Utilization of Eggshell Membrane and Olive Leaf Extract for the Preparation of Functional Materials
Abstract
1. Introduction
2. Materials and Method
2.1. Materials
2.2. Methods
2.2.1. Preparation of Olive Leaf Extract
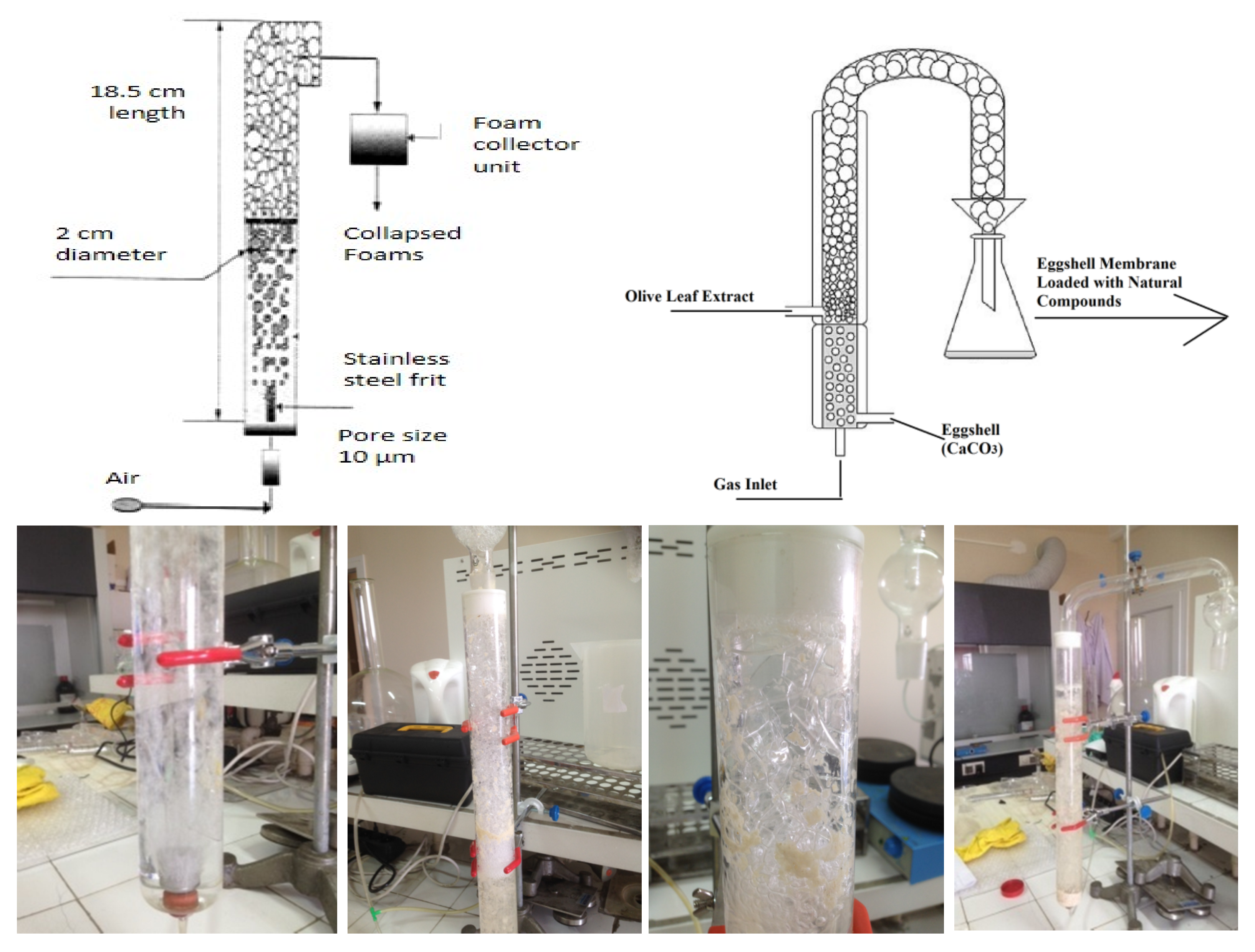
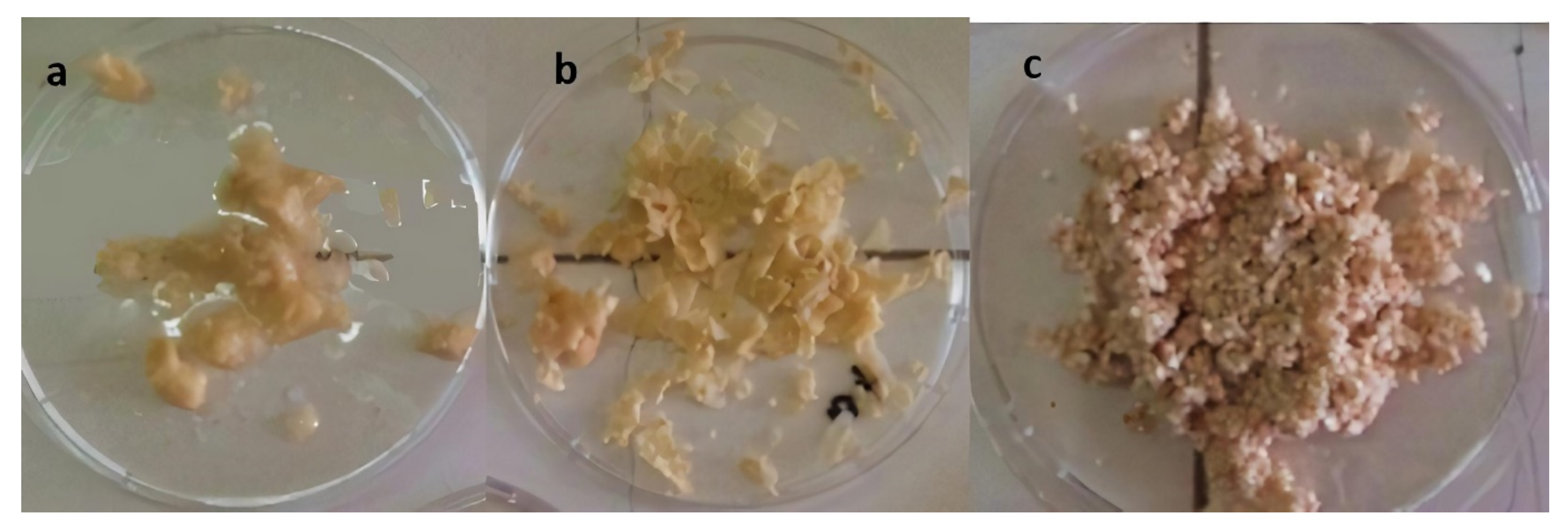
2.2.2. Separation of Eggshell Membrane from Eggshell Waste and Preparation of OLE-Loaded ESM
2.2.3. Experimental Design
2.2.4. Characterization of Eggshell Membrane and OLE-Loaded Eggshell Membrane
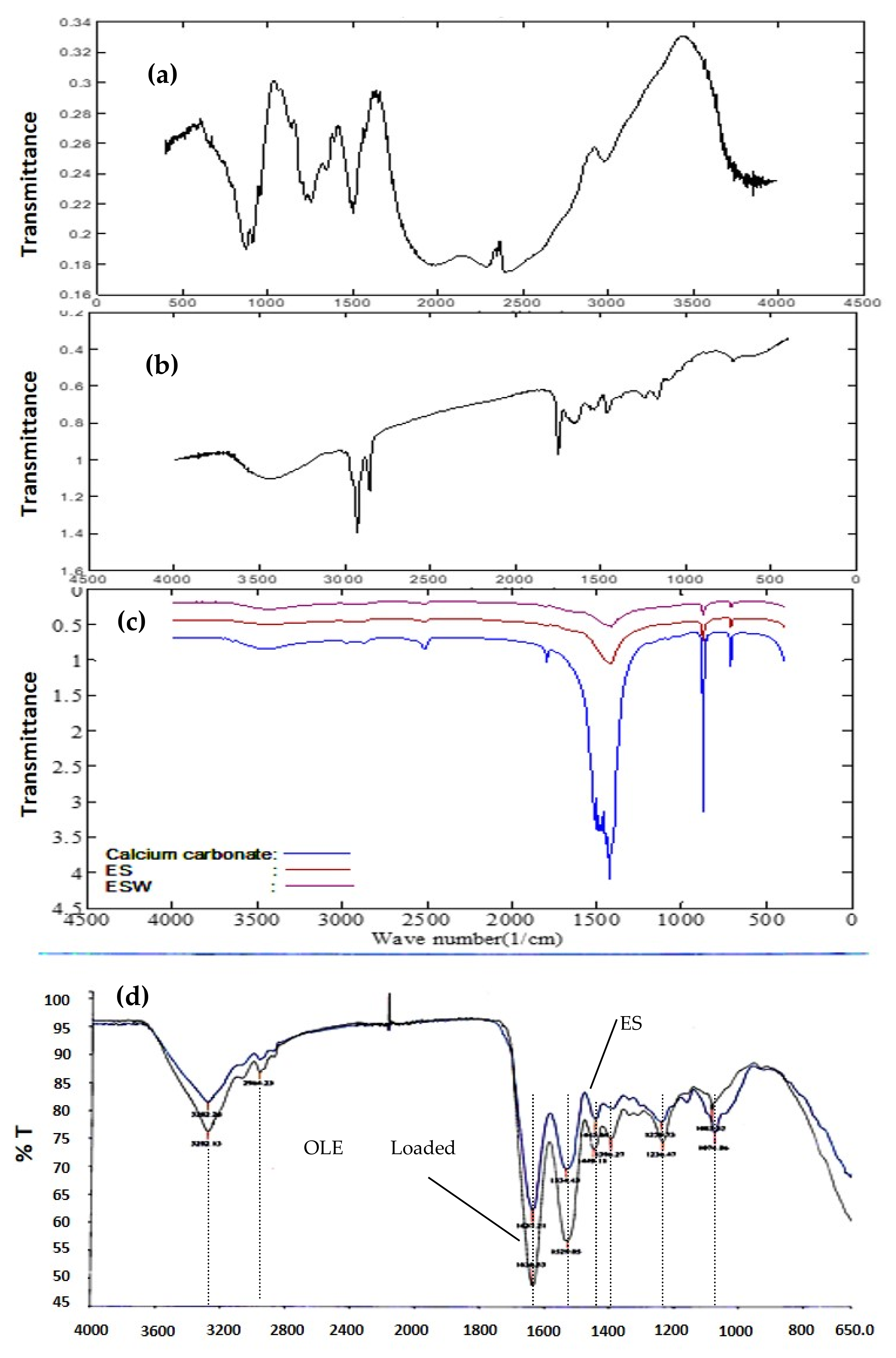
Fourier Transform Infrared (FTIR) Spectroscopy Analysis
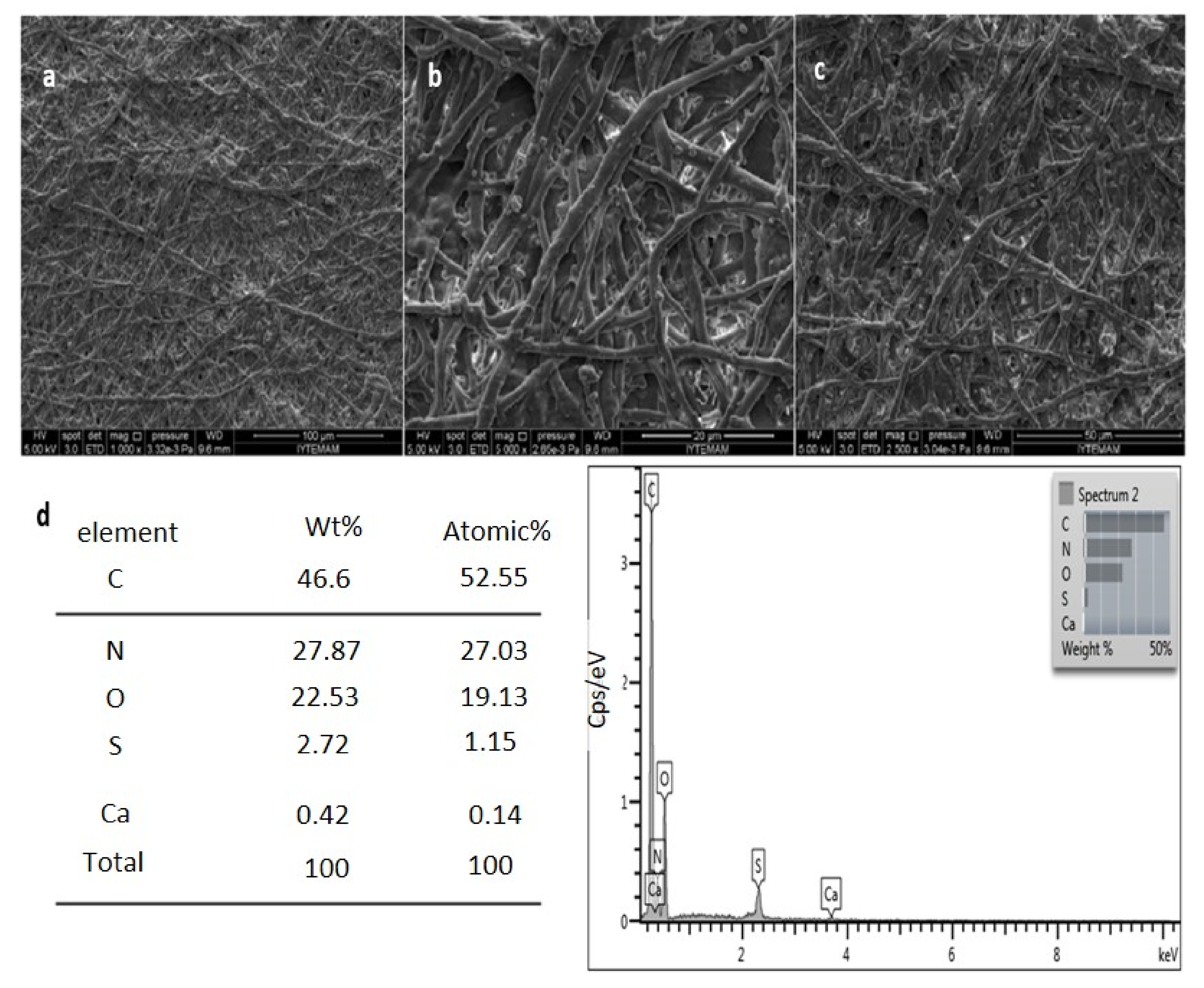
Scanning Electron Microscopy (SEM) Energy-Dispersive X-ray Spectroscopy (EDS) Analysis
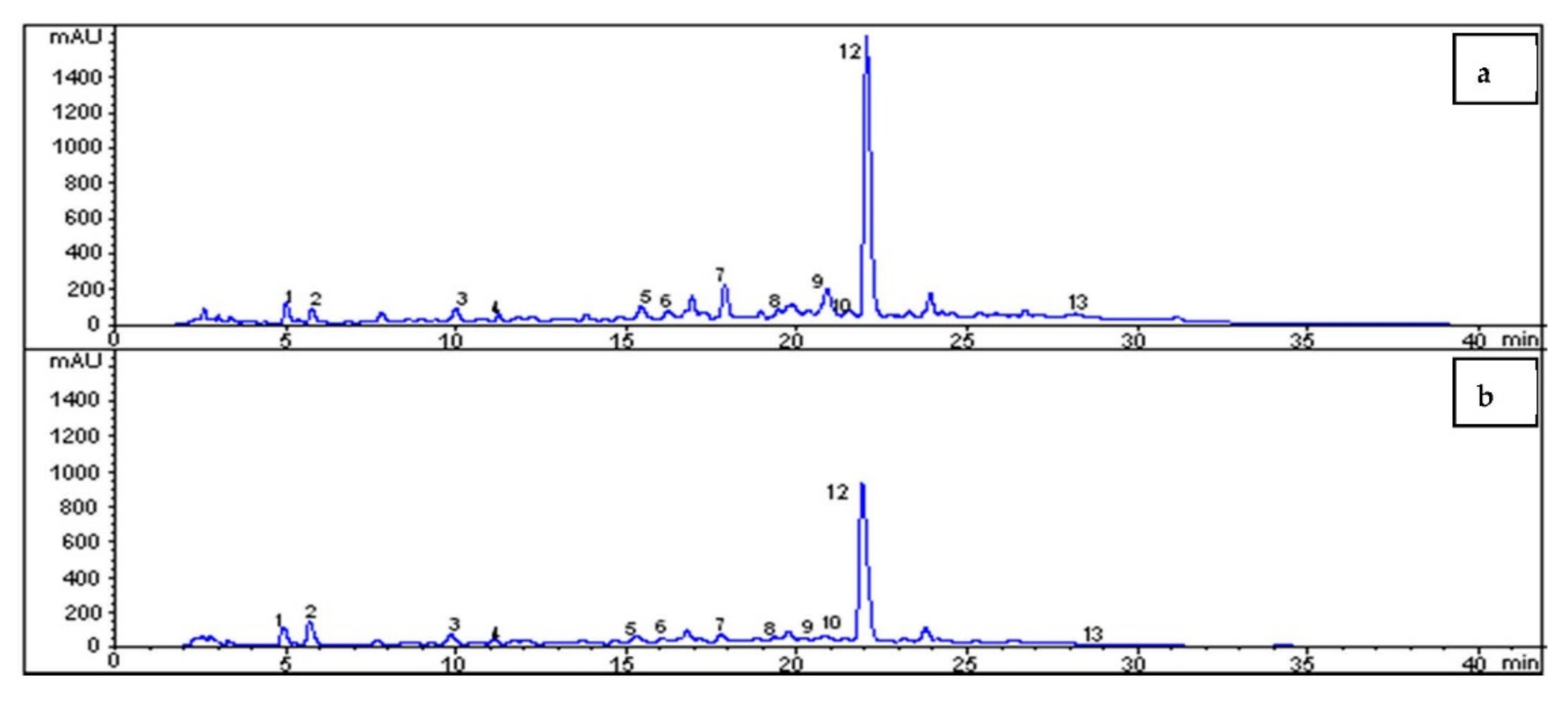
HPLC Analyses
2.2.5. Determination of Total Antioxidant Capacity (TEAC) and Total Phenol Content (TPC) for OLE
2.2.6. Determination of Cytotoxic Activity for OLE-Loaded ESM (MTT Method)
2.2.7. Determination of Antimicrobial Activity of OLE-Loaded ESM with Agar Method
3. Results and Discussion
3.1. Characterization of Eggshell Waste and Eggshell Membrane
3.2. Characterization of Olive Leaf Extract
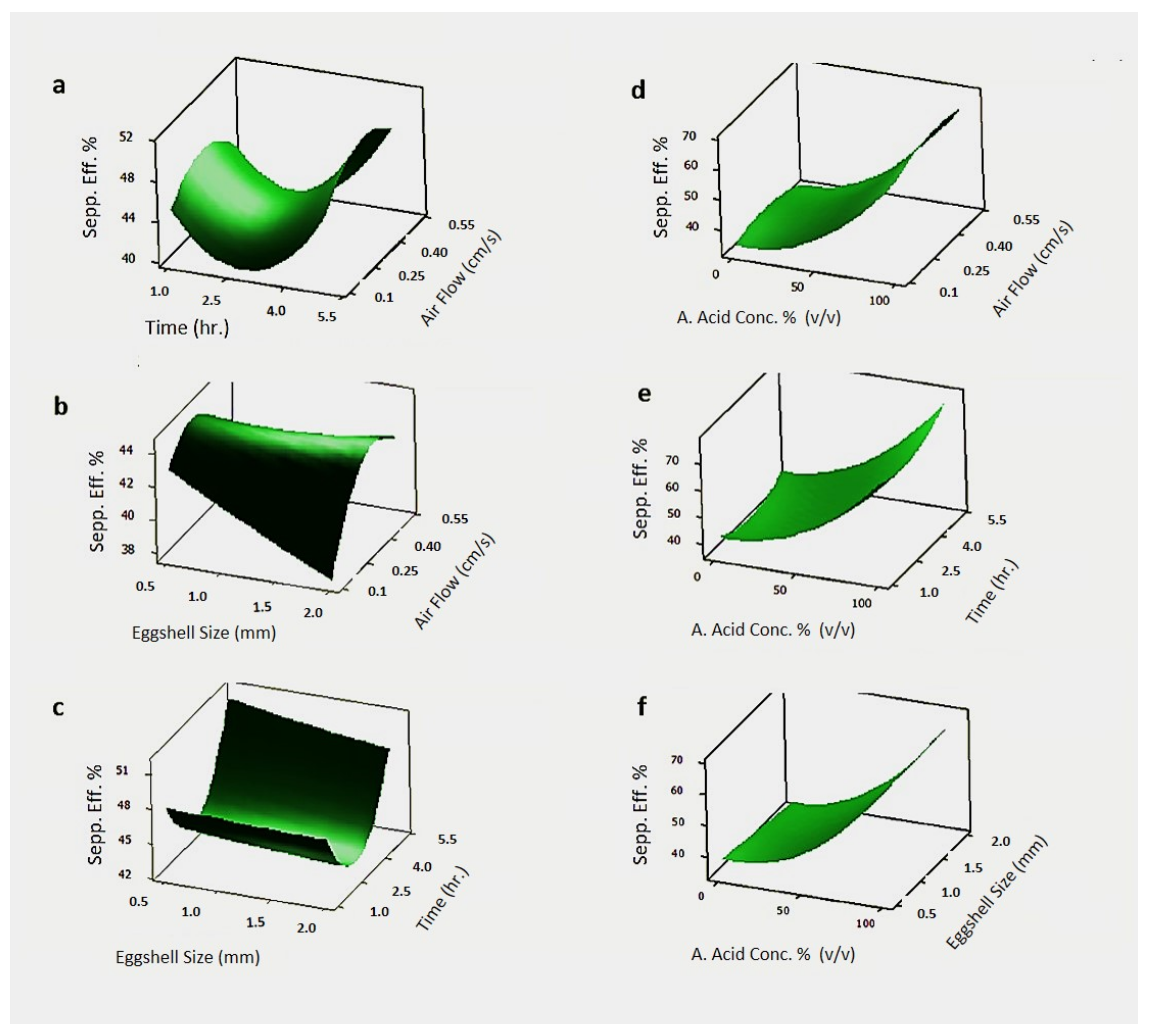
3.2.1. Response Surface Methodology (RSM) for the Separation of Eggshell Membrane
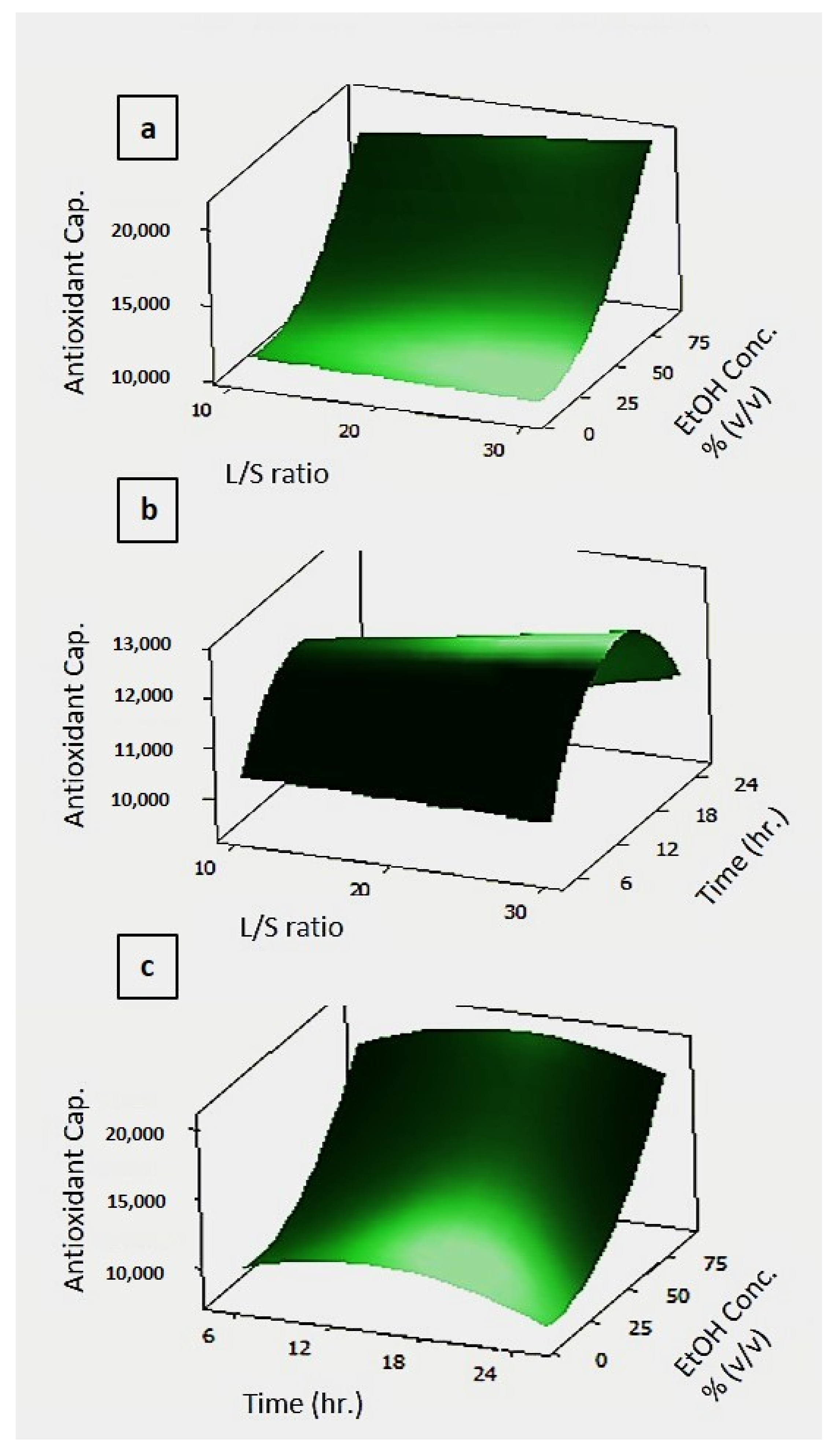
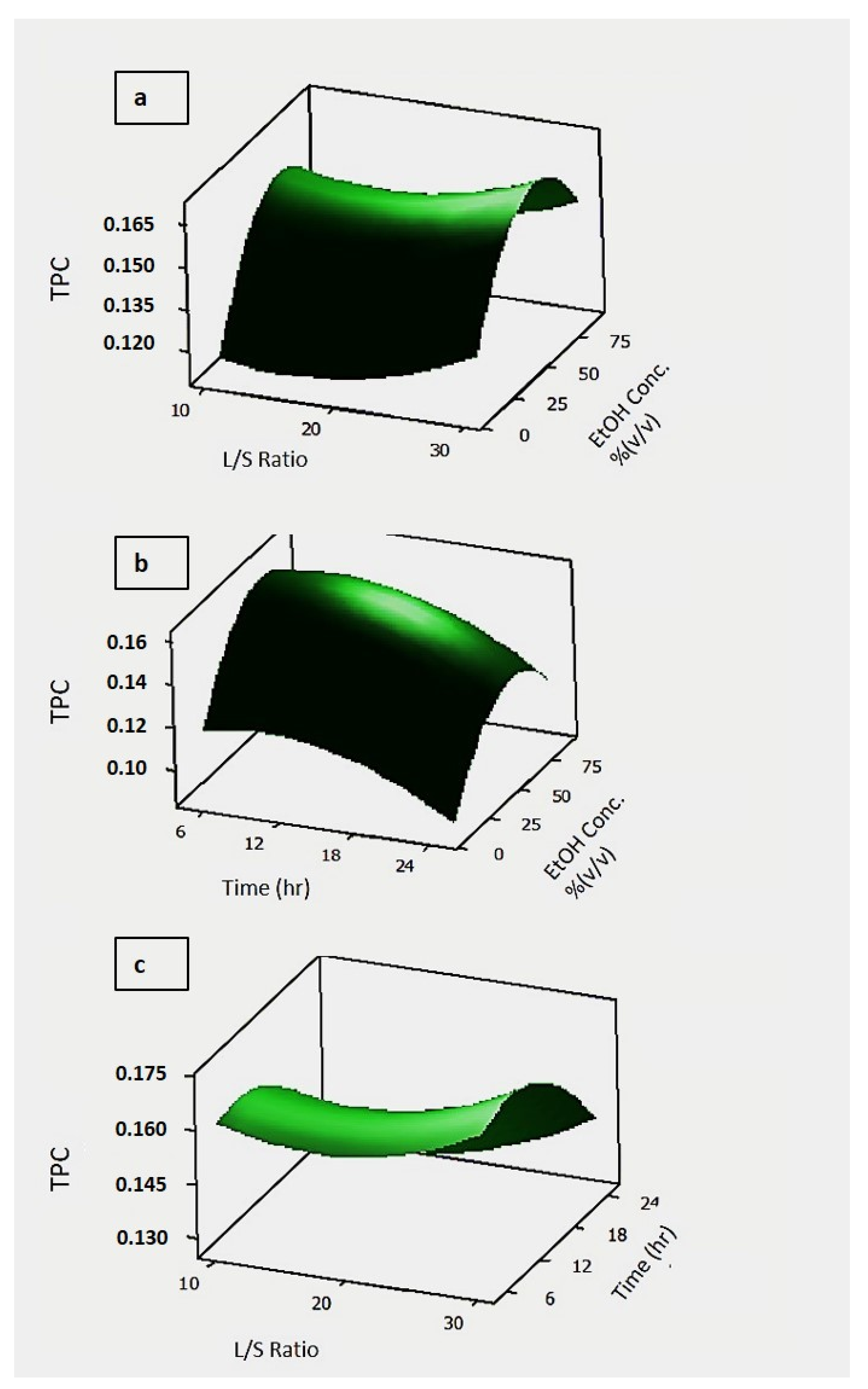
3.2.2. Effect of Process Parameters on the Total Phenolic Content and Antioxidant Capacity
3.3. Characterization of OLE-Loaded ESM
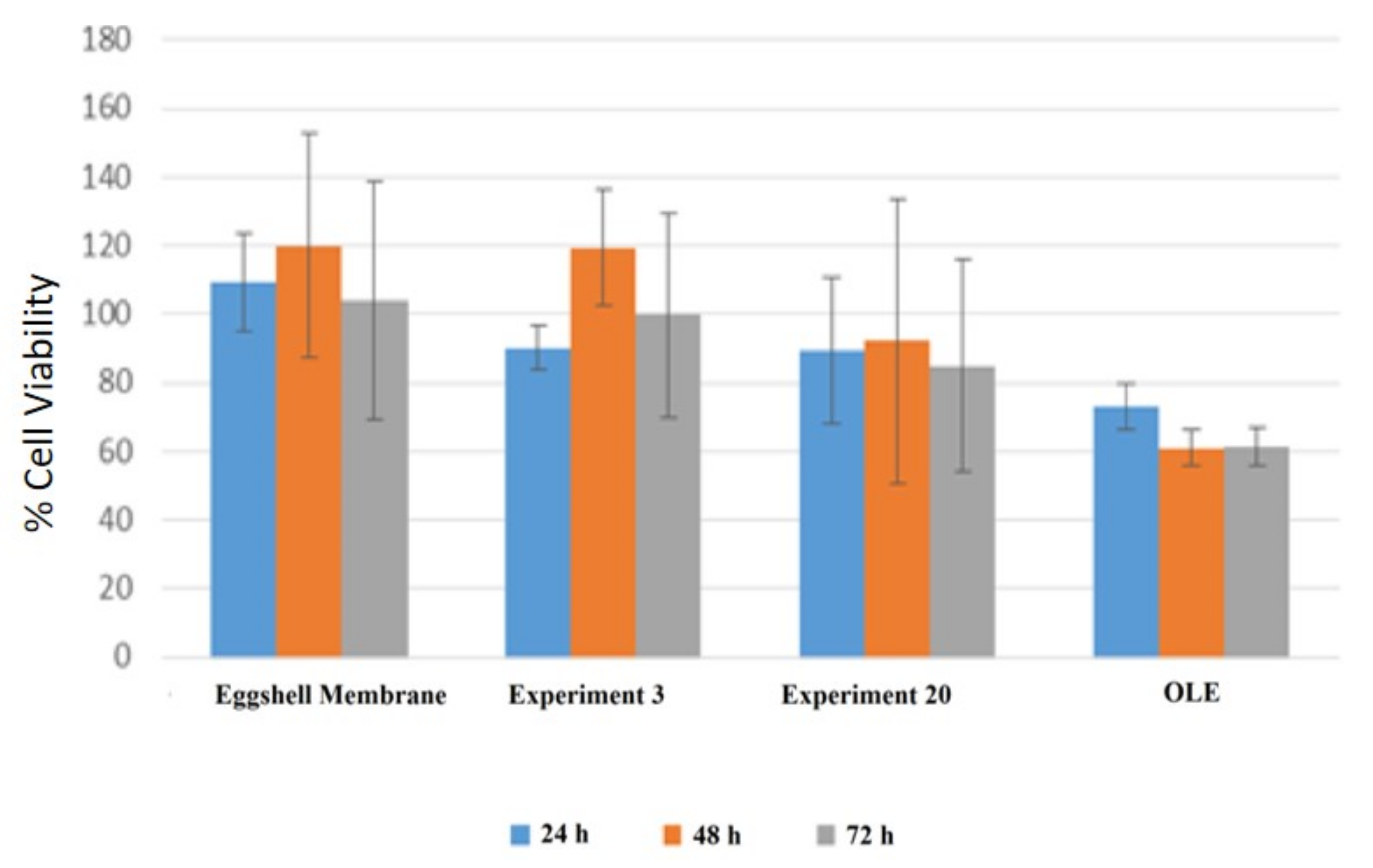
