Nutritional Quality and Safety of the Spirulina Dietary Supplements Sold on the Slovenian Market
Abstract
:1. Introduction
2. Materials and Methods
2.1. Samples
2.2. Sample Preparation
2.3. Macro-Elemental Analysis by X-ray Fluorescence Spectrometry
2.4. Iron Speciation by Fe K-Edge X-ray Absorption Near Edge Structure (XANES)
2.5. Toxic Trace Element and Se Analysis by Inductively Coupled Plasma-Mass Spectrometry
2.6. Fatty Acid Analysis by Gas Chromatography-Mass Spectrometry Method
2.6.1. Fatty Acid Extraction and Esterification
2.6.2. FAME Analysis
2.7. Amino Acid Analysis by Gas Chromatography-Mass Spectrometry
2.7.1. Liquid Phase Hydrolysis
2.7.2. Amino Acid Derivatization
2.7.3. Gas Chromatography-Mass Spectrometry Method for Amino Acid Analysis
2.8. Statistical Analysis
3. Results and Discussion
3.1. Elemental Composition
Compliance with Their Nutrient Declaration
3.2. Toxic Trace Element Content
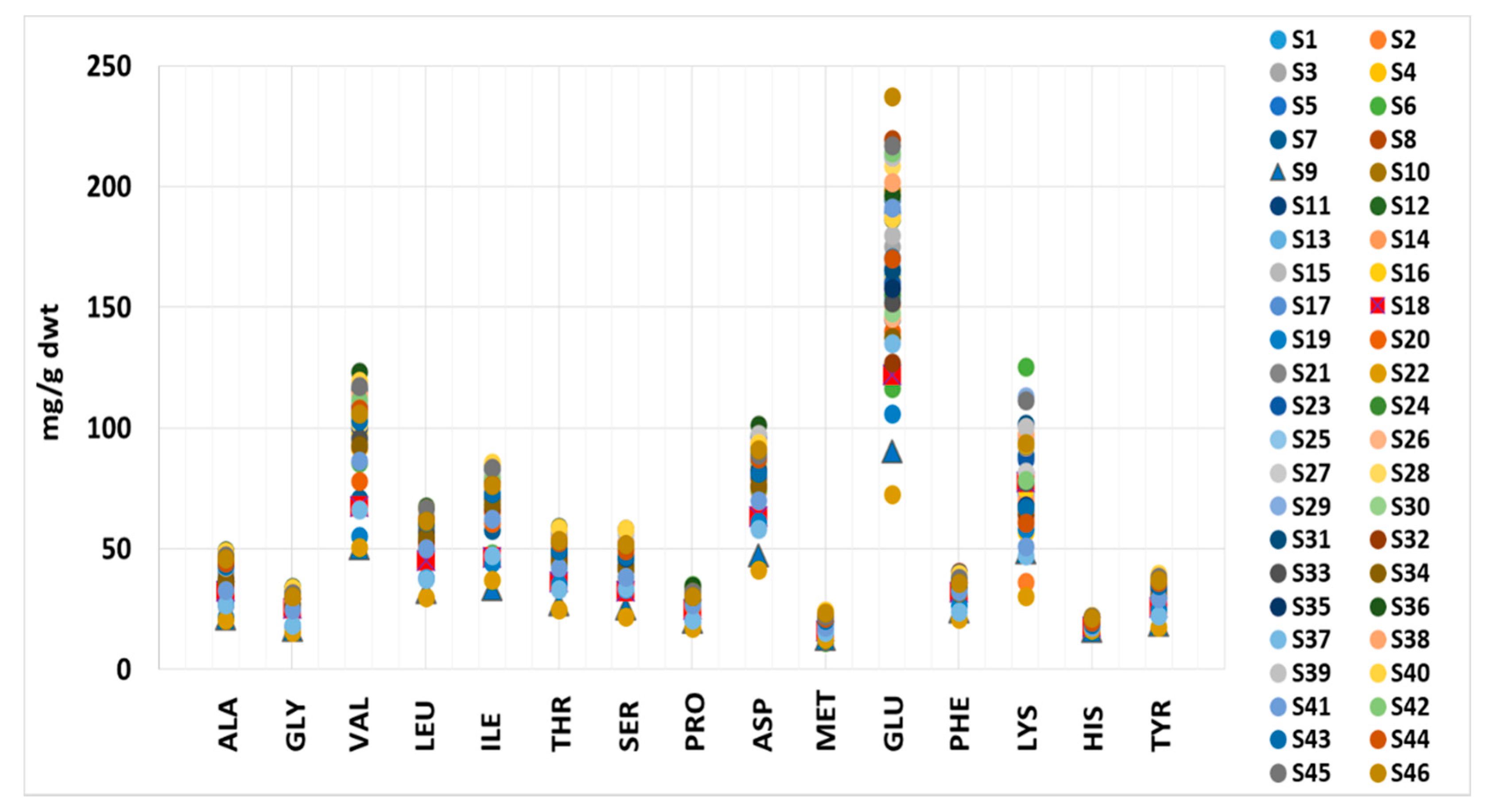
3.3. Amino Acid Content
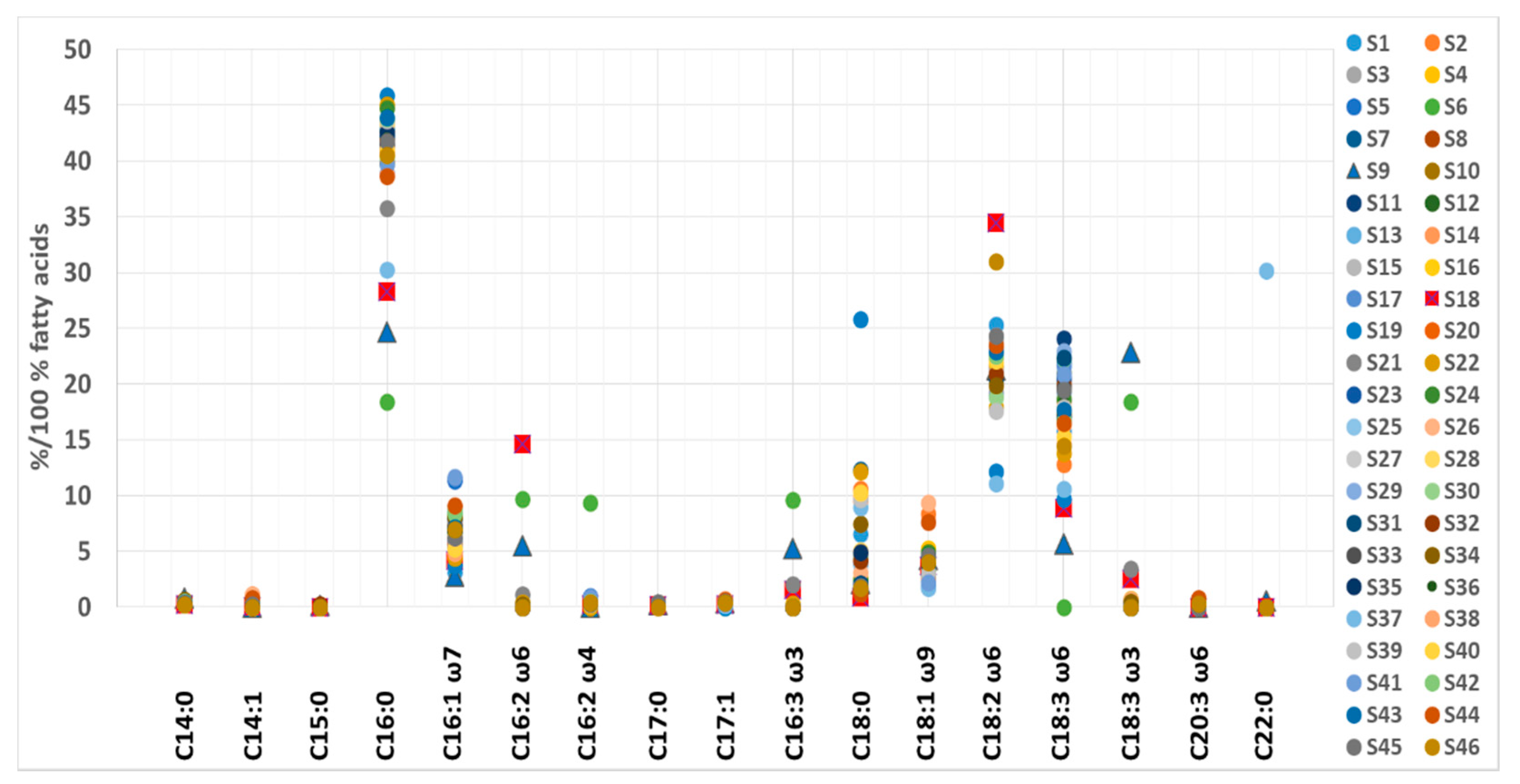
3.4. Fatty Acid Content
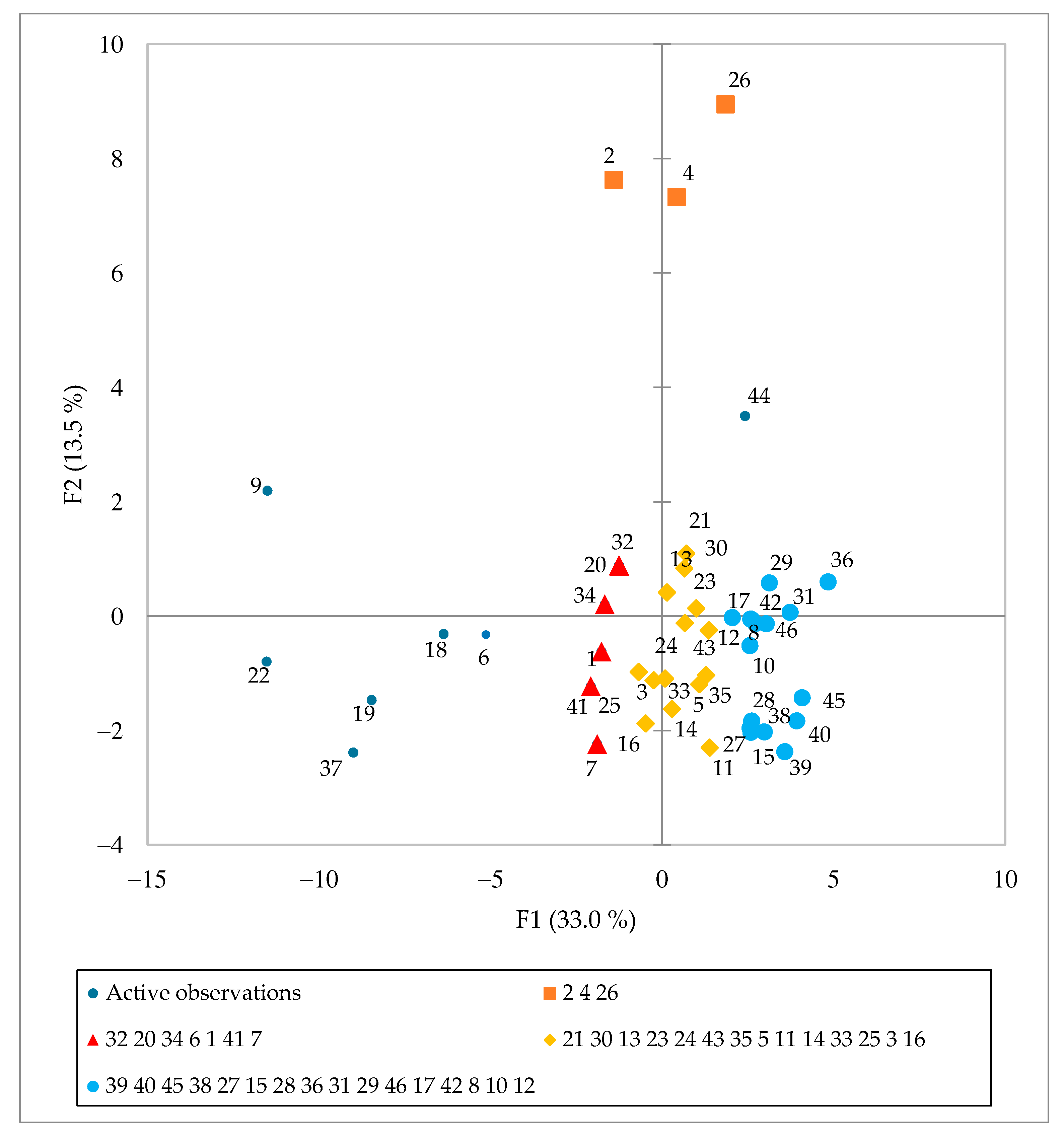
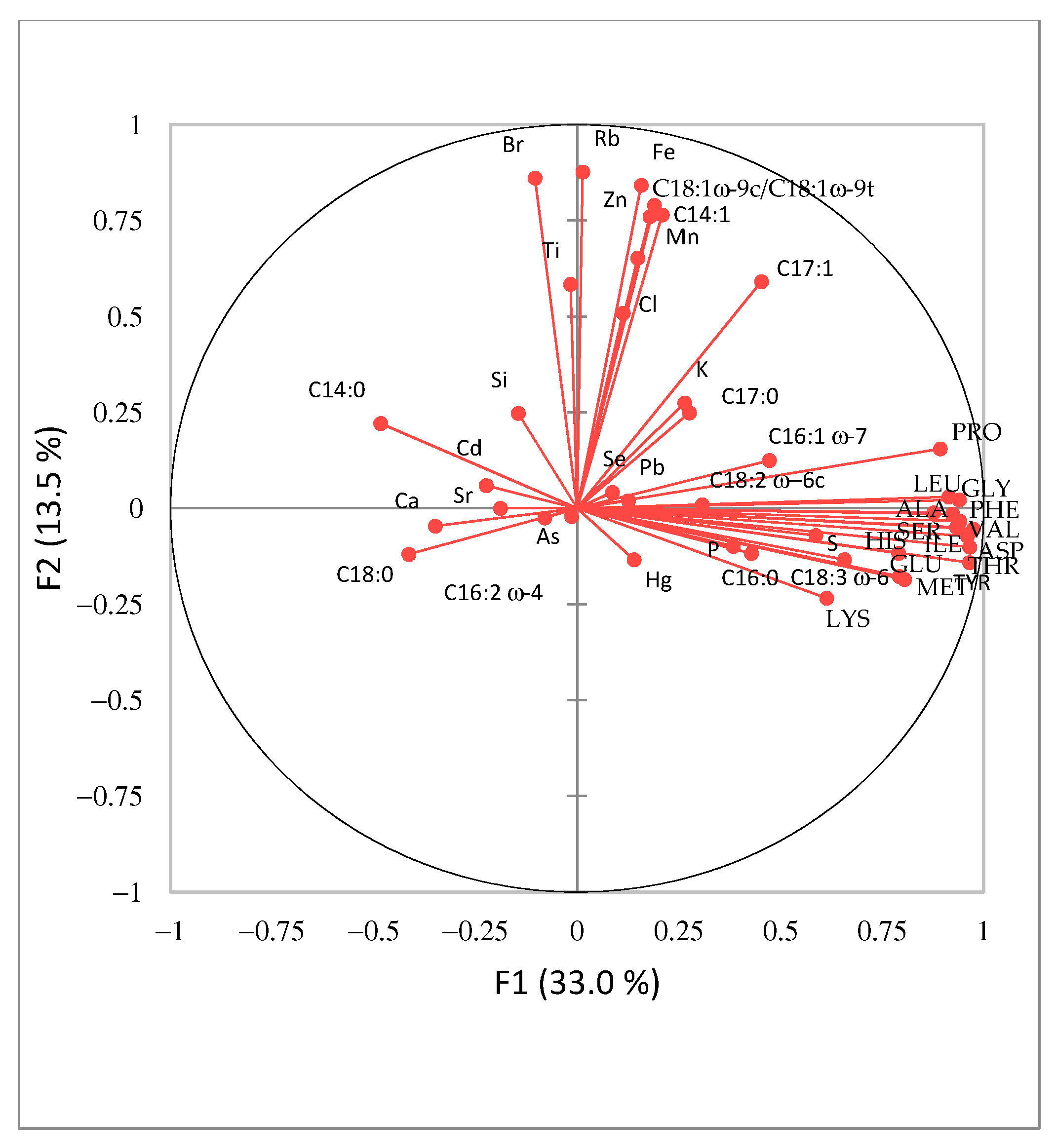
3.5. Principal Component Analysis (PCA) of Spirulina Samples from Slovenian Market Analysis Results
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Rhoades, J.D.; Kandiah, A.; Mashali, A.M. The Use of Saline Waters for Crop Production; FAO Irrigation and Drainage Paper; Food and Agriculture Organization of the United Nations: Rome, Italy, 1992; ISBN 92-5-103237-8. [Google Scholar]
- Food and Agriculture Organization of the United Nations. High Level Expert Forum-How to Feed the World in 2050; Food and Agriculture Organization of the United Nations: Rome, Italy, 2009; p. 35. [Google Scholar]
- Gardner, G.T.; Halweil, B. Underfed and Overfed: The Global Epidemic of Malnutrition; Peterson, J.A., Ed.; Worldwatch Paper; Worldwatch Institute: Washington, DC, USA, 2000; ISBN 978-1-878071-52-1. [Google Scholar]
