Sourdough Biotechnology Applied to Gluten-Free Baked Goods: Rescuing the Tradition
Abstract
:1. Introduction
2. Technological Aspects of Using Additives and Adjuvants in Gluten-Free Baked Goods and Joint Contributions with Sourdoughs
2.1. Hydrocolloids
2.2. Proteins
- Egg proteins (helped by the lecithin present in the yolk), act as foaming and emulsifying agents, and they are capable of stabilizing emulsions. These properties will improve the dough structure and gas retention, providing a softer crumb with more uniformly distributed alveoli. In addition, egg is a food with a very interesting nutritional profile, considered as a good source of high biological value proteins, fats, vitamins, and minerals (especially iron).
- The most-used milk proteins are casein, caseinates, and whey proteins. These proteins have gluten-like functional and technological properties, capable of creating cross-linked networks, and with a high capacity for swelling and water retention. Regarding GF bread, milk proteins contribute to Maillard reaction (between amino acids and reducing sugars), improving texture, roasted flavors and, mainly, both color and aroma crust quality.
- Although soybean is a protein-rich food, it is deficient in sulfur-containing amino acids, such as the essential amino acid methionine. It is used as a functional food to increase the nutritional value of GF bakery products, since it contains bioactive compounds such as isoflavones. Due to its technological properties, soybean also has a positive impact on the quality of the final product, by improving crumb, volume, water retention, and sensory assessment.
2.3. Enzymes
- Enzymes that modify starches, such as amylase and cyclodextrin glycosyltransferase; the latter degrades starch and produces dextrin that has been experimentally proven to increase the solubility of hydrophobic proteins, which in turn increases CO2 retention, providing a bigger loaf volume and a better texture [12,29,45]. Schober et al. indicated that bacterial α-amylase is used to supply sugars in the sourdough fermentation step, and also exerts an anti-staling effect in GF starch breads, so they included 0.01% of this enzyme in their sorghum sourdough formula [34].
- Enzymes that crosslink, or connect proteins, such as transglutaminase (TGase) and gluco-oxidase (GO). These enzymes, which catalyze protein polymerization and crosslinking reactions, can create a kind of network or mesh, such as the three-dimensional structure provided by gluten, that improves CO2 retention [12,30,46].
- Proteases that hydrolyze the peptide bonds of the proteins. This property can improve texture and final quality of rice-flour-based breads [12,30]. Additionally, proteolysis that occurs during the sourdough fermentation process could prevent interferences between protein aggregation upon baking and the starch gel, which seems to be desirable in GF sorghum breads [34].
2.4. Emulsifiers
- Soy lecithin, a plant origin product, which is extracted from soybeans. It has a very high concentration of phospholipids that contribute to dough extensibility, and flour hydration properties.
- Mono- and di-glycerides of fatty acids (E–471) [48] have the property of softening the dough, facilitating mixtures at an industrial level, thus achieving a crumb with more alveoli and a larger final volume. They also decrease starch retrogradation, which improves the shelf life of bakery products (especially pastries).
- Esters of mono- and di-glycerides fatty acids (E–472a–E47f) [48], are mainly used in the preparation of bread, since they provide a better “body” to the dough (an excessively liquid dough is an important defect of the GF products); this equates to a firmer dough with greater gas retention, and both texture and final volume improvements. These emulsifiers also contribute to an increased shelf life of bakery products.
3. Sourdough Biotechnology
3.1. Factors Affecting Sourdough Microbiota
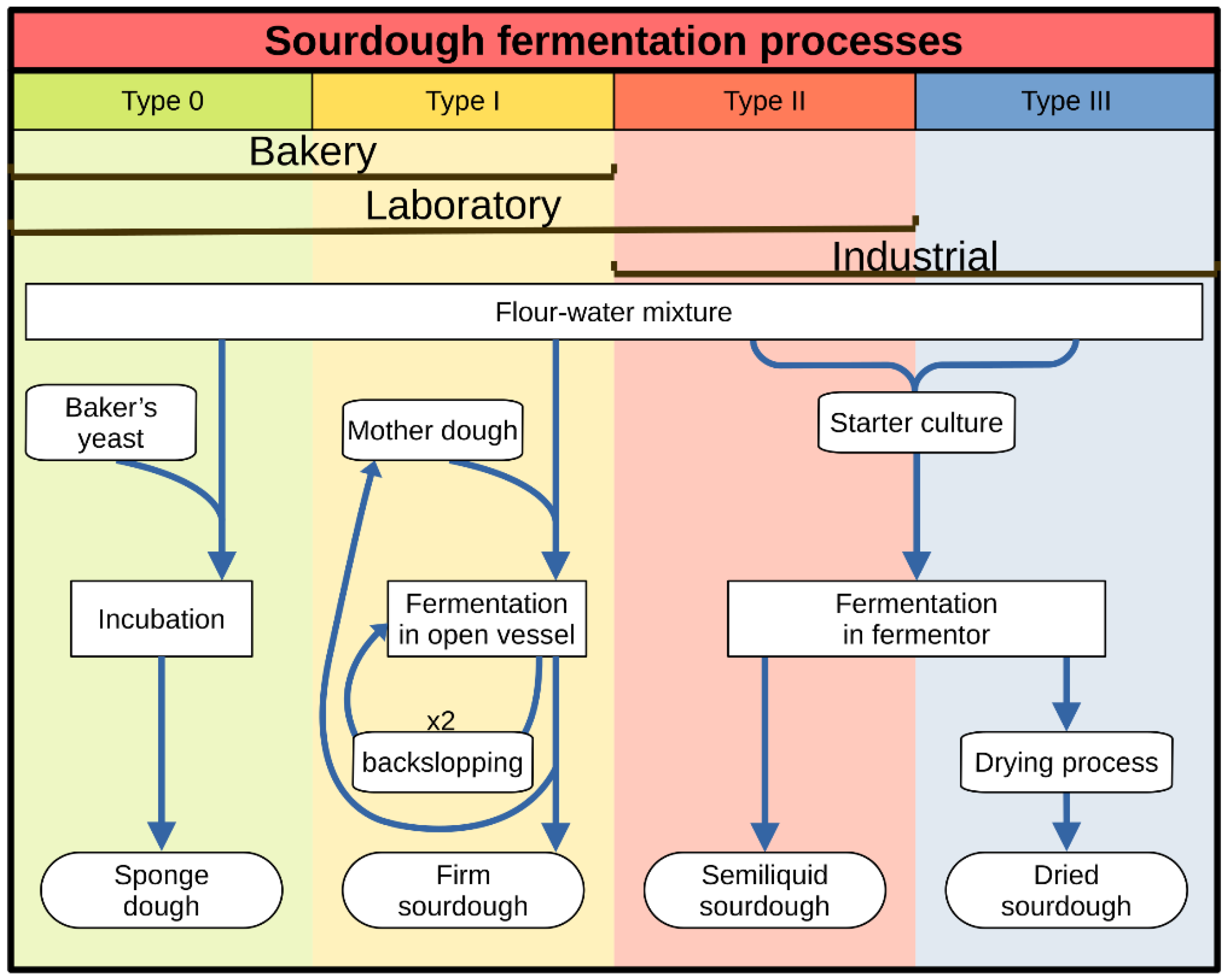
3.1.1. Sourdough Fermentation Processes
- 1.
- Sourdough Type
- Type 0 sourdough is a type of pre-dough, also known as mother sponge, characterized by a short fermentation time at room temperature (RT, <30 °C). This provokes the initial propagation of native and exogenous LAB, with a higher proliferation rate compared to yeast, producing bioactive molecules and organic acids (lactic and acetic acids) that diminish the pH (pH~4). Given the short fermentation time, yeast growing is not enough in the sourdough and it is mandatory to add commercial yeast preparations. The microbiota that can be found in type 0 sourdough is a variety of LAB species; some of them are present in other types of sourdough, and others are not usually isolated and do not contribute to the improvement of the final product. It should be noted that in this type of sourdough there is no time to select those microorganisms with a higher adaptability to sourdough ecosystems, such as the yeast Saccharomyces cerevisiae. Typical examples are solid pre-ferments, such as biga from Italy and pâte fermentêe from France; and hydrated pre-ferments, such as the levain levure from north Europe, and poolish from Poland.
- Type I sourdough can be considered as the traditional sourdough, probably the one that spontaneously emerged in antiquity. Used in artisan bakeries and domestic settings, it considerably increases the quality of the final baked good. Type I sourdoughs have a long fermentation time at RT and are composed of very few microorganism species with the highest adaptation rates, the highest resistance, being the most competitive, and capable of stablishing solid associations between them. A typical example is the sourdough from San Francisco, mainly fermented by the LAB Lactobacillus sanfranciscensis (named because it was first isolated and described in this type of sourdough—reclassified as Fructilactobacillus sanfranciscensis [55]—) and the yeast Candida humilis. The association between these two microorganisms is very stable, since Lb. sanfranciscensis use maltose and Candida humilis use glucose, so they do not compete for the carbon source. They are also very competitive, displacing other species [56].
- Type II sourdough is a semiliquid fermented dough that can be bombed and used at an industrial scale. A starter culture is usually added to this type of sourdough, which is composed of LAB species that rapidly acidify the mixture and/or generate compounds that provide the aromas and flavors of traditional sourdough. Long fermentation times are used (two to five days) in only one step and at high temperatures (>30 °C). At these conditions, LAB rapidly proliferate (due to the high temperatures that facilitate their growing), with the consequent production of organic acids, the decrease of pH (pH < 3.4), and the yeast growing inhibition at this pH. This leads to the selection of acid-tolerant and thermophilic LAB (selection that is forced when commercial starter cultures are used) and requires adding industrial yeast. Some examples of Lactobacillus species isolated from type II sourdough are Lactobacillus fermentum (pro synonymon —pro synon.—Limosilactobacillus fermentum), Lactobacillus plantarum (pro synon. Lactiplantibacillus plantarum) and Lactobacillus reuteri (pro synon. Limosilactobacillus reuteri) [55]; from rye sourdough, Lb. amylovorus is also frequently isolated [54].
