Impact of Oral Microbiota on Flavor Perception: From Food Processing to In-Mouth Metabolization
Abstract
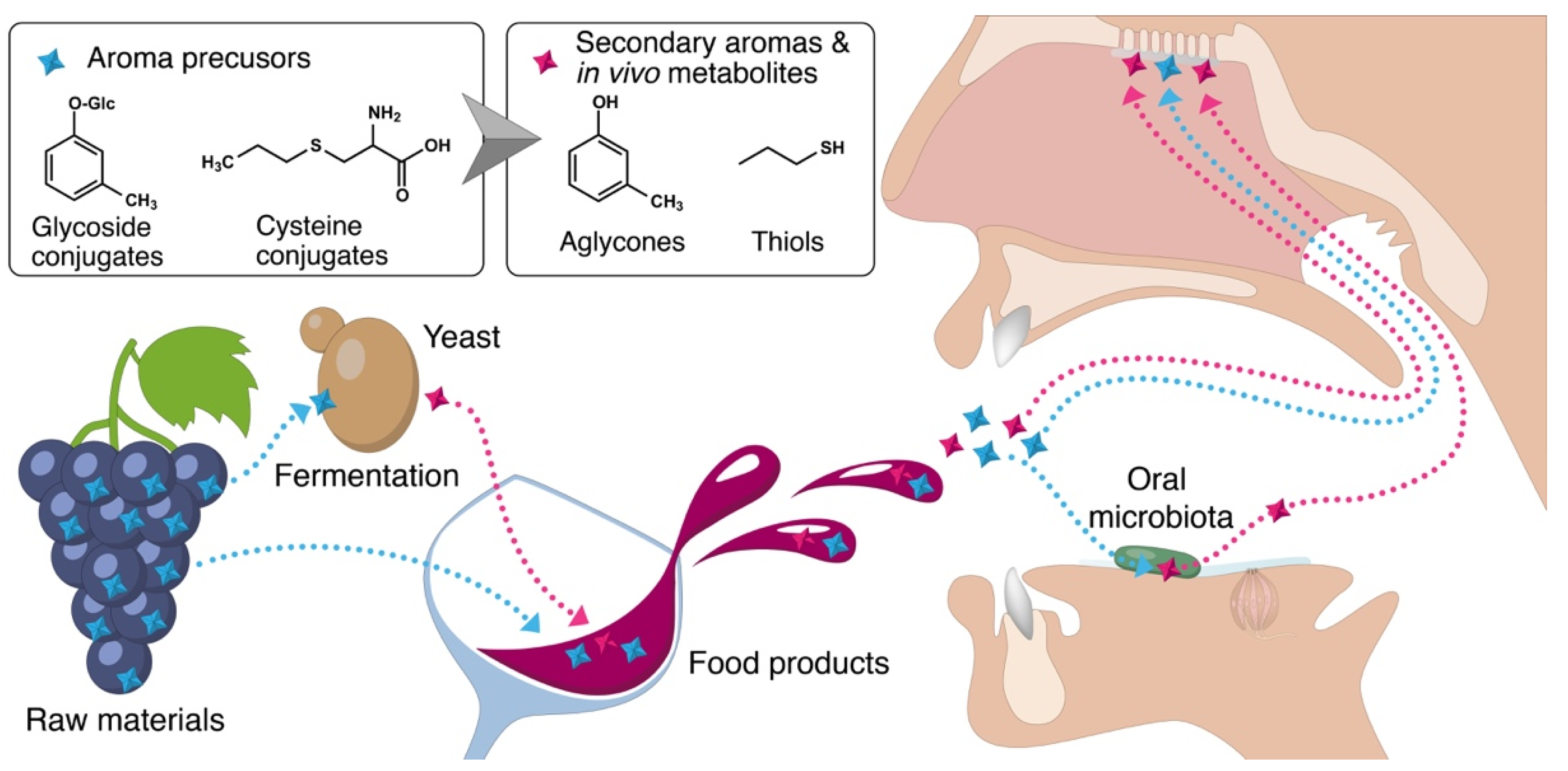
:1. Introduction
2. Characteristics of the Oral Microbiota
2.1. Development of the Microbiota throughout Age
2.2. Consequences of Oral Pathologies on the Microbiota
2.3. Saliva Microbiota
2.4. Influence of External Factors and Diet
3. Oral Microbes Modulate Chemosensory Perception
3.1. Modulation of the Host Taste and Smell Perception
3.2. Modulation of the Host Receptors Expression of Host Genes Encoding Receptors
4. Common Pathways for the Microbial Production of Flavor Compounds in Fermented Products and in the Mouth by Oral Microbiota
4.1. Glycoside Conjugates
4.1.1. Glycosides as Aroma Precursors
4.1.2. Glycosides in Wine
4.1.3. Glycosides in Beer
4.1.4. Glycosides in the Oral Cavity
4.1.5. Metabolization of Glycosides in the Oral Cavity during Alcoholic Beverage Consumption
4.2. Cysteine Conjugates
4.2.1. Cysteine Conjugates as Aroma Precursors
4.2.2. Cysteine Conjugates Metabolism and Formation of Sulfur Aroma in Fermented Beverages
4.2.3. Metabolization of Cysteine Conjugates in the Oral Cavity
5. Conclusions and Perspectives
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Canon, F.; Neiers, F.; Guichard, E. Saliva and flavor perception: Perspectives. J. Agric. Food Chem. 2018, 66, 7873–7879. [Google Scholar] [CrossRef] [PubMed]
- Munoz-Gonzalez, C.; Feron, G.; Brule, M.; Canon, F. Understanding the release and metabolism of aroma compounds using micro-volume saliva samples by ex vivo approaches. Food Chem. 2018, 240, 275–285. [Google Scholar] [CrossRef] [PubMed]
- Munoz-Gonzalez, C.; Brule, M.; Feron, G.; Canon, F. Does interindividual variability of saliva affect the release and metabolization of aroma compounds ex vivo? The particular case of elderly suffering or not from hyposalivation. J. Texture Stud. 2019, 50, 36–44. [Google Scholar] [CrossRef] [PubMed]
- Cattaneo, C.; Riso, P.; Laureati, M.; Gargari, G.; Pagliarini, E. Exploring associations between interindividual differences in taste perception, oral microbiota composition, and reported food intake. Nutrients 2019, 11, 1167. [Google Scholar] [CrossRef] [Green Version]
- Besnard, P.; Christensen, J.E.; Brignot, H.; Bernard, A.; Passilly-Degrace, P.; Nicklaus, S.; Pais de Barros, J.P.; Collet, X.; Lelouvier, B.; Servant, F.; et al. Obese subjects with specific gustatory papillae microbiota and salivary cues display an impairment to sense lipids. Sci. Rep. 2018, 8, 6742. [Google Scholar] [CrossRef]
- Gardner, A.; So, P.W.; Carpenter, G.H. Intraoral microbial metabolism and association with host taste perception. J. Dent. Res. 2020, 99, 739–745. [Google Scholar] [CrossRef] [PubMed]
- Parker, M.; Onetto, C.; Hixson, J.; Bilogrevic, E.; Schueth, L.; Pisaniello, L.; Borneman, A.; Herderich, M.; de Barros Lopes, M.; Francis, L. Factors contributing to interindividual variation in retronasal odor perception from aroma glycosides: The role of odorant sensory detection threshold, oral microbiota, and hydrolysis in saliva. J. Agric. Food Chem. 2020, 68, 10299–10309. [Google Scholar] [CrossRef]
- Starkenmann, C.; Le Calve, B.; Niclass, Y.; Cayeux, I.; Beccucci, S.; Troccaz, M. Olfactory perception of cysteine-S-conjugates from fruits and vegetables. J. Agric. Food Chem. 2008, 56, 9575–9580. [Google Scholar] [CrossRef]
