Antiviral, Antibacterial, Antifungal, and Antiparasitic Properties of Propolis: A Review
Abstract
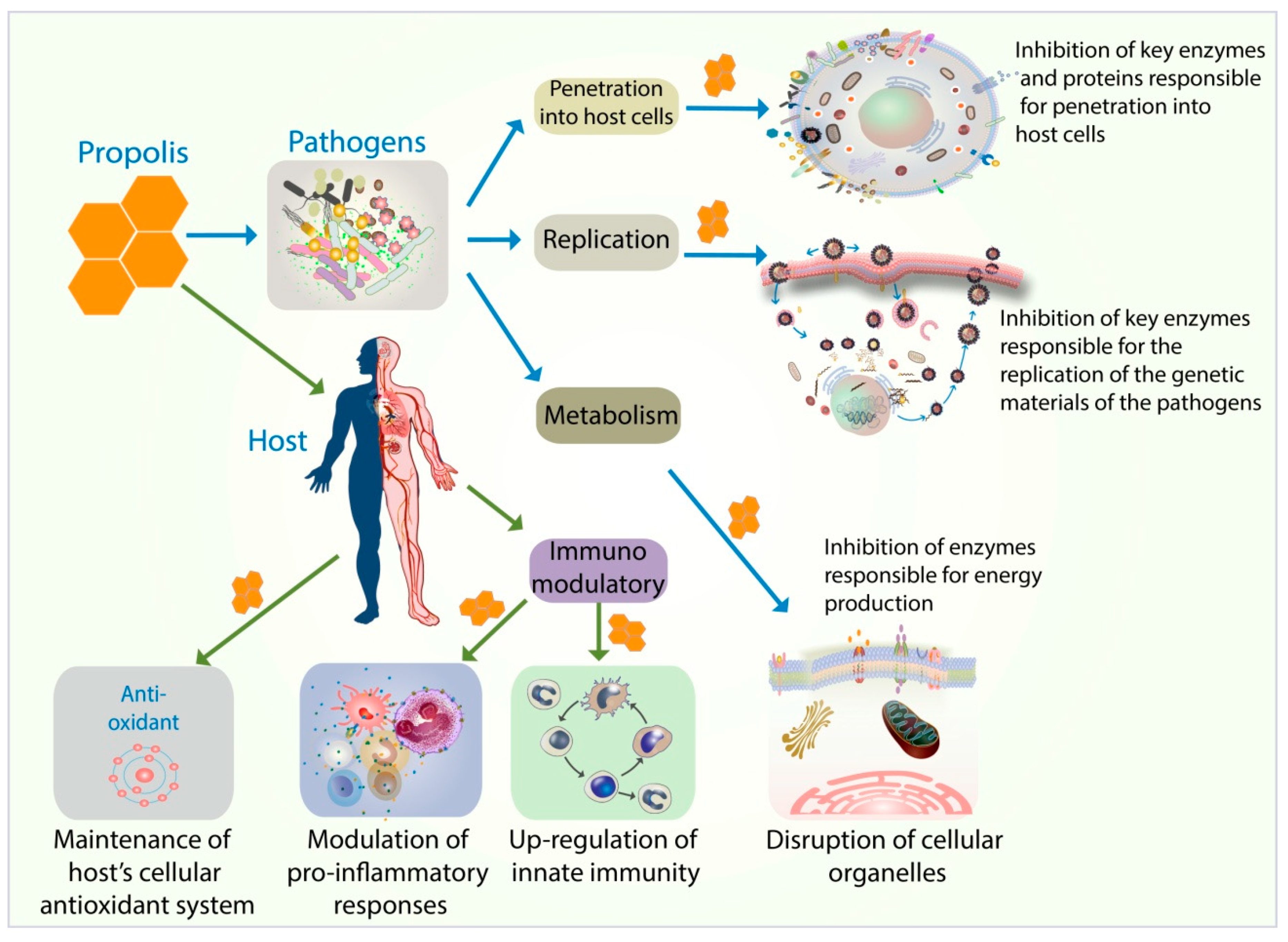
1. Introduction
2. Bioactive Compounds in Propolis
3. Antiviral Properties of Propolis
4. Antibacterial Properties of Propolis
Mechanisms of Action of Antibacterial Properties of Propolis
5. Antifungal Properties of Propolis
Antifungal Mechanisms of Action of Propolis
6. Anti-Parasitic Properties of Propolis
Antiparasitic Mechanisms of Action of Propolis
7. Human Clinical Trials
8. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Tibenderana, J.K.; D’Alessandro, U.; Erhart, A.; Rosenthal, P.J.; Achan, J.; Yeka, A.; Baliraine, F.N.; Talisuna, A.O. Quinine, an old anti-malarial drug in a modern world: Role in the treatment of malaria. Malar. J. 2011, 10, 144. [Google Scholar]
- Luz Yolanda Toro Suarez. Nobel Prize Physiol. Med. 2015, 2015, 1–17. Available online: https://www.nobelprize.org/prizes/medicine/2015/tu/facts/ (accessed on 30 January 2021).
- Fleming, A. Penicillin. Nobel Lect. 1945. Available online: https://www.nobelprize.org/uploads/2018/06/fleming-lecture.pdf (accessed on 30 January 2021).
- Niu, G.; Johnson, R.M.; Berenbaum, M.R. Toxicity of mycotoxins to honeybees and its amelioration by propolis. Apidologie 2011, 42, 79–87. [Google Scholar] [CrossRef]
- Mao, W.; Schuler, M.A.; Berenbaum, M.R. Honey constituents up-regulate detoxification and immunity genes in the western honey bee Apis mellifera. Proc. Natl. Acad. Sci. USA 2013, 110, 8842–8846. [Google Scholar] [CrossRef]
- Yemor, T.; Phiancharoen, M.; Eric Benbow, M.; Suwannapong, G. Effects of stingless bee propolis on Nosema ceranae infected Asian honey bees, Apis cerana. J. Apic. Res. 2015, 54, 468–473. [Google Scholar] [CrossRef]
- Borba, R.S.; Klyczek, K.K.; Mogen, K.L.; Spivak, M. Seasonal benefits of a natural propolis envelope to honey bee immunity and colony health. J. Exp. Biol. 2015, 218, 3689–3699. [Google Scholar] [CrossRef] [PubMed]
- Saelao, P.; Borba, R.S.; Ricigliano, V.; Spivak, M.; Simone-Finstrom, M. Honeybee microbiome is stabilized in the presence of propolis. Biol. Lett. 2020, 16, 20200003. [Google Scholar] [CrossRef]
- Dalenberg, H.; Maes, P.; Mott, B.; Anderson, K.E.; Spivak, M. Propolis envelope promotes beneficial bacteria in the honey bee (Apis mellifera) mouthpart microbiome. Insects 2020, 11, 453. [Google Scholar] [CrossRef] [PubMed]
- Wang, K.; Ping, S.; Huang, S.; Hu, L.; Xuan, H.; Zhang, C.; Hu, F. Molecular mechanisms underlying the in vitro anti-inflammatory effects of a flavonoid-rich ethanol extract from chinese propolis (poplar type). Evid. Based Complement. Altern. Med. 2013, 2013, 127672. [Google Scholar]
- Ibrahim, N.; Zakaria, A.J.; Ismail, Z.; Mohd, K.S. Antibacterial and phenolic content of propolis produced by two Malaysian stingless bees, Heterotrigona itama and Geniotrigona thoracica. Int. J. Pharmacogn. Phytochem. Res. 2016, 8, 156–161. [Google Scholar]
- Corrêa, F.R.S.; Schanuel, F.S.; Moura-Nunes, N.; Monte-Alto-Costa, A.; Daleprane, J.B. Brazilian red propolis improves cutaneous wound healing suppressing inflammation-associated transcription factor NFκB. Biomed. Pharmacother. 2017, 86, 162–171. [Google Scholar] [CrossRef]
- Zhang, W.; Cai, Y.; Chen, X.; Ji, T.; Sun, L. Optimized extraction based on the terpenoids of Heterotrigona itama propolis and their antioxidative and anti-inflammatory activities. J. Food Biochem. 2020, 44, e13296. [Google Scholar] [CrossRef] [PubMed]
- Amalia, E.; Diantini, A.; Subarnas, A. Water-soluble propolis and bee pollen of Trigona spp. From South Sulawesi Indonesia induce apoptosis in the human breast cancer MCF-7 cell line. Oncol. Lett. 2020, 20. [Google Scholar] [CrossRef] [PubMed]
- Refaat, H.; Mady, F.M.; Sarhan, H.A.; Rateb, H.S.; Alaaeldin, E. Optimization and evaluation of propolis liposomes as a promising therapeutic approach for COVID-19. Int. J. Pharm. 2021, 592, 120028. [Google Scholar] [CrossRef]
- Zulhendri, F.; Felitti, R.; Fearnley, J.; Ravalia, M. The use of propolis in dentistry, oral health, and medicine: A review. J. Oral Biosci. 2021. [Google Scholar] [CrossRef]
- Kuropatnicki, A.K.; Szliszka, E.; Krol, W. Historical aspects of propolis research in modern times. Evid. Based Complement. Altern. Med. 2013, 2013, 964149. [Google Scholar] [CrossRef] [PubMed]
- Ghisalberti, E. Propolis: A review. Bee World 1979, 60, 59–84. [Google Scholar] [CrossRef]
- Park, Y.K.; Alencar, S.M.; Aguiar, C.L. Botanical origin and chemical composition of Brazilian propolis. J. Agric. Food Chem. 2002, 50, 2502–2506. [Google Scholar] [CrossRef]
- Sahinler, N.; Kaftanoglu, O. Natural product propolis: Chemical composition. Nat. Prod. Res. 2005, 19, 183–188. [Google Scholar] [CrossRef]
- Huang, S.; Zhang, C.P.; Wang, K.; Li, G.Q.; Hu, F.L. Recent advances in the chemical composition of propolis. Molecules 2014, 19, 19610–19632. [Google Scholar] [CrossRef] [PubMed]
- Salatino, A.; Salatino, M.L.F. Scientific note: Often quoted, but not factual data about propolis composition. Apidologie 2021, 52, 312–314. [Google Scholar] [CrossRef]
- Watanabe, M.A.E.; Amarante, M.K.; Conti, B.; Sforcin, J.M. Cytotoxic constituents of propolis inducing anticancer effects: A review. J. Pharm. Pharmacol. 2011, 63, 1378–1386. [Google Scholar] [CrossRef] [PubMed]
- Braakhuis, A. Evidence on the health benefits of supplemental propolis. Nutrients 2019, 11, 2705. [Google Scholar] [CrossRef]
- Zulhendri, F.; Ravalia, M.; Kripal, K.; Chandrasekaran, K.; Fearnley, J.; Perera, C.O. Propolis in metabolic syndrome and its associated chronic diseases: A narrative review. Antioxidants 2021, 10, 348. [Google Scholar] [CrossRef] [PubMed]
- Lattanzio, V.; Kroon, P.A.; Quideau, S.; Treutter, D. Plant phenolics—Secondary metabolites with diverse functions. Recent Adv. Polyphen. Res. 2009, 1, 1–35. [Google Scholar]
- Cheynier, V.; Comte, G.; Davies, K.M.; Lattanzio, V.; Martens, S. Plant phenolics: Recent advances on their biosynthesis, genetics, andecophysiology. Plant Physiol. Biochem. 2013, 72, 1–20. [Google Scholar] [CrossRef]
- Bhattacharya, A.; Sood, P.; Citovsky, V. The roles of plant phenolics in defence and communication during Agrobacterium and Rhizobium infection. Mol. Plant Pathol. 2010, 11, 705–719. [Google Scholar] [CrossRef]
- Quideau, S.; Deffieux, D.; Douat-Casassus, C.; Pouységu, L. Plant polyphenols: Chemical properties, biological activities, and synthesis. Angew. Chemie Int. Ed. 2011, 50, 586–621. [Google Scholar] [CrossRef]
- Lattanzio, V. Phenolic Compounds: Introduction. In Natural Products; Ramawat, K.G., Merillon, J.M., Eds.; Springer: Berlin/Heidelberg, Germany, 2013; pp. 1544–1573. [Google Scholar]
- Singla, R.K.; Dubey, A.K.; Garg, A.; Sharma, R.K.; Fiorino, M.; Ameen, S.M.; Haddad, M.A.; Al-Hiary, M. Natural polyphenols: Chemical classification, definition of classes, subcategories, and structures. J. AOAC Int. 2019, 102, 1397–1400. [Google Scholar] [CrossRef]
- Kougan, G.B.; Tabopda, T.; Kuete, V.; Verpoorte, R. Simple phenols, phenolic acids, and related esters from the medicinal plants of Africa. In Medicinal Plant Research in Africa Pharmacology and Chemistry; Kuete, V., Ed.; Elsevier: Amsterdam, The Netherlands, 2013; pp. 225–249. [Google Scholar]
- Heleno, S.A.; Martins, A.; Queiroz, M.J.R.P.; Ferreira, I.C.F.R. Bioactivity of phenolic acids: Metabolites versus parent compounds: A review. Food Chem. 2015, 173, 501–513. [Google Scholar] [CrossRef] [PubMed]
- Kiokias, S.; Proestos, C.; Oreopoulou, V. Phenolic acids of plant origin-a review on their antioxidant activity in vitro (O/W emulsion systems) along with their in vivo health biochemical properties. Foods 2020, 9, 534. [Google Scholar] [CrossRef] [PubMed]
- Umezawa, T. Diversity in lignan biosynthesis. Phytochem. Rev. 2003, 2, 371–390. [Google Scholar] [CrossRef]
- Bankova, V.; Popova, M.; Bogdanov, S.; Sabatini, A.-G. Chemical composition of European propolis: Expected and unexpected results. Z. Naturforsch. C. 2002, 57, 530–533. [Google Scholar] [CrossRef] [PubMed]
- Kasiotis, K.M.; Anastasiadou, P.; Papadopoulos, A.; Machera, K. Revisiting Greek propolis: Chromatographic analysis and antioxidant activity study. PLoS ONE 2017, 12. [Google Scholar] [CrossRef] [PubMed]
- Shi, H.; Yang, H.; Zhang, X.; Yu, L. Identification and quantification of phytochemical composition and anti-inflammatory and radical scavenging properties of methanolic extracts of Chinese propolis. J. Agric. Food Chem. 2012, 60, 12403–12410. [Google Scholar] [CrossRef]
- Chen, C.; Wu, C.; Shy, H.; Lin, J. Cytotoxic prenylflavanones from Taiwanese propolis. J. Nat. Prod. 2003, 66, 503–506. [Google Scholar] [CrossRef]
- Chen, C.-N.; Weng, M.-S.; Wu, C.-L.; Lin, J.-K. Comparison of radical scavenging activity, cytotoxic effects and apoptosis induction in human melanoma cells by taiwanese propolis from different sources. Evid. Based Complement. Altern. Med. 2004, 1, 175–185. [Google Scholar] [CrossRef]
