Evaluation of Fatty Acids Profile as a Useful Tool towards Valorization of By-Products of Agri-Food Industry
Abstract
1. Introduction
2. Materials and Methods
2.1. Chemicals
2.2. Agri-Food By-Products
2.3. Extraction and Derivatization of Fatty Acids
2.4. Method Validation
2.5. Gas Chromatography–Flame Ionization Detector (GC–FID) Conditions
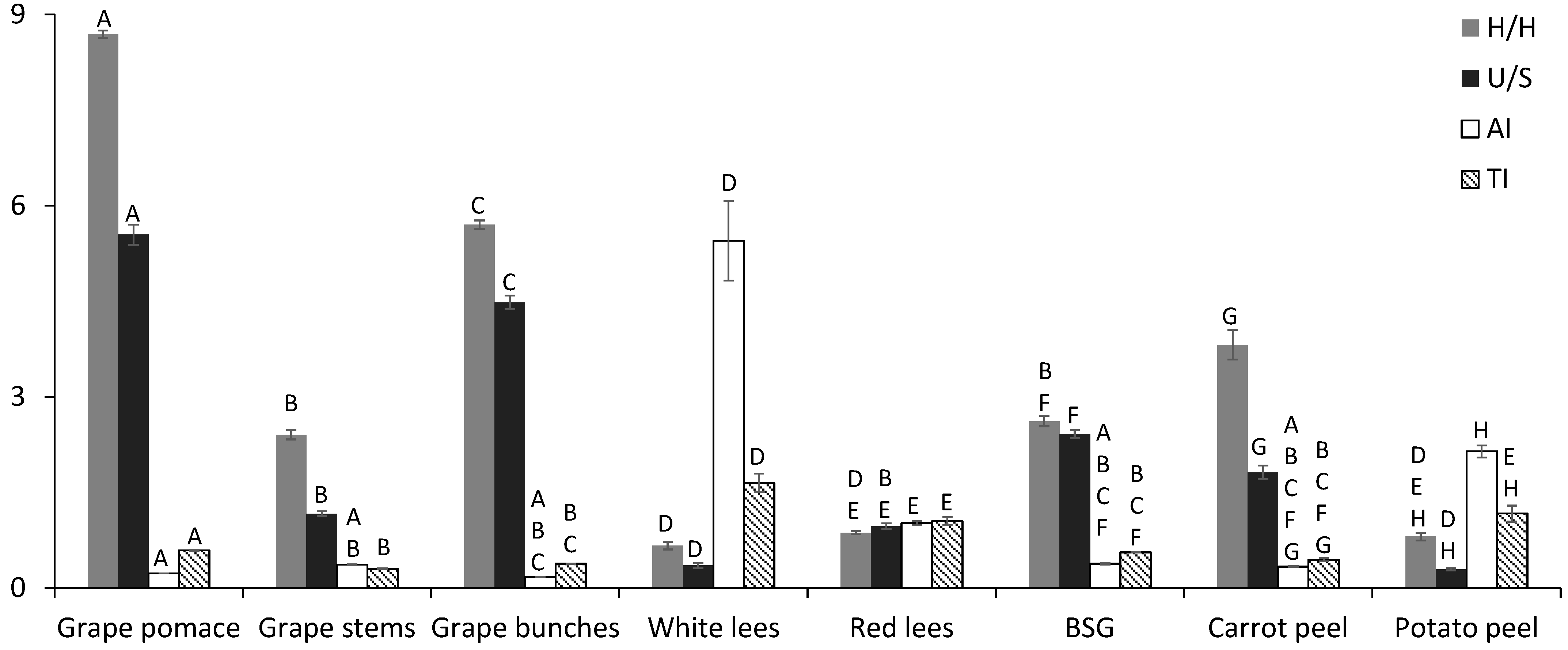
2.6. Functional Quality
AI = (C12:0 + 4 × C14:0 + C16:0)/[Σ MUFA + Σ PUFA (n6 & n3)]
TI = (C14:0 + C16:0 + C18:0)/[0.5 × Σ MUFA + 0.5 × Σ PUFA (n6) + 3 × Σ PUFA (n3) + [Σ PUFA (n3)/Σ PUFA (n6)]
2.7. Fourier Transform Infrared Spectroscopy (FTIR) Analysis of Agri-Food By-Products
2.8. Statistical Analysis
3. Results and Discussion
3.1. Method Validation
3.2. Lipid Extraction Yield and Fatty Acids Profile and Functional Quality of Agri-Food By-Products
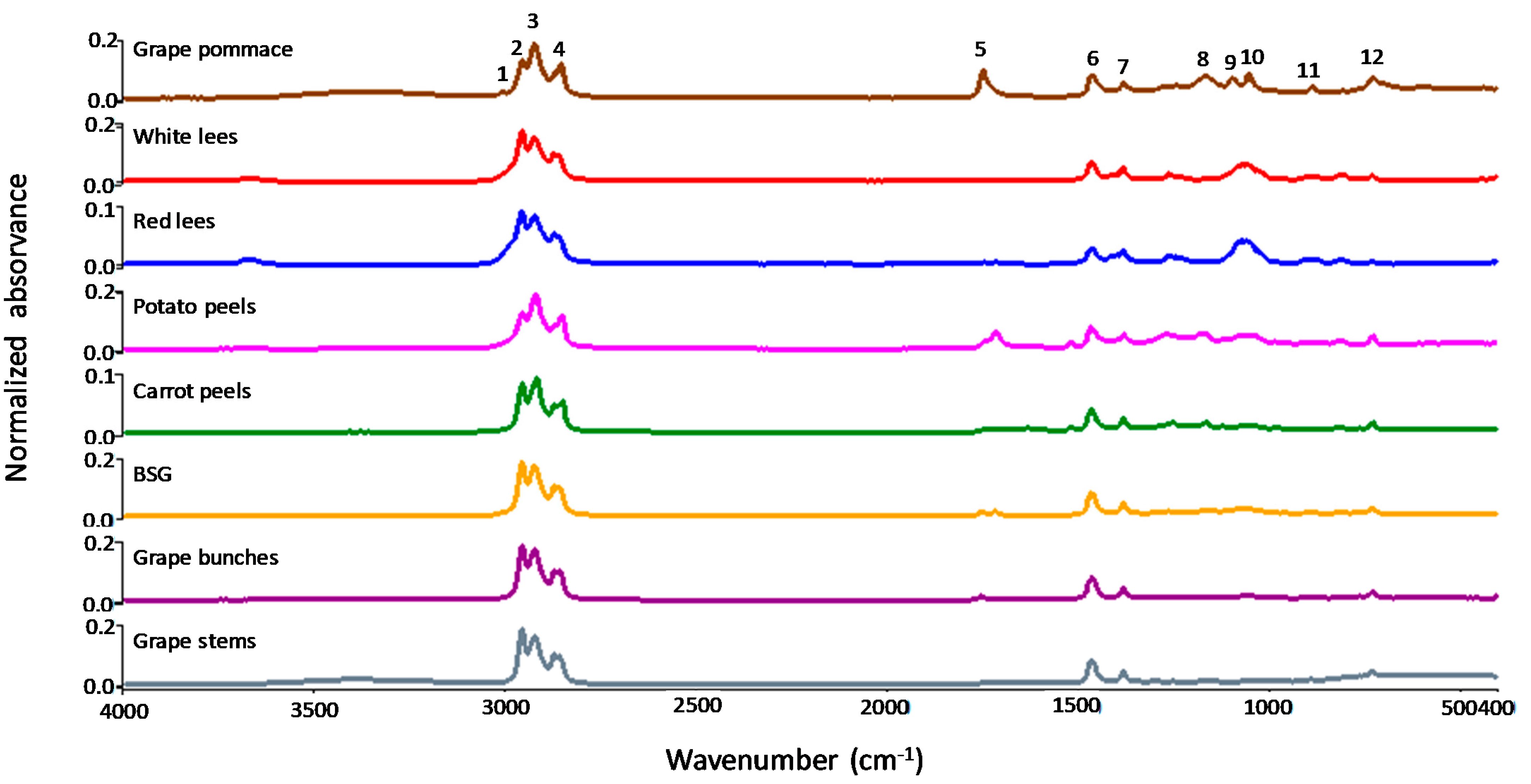
3.3. Fatty Acids Profile by Fourier Transform Infrared Spectroscopy (FTIR)
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Pfaltzgraff, L.A.; De Bruyn, M.; Cooper, E.C.; Budarin, V.; Clark, J.H. Food waste biomass: A resource for high-value chemicals. Green Chem. 2013, 15, 307–314. [Google Scholar] [CrossRef]
- Chetrariu, A.; Dabija, A. Brewer’s Spent Grains: Possibilities of Valorization, a Review. Appl. Sci. 2020, 10, 5619. [Google Scholar] [CrossRef]
- Jiménez-Moreno, N.; Esparza, I.; Bimbela, F.; Gandía, L.M.; Ancín-Azpilicueta, C. Valorization of selected fruit and vegetable wastes as bioactive compounds: Opportunities and challenges. Crit. Rev. Environ. Sci. Technol. 2020, 50, 2061–2108. [Google Scholar] [CrossRef]
- Faustino, A.M.; Veiga, M.; Sousa, P.; Costa, E.M.; Silva, S.; Pintado, M. Agro-Food Byproducts as a New Source of Natural Food Additives. Molecules 2019, 24, 1056. [Google Scholar] [CrossRef]
- Câmara, J.; Lourenço, S.; Silva, C.; Lopes, A.; Andrade, C.; Perestrelo, R. Exploring the potential of wine industry by-products as source of additives to improve the quality of aquafeed. Microchem. J. 2020, 155, 104758. [Google Scholar] [CrossRef]
- Rigano, F.; Arena, P.; Mangraviti, D.; Donnarumma, D.; Dugo, P.; Donato, P.; Mondello, L.; Micalizzi, G. Identification of high-value generating molecules from the wastes of tuna fishery industry by liquid chromatography and gas chromatography hyphenated techniques with automated sample preparation. J. Sep. Sci. 2021, 44, 1571–1580. [Google Scholar] [CrossRef]