3.4. Cytotoxic Effect of OLE Loaded Eggshell Membrane
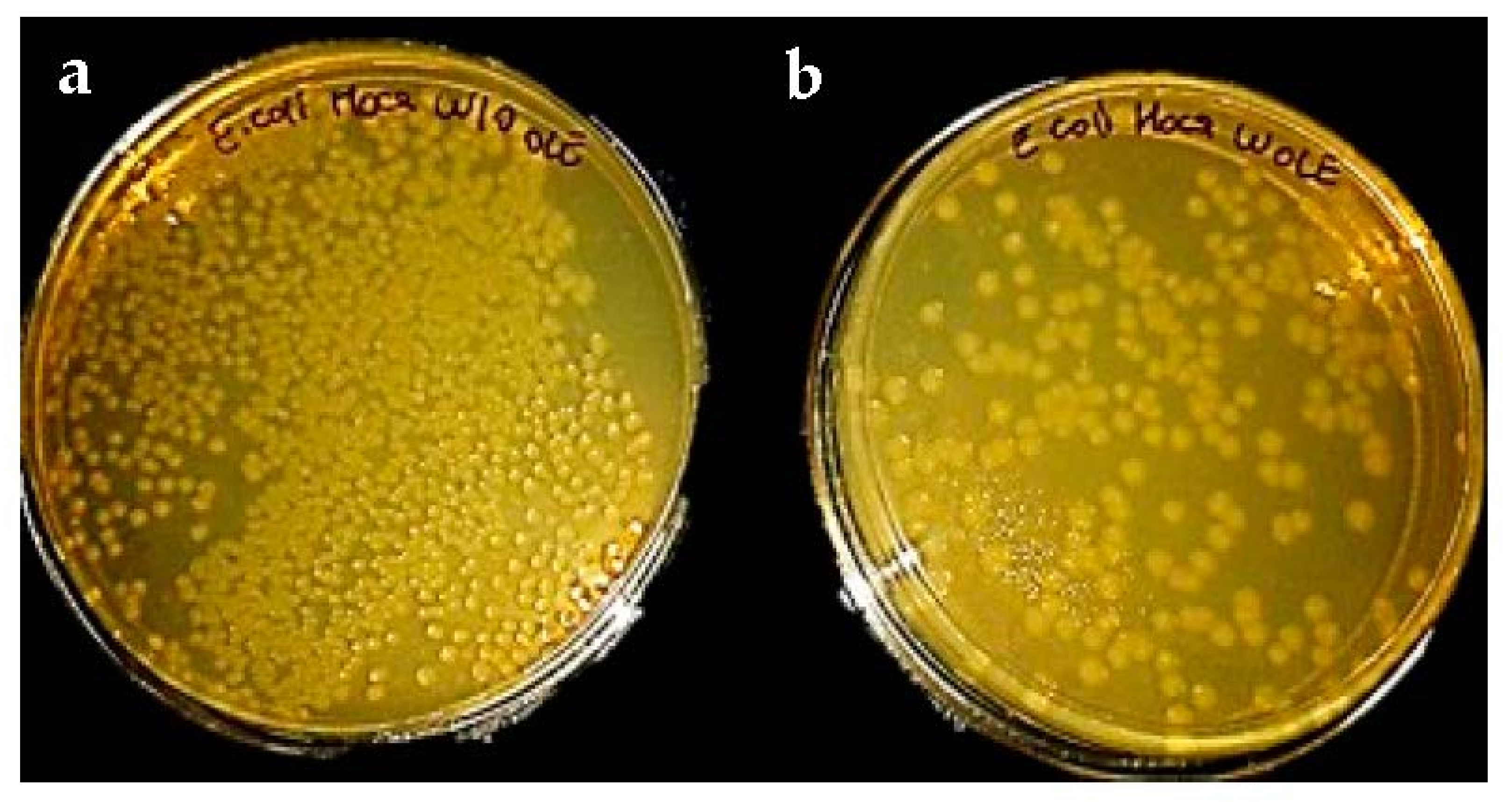
3.5. Antimicrobial Effect of OLE-Loaded Eggshell Membrane
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Amaral, M.C.; Siqueira, F.B.; Destefani, A.Z.; Holanda, J.N.F. Soil-cement bricks incorporated with eggshell waste. Proc. Inst. Civ. Eng. Waste Resour. Manag. 2013, 166, 137–141. [Google Scholar] [CrossRef]
- Pandit, P.R.; Fulekar, M. Egg shell waste as heterogeneous nanocatalyst for biodiesel production: Optimized by response surface methodology. J. Environ. Manag. 2017, 198, 319–329. [Google Scholar] [CrossRef]
- Abdulrahman, I.; Tijani, H.I.; Mohammed, B.A.; Saidu, H.; Yusuf, H.; Jibrin, M.N.; Mohammed, S. From Garbage to Biomaterials: An Overview on Egg Shell Based Hydroxyapatite. J. Mater. 2014, 2014, 802467. [Google Scholar] [CrossRef]
- Girelli, A.M.; Astolfi, M.L.; Scuto, F.R. Agro-industrial wastes as potential carriers for enzyme immobilization: A review. Chemosphere 2020, 244, 125368. [Google Scholar] [CrossRef] [PubMed]
- Sah, M.K.; Rath, S.N. Soluble eggshell membrane: A natural protein to improve the properties of biomaterials used for tissue engineering applications. Mater. Sci. Eng. C 2016, 67, 807–821. [Google Scholar] [CrossRef]
- King’ori, A.M. A Review of the Uses of Poultry Eggshells and Shell Membranes. Int. J. Poult. Sci. 2011, 10, 908–912. [Google Scholar] [CrossRef]
- Köse, M.D. Natural Bioactive Compound Loaded Eggshell Membrane from Waste Food Industry: Preparation and Characterization. Curr. Nutraceuticals 2020, 1, 1–13. [Google Scholar] [CrossRef]