- Chen, Y.W.; Wu, S.W.; Ho, K.K.; Lin, S.B.; Huang, C.Y.; Chen, C.N. Characterisation of Taiwanese propolis collected from different locations and seasons. J. Sci. Food Agric. 2008, 88, 412–419. [Google Scholar] [CrossRef]
- Trusheva, B.; Popova, M.; Koendhori, E.B.; Tsvetkova, I.; Naydenski, C.; Bankova, V. Indonesian propolis: Chemical composition, biological activity and botanical origin. Nat. Prod. Res. 2011, 25, 606–613. [Google Scholar] [CrossRef]
- Kasote, D.M.; Pawar, M.V.; Bhatia, R.S.; Nandre, V.S.; Gundu, S.S.; Jagtap, S.D.; Kulkarni, M.V. HPLC, NMR based chemical profiling and biological characterisation of Indian propolis. Fitoterapia 2017, 122, 52–60. [Google Scholar] [CrossRef]
- De Oliveira, P.F.; De Souza Lima, I.M.; Munari, C.C.; Bastos, J.K.; Da Silva Filho, A.A.; Tavares, D.C. Comparative evaluation of antiproliferative effects of brazilian green propolis, its main source baccharis dracunculifolia, and their major constituents artepillin C and baccharin. Planta Med. 2014, 80, 490–492. [Google Scholar] [CrossRef]
- Szliszka, E.; Kucharska, A.Z.; Sokół-ŁȨtowska, A.; Mertas, A.; Czuba, Z.P.; Król, W. Chemical composition and anti-inflammatory effect of ethanolic extract of Brazilian green propolis on activated J774A.1 macrophages. Evid. Based Complement. Altern. Med. 2013, 2013. [Google Scholar] [CrossRef] [PubMed]
- De Carvalho, C.; Fernandes, W.H.C.; Mouttinho, T.B.F.; De Souza, D.M.; Marcucci, M.C.; D’Alpino, P.H.P. Evidence-Based studies and perspectives of the use of brazilian green and red propolis in dentistry. Eur. J. Dent. 2019, 13, 459–469. [Google Scholar] [CrossRef] [PubMed]
- Freires, I.A.; De Alencar, S.M.; Rosalen, P.L. A pharmacological perspective on the use of Brazilian Red Propolis and its isolated compounds against human diseases. Eur. J. Med. Chem. 2016, 110, 267–279. [Google Scholar] [CrossRef]
- Rufatto, L.C.; dos Santos, D.A.; Marinho, F.; Henriques, J.A.P.; Roesch Ely, M.; Moura, S. Red propolis: Chemical composition and pharmacological activity. Asian Pac. J. Trop. Biomed. 2017, 7, 591–598. [Google Scholar] [CrossRef]
- Regueira, M.S.; Tintino, S.R.; da Silva, A.R.P.; do Socorro Costa, M.; Boligon, A.A.; Matias, E.F.F.; de Queiroz Balbino, V.; Menezes, I.R.A.; Melo Coutinho, H.D. Seasonal variation of Brazilian red propolis: Antibacterial activity, synergistic effect and phytochemical screening. Food Chem. Toxicol. 2017, 107, 572–580. [Google Scholar] [CrossRef]
- de Freitas, M.C.D.; de Miranda, M.B.; de Oliveira, D.T.; Vieira-Filho, S.A.; Caligiorne, R.B.; de Figueiredo, S.M. Biological activities of red propolis: A review. Recent Pat. Endocr. Metab. Immune Drug Discov. 2018, 11, 3–12. [Google Scholar] [CrossRef]
- Curti, V.; Zaccaria, V.; Sokeng, A.J.T.; Dacrema, M.; Masiello, I.; Mascaro, A.; D’antona, G.; Daglia, M. Bioavailability and in vivo antioxidant activity of a standardized polyphenol mixture extracted from brown propolis. Int. J. Mol. Sci. 2019, 20, 1250. [Google Scholar] [CrossRef]
- do Nascimento Araújo, C.; Mayworm, M.A.S.; Yatsuda, R.; Negri, G.; Salatino, M.L.F.; Salatino, A.; Timenetsky, J.; Campos, G.B. Chemical composition and antimycoplasma activity of a brown propolis from southern Brazil. J. Food Sci. Technol. 2020, 57, 4228–4235. [Google Scholar] [CrossRef] [PubMed]
- Popova, M.; Trusheva, B.; Bankova, V. Content of biologically active compounds in Bulgarian propolis: A basis for its standardization. Bulg. Chem. Commun. 2017, 49, 115–120. [Google Scholar]
- Popova, M.; Giannopoulou, E.; Skalicka-Wózniak, K.; Graikou, K.; Widelski, J.; Bankova, V.; Kalofonos, H.; Sivolapenko, G.; Gaweł-Bȩben, K.; Antosiewicz, B.; et al. Characterization and biological evaluation of propolis from Poland. Molecules 2017, 22, 1159. [Google Scholar] [CrossRef]
- Nazir, H.; Shahidan, W.N.S.; Ibrahim, H.A.; Ismail, T.N.N.T. Chemical constituents of Malaysian geniotrigona thoracica propolis. Pertanika J. Trop. Agric. Sci. 2018, 41, 955–962. [Google Scholar]
- Zhao, L.; Yu, M.; Sun, M.; Xue, X.; Wang, T.; Cao, W.; Sun, L. Rapid determination of major compounds in the ethanol extract of geopropolis from Malaysian stingless bees, heterotrigona itama, by UHPLC-Q-TOF/MS and NMR. Molecules 2017, 22, 1935. [Google Scholar] [CrossRef] [PubMed]
- Kraikongjit, S.; Jongjitvimol, T.; Mianjinda, N.; Sirithep, N.; Kaewbor, T.; Jumroon, N.; Jongjitvimol, J. Antibacterial effect of plant resin collected fromTetrigona apicalis (Smith, 1857) in Thung Salaeng Luang National Park, Phitsanulok. Walailak J. Sci. Technol. 2018, 15, 599–607. [Google Scholar] [CrossRef]
- Kasote, D.M.; Pawar, M.V.; Gundu, S.S.; Bhatia, R.; Nandre, V.S.; Jagtap, S.D.; Mahajan, S.G.; Kulkarni, M.V. Chemical profiling, antioxidant, and antimicrobial activities of Indian stingless bees propolis samples. J. Apic. Res. 2019, 58, 617–625. [Google Scholar] [CrossRef]
- De Souza, E.C.A.; Da Silva, E.J.G.; Cordeiro, H.K.C.; Lage Filho, N.M.; Da Silva, F.M.A.; Dos Reis, D.L.S.; Porto, C.; Pilau, E.J.; Da Costa, L.A.M.A.; De Souza, A.D.L.; et al. Chemical compositions and antioxidant and antimicrobial activities of propolis produced by frieseomelitta longipes and apis mellifera BEES. Quim. Nova 2018, 41, 485–491. [Google Scholar] [CrossRef]
- de Sousa-Fontoura, D.M.N.; Olinda, R.G.; Viana, G.A.; Kizzy, K.M.; Batista, J.S.; Serrano, R.M.O.T.; Silva, O.M.D.; Camara, C.A.; Silva, T.M.S. Wound healing activity and chemical composition of geopropolis from Melipona subnitida. Rev. Bras. Farmacogn. 2020, 30, 367–373. [Google Scholar] [CrossRef]
- dos Santos, L.; Hochheim, S.; Boeder, A.M.; Kroger, A.; Tomazzoli, M.M.; Dal Pai Neto, R.; Maraschin, M.; Guedes, A.; de Cordova, C.M.M. Chemical characterization, antioxidant, cytotoxic and antibacterial activity of propolis extracts and isolated compounds from the Brazilian stingless bees Melipona quadrifasciata and Tetragonisca angustula. J. Apic. Res. 2017, 56, 543–558. [Google Scholar] [CrossRef]
- dos Santos, H.F.; Campos, J.F.; dos Santos, C.M.; Balestieri, J.B.P.; Silva, D.B.; Carollo, C.A.; de Picoli Souza, K.; Estevinho, L.M.; dos Santos, E.L. Chemical profile and antioxidant, anti-inflammatory, antimutagenic and antimicrobial activities of geopropolis from the stingless bee Melipona orbignyi. Int. J. Mol. Sci. 2017, 18, 953. [Google Scholar] [CrossRef]
- Campos, J.F.; Das Santos, U.P.; Da Rocha, P.D.S.; Damião, M.J.; Balestieri, J.B.P.; Cardoso, C.A.L.; Paredes-Gamero, E.J.; Estevinho, L.M.; De Picoli Souza, K.; Dos Santos, E.L. Antimicrobial, antioxidant, anti-inflammatory, and cytotoxic activities of propolis from the stingless bee tetragonisca fiebrigi (Jataí). Evid. Based Complement. Altern. Med. 2015, 2015. [Google Scholar] [CrossRef] [PubMed]
- Dutra, R.P.; De Barros Abreu, B.V.; Cunha, M.S.; Batista, M.C.A.; Torres, L.M.B.; Nascimento, F.R.F.; Ribeiro, M.N.S.; Guerra, R.N.M. Phenolic acids, hydrolyzable tannins, and antioxidant activity of geopropolis from the stingless bee melipona fasciculata smith. J. Agric. Food Chem. 2014, 62, 2549–2557. [Google Scholar] [CrossRef]
- Cisilotto, J.; Sandjo, L.P.; Faqueti, L.G.; Fernandes, H.; Joppi, D.; Biavatti, M.W.; Creczynski-Pasa, T.B. Cytotoxicity mechanisms in melanoma cells and UPLC-QTOF/MS2 chemical characterization of two Brazilian stingless bee propolis: Uncommon presence of piperidinic alkaloids. J. Pharm. Biomed. Anal. 2018, 149, 502–511. [Google Scholar] [CrossRef] [PubMed]
- Massaro, F.C.; Brooks, P.R.; Wallace, H.M.; Russell, F.D. Cerumen of Australian stingless bees (Tetragonula carbonaria): Gas chromatography-mass spectrometry fingerprints and potential anti-inflammatory properties. Naturwissenschaften 2011, 98, 329–337. [Google Scholar] [CrossRef]
- Popova, M.; Gerginova, D.; Trusheva, B.; Simova, S.; Tamfu, A.N.; Ceylan, O.; Clark, K.; Bankova, V. A preliminary study of chemical profiles of honey, cerumen, and propolis of the african stingless bee meliponula ferruginea. Foods 2021, 10, 997. [Google Scholar] [CrossRef] [PubMed]
- Gajger, I.T.; Dar, S.A. Plant allelochemicals as sources of insecticides. Insects 2021, 12, 189. [Google Scholar] [CrossRef]
- Cheng, A.X.; Lou, Y.G.; Mao, Y.B.; Lu, S.; Wang, L.J.; Chen, X.Y. Plant terpenoids: Biosynthesis and ecological functions. J. Integr. Plant Biol. 2007, 49, 179–186. [Google Scholar] [CrossRef]
- Pichersky, E.; Raguso, R.A. Why do plants produce so many terpenoid compounds? New Phytol. 2018, 220, 692–702. [Google Scholar] [CrossRef] [PubMed]
- Karunanithi, P.S.; Zerbe, P. Terpene synthases as metabolic gatekeepers in the evolution of plant terpenoid chemical diversity. Front. Plant Sci. 2019, 10. [Google Scholar] [CrossRef]
