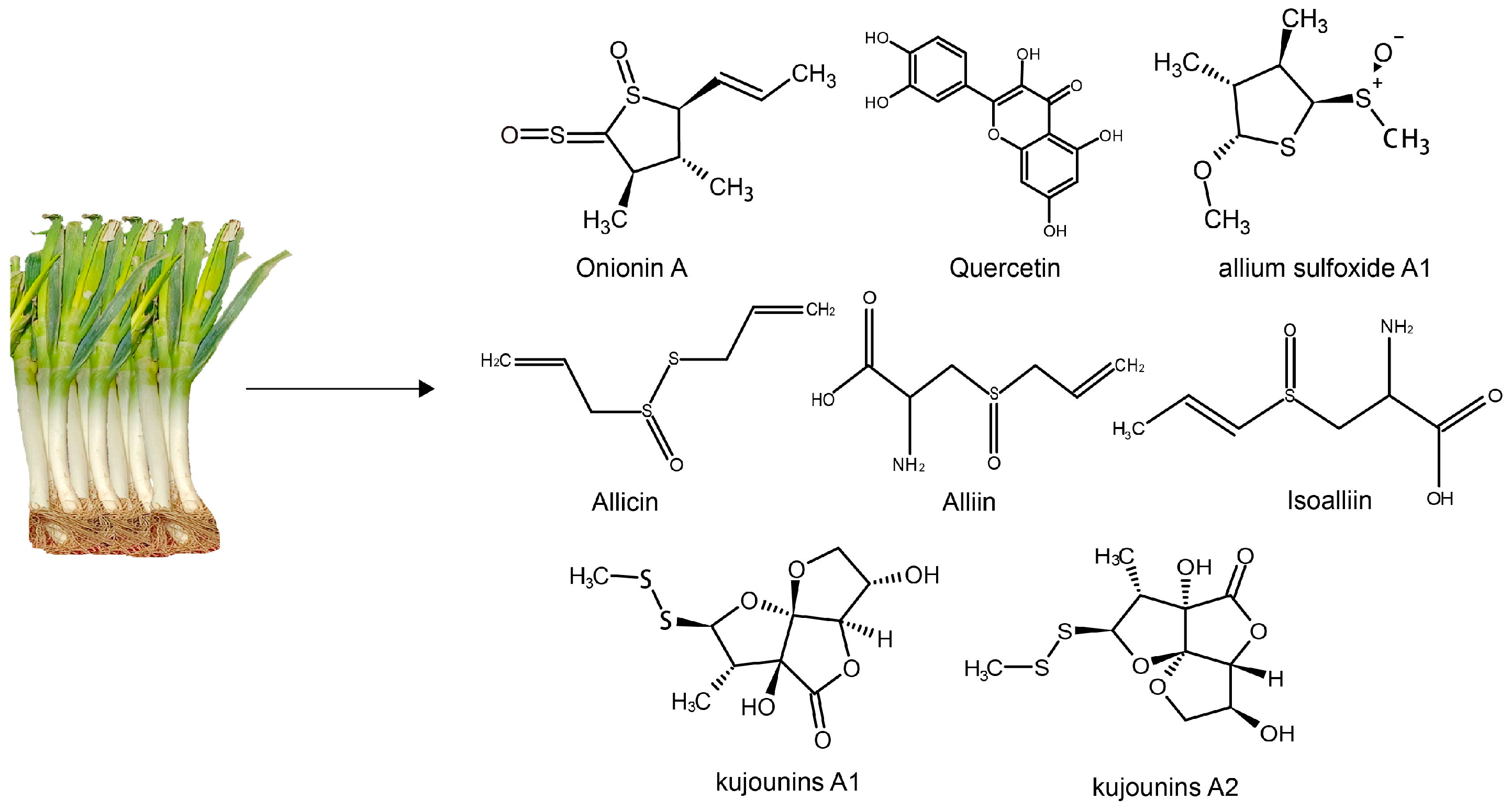
2. Characteristics and Composition of Leeks
3. Biological Functions of Leeks
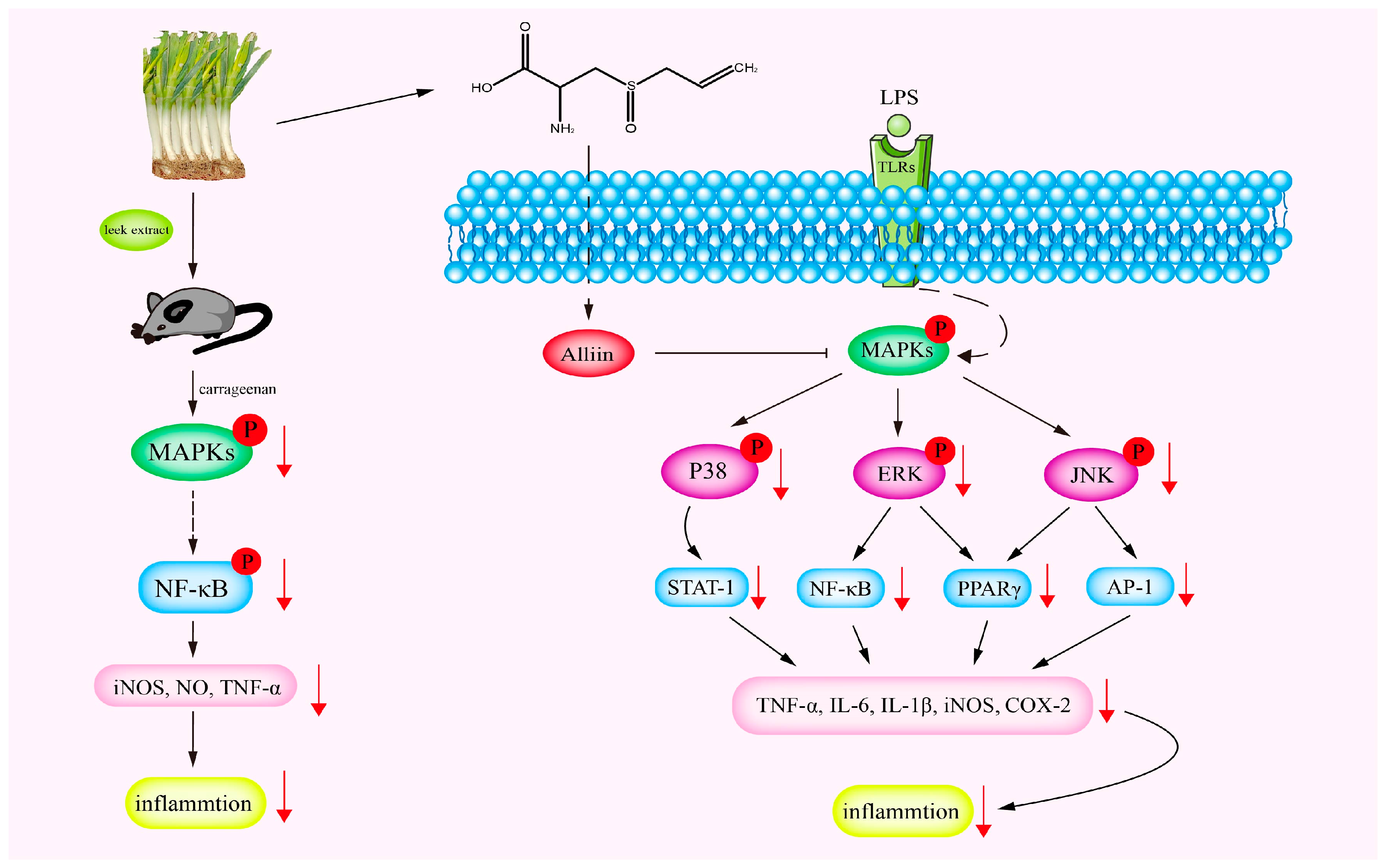
3.1. Anti-Inflammatory Activities
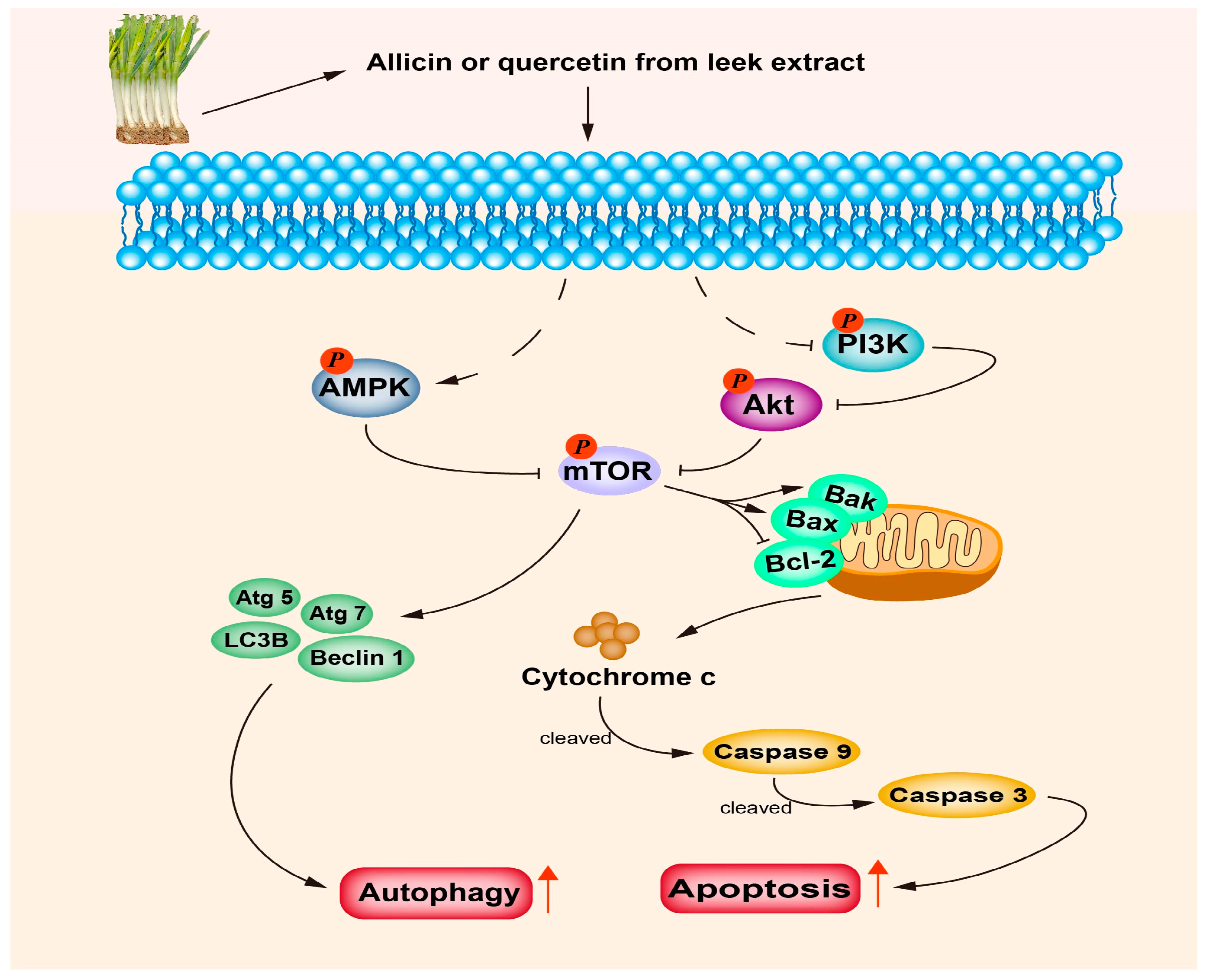
3.2. Anti-Cancer Activities
3.3. Antioxidant Activities
3.4. Anti-Obesity Activities
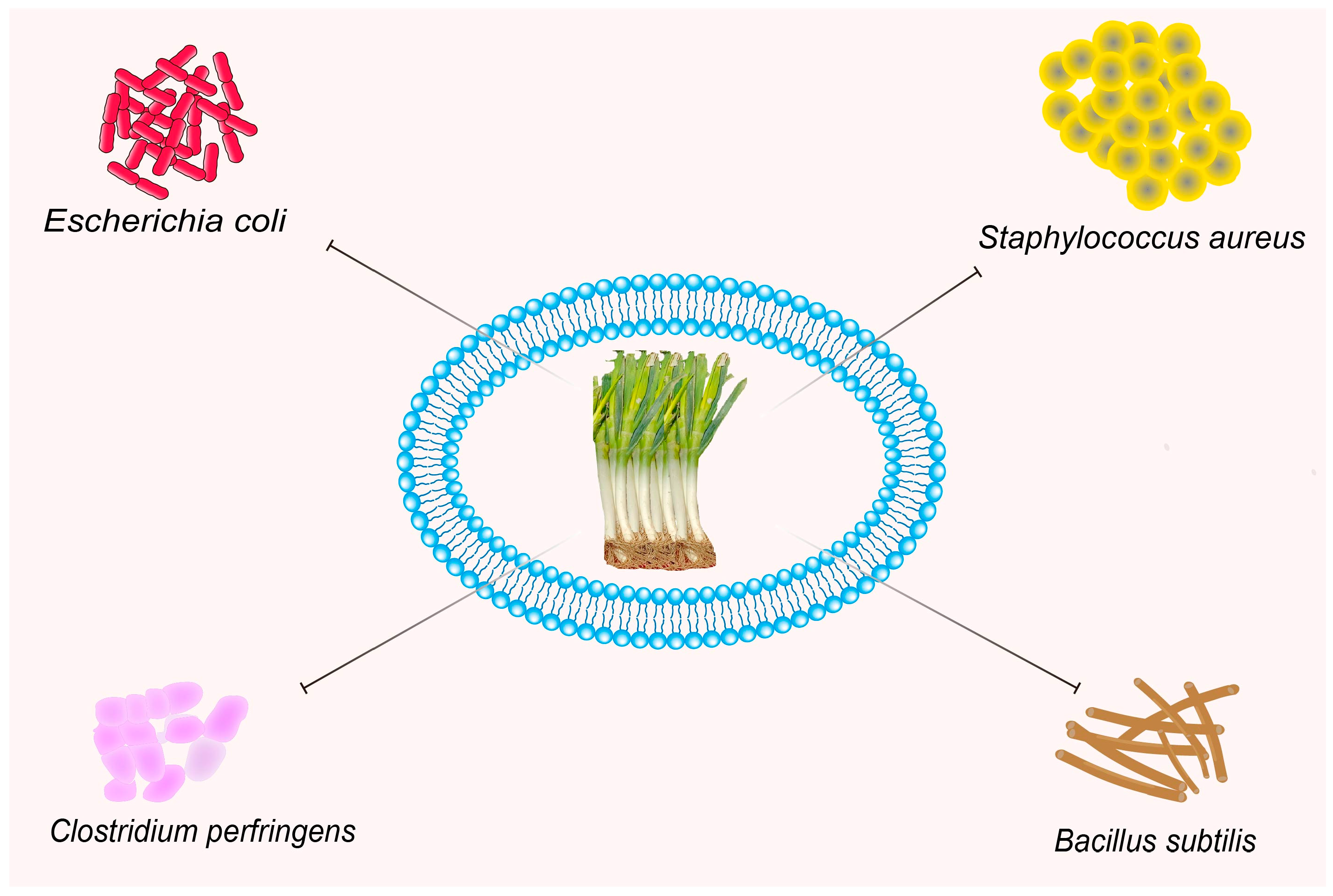
3.5. Antibacterial Activities
3.6. Other Functions
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
Abbreviations
| Akt | Protein kinase B |
| AMPK | Adenosine 5′-monophosphate (AMP)-activated protein kinase |
| Bax | Bax |
| Bcl-2 | B-cell lymphoma-2 |
| Bcl-xl | B-cell lymphoma xl |
| Caspase | Cysteinyl aspartate-specific proteinase |
| CCl4 | Carbon tetrachloride |
| COX-2 | Cyclooxygenase-2 |
| ERK | Extracellular signal-regulated kinase |
| FAS | Recombinant factor-related apoptosis |
| HIF-1α | Hypoxia inducible factor-1α |
| ICAM | Intercellular cell adhesion molecule |
| IL-10 | Interleukin-10 |
| IL-4 | Interleukin-4 |
| IL-6 | Interleukin-6 |
| iNOS | Induced nitric oxide synthase |
| LDL | Low-density lipoprotein |
| LPS | Lipopolysaccharide |
| MMP-9 | Matrix metalloprotein 9 |
| mTOR | Mammalian target of rapamycin |
| NADH | Nicotinamide adenine dinucleotide |
| NADPH | Nicotinamide adenine dinucleotide phosphate |
| NF-κB | Nuclear kappa B |
| NO | Nitric oxide |
| PCSK9 | Proprotein convertase subtilisin/kexin type 9 |
| PPARγ | Peroxisome proliferator-activated receptors γ |
| ROS | Reactive oxygen species |
| SREBP1c | Sterol regulatory element binding protein 1c |
| SREBP2 | Sterol regulatory element binding protein 2 |
| STAT3 | Signal transducer and activator of transcription-3 |
| TGF-β | Transforming growth factor β |
| TNF-α | Tumor necrosis factor α |
| UCP-2 | Uncoupling protein 2 |
| VEGF | Vascular endothelial growth factor |
References
- Bastaki, S.M.A.; Ojha, S.; Kalasz, H.; Adeghate, E. Chemical constituents and medicinal properties of allium species. Mol. Cell. Biochem. 2021, 476, 4301–4321. [Google Scholar] [CrossRef]
- Kurnia, D.; Ajiati, D.; Heliawati, L.; Sumiarsa, D. Antioxidant properties and structure-antioxidant activity relationship of allium species leaves. Molecules 2021, 26, 7175. [Google Scholar] [CrossRef] [PubMed]
- McCallum, J.; Baldwin, S.; Shigyo, M.; Deng, Y.; van Heusden, S.; Pither-Joyce, M.; Kenel, F. AlliumMap—A comparative genomics resource for cultivated allium vegetables. BMC Genom. 2012, 13, 168. [Google Scholar] [CrossRef]
- Khandagale, K.; Krishna, R.; Roylawar, P.; Ade, A.B.; Benke, A.; Shinde, B.; Singh, M.; Gawande, S.J.; Rai, A. Omics approaches in allium research: Progress and way ahead. Peer J. 2020, 8, e9824. [Google Scholar] [CrossRef]
- Yan, J.K.; Zhu, J.; Liu, Y.; Chen, X.; Wang, W.; Zhang, H.; Li, L. Recent advances in research on allium plants: Functional ingredients, physiological activities, and applications in agricultural and food sciences. Crit. Rev. Food Sci. 2022, 2022, 1–29. [Google Scholar] [CrossRef] [PubMed]
- Kothari, D.; Lee, W.D.; Niu, K.M.; Kim, S.K. The genus allium as poultry feed additive: A review. Animals 2019, 9, 1032. [Google Scholar] [CrossRef]
- Hill, C.R.; Shafaei, A.; Balmer, L.; Lewis, J.R.; Hodgson, J.M.; Millar, A.H.; Blekkenhorst, L.C. Blekkenhorst, Sulfur compounds: From plants to humans and their role in chronic disease prevention. Crit. Rev. Food Sci. 2022, 2022, 1–23. [Google Scholar]
