Resistant Starch Combined with Whey Protein Increases Postprandial Metabolism and Lowers Glucose and Insulin Responses in Healthy Adult Men
Abstract
1. Introduction
2. Materials and Methods
2.1. Participants
2.2. Pancake Test Meal
2.3. Experimental Design
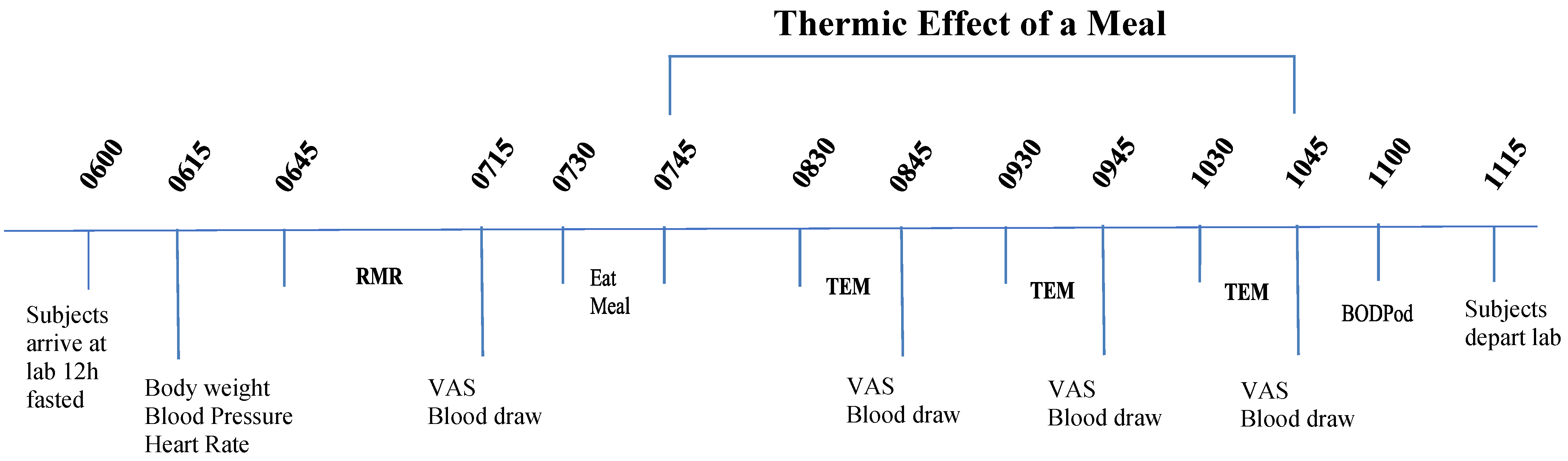
2.4. Resting Metabolic Rate (RMR) and Thermic Effect of a Meal (TEM)
2.5. Plasma Biomarkers
2.6. Feelings of Hunger, Satiation, and Desire to Eat
2.7. Heart Rate and Blood Pressure
2.8. Statistical Analyses
3. Results
3.1. Participants and Compliance
3.2. Assessment of Energy Intake
3.3. Resting Metabolic Rate, Thermic Effect of a Meal, and Substrate Utilization
3.4. Respiratory Exchange Ratio
3.5. Plasma Biomarkers
3.6. Feelings of Hunger, Satiation and Desire to Eat
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| AUC | area under the curve |
| BMI | body mass index |
| GIP | gastric inhibitory peptide |
| GLP-1 | glucagon-like polypeptide-1 |
| GRL | ghrelin |
| LEP | leptin |
| PYY | peptide YY |
| RER | respiratory exchange ratio |
| RMR | resting metabolic rate |
| RS4 | resistance starch, type 4 |
| RS | resistant starch, type 4 |
| RS + WP | resistant starch and whey protein |
| SCFA | short-chain fatty acid |
| TEM | thermic effect of a meal |
| VAS | visual analog scale |
| WMS | waxy maize starch |
| WMS + WP | waxy maize starch and whey protein |
References
- Sun, N.N.; Wu, T.Y.; Chau, C.F. Natural dietary and herbal products in anti-obesity treatment. Molecules 2016, 21, 1351. [Google Scholar] [CrossRef]
- Suh, J.H.; Wang, Y.; Ho, C.T. Natural dietary products and their effects on appetite control. J. Agric. Food Chem. 2018, 66, 36–39. [Google Scholar] [CrossRef]
- Higgins, J.A. Resistant starch and energy balance: Impact on weight loss and maintenance. Crit. Rev. Food Sci. Nutr. 2014, 54, 1158–1166. [Google Scholar] [CrossRef]
- Arciero, P.J.; Ormsbee, M.J.; Gentile, C.L.; Nindl, B.C.; Brestoff, J.R.; Ruby, M. Increased protein intake and meal frequency reduces abdominal fat during energy balance and energy deficit. Obesity 2013, 21, 1357–1366. [Google Scholar] [CrossRef]
- Arciero, P.J.; Edmonds, R.; He, F.; Ward, E.; Gumpricht, E.; Mohr, A.; Ormsbee, M.J.; Astrup, A. Protein-pacing caloric-restriction enhances body composition similarly in obese men and women during weight loss and sustains efficacy during long-term weight maintenance. Nutrients 2016, 8, 476. [Google Scholar] [CrossRef] [PubMed]
- Gentile, C.L.; Ward, E.; Holst, J.J.; Astrup, A.; Ormsbee, M.J.; Connelly, S.; Arciero, P.J. Resistant starch and protein intake enhances fat oxidation and feelings of fullness in lean and overweight/obese women. Nutr. J. 2015, 14, 113. [Google Scholar] [CrossRef] [PubMed]