- Aznar-Sánchez, J.A.; Mendoza, J.M.F.; Mendoza, F.; Ingrao, C.; Failla, S.; Bezama, A.; Nemecek, T.; Schmid, A.G. Indicators for circular economy in the agri-food sector. Resour. Conserv. Recycl. 2020, 163, 105028. [Google Scholar] [CrossRef]
- Fraga-Corral, M.; Otero, P.; Echave, J.; Garcia-Oliveira, P.; Carpena, M.; Jarboui, A.; Nuñez-Estevez, B.; Simal-Gandara, J.; Prieto, M. By-Products of Agri-Food Industry as Tannin-Rich Sources: A Review of Tannins’ Biological Activities and Their Potential for Valorization. Foods 2021, 10, 137. [Google Scholar] [CrossRef]
- Fontana, A.R.; Antoniolli, A.; Bottini, R. Grape Pomace as a Sustainable Source of Bioactive Compounds: Extraction, Characterization, and Biotechnological Applications of Phenolics. J. Agric. Food Chem. 2013, 61, 8987–9003. [Google Scholar] [CrossRef]
- Lucarini, M.; Durazzo, A.; Kiefer, J.; Santini, A.; Lombardi-Boccia, G.; Souto, E.; Romani, A.; Lampe, A.; Nicoli, S.F.; Gabrielli, P.; et al. Grape Seeds: Chromatographic Profile of Fatty Acids and Phenolic Compounds and Qualitative Analysis by FTIR-ATR Spectroscopy. Foods 2019, 9, 10. [Google Scholar] [CrossRef]
- Durazzo, A.; Lucarini, M.; Souto, E.B.; Cicala, C.; Caiazzo, E.; Izzo, A.A.; Novellino, E.; Santini, A. Polyphenols: A concise overview on the chemistry, occurrence, and human health. Phytother. Res. 2019, 33, 2221–2243. [Google Scholar] [CrossRef]
- Ferhi, S.; Santaniello, S.; Zerizer, S.; Cruciani, S.; Fadda, A.; Sanna, D.; Dore, A.; Maioli, M.; D’Hallewin, G. Total Phenols from Grape Leaves Counteract Cell Proliferation and Modulate Apoptosis-Related Gene Expression in MCF-7 and HepG2 Human Cancer Cell Lines. Molecules 2019, 24, 612. [Google Scholar] [CrossRef]
- Di Meo, F.; Aversano, R.; Diretto, G.; Demurtas, O.C.; Villano, C.; Cozzolino, S.; Filosa, S.; Carputo, D.; Crispi, S. Anti-cancer activity of grape seed semi-polar extracts in human mesothelioma cell lines. J. Funct. Foods 2019, 61, 103515. [Google Scholar] [CrossRef]
- Bijak, M.; Sut, A.; Kosiorek, A.; Saluk-Bijak, J.; Golanski, J. Dual Anticoagulant/Antiplatelet Activity of Polyphenolic Grape Seeds Extract. Nutrients 2019, 11, 93. [Google Scholar] [CrossRef]
- Tseng, A.; Zhao, Y. Wine grape pomace as antioxidant dietary fibre for enhancing nutritional value and improving storability of yogurt and salad dressing. Food Chem. 2013, 138, 356–365. [Google Scholar] [CrossRef]