- Arreola, R.; Quintero-Fabián, S.; López-Roa, R.I.; Flores-Gutiérrez, E.O.; Reyes-Grajeda, J.P.; Carrera-Quintanar, L.; Ortuño-Sahagún, D. Immunomodulation and anti-inflammatory effects of garlic compounds. J. Immunol. Res. 2015, 2015, 401630. [Google Scholar] [CrossRef]
- Tocmo, R.; Liang, D.; Lin, Y.; Huang, D. Chemical and biochemical mechanisms underlying the cardioprotective roles of dietary organopolysulfides. Front. Nutr. 2015, 2, 1. [Google Scholar] [CrossRef]
- Rodríguez Galdón, B.; Rodríguez Rodríguez, E.M.; Díaz Romero, C. Flavonoids in onion cultivars (Allium cepa L.). J. Food Sci. 2008, 73, C599–C605. [Google Scholar] [CrossRef]
- Mlcek, J.; Jurikova, T.; Skrovankova, S.; Sochor, J. Quercetin and its anti-allergic immune response. Molecules 2016, 21, 623. [Google Scholar] [CrossRef]
- Cao, Y.; Han, S.; Lu, H.; Luo, Y.; Guo, T.; Wu, Q.; Luo, F. Targeting mTOR signaling by dietary polyphenols in obesity prevention. Nutrients 2022, 14, 5171. [Google Scholar] [CrossRef] [PubMed]
- Piccolella, S.; Crescente, G.; Candela, L.; Pacifico, S. Nutraceutical polyphenols: New analytical challenges and opportunities. J. Pharm. Biomed. 2019, 175, 112774. [Google Scholar] [CrossRef] [PubMed]
- Asemani, Y.; Zamani, N.; Bayat, M.; Amirghofran, Z. Allium vegetables for possible future of cancer treatment. Phytother. Res. 2019, 33, 3019–3039. [Google Scholar] [CrossRef] [PubMed]
- Goncagul, G.; Ayaz, E. Antimicrobial effect of garlic (Allium sativum). Recent Pat. Antiinfect. Drug Discov. 2010, 5, 91–93. [Google Scholar] [CrossRef]
- Yen, D.T.H.; Tai, B.H.; Yen, P.H.; Nhiem, N.X.; Cuong, N.T.; Dung, N.V.; Huong, P.T.T.; Cuong, P.V.; Kiem, P.V. Furostane saponins from the seeds of allium ramosum and their lipid accumulation inhibitory activity. Chem. Biodivers. 2022, 19, e202200590. [Google Scholar] [CrossRef]
- Kumari, N.; Kumar, M.; Radha; Lorenzo, J.M.; Sharma, D.; Puri, S.; Pundir, A.; Dhumal, S.; Bhuyan, D.J.; Jayanthy, G.; et al. Onion and garlic polysaccharides: A review on extraction, characterization, bioactivity, and modifications. Int. J. Biol. Macromol. 2022, 219, 1047–1061. [Google Scholar] [CrossRef]
- Rauf, A.; Abu-Izneid, T.; Thiruvengadam, M.; Imran, M.; Olatunde, A.; Shariati, M.A.; Bawazeer, S.; Naz, S.; Shirooie, S.; Sanches-Silva, A.; et al. Garlic (Allium sativum L.): Its chemistry, nutritional composition, toxicity, and anticancer properties. Curr. Top. Med. Chem. 2022, 22, 957–972. [Google Scholar] [CrossRef]
- Zhang, T.R.; Zhao, C.Y.; Liu, B.; Yang, W.; Yang, L. Biological functions of welsh onion extract and its application in animal production. J. Econ. Entomol. 2015, 19, 241–244. [Google Scholar]
- Izol, E.; Temel, H.; Yilmaz, M.A.; Yener, I.; Olmez, O.T.; Kaplaner, E.; Fırat, M.; Hasimi, N.; Ozturk, M.; Ertas, A. A detailed chemical and biological investigation of twelve allium species from Eastern Anatolia with chemometric studies. Chem. Biodivers. 2021, 18, e2000560. [Google Scholar] [CrossRef]
- Arulselvan, P.; Wen, C.C.; Lan, C.W.; Chen, Y.H.; Wei, W.C.; Yang, N.S. Dietary administration of scallion extract effectively inhibits colorectal tumor growth: Cellular and molecular mechanisms in mice. PLoS ONE 2012, 7, e44658. [Google Scholar] [CrossRef]
- Sung, Y.Y.; Yoon, T.; Kim, S.J.; Yang, W.K.; Kim, H.K. Anti-obesity activity of Allium fistulosum L. extract by down-regulation of the expression of lipogenic genes in high-fat diet-induced obese mice. Mol. Med. Rep. 2011, 4, 431–435. [Google Scholar]
- Yamamoto, Y.; Aoyama, S.; Hamaguchi, N.; Rhi, G.S. Antioxidative and antihypertensive effects of welsh onion on rats fed with a high-fat high-sucrose diet. Biosci. Biotechnol. Biochem. 2005, 69, 1311–1317. [Google Scholar] [CrossRef]