- Park, S.; Choi, K.S.; Lee, D.; Kim, D.; Lim, K.T.; Lee, K.-H.; Seonwoo, H.; Kim, J. Eggshell membrane: Review and impact on engineering. Biosyst. Eng. 2016, 151, 446–463. [Google Scholar] [CrossRef]
- Sudjana, A.N.; D’Orazio, C.; Ryan, V.; Rasool, N.; Ng, J.; Islam, N.; Riley, T.V.; Hammer, K.A. Antimicrobial activity of commercial Olea europaea (olive) leaf extract. Int. J. Antimicrob. Agents 2009, 33, 461–463. [Google Scholar] [CrossRef] [PubMed]
- Korukluoglu, M.; Sahan, Y.; Yigit, A.; Yiǧit, A. Antifungal Properties of Olive Leaf Extracts and Their Phenolic Compounds. J. Food Saf. 2008, 28, 76–87. [Google Scholar] [CrossRef]
- Micol, V.; Caturla, N.; Pérez-Fons, L.; Más, V.; Pérez, L.; Estepa, A. The olive leaf extract exhibits antiviral activity against viral haemorrhagic septicaemia rhabdovirus (VHSV). Antivir. Res. 2005, 66, 129–136. [Google Scholar] [CrossRef]
- Rahmanian, N.; Jafari, S.M.; Galanakis, C.M. Recovery and Removal of Phenolic Compounds from Olive Mill Wastewater. J. Am. Oil Chem. Soc. 2014, 91, 1–18. [Google Scholar] [CrossRef]
- Galanakis, C.M. Phenols recovered from olive mill wastewater as additives in meat products. Trends Food Sci. Technol. 2018, 79, 98–105. [Google Scholar] [CrossRef]
- Galanakis, C.M.; Tsatalas, P.; Galanakis, I.M. Implementation of phenols recovered from olive mill wastewater as UV booster in cosmetics. Ind. Crop. Prod. 2018, 111, 30–37. [Google Scholar] [CrossRef]