- Iriondo-DeHond, M.; Miguel, E.; Del Castillo, M.D. Food Byproducts as Sustainable Ingredients for Innovative and Healthy Dairy Foods. Nutrients 2018, 10, 1358. [Google Scholar] [CrossRef]
- Mussatto, S.I. Brewer’s spent grain: A valuable feedstock for industrial applications. J. Sci. Food Agric. 2014, 94, 1264–1275. [Google Scholar] [CrossRef]
- Karlović, A.; Jurić, A.; Ćorić, N.; Habschied, K.; Krstanović, V.; Mastanjević, K. By-Products in the Malting and Brewing Industries—Re-Usage Possibilities. Fermentation 2020, 6, 82. [Google Scholar] [CrossRef]
- Andreu-Coll, L.; Cano-Lamadrid, M.; Sendra, E.; Carbonell-Barrachina, A.; Legua, P.; Hernández, F. Fatty acid profile of fruits (pulp and peel) and cladodes (young and old) of prickly pear [Opuntia ficus-indica (L.) Mill.] from six Spanish cultivars. J. Food Compos. Anal. 2019, 84, 103294. [Google Scholar] [CrossRef]
- Zárate, R.; El Jaber-Vazdekis, N.; Tejera, N.; Pérez, J.A.P.; Rodríguez, C. Significance of long chain polyunsaturated fatty acids in human health. Clin. Transl. Med. 2017, 6, 25. [Google Scholar] [CrossRef]
- Kumar Maurya, A.; Faizabad Uttar Pradesh, K.; Hari Om Verma, I.; Pandey, G.; Pal, J.; Shukla, B.; Om Verma, H. A review on role of fish in human nutrition with special emphasis to essential fatty acid. Int. J. Fish. Aquat. Stud. 2018, 6, 427–430. [Google Scholar]
- Nguyen, Q.V.; Malau-Aduli, B.S.; Cavalieri, J.; Malau-Aduli, A.E.O.; Nichols, P.D. Enhancing Omega-3 Long-Chain Polyunsaturated Fatty Acid Content of Dairy-Derived Foods for Human Consumption. Nutrients 2019, 11, 743. [Google Scholar] [CrossRef]
- Wołoszyn, J.; Haraf, G.; Okruszek, A.; Wereńska, M.; Goluch, Z.; Teleszko, M. Fatty acid profiles and health lipid indices in the breast muscles of local Polish goose varieties. Poult. Sci. 2020, 99, 1216–1224. [Google Scholar] [CrossRef]
- Jarukas, L.; Kuraite, G.; Baranauskaite, J.; Marksa, M.; Bezruk, I.; Ivanauskas, L. Optimization and Validation of the GC/FID Method for the Quantification of Fatty Acids in Bee Products. Appl. Sci. 2020, 11, 83. [Google Scholar] [CrossRef]
- Ribeiro, L.F.; Ribani, R.H.; Francisco, T.M.G.; Soares, A.A.; Pontarolo, R.; Haminiuk, C.W.I. Profile of bioactive compounds from grape pomace (Vitis vinifera and Vitis labrusca) by spectrophotometric, chromatographic and spectral analyses. J. Chromatogr. B 2015, 1007, 72–80. [Google Scholar] [CrossRef]