- Schrader, J. Microbial flavour production. In Flavours and Fragrances: Chemistry, Bioprocessing and Sustainability; Berger, R.G., Ed.; Springer: Berlin/Heidelberg, Germany, 2007; pp. 507–574. [Google Scholar]
- Cho, I.H.; Peterson, D.G. Chemistry of bread aroma: A review. Food Sci. Biotechnol. 2010, 19, 575–582. [Google Scholar] [CrossRef]
- Parker, M.; Capone, D.L.; Francis, I.L.; Herderich, M.J. Aroma precursors in grapes and wine: Flavor release during wine production and consumption. J. Agric. Food Chem. 2018, 66, 2281–2286. [Google Scholar] [CrossRef]
- Liu, J.; Zhu, X.-L.; Ullah, N.; Tao, Y.-S. Aroma glycosides in grapes and wine. J. Food Sci. 2017, 82, 248–259. [Google Scholar] [CrossRef] [Green Version]
- Capece, A.; Romaniello, R.; Siesto, G.; Romano, P. Conventional and non-conventional yeasts in beer production. Fermentation 2018, 4, 38. [Google Scholar] [CrossRef] [Green Version]
- Cappello, M.S.; Zapparoli, G.; Logrieco, A.; Bartowsky, E.J. Linking wine lactic acid bacteria diversity with wine aroma and flavour. Int. J. Food Microbiol. 2017, 243, 16–27. [Google Scholar] [CrossRef]
- Dewhirst, F.E.; Chen, T.; Izard, J.; Paster, B.J.; Tanner, A.C.; Yu, W.H.; Lakshmanan, A.; Wade, W.G. The human oral microbiome. J. Bacteriol. 2010, 192, 5002–5017. [Google Scholar] [CrossRef] [Green Version]
- Wilbert, S.A.; Mark Welch, J.L.; Borisy, G.G. Spatial ecology of the human tongue dorsum microbiome. Cell Rep. 2020, 30, 4003–4015.e3. [Google Scholar] [CrossRef] [PubMed]
- Lamont, R.J.; Koo, H.; Hajishengallis, G. The oral microbiota: Dynamic communities and host interactions. Nat. Rev. Microbiol. 2018, 16, 745–759. [Google Scholar] [CrossRef] [PubMed]
- Bowen, W.H.; Burne, R.A.; Wu, H.; Koo, H. Oral biofilms: Pathogens, matrix, and polymicrobial interactions in microenvironments. Trends Microbiol. 2018, 26, 229–242. [Google Scholar] [CrossRef] [PubMed]
- Ghannoum, M.A.; Jurevic, R.J.; Mukherjee, P.K.; Cui, F.; Sikaroodi, M.; Naqvi, A.; Gillevet, P.M. Characterization of the oral fungal microbiome (mycobiome) in healthy individuals. PLoS Pathog. 2010, 6, e1000713. [Google Scholar] [CrossRef] [Green Version]
- Lof, M.; Janus, M.M.; Krom, B.P. Metabolic Interactions between bacteria and fungi in commensal oral biofilms. J. Fungi 2017, 3, 40. [Google Scholar] [CrossRef] [Green Version]
- Grassl, N.; Kulak, N.A.; Pichler, G.; Geyer, P.E.; Jung, J.; Schubert, S.; Sinitcyn, P.; Cox, J.; Mann, M. Ultra-deep and quantitative saliva proteome reveals dynamics of the oral microbiome. Genome Med. 2016, 8, 44. [Google Scholar] [CrossRef] [Green Version]
- Belstrøm, D.; Holmstrup, P.; Bardow, A.; Kokaras, A.; Fiehn, N.E.; Paster, B.J. Temporal stability of the salivary microbiota in oral health. PLoS ONE 2016, 11, e0147472. [Google Scholar] [CrossRef] [PubMed]
- Kaan, A.M.M.; Kahharova, D.; Zaura, E. Acquisition and establishment of the oral microbiota. Periodontol. 2000 2021, 86, 123–141. [Google Scholar] [CrossRef]
- Dzidic, M.; Collado, M.C.; Abrahamsson, T.; Artacho, A.; Stensson, M.; Jenmalm, M.C.; Mira, A. Oral microbiome development during childhood: An ecological succession influenced by postnatal factors and associated with tooth decay. ISME J. 2018, 12, 2292–2306. [Google Scholar] [CrossRef]
- Oba, P.M.; Holscher, H.D.; Mathai, R.A.; Kim, J.; Swanson, K.S. Diet influences the oral microbiota of infants during the first six months of life. Nutrients 2020, 12, 3400. [Google Scholar] [CrossRef] [PubMed]
