Quality Attributes and Sensory Acceptance of Different Botanical Coffee Co-Products
Abstract
1. Introduction
2. Material and Methods
2.1. Roasting Process
2.2. Grinding and Extract Preparation
2.3. Determination of Water Activity (aw)
2.4. Determination of pH
2.5. Determination of Dry Matter
2.6. Determination of Total Antioxidant Capacity (TAC)
- -
- A0 is the initial absorbance of the DPPH solution;
- -
- As is the absorbance of ethanol (blank);
- -
- At is the absorbance after 10 min.
2.7. Determination of Total Polyphenols Content (TPC)
2.8. Determination of Fat and Fatty Acids Profile
Preparation of Fatty Acid Methyl Esters (FAME)
2.9. Determination of Fiber
- -
- R1 = weight of residue 1;
- -
- R2 = weight of residue 2;
- -
- ml = weight of sample 1;
- -
- m2 = weight of sample 2;
- -
- A = ash content from the R1 residue;
- -
- p = protein content of the R2 residue;
- -
- B = blank.
2.10. Determination of Chlorogenic Acids and Caffeine by HPLC-DAD
2.11. Color Determination
2.12. Sensory Analysis
- -
- X = average score awarded by evaluators;
- -
- N = highest score awarded by evaluators.
2.13. Statistical Analysis
3. Results and Discussion
3.1. Water Activity (aw)
3.2. pH
3.3. Dry Matter
3.4. Total Antioxidant Capacity (TAC) and Total Polyphenols Content (TPC)
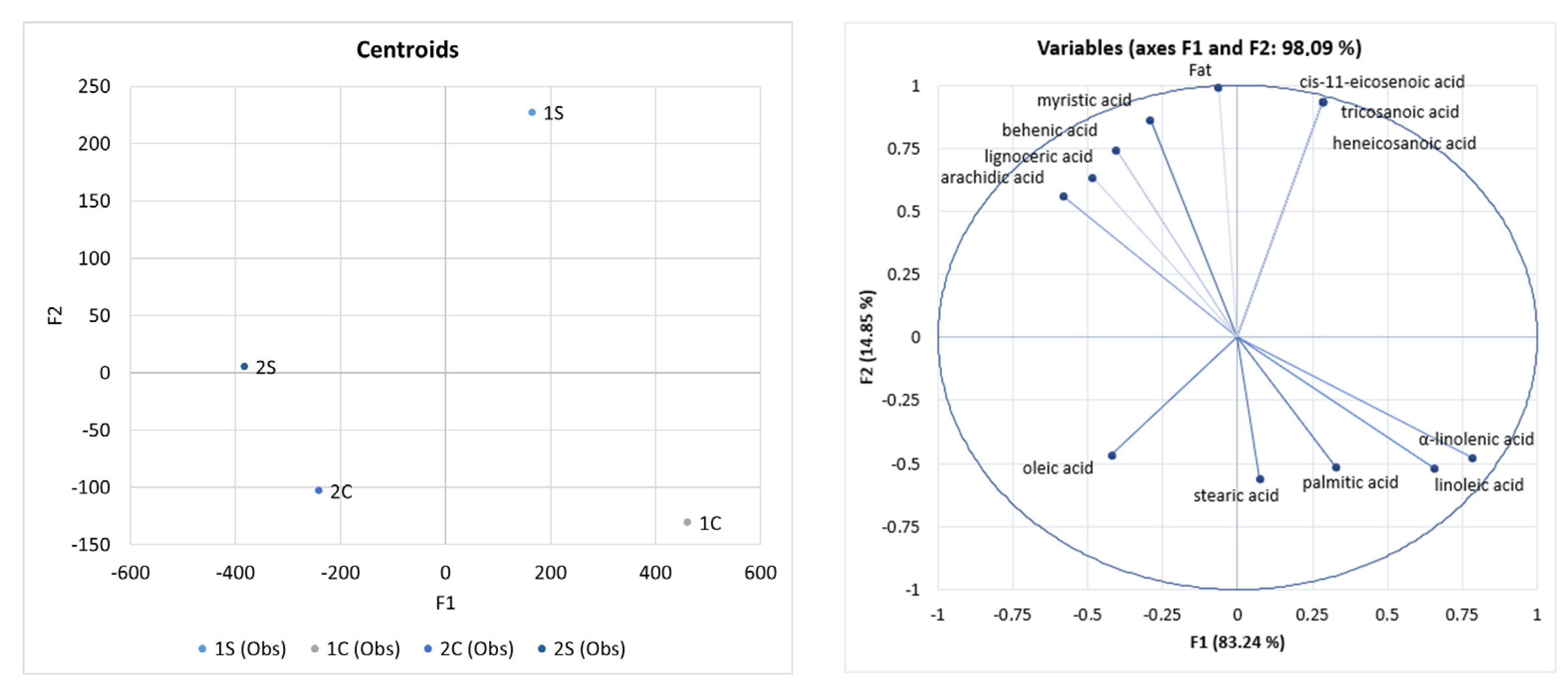
3.5. Fat and Fatty Acid
3.6. Fiber Content