- Kang, M.J.; Kim, J.H.; Choi, H.N.; Kim, M.J.; Han, J.H.; Lee, J.H.; Kim, J.I. Hypoglycemic effects of welsh onion in an animal model of diabetes mellitus. Nutr. Res. Pract. 2010, 4, 486–491. [Google Scholar] [CrossRef]
- Chen, J.H.; Chen, H.I.; Wang, J.S.; Tsai, S.J.; Jen, C.J. Effects of welsh onion extracts on human platelet function in vitro. Life Sci. 2000, 66, 1571–1579. [Google Scholar] [CrossRef] [PubMed]
- Padula, G.; Xia, X.; Hołubowicz, R. Welsh onion (Allium fistulosum L.) seed physiology, breeding, production and trade. Plants 2022, 11, 343. [Google Scholar] [CrossRef] [PubMed]
- Li, X. The Research on Hypoglycemic and Antibacterial Biological Activity of Allium fistulosum L. Master’s Thesis, Wuhan Institute of Technology, Wuhan, China, 2018. [Google Scholar]
- Li, X.; Cai, Y.; Zhou, T.T.; Zheng, W.Y.X.; Ruan, J.L. Research progress of bioactivity and chemical composition of Allium fistulosum. Chin. Condiment 2018, 43, 196–200. [Google Scholar]
- Huang, X.S. Study on the content and chemical constituents of volatile oil in welsh onion (Allium fistulosum L. var. gigantum Makino.). Food Ferment. Ind. 2004, 10, 114–117. [Google Scholar]
- Chan, J.Y.; Yuen, A.C.; Chan, R.Y.; Chan, S.W. A review of the cardiovascular benefits and antioxidant properties of allicin. Phytother. Res. 2013, 27, 637–646. [Google Scholar] [CrossRef] [PubMed]
- Yoshimoto, N.; Saito, K. S-Alk(en)ylcysteine sulfoxides in the genus allium: Proposed biosynthesis, chemical conversion, and bioactivities. J. Exp. Bot. 2019, 70, 4123–4137. [Google Scholar] [CrossRef]
- Rose, P.; Whiteman, M.; Moore, P.K.; Zhu, Y.Z. Bioactive S-alk(en)yl cysteine sulfoxide metabolites in the genus allium: The chemistry of potential therapeutic agents. Nat. Prod. Rep. 2005, 22, 351–368. [Google Scholar] [CrossRef]
- Nohara, T.; Fujiwara, Y.; Kudo, R.; Yamaguchi, K.; Ikeda, T.; Murakami, K.; Ono, M.; Kajimoto, T.; Takeya, M. Isolation and characterization of new onionins A2 and A3 from Allium cepa, and of onionins A1, A2, and A3 from Allium fistulosum. Chem. Pharm. Bull. 2014, 62, 1141–1145. [Google Scholar] [CrossRef]
- Fukaya, M.; Nakamura, S.; Nakagawa, R.; Nakashima, S.; Yamashita, M.; Matsuda, H. Rare sulfur-containing compounds, kujounins A1 and A2 and allium sulfoxide A1, from Allium fistulosum ‘Kujou’. Org. Lett. 2018, 20, 28–31. [Google Scholar] [CrossRef] [PubMed]
- Alam, A.; Al Arif Jahan, A.; Bari, M.S.; Khandokar, L.; Mahmud, M.H.; Junaid, M.; Chowdhury, M.S.; Khan, M.F.; Seidel, V.; Haque, M.A. Allium vegetables: Traditional uses, phytoconstituents, and beneficial effects in inflammation and cancer. Crit. Rev. Food Sci. 2022, 2022, 1–35. [Google Scholar] [CrossRef] [PubMed]
- Tsai, T.H.; Tsai, P.J.; Ho, S.C. Antioxidant and anti-inflammatory activities of several commonly used spices. J. Food Sci. 2005, 70, C93–C97. [Google Scholar] [CrossRef]
- Wang, B.S.; Huang, G.J.; Lu, Y.H.; Chang, L.W. Anti-inflammatory effects of an aqueous extract of welsh onion green leaves in mice. Food Chem. 2013, 138, 751–756. [Google Scholar] [CrossRef]
- Park, S.H.; Kim, J.I.; Jeong, Y.K.; Choi, Y.H. Extracts of Allium fistulosum attenuates pro-inflammatory action in the lipopolysaccharide-stimulated BV2 microglia cells. J. Life Sci. 2011, 21, 796–804. [Google Scholar] [CrossRef]