- Krespi, Y.P.; Shrime, M.G.; Kacker, A. The relationship between oral malodor and volatile sulfur compound-producing bacteria. Otolaryngol. Head Neck Surg. 2006, 135, 671–676. [Google Scholar] [CrossRef] [PubMed]
- Sarkar, P.; Malik, S.; Laha, S.; Das, S.; Bunk, S.; Ray, J.G.; Chatterjee, R.; Saha, A. Dysbiosis of oral microbiota during oral squamous cell carcinoma development. Front. Oncol. 2021, 11, 614448. [Google Scholar] [CrossRef] [PubMed]
- Wade, W.G. Resilience of the oral microbiome. Periodontol. 2000 2021, 86, 113–122. [Google Scholar] [CrossRef] [PubMed]
- Tam, J.; Hoffmann, T.; Fischer, S.; Bornstein, S.; Grassler, J.; Noack, B. Obesity alters composition and diversity of the oral microbiota in patients with type 2 diabetes mellitus independently of glycemic control. PLoS ONE 2018, 13, e0204724. [Google Scholar] [CrossRef]
- MacFarlane, T.W.; Mason, D.K. Changes in the oral flora in Sjögren’s syndrome. J. Clin. Pathol. 1974, 27, 416–419. [Google Scholar] [CrossRef] [Green Version]
- Dawes, C.; Pedersen, A.M.; Villa, A.; Ekstrom, J.; Proctor, G.B.; Vissink, A.; Aframian, D.; McGowan, R.; Aliko, A.; Narayana, N.; et al. The functions of human saliva: A review sponsored by the World Workshop on Oral Medicine VI. Arch. Oral Biol. 2015, 60, 863–874. [Google Scholar] [CrossRef] [PubMed]
- Lynge Pedersen, A.M.; Belstrom, D. The role of natural salivary defences in maintaining a healthy oral microbiota. J. Dent. 2019, 80 (Suppl. 1), S3–S12. [Google Scholar] [CrossRef]
- Schwartz, M.; Neiers, F.; Feron, G.; Canon, F. The relationship between salivary redox, diet, and food flavor perception. Front. Nutr. 2021, 7, 384. [Google Scholar] [CrossRef] [PubMed]
- Bescos, R.; Brookes, Z.L.S.; Belfield, L.A.; Fernandez-Sanjurjo, M.; Casas-Agustench, P. Modulation of oral microbiota: A new frontier in exercise supplementation. PharmaNutrition 2020, 14, 100230. [Google Scholar] [CrossRef]
- Shaw, L.; Ribeiro, A.L.R.; Levine, A.P.; Pontikos, N.; Balloux, F.; Segal, A.W.; Roberts, A.P.; Smith, A.M. The human salivary microbiome is shaped by shared environment rather than genetics: Evidence from a large family of closely related individuals. mBio 2017, 8, e01237-17. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Al-Zyoud, W.; Hajjo, R.; Abu-Siniyeh, A.; Hajjaj, S. Salivary microbiome and cigarette smoking: A first of its kind investigation in Jordan. Int. J. Environ. Res. Public Health 2019, 17, 256. [Google Scholar] [CrossRef] [Green Version]
- Liu, F.; Liang, T.; Zhang, Z.; Liu, L.; Li, J.; Dong, W.; Zhang, H.; Bai, S.; Ma, L.; Kang, L. Effects of altitude on human oral microbes. AMB Express 2021, 11, 41. [Google Scholar] [CrossRef]
- Kato, I.; Vasquez, A.; Moyerbrailean, G.; Land, S.; Djuric, Z.; Sun, J.; Lin, H.S.; Ram, J.L. Nutritional correlates of human oral microbiome. J. Am. Coll. Nutr. 2017, 36, 88–98. [Google Scholar] [CrossRef] [Green Version]
- Vach, K.; Al-Ahmad, A.; Anderson, A.; Woelber, J.P.; Karygianni, L.; Wittmer, A.; Hellwig, E. A Log ratio-based analysis of individual changes in the composition of the oral microbiota in different dietary phases. Nutrients 2021, 13, 793. [Google Scholar] [CrossRef]
- Vach, K.; Al-Ahmad, A.; Anderson, A.; Woelber, J.P.; Karygianni, L.; Wittmer, A.; Hellwig, E. Analysing the relationship between nutrition and the microbial composition of the oral biofilm-insights from the analysis of individual variability. Antibiotics 2020, 9, 479. [Google Scholar] [CrossRef]