- Birt, D.F.; Boylston, T.; Hendrich, S.; Jane, J.L.; Hollis, J.; Li, L.; McClelland, J.; Moore, S.; Phillips, G.J.; Rowling, M.; et al. Resistant starch: Promise for improving human health. Adv. Nutr. 2013, 4, 587–601. [Google Scholar] [CrossRef] [PubMed]
- Lerer-Metzger, M.; Rizkalla, S.W.; Luo, J.; Champ, M.; Kabir, M.; Bruzzo, F.; Bornet, F.; Slama, G. Effects of long-term low-glycaemic index starchy food on plasma glucose and lipid concentrations and adipose tissue cellularity in normal and diabetic rats. Br. J. Nutr. 1996, 75, 723–732. [Google Scholar] [CrossRef]
- Keenan, M.J.; Zhou, J.; McCutcheon, K.L.; Raggio, A.M.; Bateman, H.G.; Todd, E.; Jones, C.K.; Tulley, R.T.; Melton, S.; Martin, R.J.; et al. Effects of resistant starch, a non-digestible fermentable fiber, on reducing body fat. Obesity 2006, 14, 1523–1534. [Google Scholar] [CrossRef] [PubMed]
- So, P.W.; Yu, W.S.; Kuo, Y.T.; Wasserfall, C.; Goldstone, A.P.; Bell, J.D.; Frost, G. Impact of resistant starch on body fat patterning and central appetite regulation. PLoS ONE 2007, 2, e1309. [Google Scholar] [CrossRef]
- Higgins, J.A.; Jackman, M.R.; Brown, I.L.; Johnson, G.C.; Steig, A.; Wyatt, H.R.; Hill, J.O.; MacLean, P.S. Resistant starch and exercise independently attenuate weight regain on a high fat diet in a rat model of obesity. Nutr. Metab. 2011, 8, 49. [Google Scholar] [CrossRef] [PubMed]
- Emilien, C.H.; Hsu, W.H.; Hollis, J.H. Effect of resistant wheat starch on subjective appetite and food intake in healthy adults. Nutrition 2017, 43, 69–74. [Google Scholar] [CrossRef]
- Sandberg, J.C.; Björck, I.M.E.; Nilsson, A.C. Effects of whole grain rye, with and without resistant starch type 2 supplementation, on glucose tolerance, gut hormones, inflammation and appetite regulation in an 11–14.5 hour perspective; a randomized controlled study in healthy subjects. Nutr. J. 2017, 16, 25. [Google Scholar] [CrossRef]
- Al-Mana, N.M.; Robertson, M.D. Acute effect of resistant starch on food intake, appetite and satiety in overweight/obese males. Nutrients 2018, 10, 1993. [Google Scholar] [CrossRef]
- Tapsell, L.C. Diet and metabolic syndrome: Where does resistant starch fit in? J. AOAC Int. 2004, 87, 756–760. [Google Scholar] [CrossRef]
- Zhou, J.; Martin, R.J.; Tulley, R.T.; Raggio, A.M.; McCutcheon, K.L.; Shen, L.; Danna, S.C.; Tripathy, S.; Hegsted, M.; Keenan, M.J. Dietary resistant starch upregulates total GLP-1 and PYY in a sustained day-long manner through fermentation in rodents. Am. J. Physiol. Endocrinol. Metab. 2008, 295, E1160–E1166. [Google Scholar] [CrossRef]