- Melliou, E.; Stratis, E.; Chinou, I. Volatile constituents of propolis from various regions of Greece—Antimicrobial activity. Food Chem. 2007, 103, 375–380. [Google Scholar] [CrossRef]
- Popova, M.P.; Graikou, K.; Chinou, I.; Bankova, V.S. GC-MS profiling of diterpene compounds in mediterranean propolis from Greece. J. Agric. Food Chem. 2010, 58, 3167–3176. [Google Scholar] [CrossRef] [PubMed]
- Popova, M.; Trusheva, B.; Antonova, D.; Cutajar, S.; Mifsud, D.; Farrugia, C.; Tsvetkova, I.; Najdenski, H.; Bankova, V. The specific chemical profile of Mediterranean propolis from Malta. Food Chem. 2011, 126, 1431–1435. [Google Scholar] [CrossRef]
- Pujirahayu, N.; Suzuki, T.; Katayama, T. Cycloartane-type triterpenes and botanical origin of propolis of stingless Indonesian bee tetragonula sapiens. Plants 2019, 8, 57. [Google Scholar] [CrossRef]
- Iqbal, M.; Fan, T.P.; Watson, D.; Alenezi, S.; Saleh, K.; Sahlan, M. Preliminary studies: The potential anti-angiogenic activities of two Sulawesi Island (Indonesia) propolis and their chemical characterization. Heliyon 2019, 5. [Google Scholar] [CrossRef] [PubMed]
- Wen, C.C.; Kuo, Y.H.; Jan, J.T.; Liang, P.H.; Wang, S.Y.; Liu, H.G.; Lee, C.K.; Chang, S.T.; Kuo, C.J.; Lee, S.S.; et al. Specific plant terpenoids and lignoids possess potent antiviral activities against severe acute respiratory syndrome coronavirus. J. Med. Chem. 2007, 50, 4087–4095. [Google Scholar] [CrossRef] [PubMed]
- Guimarães, A.C.; Meireles, L.M.; Lemos, M.F.; Guimarães, M.C.C.; Endringer, D.C.; Fronza, M.; Scherer, R. Antibacterial activity of terpenes and terpenoids present in essential oils. Molecules 2019, 24, 2471. [Google Scholar] [CrossRef]
- Wang, C.Y.; Chen, Y.W.; Hou, C.Y. Antioxidant and antibacterial activity of seven predominant terpenoids. Int. J. Food Prop. 2019, 22, 230–238. [Google Scholar] [CrossRef]
- Chen, Y.; Zhu, Z.; Chen, J.; Zheng, Y.; Limsila, B.; Lu, M.; Gao, T.; Yang, Q.; Fu, C.; Liao, W. Terpenoids from Curcumae Rhizoma: Their anticancer effects and clinical uses on combination and versus drug therapies. Biomed. Pharmacother. 2021, 138. [Google Scholar] [CrossRef]
- Mohamed, W.A.S.; Ismail, N.Z.; Omar, E.A.; Abdul Samad, N.; Adam, S.K.; Mohamad, S. GC-MS evaluation, antioxidant content, and cytotoxic activity of propolis extract from peninsular malaysian stingless bees, tetrigona apicalis. Evid. Based Complement. Altern. Med. 2020, 2020. [Google Scholar] [CrossRef]
- Torres-González, A.; López-Rivera, P.; Duarte-Lisci, G.; López-Ramírez, Á.; Correa-Benítez, A.; Rivero-Cruz, J.F. Analysis of volatile components from Melipona beecheii geopropolis from Southeast Mexico by headspace solid-phase microextraction. Nat. Prod. Res. 2016, 30, 237–240. [Google Scholar] [CrossRef]
- Eroglu, N.; Akkus, S.; Yaman, M.; Asci, B.; Silici, S. Amino acid and vitamin content of propolis collected by native caucasican honeybees. J. Apic. Sci. 2016, 60, 101–110. [Google Scholar] [CrossRef]
- Tosic, S.; Stojanovic, G.; Mitic, S.; Pavlovic, A.; Alagic, S. Mineral composition of selected serbian propolis samples. J. Apic. Sci. 2017, 61, 5–15. [Google Scholar] [CrossRef]
- Wezgowiec, J.; Wieczynska, A.; Wieckiewicz, W.; Kulbacka, J.; Saczko, J.; Pachura, N.; Wieckiewicz, M.; Gancarz, R.; Wilk, K.A. Polish propolis-Chemical composition and biological effects in tongue cancer cells and macrophages. Molecules 2020, 25, 2426. [Google Scholar] [CrossRef] [PubMed]
- Dezmirean, D.S.; Paşca, C.; Moise, A.R.; Bobiş, O. Plant sources responsible for the chemical composition and main bioactive properties of poplar-type propolis. Plants 2021, 10, 22. [Google Scholar] [CrossRef] [PubMed]
- Wolska, K.; Górska, A.; Antosik, K.; Ługowska, K. Immunomodulatory effects of propolis and its components on basic immune cell functions. Indian J. Pharm. Sci. 2019, 81, 575–588. [Google Scholar] [CrossRef]
- Al-Hariri, M. Immune’s-boosting agent: Immunomodulation potentials of propolis. J. Fam. Community Med. 2019, 26, 57–60. [Google Scholar] [CrossRef]
- Debiaggi, M.; Tateo, F.; Pagani, L.; Luini, M.; Romero, E. Effects of propolis flavonoids on virus infectivity and replication. Microbiologica 1990, 13, 207–213. [Google Scholar]
- Forouzanfar, M.H.; Afshin, A.; Alexander, L.T.; Biryukov, S.; Brauer, M.; Cercy, K.; Charlson, F.J.; Cohen, A.J.; Dandona, L.; Estep, K.; et al. Global, regional, and national comparative risk assessment of 79 behavioural, environmental and occupational, and metabolic risks or clusters of risks, 1990–2015: A systematic analysis for the Global Burden of Disease Study 2015. Lancet 2016, 388, 1659–1724. [Google Scholar] [CrossRef]
- Harisna, A.H.; Nurdiansyah, R.; Syaifie, P.H.; Nugroho, D.W.; Saputro, K.E.; Firdayani; Prakoso, C.D.; Rochman, N.T.; Maulana, N.N.; Noviyanto, A.; et al. In silico investigation of potential inhibitors to main protease and spike protein of SARS-CoV-2 in propolis. Biochem. Biophys. Reports 2021, 26. [Google Scholar] [CrossRef]
- Khayrani, A.C.; Irdiani, R.; Aditama, R.; Pratami, D.K.; Lischer, K.; Ansari, M.J.; Chinnathambi, A.; Alharbi, S.A.; Almoallim, H.S.; Sahlan, M. Evaluating the potency of Sulawesi propolis compounds as ACE-2 inhibitors through molecular docking for COVID-19 drug discovery preliminary study. J. King Saud Univ. Sci. 2021, 33. [Google Scholar] [CrossRef]
- Dewi, L.K.; Sahlan, M.; Pratami, D.K.; Agus, A.; Agussalim; Sabir, A. Identifying propolis compounds potential to be covid-19 therapies by targeting sars-cov-2 main protease. Int. J. Appl. Pharm. 2021, 13, 103–110. [Google Scholar] [CrossRef]
- Sahlan, M.; Irdiani, R.; Flamandita, D.; Aditama, R.; Alfarraj, S.; Ansari, M.J.; Khayrani, A.C.; Pratami, D.K.; Lischer, K. Molecular interaction analysis of Sulawesi propolis compounds with SARS-CoV-2 main protease as preliminary study for COVID-19 drug discovery. J. King Saud Univ. Sci. 2021, 33. [Google Scholar] [CrossRef]
- Serkedjieva, J.; Manolova, N.; Bankova, V. Anti-influenza virus effect of some propolis constituents and their analogues (esters of substituted cinnamic acids). J. Nat. Prod. 1992, 55, 294–297. [Google Scholar] [CrossRef] [PubMed]
- Shimizu, T.; Hino, A.; Tsutsumi, A.; Yong, K.P.; Watanabe, W.; Kurokawa, M. Anti-influenza virus activity of propolis in vitro and its efficacy against influenza infection in mice. Antivir. Chem. Chemother. 2008, 19, 7–13. [Google Scholar] [CrossRef]
- Kai, H.; Obuchi, M.; Yoshida, H.; Watanabe, W.; Tsutsumi, S.; Park, Y.K.; Matsuno, K.; Yasukawa, K.; Kurokawa, M. In vitro and in vivo anti-influenza virus activities of flavonoids and related compounds as components of Brazilian propolis (AF-08). J. Funct. Foods 2014, 8, 214–223. [Google Scholar] [CrossRef]
- Kuwata, K.; Urushisaki, T.; Takemura, T.; Tazawa, S.; Fukuoka, M.; Hosokawa-Muto, J.; Araki, Y. Caffeoylquinic acids are major constituents with potent anti-influenza effects in brazilian green propolis water extract. Evid. Based Complement. Altern. Med. 2011, 2011. [Google Scholar] [CrossRef]
- Kuwata, K.; Takemura, T.; Urushisaki, T.; Fukuoka, M.; Hosokawa-Muto, J.; Hata, T.; Okuda, Y.; Hori, S.; Tazawa, S.; Araki, Y. 3,4-dicaffeoylquinic acid, a major constituent of Brazilian propolis, increases TRAIL expression and extends the lifetimes of mice infected with the influenza a virus. Evid. Based Complement. Altern. Med. 2012, 2012. [Google Scholar] [CrossRef]
- Ito, J.; Chang, F.R.; Wang, H.K.; Park, Y.K.; Ikegaki, M.; Kilgore, N.; Lee, K.H. Anti-AIDS agents. 48. Anti-HIV activity of moronic acid derivatives and the new melliferone-related triterpenoid isolated from Brazilian propolis. J. Nat. Prod. 2001, 64, 1278–1281. [Google Scholar] [CrossRef] [PubMed]
- Gekker, G.; Hu, S.; Spivak, M.; Lokensgard, J.R.; Peterson, P.K. Anti-HIV-1 activity of propolis in CD4+ lymphocyte and microglial cell cultures. J. Ethnopharmacol. 2005, 102, 158–163. [Google Scholar] [CrossRef] [PubMed]
- da Silva, C.C.F.; Salatino, A.; da Motta, L.B.; Negri, G.; Salatino, M.L.F. Chemical characterization, antioxidant and anti-HIV activities of a Brazilian propolis from Ceará state. Rev. Bras. Farmacogn. 2019, 29, 309–318. [Google Scholar] [CrossRef]
- Amoros, M.; Sauvager, F.; Girre, L.; Cormier, M. In vitro antiviral activity of propolis. Apidologie 1992, 23, 231–240. [Google Scholar] [CrossRef]
- Schnitzler, P.; Neuner, A.; Nolkemper, S.; Zundel, C.; Nowack, H.; Sensch, K.H.; Reichling, J. Antiviral activity and mode of action of propolis extracts and selected compounds. Phyther. Res. 2010, 24, 632. [Google Scholar] [CrossRef]
- Bankova, V.; Galabov, A.S.; Antonova, D.; Vilhelmova, N.; Di Perri, B. Chemical composition of Propolis Extract ACF® and activity against herpes simplex virus. Phytomedicine 2014, 21, 1432–1438. [Google Scholar] [CrossRef]