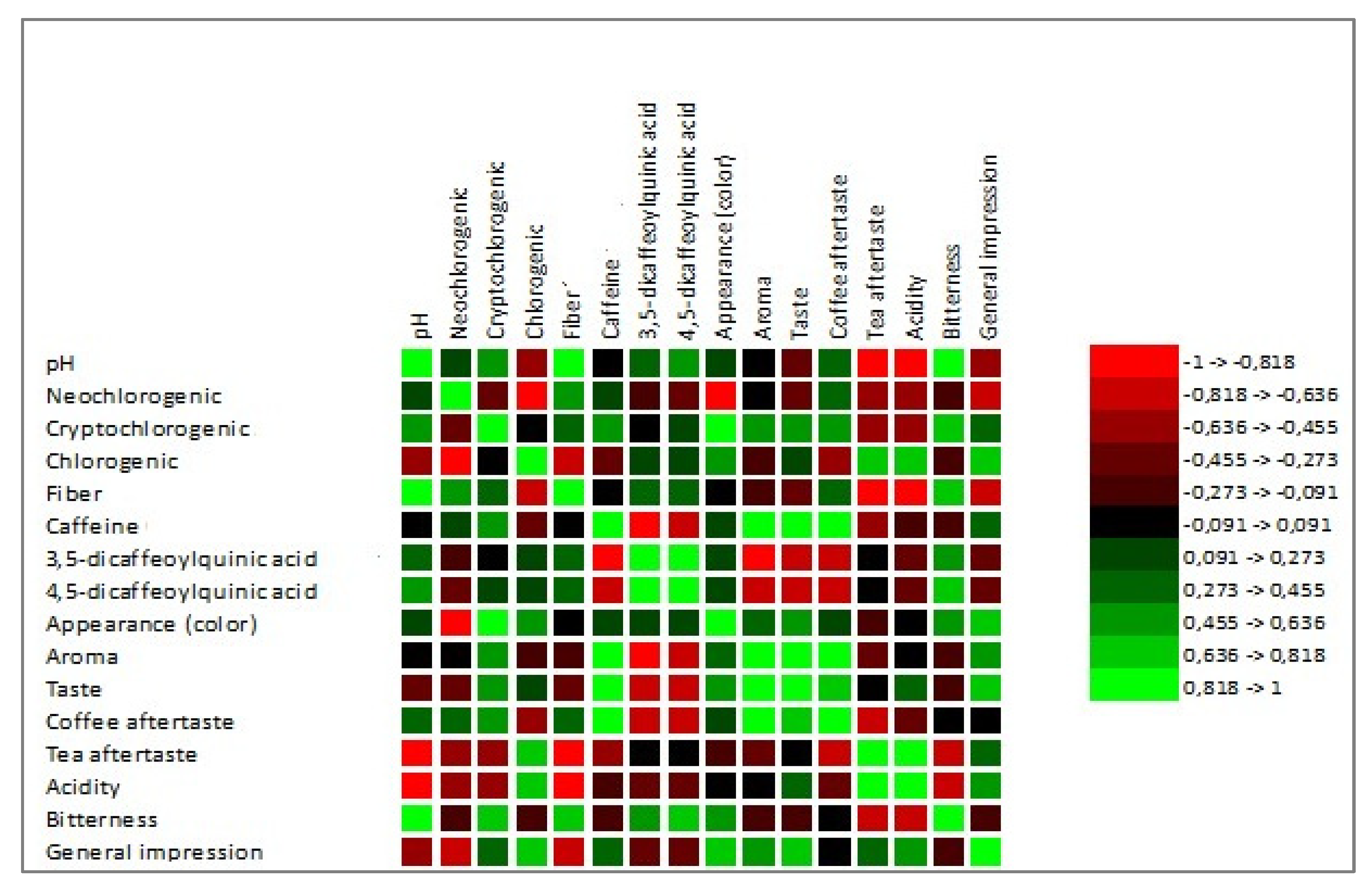
3.7. Chlorogenic Acids (CGAs) and Caffeine
3.8. Color Determination
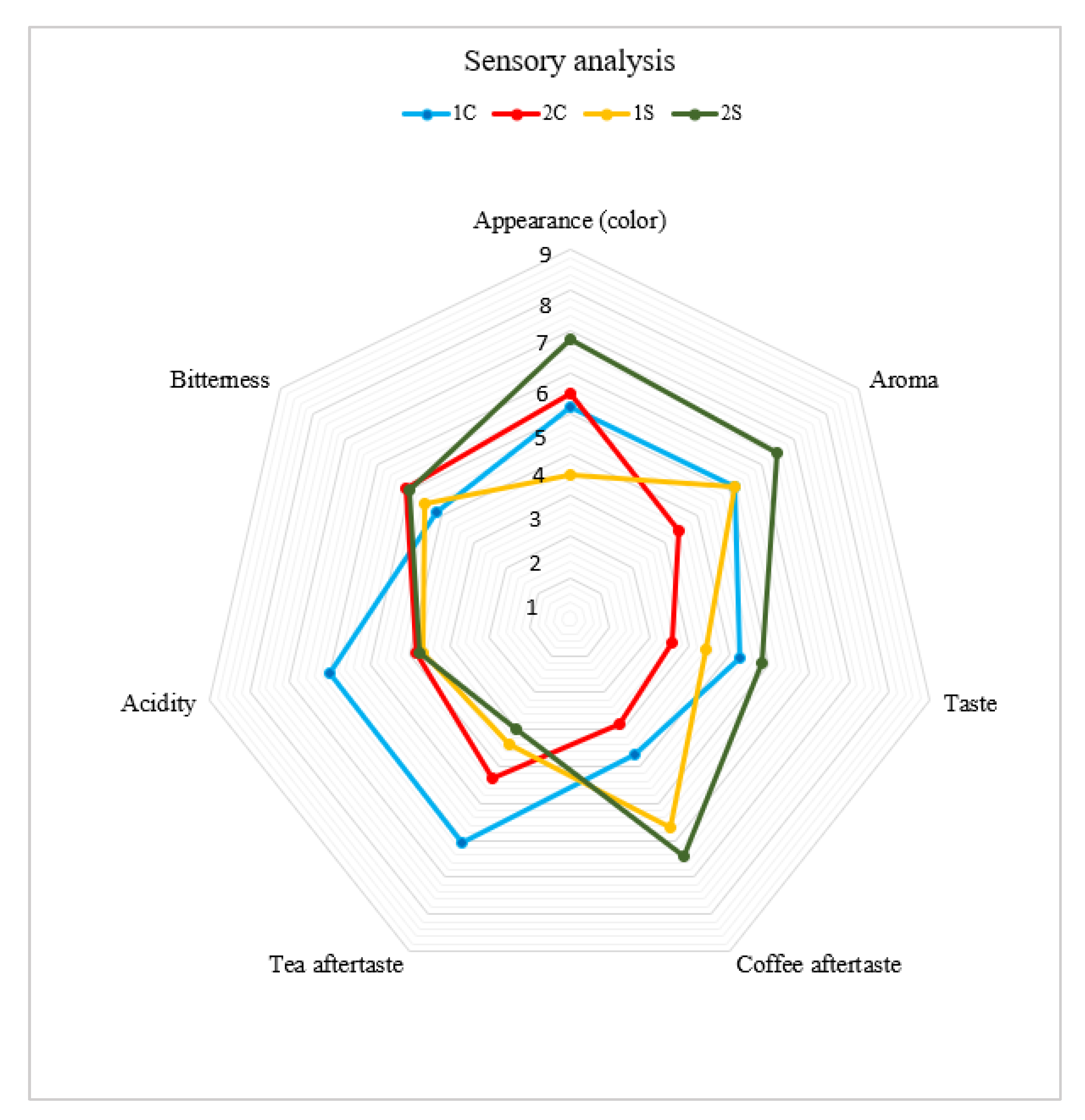
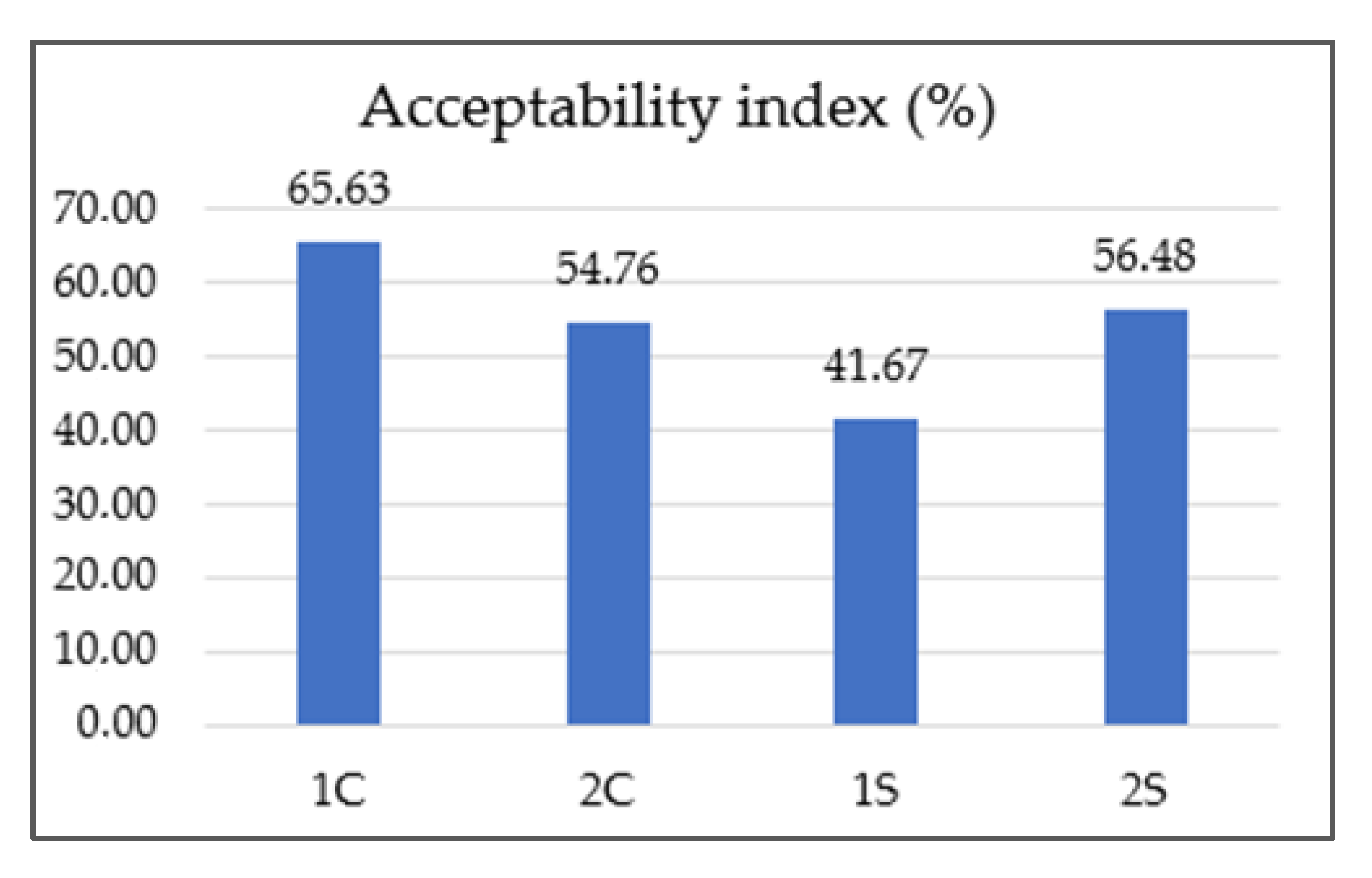
3.9. Sensory Analysis
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
References
- Toledo, C.D. Coffee-Production and Research; IntechOpen: London, UK, 2020; p. 170. ISBN 978-1-83880-886-0. [Google Scholar] [CrossRef]
- Kaplinsky, R. Competitions Policy and the Global Coffee and Cocoa Value Chains. Paper Prepared for United Nations Conference for Trade and Development (UNCTAD). 2004, p. 31. Available online: http://hubrural.org/IMG/pdf/rkaplinskycocoacoffee05.pdf (accessed on 29 April 2023).