- Nunes, M.A.; Pimentel, F.B.; Costa, A.S.; Alves, R.C.; Oliveira, M.B.P. Olive by-products for functional and food applications: Challenging opportunities to face environmental constraints. Innov. Food Sci. Emerg. Technol. 2016, 35, 139–148. [Google Scholar] [CrossRef]
- Romani, A.; Mulas, S.; Heimler, D. Polyphenols and secoiridoids in raw material (Olea europaea L. leaves) and commercial food supplements. Eur. Food Res. Technol. 2016, 243, 429–435. [Google Scholar] [CrossRef]
- Lee, O.-H.; Lee, B.-Y. Antioxidant and antimicrobial activities of individual and combined phenolics in Olea europaea leaf extract. Bioresour. Technol. 2010, 101, 3751–3754. [Google Scholar] [CrossRef] [PubMed]
- Medina, E.; Romero, C.; Garcia-Garcia, P.; Brenes, M. Characterization of bioactive compounds in commercial olive leaf extracts, and olive leaves and their infusions. Food Funct. 2019, 10, 4716–4724. [Google Scholar] [CrossRef]
- Galanakis, C.M. Separation of functional macromolecules and micromolecules: From ultrafiltration to the bor-der of nanofiltration. Trends Food Sci. Technol. 2015, 42, 44–63. [Google Scholar] [CrossRef]
- Galanakis, C.M. Recovery of high added-value components from food wastes: Conventional, emerging technologies and commercialized applications. Trends Food Sci. Technol. 2012, 26, 68–87. [Google Scholar] [CrossRef]
- Galanakis, C.M. Emerging technologies for the production of nutraceuticals from agricultural by-products: A viewpoint of opportunities and challenges. Food Bioprod. Process. 2013, 91, 575–579. [Google Scholar] [CrossRef]