- Coelho, G.R.; Mendonça, R.Z.; De SennaVilar, K.; Figueiredo, C.A.; Badari, J.C.; Taniwaki, N.; Namiyama, G.; De Oliveira, M.I.; Curti, S.P.; Evelyn Silva, P.; et al. Antiviral action of hydromethanolic extract of geopropolis from scaptotrigona postica against antiherpes simplex virus (HSV-1). Evid. Based Complement. Altern. Med. 2015, 2015. [Google Scholar] [CrossRef]
- Hochheim, S.; Guedes, A.; Faccin-Galhardi, L.; Rechenchoski, D.Z.; Nozawa, C.; Linhares, R.E.; da Filho, H.H.S.; Rau, M.; Siebert, D.A.; Micke, G.; et al. Determination of phenolic profile by HPLC–ESI-MS/MS, antioxidant activity, in vitro cytotoxicity and anti-herpetic activity of propolis from the Brazilian native bee Melipona quadrifasciata. Rev. Bras. Farmacogn. 2019, 29, 339–350. [Google Scholar] [CrossRef]
- Kurokawa, M.; Shimizu, T.; Takeshita, Y.; Takamori, Y.; Kai, H.; Sawamura, R.; Yoshida, H.; Watanabe, W.; Tsutsumi, A.; Park, Y.K.; et al. Efficacy of Brazilian propolis against herpes simplex virus type 1 infection in mice and their modes of antiherpetic efficacies. Evid. Based Complement. Altern. Med. 2011, 2011, 976196. [Google Scholar]
- Sartori, G.; Pesarico, A.P.; Pinton, S.; Dobrachinski, F.; Roman, S.S.; Pauletto, F.; Rodrigues, L.C.; Prigol, M. Protective effect of brown Brazilian propolis against acute vaginal lesions caused by herpes simplex virus type 2 in mice: Involvement of antioxidant and anti-inflammatory mechanisms. Cell Biochem. Funct. 2012, 30, 1–10. [Google Scholar] [CrossRef]
- Búfalo, M.C.; Figueiredo, A.S.; De Sousa, J.P.B.; Candeias, J.M.G.; Bastos, J.K.; Sforcin, J.M. Anti-poliovirus activity of Baccharis dracunculifolia and propolis by cell viability determination and real-time PCR. J. Appl. Microbiol. 2009, 107, 1669–1680. [Google Scholar] [CrossRef]
- Coelho, G.R.; De Senna Villar, K.; Figueiredo, C.A.; Badari, J.C.; Mendonca, R.M.Z.; Oliveira, M.I.; Curti, S.P.; Silva, P.E.S.; Do Nascimento, R.M.; Mendonca, R.Z. Antiviral effects of Scaptotrigona postica propolis and their fractions. BMC Proc. 2014, 8, 63. [Google Scholar] [CrossRef]
- Kwon, M.J.; Shin, H.M.; Perumalsamy, H.; Wang, X.; Ahn, Y.J. Antiviral effects and possible mechanisms of action of constituents from Brazilian propolis and related compounds. J. Apic. Res. 2020, 59, 413–425. [Google Scholar] [CrossRef]
- Kumar, V.; Dhanjal, J.K.; Kaul, S.C.; Wadhwa, R.; Sundar, D. Withanone and caffeic acid phenethyl ester are predicted to interact with main protease (Mpro) of SARS-CoV-2 and inhibit its activity. J. Biomol. Struct. Dyn. 2020, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Ma, X.; Guo, Z.; Zhang, Z.; Li, X.; Wang, X.; Liu, Y.; Wang, X. Ferulic acid isolated from propolis inhibits porcine parvovirus replication potentially through Bid-mediate apoptosis. Int. Immunopharmacol. 2020, 83. [Google Scholar] [CrossRef]
- Hayakari, R.; Matsumiya, T.; Xing, F.; Tayone, J.C.; Dempoya, J.; Tatsuta, T.; Aizawa-Yashiro, T.; Imaizumi, T.; Yoshida, H.; Satoh, K. Effects of Brazilian green propolis on double-stranded RNA-mediated induction of interferon-inducible gene and inhibition of recruitment of polymorphonuclear cells. J. Sci. Food Agric. 2013, 93, 646–651. [Google Scholar] [CrossRef] [PubMed]
- Verhelst, J.; Hulpiau, P.; Saelens, X. Mx proteins: Antiviral gatekeepers that restrain the uninvited. Microbiol. Mol. Biol. Rev. 2013, 77, 551–566. [Google Scholar] [CrossRef] [PubMed]
- Dabbagh-Bazarbachi, H.; Clergeaud, G.; Quesada, I.M.; Ortiz, M.; O’Sullivan, C.K.; Fernández-Larrea, J.B. Zinc ionophore activity of quercetin and epigallocatechin-gallate: From hepa 1-6 cells to a liposome model. J. Agric. Food Chem. 2014, 62, 8085–8093. [Google Scholar] [CrossRef] [PubMed]
- Kaushik, N.; Subramani, C.; Anang, S.; Muthumohan, R.; Shalimar; Nayak, B.; Ranjith-Kumar, C.T.; Surjit, M. Zinc salts block hepatitis E virus replication by inhibiting the activity of viral RNA-dependent RNA polymerase. J. Virol. 2017, 91, e00754-17. [Google Scholar] [CrossRef] [PubMed]
- Amoros, M.; Simōes, C.M.O.; Girre, L.; Sauvager, F.; Cormier, M. Synergistic effect of flavones and flavonols against herpes simplex virus type 1 in cell culture. Comparison with the antiviral activity of propolis. J. Nat. Prod. 1992, 55, 1732–1740. [Google Scholar] [CrossRef] [PubMed]
- Przybyłek, I.; Karpiński, T.M. Antibacterial properties of propolis. Molecules 2019, 24, 2047. [Google Scholar] [CrossRef]
- Almuhayawi, M.S. Propolis as a novel antibacterial agent. Saudi J. Biol. Sci. 2020, 27, 3079–3086. [Google Scholar] [CrossRef]
- Silva-Carvalho, R.; Baltazar, F.; Almeida-Aguiar, C. Propolis: A complex natural product with a plethora of biological activities that can be explored for drug development. Evid. Based Complement. Altern. Med. 2015, 2015. [Google Scholar] [CrossRef]
- Sforcin, J.M.; Bankova, V. Propolis: Is there a potential for the development of new drugs? J. Ethnopharmacol. 2011, 133, 253–260. [Google Scholar] [CrossRef]
- Bogdan Kędzia, E.H.-K. Aktywność antybiotyczna propolisu krajowego i europejskiego. The antibiotic activity of native and european propolis. Postępy Fitoter. 2013, 2, 97–107. [Google Scholar]
- Bridi, R.; Montenegro, G.; Nuñez-Quijada, G.; Giordano, A.; Fernanda Morán-Romero, M.; Jara-Pezoa, I.; Speisky, H.; Atala, E.; López-Alarcón, C. International regulations of propolis quality: Required assays do not necessarily reflect their polyphenolic-related in vitro activities. J. Food Sci. 2015, 80, C1188–C1195. [Google Scholar] [CrossRef]
- Pamplona-Zomenhan, L.C.; Pamplona, B.C.; da Silva, C.B.; Marcucci, M.C.; Mimica, L.M.J. Evaluation of the in vitro antimicrobial activity of an ethanol extract of Brazilian classified propolis on strains of Staphylococcus aureus. Brazilian J. Microbiol. 2011, 42, 1259–1264. [Google Scholar] [CrossRef]
- Ivana Tlak, G.; Iva, P.; Mirza, B.; Ivan, K.; Siniša, S.; Toni, V.; Josipa, V. Components responsible for antimicrobial activity of propolis from continental and Mediterranean regions in Croatian. Czech J. Food Sci. 2017, 35, 376–385. [Google Scholar] [CrossRef]
- Šuran, J.; Cepanec, I.; Mašek, T.; Radić, B.; Radić, S.; Tlak Gajger, I.; Vlainić, J. Propolis extract and its bioactive compounds—From traditional to modern extraction technologies. Molecules 2021, 26, 2930. [Google Scholar] [CrossRef]
- Oksuz, H.; Duran, N.; Tamer, C.; Cetin, M.; Silici, S. Effect of propolis in the treatment of experimental Staphylococcus aureus Keratitis in Rabbits. Ophthalmic Res. 2005, 37, 328–334. [Google Scholar] [CrossRef] [PubMed]
- Orsi, R.O.; Fernandes, A.; Bankova, V.; Sforcin, J.M. The effects of Brazilian and Bulgarian propolis in vitro against Salmonella Typhi and their synergism with antibiotics acting on the ribosome. Nat. Prod. Res. 2012, 26, 430–437. [Google Scholar] [CrossRef] [PubMed]
- Al-Waili, N.; Al-Ghamdi, A.; Ansari, M.J.; Al-Attal, Y.; Salom, K. Synergistic effects of honey and propolis toward drug multi-resistant Staphylococcus Aureus, Escherichia coli and Candida Albicans isolates in single and polymicrobial cultures. Int. J. Med. Sci. 2012, 9, 793–800. [Google Scholar] [CrossRef] [PubMed]
- Kowacz, M.; Pollack, G.H. Propolis-induced exclusion of colloids: Possible new mechanism of biological action. Colloids Interface Sci. Commun. 2020, 38. [Google Scholar] [CrossRef]
- Kowacz, M.; Pollack, G.H. Cells in new light: Ion concentration, voltage, and pressure gradients across a hydrogel membrane. ACS Omega 2020, 5, 21024–21031. [Google Scholar] [CrossRef]
- Sharaf, S.; Higazy, A.; Hebeish, A. Propolis induced antibacterial activity and other technical properties of cotton textiles. Int. J. Biol. Macromol. 2013, 59, 408–416. [Google Scholar] [CrossRef] [PubMed]
- Abramiuc, D.; Ciobanu, L.; Muresan, R.; Chiosac, M.; Muresan, A. Antibacterial finishing of cotton fabrics using biologically active natural compounds. Fibers Polym. 2013, 14, 1826–1833. [Google Scholar] [CrossRef]
- Arıkan, H.K.; Solak, H.H. Propolis Extract-PVA nanocomposites of textile design: Antimicrobial effect on gram positive and negative bacterias. Int. J. Second. Metab. 2017, 218–224. [Google Scholar] [CrossRef]
- Cheng, Y.; Moraru, C.I. Long-range interactions keep bacterial cells from liquid-solid interfaces: Evidence of a bacteria exclusion zone near Nafion surfaces and possible implications for bacterial attachment. Colloids Surf. B Biointerfaces 2018, 162, 16–24. [Google Scholar] [CrossRef] [PubMed]
- Dias, L.G.; Pereira, A.P.; Estevinho, L.M. Comparative study of different Portuguese samples of propolis: Pollinic, sensorial, physicochemical, microbiological characterization and antibacterial activity. Food Chem. Toxicol. 2012, 50, 4246–4253. [Google Scholar] [CrossRef] [PubMed]
- Halder, S.; Yadav, K.K.; Sarkar, R.; Mukherjee, S.; Saha, P.; Haldar, S.; Karmakar, S.; Sen, T. Alteration of Zeta potential and membrane permeability in bacteria: A study with cationic agents. Springerplus 2015, 4, 1–14. [Google Scholar] [CrossRef]