- Guo, J.; Tan, L.; Kong, L. Impact of dietary intake of resistant starch on obesity and associated metabolic profiles in human: A systematic review of the literature. Crit. Rev. Food Sci. Nutr. 2020, 23, 1–17. [Google Scholar] [CrossRef]
- Englyst, H.N.; Cummings, J.H. Resistant starch, a “new” food component: A classification of starch for nutritional purposes. In Cereals in a European Context; Morton, I.D., Ed.; Ellis Horwood: Chichester, UK, 1987; pp. 221–233. [Google Scholar]
- Shimotoyodome, A.; Suzuki, J.; Kameo, Y.; Hase, T. Dietary supplementation with hydroxypropyl-distarch phosphate from waxy maize starch increases resting energy expenditure by lowering the postprandial glucose-dependent insulinotropic polypeptide response in human subjects. Br. J. Nutr. 2011, 106, 96–104. [Google Scholar] [CrossRef]
- Stewart, M.L.; Wilcox, M.L.; Bell, M.; Buggia, M.A.; Maki, K.C. Type-4 resistant starch in substitution for available carbohydrate reduces postprandial glycemic response and hunger in acute, randomized, double-blind, controlled study. Nutrients 2018, 10, 129. [Google Scholar] [CrossRef] [PubMed]
- Wirunsawanya, K.; Upala, S.; Jaruvongvanich, V.; Sanguankeo, A. Whey protein supplementation improves body composition and cardiovascular risk factors in overweight and obese patients: A systematic review and meta-analysis. J. Am. Coll. Nutr. 2018, 37, 60–70. [Google Scholar] [CrossRef] [PubMed]
- Arciero, P.J.; Baur, D.; Connelly, S.; Ormsbee, M.J. Timed-daily ingestion of whey protein and exercise training reduces visceral adipose tissue mass and improves insulin resistance: The PRISE study. J. Appl. Physiol. 2014, 117, 1–10. [Google Scholar] [CrossRef]
- Belza, A.; Ritz, C.; Sørensen, M.Q.; Holst, J.J.; Rehfeld, J.F.; Astrup, A. Contribution of gastroenteropancreatic appetite hormones to protein-induced satiety. Am. J. Clin. Nutr. 2013, 97, 980–989. [Google Scholar] [CrossRef] [PubMed]
- Mollahosseini, M.; Shab-Bidar, S.; Rahimi, M.H.; Djafarian, K. Effect of whey protein supplementation on long and short term appetite: A meta-analysis of randomized controlled trials. Clin. Nutr. ESPEN 2017, 20, 34–40. [Google Scholar] [CrossRef]
- Arciero, P.J.; Ormsbee, M.J. Relationship of blood pressure, behavioral mood state, and physical activity following caffeine ingestion in younger and older women. Appl. Physiol. Nutr. Metab. 2009, 34, 754–762. [Google Scholar] [CrossRef]