- Sotiroudis, T.; Sotiroudis, G. Health Aspects of Spirulina (Arthrospira) Microalga Food Supplement. J. Serb. Chem. Soc. 2013, 78, 395–405. [Google Scholar] [CrossRef]
- Rogers, J.N.; Rosenberg, J.N.; Guzman, B.J.; Oh, V.H.; Mimbela, L.E.; Ghassemi, A.; Betenbaugh, M.J.; Oyler, G.A.; Donohue, M.D. A Critical Analysis of Paddlewheel-Driven Raceway Ponds for Algal Biofuel Production at Commercial Scales. Algal Res. 2014, 4, 76–88. [Google Scholar] [CrossRef] [Green Version]
- Campanella, L.; Crescentini, G.; Avino, P. Chemical Composition and Nutritional Evaluation of Some Natural and Commercial Food Products Based on Spirulina. Analusis 1999, 27, 533–540. [Google Scholar] [CrossRef]
- Chu, W.-L.; Lim, Y.-W.; Radhakrishnan, A.K.; Lim, P.-E. Protective Effect of Aqueous Extract from Spirulina Platensis against Cell Death Induced by Free Radicals. BMC Complement. Altern. Med. 2010, 10, 53. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- El-Baky, H.H.A.; El-Baz, F.K.; El-Baroty, G.S. Characterization of Nutraceutical Compounds in Blue Green Alga Spirulina Maxima. J. Med. Plant Res. 2008, 2, 292–300. [Google Scholar] [CrossRef]
- Al-Dhabi, N.A.; Arasu, M.V. Quantification of Phytochemicals from Commercial Spirulina Products and Their Antioxidant Activities. Evid.-Based Complement. Altern. Med. 2016, 2016, 13. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Capelli, B.; Cysewski, G. Potential Health Benefits of Spirulina Microalgae. Nutrafoods 2010, 9, 19–26. [Google Scholar] [CrossRef]
- Ali, S.K.; Saleh, A.M. Spirulina-an Overview. Int. J. Pharm. Pharm. Sci. 2012, 4, 9–15. [Google Scholar]
- Tokuşoglu, Ö.; Ünal, M.K. Biomass Nutrient Profiles of Three Microalgae: Spirulina Platensis, Chlorella Vulgaris, and Isochrisis Galbana. J. Food Sci. 2003, 68, 1144–1148. [Google Scholar] [CrossRef]