- Kujumgiev, A.; Tsvetkova, I.; Serkedjieva, Y.; Bankova, V.; Christov, R.; Popov, S. Antibacterial, antifungal and antiviral activity of propolis of different geographic origin. J. Ethnopharmacol. 1999, 64, 235–240. [Google Scholar] [CrossRef]
- Ota, C.; Unterkircher, C.; Fantinato, V.; Shimizu, M.T. Antifungal activity of propolis on different species of Candida. Mycoses 2001, 44, 375–378. [Google Scholar] [CrossRef]
- Murad, J.M.; Calvi, S.A.; Soares, A.M.V.C.; Bankova, V.; Sforcin, J.M. Effects of propolis from Brazil and Bulgaria on fungicidal activity of macrophages against Paracoccidioides brasiliensis. J. Ethnopharmacol. 2002, 79, 331–334. [Google Scholar] [CrossRef]
- Siqueira, A.B.S.; Gomes, B.S.; Cambuim, I.; Maia, R.; Abreu, S.; Souza-Motta, C.M.; De Queiroz, L.A.; Porto, A.L.F. Trichophyton species susceptibility to green and red propolis from Brazil. Lett. Appl. Microbiol. 2009, 48, 90–96. [Google Scholar] [CrossRef]
- Bruschi, M.L.; Dota, K.F.D.; Consolaro, M.E.L.; Svidzinski, T.I.E. Antifungal activity of brazilian propolis microparticles against yeasts isolated from vulvovaginal candidiasis. Evid. Based Complement. Altern. Med. 2011, 2011. [Google Scholar] [CrossRef]
- Bonvehí, J.S.; Gutiérrez, A.L. The antimicrobial effects of propolis collected in different regions in the Basque Country (Northern Spain). World J. Microbiol. Biotechnol. 2012, 28, 1351–1358. [Google Scholar] [CrossRef]
- Mutlu Sariguzel, F.; Berk, E.; Koc, A.N.; Sav, H.; Demir, G. Antifungal activity of propolis against yeasts isolated from blood culture: In vitro evaluation. J. Clin. Lab. Anal. 2016, 30, 513–516. [Google Scholar] [CrossRef]
- Ghaly, M.F.; Ezzat, S.M.; Sarhan, M.M. Use of propolis and ultragriseofulvin to inhibit aflatoxigenic fungi. Folia Microbiol. 1998, 43, 156–160. [Google Scholar] [CrossRef]
- Pippi, B.; Lana, A.J.D.; Moraes, R.C.; Güez, C.M.; Machado, M.; de Oliveira, L.F.S.; Lino von Poser, G.; Fuentefria, A.M. In vitro evaluation of the acquisition of resistance, antifungal activity and synergism of Brazilian red propolis with antifungal drugs on Candida spp. J. Appl. Microbiol. 2015, 118, 839–850. [Google Scholar] [CrossRef]
- Oliveira, A.C.P.; Shinobu, C.S.; Longhini, R.; Franco, S.L.; Svidzinski, T.I.E. Antifungal activity of propolis extract against yeasts isolated from onychomycosis lesions. Mem. Inst. Oswaldo Cruz 2006, 101, 493–497. [Google Scholar] [CrossRef] [PubMed]
- Quiroga, E.N.; Sampietro, D.A.; Soberón, J.R.; Sgariglia, M.A.; Vattuone, M.A. Propolis from the northwest of Argentina as a source of antifungal principles. J. Appl. Microbiol. 2006, 101, 103–110. [Google Scholar] [CrossRef] [PubMed]
- Agüero, M.B.; Gonzalez, M.; Lima, B.; Svetaz, L.; Sánchez, M.; Zacchino, S.; Feresin, G.E.; Schmeda-Hirschmann, G.; Palermo, J.; Daniel Wunderlin, A.N.D.; et al. Argentinean propolis from Zuccagnia punctata cav. (Caesalpinieae) exudates: Phytochemical characterization and antifungal activity. J. Agric. Food Chem. 2010, 58, 194–201. [Google Scholar] [CrossRef] [PubMed]
- Falcão, S.I.; Vale, N.; Cos, P.; Gomes, P.; Freire, C.; Maes, L.; Vilas-Boas, M. In vitro evaluation of portuguese propolis and floral sources for antiprotozoal, antibacterial and antifungal activity. Phyther. Res. 2014, 28, 437–443. [Google Scholar] [CrossRef] [PubMed]
- Szweda, P.; Gucwa, K.; Kurzyk, E.; Romanowska, E.; Dzierżanowska-Fangrat, K.; Zielińska Jurek, A.; Kuś, P.M.; Milewski, S. Essential oils, silver nanoparticles and propolis as alternative agents against fluconazole resistant candida albicans, candida glabrata and candida krusei clinical isolates. Indian J. Microbiol. 2015, 55, 175–183. [Google Scholar] [CrossRef] [PubMed]
- Boisard, S.; Le Ray, A.M.; Landreau, A.; Kempf, M.; Cassisa, V.; Flurin, C.; Richomme, P. Antifungal and antibacterial metabolites from a French poplar type propolis. Evid. Based Complement. Altern. Med. 2015, 2015. [Google Scholar] [CrossRef]
- Berretta, A.A.; De Castro, P.A.; Cavalheiro, A.H.; Fortes, V.S.; Bom, V.P.; Nascimento, A.P.; Marquele-Oliveira, F.; Pedrazzi, V.; Ramalho, L.N.Z.; Goldman, G.H. Evaluation of mucoadhesive gels with propolis (EPP-AF) in preclinical treatment of candidiasis vulvovaginal infection. Evid. Based Complement. Altern. Med. 2013, 2013. [Google Scholar] [CrossRef]
- Bonfim, A.P.; Sakita, K.M.; Faria, D.R.; Arita, G.S.; Vendramini, F.A.V.R.; Capoci, I.R.G.; Braga, A.G.; dos Santos, R.S.; Bruschi, M.L.; Becker, T.C.A.; et al. Preclinical approaches in vulvovaginal candidiasis treatment with mucoadhesive thermoresponsive systems containing propolis. PLoS ONE 2020, 15. [Google Scholar] [CrossRef] [PubMed]
- Wagh, V.D. Propolis: A wonder bees product and its pharmacological potentials. Adv. Pharmacol. Sci. 2013, 2013. [Google Scholar] [CrossRef] [PubMed]
- Banskota, A.H.; Tezuka, Y.; Kadota, S. Recent progress in pharmacological research of propolis. Phyther. Res. 2001, 15, 561–571. [Google Scholar] [CrossRef] [PubMed]
- De Castro, P.A.; Bom, V.L.P.; Brown, N.A.; de Almeida, R.S.C.; Ramalho, L.N.Z.; Savoldi, M.; Goldman, M.H.S.; Berretta, A.A.; Goldman, G.H. Identification of the cell targets important for propolis-induced cell death in Candida albicans. Fungal Genet. Biol. 2013, 60, 74–86. [Google Scholar] [CrossRef]
- Peng, L.; Yang, S.; Cheng, Y.J.; Chen, F.; Pan, S.; Fan, G. Antifungal activity and action mode of pinocembrin from propolis against Penicillium italicum. Food Sci. Biotechnol. 2012, 21, 1533–1539. [Google Scholar] [CrossRef]
- Siheri, W.; Zhang, T.; Ebiloma, G.U.; Biddau, M.; Woods, N.; Hussain, M.Y.; Clements, C.J.; Fearnley, J.; Edrada Ebel, R.A.; Paget, T.; et al. Chemical and antimicrobial profiling of propolis from different regions within Libya. PLoS ONE 2016, 11. [Google Scholar] [CrossRef]
- Afrouzan, H.; Zakeri, S.; Mehrizi, A.A.; Molasalehi, S.; Tahghighi, A.; Shokrgozar, M.A.; Es-Haghi, A.; Djadid, N.D. Anti-plasmodial assessment of four different Iranian propolis extracts. Arch. Iran. Med. 2017, 20, 270–281. [Google Scholar] [PubMed]
- AlGabbani, Q.; Mansour, L.; Elnakady, Y.A.; Al-Quraishy, S.; Alomar, S.; Al-Shaebi, E.M.; Abdel-Baki, A.A.S. In vivo assessment of the antimalarial and spleen-protective activities of the Saudi propolis methanolic extract. Parasitol. Res. 2017, 116, 539–547. [Google Scholar] [CrossRef] [PubMed]
- Silva, R.P.D.; Machado, B.A.S.; De Abreu Barreto, G.; Costa, S.S.; Andrade, L.N.; Amaral, R.G.; Carvalho, A.A.; Padilha, F.F.; Barbosa, J.D.V.; Umsza-Guez, M.A. Antioxidant, antimicrobial, antiparasitic, and cytotoxic properties of various Brazilian propolis extracts. PLoS ONE 2017, 12. [Google Scholar] [CrossRef]
- Otoguro, K.; Iwatsuki, M.; Ishiyama, A.; Namatame, M.; Nishihara-Tsukashima, A.; Kiyohara, H.; Hashimoto, T.; Asakawa, Y.; O’Mura, S.; Yamada, H. In vitro antitrypanosomal activity of some phenolic compounds from propolis and lactones from Fijian Kawa (Piper methysticum). J. Nat. Med. 2012, 66, 558–561. [Google Scholar] [CrossRef]
- Omar, R.M.K.; Igoli, J.; Gray, A.I.; Ebiloma, G.U.; Clements, C.; Fearnley, J.; Edrada Ebel, R.A.; Zhang, T.; De Koning, H.P.; Watson, D.G. Chemical characterisation of Nigerian red propolis and its biological activity against Trypanosoma Brucei. Phytochem. Anal. 2016, 27, 107–115. [Google Scholar] [CrossRef] [PubMed]
- Omar, R.; Igoli, J.O.; Zhang, T.; Gray, A.I.; Ebiloma, G.U.; Clements, C.J.; Fearnley, J.; Ebel, R.A.E.; Paget, T.; De Koning, H.P.; et al. The chemical characterization of nigerian propolis samples and their activity against trypanosoma brucei. Sci. Rep. 2017, 7. [Google Scholar] [CrossRef] [PubMed]
- Gressler, L.T.; Da Silva, A.S.; Machado, G.; Rosa, L.D.; Dorneles, F.; Gressler, L.T.; Oliveira, M.S.; Zanette, R.A.; de Vargas, A.C.P.; Monteiro, S.G. Susceptibility of Trypanosoma evansi to propolis extract in vitro and in experimentally infected rats. Res. Vet. Sci. 2012, 93, 1314–1317. [Google Scholar] [CrossRef]
- Nweze, N.E.; Okoro, H.O.; Al Robaian, M.; Omar, R.M.K.; Tor-Anyiin, T.A.; Watson, D.G.; Igoli, J.O. Effects of Nigerian red propolis in rats infected with Trypanosoma brucei brucei. Comp. Clin. Path. 2017, 26, 1129–1133. [Google Scholar] [CrossRef]
- da Silveira Regueira-Neto, M.; Tintino, S.R.; Rolón, M.; Coronal, C.; Vega, M.C.; de Queiroz Balbino, V.; de Melo Coutinho, H.D. Antitrypanosomal, antileishmanial and cytotoxic activities of Brazilian red propolis and plant resin of Dalbergia ecastaphyllum (L) Taub. Food Chem. Toxicol. 2018, 119, 215–221. [Google Scholar] [CrossRef]
- Alotaibi, A.; Ebiloma, G.U.; Williams, R.; Alenezi, S.; Donachie, A.M.; Guillaume, S.; Igoli, J.O.; Fearnley, J.; de Koning, H.P.; Watson, D.G. European propolis is highly active against trypanosomatids including Crithidia fasciculata. Sci. Rep. 2019, 9. [Google Scholar] [CrossRef]