- Almeida, A.D.R.; Geraldo, M.R.F.; Ribeiro, L.F.; Silva, M.V.; Maciel, M.V.D.O.B.; Haminiuk, C.W.I. Bioactive compounds from brewer’s spent grain: Phenolic compounds, fatty acids and in vitro antioxidant capacity. Acta Sci. Technol. 2017, 39, 269. [Google Scholar] [CrossRef]
- Niemi, P.; Tamminen, T.; Smeds, A.; Viljanen, K.; Ohra-Aho, T.; Holopainen-Mantila, U.; Faulds, C.B.; Poutanen, K.; Buchert, J. Characterization of Lipids and Lignans in Brewer’s Spent Grain and Its Enzymatically Extracted Fraction. J. Agric. Food Chem. 2012, 60, 9910–9917. [Google Scholar] [CrossRef]
- Yeddes, N.; Chérif, J.K.; Jrad, A.; Barth, D.; Trabelsi-Ayadi, M. Supercritical SC-CO2 and soxhlet n-hexane extract of Tunisian Opuntia ficus indica seeds and fatty acids analysis. J. Lipids 2012, 2012, 914693. [Google Scholar] [CrossRef]
- Cerón-Martínez, L.; Hurtado-Benavides, A.; Ayala-Aponte, A.; Serna-Cock, L.; Tirado, D. A Pilot-Scale Supercritical Carbon Dioxide Extraction to Valorize Colombian Mango Seed Kernel. Molecules 2021, 26, 2279. [Google Scholar] [CrossRef]
- Alinafiah, S.M.; Azlan, A.; Ismail, A.; Ab Rashid, N.-K.M. Method Development and Validation for Omega-3 Fatty Acids (DHA and EPA) in Fish Using Gas Chromatography with Flame Ionization Detection (GC-FID). Molecules 2021, 26, 6592. [Google Scholar] [CrossRef]
- Kolláthová, R.; Hanušovský, O.; Gálik, B.; Bíro, D.; Simko, M.; Juráček, M.; Rolinec, M.; Puntigam, R.; Slama, J.A.; Gierus, M. Fatty acid profile analysis of grape by-products from Slovakia and Austria. Acta Fytotech. Zootech. 2020, 23, 78–84. [Google Scholar] [CrossRef]
- Zhang, H.; Wang, Z.; Liu, O. Development and validation of a GC–FID method for quantitative analysis of oleic acid and related fatty acids. J. Pharm. Anal. 2015, 5, 223–230. [Google Scholar] [CrossRef]
- FAO. The State of World Fisheries and Aquaculture 2020. Sustainability in Action; FAO: Rome, Italy, 2020; p. 206. [Google Scholar] [CrossRef]
- Bandara, T.; Tharindu Bandara, C. Alternative feed ingredients in aquaculture: Opportunities and challenges. J. Entomol. Zool. Stud. 2018, 6, 3087. [Google Scholar]
- Tocher, D. Metabolism and Functions of Lipids and Fatty Acids in Teleost Fish. Rev. Fish. Sci. 2003, 11, 107–184. [Google Scholar] [CrossRef]
- Monroig, O.; Kabeya, N. Desaturases and elongases involved in polyunsaturated fatty acid biosynthesis in aquatic invertebrates: A comprehensive review. Fish. Sci. 2018, 84, 911–928. [Google Scholar] [CrossRef]