- Babadzhanov, A.S.; Abdusamatova, N.; Yusupova, F.M.; Faizullaeva, N.; Mezhlumyan, L.G.; Malikova, M.K. Chemical Composition of Spirulina Platensis Cultivated in Uzbekistan. Chem. Nat. Compd. 2004, 40, 276–279. [Google Scholar] [CrossRef]
- Habib, M.A.B.; Parvin, M.; Huntington, T.C.; Hasan, M.R. A Review on Culture, Production and Use of Spirulina as Food for Humans and Feeds for Domestic Animals and Fish; Food and Agriculture Organization of the United Nations: Rome, Italy, 2008. [Google Scholar]
- Gad, A.S.; Khadrawy, Y.A.; El-Nekeety, A.A.; Mohamed, S.R.; Hassan, N.S.; Abdel-Wahhab, M.A. Antioxidant Activity and Hepatoprotective Effects of Whey Protein and Spirulina in Rats. Nutrition 2011, 27, 582–589. [Google Scholar] [CrossRef] [PubMed]
- Czerwonka, A.; Kaławaj, K.; Sławińska-Brych, A.; Lemieszek, M.K.; Bartnik, M.; Wojtanowski, K.K.; Zdzisińska, B.; Rzeski, W. Anticancer Effect of the Water Extract of a Commercial Spirulina (Arthrospira Platensis) Product on the Human Lung Cancer A549 Cell Line. Biomed. Pharmacother. 2018, 106, 292–302. [Google Scholar] [CrossRef] [PubMed]
- Nagaoka, S.; Shimizu, K.; Kaneko, H.; Shibayama, F.; Morikawa, K.; Kanamaru, Y.; Otsuka, A.; Hirahashi, T.; Kato, T. A Novel Protein C-Phycocyanin Plays a Crucial Role in the Hypocholesterolemic Action of Spirulina Platensis Concentrate in Rats. J. Nutr. 2005, 135, 2425–2430. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gutiérrez-Salmeán, G.; Fabila-Castillo, L.; Chamorro-Cevallos, G. Nutritional and Toxicological Aspects of Spirulina (Arthrospira). Nutr. Hosp. 2015, 32, 34–40. [Google Scholar] [CrossRef] [PubMed]
- Meticulous Research® Spirulina Market by Distribution Channel (Consumer Channel, Business Channel), Product Type (Powder, Tablets, Capsules, Flakes, Phycocyanin Extract), Application (Nutraceuticals, Food and Beverages, Agriculture, Animal Feed)-Global Forecast to 2028. Available online: https://www.meticulousresearch.com/product/spirulina-market-5070#description (accessed on 1 March 2022).
