Analysis of Major Polyphenolic Compounds of Cydonia oblonga Miller (Quince) Fruit Extract by UPLC-MS/MS and Its Effect on Adipogenesis in 3T3-L1 Cells
Abstract
:1. Introduction
2. Materials and Methods
2.1. Reagents and Standards
2.2. Extraction of Polyphenols
2.3. Purification of Polyphenols
2.4. Determination of Total Phenolic Contents (TPC) and Total Flavonoid Contents (TFC)
2.5. Antioxidant Activity
2.6. Standard Solution Preparation for UPLC MS-MS Analysis
2.7. Sample Solution Preparation for UPLC-MS/MS Analysis
2.8. UPLC-MS/MS Analysis
2.9. Method Validation
2.9.1. Calibration Curves, Limits of Detection (LOD), and Limits of Quantification (LOQ)
2.9.2. Precision, Repeatability, and Stability
2.9.3. Recovery
2.10. Identification and Quantification
2.11. Cell Culture
2.11.1. Cell Viability Assay
2.11.2. Oil Red O Staining
2.12. Statistical Analysis
3. Results and Discussion
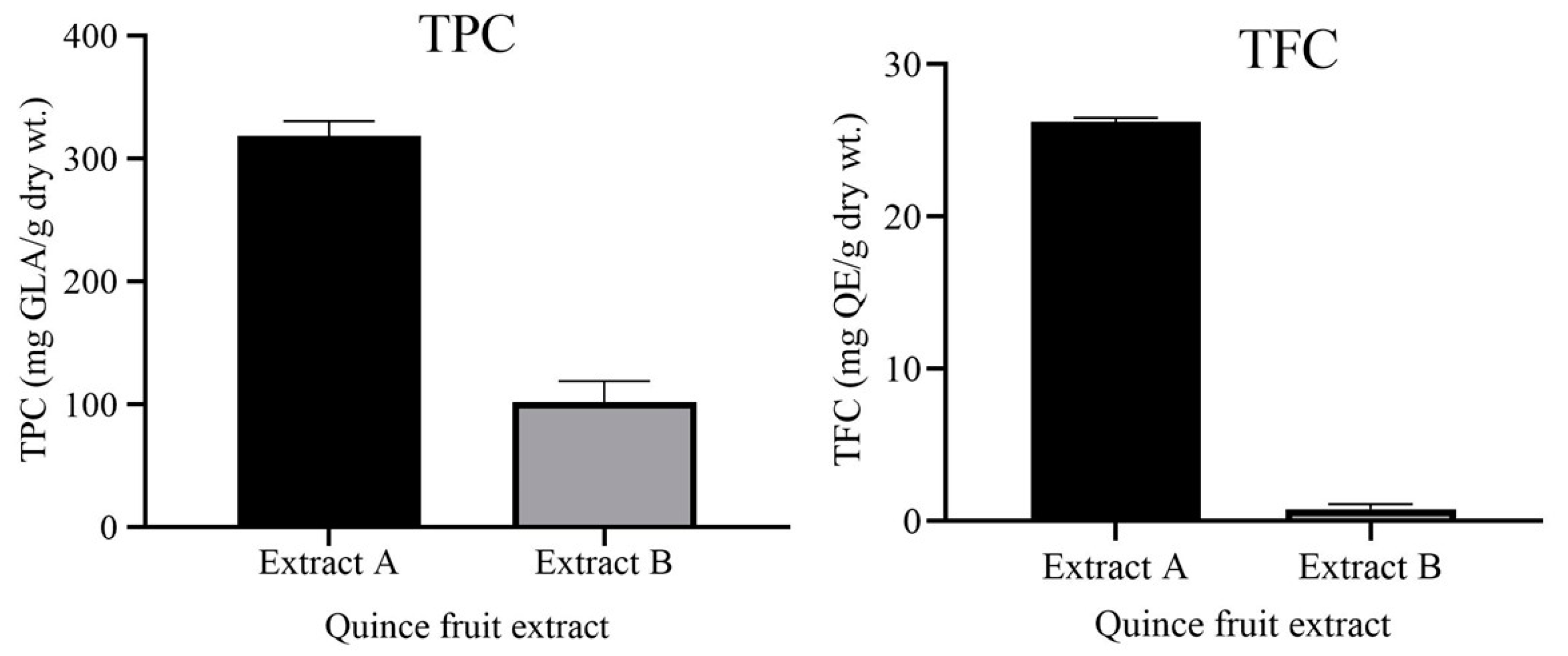
3.1. Total Phenolic Contents (TPC) and Total Flavonoid Contents (TFC)
3.2. Antioxidant Activity Assay
3.3. Optimization of the Chromatography and Mass Spectrometry System
3.4. Validation of the Method
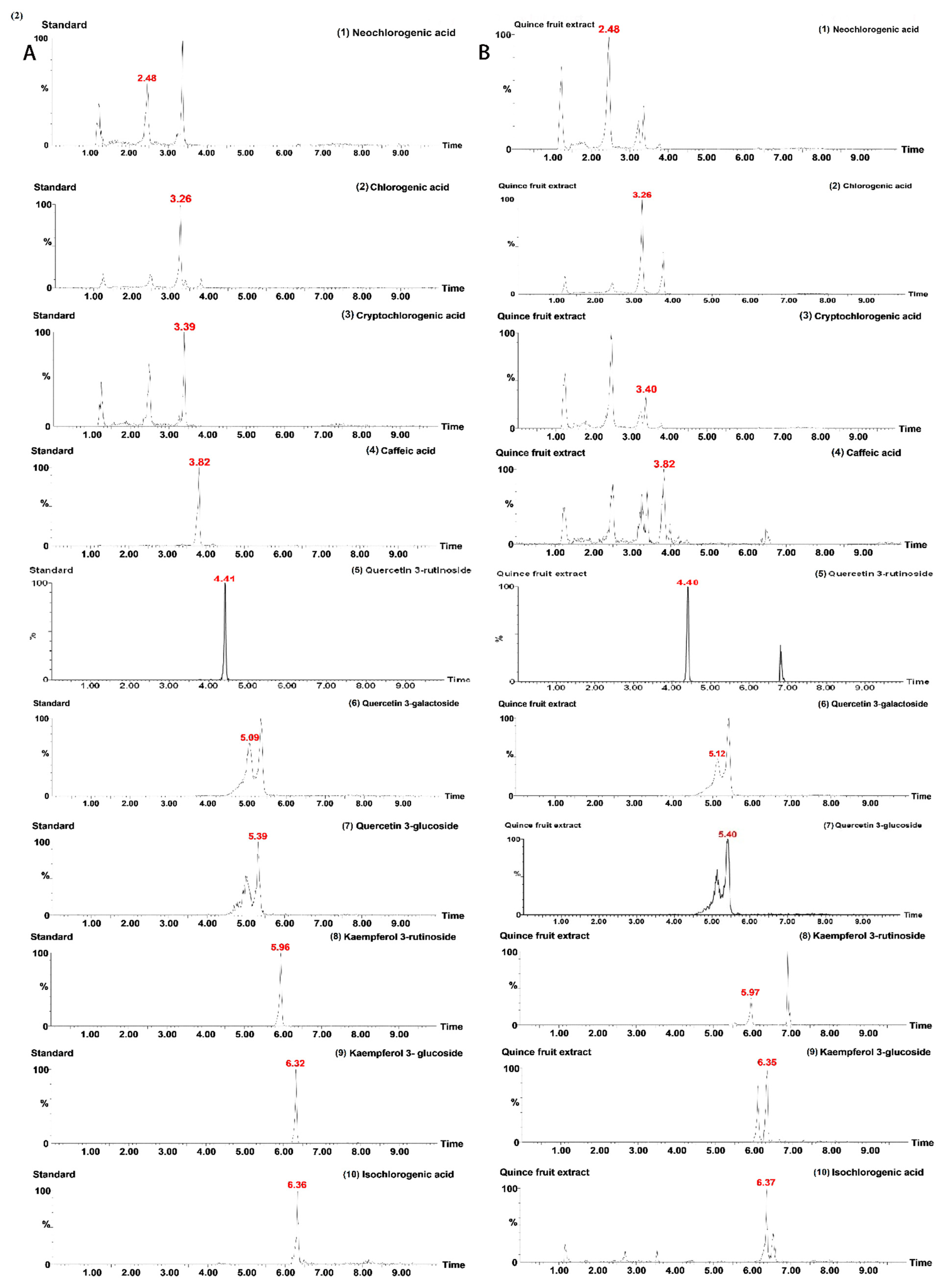
3.5. Quantitative Analysis of Quince Purified and Unpurified Extract
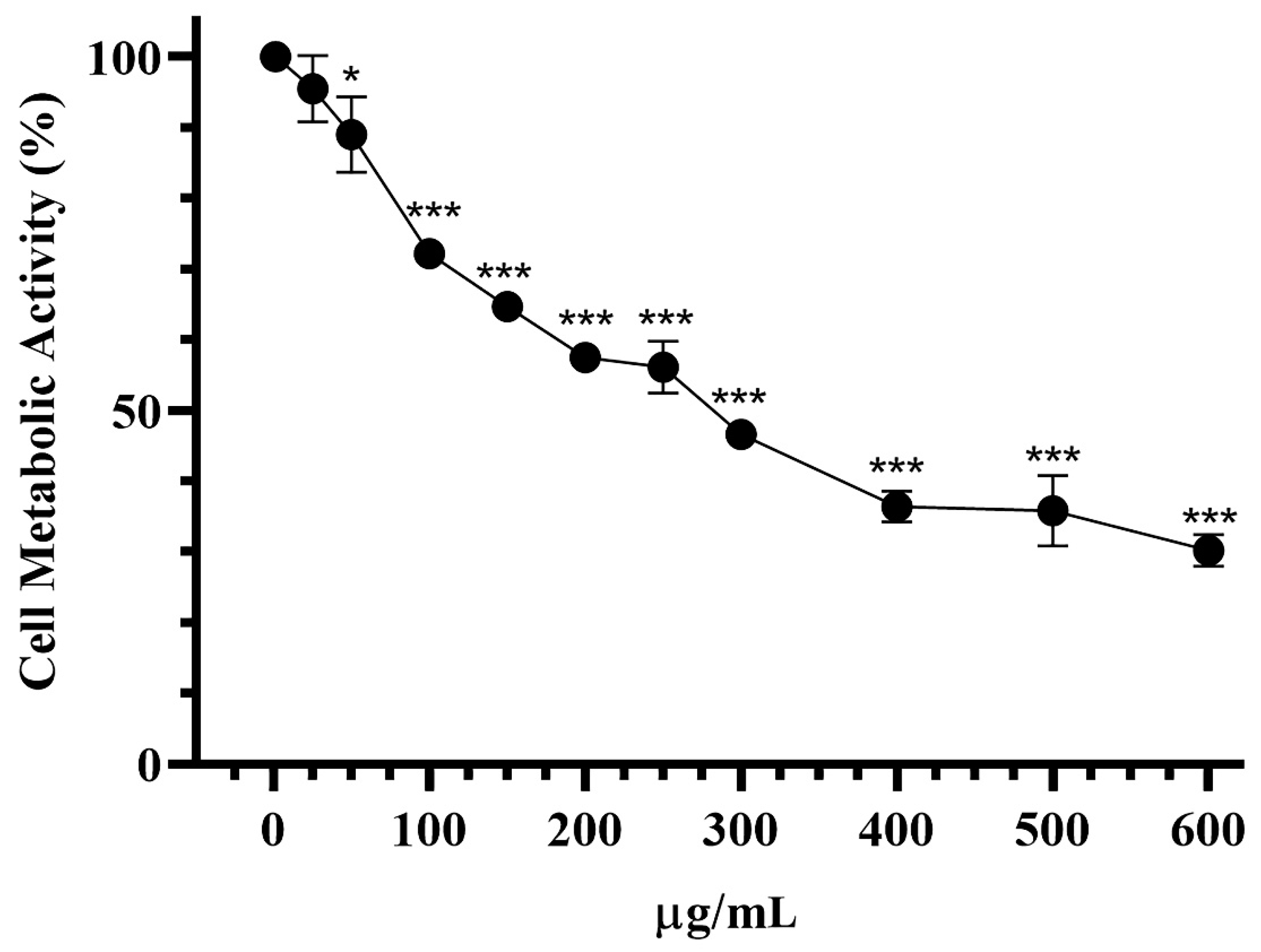
3.6. Effect on Cell Viability
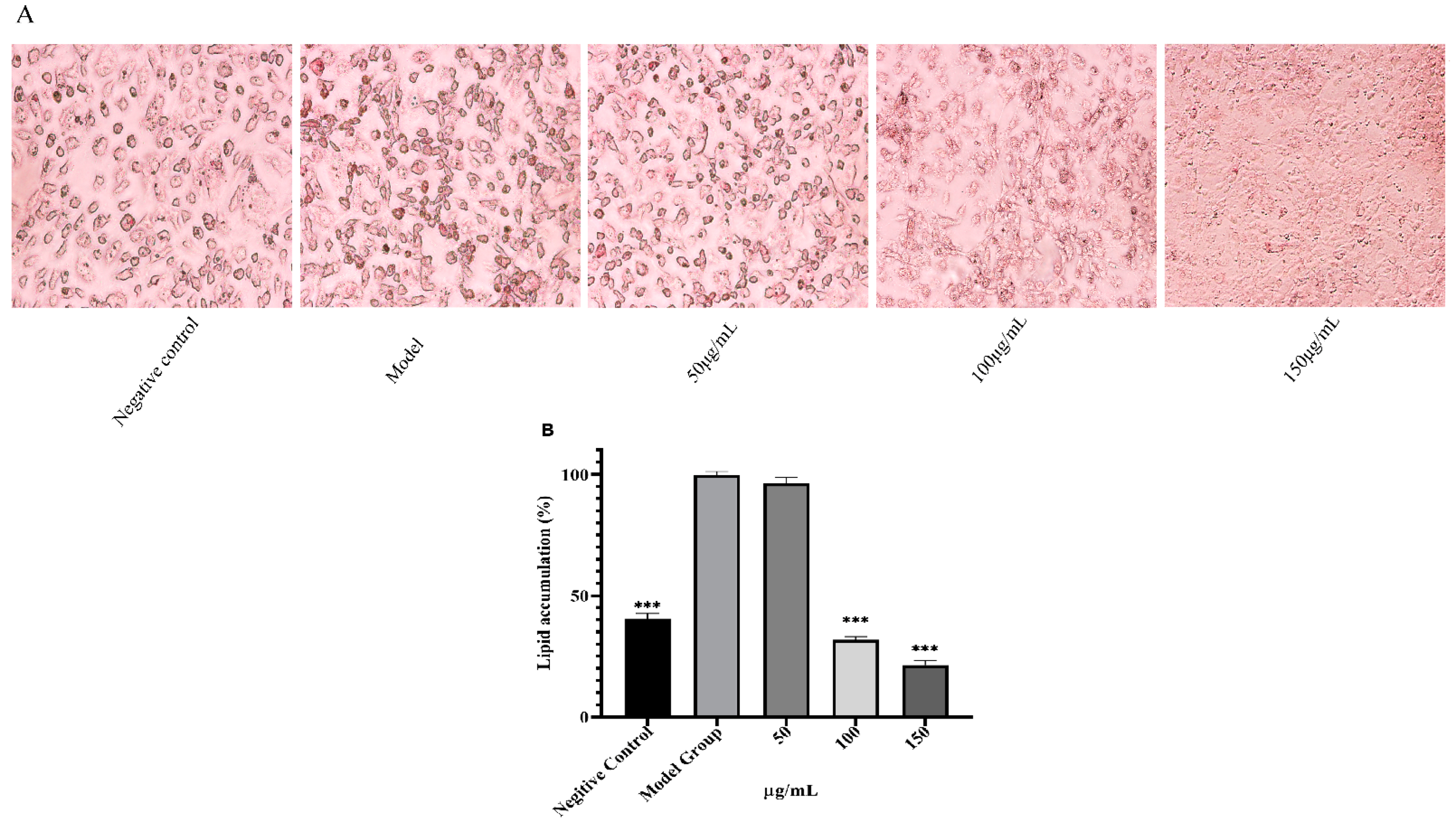
3.7. Effect on Adipogenesis