- Gocmen, D.; Sahan, Y.; Yildiz, E.; Coskun, M.; Aroufai, İ.A. Use of coffee silverskin to improve the functional properties of cookies. J. Food Sci. Technol. 2019, 56, 2979–2988. [Google Scholar] [CrossRef] [PubMed]
- Behrouzian, F.; Amini, A.M.; Alghooneh, A.; Razavi, S.M.A. Characterization of dietary fiber from coffee silverskin: An optimization study using response surface methodology. Bioact. Carbohydr. Diet. Fibre 2016, 8, 58–64. [Google Scholar] [CrossRef]
- Hu, S.; Gil-Ramírez, A.; Martín-Trueba, M.; Benítez, V.; Aguilera, Y.; Martín-Cabrejas, M.A. Valorization of coffee pulp as bioactive food ingredient by sustainable extraction methodologies. Curr. Res. Food Sci. 2023, 6, 100475. [Google Scholar] [CrossRef] [PubMed]
- Mandura, A.; Cazalens, E.; Komes, D.; Vojvodić Cebin, A.; Pudić, R.; Šeremet, D.; Natucci Pasquino, M. The assesement of bioactive potential and sensory acceptability of coffee and its byproducts-cascara and silverskin. Hrvat. Časopis Za Prehrambenu Tehnol. Biotehnol. I Nutr. 2021, 16, 35–40. [Google Scholar] [CrossRef]
- Rodrigues da Silva, M.; Sanchez Bragagnolo, F.; Lajarim Carneiro, R.; de Oliveira Carvalho Pereira, I.; Aquino Ribeiro, J.A.; Martins Rodrigues, C.; Jelley, R.E.; Fedrizzi, B.; Soleo Funari, C. Metabolite characterization of fifteen by-products of the coffee production chain: From farm to factory. Food Chem. 2022, 369, 130753. [Google Scholar] [CrossRef]
- Ferreira, T.; Shuler, J.; Guimarães, R.; Farah, A. CHAPTER 1. Introduction to Coffee Plant and Genetics. In Coffee: Production, Quality and Chemistry; Royal Society of Chemistry: London, UK, 2019; pp. 1–25. [Google Scholar] [CrossRef]
- Iriondo-DeHond, A.; Iriondo-DeHond, M.; del Castillo, M.D. Applications of Compounds from Coffee Processing By-Products. Biomolecules 2020, 10, 1219. [Google Scholar] [CrossRef]
- Esquivel, P.; Jiménez, V.M. Functional properties of coffee and coffee by-products. Food Res. Int. 2012, 46, 488–495. [Google Scholar] [CrossRef]
- Murthy, P.S.; Naidu, M. Extraction, Characterization and Bioactivity of Coffee Anthocyanins. Eur. J. Biol. Sci. 2012, 4, 13–19. [Google Scholar] [CrossRef]
- Costa, A.S.G.; Alves, R.C.; Vinha, A.F.; Costa, E.; Costa, C.S.G.; Nunes, M.A.; Almeida, A.A.; Santos-Silva, A.; Oliveira, M.B.P.P. Nutritional, chemical and antioxidant/pro-oxidant profiles of silverskin, a coffee roasting by-product. Food Chem. 2018, 267, 28–35. [Google Scholar] [CrossRef]
- Gemechu, F.G. Embracing nutritional qualities, biological activities and technological properties of coffee byproducts in functional food formulation. Trends Food Sci. Technol. 2020, 104, 235–261. [Google Scholar] [CrossRef]
- Bobková, A.; Poláková, K.; Demianová, A.; Belej, Ľ.; Bobko, M.; Jurčaga, L.; Gálik, B.; Novotná, I.; Iriondo-DeHond, A.; del Castillo, M.D. Comparative Analysis of Selected Chemical Parameters of Coffea arabica, from Cascara to Silverskin. Foods 2022, 11, 1082. [Google Scholar] [CrossRef] [PubMed]
- Brand-Williams, W.; Cuvelier, M.E.; Berset, C. Use of a free radical method to evaluate antioxidant activity. LWT Food Sci. Technol. 1995, 28, 25–30. Available online: https://www.sciencedirect.com/science/article/abs/pii/S0023643895800085 (accessed on 29 April 2023). [CrossRef]
- Fu, L.; Xu, B.-T.; Xu, X.-R.; Gan, R.-Y.; Zhang, Y.; Xia, E.-Q.; Li, H.-B. Antioxidant capacities and total phenolic contents of 62 fruits. Food Chem. 2011, 129, 345–350. Available online: https://www.sciencedirect.com/science/article/pii/S0308814611006455 (accessed on 29 April 2023). [CrossRef] [PubMed]
- Luque de Castro, M.D.; Priego-Capote, F. Soxhlet extraction: Past and present panacea. J. Chromatogr. A 2010, 1217, 2383–2389. [Google Scholar] [CrossRef]
- International Organization for Standardization. Animal and Vegetable Fats and Oils—Gas Chromatography of Fatty Acid Me-thyl Esters—Part 2: Preparation of Methyl Esters of Fatty Acids (ISO/DIS Standard No. 12966-2). 2017. Available online: https://www.iso.org/standard/72142.html (accessed on 29 April 2023).