- Arciero, P.J.; Gentile, C.L.; Pressman, R.; Everett, M.; Ormsbee, M.J.; Martin, J.; Santamore, L.G.; Fehling, P.C.; Vukovich, M.D.; Nindl, B.C. Moderate protein intake improves total and regional body composition and insulin sensitivity in overweight adults. Metabolism 2008, 57, 757–765. [Google Scholar] [CrossRef]
- Haub, M.D.; Hubach, K.L.; Al-Tamimi, E.K.; Ornelas, S.; Seib, P.A. Different types of resistant starch elicit different glucose responses in humans. J. Nutr. Metab. 2010, 2010, 230501. [Google Scholar] [CrossRef] [PubMed]
- Mah, E.; Garcia-Campayo, V.; Liska, D. Substitution of corn starch with resistant starch type 4 in a breakfast bar decreases postprandial glucose and insulin responses: A randomized, controlled, crossover study. Curr. Dev. Nutr. 2018, 2, nzy066. [Google Scholar] [CrossRef]
- Stewart, M.L.; Zimmer, J.P. A high fiber cookie made with resistant starch type 4 reduces post-prandial glucose and insulin responses in healthy adults. Nutrients 2017, 9, 237. [Google Scholar] [CrossRef] [PubMed]
- Stewart, M.L.; Zimmer, J.P. Postprandial glucose and insulin response to a high-fiber muffin top containing resistant starch type 4 in healthy adults: A double-blind, randomized, controlled trial. Nutrition 2018, 53, 59–63. [Google Scholar] [CrossRef]
- Jiang, J.; Zhao, L.; Lin, L.; Gui, M.; Aleteng, Q.; Wu, B.; Wang, S.; Pan, B.; Ling, Y.; Gao, X. Postprandial blood glucose outweighs fasting blood glucose and HbA1c in screening coronary heart disease. Sci. Rep. 2017, 7, 14212. [Google Scholar] [CrossRef]
- Ferguson, L.R.; Tasman-Jones, C.; Englyst, H.; Harris, P.J. Comparative effects of three resistant starch preparations on transit time and short-chain fatty acid production in rats. Nutr. Cancer 2000, 36, 230–237. [Google Scholar] [CrossRef]
- Upadhyaya, B.; McCormack, L.; Fardin-Kia, A.R.; Juenemann, R.; Nichenametla, S.; Clapper, J.; Specker, B.; Dey, M. Impact of dietary resistant starch type 4 on human gut microbiota and immunometabolic functions. Sci. Rep. 2016, 6, 28797. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Upadhyaya, B.; Fardin-Kia, A.R.; Juenemann, R.M.; Dey, M. Dietary resistant starch type 4-derived butyrate attenuates nuclear factor-kappa-B1 through modulation of histone H3 trimethylation at lysine 27. Food Funct. 2016, 7, 3772–3781. [Google Scholar] [CrossRef]
- Elliott, R.M.; Morgan, L.M.; Tredger, J.A.; Deacon, S.; Wright, J.; Marks, V. Glucagon-like peptide-1 (7–36)amide and glucose-dependent insulinotropic polypeptide secretion in response to nutrient ingestion in man: Acute post-prandial and 24-h secretion patterns. J. Endocrinol. 1993, 138, 159–166. [Google Scholar] [CrossRef]