- Fărcaş, A.C.; Socaci, S.; Dulf, F.; Tofană, M.; Mudura, E.; Diaconeasa, Z. Volatile profile, fatty acids composition and total phenolics content of brewers’ spent grain by-product with potential use in the development of new functional foods. J. Cereal Sci. 2015, 64, 34–42. [Google Scholar] [CrossRef]
- Sancho-Galán, P.; Amores-Arrocha, A.; Jiménez-Cantizano, A.; Palacios, V. Physicochemical and Nutritional Characterization of Winemaking Lees: A New Food Ingredient. Agronomy 2020, 10, 996. [Google Scholar] [CrossRef]
- Samotyja, U. Potato Peel as a Sustainable Resource of Natural Antioxidants for the Food Industry. Potato Res. 2019, 62, 435–451. [Google Scholar] [CrossRef]
- Ulbricht, T.L.; Southgate, D.A. Coronary heart disease: Seven dietary factors. Lancet 1991, 338, 985–992. [Google Scholar] [CrossRef]
- Guil-Guerrero, J.L.; Venegas-Venegas, E.; Rincón-Cervera, M.; Suárez, M.D. Fatty acid profiles of livers from selected marine fish species. J. Food Compos. Anal. 2011, 24, 217–222. [Google Scholar] [CrossRef]
- Rodríguez-Ruiz, J.; Belarbi, E.-H.; Sánchez, J.L.G.; Alonso, D. Rapid simultaneous lipid extraction and transesterification for fatty acid analyses. Biotechnol. Tech. 1998, 12, 689–691. [Google Scholar] [CrossRef]
- Dimić, I.; Teslić, N.; Putnik, P.; Bursać Kovačević, D.; Zeković, Z.; Šojić, B.; Mrkonjić, Ž.; Čolović, D.; Montesano, D.; Pavlić, B. Innovative and conventional valorizations of grape seeds from winery by-products as sustainable source of lipophilic antioxidants. Antioxidants 2020, 9, 568. [Google Scholar] [CrossRef]
- Worley, B.; Powers, R. Multivariate Analysis in Metabolomics. Curr. Metabolomics 2013, 1, 92–107. [Google Scholar] [CrossRef][Green Version]
- Laroche, M.; Perreault, V.; Marciniak, A.; Gravel, A.; Chamberland, J.; Doyen, A. Comparison of Conventional and Sustainable Lipid Extraction Methods for the Production of Oil and Protein Isolate from Edible Insect Meal. Foods 2019, 8, 572. [Google Scholar] [CrossRef]
- Korbecki, J.; Bajdak-Rusinek, K. The effect of palmitic acid on inflammatory response in macrophages: An overview of molecular mechanisms. Inflamm. Res. 2019, 68, 915–932. [Google Scholar] [CrossRef]
- Russo, V.M.; Jacobs, J.L.; Hannah, M.C.; Moate, P.J.; Dunshea, F.R.; Leury, B.J. In vitro evaluation of the methane mitigation potential of a range of grape marc products. Anim. Prod. Sci. 2017, 57, 1437–1444. [Google Scholar] [CrossRef]