- Kejžar, J.; Jagodic Hudobivnik, M.; Nečemer, M.; Ogrinc, N.; Masten Rutar, J.; Poklar Ulrih, N. Characterization of Algae Dietary Supplements Using Antioxidative Potential, Elemental Composition, and Stable Isotopes Approach. Front. Nutr. 2021, 7, 618503. [Google Scholar] [CrossRef] [PubMed]
- Al-Dhabi, N.A. Heavy Metal Analysis in Commercial Spirulina Products for Human Consumption. Saudi J. Biol. Sci. 2013, 20, 383–388. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gallardo-Velázquez, T.; Osorio-Revilla, G.; Zuñiga-de Loa, M.; Rivera-Espinoza, Y. Application of FTIR-HATR Spectroscopy and Multivariate Analysis to the Quantification of Adulterants in Mexican Honeys. Food Res. Int. 2009, 42, 313–318. [Google Scholar] [CrossRef]
- Wu, D.; Nie, P.; Cuello, J.; He, Y.; Wang, Z.; Wu, H. Application of Visible and Near Infrared Spectroscopy for Rapid and Non-Invasive Quantification of Common Adulterants in Spirulina Powder. J. Food Eng. 2011, 102, 278–286. [Google Scholar] [CrossRef]
- De Carvalho, L.M.D.; Cohen, P.A.; Silva, C.V.; Moreira, A.P.L.; Falcão, T.M.; Dal Molin, T.R.; Zemolin, G.; Martini, M. A New Approach to Determining Pharmacologic Adulteration of Herbal Weight Loss Products. Food Addit. Contam. Part A 2012, 29, 1661–1667. [Google Scholar] [CrossRef]
- Moreira, A.P.L.; Gobo, L.A.; Viana, C.; de Carvalho, L.M. Simultaneous Analysis of Antihypertensive Drugs and Diuretics as Adulterants in Herbal-Based Products by Ultra-High Performance Liquid Chromatography-Electrospray Tandem Mass Spectrometry. Anal. Methods 2016, 8, 1881–1888. [Google Scholar] [CrossRef]
- Nečemer, M.; Kump, P.; Vogel-Mikuš, K. Use of X-Ray fluorescence-based analytical techniques in phytoremediation. In Handbook of Phytoremediation; Golubev, I.A., Ed.; Environmental Science, Engineering and Technology; Nova Science Publishers Inc.: New York, NY, USA, 2011; pp. 331–358. ISBN 978-1-61728-753-4. [Google Scholar]
- Nečemer, M.; Kump, P.; Ščančar, J.; Jaćimović, R.; Simčič, J.; Pelicon, P.; Budnar, M.; Jeran, Z.; Pongrac, P.; Regvar, M.; et al. Application of X-ray Fluorescence Analytical Techniques in Phytoremediation and Plant Biology Studies. Spectrochim. Acta B At. Spectrosc. 2008, 63, 1240–1247. [Google Scholar] [CrossRef]
- Ravel, B.; Newville, M. ATHENA, ARTEMIS, HEPHAESTUS: Data Analysis for X-Ray Absorption Spectroscopy Using IFEFFIT. J. Synchrotron Radiat. 2005, 12, 537–541. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pongrac, P.; Arčon, I.; Castillo-Michel, H.; Vogel-Mikuš, K. Mineral Element Composition in Grain of Awned and Awnletted Wheat (Triticum Aestivum L.) Cultivars: Tissue-Specific Iron Speciation and Phytate and Non-Phytate Ligand Ratio. Plants 2020, 9, 79. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Christie, W.W.; Han, X. Lipid Analysis: Isolation, Separation, Identification and Lipidomic Analysis, 4th ed.; The Oily Press: Cambridge, UK, 2010; ISBN 978-0-85709-786-6. [Google Scholar]
- Paniagua-Michel, J. Chapter 16-Microalgal Nutraceuticals. In Handbook of Marine Microalgae; Kim, S.-K., Ed.; Academic Press: Boston, MA, USA, 2015; pp. 255–267. ISBN 978-0-12-800776-1. [Google Scholar]
- EFSA Panel on Dietetic Products, Nutrition, and Allergies (NDA). Scientific Opinion on Principles for Deriving and Applying Dietary Reference Values. EFSA J. 2010, 8, 1–30. [Google Scholar] [CrossRef] [Green Version]
- EFSA (European Food Safety Authority). Dietary Reference Values for Nutrients. Summary Report. EFSA Supporting Publ. 2019, 14, e15121. [Google Scholar] [CrossRef] [Green Version]
- Sawai, J. Antimicrobial Characteristics of Heated Scallop Shell Powder and Its Application. Biocontrol Sci. 2011, 16, 95–102. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- da Silva, R.P.; Kawai, G.S.D.; de Andrade, F.R.D.; Bezzon, V.D.N.; Ferraz, H.G. Characterisation and Traceability of Calcium Carbonate from the Seaweed Lithothamnium Calcareum. Solids 2021, 2, 192–211. [Google Scholar] [CrossRef]
- EFSA (European Food Safety Authority). Tolerable Upper Intake Levels for Vitamins and Minerals, 1st ed.; European Food Safety Authority: Parma, Italy, 2006; ISBN 92-9199-014-0.
- Qamar, A.; Saeed, F.; Tahir-Nadeem, M.; Hussain, A.I.; Niaz, B.; Ullah Khan, A.; Afzaal, M.; Badar Ul Ain, H.; Imran, M. Exploring the Phytochemical Profile of Green Grasses with Special Reference to Antioxidant Properties. Int. J. Food Prop. 2018, 21, 2566–2577. [Google Scholar] [CrossRef] [Green Version]
- Puyfoulhoux, G.; Rouanet, J.-M.; Besançon, P.; Baroux, B.; Baccou, J.-C.; Caporiccio, B. Iron Availability from Iron-Fortified Spirulina by an in Vitro Digestion/Caco-2 Cell Culture Model. J. Agric. Food Chem. 2001, 49, 1625–1629. [Google Scholar] [CrossRef]
- Sukumaran, P.; Dahlan, F.L.; Omar, H.; Ismail, A. Macro- and Micronutrients Status in Arthrospira Platensis Grown in Freshwater and Brackish Water Medium. Int. J. Curr. Microbiol. Appl. Sci. 2014, 3, 384–391. [Google Scholar]
- Scheers, N.; Andlid, T.; Alminger, M.; Sandberg, A.-S. Determination of Fe2+ and Fe3+ in Aqueous Solutions Containing Food Chelators by Differential Pulse Anodic Stripping Voltammetry. Electroanalysis 2010, 22, 1090–1096. [Google Scholar] [CrossRef]
- Chidambaram, M.V.; Reddy, M.B.; Thompson, J.L.; Bates, G.W. In Vitro Studies of Iron Bioavailability. Probing the Concentration and Oxidation-Reduction Reactivity of Pinto Bean Iron with Ferrous Chrornogens. Biol. Trace. Elem. Res. 1989, 19, 25–40. [Google Scholar] [CrossRef] [PubMed]
- Hathcock, J.N. Vitamin and Mineral Safety, 3rd ed.; MacKay, D., Ed.; Council for Responsible Nutrition (CRN): Washington, DC, USA, 2014. [Google Scholar]
- Cases, J.; Vacchina, V.; Napolitano, A.; Caporiccio, B.; Besançon, P.; Lobinski, R.; Rouanet, J.-M. Selenium from Selenium-Rich Spirulina Is Less Bioavailable than Selenium from Sodium Selenite and Selenomethionine in Selenium-Deficient Rats. J. Nutr. 2001, 131, 2343–2350. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Falquet, J. The Nutritional Aspects of Spirulina. Antenna Technologies. Available online: https://iceagefarmer.com/docs/Crop%20Focus/Spirulina/AspectNut_UK.pdf (accessed on 3 October 2021).
- Frontasyeva, M.V.; Pavlov, S.S.; Mosulishvili, L.; Kirkesali, E.; Ginturi, E.; Kuchava, N. Accumulation of Trace Elements by Biological Matrice of Spirulina Platensis. Ecol. Chem. Eng. S 2009, 16, 277–285. [Google Scholar]
- Dmytryk, A.; Saeid, A.; Chojnacka, K. Biosorption of Microelements by Spirulina: Towards Technology of Mineral Feed Supplements. Sci. World J. 2014, 2014, 1–15. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Craig, W.J.; Mangels, A.R. Position of the American Dietetic Association: Vegetarian Diets. J. Am. Diet. Assoc. 2009, 109, 1266–1282. [Google Scholar] [CrossRef] [PubMed]
- European Commission. Guidance Document for Competent Authorities for the Control of Compliance with EU Legislation on: Regulation (EU) No 1169/2011 of the European Parliament and of the Council of 25 October 2011 on the Provision of Food Information to Consumers, Amending Regulations (EC) No 1924/2006 and (EC) No 1925/2006 of the European Parliament and of the Council, and Repealing Commission Directive 87/250/EEC, Council Directive 90/496/EEC, Commission Directive 1999/10/EC, Directive 2000/13/EC of the European Parliament and of the Council, Commission Directives 2002/67/EC and 2008/5/EC and Commission Regulation (EC) No 608/2004 and Council Directive 90/496/EEC of 24 September 1990 on Nutrition Labelling of Foodstuffs and Directive 2002/46/EC of the European Parliament and of the Council of 10 June 2002 on the Approximation of the Laws of the Member States Relating to Food Supplements with Regard to the Setting of Tolerances for Nutrient Values Declared on a Label; European Commission: Brussels, Belgium, 2012; p. 15. [Google Scholar]
- European Commission. Commission Regulation (EC) No 1881/2006 of 19 December 2006 Setting Maximum Levels for Certain Contaminants in Foodstuffs (Last Amended by Commission Regulation (EU) 2021/1323 of 10 August 2021); European Commission: Brussels, Belgium, 2021; Volume 364, p. 33. [Google Scholar]
- European Commission. Commission Regulation (EU) 2015/1006 of 25 June 2015 Amending Regulation (EC) No 1881/2006 as Regards Maximum Levels of Inorganic Arsenic in Foodstuffs; European Commission: Brussels, Belgium, 2015; Volume 161, pp. 14–16. [Google Scholar]
- SURS. Statistical Office of Republic of Slovenia. Available online: https://www.stat.si/StatWeb/en (accessed on 6 October 2021).