- Meier, J.J.; Nauck, M.A. Clinical endocrinology and metabolism. Glucose-dependent insulinotropic polypeptide/gastric inhibitory polypeptide. Best Pract. Res. Clin. Endocrinol. Metab. 2004, 18, 587–606. [Google Scholar] [CrossRef]
- Shimotoyodome, A.; Fukuoka, D.; Suzuki, J.; Fujii, Y.; Mizuno, T.; Meguro, S.; Tokimitsu, I.; Hase, T. Coingestion of acylglycerols differentially affects glucose-induced insulin secretion via glucose-dependent insulinotropic polypeptide in C57BL/6J mice. Endocrinology 2009, 150, 2118–2126. [Google Scholar] [CrossRef]
- Irwin, N.; Flatt, P.R. Therapeutic potential for GIP receptor agonists and antagonists. Best Pract. Res. Clin. Endocrinol. Metab. 2009, 23, 499–512. [Google Scholar] [CrossRef] [PubMed]
- Song, D.H.; Wolfe, M.M. Glucose-dependent insulinotropic polypeptide and its role in obesity. Curr. Opin. Endocrinol. Diabetes Obes. 2007, 14, 46–51. [Google Scholar] [CrossRef] [PubMed]
- Althage, M.C.; Ford, E.L.; Wang, S.; Tso, P.; Polonsky, K.S.; Wice, B.M. Targeted ablation of glucose-dependent insulinotropic polypeptide-producing cells in transgenic mice reduces obesity and insulin resistance induced by a high fat diet. J. Biol. Chem. 2008, 283, 18365–18376. [Google Scholar] [CrossRef] [PubMed]
- Miyawaki, K.; Yamada, Y.; Ban, N.; Ihara, Y.; Tsukiyama, K.; Zhou, H.; Fujimoto, S.; Oku, A.; Tsuda, K.; Toyokuni, S.; et al. Inhibition of gastric inhibitory polypeptide signaling prevents obesity. Nat. Med. 2002, 8, 738–742. [Google Scholar] [CrossRef]
- Daousi, C.; Wilding, J.P.; Aditya, S.; Durham, B.H.; Cleator, J.; Pinkney, J.H.; Ranganath, L.R. Effects of peripheral administration of synthetic human glucose-dependent insulinotropic peptide (GIP) on energy expenditure and subjective appetite sensations in healthy normal weight subjects and obese patients with type 2 diabetes. Clin. Endocrinol. 2009, 71, 195–201. [Google Scholar] [CrossRef]
- Giezenaar, C.; Hutchison, A.T.; Luscombe-Marsh, N.D.; Chapman, I.; Horowitz, M.; Soenen, S. Effect of age on blood glucose and plasma insulin, glucagon, ghrelin, CCK, GIP, and GLP-1 responses to whey protein ingestion. Nutrients 2017, 10, 2. [Google Scholar] [CrossRef]
- Giezenaar, C.; Luscombe-Marsh, N.D.; Hutchison, A.T.; Standfield, S.; Feinle-Bisset, C.; Horowitz, M.; Chapman, I.; Soenen, S. Dose-dependent effects of randomized intraduodenal whey-protein loads on glucose, gut hormone, and amino acid concentrations in healthy older and younger men. Nutrients 2018, 10, 78. [Google Scholar] [CrossRef]
- Rigamonti, A.E.; Leoncini, R.; De Col, A.; Tamini, S.; Cicolini, S.; Abbruzzese, L.; Cella, S.G.; Sartorio, A. The appetite-suppressant and GLP-1-stimulating effects of whey proteins in obese subjects are associated with increased circulating levels of specific amino acids. Nutrients 2020, 12, 775. [Google Scholar] [CrossRef] [PubMed]