- Wang, B.S.; Chen, J.H.; Liang, Y.C.; Duh, P.D. Effects of welsh onion on oxidation of low-density lipoprotein and nitric oxide production in macrophage cell line RAW 264.7. Food Chem. 2005, 91, 147–155. [Google Scholar] [CrossRef]
- Țigu, A.B.; Moldovan, C.S.; Toma, V.A.; Farcaș, A.D.; Moț, A.C.; Jurj, A.; Fischer-Fodor, E.; Mircea, C.; Pârvu, M. Phytochemical analysis and in vitro effects of Allium fistulosum L. and Allium sativum L. extracts on human normal and tumor cell lines: A comparative study. Molecules 2021, 26, 574. [Google Scholar] [CrossRef]
- Wang, M.; Chen, X.; Yu, F.; Zhang, L.; Zhang, Y.; Chang, W. The targeting of noncoding RNAs by quercetin in cancer prevention and therapy. Oxidative Med. Cell. Longev. 2022, 2022, 4330681. [Google Scholar] [CrossRef]
- Reyes-Farias, M.; Carrasco-Pozo, C. The anti-cancer effect of quercetin: Molecular implications in cancer metabolism. Int. J. Mol. Sci. 2019, 20, 3177. [Google Scholar] [CrossRef] [PubMed]
- Wang, B.S.; Lin, S.S.; Hsiao, W.C.; Fan, J.J.; Fuh, L.F.; Duh, P.D. Protective effects of an aqueous extract of welsh onion green leaves on oxidative damage of reactive oxygen and nitrogen species. Food Chem. 2006, 98, 149–157. [Google Scholar] [CrossRef]
- Chang, T.C.; Jang, H.D.; Lin, W.D.; Duan, P.F. Antioxidant and antimicrobial activities of commercial rice wine extracts of Taiwanese Allium fistulosum. Food Chem. 2016, 190, 724–729. [Google Scholar] [CrossRef]
- Yamamoto, Y.; Yasuoka, A. Welsh onion attenuates hyperlipidemia in rats fed on high-fat high-sucrose diet. Biosci. Biotechnol. Biochem. 2010, 74, 402–404. [Google Scholar] [CrossRef]
- Sung, Y.Y.; Kim, D.S.; Kim, S.H.; Kim, H.K. Aqueous and ethanolic extracts of welsh onion, Allium fistulosum, attenuate high-fat diet-induced obesity. BMC Complement. Altern. Med. 2018, 18, 105. [Google Scholar] [CrossRef] [PubMed]
- Choi, H.K.; Hwang, J.T.; Nam, T.G.; Kim, S.H.; Min, D.K.; Park, S.W.; Chung, M.Y. Welsh onion extract inhibits PCSK9 expression contributing to the maintenance of the LDLR level under lipid depletion conditions of HepG2 cells. Food Funct. 2017, 8, 4582–4591. [Google Scholar] [CrossRef]
- Zhai, M.M.; Niu, H.T.; Li, J.; Xiao, H.; Shi, Y.P.; Di, D.L.; Crews, P.; Wu, Q.X. Talaromycolides A-C, novel phenyl-substituted phthalides isolated from the green Chinese onion-derived fungus talaromyces pinophilus AF-02. J. Agric. Food Chem. 2015, 63, 9558–9564. [Google Scholar] [CrossRef] [PubMed]
- Zolfaghari, B.; Yazdiniapour, Z.; Sadeghi, M.; Akbari, M.; Troiano, R.; Lanzotti, V. Cinnamic acid derivatives from welsh onion (Allium fistulosum) and their antibacterial and cytotoxic activities. Phytochem. Anal. 2021, 32, 84–90. [Google Scholar] [CrossRef]
- Han, C. Study on the antimicrobial activity of the extract from welsh onion. Food Res. Dev. 2007, 6, 65–67. [Google Scholar]
- Dalgleish, A.G.; O’Byrne, K.J. Chronic immune activation and inflammation in the pathogenesis of AIDS and cancer. Adv. Cancer Res. 2002, 84, 231–276. [Google Scholar]
- Liu, C.; Chu, D.; Kalantar-Zadeh, K.; George, J.; Young, H.A.; Liu, G. Cytokines: From clinical significance to quantification. Adv. Sci. 2021, 8, e2004433. [Google Scholar] [CrossRef] [PubMed]
- Netea, M.G.; Balkwill, F.; Chonchol, M.; Cominelli, F.; Donath, M.Y.; Giamarellos-Bourboulis, E.J.; Golenbock, D.; Gresnigt, M.S.; Heneka, M.T.; Hoffman, H.M.; et al. A guiding map for inflammation. Nat. Immunol. 2017, 18, 826–831. [Google Scholar] [CrossRef] [PubMed]