- Rowinska, I.; Szyperska-Slaska, A.; Zariczny, P.; Paslawski, R.; Kramkowski, K.; Kowalczyk, P. The influence of diet on oxidative stress and inflammation induced by bacterial biofilms in the human oral cavity. Materials 2021, 14, 1444. [Google Scholar] [CrossRef]
- Peters, B.A.; McCullough, M.L.; Purdue, M.P.; Freedman, N.D.; Um, C.Y.; Gapstur, S.M.; Hayes, R.B.; Ahn, J. Association of coffee and tea intake with the oral microbiome: Results from a large cross-sectional study. Cancer Epidemiol. Biomark. Prev. 2018, 27, 814–821. [Google Scholar] [CrossRef] [Green Version]
- Liu, Z.; Guo, H.; Zhang, W.; Ni, L. Salivary microbiota shifts under sustained consumption of oolong tea in healthy adults. Nutrients 2020, 12, 966. [Google Scholar] [CrossRef] [Green Version]
- Stapleton, P.D.; Shah, S.; Anderson, J.C.; Hara, Y.; Hamilton-Miller, J.M.; Taylor, P.W. Modulation of beta-lactam resistance in Staphylococcus aureus by catechins and gallates. Int. J. Antimicrob. Agents 2004, 23, 462–467. [Google Scholar] [CrossRef]
- Kong, J.; Zhang, G.; Xia, K.; Diao, C.; Yang, X.; Zuo, X.; Li, Y.; Liang, X. Tooth brushing using toothpaste containing theaflavins reduces the oral pathogenic bacteria in healthy adults. 3 Biotech 2021, 11, 150. [Google Scholar] [CrossRef] [PubMed]
- Esberg, A.; Haworth, S.; Hasslof, P.; Lif Holgerson, P.; Johansson, I. Oral microbiota profile associates with sugar intake and taste preference genes. Nutrients 2020, 12, 681. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ijichi, C.; Wakabayashi, H.; Sugiyama, S.; Ihara, Y.; Nogi, Y.; Nagashima, A.; Ihara, S.; Niimura, Y.; Shimizu, Y.; Kondo, K.; et al. Metabolism of odorant molecules in human nasal/oral cavity affects the odorant perception. Chem. Senses 2019, 44, 465–481. [Google Scholar] [CrossRef] [PubMed]
- François, A.; Grebert, D.; Rhimi, M.; Mariadassou, M.; Naudon, L.; Rabot, S.; Meunier, N. Olfactory epithelium changes in germfree mice. Sci. Rep. 2016, 6, 24687. [Google Scholar] [CrossRef] [PubMed]
- Cohn, Z.J.; Kim, A.; Huang, L.; Brand, J.; Wang, H. Lipopolysaccharide-induced inflammation attenuates taste progenitor cell proliferation and shortens the life span of taste bud cells. BMC Neurosci. 2010, 11, 72. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wang, H.; Zhou, M.; Brand, J.; Huang, L. Inflammation and taste disorders: Mechanisms in taste buds. Ann. N. Y. Acad. Sci. 2009, 1170, 596–603. [Google Scholar] [CrossRef]
- Gardner, A.; Parkes, H.G.; So, P.-W.; Carpenter, G.H. Determining bacterial and host contributions to the human salivary metabolome. J. Oral Microbiol. 2019, 11, 1617014. [Google Scholar] [CrossRef] [Green Version]
- Bartoshuk, L.M. The psychophysics of taste. Am. J. Clin. Nutr. 1978, 31, 1068–1077. [Google Scholar] [CrossRef]
- Feron, G. Unstimulated saliva: Background noise in taste molecules. J. Texture Stud. 2019, 50, 6–18. [Google Scholar] [CrossRef] [Green Version]
- Neyraud, E.; Cabaret, S.; Brignot, H.; Chabanet, C.; Labouré, H.; Guichard, E.; Berdeaux, O. The basal free fatty acid concentration in human saliva is related to salivary lipolytic activity. Sci. Rep. 2017, 7, 5969. [Google Scholar] [CrossRef] [Green Version]
- Komiyama, E.Y.; Lepesqueur, L.S.; Yassuda, C.G.; Samaranayake, L.P.; Parahitiyawa, N.B.; Balducci, I.; Koga-Ito, C.Y. Enterococcus species in the oral cavity: Prevalence, virulence factors and antimicrobial susceptibility. PLoS ONE 2016, 11, e0163001. [Google Scholar] [CrossRef] [Green Version]