- Type III sourdough is a freeze-dried type II sourdough to facilitate its commercialization and later industrial use.
- 2.
- Temperature of Fermentation
- 3.
- Dough Yield
- 4.
- Other Factors
- The pH of the sourdough, affected by LAB or yeast presence and fermentation stage [58].
- Additional nutrient sources: traditional ingredients added to sourdough final mixes complement the nutrient content of the sourdough—e.g., adding mono- and disaccharides or different amino acid sources, thus affecting the intrinsic parameters for microbial growth [58,59] and the microbial composition itself [60].
- Ash content in the bran fraction of the flour. The bran fraction contains several minerals and micronutrients that can promote the growth of LAB in the sourdough. The ash content also influences the buffering capacity of the sourdough system that makes it possible to reach a higher total titratable activity [58].
- The resting time of the dough and its temperature; if it is performed at cold temperatures, it will favor microorganisms that are resistant to cold stress and to the absence of substratum [54].
3.1.2. Instrumental Techniques for the Isolation and Identification of Microorganisms
- 1.
- Sampling
- 2.
- Fermentation Place
- 3.
- Isolation and Identification Techniques
3.2. Sourdough Autochthonous Microbiota
3.2.1. Gluten-Containing Sourdough
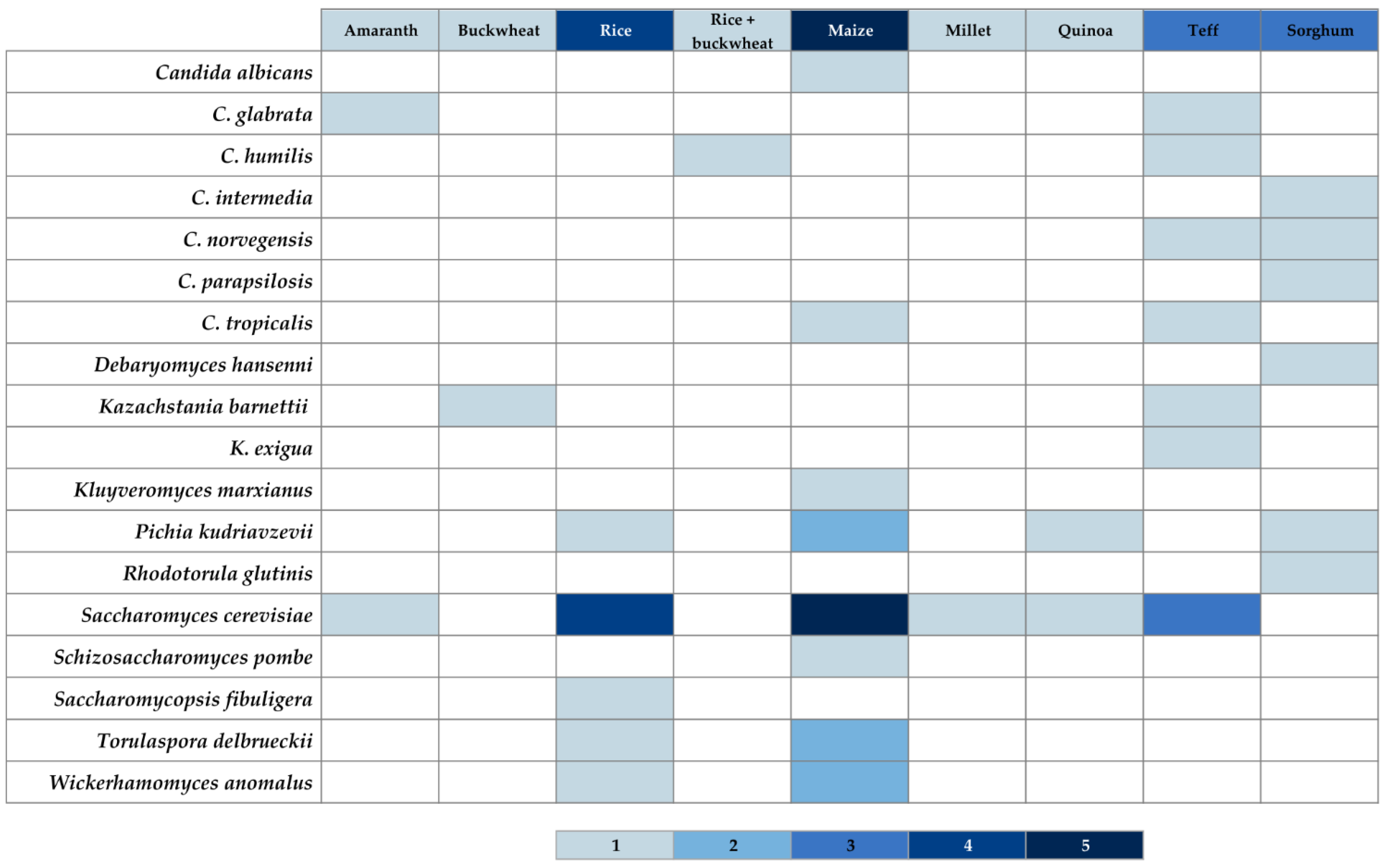
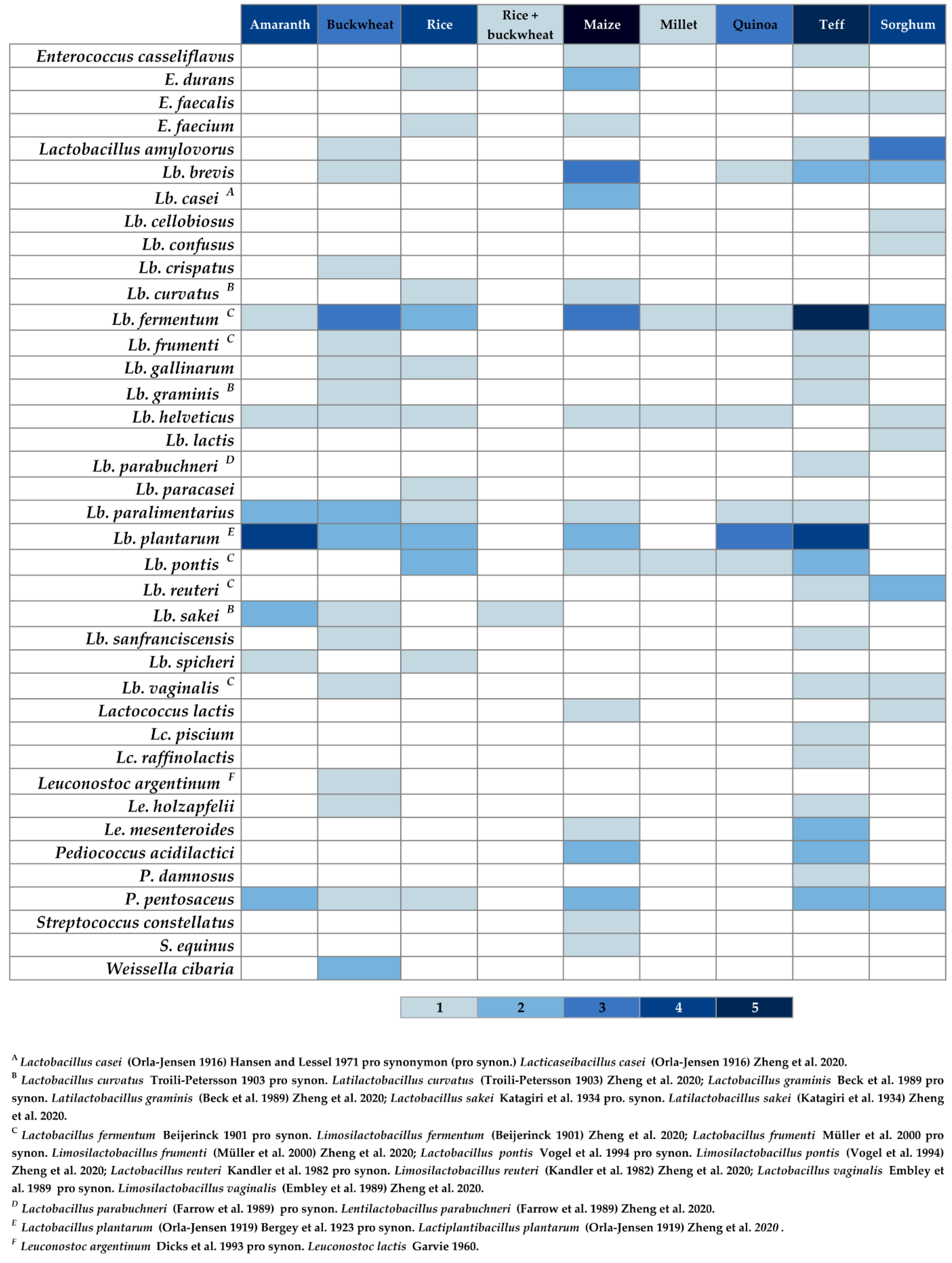
3.2.2. Gluten-Free Sourdough
| Country 1 | Flour Type 1 | Propagation Method 1 | Identification Method 1 | Microorganisms Reported (LAB 2/Y 3) | Reference(s) |
|---|---|---|---|---|---|
| Argentina | Amaranth | Laboratory | Molecular | LAB: Lactobacillus plantarum 4 | [59,69] |
| Quinoa | Laboratory | Molecular | LAB: Lb. brevis 5, Lb. plantarum | [58,59] | |
| n.i. | n.i. | LAB: Lb. plantarum | [17,64] | ||
| Belgium | Teff | Bakery | Molecular | LAB: L. brevis, L. helveticus, Lb. plantarum, L. sanfranciscensis, P. pentosaceus | [70] |
| Y: K. exigua | |||||
| Laboratory | Molecular | LAB: L. fermentum, Lb. plantarum, L. sanfranciscensis, W. cibaria, and P. pentosaceus | |||
| Y: S. cerevisiae | |||||
| Botswana | Sorghum | n.i. | n.i. | LAB: Lb. harbinensis 6, Lb. parabuchneri 7, Lb. plantarum | [64,71] |
| China | Rice | Bakery | Molecular | LAB: Enterococcus durans, E. faecium, Lb. plantarum, Pediococcus pentosaceus | [59,72] |
| Y: Saccharomyces cerevisiae, Saccharomycopsis fibuligera, Torulaspora delbrueckii, Wickerhamomyces anomalus | |||||
| Maize | Bakery | Molecular | LAB: E. durans, Lb. plantarum, P. pentosaceus | ||
| Y: S. cerevisiae, T. delbrueckii, W. anomalus | |||||
| Ethiopia | Teff | Laboratory | Phenotypic | LAB: E. faecalis, Lb. brevis, Lb. fermentum 8, Lb. plantarum, Leuconostoc mesenteroides | [59,73] |
| Laboratory | Molecular + phenotypic | LAB: Lb. fermentum, Lb. graminis 9, Lb. parabuchneri, Lb. plantarum | [59,74] | ||
| Laboratory | Phenotypic | LAB: E. casseliflavus, Lb. fermentum, Lactococcus piscium, Lc. plantarum, Lc. raffinolactis, Le. mesenteroides, P. acidilactici, P. pentosaceus | [59,75] | ||
| Y: Candida humilis, C. tropicalis, Kazachstania exigua, Pichia norvegensis, S. cerevisiae | |||||
| Laboratory | Molecular + phenotypic | LAB: Lb. fermentum, Lb. graminis, Lb. parabuchneri, Lb. plantarum | [64,74] | ||
| France | Rice + buckwheat | Laboratory | Molecular | LAB: Lb. sakei 10 | [59,76] |
| Y: C. humilis | |||||
| Ghana | Maize | Bakery | Phenotypic | Y: C. tropicalis, Kluyveromyces marxianus, P. kudriavzevii, S. cerevisiae | [59,77] |
| Germany | Buckwheat | Laboratory | Molecular | LAB: Lb. fermentum, Lb. helveticus, Lb. paralimentarius, Lb. plantarum | [59,78] |
| Y: not detected | |||||
| Amaranth | Laboratory | Molecular | LAB: Lb. paralimentarius, Lb. plantarum, Lb. sakei, P. pentosaceus | [59,79] | |
| Laboratory, use of a starter including all LAB species on the right column | Molecular | LAB: Lb. plantarum, Lb. sakei, P. pentosaceus | |||
| Laboratory, use of a starter including all LAB species on the right column and Lb. acetotolerans, Lb. brevis, Lb. casei, Lb. curvatus, Lb. sanfranciscensis, Lb. spicheri, Lc. lactis, Le. paramesenteroides and yeast species C. humilis, W. anomalus, P. kudriavzevii, S. cerevisiae, Torulaspora sp | Molecular | LAB: Lb. fermentum, Lb. helveticus, Lb. paralimentarius, Lb. plantarum, Lb. spicheri 11 | [59,78] | ||
| Y: C. glabrata, S. cerevisiae | |||||
| Laboratory | Molecular | LAB: Lb. plantarum, Lb. sakei | [64,79] | ||
| Rice | Laboratory, use of a starter (mother sponge) including underlined species on the right column and Lb. perolens | Molecular + phenotypic | LAB: Lb. paracasei, Lb. paralimentarius, Lb. spicheri | [59,80] | |
| Y: S. cerevisiae | |||||
| Laboratory, use of a starter including underlined species on the right column and yeast specie P. membranifaciens. | Molecular + phenotypic | LAB: Lb. curvatus, Lb. fermentum, Lb. gallinarum, Lb. kimchii 12, Lb. plantarum, Lb. pontis 13 | |||
| Y: P. kudriavzevii, S. cerevisiae | |||||
| Laboratory | Molecular | LAB: Lb. fermentum, Lb. helveticus, Lb. plantarum, Lb. pontis | [59,78] | ||