- Zuo, L.; He, F.; Tinsley, G.M.; Pannell, B.K.; Ward, E.; Arciero, P.J. Comparison of high-protein, intermittent fasting low-calorie diet and heart healthy diet for vascular health of the obese. Front. Physiol. 2016, 7, 350. [Google Scholar] [CrossRef]



| WMS (Control) | WMS + WP | RS | RS + WP | |
|---|---|---|---|---|
| Energy (kcal) | 397.0 | 397.0 | 397.0 | 397.0 |
| Waxy maize starch (g) | 45.0 | 45.0 | – | – |
| Resistant starch a from waxy maize starch (g) | – | – | 40.0 | 40.0 |
| Whey protein (g) | – | 20.5 | – | 20.5 |
| Sucrose (g) | 4.5 | 4.5 | 4.5 | 4.5 |
| Maltodextrin (g) | 10.7 | 10.7 | 10.7 | 10.7 |
| Milk powder (g) | 21.1 | 0.90 | 21.1 | 0.90 |
| Egg (g) | 50.0 | 50.0 | 50.0 | 50.0 |
| Baking powder (g) | 4.8 | 4.8 | 4.8 | 4.8 |
| Total carbohydrate (g) | 73.0 | 60.0 | 73.0 | 60.0 |
| Total fat (g) | 5.0 | 5.5 | 5.0 | 5.5 |
| Total protein (g) | 15.0 | 26.8 | 15.0 | 26.8 |
| Total fiber (g) | 0.0 | 0.0 | 0.0 | 0.0 |
| N | 8 |
|---|---|
| Age (years) | 49.00 ± 13.61 |
| Weight (kg) | 91.34 ± 11.77 |
| Height (cm) | 180.49 ± 5.83 |
| BMI | 28.16 ± 5.071 |
| Percent fat mass (%) | 26.85 ± 10.17 |
| Systolic BP (mmHg) | 123.25 ± 8.65 |
| Diastolic BP (mmHg) | 76.50 ± 4.75 |
| Resting HR (bpm) | 58.75 ± 8.12 |
| Glucose (mg/dL) | 91.75 ± 10.38 |
| Total Cholesterol (mg/dL) | 181.50 ± 27.88 |
| HDL (mg/dL) | 44.75 ± 7.78 |
| LDL (mg/dL) | 114.50 ± 29.41 |
| Triglycerides (mg/dL) | 109.63 ± 55.67 |
| TC/HDL ratio | 4.12 ± 0.87 |
| Outcome Variable | Meal | Baseline | 60 min | 120 min | 180 min | ||||
|---|---|---|---|---|---|---|---|---|---|
| Mean | 95% CI | Mean | 95% CI | Mean | 95% CI | Mean | 95% CI | ||
| RMR and TEM (kcal/minute) b | WMS | 1.17 | (1.05–1.29) | 1.33 | (1.21–1.45) | 1.31 | (1.19–1.43) | 1.23 | (1.11–1.35) |
| RS | 1.17 | (1.05–1.29) | 1.38 | (1.26–1.49) | 1.29 | (1.17–1.41) | 1.29 | (1.18–1.42) | |
| WMS + WP | 1.15 | (1.03–1.27) | 1.34 | (1.22–1.46) | 1.28 | (1.16–1.40) | 1.26 | (1.14–1.38) | |
| RS + WP * | 1.25 | (1.13–1.37) | 1.42 | (1.29–1.54) | 1.41 | (1.29–1.53) | 1.34 | (1.22–1.46) | |
| RER | WMS | 0.85 | (0.83–0.88) | 0.86 | (0.83–0.89) | 0.9 | (0.87–0.93) | 0.89 | (0.86–0.92) |
| RS | 0.81 | (0.79–0.84) | 0.88 | (0.85–0.90) | 0.87 | (0.85–0.90) | 0.87 | (0.84–0.89) | |
| WMS + WP | 0.83 | (0.80–0.86) | 0.86 | (0.83–0.88) | 0.88 | (0.85–0.91) | 0.87 | (0.84–0.89) | |
| RS + WP * | 0.82 | (0.79–0.85) | 0.84 | (0.81–0.87) | 0.85 | (0.82–0.88) | 0.86 | (0.81–0.87) | |
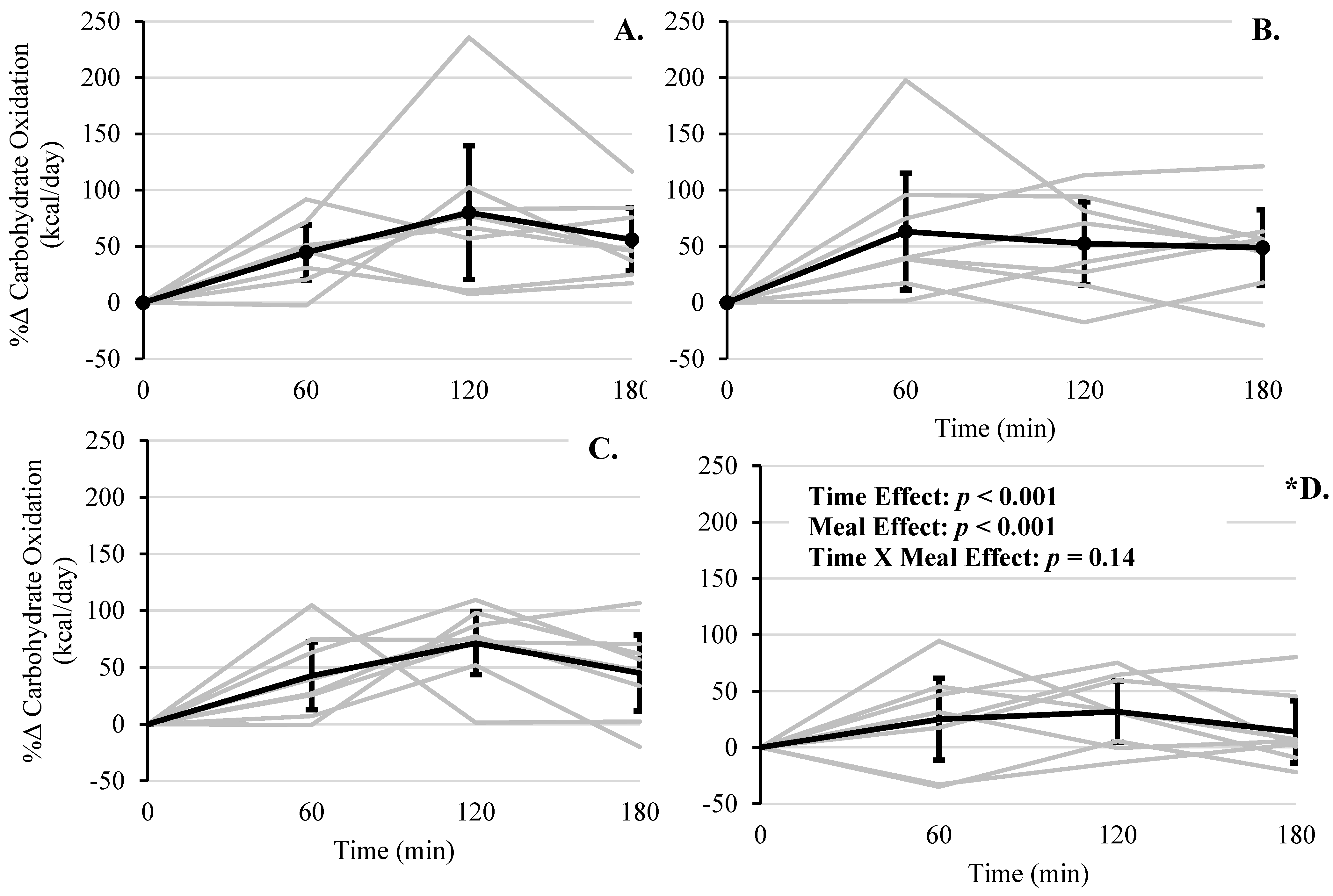