- Hsu, Y.-M.; Hwang, J.-M.; Yeh, T.-R. Inorganic Elements Determination for Algae/Spirulina Food Marketed in Taiwan. J. Food Drug. Anal. 2001, 9, 178–182. [Google Scholar] [CrossRef]
- Al-Homaidan, A. Heavy Metal Levels in Saudi Arabian Spirulina. Pak. J. Biol. Sci. 2006, 9, 2693–2695. [Google Scholar] [CrossRef] [Green Version]
- Rzymski, P.; Budzulak, J.; Niedzielski, P.; Klimaszyk, P.; Proch, J.; Kozak, L.; Poniedziałek, B. Essential and Toxic Elements in Commercial Microalgal Food Supplements. J. Appl. Phycol. 2019, 31, 3567–3579. [Google Scholar] [CrossRef] [Green Version]
- Rzymski, P.; Niedzielski, P.; Karczewski, J.; Poniedziałek, B. Biosorption of Toxic Metals Using Freely Suspended Microcystis Aeruginosa Biomass. Cent. Eur. J. Chem. 2014, 12, 1232–1238. [Google Scholar] [CrossRef]
- Kuhnlein, H.V.; Chan, H.M. Environment and Contaminants in Traditional Food Systems of Northern Indigenous Peoples. Annu. Rev. Nutr. 2000, 20, 595–626. [Google Scholar] [CrossRef] [PubMed]
- Kotangele, L.R.; Sarkar, R.; Krishnamoorthi, K.P. Toxicity of Mercury and Zinc to Spirulina Platensis. Indian J. Environ. Health 1984, 26, 41–46. [Google Scholar]
- Pande, A.S.; Sarkar, R.; Krishnamoorthi, K.P. Toxicity of Copper Sulphate to the Alga Spirulina Platensis and the Ciliate Tetrahymena Pyriformis. Indian J. Exp. Biol. 1981, 19, 500–502. [Google Scholar]
- Badawy, A.A.-B.; Morgan, C.J.; Turner, J.A. Application of the Phenomenex EZ:FaastTM Amino Acid Analysis Kit for Rapid Gas-Chromatographic Determination of Concentrations of Plasma Tryptophan and Its Brain Uptake Competitors. Amino Acids 2008, 34, 587–596. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kaspar, H.; Dettmer, K.; Gronwald, W.; Oefner, P.J. Automated GC–MS Analysis of Free Amino Acids in Biological Fluids. J. Chromatogr. B 2008, 870, 222–232. [Google Scholar] [CrossRef] [PubMed]
- Food and Agriculture Organization of the United Nations; World Health Organization; United Nations University. Protein and Amino Acid Requirements in Human Nutrition: Report of a Joint FAO/WHO/UNU Expert Consultation; WHO Technical Report Series; World Health Organization: Geneva, Switzerland, 2007; p. 265.
- Dewi, E.N.; Amalia, U.; Mel, M. The Effect of Different Treatments to the Amino Acid Contents of Micro Algae Spirulina sp. Aquat. Procedia 2016, 7, 59–65. [Google Scholar] [CrossRef]
- Bashir, S.; Sharif, M.K.; Butt, M.S.; Shahid, M. Functional Properties and Amino Acid Profile of Spirulina Platensis Protein Isolates. Pak. J. Sci. Ind. Res. Ser. B Biol. Sci. 2016, 59, 12–19. [Google Scholar] [CrossRef]
- Ghumman, A.; Singh, N.; Kaur, A. Chemical, Nutritional and Phenolic Composition of Wheatgrass and Pulse Shoots. Int. J. Food Sci. Technol. 2017, 52, 2191–2200. [Google Scholar] [CrossRef]
- Muys, M.; Sui, Y.; Schwaiger, B.; Lesueur, C.; Vandenheuvel, D.; Vermeir, P.; Vlaeminck, S.E. High Variability in Nutritional Value and Safety of Commercially Available Chlorella and Spirulina Biomass Indicates the Need for Smart Production Strategies. Bioresour. Technol. 2019, 275, 247–257. [Google Scholar] [CrossRef] [PubMed]
- Volkmann, H.; Imianovsky, U.; Oliveira, J.L.B.; Sant’Anna, E.S. Cultivation of Arthrospira (Spirulina) 11 Platensis in Desalinator Wastewater and Salinated Synthetic Medium: Protein Content and Amino-Acid Profile. Braz. J. Microbiol. 2008, 39, 98–101. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zeng, M.-T.; Vonshak, A. Adaptation of Spirulina Platensis to Salinity-Stress. Comp. Biochem. Physiol. Part A Mol. Integr. Physiol. 1998, 120, 113–118. [Google Scholar] [CrossRef]
- Desmorieux, H.; Hernandez, F. Biochemical and Physical Criteria of Spirulina after Different Drying Processes. In Proceedings of the 14th International Drying Symposium, Sao Paulo, Brazil, 22–25 August 2004; Volume B, pp. 900–907. [Google Scholar]
- Agustini, T.W.; Suzery, M.; Sutrisnanto, D.; Ma’ruf, W.F. Hadiyanto Comparative Study of Bioactive Substances Extracted from Fresh and Dried Spirulina sp. Procedia Environ. Sci. 2015, 23, 282–289. [Google Scholar] [CrossRef] [Green Version]
- de Jesus, C.S.; da Silva Uebel, L.; Costa, S.S.; Miranda, A.L.; de Morais, E.G.; de Morais, M.G.; Costa, J.A.V.; Nunes, I.L.; de Souza Ferreira, E.; Druzian, J.I. Outdoor Pilot-Scale Cultivation of Spirulina Sp. LEB-18 in Different Geographic Locations for Evaluating Its Growth and Chemical Composition. Bioresour. Technol. 2018, 256, 86–94. [Google Scholar] [CrossRef] [PubMed]
- Diraman, H.; Koru, E.; Dibeklioglu, H. Fatty Acid Profile of Spirulina Platensis Used as a Food Supplement. Isr. J. Aquac. 2009, 61, 134–142. [Google Scholar]
- Bhakar, R.; Kumar, R.; Pabbi, S. Total Lipids and Fatty Acid Profile of Different Spirulina Strains as Affected by Salinity and Incubation Time. Int. J. Plant Res. 2013, 26, 148–154. [Google Scholar] [CrossRef]