| Y: S. cerevisiae | |||||
| Laboratory | Molecular | LAB: Lb. kimchii, Lb. paralimentarius, Lb. perolens 14 | [64,80] | ||
| Maize | Laboratory, use of a starter including all species on the right column and Lb. acetotolerans, Lb. brevis, Lb. casei, Lb. curvatus, Lb. sanfranciscensis, Lb. spicheri, Lc. lactis, Le. paramesenteroides and yeast species C. humilis, W. anomalus, Torulaspora sp. | Molecular | LAB: Lb. fermentum, Lb. helveticus, Lb. paralimentarius, Lb. pontis | [59,78] | |
| Y: P. kudriavzevii, S. cerevisiae | |||||
| Millet | Molecular | LAB: Lb. fermentum, Lb. helveticus, Lb. pontis | |||
| Y: S. cerevisiae | |||||
| Quinoa | Molecular | LAB: Lb. fermentum, Lb. helveticus, Lb. paralimentarius, Lb. plantarum, Lb. pontis | |||
| Y: P. kudriavzevii, S. cerevisiae | |||||
| Italy | Quinoa | Laboratory | Molecular | LAB: Lb. plantarum | [17,59] |
| Teff | Laboratory | Molecular | LAB: Lb. plantarum, Lb. fermentum. | [81] | |
| Y: S. cerevisiae | |||||
| Ireland | Buckwheat | Laboratory use of a starter use of a starter including all LAB species on the right column and Lb. helveticus, Lb. paracasei, Lb. pontis, Lb. reuteri, and yeast species C. humilis and S. pastorianus | Molecular | LAB: Lb. amylovorus, Lb. brevis, Lb. fermentum, Lb, frumenti 15, Lb. paralimentarius, Lb. plantarum, Lb. sanfranciscensis 16, Leuconostoc argentinum 17, Weissella cibaria | [59,82] |
| Y: not detected | |||||
| Laboratory | Molecular + phenotypic | LAB: Lb. acidophilus, Lb. amylovorus, Lb. crispatus, Lb. fermentum, Lb. gallinarum, Lb. graminis, Lb. helveticus, Lb. plantarum, Lb.sakei, Lb. vaginalis | [64,83] | ||
| Laboratory | Molecular | LAB: Lb. crispatus, Lb. fermentum, Lb. gallinarum, Lb. graminis, Lb. plantarum, Lb. sakei, Lb. vaginalis, Le. holzapfelii, P. pentosaceus, W. cibaria | [59,83] | ||
| Y: K. barnetti | |||||
| Teff | Laboratory, use of a starter use of a starter including all LAB species on the right column and Lb. helveticus, Lb. paracasei, Lb. pontis, Lb. reuteri, and yeast species C. humilis and S. pastorianus | Molecular | LAB: Lb. amylovorus, Lb. brevis, Lb. fermentum, Lb. frumenti, Lb. paralimentarius, Lb. plantarum, Lb. pontis, Lb. reuteri 18, Lb. sanfranciscensis, P. acidilactici | ||
| Y: K. barnettii, S. cerevisiae | |||||
| Laboratory | Molecular + phenotypic | LAB: Lb. amylovorus, Lb. fermentum, Lb. gallinarum, Lb. plantarum, Lb. vaginalis 19 | [64,83] | ||
| Laboratory | Molecular | LAB: Lb. fermentum, Lb. gallinarum, Lb. pontis, Lb. vaginalis, Le. holzapfelii, P. pentosaceus | [59,83] | ||
| Y: C. glabrata, S. cerevisiae | |||||
| Morocco | Maize | n.i. | n.i. | LAB: Lb. alimentarius, Lb. casei 20 | [64,84] |
| Nigeria | Maize | Laboratory | Molecular | LAB: Lb. brevis, Lb. casei, Lb. fermentum, Lb. plantarum, Le. mesenteroides, P. acidilactici | [59,85] |
| Y: C. albicans, S. cerevisiae, Schizosaccharomyces pombe | |||||
| Laboratory | Phenotypic | LAB: Lb. brevis, Lb. casei, Lb. fermentum, P. acidilactici, P. pentosaceus | [59,86] | ||
| Laboratory | Molecular | LAB: Lb. acidophilus, Lb. brevis, Lb. casei, Lb. fermentum, Lb. plantarum | [64,85] | ||
| Portugal | Maize | Bakery | Phenotypic | LAB: E. casseliflavus, E. durans, E. faecium, Lb. brevis, Lb. curvatus, Lc. lactis subsp. lactis, Leuconostoc spp., Streptococcus constellatus, S. equinus | [59,64,87] |
| Y: S. cerevisiae, T. delbrueckii, W. anomalus | |||||
| Saudi Arabia | Sorghum | Bakery | Phenotypic | LAB: Lb. brevis, Lb. cellobiosus 21, Lb. lactis, P. pentosaceus | [59,64,88] |
| Y: C. norvegensis, C. parapsilosis, Rhodotorula glutinis | |||||
| Sudan | Sorghum | Laboratory | Phenotypic | LAB: Lb. brevis, Lb. confusus 22, Lactobacillus spp., P. pentosaceus | [59,89] |
| Y: C. intermedia, Debaromyces hansenni | |||||
| Laboratory | Phenotypic | LAB: Lb. amylovorus, Lb. fermentum, Lb. reuteri | [59,90] | ||
| Y: P. kudriavzevii | |||||
| Laboratory | Molecular + phenotypic | LAB: E. faecalis, Lb. fermentum, Lb. helveticus, Lb. reuteri, Lb. vaginalis, Lc. lactis | [59,64,90,91] |
4. Identification of Microorganisms Capable of Producing Hydrocolloid-Like Compounds
- HePS are described as such because the sugar polymer chain is made of different monosaccharides, usually D-galactose, D-glucose, R-rhamnose and, to a lesser extent, other N-acetylated monosaccharides, varying from two to eight different monomers, and with a molecular weight up to 106 Da. A large variety of HePS can be synthesized by LAB, depending on the type of monosaccharides, bonds between these monosaccharides, and spatial configurations (linear vs. branched). As an example, Suzuki et al. studied how Lactococcus lactis can synthesize a high number of different HePS [102]. HePS are synthesized from sugar–nucleotide precursors, intracellularly (in the cytoplasm), and in small quantities, usually between 10 and 166 mg/L. The yield of this synthesis depends on several factors: by optimizing some culture parameters of Lb. plantarum, Ismail and Nampoothiri achieved a final EPS concentration of 1.2 g/L [103]. Xanthan and gellan gums are HePS synthesized by bacteria belonging to phylum “Proteobacteria”.