- Ahmed, S.T.; Islam, M.; Bostami, A.R.; Mun, H.-S.; Kim, Y.-J.; Yang, C.-J. Meat composition, fatty acid profile and oxidative stability of meat from broilers supplemented with pomegranate (Punica granatum L.) by-products. Food Chem. 2015, 188, 481–488. [Google Scholar] [CrossRef]
- Simopoulos, A.P. An Increase in the Omega-6/Omega-3 Fatty Acid Ratio Increases the Risk for Obesity. Nutrients 2016, 8, 128. [Google Scholar] [CrossRef]
- Shiroma, C.; Rodriguez-Saona, L. Application of NIR and MIR spectroscopy in quality control of potato chips. J. Food Compos. Anal. 2009, 22, 596–605. [Google Scholar] [CrossRef]
- Moreira, J.L.; Santos, L. Spectroscopic Interferences in Fourier Transform Infrared Wine Analysis; Elsevier: Amsterdam, The Netherlands, 2004; Volume 513, pp. 263–268. [Google Scholar]
| FAMEs | RT (min) | R2 | Range (µg/mL) | Equation | LOD (µg/mL) | LOQ (µg/mL) | Precision (%RSD) | |
|---|---|---|---|---|---|---|---|---|
| Intra-Day | Inter-Day | |||||||
| C4:0 | 4.25 | 0.993 | 1.77–39.1 | y = 0.069x + 0.354 | 0.41 | 1.38 | 1.88 | 5.02 |
| C8:0 | 11.0 | 0.982 | 1.19–39.3 | y = 0.119x + 1.472 | 0.20 | 0.66 | 1.47 | 8.66 |
| C10:0 | 13.9 | 0.993 | 1.19–39.8 | y = 0.136x + 0.708 | 0.01 | 0.03 | 1.15 | 10.2 |
| C12:0 | 16.5 | 0.993 | 1.19–39.5 | y = 0.136x + 0.870 | 0.02 | 0.07 | 1.01 | 2.64 |
| C14:0 | 18.9 | 0.999 | 1.19–39.6 | y = 0.151x + 1.112 | 0.22 | 0.74 | 1.32 | 3.73 |
| C14:1n5 | 19.3 | 0.988 | 1.76–19.5 | y = 0.156x + 0.155 | 0.19 | 0.63 | 2.56 | 12.6 |
| C15:0 | 20.3 | 0.998 | 1.76–19.6 | y = 0.158x + 0.756 | 0.14 | 0.48 | 1.52 | 10.4 |
| C15:1n5 | 20.7 | 0.994 | 1.80–20.0 | y = 0.139x + 0.838 | 0.40 | 1.36 | 1.84 | 7.79 |
| C16:0 | 21.8 | 0.995 | 0.60–60.3 | y = 0.201x + 1.395 | 0.12 | 0.40 | 3.09 | 5.35 |
| C16:1n7 | 22.2 | 0.995 | 0.62–20.5 | y = 0.146x + 1.003 | 0.07 | 0.24 | 2.76 | 4.17 |
| C18:0 | 26.3 | 0.996 | 1.19–39.6 | y = 0.234x + 0.846 | 0.35 | 1.15 | 3.25 | 5.96 |
| C18:1n9 | 26.7 | 0.996 | 1.19–39.6 | y = 0.379x + 1.176 | 0.33 | 1.10 | 4.89 | 7.28 |
| C18:2n6 | 27.9 | 0.993 | 0.58–19.9 | y = 0.302x + 0.9574 | 0.06 | 0.21 | 1.69 | 2.98 |
| C18:3n3 | 29.9 | 0.983 | 1.73–19.2 | y = 0.254x + 0.270 | 0.28 | 0.92 | 2.14 | 3.65 |
| C20:0 | 34.1 | 0.981 | 1.19–39.6 | y = 0.182x + 0.901 | 0.16 | 0.53 | 1.62 | 5.11 |
| C20:1n9 | 34.8 | 0.992 | 1.79–19.9 | y = 0.185x + 0.813 | 0.25 | 0.82 | 1.37 | 12.9 |
| C22:0 | 48.2 | 0.992 | 1.18–39.4 | y = 0.181x + 0.619 | 0.10 | 0.33 | 1.04 | 7.02 |