- Brignot, H.; Feron, G. Oral lipolysis and its association with diet and the perception and digestion of lipids: A systematic literature review. Arch. Oral Biol. 2019, 108, 104550. [Google Scholar] [CrossRef]
- Voigt, N.; Stein, J.; Galindo, M.M.; Dunkel, A.; Raguse, J.D.; Meyerhof, W.; Hofmann, T.; Behrens, M. The role of lipolysis in human orosensory fat perception. J. Lipid Res. 2014, 55, 870–882. [Google Scholar] [CrossRef] [Green Version]
- Scinska-Bienkowska, A.; Wrobel, E.; Turzynska, D.; Bidzinski, A.; Jezewska, E.; Sienkiewicz-Jarosz, H.; Golembiowska, K.; Kostowski, W.; Kukwa, A.; Plaznik, A.; et al. Glutamate concentration in whole saliva and taste responses to monosodium glutamate in humans. Nutr. Neurosci. 2006, 9, 25–31. [Google Scholar] [CrossRef] [PubMed]
- Takahashi, N. Oral microbiome metabolism: From “Who Are They?” to "What Are They Doing?". J. Dent. Res. 2015, 94, 1628–1637. [Google Scholar] [CrossRef] [PubMed]
- Ployon, S.; Brule, M.; Andriot, I.; Morzel, M.; Canon, F. Understanding retention and metabolization of aroma compounds using an in vitro model of oral mucosa. Food Chem. 2020, 318, 126468. [Google Scholar] [CrossRef] [PubMed]
- Mallery, S.R.; Budendorf, D.E.; Larsen, M.P.; Pei, P.; Tong, M.; Holpuch, A.S.; Larsen, P.E.; Stoner, G.D.; Fields, H.W.; Chan, K.K.; et al. Effects of human oral mucosal tissue, saliva, and oral microflora on intraoral metabolism and bioactivation of black raspberry anthocyanins. Cancer Prev. Res. 2011, 4, 1209–1221. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Walle, T.; Browning, A.M.; Steed, L.L.; Reed, S.G.; Walle, U.K. Flavonoid glucosides are hydrolyzed and thus activated in the oral cavity in humans. J. Nutr. 2005, 135, 48–52. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Heydel, J.M.; Menetrier, F.; Belloir, C.; Canon, F.; Faure, P.; Lirussi, F.; Chavanne, E.; Saliou, J.M.; Artur, Y.; Canivenc-Lavier, M.C.; et al. Characterization of rat glutathione transferases in olfactory epithelium and mucus. PLoS ONE 2019, 14, e0220259. [Google Scholar] [CrossRef] [Green Version]
- Schwartz, M.; Menetrier, F.; Heydel, J.-M.; Chavanne, E.; Faure, P.; Labrousse, M.; Lirussi, F.; Canon, F.; Mannervik, B.; Briand, L.; et al. Interactions between odorants and glutathione transferases in the human olfactory cleft. Chem. Senses 2020, 45, 645–654. [Google Scholar] [CrossRef] [PubMed]
- Duca, F.A.; Swartz, T.D.; Sakar, Y.; Covasa, M. Increased oral detection, but decreased intestinal signaling for fats in mice lacking gut microbiota. PLoS ONE 2012, 7, e39748. [Google Scholar] [CrossRef] [Green Version]
- Laugerette, F.; Passilly-Degrace, P.; Patris, B.; Niot, I.; Febbraio, M.; Montmayeur, J.P.; Besnard, P. CD36 involvement in orosensory detection of dietary lipids, spontaneous fat preference, and digestive secretions. J. Clin. Investig. 2005, 115, 3177–3184. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jurczak, A.; Jamka-Kasprzyk, M.; Bębenek, Z.; Staszczyk, M.; Jagielski, P.; Kościelniak, D.; Gregorczyk-Maga, I.; Kołodziej, I.; Kępisty, M.; Kukurba-Setkowicz, M.; et al. Differences in sweet taste perception and its association with the streptococcus mutans cariogenic profile in preschool children with caries. Nutrients 2020, 12, 2592. [Google Scholar] [CrossRef]