- HoPS are polymers based on a single type of monosaccharide (glucose or fructose), and, because of this, they are recognized as glucans or fructans (also designated as fructooligosaccharides or FOS) [96,100]. Its synthesis is extracellular, from sucrose, by the action of enzymes (glycosyl hydroxylases), and with a molecular weight greater than HePS (>106 Da). For the polymerization of glucose or fructose, these enzymes employ the energy of the glycosidic bond. HoPS are synthesized by different genera of LAB (mainly, Lactobacillus, Streptococcus, Leuconostoc, Oenococcus and Weissella) and in an amount greater than HePS, reaching up to 10 g/L. In addition to this first classification of HoPS (in glucans and fructans), these compounds are also classified based on the carbons involved in the glycosidic linkages of the backbone chain of the polymer.
- ○
- Within the group of glucans, the following types are recognized: dextrans, mutans, reuterans, and alternans. Dextrans are the HoPS with the most technological relevance, being the only EPS synthesized at an industrial level, widely used as, for example, a thickener for jams and ice cream: they reduce crystallization, increase moisture retention, and do not affect taste.
- ○
- Two types of fructans can be distinguished: inulin and levan. As its prebiotic properties, inulin is acquiring a greater role in the current market. Recently, it has been reported that fructans can induce gastrointestinal symptoms in individuals with self-reported non-celiac gluten sensitivity [104].
- Using mixtures of GF flours, where each flour supplies a type of bacteria that produces the EPS that we are looking for.
- Using controlled fermentation processes oriented to the development of the microbiota of interest.
- Using commercial starters based on bacteria strains selected because of their technological potential.
5. Concluding Remarks and Future Perspectives
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Ribeiro, M.; Nunes, F.M.; Rodriguez-Quijano, M.; Carrillo, J.M.; Branlard, G.; Igrejas, G. Next-generation therapies for celiac disease: The gluten-targeted approaches. Trends Food Sci. Technol. 2018, 75, 56–71. [Google Scholar] [CrossRef]
- Xhakollari, V.; Canavari, M.; Osman, M. Factors affecting consumers’ adherence to gluten-free diet, a systematic review. Trends Food Sci. Technol. 2019, 85, 23–33. [Google Scholar] [CrossRef]
- Scherf, K.A.; Catassi, C.; Chirdo, F.; Ciclitira, P.J.; Feighery, C.; Gianfrani, C.; Koning, F.; Lundin, K.E.A.; Schuppan, D.; Smulders, M.J.M.; et al. Recent Progress and Recommendations on Celiac Disease from the Working Group on Prolamin Analysis and Toxicity. Front. Nutr. 2020, 7, 14. [Google Scholar] [CrossRef] [Green Version]
- Lundin, K.E.A.; Wijmenga, C. Coeliac disease and autoimmune disease—genetic overlap and screening. Nat. Rev. Gastroenterol. Hepatol. 2015, 12, 507–515. [Google Scholar] [CrossRef]
- Singh, P.; Arora, A.; Strand, T.A.; Leffler, D.A.; Catassi, C.; Green, P.H.; Kelly, C.P.; Ahuja, V.; Makharia, G.K. Global Prevalence of Celiac Disease: Systematic Review and Meta-analysis. Clin. Gastroenterol. Hepatol. 2018, 16, 823–836.e2. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ciccocioppo, R.; Kruzliak, P.; Cangemi, G.C.; Pohanka, M.; Betti, E.; Lauret, E.; Rodrigo, L. The Spectrum of Differences between Childhood and Adulthood Celiac Disease. Nutrients 2015, 7, 8733–8751. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lionetti, E.; Gatti, S.; Pulvirenti, A.; Catassi, C. Celiac disease from a global perspective. Best Pract. Res. Clin. Gastroenterol. 2015, 29, 365–379. [Google Scholar] [CrossRef] [PubMed]
- Fedorak, R.N.; Switzer, C.M.; Bridges, R.J. Canadian Digestive Health Foundation Public Impact Series 4: Celiac disease in Canada. Incidence, prevalence, and direct and indirect economic impact. Can. J. Gastroenterol. 2012, 26, 350–352. [Google Scholar] [CrossRef] [PubMed]
- Catassi, C.; Fabiani, E.; Ratsch, I.M.; Coppa, G.V.; Giorgi, P.L.; Pierdomenico, R.; Alessandrini, S.; Iwanejko, G.; Domenici, R.; Mei, E.; et al. The coeliac iceberg in Italy. A multicentre antigliadin antibodies screening for coeliac disease in school-age subjects. Acta Paediatr. Suppl. 1996, 412, 29–35. [Google Scholar] [CrossRef]
- Capriles, V.D.; dos Santos, F.G.; Arêas, J.A.G. Gluten-free breadmaking: Improving nutritional and bioactive compounds. J. Cereal Sci. 2016, 67, 83–91. [Google Scholar] [CrossRef]
- Masure, H.G.; Fierens, E.; Delcour, J.A. Current and forward looking experimental approaches in gluten-free bread making research. J. Cereal Sci. 2016, 67, 92–111. [Google Scholar] [CrossRef]
- Capriles, V.D.; Arêas, J.A.G. Novel Approaches in Gluten-Free Breadmaking: Interface between Food Science, Nutrition, and Health. Compr. Rev. Food Sci. Food Saf. 2014, 13, 871–890. [Google Scholar] [CrossRef]
- do Nascimento, A.B.; Fiates, G.M.R.; Dos Anjos, A.; Teixeira, E. Analysis of ingredient lists of commercially available gluten-free and gluten-containing food products using the text mining technique. Int. J. Food Sci. Nutr. 2013, 64, 217–222. [Google Scholar] [CrossRef] [PubMed]
- Hüttner, E.K.; Arendt, E.K. Recent advances in gluten-free baking and the current status of oats. Trends Food Sci. Technol. 2010, 21, 303–312. [Google Scholar] [CrossRef]
- Naqash, F.; Gani, A.; Gani, A.; Masoodi, F.A. Gluten-free baking: Combating the challenges - A review. Trends Food Sci. Technol. 2017, 66, 98–107. [Google Scholar] [CrossRef]
- Moroni, A.V.; Dal Bello, F.; Arendt, E.K. Sourdough in gluten-free bread-making: An ancient technology to solve a novel issue? Food Microbiol. 2009, 26, 676–684. [Google Scholar] [CrossRef]
- Rizzello, C.G.; Lorusso, A.; Montemurro, M.; Gobbetti, M. Use of sourdough made with quinoa (Chenopodium quinoa) flour and autochthonous selected lactic acid bacteria for enhancing the nutritional, textural and sensory features of white bread. Food Microbiol. 2016, 56, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Novotni, D.; Čukelj, N.; Smerdel, B.; Bituh, M.; Dujmić, F.; Ćurić, D. Glycemic index and firming kinetics of partially baked frozen gluten-free bread with sourdough. J. Cereal Sci. 2012, 55, 120–125. [Google Scholar] [CrossRef]
- Polanco Allué, I. Libro Blanco de la Enfermedad Celíaca, 1st ed.; ICM: Madrid, Spain, 2008; ISBN 978-84-936109-4-4. [Google Scholar]
- Nionelli, L.; Rizzello, C.G. Sourdough-Based Biotechnologies for the Production of Gluten-Free Foods. Foods 2016, 5, 65. [Google Scholar] [CrossRef]
- Galle, S.; Schwab, C.; Arendt, E.; Ganzle, M. Exopolysaccharide-forming Weissella strains as starter cultures for sorghum and wheat sourdoughs. J. Agric. Food Chem. 2010, 58, 5834–5841. [Google Scholar] [CrossRef]
- BeMiller, J.N. 9-Hydrocolloids. In Gluten-Free Cereal Products and Beverages; Arendt, E.K., Dal Bello, F., Eds.; Food Science and Technology; Academic Press: San Diego, CA, USA, 2008; pp. 203–215. ISBN 978-0-12-373739-7. [Google Scholar]
- Sabanis, D.; Tzia, C. Effect of hydrocolloids on selected properties of gluten-free dough and bread. Food Sci. Technol. Int. 2011, 17, 279–291. [Google Scholar] [CrossRef]
- Zannini, E.; Jones, J.M.; Renzetti, S.; Arendt, E.K. Functional Replacements for Gluten. Annu. Rev. Food Sci. Technol. 2012, 3, 227–245. [Google Scholar] [CrossRef]
- Mir, S.A.; Shah, M.A.; Naik, H.R.; Zargar, I.A. Influence of hydrocolloids on dough handling and technological properties of gluten-free breads. Trends Food Sci. Technol. 2016, 51, 49–57. [Google Scholar] [CrossRef]
- Demirkesen, I.; Kelkar, S.; Campanella, O.H.; Sumnu, G.; Sahin, S.; Okos, M. Characterization of structure of gluten-free breads by using X-ray microtomography. Food Hydrocoll. 2014, 36, 37–44. [Google Scholar] [CrossRef]
- Hager, A.-S.; Arendt, E.K. Influence of hydroxypropylmethylcellulose (HPMC), xanthan gum and their combination on loaf specific volume, crumb hardness and crumb grain characteristics of gluten-free breads based on rice, maize, teff and buckwheat. Food Hydrocoll. 2013, 32, 195–203. [Google Scholar] [CrossRef]
- O’Shea, N.; Arendt, E.; Gallagher, E. State of the art in gluten-free research. J. Food Sci. 2014, 79, R1067-76. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Drabińska, N.; Zieliński, H.; Krupa-Kozak, U. Technological benefits of inulin-type fructans application in gluten-free products—A review. Trends Food Sci. Technol. 2016, 56, 149–157. [Google Scholar] [CrossRef]