- De Meyer, G.R.; De Cleen, D.M.; Cooper, S.; Knaapen, M.W.; Jans, D.M.; Martinet, W.; Herman, A.G.; Bult, H.; Kockx, M.M. Platelet phagocytosis and processing of beta-amyloid precursor protein as a mechanism of macrophage activation in atherosclerosis. Circ. Res. 2002, 90, 1197–1204. [Google Scholar] [CrossRef]
- Kay, J.; Thadhani, E.; Samson, L.; Engelward, B. Inflammation-induced DNA damage, mutations and cancer. DNA Repair 2019, 83, 102673. [Google Scholar] [CrossRef]
- Wang, X.; Feng, Y.; Zhou, C.; Sun, Y.; Wu, B.; Yagoub, A.E.A.; Aboagarib, E.A.A. Effect of vacuum and ethanol pretreatment on infrared-hot air drying of scallion (Allium fistulosum). Food Chem. 2019, 295, 432–440. [Google Scholar] [CrossRef]
- Tenopoulou, M.; Doulias, P.T. Endothelial nitric oxide synthase-derived nitric oxide in the regulation of metabolism. F1000Research 2020, 9, 1190. [Google Scholar] [CrossRef] [PubMed]
- Kaur, B.; Singh, P. Inflammation: Biochemistry, cellular targets, anti-inflammatory agents and challenges with special emphasis on cyclooxygenase-2. Bioorg. Chem. 2022, 121, 105663. [Google Scholar] [CrossRef] [PubMed]
- Shi, L.; Lin, Q.; Li, X.; Nie, Y.; Sun, S.; Deng, X.; Wang, L.; Lu, J.; Tang, Y.; Luo, F. Alliin, a garlic organosulfur compound, ameliorates gut inflammation through MAPK-NF-κB/AP-1/STAT-1 inactivation and PPAR-γ activation. Mol. Nutr. Food Res. 2017, 61, 9. [Google Scholar] [CrossRef]
- Liu, M.; Lu, J.; Yang, S.; Chen, Y.; Yu, J.; Guan, S. Alliin alleviates LPS-induced pyroptosis via promoting mitophagy in THP-1 macrophages and mice. Food Chem. Toxicol. 2022, 160, 112811. [Google Scholar] [CrossRef]
- Jang, J.Y.; Lee, M.J.; You, B.R.; Jin, J.S.; Lee, S.H.; Yun, Y.R.; Kim, J. Allium hookeri root extract exerts anti-inflammatory effects by nuclear factor-κB down-regulation in lipopolysaccharide-induced RAW264.7 cells. BMC Complement. Altern. Med. 2017, 17, 126. [Google Scholar]
- Zhang, M.; Pan, H.; Xu, Y.; Wang, X.; Qiu, Z.; Jiang, L. Allicin decreases lipopolysaccharide-induced oxidative stress and inflammation in human umbilical vein endothelial cells through suppression of mitochondrial dysfunction and activation of Nrf2. Cell Physiol. Biochem. 2017, 41, 2255–2267. [Google Scholar] [CrossRef] [PubMed]
- Barnabei, L.; Laplantine, E.; Mbongo, W.; Rieux-Laucat, F.; Weil, R. NF-κB: At the borders of autoimmunity and inflammation. Front. Immunol. 2021, 12, 716469. [Google Scholar] [CrossRef] [PubMed]
- Huxford, T.; Huang, D.B.; Malek, S.; Ghosh, G. The crystal structure of the IkappaBalpha/NF-kappaB complex reveals mechanisms of NF-kappaB inactivation. Cell 1998, 95, 759–770. [Google Scholar] [CrossRef]
- Torre, L.A.; Siegel, R.L.; Ward, E.M.; Jemal, A. Global cancer incidence and mortality rates and trends—An update. Cancer Epidemiol. Biomark. 2016, 25, 16–27. [Google Scholar] [CrossRef] [PubMed]
- Sung, H.; Ferlay, J.; Siegel, R.L.; Laversanne, M.; Soerjomataram, I.; Jemal, A.; Bray, F. Global cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J. Clin. 2021, 71, 209–249. [Google Scholar] [CrossRef]
- López-Plaza, B.; Loria-Kohen, V.; González-Rodríguez, L.G.; Fernández-Cruz, E. Alimentación y estilo de vida en la prevención del cáncer [Diet and lifestyle in cancer prevention]. Nutr. Hosp. 2022, 39, 74–77. [Google Scholar]
- Kamal, N.; Ilowefah, M.A.; Hilles, A.R.; Anua, N.A.; Awin, T.; Alshwyeh, H.A.; Aldosary, S.K.; Jambocus, N.G.S.; Alosaimi, A.A.; Rahman, A.; et al. Genesis and mechanism of some cancer types and an overview on the role of diet and nutrition in cancer prevention. Molecules 2022, 27, 1794. [Google Scholar] [CrossRef]