- Decree of the Ministry of Agriculture of the Slovak Republic of 23 August 2004 No. 2145/2004-100, Which Amends the Decree of the Ministry of Agriculture of the Slovak Republic of 7 October 1997 no. 1497/4/1997-100 on Official Sampling and Laboratory Testing and Evaluation of Feed, as Amended by the Decree of the Ministry of Agriculture of the Slovak Republic of 12 February 2003 no. 149/2/2003-100. Available online: https://www.mpsr.sk/index.php?navID=126&year=2004 (accessed on 29 April 2023).
- AACC. International Approved Methods Technical Committee Report: Collaborative Study on Determination of Total Dietary Fiber (Digestion-Resistant Carbohydrates per Codex Definition) by a Rapid Enzymatic-Gravimetric Method and Liquid Chromatography. Cereal Foods World 2018, 63, 80–84. [Google Scholar] [CrossRef]
- Bobková, A.; Demianová, A.; Belej, Ľ.; Harangozo, Ľ.; Bobko, M.; Jurčaga, L.; Poláková, K.; Božiková, M.; Bilčík, M.; Árvay, J. Detection of Changes in Total Antioxidant Capacity, the Content of Polyphenols, Caffeine, and Heavy Metals of Teas in Relation to Their Origin and Fermentation. Foods 2021, 10, 1821. [Google Scholar] [CrossRef]
- CIE 041-1978; Light as a True Visual Quantity: Principles of Measurement. Commission Internationale de L’Eclairage: Vienna, Austria, 1978. [CrossRef]
- Barahona, I.; Sanmiguel Jaimes, E.M.; Yang, J. Sensory attributes of coffee beverages and their relation to price and package information: A case study of Colombian customers’ preferences. Food Sci. Nutr. 2020, 8, 1173–1186. [Google Scholar] [CrossRef]
- Pereira, L.L.; Guarçoni, R.C.; da Luz, J.M.R.; de Oliveira, A.C.; Moreli, A.P.; Filete, C.A.; Paiva, G.; Debona, D.G.; Gomes, W.d.S.; Cardoso, W.S.; et al. Impacts of brewing methods on sensory perception and organoleptic compounds of coffee. Food Chem. Adv. 2023, 2, 100185. [Google Scholar] [CrossRef]
- de Sousa, L.H.B.P.; da Luz, J.M.R.; da Silva, M.d.C.S.; Moreli, A.P.; Veloso, T.G.R.; Guarçoni, R.C.; Moreira, T.R.; Barros, M.V.P.; Kasuya, M.C.M.; Marcate, J.P.P.; et al. Relationship between sensory and microbial profiles of fermented coffee by dry and washed methods. Food Chem. Adv. 2023, 2, 100259. [Google Scholar] [CrossRef]
- Riandani, A.P.; Prangdimurti, E.; Herawati, D. Profiling the chemical and sensory properties of cascara beverages from different locations in Indonesia. Food Res. 2022, 6, 388–398. [Google Scholar] [CrossRef]
- Meilgaard, M.; Vance Civille, G.; Thomas Carr, B. Sensory Evaluation Techniques, 3rd ed.; CRC Press: Boca Raton, FL, USA, 1999. [Google Scholar] [CrossRef]
- DePaula, J.; Cunha, S.C.; Cruz, A.; Sales, A.L.; Revi, I.; Fernandes, J.; Ferreira, I.M.P.L.V.O.; Miguel, M.A.L.; Farah, A. Volatile Fingerprinting and Sensory Profiles of Coffee Cascara Teas Produced in Latin American Countries. Foods 2022, 11, 3144. [Google Scholar] [CrossRef] [PubMed]
- XLSTAT Addinsoft, Statistical and Data Analysis Solution; XLSTAT, PREMIUM Verion: New York, NY, USA, 2021.
- Zhang, D.; Lu, G. Shape-based image retrieval using generic Fourier descriptor. Signal Processing: Image Communication. Signal Process. Image Commun. 2002, 17, 825–848. [Google Scholar] [CrossRef]
- Kumar, A. PCA vs LDA Differences, Plots, Examples. 2023. Available online: https://vitalflux.com/pca-vs-lda-differences-plots-examples/ (accessed on 13 April 2023).
- Troller, J.A.; Christian, J.H.B. Water Activity—Basic Concepts. In Water Activity and Food; Elsevier: Amsterdam, The Netherlands, 1978; pp. 1–12. [Google Scholar] [CrossRef]
- Slade, L.; Levine, H.; Reid, D.S. Beyond water activity: Recent advances based on an alternative approach to the assessment of food quality and safety. Critical Reviews. Food Sci. Nutr. 1991, 30, 115–360. [Google Scholar] [CrossRef] [PubMed]
- Bicho, N.C.; Leitão, A.E.; Ramalho, J.C.; De Alvarenga, N.B.; Lidon, F.C. Identification of nutritional descriptors of roasting intensity in beverages of Arabica and Robusta coffee beans. Int. J. Food Sci. Nutr. 2011, 62, 865–871. [Google Scholar] [CrossRef]
- Martuscelli, M.; Esposito, L.; Mastrocola, D. The Role of Coffee Silver Skin against Oxidative Phenomena in Newly Formulated Chicken Meat Burgers after Cooking. Foods 2021, 10, 1833. [Google Scholar] [CrossRef]