- Roselló-Soto, E.; Barba, F.J.; Parniakov, O.; Galanakis, C.M.; Lebovka, N.; Grimi, N.; Vorobiev, E. High Voltage Electrical Discharges, Pulsed Electric Field, and Ultrasound Assisted Extraction of Protein and Phenolic Compounds from Olive Kernel. Food Bioprocess Technol. 2015, 8, 885–894. [Google Scholar] [CrossRef]
- Galanakis, C.M. The Food Systems in the Era of the Coronavirus (COVID-19) Pandemic Crisis. Foods 2020, 9, 523. [Google Scholar] [CrossRef] [PubMed]
- Galanakis, C.M. Functionality of Food Components and Emerging Technologies. Foods 2021, 10, 128. [Google Scholar] [CrossRef] [PubMed]
- Galanakis, C.M.; Aldawoud, T.M.S.; Rizou, M.; Rowan, N.; Ibrahim, S. Food Ingredients and Active Com-pounds against the Coronavirus Disease (COVID-19) Pandemic: A Comprehensive Review. Foods 2020, 9, 1701. [Google Scholar] [CrossRef]
- Bayraktar, O.; Köse, M.D.; Baspinar, Y. Development of olive leaf extract loaded fibroin microparticles by spray drying. Drug Discov. 2019, 13, 39–45. [Google Scholar]
- Ananey-Obiri, D.; Matthews, L.; Azahrani, M.H.; Ibrahim, S.A.; Galanakis, C.M.; Tahergorabi, R. Applica-tion of Protein-based Edible Coatings for Fat Uptake Reduction in Deep-Fat Fried Foods with an Emphasis on Muscle Food Proteins. Trends Food Sci. Technol. 2018, 80, 167–174. [Google Scholar] [CrossRef]
- Barth, A. Infrared spectroscopy of proteins. Biochim. Biophys. Acta (BBA) Bioenerg. 2007, 1767, 1073–1101. [Google Scholar] [CrossRef]