- Cohen, Z.; Vonshak, A.; Richmond, A. Fatty Acid Composition of Spirulina Strains Grown under Various Environmental Conditions. Phytochemistry 1987, 26, 2255–2258. [Google Scholar] [CrossRef]
- Ötleş, S.; Pire, R. Fatty Acid Composition of Chlorella and Spirulina Microalgae Species. J. AOAC Int. 2001, 84, 1708–1714. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Carcea, M.; Sorto, M.; Batello, C.; Narducci, V.; Aguzzi, A.; Azzini, E.; Fantauzzi, P.; Finotti, E.; Gabrielli, P.; Galli, V.; et al. Nutritional Characterization of Traditional and Improved Dihé, Alimentary Blue-Green Algae from the Lake Chad Region in Africa. LWT Food Sci. Technol. 2015, 62, 753–763. [Google Scholar] [CrossRef]
- Wang, Y.-Z.; Hallenbeck, P.C.; Leite, G.B.; Paranjape, K.; Huo, D.-Q. Growth and Lipid Accumulation of Indigenous Algal Strains under Photoautotrophic and Mixotrophic Modes at Low Temperature. Algal Res. 2016, 16, 195–200. [Google Scholar] [CrossRef]
- Renaud, S.M.; Thinh, L.-V.; Lambrinidis, G.; Parry, D.L. Effect of Temperature on Growth, Chemical Composition and Fatty Acid Composition of Tropical Australian Microalgae Grown in Batch Cultures. Aquaculture 2002, 211, 195–214. [Google Scholar] [CrossRef]
- de Oliveira, M.A.C.L.; Monteiro, M.P.C.; Robbs, P.G.; Leite, S.G.F. Growth and Chemical Composition of Spirulina Maxima and Spirulina Platensis Biomass at Different Temperatures. Aquac. Int. 1999, 7, 261–275. [Google Scholar] [CrossRef]
- Shekarabi, S.P.H.; Mehrgan, M.S.; Razi, N.; Sabzi, S. Biochemical Composition and Fatty Acid Profile of the Marine Microalga Isochrysis Galbana Dried with Different Methods. J. Microbiol. Biotechnol. Food Sci. 2019, 9, 521–524. [Google Scholar] [CrossRef]
- Sharoba, A.M. Nutritional Value of Spirulina and Its Use in the Preparation of Some Complementary Baby Food Formulas. J. Dairy Sci. 2014, 5, 517–538. [Google Scholar] [CrossRef]
| Sample | Form | Declared Origin | Declared Product Content |
|---|---|---|---|
| S1 | Tablets | Japan | Spirulina, edible scallop shell powder, edible refined processing fat |
| S4 | Tablets | Hawaii | Spirulina pacifica, silicon dioxide, chicory inulin, magnesium stearate |
| S26 | Tablets | Hawaii | Spirulina pacifica, silicon dioxide |
| S7 | Tablets | India | Spirulina platensis |
| S19 | Tablets | India | Spirulina platensis, calcium carbonate, micro-crystalline cellulose, stearic acid, croscarmellose sodium, silica |
| S34 | Tablets | India | Spirulina platensis |
| S38 | Tablets | India | Spirulina platensis |
| S9 | Powder | Mongolia-China | Wheatgrass, barley grass, Spirulina, Chlorella |
| S8 | Tablets | China | Spirulina platensis |
| S10 | Powder | China | Spirulina |
| S11 | Powder | China | Spirulina platensis |
| S12 | Powder | China | Spirulina |
| S13 | Tablets | China | Spirulina |
| S17 | Powder | China | Spirulina platensis |
| S23 | Tablets | China | Spirulina |
| S24 | Powder | China | Spirulina |
| S25 | Tablets | China | Spirulina |
| S27 | Powder | China | Spirulina |
| S31 | Powder | China | Spirulina platensis |
| S32 | Tablets | China | Spirulina |
| S33 | Powder | China | Spirulina |
| S41 | Powder | China | Spirulina platensis |
| S43 | Powder | China | Spirulina |
| S3 | Powder | Outside EU | Spirulina platensis |
| S5 | Powder | Outside EU | Spirulina |
| S6 | Powder | Outside EU | Spirulina platensis |
| S16 | Powder | Outside EU | Spirulina |
| S35 | Tablets | Outside EU | Spirulina |
| S36 | Powder | Outside EU | Spirulina |
| S14 | Tablets | Taiwan | Spirulina platensis |
| S15 | Powder | Taiwan | Spirulina platensis |
| S40 | Tablets | Taiwan | Spirulina platensis |
| S29 | Tablets | Portugal | Spirulina platensis, silicon dioxide, magnesium stearate |
| S30 | Tablets | Portugal | Spirulina platensis, silicon dioxide, magnesium stearate |
| S44 | Flakes | Italy | Spirulina platensis |
| S46 | Fresh | Italy | Spirulina platensis |
| S18 | Capsules | EU | Spirulina, Chlorella, Lithothamnium |
| S2 | Capsules | NS 1 | Spirulina pacifica, magnesium stearate |
| S20 | Tablets | NS | Spirulina, silicon dioxide, magnesium stearate |
| S21 | Tablets | NS | Spirulina |
| S22 | Tablets | NS | Spirulina platensis, maltodextrin, silicon dioxide, magnesium stearate, hydroxypropyl methyl cellulose |
| S28 | Tablets | NS | Spirulina |
| S37 | Tablets | NS | Spirulina maxima, corn maltodextrin, magnesium stearate |
| S39 | Tablets | NS | Spirulina |
| S42 | Capsules | NS | Spirulina |
| S45 | Powder | NS 1 | Spirulina |
| S. No. 1 | Si (g/kg) | P (g/kg) | S (g/kg) | Cl (g/kg) | K (g/kg) | Ca (g/kg) | Ti (mg/kg) | Mn (mg/kg) | Fe (g/kg) | Zn (mg/kg) | Se (ug/g) | Br (mg/kg) | Rb (mg/kg) | Sr (mg/kg) |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| S1 | 1.57 | 10.5 | 7.53 | 0.66 | 16.1 | 8.18 | 18.2 | 43.4 | 0.81 | 14.0 | 0.06 | 1.29 | 1.55 | 37.9 |
| S2 | 7.69 | 11.4 | 7.53 | 3.07 | 14.8 | 1.32 | 56.9 | 159 | 3.29 | 43.6 | 0.59 | 11.2 | 7.47 | 10.0 |
| S3 | 1.16 | 10.6 | 8.64 | 3.18 | 15.4 | 0.82 | 5.45 | 29.4 | 0.49 | 8.33 | 0.12 | 1.91 | 1.67 | 17.8 |