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Piątczak, E.; Dybowska, M.; Płuciennik, E.; Kośla, K.; Kolniak-Ostek, J.; Kalinowska-Lis, U. Identification and accumulation of phenolic compounds in the leaves and bark of Salix alba (L.) and their biological potential. Biomolecules 2020, 10, 1391. [Google Scholar] [CrossRef]
- Pye, C.R.; Bertin, M.J.; Lokey, R.S.; Gerwick, W.H.; Linington, R.G. Retrospective analysis of natural products provides insights for future discovery trends. Proc. Natl. Acad. Sci. USA 2017, 114, 5601–5606. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Smith-Hall, C.; Larsen, H.O.; Pouliot, M. People, plants and health: A conceptual framework for assessing changes in medicinal plant consumption. J. Ethnobiol. Ethnomed. 2012, 8, 43. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Süntar, I. Importance of ethnopharmacological studies in drug discovery: Role of medicinal plants. Phytochem. Rev. 2020, 19, 1199–1209. [Google Scholar] [CrossRef]
- Zhang, J.; Onakpoya, I.J.; Posadzki, P.; Eddouks, M. The safety of herbal medicine: From prejudice to evidence. Altern. Med. 2015, 2015, 316706. [Google Scholar] [CrossRef] [Green Version]
- Ruhsam, M.; Hollingsworth, P.M. Authentication of Eleutherococcus and Rhodiola herbal supplement products in the United Kingdom. J. Pharm. Biomed. Anal. 2018, 149, 403–409. [Google Scholar] [CrossRef]
- Ji, S.; Fattahi, A.; