1. Introduction
Prevention of type 2 diabetes mellitus (T2DM) has become a major health challenge given its high global prevalence, which has reached 8.5%, according to the latest report on diabetes published by the World Health Organization (WHO) in 2016 [
1]. Impaired glucose regulation (IGR), also known as prediabetes, including impaired glucose tolerance (IGT) and/or impaired fasting glycemia (IFG), are insidious intermediate conditions before the occurrence of T2DM [
2]. Considering China’s high prevalence of IGR, which has reached 35.7% (95% confidence interval: 34.1–37.4%) in 2013 and been maintained at 35.2% (95% confidence interval: 33.5–37.0%) in 2017 [
3,
4], it is of practical significance in preventing the conversion of prediabetes to T2DM.
Dietary and/or another lifestyle intervention has been listed as effective strategies in the international guidelines on the management of prediabetes [
5,
6,
7]. As a kind of food rich in bioactive compounds, berries are considered “promising functional fruits” and have been widely studied in recent years [
8,
9,
10]. A systematic review and meta-analysis of prospective cohort studies showed that consumption of berries was associated with an 18% reduced risk of T2DM [
11]. The incorporation of berries or berries-derived byproducts into high-carbohydrate (HCD) and high-fat (HFD) diets, has also been found to contribute to the reversion/reduction of the HCD/HFD-induced alterations in glucose metabolism-related pathways and inflammation in diabetic subjects [
8].
Hippophae rhamnoides L., commonly referred to as sea buckthorn (SB), belongs to the family Elaeagnaceae and genus
Hippophae [
12]. At present, increasing attention on the possible positive effects of SB berries for glycemic control was also noted. Several animal studies have shown positive effects of SB protein/fruit oil extract on reducing blood glucose, as well as alleviating insulin resistance [
13,
14,
15]. In human trials, for example, compared with strawberry, SB decreased and delayed insulin response and improved glycemic profile following a sucrose-containing berry meal [
16]. In another randomized study, a daily dose of 100 g fresh SB reduced fasting plasma glucose (FPG) after intervention for 33–35 days, although the proportion of glycated hemoglobin A1c (HbA1c) was increased contrarily [
9]. Elsewhere, 40 g of dried SB together with yogurt (200 g) and glucose (50 g) has been suggested to stabilize postprandial hyperglycemia and suppress peak insulin response [
17].
As reviewed by Padwad et al., all parts of SB have been found to be a rich source of bioactive substances, such as fat-soluble vitamins (A, E, and K) and flavonoids (quercetin, kaempferol, isorhamnetin, myricetin) [
18]. The flavonoids, in particular, have been indicated to potentially exhibit hypoglycemic activities through reducing glucose absorption, enhancing insulin secretion and sensitivity, regulating hepatic glucose output, among others [
19,
20]. In addition, SB may also play a role in regulating neuroendocrine, immune, antioxidant, and anti-inflammatory functions [
21,
22,
23], which might contribute to the control of blood glucose.
Although the studies above showed positive effects of SB on glycemic control, it should be noted that most of these studies were conducted in overweight and/or obese subjects [
9,
16]. Furthermore, previous studies have been carried out mainly in Europe, using European SB berries as the intervention [
16,
17]. According to records, SB has a long history of cultivation and edible or medicinal use in China [
24]. In addition, five of the nine subspecies of SB are distributed in China, which suggests great research potential [
25]. Recently, a previous double-blind, randomized, placebo-controlled trial conducted by our team showed that the high-sensitivity C-reactive protein concentration was decreased in the SB group in patients with hypercholesterolemia, indicating an anti-inflammatory effect [
26]. However, the possible benefits of SB in IGR subjects are still unknown.
Thus, this study aimed to assess the effects of SB grown in China on glycemia indicators, including FPG, postprandial plasma glucose (PPG) and glycated serum protein (GSP) in subjects with IGR.
4. Discussion
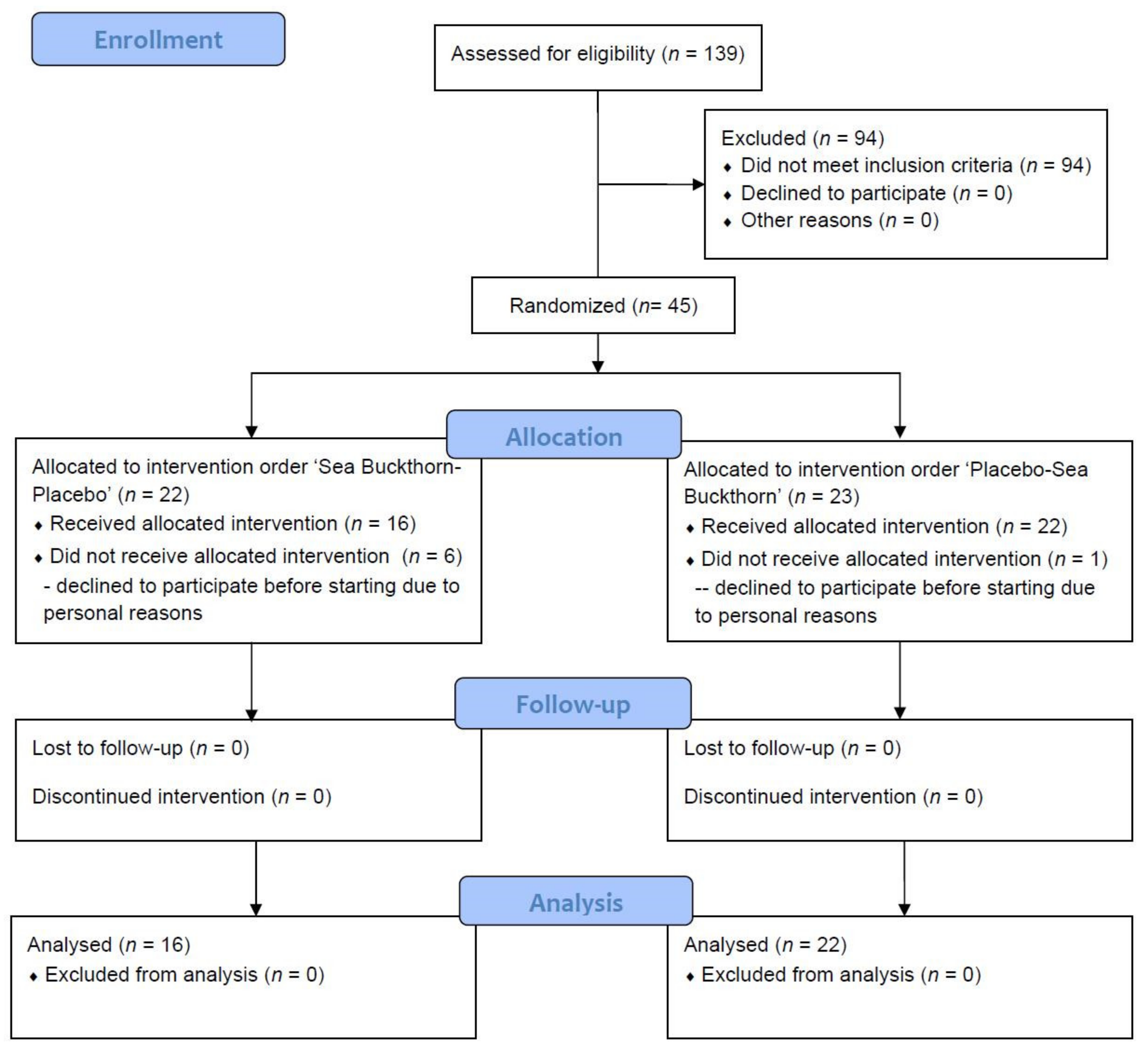
According to the screening data, the detection rate of IGR was 32.4% (45/139), which was close to the prevalence of 35.2% reported in a nationally representative cross-sectional survey in China in 2017 [
4]. Considering that IGR contributes to a high risk of type 2 diabetes and other metabolic diseases [
2,
32,
33], it is crucial to investigate more effective prevention strategies [
34,
35]. The positive effect of functional foods rich in bioactive ingredients on preventing and managing chronic diseases, for example, is commonly accepted [
7,
36]. In the present study, consumption of 90 mL SBFP for 35 days led to a tendency for FPG to decrease in people with IGR, but did not affect the PPG or GSP.
To date, evidence of the ability of SB to alter FPG is insufficient and mainly produced by animal trials. Consistent with the positive effect found in this study, Lehtonen et al. reported that there was a small, but significant, decrease in FPG (−0.1 mmol/L;
p = 0.002) after the inclusion of a certain amount of air-dried SB (equivalent to 100 g/day fresh berries) for 33–35 days in the diet of 110 overweight and obese women [
9]. In a recent animal trial, after a four-week treatment period (100–200 mg/kg/day), the FPG of diabetic mice was significantly reduced in the SB seed protein (SSP) and SB polysaccharide (SPO) group compared with the module control group, whereas SB procyanidins (SPR) showed no effect [
14]. Administration of flavonoids from sea buckthorn pomace (SBP) for four weeks also resulted in significantly hypoglycemic effects in ICR mice with alloxan-induced diabetes, even in the low dose group (3.0 mg/kg/day) [
37]. In the study by Gao et al., it was suggested that a middle or high dose of sea buckthorn fruit oil (SBFO, 200 or 300 mg/kg/day) could lower the FPG at a rate of 10.47% and 13.79% in T2DM SD rats, respectively [
13]. In addition, the continuous intervention of methanolic extract from
Hippophae salicifolia D.Don (a species of SB) leaves at 200 or 400 mg/kg for 45 days both exhibited significant reduction (22% and 39%, respectively) in FPG compared to the diabetic control rats [
38].
Prior research suggested that SB and its extracts might have a hypoglycemic effect. However, the mechanism of this effect was unclear. Protein included in SB is considered to be a high-quality resource of essential and semi-essential amino acids [
14]. A study by Yuan et al. confirmed an increase of hypoglycemic-related beneficial bacteria (
Bifidobacterium and
Lactobacillus) in diabetic mice with consumption of SSP, which could be due to the higher concentrations of short-chain fatty acids and lower pH produced by the metabolism of SSP [
39]. An in vitro experiment conducted in insulin-resistant HepG2 cells revealed that the SBFO extract rich in palmitoleic acid (POA) could increase the expression of glucose transporter type 4 through the activation of the PI3K/Akt signaling pathway [
2]. In addition to the macronutrients mentioned above, evidence accumulated over the years indicates that biologically active substances rich in SB may also play an important role, especially polyphenols. The mechanisms involved in relevant studies, including the inhibition of glucose absorption, stimulation of insulin secretion and activation of insulin receptors, modulation of hepatic glucose output, etc. [
7,
19,
20]. Based on these studies, abundant unsaturated fatty acids (e.g., POA) and flavonoids (especially Isorhamnetin and Quercetin, see
Table 1) in the SB we used in this study may explain its positive role in hypoglycemic.
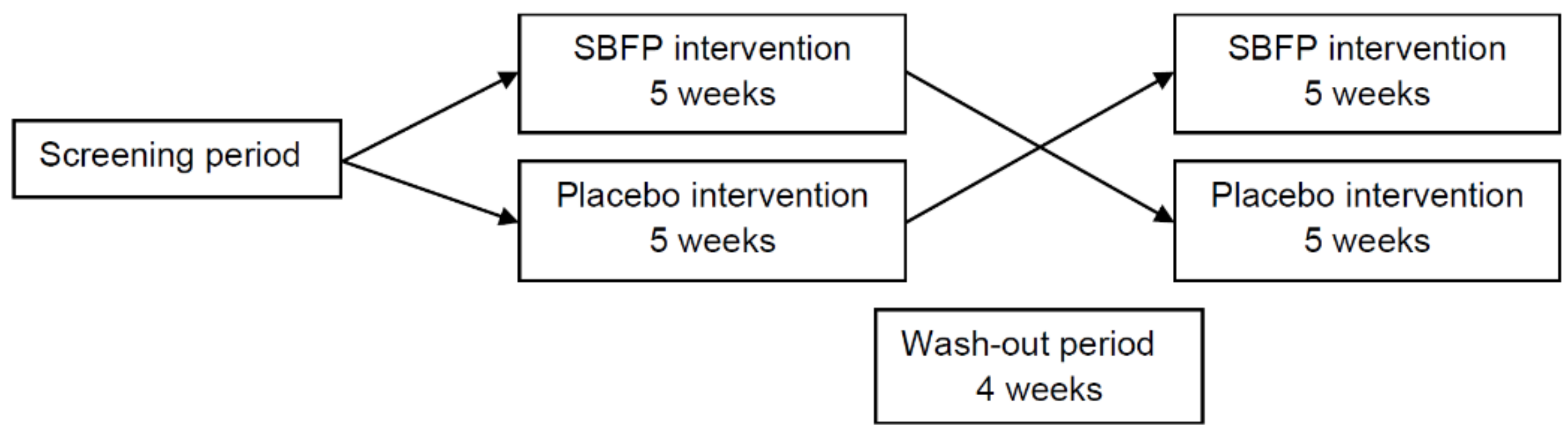
In line with expectations, the intervention of SBFP at five weeks showed a positive effect on FPG. However, during the wash-out period, subjects who received SBFP also had a continued decline of FPG, which might suggest long-term effects. Since the statistic carryover effects were not significant in our study, the wash-out period set in our study was considered reasonable. In future studies, the intervention can be extended appropriately, and the long-term effects of SB need to be examined.
When it comes to PPG, the results of previous studies were not completely consistent. The study by Gao et al. showed that four weeks’ treatment of SBFO extracts improved the oral glucose tolerance test (OGTT) in T2DM SD rats in a dose-dependent manner [
13]. A study based on ten healthy volunteers consuming one control meal and three experimental meals with dried and crushed whole SB berries, supercritical fluid (SF)-carbon dioxide (CO2)-extracted oil-free berries, and ethanol-extracted SF-CO2-extraction residue, respectively, suggested that meal with dried and crushed whole berries stabilize postprandial hyperglycemia [
17]. However, in the study by Mortensen et al., no difference between control and SB was observed for PPG following a sucrose-containing berry meal [
16]. It needs to be noted that, distinct from the above studies, we measured PPG after a standard meal without SBFP, so the effect we detected was not immediate, and further studies are still needed.
To date, there are few studies concerning the effect of SB on HbA1c or GSP, which reflects the average PG level over three months or 2–3 weeks, respectively. Nemes-Nagy et al. treated 30 type 1 diabetic children with a dietary supplement containing blueberry and SB for two months, and levels of HbA1c were significantly lowered in this group [
40]. However, it is hard to distinguish whether the effects are a consequence of SB or blueberry treatment. Regretfully, the intervention of our study was not long enough to reflect the status of blood glucose in the last three months. But to the best of our knowledge, this is the first study to report the effect of SB on GSP. Reflecting a short-term glycemic regulation, GSP increased in the intervention period, but decreased in the wash-out period, and it seems to have no significant relevance to different interventions.
Previous studies indicated that berries may help improve appetite control, due to their fiber content, which in turn facilitates the control of weight [
41]. An increased desire for something sweet was observed in the study by Mortensen et al. after administering a test meal containing added sucrose and SB [
16]. In the present study, no differences for SB or placebo were observed for any nutritional intakes. However, whether the one-off 24 h food recall represents relevant nutrient intake is unclear. The effects of SB on appetite or dietary intakes, and its association with PG, need further study.
The strength of this study lies in the crossover design and the zero-dropout rate after the start of this study. To our knowledge, this is the first study to use SBFP for patients with IGR, and we evaluated relatively comprehensive glycemic markers, including FPG, 2 h PG-AUC and GSP. However, there are some limitations that should be considered. The distribution of genders was not balanced—although most women in our study had reached menopause, the differences in biology and physiology between men and women cannot be ignored. Furthermore, we did not provide long-term follow-up to detect the HbA1c and the progression from IGR to diabetes or normal glucose regulation, highlighting potential directions for future research.