- Wang, K.; Lu, F.; Li, Z.; Zhao, L.; Han, C. Recent developments in gluten-free bread baking approaches: A review. Food Sci. Technol. 2017, 37, 1–9. [Google Scholar] [CrossRef] [Green Version]
- Tsatsaragkou, K.; Gounaropoulos, G.; Mandala, I. Development of gluten free bread containing carob flour and resistant starch. LWT - Food Sci. Technol. 2014, 58, 124–129. [Google Scholar] [CrossRef]
- Gao, Y.; Janes, M.E.; Chaiya, B.; Brennan, M.A.; Brennan, C.S.; Prinyawiwatkul, W. Gluten-free bakery and pasta products: Prevalence and quality improvement. Int. J. Food Sci. Technol. 2017, 53, 19–32. [Google Scholar] [CrossRef] [Green Version]
- Costa, A.F.S.; Almeida, F.C.G.; Vinhas, G.M.; Sarubbo, L.A. Production of Bacterial Cellulose by Gluconacetobacter hansenii Using Corn Steep Liquor As Nutrient Sources. Front. Microbiol. 2017, 8, 2027. [Google Scholar] [CrossRef]
- Schober, T.J.; Bean, S.R.; Boyle, D.L. Gluten-Free Sorghum Bread Improved by Sourdough Fermentation: Biochemical, Rheological, and Microstructural Background. J. Agric. Food Chem. 2007, 55, 5137–5146. [Google Scholar] [CrossRef]
- Campo, E.; del Arco, L.; Urtasun, L.; Oria, R.; Ferrer-Mairal, A. Impact of sourdough on sensory properties and consumers’ preference of gluten-free breads enriched with teff flour. J. Cereal Sci. 2016, 67, 75–82. [Google Scholar] [CrossRef]
- Demirkesen, I.; Mert, B.; Sumnu, G.; Sahin, S. Rheological properties of gluten-free bread formulations. J. Food Eng. 2010, 96, 295–303. [Google Scholar] [CrossRef]
- Cato, L.; Gan, J.J.; Rafael, L.G.B.; Small, D.M. Gluten free breads using rice flour and hydrocolloid gums. Food Aust. 2004, 56, 75–78. [Google Scholar]
- Perez-Quirce, S.; Caballero, P.A.; Vela, A.J.; Villanueva, M.; Ronda, F. Impact of yeast and fungi (1 → 3)(1 → 6)-β-glucan concentrates on viscoelastic behavior and bread making performance of gluten-free rice-based doughs. Food Hydrocoll. 2018, 79, 382–390. [Google Scholar] [CrossRef]
- Gularte, M.A.; de la Hera, E.; Gómez, M.; Rosell, C.M. Effect of different fibers on batter and gluten-free layer cake properties. LWT Food Sci. Technol. 2012, 48, 209–214. [Google Scholar] [CrossRef] [Green Version]
- Korus, J.; Grzelak, K.; Achremowicz, K.; Sabat, R. Influence of prebiotic additions on the quality of gluten-free bread and on the content of inulin and fructooligosaccharides. Food Sci. Technol. Int. 2006, 12, 489–495. [Google Scholar] [CrossRef]
- Deora, N.S.; Deswal, A.; Mishra, H.N. Functionality of alternative protein in gluten-free product development. Food Sci. Technol. Int. 2015, 21, 364–379. [Google Scholar] [CrossRef] [PubMed]
- Fasano, A.; Catassi, C. Celiac Disease. N. Engl. J. Med. 2012, 367, 2419–2426. [Google Scholar] [CrossRef]
- Renzetti, S.; Dal Bello, F.; Arendt, E.K. Microstructure, fundamental rheology and baking characteristics of batters and breads from different gluten-free flours treated with a microbial transglutaminase. J. Cereal Sci. 2008, 48, 33–45. [Google Scholar] [CrossRef]
- Steffolani, M.E.; Ribotta, P.D.; Pérez, G.T.; León, A.E. Effect of glucose oxidase, transglutaminase, and pentosanase on wheat proteins: Relationship with dough properties and bread-making quality. J. Cereal Sci. 2010, 51, 366–373. [Google Scholar] [CrossRef]
- Moore, M.M.; Heinbockel, M.; Dockery, P.; Ulmer, H.M.; Arendt, E.K. Network formation in gluten-free bread with application of transglutaminase. Cereal Chem. 2006, 83, 28–36. [Google Scholar] [CrossRef]
- Storck, C.R.; da Rosa Zavareze, E.; Gularte, M.A.; Elias, M.C.; Rosell, C.M.; Guerra Dias, A.R. Protein enrichment and its effects on gluten-free bread characteristics. LWT Food Sci. Technol. 2013, 53, 346–354. [Google Scholar] [CrossRef]
- Lerner, A.; Matthias, T. Possible association between celiac disease and bacterial transglutaminase in food processing: A hypothesis. Nutr. Rev. 2015, 73, 544–552. [Google Scholar] [CrossRef]
- European Commission Commission Regulation (EU). No 1129/2011 of 11 November 2011 amending Annex II to Regulation (EC) No 1333/2008 of the European Parliament and of the Council by establishing a Union list of food additives. Off. J. Eur. Union 2011, L, 1–177. [Google Scholar] [CrossRef]
- Mohammadi, M.; Azizi, M.H.; Neyestani, T.R.; Hosseini, H.; Mortazavian, A.M. Development of gluten-free bread using guar gum and transglutaminase. J. Ind. Eng. Chem. 2015, 21, 1398–1402. [Google Scholar] [CrossRef]
- López-Tenorio, J.A.; Rodríguez-Sandoval, E.; Sepúlveda-Valencia, J.U. The Influence of Different Emulsifiers on the Physical and Textural Characteristics of Gluten-Free Cheese Bread. J. Texture Stud. 2015, 46, 227–239. [Google Scholar] [CrossRef]
- Rubel, W. Bread: A Global History; Reaktion Books: London, UK, 2011; ISBN 9781861898548. [Google Scholar]
- Gänzle, M.; Ripari, V. Composition and function of sourdough microbiota: From ecological theory to bread quality. Int. J. Food Microbiol. 2016, 239, 19–25. [Google Scholar] [CrossRef] [PubMed]
- Ruiz Rodríguez, L.; Vera Pingitore, E.; Rollan, G.; Cocconcelli, P.S.; Fontana, C.; Saavedra, L.; Vignolo, G.; Hebert, E.M. Biodiversity and technological-functional potential of lactic acid bacteria isolated from spontaneously fermented quinoa sourdoughs. J. Appl. Microbiol. 2016, 120, 1289–1301. [Google Scholar] [CrossRef] [Green Version]
- De Vuyst, L.; Van Kerrebroeck, S.; Leroy, F. Chapter Two - Microbial Ecology and Process Technology of Sourdough Fermentation. Adv. Appl. Microbiol. 2017, 100, 49–160. [Google Scholar] [CrossRef]
- Zheng, J.; Wittouck, S.; Salvetti, E.; Franz, C.M.A.P.; Harris, H.M.B.; Mattarelli, P.; O’toole, P.W.; Pot, B.; Vandamme, P.; Walter, J.; et al. A taxonomic note on the genus Lactobacillus: Description of 23 novel genera, emended description of the genus Lactobacillus beijerinck 1901, and union of Lactobacillaceae and Leuconostocaceae. Int. J. Syst. Evol. Microbiol. 2020, 70, 2782–2858. [Google Scholar] [CrossRef]
- Michel, E.; Monfort, C.; Deffrasnes, M.; Guezenec, S.; Lhomme, E.; Barret, M.; Sicard, D.; Dousset, X.; Onno, B. Characterization of relative abundance of lactic acid bacteria species in French organic sourdough by cultural, qPCR and MiSeq high-throughput sequencing methods. Int. J. Food Microbiol. 2016, 239, 35–43. [Google Scholar] [CrossRef] [Green Version]
- Coda, R.; Di Cagno, R.; Gobbetti, M.; Rizzello, C.G. Sourdough lactic acid bacteria: Exploration of non-wheat cereal-based fermentation. Food Microbiol. 2014, 37, 51–58. [Google Scholar] [CrossRef]
- Chavan, R.; Chavan, R. Sourdough Technology—A Traditional Way for Wholesome Foods: A Review. Compr. Rev. Food Sci. Food Saf. 2011, 10, 169–182. [Google Scholar] [CrossRef]
- Gobbetti, M.; Minervini, F.; Pontonio, E.; Di Cagno, R.; De Angelis, M. Drivers for the establishment and composition of the sourdough lactic acid bacteria biota. Int. J. Food Microbiol. 2016, 239, 3–18. [Google Scholar] [CrossRef] [PubMed]
- Minervini, F.; De Angelis, M.; Di Cagno, R.; Gobbetti, M. Ecological parameters influencing microbial diversity and stability of traditional sourdough. Int. J. Food Microbiol. 2014, 171, 136–146. [Google Scholar] [CrossRef]
- De Vuyst, L.; Van Kerrebroeck, S.; Harth, H.; Huys, G.; Daniel, H.-M.; Weckx, S. Microbial ecology of sourdough fermentations: Diverse or uniform? Food Microbiol. 2014, 37, 11–29. [Google Scholar] [CrossRef]
- Scheirlinck, I.; Van der Meulen, R.; De Vuyst, L.; Vandamme, P.; Huys, G. Molecular source tracking of predominant lactic acid bacteria in traditional Belgian sourdoughs and their production environments. J. Appl. Microbiol. 2009, 106, 1081–1092. [Google Scholar] [CrossRef] [PubMed]
- Minervini, F.; Lattanzi, A.; De Angelis, M.; Celano, G.; Gobbetti, M. House microbiotas as sources of lactic acid bacteria and yeasts in traditional Italian sourdoughs. Food Microbiol. 2015, 52, 66–76. [Google Scholar] [CrossRef]
- Huys, G.; Daniel, H.-M.; De Vuyst, L. Taxonomy and Biodiversity of Sourdough Yeasts and Lactic Acid Bacteria. In Handbook on Sourdough Biotechnology; Gobbetti, M., Gänzle, M., Eds.; Springer: Boston, MA, USA, 2013; pp. 105–154. ISBN 978-1-4614-5425-0. [Google Scholar]
- Puratos Sourdough Library—A Unique Collection of Natural Ferments Providing a Range of Flavours. Available online: https://www.puratos.com/commitments/next-generation/product-heritage/sourdough-library (accessed on 30 May 2018).