- Alcock, J.; Maley, C.C.; Aktipis, C.A. Is eating behavior manipulated by the gastrointestinal microbiota? Evolutionary pressures and potential mechanisms. Bioessays 2014, 36, 940–949. [Google Scholar] [CrossRef]
- Holt, S.; Miks, M.H.; de Carvalho, B.T.; Foulquie-Moreno, M.R.; Thevelein, J.M. The molecular biology of fruity and floral aromas in beer and other alcoholic beverages. FEMS Microbiol. Rev. 2019, 43, 193–222. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Munoz-Gonzalez, C.; Cueva, C.; Angeles Pozo-Bayon, M.; Victoria Moreno-Arribas, M. Ability of human oral microbiota to produce wine odorant aglycones from odourless grape glycosidic aroma precursors. Food Chem. 2015, 187, 112–119. [Google Scholar] [CrossRef] [PubMed]
- Alegre, Y.; Sáenz-Navajas, M.-P.; Hernández-Orte, P.; Ferreira, V. Sensory, olfactometric and chemical characterization of the aroma potential of Garnacha and Tempranillo winemaking grapes. Food Chem. 2020, 331, 127207. [Google Scholar] [CrossRef]
- Gamero, A.; Hernandez-Orte, P.; Querol, A.; Ferreira, V. Effect of aromatic precursor addition to wine fermentations carried out with different Saccharomyces species and their hybrids. Int. J. Food Microbiol. 2011, 147, 33–44. [Google Scholar] [CrossRef] [PubMed]
- Van Wyk, N.; Grossmann, M.; Wendland, J.; von Wallbrunn, C.; Pretorius, I.S. The whiff of wine yeast innovation: Strategies for enhancing aroma production by yeast during wine fermentation. J. Agric. Food Chem. 2019, 67, 13496–13505. [Google Scholar] [CrossRef]
- Morata, A.; Escott, C.; Banuelos, M.A.; Loira, I.; Fresno, J.M.D.; Gonzalez, C.; Suarez-Lepe, J.A. Contribution of non-saccharomyces yeasts to wine freshness. A review. Biomolecules 2019, 10, 34. [Google Scholar] [CrossRef] [Green Version]
- Manzanares, P.; Valles, S.; Viana, F. Non-Saccharomyces Yeasts in the Winemaking Process; Academic Press: Amsterdam, The Netherlands, 2011. [Google Scholar]
- Schmidt, S.; Rainieri, S.; Witte, S.; Matern, U.; Martens, S. Identification of a Saccharomyces cerevisiae glucosidase that hydrolyzes flavonoid glucosides. Appl. Environ. Microbiol. 2011, 77, 1751–1757. [Google Scholar] [CrossRef] [Green Version]
- Morcol, T.B.; Negrin, A.; Matthews, P.D.; Kennelly, E.J. Hop (Humulus lupulus L.) terroir has large effect on a glycosylated green leaf volatile but not on other aroma glycosides. Food Chem. 2020, 321, 126644. [Google Scholar] [CrossRef] [PubMed]
- Daenen, L.; Sterckx, F.; Delvaux, F.R.; Verachtert, H.; Derdelinckx, G. Evaluation of the glycoside hydrolase activity of a Brettanomyces strain on glycosides from sour cherry (Prunus cerasus L.) used in the production of special fruit beers. FEMS Yeast Res. 2008, 8, 1103–1114. [Google Scholar] [CrossRef] [Green Version]
- Chauncey, H.H.; Lionetti, F.; Winer, R.A.; Lisanti, V.F. Enzymes of human saliva. I. The determination, distribution, and origin of whole saliva enzymes. J. Dent. Res. 1954, 33, 321–334. [Google Scholar] [CrossRef] [PubMed]
- Hemingway, K.M.; Alston, M.J.; Chappell, C.G.; Taylor, A.J. Carbohydrate-flavour conjugates in wine. Carbohydr. Polym. 1999, 38, 283–286. [Google Scholar] [CrossRef]
- Mayr, C.M.; Parker, M.; Baldock, G.A.; Black, C.A.; Pardon, K.H.; Williamson, P.O.; Herderich, M.J.; Francis, I.L. Determination of the importance of in-mouth release of volatile phenol glycoconjugates to the flavor of smoke-tainted wines. J. Agric. Food Chem. 2014, 62, 2327–2336. [Google Scholar] [CrossRef]