| C22:1n9 | 49.6 | 0.999 | 0.60–20.0 | y = 0.112x + 0.991 | 0.17 | 0.56 | 2.42 | 5.02 |
| C24:0 | 74.1 | 0.990 | 2.52–39.1 | y = 0.183x + 0.445 | 0.64 | 2.13 | 1.98 | 9.73 |
| FAMEs | Grape Pomace | Grape Stems | Grape Bunches | White Lees | Red Lees | BSG | Carrot Peel | Potato Peel |
|---|---|---|---|---|---|---|---|---|
| Saturated fatty acids | ||||||||
| Methyl butanoate C4:0) | - | - | - | - | - | - | - | 4.67 ± 0.43 |
| Methyl octanoate (C8:0) | - | - | - | - | - | - | 2.62 ± 0.03 | - |
| Methyl decanoate (C10:0) | - | 2.43 ± 0.31 A | - | 15.3 ± 0.96 B | - | - | 2.03 ± 0.16 A | - |
| Methyl dodecanoate (C12:0) | - | - | - | 18.5 ± 1.09 A | 5.64 ± 0.92 B | - | 2.77 ± 0.12 C | 5.51 ± 0.66 B,D |
| Methyl tetradecanoate (C14:0) | - | - | - | 27.6 ± 1.01 A | 3.10 ± 0.33 B | - | 1.43 ± 0.17 C | 5.02 ± 0.46 D |
| Methyl pentadecanoate (C15:0) | - | - | - | - | - | - | 1.33 ± 0.16 | - |
| Methyl hexadecanoate (C16:0) | 9.69 ± 0.13 A | 19.7 ± 0.67 B | 14.3 ± 0.30 C | 11.3 ± 1.18 A,C,D | 32.3 ± 0.31 E | 27.0 ± 0.66 F | 13.2 ± 0.69 C,D,G | 23.3 ± 1.36 H |
| Methyl stearate (C18:0) | 4.57 ± 0.34 A | 4.52 ± 0.06 A,B | 3.90 ± 0.57 A,B,C | 1.38 ± 0.10 D | 9.77 ± 0.59 E | 1.59 ± 0.24 D,F | 2.79 ± 0.17 G | 8.50 ± 0.35 H |
| Methyl eicosanoate (C20:0) | 1.04 ± 0.18 A | 2.91 ±0.14 B | - | - | - | 0.69 ± 0.17 A,C | 3.02 ± 0.30 B,D | - |
| Methyl behenate (C22:0) | - | 7.61 ± 0.82 A | - | - | - | - | 3.39 ± 0.20 B | - |
| Methyl lignocerate (C24:0) | - | 9.02 ± 0.79 A | - | - | - | - | 2.90 ± 0.15 B | 30.2 ± 0.71 C |
| Monounsaturated fatty acids | ||||||||
| Methyl 9-tetradecenoate (C14:1n5) | - | - | - | - | 3.16 ± 0.59 A | - | 1.65 ± 0.30 B | - |
| Methyl pentadecenoate (C15:1n5) | - | - | - | - | - | - | 1.28 ± 0.06 | - |
| Methyl palmitoleate (C16:1n7) | 0.57 ± 0.07 A | - | - | - | 4.90 ± 0.61 B | - | 2.25 ± 0.30 C | - |
| Methyl oleate (C18:1n9) | 11.5 ± 1.85 A | 4.82 ± 0.24 B | 27.4 ± 0.38 C | 3.88 ± 0.58 B,D | 8.44 ± 0.43 E | 11.7 ± 0.12 A,F | 38.6 ± 0.85 G | 5.06 ± 0.43 B,D,H |
| Methyl 11-eicosatrienoate (C20:1n9) | - | - | - | - | - | - | 2.00 ± 0.05 | - |
| Methyl erucate (C22:1n9) | - | 6.53 ± 0.73 A | - | - | 10.7 ± 0.07 B | - | 1.44 ± 0.04 C | - |
| Polyunsaturated fatty acids | ||||||||
| Methyl linoleate (C18:2n6) | 70.8 ± 1.53 A | 25.1 ± 0.39 B | 52.0 ± 0.48 C | 18.3 ± 1.47 D | 16.2 ± 0.59 D,E | 53.9 ± 0.89 C,F | 14.7 ± 0.51 E,G | 11.2 ± 0.03 H |