- Singh, B.R.; DeOliveira, D.B.; Fu, F.-N.; Fuller, M.P. Fourier transform infrared analysis of amide III bands of proteins for the secondary structure estimation. In Biomolecular Spectroscopy III; Nafie, L.A., Mantsch, H.H., Eds.; SPIE: Bellingham, DC, USA, 1993; Volume 1890, pp. 47–55. [Google Scholar] [CrossRef]
- Tsai, W.; Yang, J.; Lai, C.; Cheng, Y.; Lin, C.; Yeh, C. Characterization and adsorption properties of eggshells and eggshell membrane. Bioresour. Technol. 2006, 97, 488–493. [Google Scholar] [CrossRef]
- Altıok, E.; Bayçın, D.; Bayraktar, O.; Ülkü, S. Isolation of polyphenols from the extracts of olive leaves (Olea europaea L.) by adsorption on silk fibroin. Sep. Purif. Technol. 2008, 62, 342–348. [Google Scholar] [CrossRef]
- Bayçın, D.; Altıok, E.; Ülkü, S.; Bayraktar, O.; Baycin, D. Adsorption of Olive Leaf (Olea europaea L.) Antioxidants on Silk Fibroin. J. Agric. Food Chem. 2007, 55, 1227–1236. [Google Scholar] [CrossRef]
- Talhaoui, N.; Taamalli, A.; Gómez-Caravaca, A.M.; Fernández-Gutiérrez, A.; Segura-Carretero, A. Phenolic compounds in olive leaves: Analytical determination, biotic and abiotic influence, and health benefits. Food Res. Int. 2015, 77, 92–108. [Google Scholar] [CrossRef]
- Basal, G.; Tetik, G.D.; Kurkcu, G.; Bayraktar, O.; Gurhan, I.D.; Atabey, A. Olive leaf extract loaded silk fibro-in/hyaluronic acid nanofiber webs for wound dressing applications. Dig. J. Nanomater. Biostruct. 2016, 11, 1113–1123. [Google Scholar]
- Stuart, B.H. Infrared Spectroscopy Fundamentals and Applications; John Wiley and Sons Ltd.: Hoboken, NJ, USA, 2004; pp. 47–80. [Google Scholar]