- Narimatsu, H.; Yaguchi, Y.T. The role of diet and nutrition in cancer: Prevention, treatment, and survival. Nutrients 2022, 14, 3329. [Google Scholar] [CrossRef]
- Zhang, Y.; Liu, K.; Yan, C.; Yin, Y.; He, S.; Qiu, L.; Li, G. Natural polyphenols for treatment of colorectal cancer. Molecules 2022, 27, 8810. [Google Scholar] [CrossRef]
- Li, M.; Hu, Z.; Guo, T.; Xie, T.; Tang, Y.; Wu, X.; Luo, F. Targeting mTOR signaling by dietary polysaccharides in cancer prevention: Advances and challenges. J. Agric. Food Chem. 2023, 71, 96–109. [Google Scholar] [CrossRef]
- Hu, Z.; Li, M.; Cao, Y.; Akan, O.D.; Guo, T.; Luo, F. Targeting AMPK signaling by dietary polyphenols in cancer prevention. Mol. Nutr. Food Res. 2022, 66, e2100732. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.; Rauch, J.; Kolch, W. Targeting MAPK signaling in cancer: Mechanisms of drug resistance and sensitivity. Int. J. Mol. Sci. 2020, 21, 1102. [Google Scholar] [CrossRef] [PubMed]
- Xu, J.; Wang, Y.; Wang, Y.; Wang, Z.; He, X. A-24, a steroidal saponin from allium chinense, induced apoptosis, autophagy and migration inhibition in p53 wild-type and p53-deficient gastric cancer cells. Chem. Biol. Interact. 2021, 348, 109648. [Google Scholar] [CrossRef] [PubMed]
- Li, C.; Lun, W.; Zhao, X.; Lei, S.; Guo, Y.; Ma, J.; Zhi, F. Allicin alleviates inflammation of trinitrobenzenesulfonic acid-induced rats and suppresses P38 and JNK pathways in Caco-2 cells. Mediat. Inflamm. 2015, 2015, 434692. [Google Scholar] [CrossRef]
- Wang, Y.; Yi, X.; Xiang, L.; Huang, Y.; Wang, Z.; He, X. Furostanol saponins from chinese onion induce G2/M cell-cycle arrest and apoptosis through mitochondria-mediate pathway in HepG2 cells. Steroids 2019, 148, 11–18. [Google Scholar] [CrossRef]
- Park, H.S.; Choi, E.J.; Lee, J.H.; Kim, G.H. Evaluation of allium vegetables for anti-adipogenic, anti-cancer, and anti-inflammatory activities in vitro. J. Life Sci. 2013, 5, 127–132. [Google Scholar]
- Rosas-González, V.C.; Téllez-Bañuelos, M.C.; Hernández-Flores, G.; Bravo-Cuellar, A.; Aguilar-Lemarroy, A.; Jave-Suárez, L.F.; Haramati, J.; Solorzano-Ibarra, F.; Ortiz-Lazareno, P.C. Differential effects of alliin and allicin on apoptosis and senescence in luminal A and triple-negative breast cancer: Caspase, ΔΨm, and pro-apoptotic gene involvement. Fundam. Clin. Pharmacol. 2020, 34, 671–686. [Google Scholar] [CrossRef]
- Di Petrillo, A.; Orrù, G.; Fais, A.; Fantini, M.C. Quercetin and its derivates as antiviral potentials: A comprehensive review. Phytother. Res. 2022, 36, 266–278. [Google Scholar] [CrossRef]
- Pan, Y.; Zheng, Y.M.; Ho, W.S. Effect of quercetin glucosides from allium extracts on HepG2, PC-3 and HT-29 cancer cell lines. Oncol. Lett. 2018, 15, 4657–4661. [Google Scholar] [CrossRef]
- Srinivas, U.S.; Tan, B.W.Q.; Vellayappan, B.A.; Jeyasekharan, A.D. ROS and the DNA damage response in cancer. Redox. Biol. 2019, 25, 101084. [Google Scholar] [CrossRef]
- Prasad, S.; Gupta, S.C.; Tyagi, A.K. Reactive oxygen species (ROS) and cancer: Role of antioxidative nutraceuticals. Cancer Lett. 2017, 387, 95–105. [Google Scholar] [CrossRef]
- White, P.A.; Oliveira, R.C.; Oliveira, A.P.; Serafini, M.R.; Araújo, A.A.; Gelain, D.P.; Moreira, J.C.; Almeida, J.R.; Quintans, J.S.; Quintans-Junior, L.J.; et al. Antioxidant activity and mechanisms of action of natural compounds isolated from lichens: A systematic review. Molecules 2014, 19, 14496–14527. [Google Scholar] [CrossRef] [PubMed]