- Muñoz-González, C.; Feron, G.; Guichard, E.; Rodríguez-Bencomo, J.J.; Martín-Álvarez, P.J.; Moreno-Arribas, M.V.; Pozo-Bayón, M.Á. Understanding the role of saliva in aroma release from wine by using static and dynamic headspace conditions. J. Agric. Food Chem. 2014, 62, 8274–8288. [Google Scholar] [CrossRef] [Green Version]
- Aybeke, E.N.; Ployon, S.; Brulé, M.; De Fonseca, B.; Bourillot, E.; Morzel, M.; Lesniewska, E.; Canon, F. Nanoscale mapping of the physical surface properties of human buccal cells and changes induced by saliva. Langmuir 2019, 35, 12647–12655. [Google Scholar] [CrossRef]
- McGorrin, R.J. The significance of volatile sulfur compounds in food flavors. In Volatile Sulfur Compounds in Food; American Chemical Society: Washington, DC, USA, 2011; Volume 1068, pp. 3–31. [Google Scholar]
- Landaud, S.; Helinck, S.; Bonnarme, P. Formation of volatile sulfur compounds and metabolism of methionine and other sulfur compounds in fermented food. Appl. Microbiol. Biotechnol. 2008, 77, 1191–1205. [Google Scholar] [CrossRef] [PubMed]
- Cannon, R.J.; Ho, C.T. Volatile sulfur compounds in tropical fruits. J. Food Drug Anal. 2018, 26, 445–468. [Google Scholar] [CrossRef]
- Ferreira, V.; Lopez, R. The actual and potential aroma of winemaking grapes. Biomolecules 2019, 9, 818. [Google Scholar] [CrossRef] [Green Version]
- Gros, J.; Tran, T.T.H.; Collin, S. Enzymatic release of odourant polyfunctional thiols from cysteine conjugates in hop. J. Inst. Brew 2013, 119, 221–227. [Google Scholar] [CrossRef]
- Roncoroni, M.; Santiago, M.; Hooks, D.O.; Moroney, S.; Harsch, M.J.; Lee, S.A.; Richards, K.D.; Nicolau, L.; Gardner, R.C. The yeast IRC7 gene encodes a beta-lyase responsible for production of the varietal thiol 4-mercapto-4-methylpentan-2-one in wine. Food Microbiol. 2011, 28, 926–935. [Google Scholar] [CrossRef]
- Allegrini, A.; Astegno, A.; La Verde, V.; Dominici, P. Characterization of C-S lyase from Lactobacillus delbrueckii subsp. bulgaricus ATCC BAA-365 and its potential role in food flavour applications. J. Biochem. 2017, 161, 349–360. [Google Scholar] [CrossRef]
- Tominaga, T.; Peyrot des Gachons, C.; Dubourdieu, D. A New Type of Flavor Precursors in Vitis vinifera L. cv. Sauvignon Blanc: S-Cysteine Conjugates. J. Agric. Food Chem. 1998, 46, 5215–5219. [Google Scholar] [CrossRef]
- Peña-Gallego, A.; Hernández-Orte, P.; Cacho, J.; Ferreira, V. S-Cysteinylated and S-glutathionylated thiol precursors in grapes. A review. Food Chem. 2012, 131, 1–13. [Google Scholar] [CrossRef]
- Peyrot des Gachons, C.; Tominaga, T.; Dubourdieu, D. Sulfur Aroma Precursor Present in S-glutathione Conjugate Form: Identification of S-3-(Hexan-1-ol)-glutathione in Must from Vitis vinifera L. cv. Sauvignon Blanc. J. Agric. Food Chem. 2002, 50, 4076–4079. [Google Scholar] [CrossRef] [PubMed]
- Vermeulen, C.; Lejeune, I.; Tran, T.T.H.; Collin, S. Occurrence of Polyfunctional Thiols in Fresh Lager Beers. J. Agric. Food Chem. 2006, 54, 5061–5068. [Google Scholar] [CrossRef] [PubMed]
- Eriksen, R.L.; Padgitt-Cobb, L.K.; Townsend, M.S.; Henning, J.A. Gene expression for secondary metabolite biosynthesis in hop (Humulus lupulus L.) leaf lupulin glands exposed to heat and low-water stress. Sci. Rep. 2021, 11, 5138. [Google Scholar] [CrossRef]