- Tang, H.R.; Covington, A.D.; Hancock, R.A. Structure-activity relationships in the hydrophobic ınteractions of polyphenols with cellulose and collagen. Biopolymers 2003, 70, 403–413. [Google Scholar] [CrossRef] [PubMed]
- Liao, X.P.; Lu, Z.B.; Shi, B. Selective Adsorption of Vegetable Tannins onto Collagen Fi-bers. Ind. Eng. Chem. Res. 2003, 42, 3397–3402. [Google Scholar] [CrossRef]
- Zhao, Y.-H.; Chi, Y.-J. Characterization of Collagen from Eggshell Membrane. Biotechnology 2009, 8, 254–258. [Google Scholar] [CrossRef]


| Independent Variable | Unit | Factor Symbol | Coded Levels | |
|---|---|---|---|---|
| −1 | 1 | |||
| Acetic acid concentration | % | A | 0 | 100 |
| Average eggshell size | mm | B | 0.5 | 2 |
| Treatment time in the column | h | C | 1 | 5 |
| Air flow rate | cm/s | D | 0.1 | 0.5 |
| Exp No | Acetic Acid Concentration % (v/v) | Average Eggshell Size (mm) | Time in the Column (h) | Flow Rate of the Air in the Column (cm/s) | Eggshell and Membrane Separation Efficiency (%) |
|---|---|---|---|---|---|
| 1 | 0 | 0.5 | 1 | 0.1 | 45 |
| 2 | 100 | 0.5 | 1 | 0.1 | 66 |
| 3 | 0 | 2.0 | 1 | 0.1 | 38 |
| 4 | 100 | 2.0 | 1 | 0.1 | 68 |
| 5 | 0 | 0.5 | 5 | 0.1 | 42 |
| 6 | 100 | 0.5 | 5 | 0.1 | 77 |
| 7 | 0 | 2.0 | 5 | 0.1 | 36 |
| 8 | 100 | 2.0 | 5 | 0.1 | 65 |
| 9 | 0 | 0.5 | 1 | 0.5 | 40 |
| 10 | 100 | 0.5 | 1 | 0.5 | 69 |
| 11 | 0 | 5.0 | 1 | 0.5 | 38 |
| 12 | 100 | 5.0 | 1 | 0.5 | 71 |
| 13 | 0 | 0.5 | 5 | 0.5 | 42 |
| 14 | 100 | 0.5 | 5 | 0.5 | 70 |
| 15 | 0 | 2.0 | 5 | 0.5 | 38 |
| 16 | 100 | 2.0 | 5 | 0.5 | 81 |
| 17 | 0 | 1.25 | 3 | 0.3 | 34 |
| 18 | 100 | 1.25 | 3 | 0.3 | 74 |