- Lorbeer, L.; Schwarz, S.; Franke, H.; Lachenmeier, D.W. Toxicological Assessment of Roasted Coffee Silver Skin (Testa of Coffea sp.) as Novel Food Ingredient. Molecules 2022, 27, 6839. [Google Scholar] [CrossRef]
- Murlida, E.; Noviasari, S.; Nilda, C.; Rohaya, S.; Rahmi, F.; Muzaifa, M. Chemical characteristics of cascara tea from several varieties of coffee in Aceh Province. IOP Conf. Ser. Earth Environ. Sci. 2021, 667, 012078. [Google Scholar] [CrossRef]
- Prono-Widayat, H.; Arpi, N.; Andini, R.; Muzaifa, M.; Gunawan, F. Chemical analysis of cascara tea from wine coffee processing with a different fermentation time. IOP Conf. Ser. Earth Environ. Sci. 2021, 667, 012104. [Google Scholar] [CrossRef]
- Maharani, S.; Mustikawati, I.; Nailufhar, L.; Istiqomah, S. The effect of brewing time on pH values, polyphenols content, and antioxidant activities of coffee husk tea (cascara tea). J. Phys. Conf. Ser. 2021, 1869, 012050. [Google Scholar] [CrossRef]
- Gottstein, V.; Bernhardt, M.; Dilger, E.; Keller, J.; Breitling-Utzmann, C.M.; Schwarz, S.; Kuballa, T.; Lachenmeier, D.W.; Bunzel, M. Coffee Silver Skin: Chemical Characterization with Special Consideration of Dietary Fiber and Heat-Induced Contaminants. Foods 2021, 10, 1705. [Google Scholar] [CrossRef]
- Bessada, S.M.F.; Alves, R.C.; Costa, A.S.G.; Nunes, M.A.; Oliveira, M.B.P.P. Coffea canephora silverskin from different geographical origins: A comparative study. Sci. Total Environ. 2018, 645, 1021–1028. [Google Scholar] [CrossRef] [PubMed]
- Bertolino, M.; Barbosa-Pereira, L.; Ghirardello, D.; Botta, C.; Rolle, L.; Guglielmetti, A.; Borotto Dalla Vecchia, S.; Zeppa, G. Coffee silverskin as nutraceutical ingredient in yogurt: Its effect on functional properties and its bioaccessibility. J. Sci. Food Agric. 2019, 99, 4267–4275. [Google Scholar] [CrossRef] [PubMed]
- EFSA. Technical Report on the notification of dried cherry pulp from Coffea arabica L. and Coffea canephora Pierre ex A. Froehner as a traditional food from a third country pursuant to Article 14 of Regulation (EU) 2015/2283. EFSA Support. Publ. 2021, 18, 6808E. [Google Scholar] [CrossRef]
- Braham, J.E.; Bressami, R. Coffee Pulp Composition, Technology, and Utilization; Institute of Nutrition of Central America and Panama: Guatemala City, Guatemala, 1979; ISBN 0-88936-190-8; Available online: https://doanchuyennganh.weebly.com/uploads/5/3/5/8/53582365/book_coffee_pulp_composition_technology_and_utilization.pdf (accessed on 29 April 2023)ISBN 0-88936-190-8.
- Sholichah, E.; Desnilasari, D.; Subekti, R.J.; Karim, M.A.; Purwono, B. The influence of coffee cherry fermentation on the properties of Cascara arabica from Subang, West Java. IOP Conf. Ser. Mater. Sci. Eng. 2021, 1011, 012006. [Google Scholar] [CrossRef]
- Arpi, N.; Muzaifa, M.; Sulaiman, M.I.; Andini, R.; Kesuma, S.I. Chemical Characteristics of Cascara, Coffee Cherry Tea, Made of Various Coffee Pulp Treatments. IOP Conf. Ser. Earth Environ. Sci. 2021, 709, 012030. [Google Scholar] [CrossRef]
- Angeloni, S.; Scortichini, S.; Fiorini, D.; Sagratini, G.; Vittori, S.; Neiens, S.D.; Steinhaus, M.; Zheljazkov, V.D.; Maggi, F.; Caprioli, G. Characterization of Odor-Active Compounds, Polyphenols, and Fatty Acids in Coffee Silverskin. Molecules 2020, 25, 2993. [Google Scholar] [CrossRef]
- Rios, M.B.; Iriondo-DeHond, A.; Iriondo-DeHond, M.; Herrera, T.; Velasco, D.; Gómez-Alonso, S.; Callejo, M.J.; del Castillo, M.D. Effect of Coffee Cascara Dietary Fiber on the Physicochemical, Nutritional and Sensory Properties of a Gluten-Free Bread Formulation. Molecules 2020, 25, 1358. [Google Scholar] [CrossRef]
- El-Badry, A.M.; Graf, R.; Clavien, P.-A. Omega 3–Omega 6: What is right for the liver? J. Hepatol. 2007, 47, 718–725. [Google Scholar] [CrossRef]
- Mota, D.; Barbosa, M.; Schneider, J.; Lima, Á.; Pereira, M.; Krause, L.; Soares, C.M. Potential Use of Crude Coffee Silverskin Oil in Integrated Bioprocess for Fatty Acids Production. J. Am. Oil Chem. Soc. 2021, 98, 519–529. [Google Scholar] [CrossRef]
- Efhymiopoulos, I.; Hellier, P.; Ladommatos, N.; Eveleigh, A.; Mills-Lamptey, B. Factors affecting the efficiency of pressurized solvent extraction of oil from spent coffee grounds. Detritus 2019, 5, 75–83. [Google Scholar] [CrossRef]