- Belda, I.; Ruiz, J.; Navascues, E.; Marquina, D.; Santos, A. Improvement of aromatic thiol release through the selection of yeasts with increased beta-lyase activity. Int. J. Food Microbiol. 2016, 225, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Holt, S.; Cordente, A.G.; Williams, S.J.; Capone, D.L.; Jitjaroen, W.; Menz, I.R.; Curtin, C.; Anderson, P.A. Engineering Saccharomyces cerevisiae to release 3-Mercaptohexan-1-ol during fermentation through overexpression of an S. cerevisiae Gene, STR3, for improvement of wine aroma. Appl. Environ. Microbiol. 2011, 77, 3626–3632. [Google Scholar] [CrossRef] [Green Version]
- Cerny, C.; Guntz-Dubini, R. Formation of cysteine-S-conjugates in the Maillard reaction of cysteine and xylose. Food Chem. 2013, 141, 1078–1086. [Google Scholar] [CrossRef]
- Basic, A.; Blomqvist, M.; Dahlen, G.; Svensater, G. The proteins of Fusobacterium spp. involved in hydrogen sulfide production from L-cysteine. BMC Microbiol. 2017, 17, 61. [Google Scholar] [CrossRef] [Green Version]
- Frank, D.; Piyasiri, U.; Archer, N.; Jenifer, J.; Appelqvist, I. Influence of saliva on individual in-mouth aroma release from raw cabbage (Brassica oleracea var. capitata F. rubra L.) and links to perception. Heliyon 2018, 4, e01045. [Google Scholar] [CrossRef] [Green Version]
| Localization | Flavor Compounds | Producing Microbial Species | Food Product |
|---|---|---|---|
| Fermented beverages | Terpenes C13-norisoprenoids Volatile phenols C6 compounds Aliphatic alcohols Aliphatic acids Benzenic compounds Phenolic acid derivatives | Saccharomyces hybrids, Pichia anomala, Candida molischiana, Candida wickerhamii, Hanseniaspora uvarum, Metschnikowia pulcherrima | Wine [12,70,71] |
| Terpenes C13-norisoprenoids Benzenic compounds | Oenococcus oenii | Red wine [14] | |
| Terpenes Aliphatic alcohols | Brettanomyces bruxellensis, B. custersii, B. anomalus | Beer, special fruit beers [13,78] | |
| Oral cavity | Volatile phenols | unknown | Smoke affected wines [81] |
| Terpenes Benzenic compounds Lipid derivatives | unknown | White grapes [82] |
| Localization | Flavor Compound | Producing Microbial Species | Food Product |
|---|---|---|---|
| Fermented beverages | 4-mercapto-4-methylpentan-2-one | S. cerevisiae, E. limosum | Sauvignon wine [91,92] |
| 4-mercapto-4-methylpentan-2-ol | |||
| 3-mercaptohexan-1-ol | |||
| 3-methyl-2-buten-1-thiol | S. cerevisiae, P. kluyveri | Lager beer [69,94] | |
| 2-mercapto-3-methylbutanol | |||
| 3-mercapto-3-methylbutanol | |||
| Oral cavity | (R/S)-3-sulfanylhexan-1-ol | F. nucleatum | Grapes [8] |
| 1-propanethiol | Onion [8] | ||
| 2-heptanethiol | Bell pepper [8] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Schwartz, M.; Canon, F.; Feron, G.; Neiers, F.; Gamero, A. Impact of Oral Microbiota on Flavor Perception: From Food Processing to In-Mouth Metabolization. Foods 2021, 10, 2006. https://doi.org/10.3390/foods10092006
Schwartz M, Canon F, Feron G, Neiers F, Gamero A. Impact of Oral Microbiota on Flavor Perception: From Food Processing to In-Mouth Metabolization. Foods. 2021; 10(9):2006. https://doi.org/10.3390/foods10092006
Chicago/Turabian StyleSchwartz, Mathieu, Francis Canon, Gilles Feron, Fabrice Neiers, and Amparo Gamero. 2021. "Impact of Oral Microbiota on Flavor Perception: From Food Processing to In-Mouth Metabolization" Foods 10, no. 9: 2006. https://doi.org/10.3390/foods10092006
APA StyleSchwartz, M., Canon, F., Feron, G., Neiers, F., & Gamero, A. (2021). Impact of Oral Microbiota on Flavor Perception: From Food Processing to In-Mouth Metabolization. Foods, 10(9), 2006. https://doi.org/10.3390/foods10092006