| Carbohydrate Oxidation Rate (kcal/day) | WMS | 892 | (660–1124) | 1076 | (844–1308) | 1282 | (1050–1514) | 1135 | (903–1367) |
| RS | 632 | (400–864) | 1227 | (995–1458) | 1103 | (871–1335) | 1061 | (829–1293) | |
| WMS + WP | 821 | (589–1053) | 1110 | (878–1342) | 1273 | (1041–1505) | 1095 | (863–1327) | |
| RS + WP * | 664 | (432–896) | 941 | (709–1173) | 957 | (725–1189) | 839 | (607–1071) | |
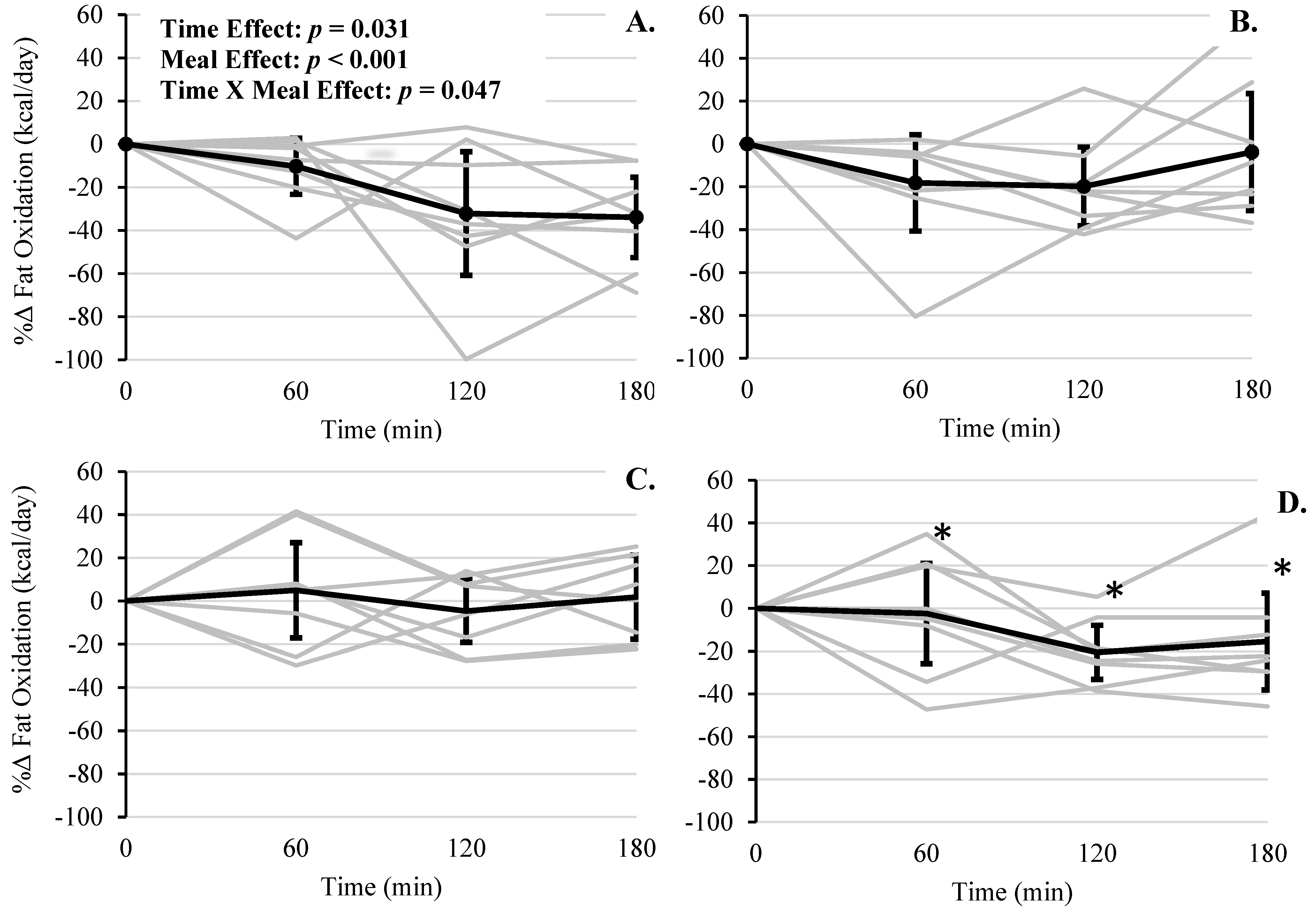
| Fat Oxidation Rate (kcal/day) | WMS | 806 | (636–977) | 874 | (704–1045) | 615 | (445–786) | 640 | (470–811) |
| RS | 1057 | (886–1227) | 767 | (596–937) | 756 | (585–926) | 880 | (710–1051) | |
| WMS + WP | 982 | (812–1153) | 934 | (763–1105) | 768 | (597–938) | 810 | (640–981) | |
| RS + WP | 1003 | (832–1173) | 993 ** | (822–1163) | 895 ** | (724–1065) | 982 ** | (811–1153) | |
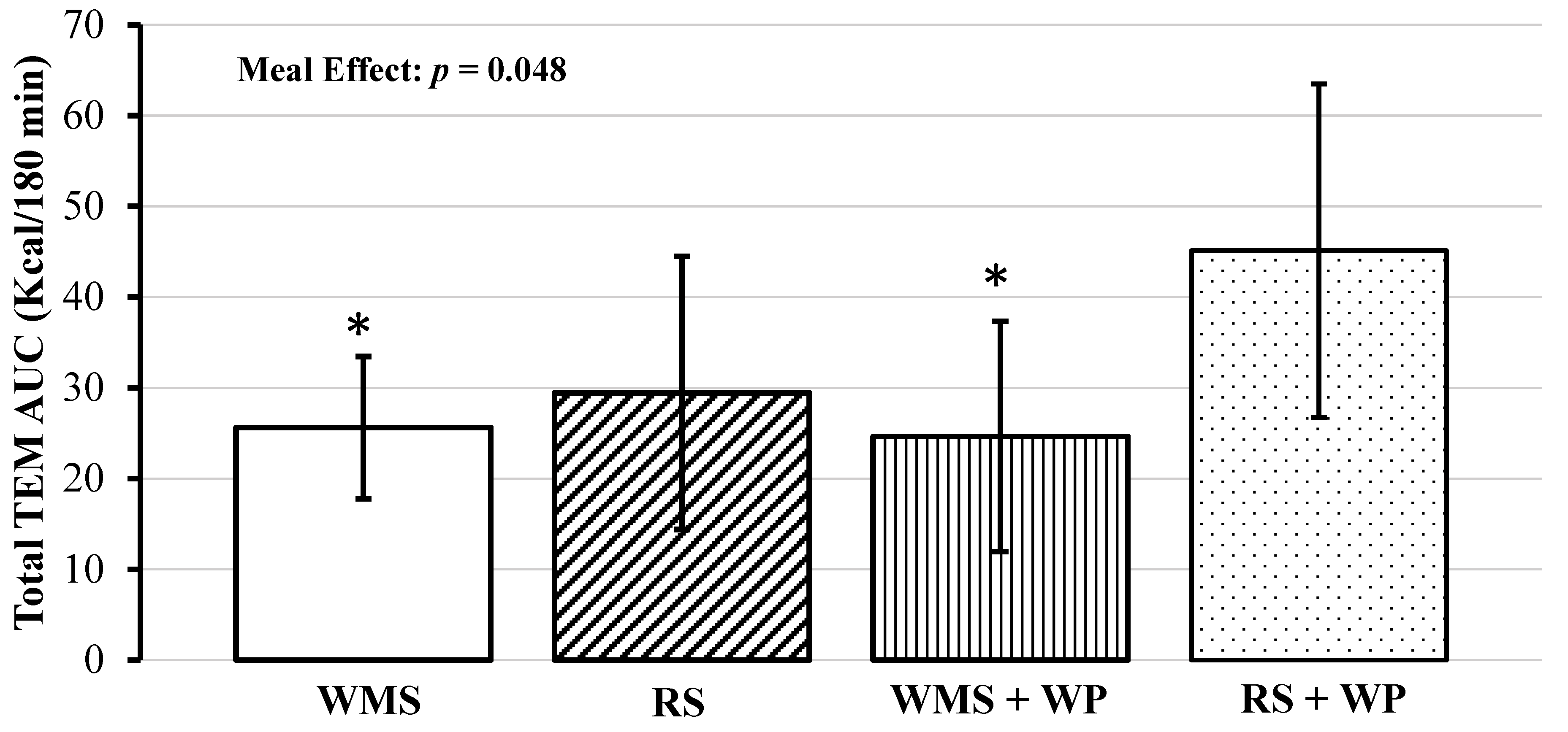
| Meal | Kcal Consumed | RMR b | 60 min iAUC TEM c | 120 min iAUC TEM | 180 min iAUC TEM | Total AUC TEM d |
|---|---|---|---|---|---|---|
| WMS | 397 | 1686 (1490–1881) | 4.39 (−0.29–9.08) | 12.53 (7.84–17.21) | 8.70 (4.02–13.39) | 25.61 (14.31–36.91) |
| RS | 397 | 1682 (1507–1858) | 5.74 (1.05–10.42) | 14.48 (9.79–19.16) | 9.23 (4.54–13.91) | 29.44 (18.14–40.74) |
| WMS + WP | 397 | 1659 (1447–1871) | 4.54 (−0.15–9.22) | 12.00 (7.32–6.69) | 8.10 (3.42–12.79) | 24.64 (13.34–35.94) |
| RS + WP * | 397 | 1692 (1513–1870) | 6.83 (2.14–11.51) | 20.36 (15.68–25.05) | 17.93 (13.24–22.61) | 45.11 (33.81–56.41) * |
| Outcome Variable | Meal | Baseline | 60 min | 120 min | 180 min | ||||
|---|---|---|---|---|---|---|---|---|---|
| Mean | 95% CI | Mean | 95% CI | Mean | 95% CI | Mean | 95% CI | ||
| Glucose (mg/dL) | WMS | 110.41 | (94.68–126.13) | 133.11 | (117.39–148.84) | 99.95 | (83.49–116.40) | 101.42 | (84.96–117.87) |
| RS | 105.06 | (89.34–120.79) | 121.64 | (105.18–138.09) | 99.87 | (84.15–115.59) | 96.65 | (79.26–114.05) | |
| RS + WP * | 100.86 | (85.14–116.89) | 97.57 | (81.14–116.59) | 93.83 | (78.10–109.55) | 95.7 | (79.98–111.43) | |
| Insulin (units) | WMS | 3.52 | (−1.19–8.23) | 29.43 | (24.72–34.14) | 12.53 | (7.64–17.41) | 7.83 | (2.95–12.72) |
| RS | 2.87 | (−1.84–7.58) | 16.89 | (12.01–21.78) | 8.52 | (3.64–13.41) | 3.51 | (–1.59–8.63) | |
| RS + WP * | 2.86 | (−1.85–7.57) | 14.80 ** | (10.09–19.51) | 5.94 ** | (1.23–10.65) | 3.14 ** | (–1.57–7.85) | |
| GIP (pmol/L) | WMS | 35.26 | (0.50–70.01) | 184.98 | (150.23–219.73) | 148.32 | (112.57–184.06) | 89.31 | (54.56–124.06) |
| RS | 36.36 | (1.61–71.11) | 169.97 | (134.23–205.71) | 131.84 | (97.08–166.59) | 80.66 | (43.62–117.69) | |
| RS + WP | 26.69 | (−8.06–61.45) | 157.42 | (122.66–192.17) | 133.00 | (98.25–167.76) | 74.78 | (40.03–109.54) | |
| GLP-1 (pM) | WMS | 6.63 | (3.03–10.22) | 11.71 | (7.85–15.77) | 11.32 | (7.62–15.03) | 9.93 | (6.22–13.63) |
| RS | 7.44 | (3.85–11.03) | 13.51 | (9.80–17.22) | 15.08 | (11.22–18.93) | 10.73 | (6.87–14.59) | |
| RS + WP | 6.13 | (2.53–9.72) | 10.13 | (6.53–13.72) | 10.25 | (6.66–13.84) | 8.88 | (5.28–12.47) | |
| GRL (pM) | WMS | 375.47 | (253.34–497.59) | 241.84 | (118.37–365.31) | 321.06 | (197.62–444.49) | 377.53 | (255.41–499.66) |
| RS | 413.76 | (291.64–535.89) | 278.22 | (154.77–401.66) | 367.44 | (245.31–489.56) | 411.33 | (286.09–536.57) | |
| RS + WP | 400.08 | (277.96–522.20) | 289.58 | (166.11–413.05) | 330.52 | (208.39–452.64) | 426.69 | (304.57–548.81) | |
| PYY (pg/mL) | WMS | 32.63 | (14.25–51.02) | 45.16 | (26.57–63.76) | 41.87 | (23.49–60.26) | 44.38 | (26.01–62.77) |
| RS | 27.86 | (9.62–46.08) | 40.78 | (22.39–59.16) | 44.48 | (26.76–64.56) | 45.66 | (26.76–64.56) | |
| RS + WP | 29.36 | (11.13–47.59) | 34.11 | (15.88–52.34) | 36.36 | (18.13–54.59) | 43.34 | (24.96–61.72) | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mohr, A.E.; Minicucci, O.; Long, D., Jr.; Miller, V.J.; Keller, A.; Sheridan, C.; O’brien, G.; Ward, E.; Schuler, B.; Connelly, S.; et al. Resistant Starch Combined with Whey Protein Increases Postprandial Metabolism and Lowers Glucose and Insulin Responses in Healthy Adult Men. Foods 2021, 10, 537. https://doi.org/10.3390/foods10030537
Mohr AE, Minicucci O, Long D Jr., Miller VJ, Keller A, Sheridan C, O’brien G, Ward E, Schuler B, Connelly S, et al. Resistant Starch Combined with Whey Protein Increases Postprandial Metabolism and Lowers Glucose and Insulin Responses in Healthy Adult Men. Foods. 2021; 10(3):537. https://doi.org/10.3390/foods10030537
Chicago/Turabian StyleMohr, Alex E., Olivia Minicucci, Dale Long, Jr., Vincent J. Miller, Allison Keller, Caitlin Sheridan, Gabriel O’brien, Emery Ward, Brad Schuler, Scott Connelly, and et al. 2021. "Resistant Starch Combined with Whey Protein Increases Postprandial Metabolism and Lowers Glucose and Insulin Responses in Healthy Adult Men" Foods 10, no. 3: 537. https://doi.org/10.3390/foods10030537
APA StyleMohr, A. E., Minicucci, O., Long, D., Jr., Miller, V. J., Keller, A., Sheridan, C., O’brien, G., Ward, E., Schuler, B., Connelly, S., Holst, J. J., Astrup, A., He, F., Gentile, C. L., & Arciero, P. J. (2021). Resistant Starch Combined with Whey Protein Increases Postprandial Metabolism and Lowers Glucose and Insulin Responses in Healthy Adult Men. Foods, 10(3), 537. https://doi.org/10.3390/foods10030537