- Spicher, G. Die Mikroflora des Sauerteiges. I. Mitteilung: Untersuchungen über die Art der in Sauerteigen anzutreffenden stäbchenförmigen Milchsäurebakterien (Genus Lactobacillus Beijerinck). Zeitblatt fur Bakteriol. II Abt 1959, 113, 80–106. [Google Scholar]
- Barber, S.; Torner, M.J.; Martínez-Anaya, M.A.; de Barber, C.B. Microflora of the sour dough of wheat flour bread IX. Biochemical characteristics and baking performance of wheat doughs elaborated with mixtures of pure microorganisms. Z. Lebensm. Unters. Forsch. 1989, 189, 6–11. [Google Scholar] [CrossRef]
- Van Kerrebroeck, S.; Maes, D.; De Vuyst, L. Sourdoughs as a function of their species diversity and process conditions, a meta-analysis. Trends Food Sci. Technol. 2017, 68, 152–159. [Google Scholar] [CrossRef]
- Ruiz Rodríguez, L.; Vera Pingitore, E.; Rollan, G.; Martos, G.; Saavedra, L.; Fontana, C.; Hebert, E.M.; Vignolo, G. Biodiversity and technological potential of lactic acid bacteria isolated from spontaneously fermented amaranth sourdough. Lett. Appl. Microbiol. 2016, 63, 147–154. [Google Scholar] [CrossRef]
- Harth, H.; Van Kerrebroeck, S.; De Vuyst, L. Impact of process conditions on the microbial community dynamics and metabolite production kinetics of teff sourdough fermentations under bakery and laboratory conditions. Food Sci. Nutr. 2018, 6, 1438–1455. [Google Scholar] [CrossRef]
- Sekwati-Monang, B.; Gänzle, M.G. Microbiological and chemical characterisation of ting, a sorghum-based sourdough product from Botswana. Int. J. Food Microbiol. 2011, 150, 115–121. [Google Scholar] [CrossRef]
- Obiri-Danso, K. Microbiological studies on corn dough fermentation. Ceral Chem. 1994, 71, 186–188. [Google Scholar]
- Gashe, B.A. Involvement of Lactic Acid Bacteria in the Fermentation of TEF (Eragrosfis tef), an Ethiopian Fermented Food. J. Food Sci. 1985, 50, 800–801. [Google Scholar] [CrossRef]
- Sterr, Y.; Weiss, A.; Schmidt, H. Evaluation of lactic acid bacteria for sourdough fermentation of amaranth. Int. J. Food Microbiol. 2009, 136, 75–82. [Google Scholar] [CrossRef]
- Meroth, C.B.; Hammes, W.P.; Hertel, C. Characterisation of the Microbiota of Rice Sourdoughs and Description of Lactobacillus spicheri sp. nov. Syst. Appl. Microbiol. 2004, 27, 151–159. [Google Scholar] [CrossRef]
- Dingeo, C.; Difonzo, G.; Paradiso, V.M.; Rizzello, C.G.; Pontonio, E. Teff type-i sourdough to produce gluten-free muffin. Microorganisms 2020, 8, 1149. [Google Scholar] [CrossRef] [PubMed]
- Moroni, A.V.; Arendt, E.K.; Morrissey, J.P.; Bello, F.D. Development of buckwheat and teff sourdoughs with the use of commercial starters. Int. J. Food Microbiol. 2010, 142, 142–148. [Google Scholar] [CrossRef] [PubMed]
- Moroni, A.V.; Arendt, E.K.; Bello, F.D. Biodiversity of lactic acid bacteria and yeasts in spontaneously-fermented buckwheat and teff sourdoughs. Food Microbiol. 2011, 28, 497–502. [Google Scholar] [CrossRef] [PubMed]
- Boraam, F.; Faid, M.; Larpent, J.; Breton, A. Lactic acid bacteria and yeast associated with traditional Moroccan sour-dough bread fermentation. Sci. Aliments 1993, 13, 501–509. [Google Scholar]
- Edema, M.; Sanni, A.I. Micro-population of fermenting maize meal for sour maize bread production in Nigeria. Niger. J. Microbiol. 2006, 20, 937–946. [Google Scholar]
- Sanni, A.I.; Onilude, A.A.; Fatungase, M.O. Production of sour maize bread using starter-cultures. World J. Microbiol. Biotechnol. 1997, 14, 101–106. [Google Scholar] [CrossRef]
- Mohammed, S.I.; Steenson, L.R.; Kirleis, A.W. Isolation and characterization of microorganisms associated with the traditional sorghum fermentation for production of Sudanese Kisra. Appl. Environ. Microbiol. 1991, 57, 2529–2533. [Google Scholar] [CrossRef] [Green Version]
- Hamad, S.H.; Böcker, G.; Vogel, R.F.; Hammes, W.P. Microbiological and chemical analysis of fermented sorghum dough for Kisra production. Appl. Microbiol. Biotechnol. 1992, 37, 728–731. [Google Scholar] [CrossRef]
- Vogelmann, S.A.; Hertel, C. Impact of ecological factors on the stability of microbial associations in sourdough fermentation. Food Microbiol. 2011, 28, 583–589. [Google Scholar] [CrossRef]
- Parte, A.C.; Carbasse, J.S.; Meier-Kolthoff, J.P.; Reimer, L.C.; Göker, M. List of prokaryotic names with standing in nomenclature (LPSN) moves to the DSMZ. Int. J. Syst. Evol. Microbiol. 2020, 70, 5607–5612. [Google Scholar] [CrossRef]
- Real Decreto 308/2019, de 26 de abril, por el que se aprueba la norma de calidad para el pan. Available online: https://www.boe.es/eli/es/rd/2019/04/26/308 (accessed on 28 June 2021).
- Wolter, A.; Hager, A.-S.; Zannini, E.; Czerny, M.; Arendt, E.K. Influence of dextran-producing Weissella cibaria on baking properties and sensory profile of gluten-free and wheat breads. Int. J. Food Microbiol. 2014, 172, 83–91. [Google Scholar] [CrossRef]
- Aguilar, N.; Albanell, E.; Miñarro, B.; Capellas, M. Chestnut flour sourdough for gluten-free bread making. Eur. Food Res. Technol. 2016, 242, 1795–1802. [Google Scholar] [CrossRef]
- Różyło, R.; Rudy, S.; Krzykowski, A.; Dziki, D.; Siastała, M.; Polak, R. Gluten-Free Bread Prepared with Fresh and Freeze-Dried Rice Sourdough-Texture and Sensory Evaluation. J. Texture Stud. 2016, 47, 443–453. [Google Scholar] [CrossRef]
- Różyło, R.; Rudy, S.; Krzykowski, A.; Dziki, D.; Gawlik-Dziki, U.; Różyło, K.; Skonecki, S. Effect of adding fresh and freeze-dried buckwheat sourdough on gluten-free bread quality. Int. J. Food Sci. Technol. 2015, 50, 313–322. [Google Scholar] [CrossRef]
- Różyło, R.; Rudy, S.; Krzykowski, A.; Dziki, D. Novel Application of Freeze-Dried Amaranth Sourdough in Gluten-Free Bread Production. J. Food Process Eng. 2015, 38, 135–143. [Google Scholar] [CrossRef]
- Li, Z.; Li, H.; Bian, K. Microbiological characterization of traditional dough fermentation starter (Jiaozi) for steamed bread making by culture-dependent and culture-independent methods. Int. J. Food Microbiol. 2016, 234, 9–14. [Google Scholar] [CrossRef] [PubMed]
- De Vuyst, L.; Harth, H.; Van Kerrebroeck, S.; Leroy, F. Yeast diversity of sourdoughs and associated metabolic properties and functionalities. Int. J. Food Microbiol. 2016, 239, 26–34. [Google Scholar] [CrossRef]
- Arendt, E.K.; Moroni, A.; Zannini, E. Medical nutrition therapy: Use of sourdough lactic acid bacteria as a cell factory for delivering functional biomolecules and food ingredients in gluten free bread. Microb. Cell Fact. 2011, 10, S15. [Google Scholar] [CrossRef] [Green Version]
- Nácher-Vázquez, M. Dextranos de Bacterias Lácticas Aisladas de Productos Cárnicos: Caracterización y Aplicaciones. Ph.D. Thesis, Universitat de València, Valencia, Spain, 2015. [Google Scholar]
- Zannini, E.; Waters, D.M.; Coffey, A.; Arendt, E.K. Production, properties, and industrial food application of lactic acid bacteria-derived exopolysaccharides. Appl. Microbiol. Biotechnol. 2016, 100, 1121–1135. [Google Scholar] [CrossRef] [PubMed]
- Laiño, J.; Villena, J.; Kanmani, P.; Kitazawa, H. Immunoregulatory Effects Triggered by Lactic Acid Bacteria Exopolysaccharides: New Insights into Molecular Interactions with Host Cells. Microorganisms 2016, 4, 27. [Google Scholar] [CrossRef] [Green Version]
- Wang, K.; Li, W.; Rui, X.; Chen, X.; Jiang, M.; Dong, M. Characterization of a novel exopolysaccharide with antitumor activity from Lactobacillus plantarum 70810. Int. J. Biol. Macromol. 2014, 63, 133–139. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Ganzle, M.G.; Schwab, C. Exopolysaccharide synthesized by Lactobacillus reuteri decreases the ability of enterotoxigenic Escherichia coli to bind to porcine erythrocytes. Appl. Environ. Microbiol. 2010, 76, 4863–4866. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Galle, S.; Arendt, E.K. Exopolysaccharides from sourdough lactic acid bacteria. Crit. Rev. Food Sci. Nutr. 2014, 54, 891–901. [Google Scholar] [CrossRef]
- Lynch, K.M.; Coffey, A.; Arendt, E.K. Exopolysaccharide producing lactic acid bacteria: Their techno-functional role and potential application in gluten-free bread products. Food Res. Int. 2017. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, C.; Kobayashi, M.; Kimoto-Nira, H. Novel exopolysaccharides produced by Lactococcus lactis subsp. lactis, and the diversity of epsE genes in the exopolysaccharide biosynthesis gene clusters. Biosci. Biotechnol. Biochem. 2013, 77, 2013–2018. [Google Scholar] [CrossRef]