| S4 | 13.5 | 10.0 | 7.57 | 5.77 | 17.1 | 2.20 | 46.5 | 128 | 3.48 | 52.7 | 0.42 | 16.5 | 11.9 | 24.3 |
| S5 | 1.34 | 12.4 | 9.04 | 3.78 | 20.8 | 0.74 | 8.77 | 22.1 | 0.48 | 14.6 | 0.06 | 1.77 | 1.23 | 25.1 |
| S6 | 21.7 | 12.7 | 6.47 | 0.09 | 11.0 | 1.28 | 9.31 | 54.9 | 0.90 | 13.9 | 0.02 | 0.50 | 4.12 | 10.2 |
| S7 | 10.7 | 11.8 | 7.77 | 0.60 | 14.3 | 5.10 | 3.96 | 32.8 | 0.37 | 10.4 | 0.03 | 0.47 | 0.81 | 22.1 |
| S8 | 16.0 | 11.5 | 7.53 | 1.03 | 14.7 | 2.04 | 60.6 | 35.0 | 1.39 | 16.0 | 0.40 | 0.91 | 0.91 | 27.3 |
| S9 | 5.21 | 5.06 | 3.14 | 2.61 | 19.7 | 2.83 | 10.4 | 43.0 | 0.44 | 19.1 | 0.03 | 10.8 | 3.75 | 12.7 |
| S10 | 2.34 | 14.1 | 9.38 | 2.11 | 18.5 | 3.09 | 35.5 | 36.6 | 1.68 | 18.5 | 0.37 | 1.34 | 1.58 | 34.7 |
| S11 | 1.42 | 12.9 | 8.29 | 0.48 | 15.2 | 1.20 | 4.42 | 26.5 | 0.57 | 11.1 | 0.10 | 1.19 | 1.07 | 28.2 |
| S12 | 1.63 | 13.9 | 8.42 | 1.77 | 16.8 | 5.24 | 12.3 | 38.2 | 1.38 | 33.0 | 0.15 | 1.39 | 1.48 | 31.8 |
| S13 | 16.6 | 12.6 | 7.79 | 1.97 | 15.5 | 5.39 | 14.8 | 34.4 | 1.79 | 33.6 | 0.14 | 1.47 | 0.92 | 31.1 |
| S14 | 7.94 | 12.2 | 7.54 | 0.19 | 13.6 | 1.00 | 9.91 | 34.9 | 0.69 | 16.5 | 2.70 | 0.57 | 2.23 | 7.41 |
| S15 | 1.43 | 11.9 | 7.32 | 0.21 | 13.7 | 0.89 | 5.07 | 33.3 | 0.66 | 15.7 | 2.63 | 0.48 | 1.60 | 6.82 |
| S16 | 1.59 | 14.7 | 7.50 | 0.52 | 14.3 | 2.78 | 6.08 | 36.3 | 0.65 | 17.5 | 0.07 | 0.91 | 0.50 | 12.7 |
| S17 | 1.61 | 13.5 | 8.64 | 2.21 | 16.6 | 5.34 | 12.3 | 30.9 | 1.74 | 34.9 | 0.13 | 1.88 | 1.20 | 32.3 |
| S18 | 2.74 | 12.6 | 6.17 | 0.60 | 8.63 | 63.5 | 43.1 | 47.1 | 0.75 | 11.1 | 0.11 | 9.11 | 2.55 | 478 |
| S19 | 19.4 | 12.2 | 9.29 | 2.55 | 18.4 | 28.0 | 15.9 | 30.3 | 0.56 | 9.74 | 0.10 | 2.26 | 1.24 | 27.6 |
| S20 | 16.8 | 12.1 | 8.39 | 3.04 | 16.3 | 2.43 | 47.5 | 29.1 | 1.13 | 23.0 | 0.92 | 2.00 | 1.91 | 32.2 |
| S21 | 14.7 | 9.77 | 7.30 | 1.94 | 16.2 | 1.35 | 28.2 | 150 | 0.69 | 22.4 | 0.07 | 3.09 | 2.97 | 9.66 |
| S22 | 1.85 | 6.82 | 3.60 | 0.55 | 7.40 | 1.37 | 11.1 | 21.1 | 0.39 | 7.69 | 0.07 | 1.24 | 1.06 | 8.00 |
| S23 | 15.1 | 11.2 | 7.12 | 1.12 | 14.2 | 2.03 | 65.8 | 28.3 | 1.39 | 14.0 | 0.44 | 0.88 | 0.78 | 28.0 |
| S24 | 1.40 | 9.27 | 6.05 | 0.87 | 12.5 | 2.45 | 19.0 | 88.3 | 0.77 | 15.4 | 0.06 | 1.04 | 1.16 | 11.2 |
| S25 | 15.4 | 9.61 | 8.32 | 2.68 | 17.3 | 1.02 | 9.37 | 51.5 | 0.60 | 10.0 | 0.08 | 1.60 | 1.43 | 18.7 |
| S26 | 15.0 | 10.9 | 7.91 | 5.63 | 17.5 | 2.28 | 42.3 | 185 | 3.09 | 35.5 | 0.55 | 17.4 | 9.96 | 14.1 |
| S27 | 1.79 | 11.2 | 8.30 | 2.07 | 16.8 | 0.80 | 8.61 | 22.8 | 0.72 | 15.6 | 0.08 | 1.27 | 1.04 | 22.3 |
| S28 | 4.91 | 10.3 | 8.66 | 3.47 | 17.4 | 0.72 | 2.58 | 26.2 | 0.47 | 7.27 | 0.11 | 2.54 | 2.44 | 15.4 |
| S29 | 15.6 | 10.9 | 7.24 | 1.67 | 15.6 | 1.62 | 35.7 | 192 | 1.14 | 20.9 | 0.07 | 2.71 | 2.06 | 15.6 |
| S30 | 15.1 | 10.1 | 6.99 | 1.63 | 15.2 | 1.50 | 34.5 | 195 | 1.12 | 21.7 | 0.07 | 3.21 | 1.33 | 15.2 |
| S31 | 1.53 | 11.4 | 6.72 | 1.36 | 14.7 | 1.67 | 13.0 | 178 | 0.78 | 22.0 | 0.10 | 1.82 | 2.85 | 7.85 |
| S32 | 16.1 | 10.2 | 8.15 | 2.62 | 14.2 | 2.54 | 39.4 | 35.9 | 1.39 | 22.9 | 1.23 | 1.86 | 2.30 | 35.5 |
| S33 | 1.06 | 10.2 | 7.99 | 2.18 | 14.9 | 1.00 | 5.84 | 28.0 | 0.64 | 5.42 | 0.06 | 1.62 | 1.55 | 29.7 |
| S34 | 16.9 | 10.9 | 8.09 | 4.34 | 15.9 | 0.91 | 7.82 | 27.8 | 0.52 | 11.3 | 0.28 | 4.47 | 0.57 | 31.2 |
| S35 | 7.52 | 10.1 | 7.85 | 2.20 | 14.4 | 0.96 | 6.85 | 26.5 | 0.63 | 7.18 | 0.11 | 2.38 | 1.27 | 26.3 |
| S36 | 1.97 | 11.4 | 7.10 | 2.58 | 14.9 | 2.17 | 24.7 | 34.5 | 1.01 | 17.0 | 0.21 | 6.74 | 1.23 | 55.0 |
| S37 | 6.43 | 6.16 | 3.88 | 0.91 | 5.83 | 0.75 | 12.8 | 19.3 | 0.28 | 6.02 | 0.07 | 1.67 | 0.55 | 9.75 |
| S38 | 7.56 | 10.3 | 8.04 | 0.30 | 9.00 | 4.93 | 2.81 | 24.9 | 0.41 | 13.3 | 0.02 | 0.67 | 1.33 | 26.1 |
| S39 | 12.1 | 9.93 | 6.71 | 1.76 | 12.7 | 0.97 | 8.88 | 14.7 | 0.44 | 13.1 | 0.14 | 1.86 | 0.99 | 19.8 |
| S40 | 8.49 | 10.2 | 8.06 | 0.34 | 8.84 | 5.52 | 4.39 | 28.3 | 0.42 | 14.7 | 0.03 | 0.85 | 1.09 | 29.3 |
| S41 | 0.94 | 8.64 | 7.18 | 5.03 | 16.4 | 0.52 | 3.58 | 18.6 | 0.38 | 10.3 | 0.05 | 2.93 | 2.42 | 15.9 |
| S42 | 1.07 | 10.1 | 7.72 | 4.70 | 15.1 | 0.69 | 8.89 | 29.1 | 0.55 | 9.91 | 0.13 | 3.78 | 1.21 | 23.7 |
| S43 | 1.34 | 10.1 | 9.91 | 5.34 | 15.7 | 1.42 | 3.16 | 23.2 | 0.41 | 2.30 | 0.26 | 5.51 | 1.34 | 71.8 |
| S44 | 0.78 | 8.56 | 6.63 | 2.75 | 20.6 | 3.45 | 10.4 | 84.9 | 0.69 | 24.9 | 0.00 | 8.00 | 6.55 | 86.2 |
| S45 | 1.02 | 11.0 | 7.87 | 1.13 | 14.3 | 1.26 | 3.18 | 27.4 | 0.45 | 18.1 | 0.10 | 1.43 | 1.47 | 22.0 |
| S46 | 0.68 | 6.64 | 7.38 | 5.36 | 26.9 | 0.46 | 5.36 | 32.9 | 0.93 | 7.59 | 0.00 | 7.07 | 4.21 | 4.39 |
| Sample | Fe3+ (%) | Fe2+ (%) |
|---|---|---|
| S4 | 92 | 8 |
| S17 | 87 | 13 |
| S22 | 85 | 15 |
| S41 | 82 | 18 |
| S46 | 88 | 12 |
| S. No. 1 | Fe (g/kg) | Mn (mg/kg) | Ca (g/kg) | Zn (mg/kg) | P (g/kg) | |||||
|---|---|---|---|---|---|---|---|---|---|---|
| DV 2 | MV 3 (Deviation (%)) | DV | MV (Deviation (%)) | DV | MV (Deviation (%)) | DV | MV (Deviation (%)) | DV | MV (Deviation (%)) | |