- Giordano, M.; Bertolino, M.; Belviso, S.; Ghirardello, D.; Zeppa, G. Effects of Species, Post-Harvest Treatment, and Roasting on Fibre, Volatile Compounds, and Polyphenol Contents in Coffee Silverskin. Foods 2022, 11, 3132. [Google Scholar] [CrossRef]
- Santos, D.E.M.; Macedo, L.M.; Tundisi, L.L.; Ataide, A.J.; Camargo, A.G.; Alves, R.C.; Oliveira, M.B.; Mazzola, G.P. Coffee by-products in topical formulations. Trends Food Sci. Technol. 2021, 11, 280–291. [Google Scholar] [CrossRef]
- Rodrigues, R.; Oliveira, M.B.P.P.; Alves, R.C. Chlorogenic Acids and Caffeine from Coffee By-Products: A Review on Skincare Applications. Cosmetics. 2023, 10, 12. [Google Scholar] [CrossRef]
- Jiamjariyatam, R.; Samosorn, S.; Dolsophon, K.; Tantayotai, P.; Lorliam, W.; Krajangsang, S. Development of Cascara Tea from Coffee Cherry Pulp. J. Culin. Sci. Technol. 2020, 18. [Google Scholar] [CrossRef]
- Mindarti, S.; Zalizar, L.; Damat Saati, E.A.; Fajriani, S. Characterization of fiber fraction, physical and chemical properties of coffee flour (Coffea sp.) as functional foodstuff for diabetes mellitus patient. IOP Conf. Ser. Earth Environ. Sci. 2020, 462, 012017. [Google Scholar] [CrossRef]
- Hejna, A. Potential applications of by-products from the coffee industry in polymer technology–Current state and perspectives. Waste Manag. 2021, 121, 296–330. [Google Scholar] [CrossRef] [PubMed]
- Cantele, C.; Tedesco, M.; Ghirardello, D.; Zeppa, G.; Bertolino, M. Coffee Silverskin as a Functional Ingredient in Vegan Biscuits: Physicochemical and Sensory Properties and In Vitro Bioaccessibility of Bioactive Compounds. Foods 2022, 11, 717. [Google Scholar] [CrossRef]
- Farah, A.; de Paula Lima, J. CHAPTER 16. Chlorogenic Acids: Daily Consumption through Coffee, Metabolism and Potential Health Effects. In Coffee; Royal Society of Chemistry: London, UK, 2019; pp. 364–415. [Google Scholar] [CrossRef]
- Król, K.; Gantner, M.; Tatarak, A.; Hallmann, E. The content of polyphenols in coffee beans as roasting, origin and storage effect. Eur. Food Res. Technol. 2019, 246, 33–39. [Google Scholar] [CrossRef]
- Fuller, M.; Rao, N.Z. The Effect of Time, Roasting Temperature, and Grind Size on Caffeine and Chlorogenic Acid Concentrations in Cold Brew Coffee. Sci. Rep. 2017, 7, 17979. [Google Scholar] [CrossRef]
- Eckhardt, S.; Franke, H.; Schwarz, S.; Lachenmeier, D.W. Risk Assessment of Coffee Cherry (Cascara) Fruit Products for Flour Replacement and Other Alternative Food Uses. Molecules 2022, 27, 8435. [Google Scholar] [CrossRef]
- Xu, Y.Q.; Zou, C.; Gao, Y.; Chen, J.X.; Wang, F.; Chen, G.S.; Yin, J.F. Effect of the type of brewing water on the chemical composition, sensory quality, and antioxidant capacity of Chinese teas. Food Chem. 2017, 236, 142–151. [Google Scholar] [CrossRef]
- Ateş, G.; Elmacı, Y. Coffee silverskin as fat replacer in cake formulations and its effect on physical, chemical, and sensory attributes of cakes. LWT 2018, 90, 519–525. [Google Scholar] [CrossRef]
- Garcia-Serna, E.; Martinez-Saez, N.; Mesias, M.; Morales, F.; Castillo, M. Use of Coffee Silverskin and Stevia to Improve the Formulation of Biscuits. Pol. J. Food Nutr. Sci. 2014, 64, 243–251. [Google Scholar] [CrossRef]
- de Paula, J.; Farah, A. Phenolic compounds in coffee seeds and by-products. In Coffee Science; CRC Press: Boca Raton, FL, USA, 2022; pp. 129–147. [Google Scholar] [CrossRef]
- Abduh, M.Y.; Nofitasari, D.; Rahmawati, A.; Eryanti, A.Y.; Rosmiati, M. Effects of brewing conditions on total phenolic content, antioxidant activity and sensory properties of cascara. Food Chem. Adv. 2023, 2, 100183. [Google Scholar] [CrossRef]
- Martinez-Saez, N.; Ullate, M.; Martin-Cabrejas, M.A.; Martorell, P.; Genovés, S.; Ramon, D.; del Castillo, M.D. A novel antioxidant beverage for body weight control based on coffee silverskin. Food Chem. 2014, 150, 227–234. [Google Scholar] [CrossRef]
- Neves, J.V.G.; Borges, M.V.; Silva, D.M.; Leite, C.X.S.; Santos, M.R.C.; Lima, N.G.B.; Lannes, S.C.S.; Silva, M.V. Total phenolic content and primary antioxidant capacity of aqueous extracts of coffee husk: Chemical evaluation and beverage development. Food Sci. Technol. 2019, 39 (Suppl. S1), 348–353. [Google Scholar] [CrossRef]