- Qi, J.H.; Dong, F.X. The relevant targets of anti-oxidative stress: A review. J. Drug Target. 2021, 29, 677–686. [Google Scholar] [CrossRef] [PubMed]
- Neha, K.; Haider, M.R.; Pathak, A.; Yar, M.S. Medicinal prospects of antioxidants: A review. Eur. J. Med. Chem. 2019, 178, 687–704. [Google Scholar] [CrossRef]
- Aoyama, S.; Mamoto, Y. Antioxidant activity and flavonoid content of welsh onion (Allium fistulosum) and the effect of thermal treatment. Food Sci. Technol. Res. 2007, 13, 67–72. [Google Scholar] [CrossRef]
- EI-Hadidy, E.M.; Mossa, M.E.A.; Habashy, H.N. Effect of freezing on the pungency and antioxidants activity in leaves and bulbs of green onion in Giza 6 and Photon varieties. Ann. Agric. Sci. Cairo 2014, 59, 33–39. [Google Scholar] [CrossRef]
- Zhao, C.; Wang, Z.; Cui, R.; Su, L.; Sun, X.; Borras-Hidalgo, O.; Li, K.; Wei, J.; Yue, Q.; Zhao, L. Effects of nitrogen application on phytochemical component levels and anticancer and antioxidant activities of Allium fistulosum. Peer J. 2021, 9, e11706. [Google Scholar] [CrossRef]
- Medina-Jaramillo, C.; Gomez-Delgado, E.; López-Córdoba, A. Improvement of the ultrasound-assisted extraction of polyphenols from welsh onion (Allium fistulosum) leaves using response surface methodology. Foods 2022, 11, 2425. [Google Scholar] [CrossRef]
- Harasym, J.; Oledzki, R. Effect of fruit and vegetable antioxidants on total antioxidant capacity of blood plasma. Nutrition 2014, 30, 511–517. [Google Scholar] [CrossRef]
- Marcelin, G.; Silveira, A.L.M.; Martins, L.B.; Ferreira, A.V.; Clément, K. Deciphering the cellular interplays underlying obesity-induced adipose tissue fibrosis. J. Clin. Investig. 2019, 129, 4032–4040. [Google Scholar] [CrossRef]
- Seravalle, G.; Grassi, G. Obesity and hypertension. Pharmacol. Res. 2017, 122, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Talaat, M.; Zayed, B.; Tolba, S.; Abdou, E.; Gomaa, M.; Itani, D.; Hutin, Y.; Hajjeh, R. Increasing antimicrobial resistance in World Health Organization Eastern Mediterranean Region, 2017-2019. Emerg. Infect. Dis. 2022, 28, 717–724. [Google Scholar] [CrossRef]
- Vinodhini, S.; Vithiya, B.S.M.; Prasad, T.A.A. Green synthesis of silver nanoparticles by employing the Allium fistulosum, Tabernaemontana divaricate and Basella alba leaf extracts for antimicrobial applications. J. King Saud. Univ. Sci. 2022, 34, 101939. [Google Scholar] [CrossRef]
- Cha, H.S.; Seong, K.S.; Kim, S.H.; Seo, J.W.; Park, S.J.; Kim, S.I.; Han, D.U. Protective effects of welsh onion (Allium fistulosum L.) on drug-induced hepatotoxicity in rats. J. Korean Soc. Food Sci. Nutr. 2005, 34, 9. [Google Scholar]
- Lee, Y.H.; Yang, H.; Lee, S.R.; Kwon, S.W.; Hong, E.J.; Lee, H.W. Welsh Onion root (Allium fistulosum) restores ovarian functions from letrozole induced-polycystic ovary syndrome. Nutrients 2018, 10, 1430. [Google Scholar] [CrossRef] [PubMed]
- Yang, H.J.; Kim, M.J.; Qiu, J.Y.; Zhang, T.; Wu, X.; Jang, D.J.; Park, S. Rice porridge containing welsh onion root water extract alleviates osteoarthritis-related pain behaviors, glucose levels, and bone metabolism in osteoarthritis-induced ovariectomized rats. Nutrients 2019, 11, 1503. [Google Scholar] [CrossRef]
- Lee, J.B.; Miyake, S.; Umetsu, R.; Hayashi, K.; Chijimatsu, T.; Hayashi, T. Anti-influenza a virus effects of fructan from welsh onion (Allium fistulosum L.). Food Chem. 2012, 134, 2164–2168. [Google Scholar] [CrossRef]
| Bioactivities | Extraction Method (Extract) | Model | Molecular Mechanisms | References |
|---|---|---|---|---|