| Methyl α-linolenate (C18:3n3) | 1.94 ± 0.32 A | 17.4 ± 0.42 B | 2.29 ± 0.39 A,C | 3.80 ± 0.28 D | 6.00 ± 0.63 E | 5.17 ± 0.42 E,F | 2.28 ± 0.05 A,C,G | 6.58 ± 0.80 E,H |
| Σ SFA | 15.3 A | 46.2 B | 18.2 A,C | 74.1 D | 50.8 E | 29.3 F | 35.5 G | 77.2 D,H |
| Σ MUFA | 12.1 A | 11.3 A,B | 27.4 C | 3.88 D | 27.2 C,E | 11.7 A,B,F | 47.3 G | 5.06 D,H |
| Σ PUFA | 72.7 A | 42.5 B | 54.3 C | 22.0 D | 22.2 D,E | 59.0 F | 17.0 G | 17.8 G,H |
| PUFA n3/PUFA n6 | 0.03 A | 0.70 B | 0.04 A,C | 0.21 D | 0.37 E | 0.10 A,C,F | 0.16 D,F,G | 0.59 H |
| PUFA n6/PUFA n3 | 37.3 A | 1.44 B | 23.2 C | 4.81 B,D | 2.72 B,D,E | 10.5 D,E,F | 6.42 D,E,F,G | 1.72 B,D,E,G,H |
| Band | Frequency (cm−1) | Functional Group | Mode of Vibration |
|---|---|---|---|
| 1 | 3014 | CH=CH (cis) | Stretching |
| 2 | 2953 | R-CH3 (methyl) | Stretching (asym) |
| 3 | 2925 | R=CH2 (methylene) | Stretching (asym) |
| 4 | 2856 | R=CH2 | Stretching (sym) |
| 5 | 1748 | C=O (ester-FA linkage) | Stretching |
| 6 | 1458 | R=CH2 | Bending (scissoring) |
| 7 | 1378 | R-CH3 | Bending (sym) |
| 8 | 1163 | R =CH-R | Stretching |
| 9–10 | 1100–1050 | C-O (ester) | Stretching (asym) |
| 11 | 880 | R=CH (cis or trans) | Bending out of plate |
| 12 | 725 | R=CH2 or R-(CH2)n | Rocking (overlapping) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ferreira, R.; Lourenço, S.; Lopes, A.; Andrade, C.; Câmara, J.S.; Castilho, P.; Perestrelo, R. Evaluation of Fatty Acids Profile as a Useful Tool towards Valorization of By-Products of Agri-Food Industry. Foods 2021, 10, 2867. https://doi.org/10.3390/foods10112867
Ferreira R, Lourenço S, Lopes A, Andrade C, Câmara JS, Castilho P, Perestrelo R. Evaluation of Fatty Acids Profile as a Useful Tool towards Valorization of By-Products of Agri-Food Industry. Foods. 2021; 10(11):2867. https://doi.org/10.3390/foods10112867
Chicago/Turabian StyleFerreira, Rui, Sílvia Lourenço, André Lopes, Carlos Andrade, José S. Câmara, Paula Castilho, and Rosa Perestrelo. 2021. "Evaluation of Fatty Acids Profile as a Useful Tool towards Valorization of By-Products of Agri-Food Industry" Foods 10, no. 11: 2867. https://doi.org/10.3390/foods10112867
APA StyleFerreira, R., Lourenço, S., Lopes, A., Andrade, C., Câmara, J. S., Castilho, P., & Perestrelo, R. (2021). Evaluation of Fatty Acids Profile as a Useful Tool towards Valorization of By-Products of Agri-Food Industry. Foods, 10(11), 2867. https://doi.org/10.3390/foods10112867