| S4 | 1.7 | 3.48 (+105) | 67.0 | 128 (+91.1) | - | - | - | - | - | - |
| S5 | 1.04 | 0.48 (−53.7) | - | - | - | - | 15 | 14.6 (−2.67) | - | - |
| S8 | 0.78 | 1.39 (+78.2) | - | - | - | - | - | - | - | - |
| S10 | 0.07 | 1.68 (+2453) | - | - | 3.33 | 3.09 (−7.07) | - | - | - | - |
| S12 | 1.23 | 1.38 (+12.2) | 67 | 38.2 (−43.0) | - | - | - | - | 9.33 | 13.9 (+49.0) |
| S13 | 0.07 | 1.79 (+2457) | - | - | 3.33 | 5.39 (+61.9) | - | - | - | - |
| S14 | 0.40 | 0.69 (+72.0) | 40 | 34.9 (−12.8) | - | - | - | - | - | - |
| S15 | 0.40 | 0.69 (+64.5) | 40 | 33.3 (−16.8) | - | - | - | - | - | - |
| S17 | 0.30 | 1.74 (+480) | 30 | 30.9 (+3.00) | 1.20 | 5.34 (+345) | 180 | 34.9 (−80.6) | 12.0 | 13.5 (+12.5) |
| S23 | 1.23 | 1.39 (+13.0) | - | - | - | - | 360 | 14.0 (−96.1) | - | - |
| S24 | 1.23 | 0.77 (−37.6) | - | - | - | - | 360 | 15.4 (−95.7) | - | - |
| S26 | 2.30 | 3.09 (+34.4) | 130 | 185 (+42.3) | - | - | - | - | - | - |
| S28 | 0.6 | 0.47 (−22.3) | - | - | 6.67 | 0.72 (−89.2) | - | - | - | - |
| S35 | 0.62 | 0.63 (+1.29) | - | - | - | - | - | - | - | - |
| S36 | 0.62 | 1.01 (+62.9) | - | - | - | - | - | - | - | - |
| Element | ML 1/BMDL01 2 | Min | Max | Median (IR 3) | |
|---|---|---|---|---|---|
| Cd (μg/kg dwt) | 3000 | 1.22 | 226 | 22.6 (13.6–53.9) | |
| Hg (μg/kg dwt) | 100 | 0.84 | 101 | 5.57 (3.57–7.63) | |
| Pb (μg/kg dwt) | 3000 | 47.4 | 1320 | 355 (167–611) | |
| As (ug/day) | Male (87 kg) | Female (68 kg) | |||
| 26.1–696 | 20.4–544 | ||||
| RDI 4 3 g | 0.04 | 8.11 | 0.82 (0.49–1.10) | ||
| RDI 4 10 g | 0.15 | 27.0 | 2.72 (1.63–3.66) | ||
| Amino Acid | ADR 1 (mg/Day) | Min (mg/Day) | Max (mg/Day) | Median (IR 2) (mg/Day) | ||||
|---|---|---|---|---|---|---|---|---|
| Male (87 kg) | Female (68 kg) | 3 g | 10 g | 3 g | 10 g | 3 g | 10 g | |
| ALA | - | - | 62.5 | 208 | 148 | 493 | 124 (112–132) | 414 (374–439) |
| GLY | - | - | 46.9 | 156 | 102 | 341 | 89.9 (83.1–94.1) | 300 (277–314) |
| VAL | 2262 | 1768 | 153 | 509 | 369 | 1231 | 307 (279–325) | 1022 (930–1083) |
| LEU | 3393 | 2652 | 90.1 | 300 | 202 | 674 | 169 (160–182) | 565 (532–606) |
| ILE | 1740 | 1360 | 111 | 370 | 257 | 858 | 220 (203–231) | 733 (677–771) |
| THR | 1305 | 1020 | 74.1 | 247 | 177 | 589 | 149 (137–160) | 497 (458–533) |
| SER | - | - | 65.5 | 218 | 175 | 582 | 140 (127–154) | 466 (423–513) |
| PRO | - | - | 52.0 | 173 | 105 | 349 | 91.6 (85.3–93.9) | 305 (284–313) |
| ASP | - | - | 125 | 415 | 304 | 1012 | 255 (239–270) | 850 (796–900) |
| MET | 870 | 680 | 34.7 | 116 | 74.0 | 247 | 61.4 (56.3–66.2) | 205 (188–221) |
| GLU | - | - | 218 | 725 | 712 | 2374 | 498 (455–585) | 1661 (1517–1949) |
| PHE 3 | 2175 | 1700 | 63.3 | 211 | 121 | 403 | 109 (102–112) | 362 (341–374) |
| LYS | 2610 | 2040 | 90.9 | 303 | 377 | 1256 | 231 (199–280) | 770 (662–934) |
| HIS | 870 | 680 | 48.9 | 163 | 66.6 | 222 | 58.7 (56.3–62.4) | 196 (188–208) |
| TYR 3 | 2175 | 1700 | 52.6 | 175 | 119 | 397 | 104 (96.8–111) | 347 (323–371) |
| Fatty Acid | Min | Max | Median (IR 1) |
|---|---|---|---|
| SFA 2 | |||
| C14:0 | 0.23 | 0.63 | 0.35 (0.31–0.40) |
| C15:0 | <LOD | 0.22 | <LOD |
| C16:0 | 18.4 | 45.9 | 41.8 (40.6–42.9) |
| C17:0 | <LOD | 0.42 | 0.33 (0.27–0.36) |
| C18:0 | 0.97 | 25.8 | 3.90 (1.70–5.46) |
| C22:0 | <LOD | 30.2 | <LOD |
| MUFA 3 | |||
| C14:1 | <LOD | 1.12 | 0.33 (0.09–0.41) |
| C16:1 ω-7 | 3.07 | 11.7 | 6.93 (5.96–7.94) |
| C17:1 | <LOD | 0.72 | 0.35 (0.31–0.42) |
| C18:1 ω-9c/9t | 1.72 | 9.34 | 3.78 (3.24–4.38) |
| PUFA 4 | |||
| C16:2 ω-4 | <LOD | 9.38 | 0.42 (0.36–0.49) |
| ω-6 PUFA | |||
| C16:2 ω-6 | <LOD | 9.71 | <LOD |
| LA, C18:2 ω-6c | 11.1 | 31.0 | 22.0 (20.6–22.7) |
| GLA, C18:3 ω-6 | <LOD | 24.1 | 19.4 (16.7–20.3) |
| C20:3 ω-6 | <LOD | 0.82 | <LOD (<LOD–0.33) |
| ω-3 PUFA | |||
| C16:3 ω-3 | <LOD | 9.58 | <LOD |
| C18:3 ω-3 | <LOD | 18.4 | <LOD |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Masten Rutar, J.; Jagodic Hudobivnik, M.; Nečemer, M.; Vogel Mikuš, K.; Arčon, I.; Ogrinc, N. Nutritional Quality and Safety of the Spirulina Dietary Supplements Sold on the Slovenian Market. Foods 2022, 11, 849. https://doi.org/10.3390/foods11060849
Masten Rutar J, Jagodic Hudobivnik M, Nečemer M, Vogel Mikuš K, Arčon I, Ogrinc N. Nutritional Quality and Safety of the Spirulina Dietary Supplements Sold on the Slovenian Market. Foods. 2022; 11(6):849. https://doi.org/10.3390/foods11060849
Chicago/Turabian StyleMasten Rutar, Jasmina, Marta Jagodic Hudobivnik, Marijan Nečemer, Katarina Vogel Mikuš, Iztok Arčon, and Nives Ogrinc. 2022. "Nutritional Quality and Safety of the Spirulina Dietary Supplements Sold on the Slovenian Market" Foods 11, no. 6: 849. https://doi.org/10.3390/foods11060849
APA StyleMasten Rutar, J., Jagodic Hudobivnik, M., Nečemer, M., Vogel Mikuš, K., Arčon, I., & Ogrinc, N. (2022). Nutritional Quality and Safety of the Spirulina Dietary Supplements Sold on the Slovenian Market. Foods, 11(6), 849. https://doi.org/10.3390/foods11060849