| Anti-inflammatory activities | The aqueous extract of A. fistulosum | LPS-activated macrophages | NO↓ | [35] |
| The aqueous extract of A. fistulosum | LPS-activated RAW 264.7 | NO↓, iNOS↓ | [36] | |
| The water extract of A. fistulosum | Carrageenan-induced edema in mice | NO↓, iNOS↓, TNF-α↓ | [37] | |
| Water extract from the whole A. fistulosum Ethanol extract from the whole A. fistulosum Water extract from the root of A. fistulosum Ethanol extract from the root of A. fistulosum | LPS-stimulated BV2 | iNOS↓, COX-2↓, TNF-α↓, IL-6↓, IL-1β↓ | [38] | |
| Aqueous extracts of Welsh onion green leaves | LPS-activated RAW 264.7 | NF-κB↓, NO↓, iNOS↓, COX-2↓ | [39] | |
| Anti-cancer activities | Hot water extract of scallion Cold water extract of scallion Ethanol extract of scallion | A mouse model of colon carcinoma (CT-26 cells) | cyclin D1↓, c-Myc↓, MMP-9↓, ICAM↓, VEGF↓, HIF-1α↓, iNOS↓, COX-2↓, IL-6↓, TNF-α↓ | [21] |
| Anti-cancer activities | Aqueous extracts of Welsh onion | DLD-1, MDA-MB-231, MCF-7, SK-MES-1 | Bax↑, Bcl-2↓, Caspase 3↑ | [40] |
| Alliin, Allicin, Quercetin | MCF-7 and HCC-70 | Caspase 3↑, Caspase 8↑, Caspase 9↑, P21↑, NOXA↑, Bax↑, ΔΨm↓, Bcl-xl↓ | [41] | |
| Quercetin | Breast cancer cell lines (MCF-7, MDA-MB-231, HBL100 and BT549) | β-catenin↓, HIF-1α↓, Caspase 3↑, p-Akt↓, p-mTor↓, p-ERK↓ | [42] | |
| Antioxidant activities | Water-soluble extract of Welsh onion Lipid-soluble extract of Welsh onion | Rats fed a high-fat, high-sucrose diet | angiotensin II↓, NADH/NADPH↓, TBARS↓, | [23] |
| Aqueous extract of Welsh onion green leaves | — | NO↓, O2−↓, ·OH↓ | [43] | |
| Aqueous extract of Welsh onion green leaves | Macrophage cell line (RAW 264.7) | LDL↓, NF-κB↓, NO↓, iNOS↓, COX-2↓ | [39] | |
| Rice wine extracts of Taiwanese Allium fistulosum | DPPH ethanolic solution; ABTS solution diluted with water | — | [44] | |
| Anti-obesity activities | 70% ethanol extract from Allium fistulosum L. | High-fat, diet-induced obese mice | SREBP1c↓, PPARγ↓, FAS↓ | [22] |
| Welsh onions were crushed and heated to denature enzyme activity, freeze dried, and ground | High-fat, high-sugar fed rats | — | [45] | |
| Anti-obesity activities | The aqueous and ethanolic extracts from the Allium fistulosum | High-fat, diet-induced obese mice | IGF-1↓, PPAR-γ↓, AMPK-α↑, UCP-2↑ | [46] |
| 70% ethanol extract from Welsh onion | HepG2 cells | SREBP2↓, LDLR↓, PCSK9↓, HNF1α↓ | [47] | |
| Antibacterial activities | Extraction separation | Gram-positive and Gram-negative (Candida cyclic, Bacillus subtilis, Escherichia coli and Staphylococcus aureus) | — | [48,49,50] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Xie, T.; Wu, Q.; Lu, H.; Hu, Z.; Luo, Y.; Chu, Z.; Luo, F. Functional Perspective of Leeks: Active Components, Health Benefits and Action Mechanisms. Foods 2023, 12, 3225. https://doi.org/10.3390/foods12173225
Xie T, Wu Q, Lu H, Hu Z, Luo Y, Chu Z, Luo F. Functional Perspective of Leeks: Active Components, Health Benefits and Action Mechanisms. Foods. 2023; 12(17):3225. https://doi.org/10.3390/foods12173225
Chicago/Turabian StyleXie, Tiantian, Qi Wu, Han Lu, Zuomin Hu, Yi Luo, Zhongxing Chu, and Feijun Luo. 2023. "Functional Perspective of Leeks: Active Components, Health Benefits and Action Mechanisms" Foods 12, no. 17: 3225. https://doi.org/10.3390/foods12173225
APA StyleXie, T., Wu, Q., Lu, H., Hu, Z., Luo, Y., Chu, Z., & Luo, F. (2023). Functional Perspective of Leeks: Active Components, Health Benefits and Action Mechanisms. Foods, 12(17), 3225. https://doi.org/10.3390/foods12173225