- Alanazi, S.; Alenzi, N.; Alenazi, F.; Tabassum, H.; Watson, D. Chemical characterization of Saudi propolis and its antiparasitic and anticancer properties. Sci. Rep. 2021, 11, 5390. [Google Scholar] [CrossRef] [PubMed]
- Pontin, K.; Da Silva Filho, A.A.; Santos, F.F.; Silva, M.L.A.E.; Cunha, W.R.; Nanayakkara, N.P.D.; Bastos, J.K.; De Albuquerque, S. In vitro and in vivo antileishmanial activities of a Brazilian green propolis extract. Parasitol. Res. 2008, 103, 487–492. [Google Scholar] [CrossRef] [PubMed]
- Hegazi, A.G.; El-fadaly, H.A.; Barakat, A.M.; Abou-el-doubal, S.K.A. In vitro Effects of Some Bee Products on T. gondii Tachyzoites. Glob. Vet. 2014, 13, 1043–1050. [Google Scholar]
- Freitas, S.F.; Shinohara, L.; Sforcin, J.M.; Guimarães, S. In vitro effects of propolis on Giardia duodenalis trophozoites. Phytomedicine 2006, 13, 170–175. [Google Scholar] [CrossRef]
- Mokhtar, A.B.; El-Gayar, E.K.; Habib, E.S. In vitro anti-protozoal activity of propolis extract and cysteine proteases inhibitor (phenyl vinyl sulfone) on blastocystis species. J. Egypt. Soc. Parasitol. 2016, 46, 261–272. [Google Scholar] [CrossRef] [PubMed]
- Asfaram, S.; Fakhar, M.; Keighobadi, M.; Akhtari, J. Promising anti-protozoan activities of propolis (bee glue) as natural product: A review. Acta Parasitol. 2020. [Google Scholar] [CrossRef]
- Fidalgo, L.M.; Ramos, I.S.; Parra, M.G.; Cuesta-Rubio, O.; Hernández, I.M.; Fernández, M.C.; Piccinelli, A.L.; Rastrelli, L. Activity of Cuban propolis extracts on Leishmania amazonensis and Trichomonas vaginalis. Nat. Prod. Commun. 2011, 6, 973–976. [Google Scholar] [CrossRef]
- Siheri, W.; Ebiloma, G.U.; Igoli, J.O.; Gray, A.I.; Biddau, M.; Akrachalanont, P.; Alenezi, S.; Alwashih, M.A.; Edrada-Ebel, R.A.; Muller, S.; et al. Isolation of a novel flavanonol and an alkylresorcinol with highly potent anti-trypanosomal activity from libyan propolis. Molecules 2019, 24, 1041. [Google Scholar] [CrossRef] [PubMed]
- Antwi, C.A.; Amisigo, C.M.; Adjimani, J.P.; Gwira, T.M. In vitro activity and mode of action of phenolic compounds on leishmania donovani. PLoS Negl. Trop. Dis. 2019, 13. [Google Scholar] [CrossRef]
- Volpi, N. Separation of flavonoids and phenolic acids from propolis by capillary zone electrophoresis. Electrophoresis 2004, 25, 1872–1878. [Google Scholar] [CrossRef]
- Mallo, N.; Lamas, J.; Leiro, J.M. Hydrogenosome metabolism is the key target for antiparasitic activity of resveratrol against trichomonas vaginalis. Antimicrob. Agents Chemother. 2013, 57, 2476–2484. [Google Scholar] [CrossRef]
- Duca, A.; Sturza, A.; Moacă, E.A.; Negrea, M.; Lalescu, V.D.; Lungeanu, D.; Dehelean, C.A.; Muntean, D.M.; Alexa, E. Identification of resveratrol as bioactive compound of propolis from western Romania and characterization of phenolic profile and antioxidant activity of ethanolic extracts. Molecules 2019, 24, 3368. [Google Scholar] [CrossRef] [PubMed]
- Embley, T.M.; Van Der Giezen, M.; Horner, D.S.; Dyal, P.L.; Foster, P.; Tielens, A.G.M.; Martin, W.; Tovar, J.; Douglas, A.E.; Cavalier-Smith, T.; et al. Mitochondria and hydrogenosomes are two forms of the same fundamental organelle. Philos. Trans. R. Soc. B Biol. Sci. 2003, 358, 191–203. [Google Scholar] [CrossRef] [PubMed]
- Bolaños, V.; Díaz-Martínez, A.; Soto, J.; Marchat, L.A.; Sanchez-Monroy, V.; Ramírez-Moreno, E. Kaempferol inhibits Entamoeba histolytica growth by altering cytoskeletal functions. Mol. Biochem. Parasitol. 2015, 204, 16–25. [Google Scholar] [CrossRef]
- Bolaños, V.; Díaz-Martínez, A.; Soto, J.; Rodríguez, M.A.; López-Camarillo, C.; Marchat, L.A.; Ramírez-Moreno, E. The flavonoid (-)-epicatechin affects cytoskeleton proteins and functions in Entamoeba histolytica. J. Proteomics 2014, 111, 74–85. [Google Scholar] [CrossRef] [PubMed]
- Fonseca-Silva, F.; Canto-Cavalheiro, M.M.; Menna-Barreto, R.F.S.; Almeida-Amaral, E.E. Effect of apigenin on leishmania amazonensis is associated with reactive oxygen species production followed by mitochondrial dysfunction. J. Nat. Prod. 2015, 78, 880–884. [Google Scholar] [CrossRef] [PubMed]
- Fonseca-Silva, F.; Inacio, J.D.F.; Canto-Cavalheiro, M.M.; Almeida-Amaral, E.E. Reactive oxygen species production and mitochondrial dysfunction contribute to quercetin induced death in Leishmania amazonensis. PLoS ONE 2011, 6. [Google Scholar] [CrossRef]
- Sen, G.; Mukhopadhyay, S.; Ray, M.; Biswas, T. Quercetin interferes with iron metabolism in Leishmania donovani and targets ribonucleotide reductase to exert leishmanicidal activity. J. Antimicrob. Chemother. 2008, 61, 1066–1075. [Google Scholar] [CrossRef] [PubMed]
- da Silva Bortoleti, B.T.; Tomiotto-Pellissier, F.; Gonçalves, M.D.; Miranda-Sapla, M.M.; Assolini, J.P.; Carloto, A.C.; Lima, D.M.; Silveira, G.F.; Almeida, R.S.; Costa, I.N.; et al. Caffeic acid has antipromastigote activity by apoptosis-like process; and anti-amastigote by TNF-α/ROS/NO production and decreased of iron availability. Phytomedicine 2019, 57, 262–270. [Google Scholar] [CrossRef]
- Teles, C.B.G.; Moreira-Dill, L.S.; de Almeida Silva, A.; Facundo, V.A.; de Azevedo, W.F.; da Silva, L.H.P.; Motta, M.C.M.; Stábeli, R.G.; Silva-Jardim, I. A lupane-triterpene isolated from Combretum leprosum Mart. fruit extracts that interferes with the intracellular development of Leishmania (L.) amazonensis in vitro. BMC Complement. Altern. Med. 2015, 15. [Google Scholar] [CrossRef]
- Sanpa, S.; Popova, M.; Bankova, V.; Tunkasiri, T.; Eitssayeam, S.; Chantawannakul, P. Antibacterial compounds from propolis of Tetragonula laeviceps and Tetrigona melanoleuca (Hymenoptera: Apidae) from Thailand. PLoS ONE 2015, 10. [Google Scholar] [CrossRef]
- De Pablos, L.M.; González, G.; Rodrigues, R.; García Granados, A.; Parra, A.; Osuna, A. Action of a pentacyclic triterpenoid, maslinic acid, against Toxoplasma gondii. J. Nat. Prod. 2010, 73, 831–834. [Google Scholar] [CrossRef]
- Moneriz, C.; Mestres, J.; Bautista, J.M.; Diez, A.; Puyet, A. Multi-targeted activity of maslinic acid as an antimalarial natural compound. FEBS J. 2011, 278, 2951–2961. [Google Scholar] [CrossRef]
- Bero, J.; Beaufay, C.; Hannaert, V.; Hérent, M.F.; Michels, P.A.; Quetin-Leclercq, J. Antitrypanosomal compounds from the essential oil and extracts of Keetia leucantha leaves with inhibitor activity on Trypanosoma brucei glyceraldehyde-3-phosphate dehydrogenase. Phytomedicine 2013, 20, 270–274. [Google Scholar] [CrossRef] [PubMed]
- Yamamoto, E.S.; Campos, B.L.S.; Jesus, J.A.; Laurenti, M.D.; Ribeiro, S.P.; Kallás, E.G.; Rafael-Fernandes, M.; Santos-Gomes, G.; Silva, M.S.; Sessa, D.P.; et al. The effect of ursolic acid on leishmania (Leishmania) amazonensis is related to programed cell death and presents therapeutic potential in experimental cutaneous leishmaniasis. PLoS ONE 2015, 10. [Google Scholar] [CrossRef] [PubMed]
- Roberto, M.; Junior, M.; Daugsch, A.; Moraes, C.S.; Queiroga, C.L.; Pastore, G.M.; Park, Y.K. Comparison of volatile and polyphenolic compounds in Brazilian green propolis and its botanical origin Baccharis dracunculifolia. Cienc. Tecnol. Aliment. 2008, 28, 178–181. [Google Scholar]
- Mohtar, L.G.; Rodríguez, S.A.; Nazareno, M.A. Comparative analysis of volatile compound profiles of propolis from different provenances. J. Sci. Food Agric. 2018, 98, 3409–3415. [Google Scholar] [CrossRef]
- Bankova, V.; Popova, M.; Trusheva, B. Propolis volatile compounds: Chemical diversity and biological activity: A review. Chem. Cent. J. 2014, 8. [Google Scholar] [CrossRef]
- Camargos, H.S.; Moreira, R.A.; Mendanha, S.A.; Fernandes, K.S.; Dorta, M.L.; Alonso, A. Terpenes increase the lipid dynamics in the Leishmania plasma membrane at concentrations similar to their IC50 values. PLoS ONE 2014, 9. [Google Scholar] [CrossRef]
- Moura, I.C.; Wunderlich, G.; Uhrig, M.L.; Couto, A.S.; Peres, V.J.; Katzin, A.M.; Kimura, E.A. Limonene arrests parasite development and inhibits isoprenylation of proteins in Plasmodium falciparum. Antimicrob. Agents Chemother. 2001, 45, 2553–2558. [Google Scholar] [CrossRef] [PubMed]
- Rosa, M.D.S.S.; Mendonça-Filho, R.R.; Bizzo, H.R.; Rodrigues, I.D.A.; Soares, R.M.A.; Souto-Padrón, T.; Alviano, C.S.; Lopes, A.H.C.S. Antileishmanial activity of a linalool-rich essential oil from Croton cajucara. Antimicrob. Agents Chemother. 2003, 47, 1895–1901. [Google Scholar] [CrossRef] [PubMed]
- Silveira, M.A.D.; De Jong, D.; Berretta, A.A.; dos Santos Galvão, E.B.; Ribeiro, J.C.; Cerqueira-Silva, T.; Amorim, T.C.; da Conceição, L.F.M.R.; Gomes, M.M.D.; Teixeira, M.B.; et al. Efficacy of Brazilian Green Propolis (EPP-AF®) as an adjunct treatment for hospitalized COVID-19 patients: A randomized, controlled clinical trial. Biomed. Pharmacother. 2021, 111526. [Google Scholar] [CrossRef]