| 19 | 50 | 0.5 | 3 | 0.3 | 43 |
| 20 | 50 | 2.0 | 3 | 0.3 | 47 |
| 21 | 50 | 1.25 | 1 | 0.3 | 45 |
| 22 | 50 | 1.25 | 5 | 0.3 | 56 |
| 23 | 50 | 1.25 | 3 | 0.1 | 44 |
| 24 | 50 | 1.25 | 3 | 0.5 | 40 |
| 25 | 50 | 1.25 | 3 | 0.3 | 47 |
| 26 | 50 | 1.25 | 3 | 0.3 | 34 |
| 27 | 50 | 1.25 | 3 | 0.3 | 45 |
| 28 | 50 | 1.25 | 3 | 0.3 | 46 |
| 29 | 50 | 1.25 | 3 | 0.3 | 47 |
| 30 | 50 | 1.25 | 3 | 0.3 | 38 |
| 31 | 50 | 1.25 | 3 | 0.3 | 40 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bayraktar, O.; Galanakis, C.M.; Aldawoud, T.M.S.; Ibrahim, S.A.; Köse, M.D.; Uslu, M.E. Utilization of Eggshell Membrane and Olive Leaf Extract for the Preparation of Functional Materials. Foods 2021, 10, 806. https://doi.org/10.3390/foods10040806
Bayraktar O, Galanakis CM, Aldawoud TMS, Ibrahim SA, Köse MD, Uslu ME. Utilization of Eggshell Membrane and Olive Leaf Extract for the Preparation of Functional Materials. Foods. 2021; 10(4):806. https://doi.org/10.3390/foods10040806
Chicago/Turabian StyleBayraktar, Oguz, Charis M. Galanakis, Turki M. S. Aldawoud, Salam A. Ibrahim, Merve Deniz Köse, and Mehmet Emin Uslu. 2021. "Utilization of Eggshell Membrane and Olive Leaf Extract for the Preparation of Functional Materials" Foods 10, no. 4: 806. https://doi.org/10.3390/foods10040806
APA StyleBayraktar, O., Galanakis, C. M., Aldawoud, T. M. S., Ibrahim, S. A., Köse, M. D., & Uslu, M. E. (2021). Utilization of Eggshell Membrane and Olive Leaf Extract for the Preparation of Functional Materials. Foods, 10(4), 806. https://doi.org/10.3390/foods10040806