- Ismail, B.; Nampoothiri, K.M. Production, purification and structural characterization of an exopolysaccharide produced by a probiotic Lactobacillus plantarum MTCC 9510. Arch. Microbiol. 2010, 192, 1049–1057. [Google Scholar] [CrossRef]
- Verbeke, K. Nonceliac Gluten Sensitivity: What Is the Culprit? Gastroenterology 2018, 154, 471–473. [Google Scholar] [CrossRef] [Green Version]
- Galle, S.; Schwab, C.; Dal Bello, F.; Coffey, A.; Gänzle, M.G.; Arendt, E.K. Influence of in-situ synthesized exopolysaccharides on the quality of gluten-free sorghum sourdough bread. Int. J. Food Microbiol. 2012, 155, 105–112. [Google Scholar] [CrossRef]
- Nami, Y.; Gharekhani, M.; Aalami, M.; Hejazi, M.A. Lactobacillus-fermented sourdoughs improve the quality of gluten-free bread made from pearl millet flour. J. Food Sci. Technol. 2019, 56, 4057–4067. [Google Scholar] [CrossRef]
- Dertli, E.; Mercan, E.; Arıcı, M.; Yılmaz, M.T.; Sağdıç, O. Characterisation of lactic acid bacteria from Turkish sourdough and determination of their exopolysaccharide (EPS) production characteristics. LWT Food Sci. Technol. 2016, 71, 116–124. [Google Scholar] [CrossRef]
- Franco, W.; Pérez-Díaz, I.M.; Connelly, L.; Diaz, J.T. Isolation of exopolysaccharide-producing yeast and lactic acid bacteria from quinoa (Chenopodium quinoa) sourdough fermentation. Foods 2020, 9, 337. [Google Scholar] [CrossRef] [Green Version]
- Rühmkorf, C.; Jungkunz, S.; Wagner, M.; Vogel, R.F. Optimization of homoexopolysaccharide formation by lactobacilli in gluten-free sourdoughs. Food Microbiol. 2012, 32, 286–294. [Google Scholar] [CrossRef] [PubMed]
- Montemurro, M.; Pontonio, E.; Rizzello, C.G. Design of a “clean-label” gluten-free bread to meet consumers demand. Foods 2021, 10, 462. [Google Scholar] [CrossRef] [PubMed]
- Coda, R.; Nionelli, L.; Rizzello, C.G.; De Angelis, M.; Tossut, P.; Gobbetti, M. Spelt and emmer flours: Characterization of the lactic acid bacteria microbiota and selection of mixed starters for bread making. J. Appl. Microbiol. 2010, 108, 925–935. [Google Scholar] [CrossRef]
- Nionelli, L.; Curri, N.; Curiel, J.A.; Di Cagno, R.; Pontonio, E.; Cavoski, I.; Gobbetti, M.; Rizzello, C.G. Exploitation of Albanian wheat cultivars: Characterization of the flours and lactic acid bacteria microbiota, and selection of starters for sourdough fermentation. Food Microbiol. 2014, 44, 96–107. [Google Scholar] [CrossRef] [PubMed]
- Galle, S.; Schwab, C.; Arendt, E.K.; Gänzle, M.G. Structural and rheological characterisation of heteropolysaccharides produced by lactic acid bacteria in wheat and sorghum sourdough. Food Microbiol. 2011, 28, 547–553. [Google Scholar] [CrossRef] [PubMed]
- Mamhoud, A.; Nionelli, L.; Bouzaine, T.; Hamdi, M.; Gobbetti, M.; Rizzello, C.G. Selection of lactic acid bacteria isolated from Tunisian cereals and exploitation of the use as starters for sourdough fermentation. Int. J. Food Microbiol. 2016, 225, 9–19. [Google Scholar] [CrossRef]

| Sourdough Property/Function | References |
|---|---|
| Sensory improvements | [17] |
| Nutritional improvements | [18,19] |
| Functional improvements | [20] |
| Shelf-life extension | [21] |
| Food Product | Cereal(s) or Pseudo-Cereal(s) Used in the Product | Main Flour(s) | Hydrocolloids | Technological Outcome | Reference(s) |
|---|---|---|---|---|---|
| GF bread | Brown rice | Brown rice | Xanthan gum, guar gum, xanthan-locust bean gum, MC 1, CMC 2, HPMC 3 | ↑4 Porosity, ↑ cohesiveness and elasticity | [25,26] |
| GF bread | Buckwheat | Buckwheat flour | 0.14% xanthan gum | ↑ Bread volume ↓5 Crumb hardness/firmness | [12,27] |
| GF bread | Buckwheat | Buckwheat flour | Guar gum, HPMC, tragacanth gum | ↑ Crumb alveoli resistance, ↑ elasticity | [25] |
| GF 7 bread | Teff, buckwheat, rice maize | Teff, buckwheat, rice, or maize flours | 1.5% HPMC | dov 6 | [12] |
| Rice | Rice flour and potato starch | Fructans (such as inulin) | ↑ Bread volume ↓ Crumb hardness | [28] | |
| Maize | Maize starch, potato starch | Inulin (<10 polymerization degrees) | ↑ Bread volume ↓ Crumb hardness | ||
| Maize | Maize starch, zein | HPMC, high β-glucan oat bran | Positive rheology, good crumb structure | ||
| Maize | Maize starch, potato starch | 5% Inulin | ↑ Bread volume (4%) ↓ Crumb hardness | [29] | |
| Maize | Maize starch, potato starch | 8% Inulin | ↑ Bread volume (9%) ↓ Crumb hardness Wrinkling of the crust | ||
| Rice | Rice flour and potato starch | 4% to 12% ITFs 8 (Raftilose® Synergy1) | ↑ Specific volume, darker crust, appealing crust and crumb | ||
| Rice | Rice flour | Inulin | ↑ Volume, delayed staling, improved crumb, smoother crust | ||
| Rice | Rice flour, potato starch, cassava starch, sour tapioca flour | ITFs (inulin, FOS 9) | Color and porosity improvements Improved texture, taste and flavor | ||
| Maize, rice | Maize flour, rice flour, inactive soy flour | CMC or xanthan gum | dov | [30,31] | |
| Rice | Maize flour, carob flour, resistant starch (RS) | Carob flour, resistant starch (RS) | Low crumb firmness and improved porosity values with 15 g carob flour, 10 g RS, 10 g protein and 140 g water/100 g flour | ||
| Maize | Maize starch, potato starch | Flaxseed mucilage | Improved sensory acceptance | [15] | |
| Amaranth | Maize starch, amaranth flour, pea isolate | Psyllium | Improved final product quality | ||
| Rice, quinoa | Rice flour, quinoa flour | Xanthan gum | dov | [32] | |
| Rice, buckwheat | Rice flour, buckwheat flour | ||||
| GF layer cakes | Rice | Rice flour | Inulin, oat fibers, guar gum | Same volume as control ↑ Crumb firmness ↓ Elasticity | [28] |
| GF cheese bread | Maize | Pre-cooked cornflour, cassava starch | 9% FOS | ↓ Hydration; ↑ solubility of starch–FOS mixtures | [29] |
| GF bread | Maize | Maize flour | 1.77% HPMC | ↑ Bread volume ↓ Crumb hardness/firmness | [12] |
| “Empanadas” and piecrusts | Maize | Maize starch | Guar gum, HPMC, xanthan gum | ↑ Elasticity | [25] |
| GF bread | Maize | Maize flour, maize starch | Xanthan gum | ↑ Specific volume ↓ Crumb hardness | |
| GF bread | Maize | Maize starch, potato starch | Pectin, whey protein | dov | [32] |
| GF bread | Maize | Maize flour, maize starch | Guar gum, pectin | ↓ Firmness, ↓ crumb hardening | [25] |
| GF bread | Rice | Rice flour | 2.2% HPMC | dov | [12] |
| GF bread | Rice | Rice flour | HPMC | ↑ Elasticity and viscosity | [25] |
| GF bread | Rice | Rice flour | HPMC | dov | |
| GF bread | Rice | Rice flour | Xanthan gum, carob gum, guar gum, HPMC | ↑ Viscoelasticity | |
| GF bread | Rice | Rice flour | HPMC | ↑ Specific volume | |
| GF bread | Rice | Rice flour | HPMC, xanthan gum | ↑ Specific volume | |
| GF bread | Rice | Rice flour | HPMC | ↓ Crumb firmness | |
| GF bread | Rice | Rice flour | HPMC | ↑ Moisture content Enhanced sensory properties | |
| GF bread | Rice | Rice flour | HPMC, guar gum, CMC | ↑ Specific volume | [25] |
| GF flat bread | Rice | Rice flour | 15 g/kg xanthan gum 10 g/kg CMC 10 g/kg xanthan gum | ↑ Crumb alveoli size ↑ Crumb porosity ↑ Dough yield | [15] |
| GF bread | Rice | Rice flour | HPMC | dov | [32] |
| GF bread | Rice | Rice flour | HPMC, β-glucan | dov | |
| GF bread | Rice | Rice flour | Xanthan gum, guar gum, carob gum | dov | |
| GF cake and muffin products | Rice | Rice flour | Tragacanth gum, xanthan gum | dov | |
| GF bread | Rice | Rice flour, carob flour, resistant starch | Carob gum, DATEM®, whey protein concentrate, α-amylase, transglutaminase, hemicellulase | dov | |
| GF bread | Rice, buckwheat | Rice flour, buckwheat flour | Xanthan gum | dov | |
| GF bread | Rice, maize | Rice flour, maize flour, soy flour | Carrageenan, alginate, xanthan gum, CMC | ↑ Consistency, ↑ starch retrogradation, ↑ amylopectin retrogradation | [25] |
| GF bread | Rice, maize | Rice flour, maize starch | Xanthan gum | dov | |
| CMC, pectin, agarose, xanthan gum | ↑ Elasticity ↑ Dough strength | ||||
| CMC, xanthan gum | ↓ Crumb firmness ↑ Crumb porosity | ||||