- Esposito, C.; Garzarella, E.U.; Bocchino, B.; D’Avino, M.; Caruso, G.; Buonomo, A.R.; Sacchi, R.; Galeotti, F.; Tenore, G.C.; Zaccaria, V.; et al. A standardized polyphenol mixture extracted from poplar-type propolis for remission of symptoms of uncomplicated upper respiratory tract infection (URTI): A monocentric, randomized, double-blind, placebo-controlled clinical trial. Phytomedicine 2021, 80. [Google Scholar] [CrossRef] [PubMed]
- Cohen, H.A.; Varsano, I.; Kahan, E.; Sarrell, E.M.; Uziel, Y. Effectiveness of an herbal preparation containing echinacea, propolis, and vitamin C in preventing respiratory tract infections in children. Arch. Pediatr. Adolesc. Med. 2004, 158, 217. [Google Scholar] [CrossRef] [PubMed]
- Marchisio, P.; Esposito, S.; Bianchini, S.; Desantis, C.; Galeone, C.; Nazzari, E.; Pignataro, L.; Principi, N. Effectiveness of a propolis and zinc solution in preventing acute otitis media in children with a history of recurrent acute otitis media. Int. J. Immunopathol. Pharmacol. 2010, 23, 567–575. [Google Scholar] [CrossRef] [PubMed]
- Vekic, J.; Ivanisevic, J.; Zeljkovic, A.; Spasojevic-Kalimanovska, V.; Bogavac-Stanojevic, N.; Mihajlovic, M.; Janac, J.; Vujcic, S.; Miljkovic, M.; Zujovic, D.; et al. Effect of propolis and N-acetylcysteine supplementation on lipoprotein subclasses distribution and paraoxonase 1 activity in subjects with acute respiratory infection. J. Med. Biochem. 2020, 39. [Google Scholar] [CrossRef]
- Kosari, M.; Noureddini, M.; Khamechi, S.P.; Najafi, A.; Ghaderi, A.; Sehat, M.; Banafshe, H.R. The effect of propolis plus Hyoscyamus niger L. methanolic extract on clinical symptoms in patients with acute respiratory syndrome suspected to COVID-19: A clinical trial. Phyther. Res. 2021. [Google Scholar] [CrossRef]
| Country | Extracts | Species | Chemical Composition Profile | References |
|---|---|---|---|---|
| European honey bees | ||||
| Bulgaria | hydroethanolic | Apis mellifera | Pinocembrin, pinobanksin, pinobanksin-3-O-acetate, chrysin, galangin, prenyl esters of caffeic acid and ferulic acid | [36,53] |
| Italy | hydroethanolic | Apis mellifera | Pinocembrin, pinobanksin-3-O-acetate, chrysin, galangin, benzyl caffeate, and caffeic acid phenethyl ester | [36] |
| Switzerland | hydroethanolic | Apis mellifera | Pinocembrin, pinobanksin-3-O-acetate, galangin, phenolic glycerides | [36] |
| Poland | hydroethanolic | Apis mellifera | Benzoic acid, p-coumaric acid, ferullic acid, butyl p-coumarate, pentyl p-coumarate, pentenyl p-coumarate, pinocembrin, pinobanksin, chrysin, galangin, and kaempferol | [54] |
| Greece | hydroethanolic | Apis mellifera | Pinocembbrin, apigenin, chrysin, galangin, ellagic acid, tectochrysin, syringic acid, ferullic acid, gallic acid, hesperetin, luteolin, p-coumaric acid, pinobanksin, caffeic acid, pinostrobin, caffeic acid phenethyl ester, quercetin, rhamnetin, kaempferol, chlorogenic acid, protocatechuic acid, kaempferide, acacetin, resveratrol, eriodictyol, naringenin, pinobanksin-3-O-acetate, catechin, and rutin | [37] |
| China | hydroethanolic | Apis mellifera | Caffeic acid, p-coumaric acid, ferulic acid, isoferulic acid, 3,4-dimethylcaffeic acid, pinobanksin, chrysin, pinocembrin, galangin, pinobanksin-3-acetate, and caffeic acid phenethyl ester | [38] |
| Taiwan | hydroethanolic | Apis mellifera | Propolin A-F (prenylflavanones) | [41] |
| India | hydroethanolic | Apis mellifera | Gallic acid, naringin, caffeic acid, p-coumaric acid, ferulic acid, quercetin, cinnamic acid, kaempferol, chrysin, galanginin, and caffeic acid phenethyl ester | [43] |
| Brazil | hydroethanolic | Apis mellifera | Artepillin C, kaempferol, kaempferide, p-coumaric acid, 3-prenyl-4-hydroxycinnamic acid, retusapurpurin A and B, formononetin, biochanin A, vestitol, neovestitol, daidzein, galangin, pinocembrin, chrysin, apigenin, and pinobanksin | [19,44,45,46,47,48,49,50,51,52] |
| Indonesia | hydroethanolic and chloroform | Apis mellifera | Alk(en)ylresorcinols, propolin C, D, F, and G | [42] |
| Stingless bees | ||||
| Malaysia | hydroethanolic | Geniotrigona thoracia | Caffeic acid, p-coumaric acid, quercetin, myricetin, naringenin, hesperitin, kaempferol, and baicaline | [55] |
| Malaysia | ethanolic | Heterotrigona itama | Gallic acid and its derivatives, caffeic acid and its derivatives, vanillic acid, syringic acid, protocatechuic acid, benzoic acid, vitexin-O-gallate, pinobanksin, lapachol, acetyleugenol, kaempferol, and mangostin | [56] |
| Thailand | hydroethanolic | Tetrigona apicalis | Gallic acid, eriodictyol, isoquercetin, quercetin, hydroquinin, catechin | [57] |
| India | ethanolic | Not determined | Gallic acid, naringin, caffeic acid, p-coumaric acid, ferullic acid, quercetin, cinnamic acid, kaempferol, and caffeic acid phenethyl ester | [58] |
| Brazil | hydroethanolic | Frieseomelitta longipes | Xanthochymol and gambogenone | [59] |
| Brazil | hydroethanolic | Melipona subnitida | Gallic acid and its derivatives, p-coumaric acid and its derivatives, cinnamic acid and its derivatives, kaempferol and its derivatives, quercetin and its derivatives, naringenin and its derivatives, ellagic acid, aromadendrin, myricetin dimethyl-ether, and herbacetin | [60] |
| Brazil | hydroethanolic hydroethanolic aqueous aqueous | Melipona quadrifasciata Tetragonisca angustula Melipona quadrifasciata Tetragonisca angustula | Quercetin, epigallocatechin, p-OH-benzoic acid, epigallocatecchin gallate, and coumaric acid Quercetin, p-OH-benzoic acid, caffeic acid, and coumaric acid Rutin, gallic acid, gallocatechin, epicatechin gallate, and syringic acid Quercetin, gallic acid, and gallocatechin | [61] |
| Brazil | hydroethanolic | Melipona orbignyi | Gallic acid and its derivatives, coumaric acid and its derivatives, aromadendrin, naringenin | [62] |
| Brazil | hydroethanolic | Tetragonisca fiebrigi | Benzoic acid, caffeic acid and its derivatives, cinnamic acid and its derivatives, p-coumaric acid and its derivatives | [63] |
| Brazil | hydroethanolic | Melipona fasciculata | Gallic acid and its derivatives, ellagic acid, and valoneic acid dilactone | [64] |
| Brazil | hydroethanolic | Scaptotrigona Bipunctata Melipona quadrifasciata anthidioides | Vicenin-1, -2, and -3Mepuberin | [65] |
| Australia | methanol, followed by diethyl ether and ethyl-O-acetate | Tetragonula carbonaria | Cinnamic acid, p-coumaric acid, phenolic acid, and gallic acid | [66] |
| Tanzania | hydroethanolic | Meliponula ferruginea | p-hydroxybenzoic acid, vanillic acid, p-coumaric acid, caffeic acid, resorcinol, cardanol, and anacardic acid | [67] |
| Country | Extracts | Species | Terpenoid Profile | References |
|---|---|---|---|---|
| Italy | hydroethanolic | Apis mellifera | Diterpenic acids | [36] |
| Greece | hydroethanolic | Apis mellifera | α-pinene, junipene, and δ-cadinene, isocupressic acid, pimaric acid, communic acid, and 14,15-dinor-13-oxo-8(17)-labden-19-oic acid | [72,73] |
| Malta | hydroethanolic | Apis mellifera | 2-acetoxy-6-p-methoxybenzoyljaeschkeanadiol, 2-acetoxy-6-p-methoxybenzoyljaeschkeanadiol, ferutinin, and teferin | [74] |
| Malaysia | hydroethanolic | Geniotrigona thoracia | fren-9(11)-en-2-alpha-ol, lup-20(29)-ene-3,21-dione, 28-hydroxy-, beta-amyrenol, and friedelan-y-al | [55] |
| Malaysia | hydroethanolic | Tetrigona apicalis | α-cubebene, copaene, caryophyllene, bicyclogermacrene, caryophyllene oxide, α-cadinol, α-amyrin, and β-amyrin | [81] |
| Malaysia | ethanolic | Heterotrigona itama | 24(E)-cycloart-24-ene-26-ol-3-one, 20-hydroxy-24-dammaren-3-one, | [56] |
| Indonesia | mangiferolic acid, cycloartenol, and ambolic acid | [75] | ||
| Mexico | solid-phase microextraction | Melipona beecheii | (Z)-ocimenone, α-pinene, m-cymene, trans-isocarveol, limonene, verbenone, β-pinene, acampholenal, m-cymenene, trans-pinocamphone and trans-pulegol | [82] |
| Brazil | hydroethanolic | Frieseomelitta longipes | Pseudolimonene, β-phellandrene, (Z)-β-ocimene, α-cubebene, α-copaene, β-bourbonene, β-longipinene, α-gurjunene, α-cis-bergamotene, β-caryophyllene, β-copaene, β-trans-bergamotene, α-humulene, γ-muurolene, germacrene D, β-chamigrene, valencene, β-bisabolene, γ-cadinene, δ-cadinene, germacrene B | [59] |
| Brazil | hydroethanolic | Melipona orbignyi | Diterpenes, sequisterpenes, and triterpenes | [62] |
| Brazil | hydroethanolic | Scaptotrigona bipunctata Melipona quadrifasciata anthidioides | Triterpene (related to α-amyrin or β-amyrin) 7-O-methyl aromadendrin, abietic acid and its derivatives, | [65] |
| Australia | methanol, followed by diethyl ether and ethyl-O-acetate | Tetragonula carbonaria | Abietic acid, dehydroabietic acid, pimaric acid, and β-amyrin | [66] |
| Tanzania | hydroethanolic | Meliponula ferruginea | Diterpenic acid (pimaric), communic acid, 13-epi-cupressic acid, imbricataloic acid, abietic acid, dehydroabietic acid, acetylisocupressic acid, β-amyrin, cycloartenol, lupeol, β-amyrenone, triterpenic acid, triterpene acetate (betulin), lupenon, dammarenone, mangiferolic acid | [67] |