- Rao, N.Z.; Fuller, M. Acidity and Antioxidant Activity of Cold Brew Coffee. Sci. Rep. 2018, 8, 16030. [Google Scholar] [CrossRef]
- Basheer, B.; Albaqami, N.N.; Almogble, E.A.; Alsaqabi, D.K.; Alkhneen, S.M.S.; Alenazi, A.S.; Ghilan, M.A.; Alangari, S.S.; Ali, R. Evaluation of Titratable Acidity and pH Level of Different Coffee Drinks—An In-vitro Study. Pak. J. Med. Health Sci. 2022, 16, 767–770. [Google Scholar] [CrossRef]
| Parameter | 1S | 2S | 1C | 2C |
|---|---|---|---|---|
| Cultivar | C. arabica | C. canephora | C. arabica | C. canephora |
| Date of roast *, harvest ** | 2021 * | 2023 * | 2020 ** | 2020 ** |
| Roasting *, post-harvest ** process | Medium * | Medium * | Natural/dry ** | Natural/dry ** |
| Geographical origin | Panama | India | Panama | India |
| Variety | mix | mix | Maragogype | Catmar |
| Altitude | - | - | 1300 mamsl | 600–850 mamsl |
| Parameter | 1S | 2S | 1C | 2C |
|---|---|---|---|---|
| aw | 0.18 ± 0.01 a | 0.28 ± 0.01 b | 0.64 ± 0.04 d | 0.39 ± 0.01 c |
| pH | 4.62 ± 0.03 b | 5.90 ± 0.02 c | 4.22 ± 0.03 a | 5.97 ± 0.02 d |
| Dry matter (%) | 92.70 ± 0.11 c | 93.91 ± 0.14 d | 85.16 ± 0.12 a | 89.60 ± 0.01 b |
| TAC (%) | 58.17 ± 1.28 d | 46.65 ± 1.20 c | 36.54 ± 1.84 a | 41.12 ± 2.11 b |
| TPC (g GAE. kg−1) | 1.64 ± 0.18 a | 11.15 ± 0.69 b | 2.59 ± 0.17 a | 49.14 ± 1.35 c |
| Fiber (%) | 64.73 ± 0.19 d | 61.06 ± 0.04 b | 37.57 ± 0.37 a | 63.75 ± 0.04 c |
| Neochlorogenic acid (mg·g−) | 0.34 ± 0.01 c | 0.11 ± 0.01 b | 0.09 ± 0.01 a | 0.12 ± 0.01 b |
| Cryptochlorogenic acid (mg·g−1) | 0.21 ± 0.09 ab | 0.80 ± 0.09 c | 0.18 ± 0.07 a | 0.38 ± 0.09 b |
| Chlorogenic acid (mg·g−1) | 0.38 ± 0.09 a | 0.50 ± 0.09 ab | 0.60 ± 0.09 b | 0.52 ± 0.09 ab |
| Caffeine (mg·g−1) | 5.77 ± 0.09 c | 8.01 ± 0.09 d | 4.12 ± 0.09 b | 0.48 ± 0.09 a |
| 3,5-dicaffeoylquinic acid (mg·g−1) | n.d. | n.d. | n.d. | 0.27 ± 0.02 d |
| 4,5-dicaffeoylquinic acid (mg·g−1) | n.d. | 0.07 ± 0.02 b | n.d. | 0.03 ± 0.01 a |
| Sample ID | PUFA | MUFA | SFA | Ratio Σn3/Σn6 | Ratio Σn6/Σn3 |
|---|---|---|---|---|---|
| 1S | 26.81 ± 0.25 c | 5.11 ± 0.27 b | 67.53 ± 0.03 b | 0.08 ± 0.01 c | 12.29 ± 0.08 a |
| 2S | 22.74 ± 0.03 a | 5.49 ± 0.17 b | 71.77 ± 0.14 a | 0.17 ± 0.01 b | 6.06 ± 0.05 b |
| 1C | 51.91 ± 0.30 a | 5.55 ± 0.07 b | 42.54 ± 0.37 d | 0.67 ± 0.02 a | 1.50 ± 0.03 c |
| 2C | 29.12 ± 0.12 b | 19.45 ± 0.79 a | 51.44 ± 0.66 c | n.d. | n.d. |
| Sample | L* (D65) | a* (D65) | b* (D65) | C* | h˚ |
|---|---|---|---|---|---|
| 1S | 43.10 ± 0.07 a | 9.48 ± 0.06 b | 25.91 ± 0.08 a | 27.58 ± 0.09 a | 69.91 ± 0.05 b |
| 2S | 36.36 ± 0.05 b | 6.38 ± 0.03 c | 19.99 ± 0.07 c | 19.08 ± 0.07 d | 70.47 ± 0.02 a |
| 1C | 30.07 ± 0.08 c | 10.44 ± 0.09 a | 19.29 ± 0.11 b | 21.93 ± 0.14 b | 61.58 ± 0.05 c |
| 2C | 28.65 ± 0.11 d | 10.35 ± 0.01 a | 17.52 ± 0.06 d | 20.34 ± 0.06 c | 59.42 ± 0.09 d |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Poláková, K.; Bobková, A.; Demianová, A.; Bobko, M.; Lidiková, J.; Jurčaga, L.; Belej, Ľ.; Mesárošová, A.; Korčok, M.; Tóth, T. Quality Attributes and Sensory Acceptance of Different Botanical Coffee Co-Products. Foods 2023, 12, 2675. https://doi.org/10.3390/foods12142675
Poláková K, Bobková A, Demianová A, Bobko M, Lidiková J, Jurčaga L, Belej Ľ, Mesárošová A, Korčok M, Tóth T. Quality Attributes and Sensory Acceptance of Different Botanical Coffee Co-Products. Foods. 2023; 12(14):2675. https://doi.org/10.3390/foods12142675
Chicago/Turabian StylePoláková, Katarína, Alica Bobková, Alžbeta Demianová, Marek Bobko, Judita Lidiková, Lukáš Jurčaga, Ľubomír Belej, Andrea Mesárošová, Melina Korčok, and Tomáš Tóth. 2023. "Quality Attributes and Sensory Acceptance of Different Botanical Coffee Co-Products" Foods 12, no. 14: 2675. https://doi.org/10.3390/foods12142675
APA StylePoláková, K., Bobková, A., Demianová, A., Bobko, M., Lidiková, J., Jurčaga, L., Belej, Ľ., Mesárošová, A., Korčok, M., & Tóth, T. (2023). Quality Attributes and Sensory Acceptance of Different Botanical Coffee Co-Products. Foods, 12(14), 2675. https://doi.org/10.3390/foods12142675