| Egyptian balady flat bread | Rice, maize | Rice flour, maize starch, potato starch | Xanthan gum, guar gum | ↓ Loss of moisture ↓ Hardness/firmness | |
| GF bread | Brown rice, maize, buckwheat | Brown rice flour, maize starch, soybean flour, buckwheat flour | Xanthan gum, Konjac gum | ↓ Elasticity, cohesiveness, and resilience | |
| Rice, maize | Rice flour, maize starch, chestnut flour | HPMC, lupine protein, vegetable fiber; guar gum, skimmed milk, cellulose | dov | [32] | |
| Rice, maize | Rice flour and maize starch | HPMC, skimmed milk, egg powder, soy protein, xanthan gum | dov | ||
| HPMC, vegetable fiber (bamboo, oat, pea, potato) | dov | ||||
| GF bread | Rice, maize | Rice, maize, maize starch | Xanthan gum | ↑ Color improvements, ↑ Volume, hydration | [25] |
| GF bread | Rice, maize, quinoa | Rice flour, maize flour, maize starch, quinoa flour | HPMC, amyloglucosidase, α-amylase | ↑ Volume, ↑ firmness | [32] |
| GF bread | Rice, maize, rice | Rice flour, maize flour, rice starch, rice protein | HPMC, carob gum, guar gum, psyllium, beetroot fiber, amylase | dov | |
| GF bread | Sorghum, maize | Decorticated sorghum flour, maize starch | Xanthan gum | dov | [25] |
| GF bread | Teff | Teff flour | 0.04% xanthan gum 2% HPMC | ↑ Bread volume ↓ Crumb hardness/firmness | [12] |
| Food Product | Cereal(s) or Pseudo-Cereal(s) Used in the Product | Main Flour(s) Used in the Product | Protein Supplementation/Additives | Technological Outcome | Reference(s) |
|---|---|---|---|---|---|
| GF 1 bread | Rice | Rice flour, cassava starch, soy flour | 7.5% soy 7.8% milk powder | ↑ 2 Nutritive value, without sensorial changes | [12] |
| Rice | Rice flour | Bovine plasma protein | dov 3 | [30] | |
| Rice | Rice flour | Bovine serum albumin | dov | ||
| Maize | Maize starch, potato starch | Collagen | dov | ||
| Variable | Variable | Egg, caseinate, whey protein, milk protein | dov | [30,41] | |
| Variable | Variable | Egg | Improved structure, stable foaming, and gas retention | [12] | |
| Variable | Variable | Lactose free milk powder | dov, darkening of the crumb | ||
| Precooked rice pasta | Rice | Rice flour, yellow pea flour, chickpea flour, lentil flour | Legume protein | Increased protein and dietary fiber content | [15] |
| GF bread | Rice, buckwheat, quinoa | Rice flour, quinoa flour, buckwheat flour, potato starch | Quinoa protein | ↑ Elasticity and dietary fiber content improved dough structure | |
| GF bread | Rice | Rice flour | Rice bran protein concentrate | ↑ Elasticity, shear strength, volume, gas retention and shelf life | |
| GF bread | - | Soy flour | Soy | dov (↑ loaf volume, ↓ 4 crumb hardness) | [12,42,43] |
| GF bread | - | Soy flour | Soy protein | dov | |
| GF bread | Variable | Soy flour | Soy protein, milk powder | dov | |
| GF bread | Variable | Soy flour | Soy, pea | dov | |
| GF bread | Variable | Starch from different sources | Whey protein | dov | [12,30] |
| GF bread | Maize | Maize flour | Zein | dov | [12,30] |
| Egg yolk muffins | Maize | Maize | Egg yolk granulates, apple pectins, gelatine | dov | [32] |
| GF bread | Maize | Maize starch, carob germ flour | Carob protein, HPMC 5 | dov | [12] |
| GF doughs | Maize | Unmodified maize starch | Zein, HPMC | dov | |
| GF muffins | Maize | Maize starch, kidney bean flour, field pea flour, amaranth flour | Protein isolates | dov | [32,41,44] |
| GF muffins | Rice | Egg, fructose, inulin, sucralose | dov | ||
| Rice | Jambolan fruit pulp, soy Protein isolates, glycerol monostearate, xanthan gum | dov | |||
| Rice | Soya bean protein isolate, pea protein isolate, egg white isolate, casein, xanthan gum | dov | |||
| Rice | Soy protein isolates, glycerol monostearate, xanthan gum, black carrot dietary fiber concentrate | dov | |||
| GF bread | Buckwheat, rice | Buckwheat flour, rice flour, chickpea flour | Green mussel protein hydrolysates | dov | |
| GF bread | Wheat | Wheat starch | 6% whey protein | Darker crust, white crumb, ↑ volume, improved texture | [12] |
| Wheat | Wheat starch | Whey protein | dov |
| Food Product | Cereal(s) or Pseudo-Cereal(s) Used in the Product | Main Flour(s) | Enzymes/Additives | Technological Outcome | Reference(s) |
|---|---|---|---|---|---|
| GF 1 bread | Brown rice, buckwheat, maize, oat sorghum or teff | Brown rice, buckwheat, maize, oat, sorghum or teff flours | 0.1 or 10 U 2 of TGase 3/g of protein | Depending on protein source and enzyme dosage | [12,47] |
| GF bread | Buckwheat, brown rice | Buckwheat flour, brown rice flour | 0.1 to 10 U of TGase/g protein | ↑ 4 Increased batter pseudoplasticity, ↑ water holding capacity, improved crumb texture and structure | [15] |
| GF bread | Buckwheat, sorghum, or maize | Buckwheat, sorghum, or maize flours | 0.01% or 0.1% proteases | Liquid-like batters, poor viscoelastic behavior, ↓ 5 gas-holding capacity | [12] |
| GF bread | Buckwheat, rice | Buckwheat flour, rice flour | Amylase | dov 6 | [30] |
| GF bread | Rice | Rice flour | Cyclodextrinase | dov | [12,30] |
| GF bread | Rice, sorghum, maize | Rice, sorghum, maize flours | GO 7 | dov | [12,30] |
| GF bread | Rice | Jasmine rice flour, pregelatinized tapioca starch | TGase | dov, TGase increased loaf volume and softened bread crumb. | [30,46,48] |
| GF bread | Oat | Oat flour | Tyrosinase, laccase, xylanase | dov, tyrosinase increased firmness of the dough, laccase and xylanase improved specific volume | |
| GF cake and muffin products | Rice | Rice flour, legume flour, chickpea flour, pea flour, lentil flour, bean flour | α-amylase, amyloglucosidase, trypsin, GO | dov | [32] |
| GF bread | Rice | Rice flour | 0.01% GO 2% HPMC 8 | ↑ Final volume, smoother crumb | [12] |
| GF bread | Rice | Rice flour | 1 U TGase/g | Improved crumb texture | [15] |
| GF bread | Rice | Rice flour | 1.35 U of TGase/g rice flour protein 0.67% albumin 0.67% casein | ↑ Final volume, less compact crumb | [12] |
| GF dough and bread | Rice | Rice flour | Aspergillus oryzae protease | ↑ Viscosity, improved gas-holding capacity, volume improvements | |
| GF bread | Rice | Rice flour | Glutathione oxidase | ↑ Elasticity and volume improved gas-holding capacity | [15] |
| GF bread | Rice | Rice flour | Microbial TGase HPMC | dov | [12] |
| GF bread | Rice | Rice flour | Proteases | Depending on protease amount added |
| Food Product | Cereal(s) or Pseudo-Cereal(s) Used in the Product | Main Flour(s) | Emulsifiers | Technological Outcome | Reference |
|---|---|---|---|---|---|
| GF 1 dough | Buckwheat | Buckwheat flour | DATEM® | dov 2 | [12] |
| GF cheese bread | - | Cassava starch | DATEM® | dov | [50] |
| GF bread formulas | Rice | Rice flour | 0.5% DATEM® 0.5% (xanthan gum/guar) | Improved final product (with highest scores for texture acceptability) | [12] |
| GF bread | Rice | Rice flour, tigernut flour | DATEM®, xanthan gum, guar gum | dov | [32] |
| GF cake and muffin products | Rice, maize | Rice flour, maize flour | Lecithin | dov |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ramos, L.; Alonso-Hernando, A.; Martínez-Castro, M.; Morán-Pérez, J.A.; Cabrero-Lobato, P.; Pascual-Maté, A.; Téllez-Jiménez, E.; Mujico, J.R. Sourdough Biotechnology Applied to Gluten-Free Baked Goods: Rescuing the Tradition. Foods 2021, 10, 1498. https://doi.org/10.3390/foods10071498
Ramos L, Alonso-Hernando A, Martínez-Castro M, Morán-Pérez JA, Cabrero-Lobato P, Pascual-Maté A, Téllez-Jiménez E, Mujico JR. Sourdough Biotechnology Applied to Gluten-Free Baked Goods: Rescuing the Tradition. Foods. 2021; 10(7):1498. https://doi.org/10.3390/foods10071498
Chicago/Turabian StyleRamos, Laura, Alicia Alonso-Hernando, Miriam Martínez-Castro, Jose Alejandro Morán-Pérez, Patricia Cabrero-Lobato, Ana Pascual-Maté, Eduardo Téllez-Jiménez, and Jorge R. Mujico. 2021. "Sourdough Biotechnology Applied to Gluten-Free Baked Goods: Rescuing the Tradition" Foods 10, no. 7: 1498. https://doi.org/10.3390/foods10071498
APA StyleRamos, L., Alonso-Hernando, A., Martínez-Castro, M., Morán-Pérez, J. A., Cabrero-Lobato, P., Pascual-Maté, A., Téllez-Jiménez, E., & Mujico, J. R. (2021). Sourdough Biotechnology Applied to Gluten-Free Baked Goods: Rescuing the Tradition. Foods, 10(7), 1498. https://doi.org/10.3390/foods10071498