| Propolis/Propolis-Derived Compounds | Types of Virus | Mechanisms of Action | References |
|---|---|---|---|
| Chrysin, kaempferol, acacetin, galangin, and quercetin | herpesvirus, adenovirus, rotavirus, and coronavirus | Not determined | [89] |
| Kaempferol and p-coumaric acid | rhinovirus | Prevention of the entry of human rhinovirus and inhibition of the viral replication. | [112] |
| Liposomal propolis | Sars Cov 2 | Interfering with 3CL-protease and S1 spike protein of Sars-Cov 2. | [15] |
| Withanone, caffeic acid phenethyl ester, sulabiroins A, (2S)-5,7-dihydroxy-4’-methoxy-8-prenylflavanone acid, glyasperin A, and broussoflavonol F | Sars Cov 2 | Interfering with the highly conserved residues (substrate-binding pocket) of Mpro protein of Sars-Cov 2. | [93,94,113] |
| Isopentyl ferulate | influenza virus A (H3N2) | Not determined | [95] |
| Ethanolic extract of propolis | influenza virus A/PR/8/34 (H1N1) | Reducing the viral load in the bronchoalveolar lavage fluids of the lungs. | [96] |
| Apigenin, kaempferol, and coumaric acid | Influenza A/PR/8/34(H1N1) influenza A/Toyama/26/2011 (H1N1) | Reducing the viral load in the bronchoalveolar lavage fluids of the lungs. | [97] |
| Water extract of propolis, caffeoylquinic acids, and 3,4-dicaffeoylquinic acid | influenza virus A/WSN/33 (H1N1) | Increase in the expression of tumor necrosis factor-related apoptosis-inducing ligand (TRAIL) which expedited viral clearance. | [98,99] |
| Moronic acid | HIV | Inhibition of HIV in H9 lymphocytes. | [100] |
| Hydroalcoholic extract of propolis | HIV | Inhibition of HIV-1 infected CD4+ lymphocyte and microglial cell cultures. | [101] |
| Ethyl acetate extract of propolis, naringenin, quercetin, and diprenylcinnamic acid | HIV | Not determined | [102] |
| Galangin, kaempferol, and quercetin | Herpesvirus | Not determined | [119] |
| Aqueous and ethanolic extracts of propolis | Herpesvirus | Not determined | [104] |
| Poplar propolis extract (ACF®) | Herpesvirus | Not determined | [105] |
| Hydromethanolic extract of propolis, pyrrolizidine alkaloids, and C-glycosyl flavones | Herpesvirus | Not determined | [106] |
| Ethanolic extracts of propolis | Herpesvirus | Significant reduction of the viral load in brains and skins of mice infected with HSV-1. | [108] |
| Hydroalcoholic extract (70% ethanol) of propolis | Herpesvirus | Reduction of the severity of extravaginal lesions and histological damage in the vaginal tissue infected with HSV-2. | [109] |
| Ferulic acid | parvovirus | Inhibition and reversal of the parvovirus-induced expression of pro-apoptotic genes Bid, Bad, Bim, and Bak. | [114] |
| Quercetin and epigallocatechin gallate | Not determined | Zinc ionophore | [117] |
| Propolis/Propolis-Derived Compounds | Types of Fungi and Yeasts | Mechanisms of Action | References |
|---|---|---|---|
| Ethanolic extract of propolis | 20 strains each of Candida albicans, Candida tropicalis, Candida krusei and 15 strainsof Candida guilliermondii. | Not determined | [141] |
| Ethanolic extract of propolis and propolis microparticles | clinical yeast isolates of vulvovaginal candidiasis | Not determined | [144] |
| n-hexane extract of propolis | Candida spp. | Not determined | [148] |
| Hydroethanolic extract of propolis | C. parapsilosis, C. tropicalis, C. albicans, and other species | Not determined | [149] |
| Methanolic extract of propolis, 2′,4′-dihydroxy-3-methoxychalcone and 2′,4′-dihydroxychalcone | M. gypseum, T. mentagrophytes, and T. rubrum | Not determined | [151] |
| Hydroethanolic extract of propolis | C. albicans, T.rubrum, and A. fumigatus | Not determined | [152] |
| Hydroethanolic, methanolic, aqueous, and dichloromethane extracts of propolis | C. albicans and C. glabrata | Not determined | [154] |
| Propolis Standardized Extract (EPP-AF®) | C. abicans | Induction of apoptosis through metacaspase and Ras signaling. Disruption the expression of various genes involved in pathogenesis, cell adhesion, biofilm formation, filamentous growth, and phenotypic switching. | [159] |
| Pinocembrin | P. italicum | Disruption of energy homeostasis, mycelia growth, the structure of the hyphae and the cell membrane. Reduction of the phosphorylated adenosine nucleotides levels. | [160] |
| Propolis/Propolis-Derived Compounds | Types of Parasites | Mechanisms of Action | References |
|---|---|---|---|
| Ethanolic extracts of propolis | Trypanosomabrucei, Leishmania donovani, Plasmodium falciparum, Crithidiafasciculata and Mycobacterium marinum | Not determined | [161] |
| Hydroethanolic, ethyl acetate, and dichloromethane extracts of propolis | Chloroquine (CQ)-sensitive Plasmodium falciparum 3D7 and Plasmodium berghei (ANKA strain) | Not determined | [162] |
| Methanolic extract of propolis | P. chabaudin | Increase in TNF-α and interferon-γ | [163] |
| Ethanolic extract of propolis and supercritically extracted propolis extract | T. cruzi Y strain | Not determined | [164] |
| Ethanolic extract of propolis liquiritigenin, pinocembrin, vestitol, medicarpin, 8-prenylnaringenin, 6-prenylnaringenin, propolin D, macarangin, and dihydrobenzofuran | Standarddrug-sensitive T. brucei brucei clone andtwo pentamidine-resistant types | Not determined | [166] |
| 1,3,7-trihydroxy-2,8-di-(3-methylbut-2-enyl)xanthone, 1,3,7-trihydroxy-4,8-di-(3-methylbut-2-enyl)xanthone 1,7-dihydroxy-8-(3-methylbut-2-enyl)-3-(methylbut-2-enyloxy) xanthone, ambonic acid, mangiferonic acid and a mixture of α-amyrin with mangiferonic acid | T. brucei brucei | Not determined | [167] |
| β-phenethyl caffeate, farnesyl caffeate | T. brucei brucei | Not determined | [165] |
| Taxifolin-3-acetyl-4′-methyl ether and bilobol | T. brucei, P. falciparum T. spiralis,and C. elegans | Induction of cell lysis, disruption phospholipid metabolism and depletion of lipids such as phosphatidyl glycerol (PG) and phosphatidyl inositol (PI) lipids. | [179] |
| Rosmarinic acid and apigenin | L. donovani | Promotion of cell lysis, cytoplasmic condensation, and kinetoplast and nuclear DNA aggregation. Promotion of cell arrest at the G0/G1 phase and induced iron chelation. | [180] |
| Resveratrol | T. vaginalis | Disruption of hydrogenosome metabolism, by affecting [Fe]-hydrogenase (Tvhyd), pyruvate-ferredoxin oxidoreductase, and heat shock protein 70 (Hsp70). | [182] |
| Kaempferol | Entamoeba histolityca | Modification of the expression of actin, myosin II heavy chain and cortexillin II. | [185] |
| Epicathechin | E. histolytica | Disruption the expression of the heat shock protein 70, myosin II heavy chain, and actin. Disruption of the expression offructose-1,6-biphosphate aldolase and glyceraldehyde-phosphate dehydrogenase. | [186] |
| Apigenin and quercetin | L. amazonensis | Upregulation of the expression of reactive oxygen species (ROS), induction of mitochondrial dysfunction and membrane potential disruption, and the inhibition of ribonucleotide reductase. | [187,188,189] |
| Caffeic acid | L. amazonensis | Induction of morphological changes, disruption of the integrity of cellular plasma membrane and mitochondria, and consequently promotion ofapoptosis. Upregulation of the inflammatory response of macrophages by promoting the expression of ROS and TNF-α, while reducing the expression of IL-10 and the availability of iron. | [190] |
| Lupane, maslinic acid and ursolic acid, limonene, α-terpineol, 1,8-cineole, and linalool | L. amazonensis, Toxoplasma gondii, and T. brucei | Induction of morphological changes, promotion of apoptosis, and inhibition of crucial metabolic proteases and enzymes. | [191,193,194,195,196,200,201,202] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zulhendri, F.; Chandrasekaran, K.; Kowacz, M.; Ravalia, M.; Kripal, K.; Fearnley, J.; Perera, C.O. Antiviral, Antibacterial, Antifungal, and Antiparasitic Properties of Propolis: A Review. Foods 2021, 10, 1360. https://doi.org/10.3390/foods10061360
Zulhendri F, Chandrasekaran K, Kowacz M, Ravalia M, Kripal K, Fearnley J, Perera CO. Antiviral, Antibacterial, Antifungal, and Antiparasitic Properties of Propolis: A Review. Foods. 2021; 10(6):1360. https://doi.org/10.3390/foods10061360
Chicago/Turabian StyleZulhendri, Felix, Kavita Chandrasekaran, Magdalena Kowacz, Munir Ravalia, Krishna Kripal, James Fearnley, and Conrad O. Perera. 2021. "Antiviral, Antibacterial, Antifungal, and Antiparasitic Properties of Propolis: A Review" Foods 10, no. 6: 1360. https://doi.org/10.3390/foods10061360
APA StyleZulhendri, F., Chandrasekaran, K., Kowacz, M., Ravalia, M., Kripal, K., Fearnley, J., & Perera, C. O. (2021). Antiviral, Antibacterial, Antifungal, and Antiparasitic Properties of Propolis: A Review. Foods, 10(6), 1360. https://doi.org/10.3390/foods10061360








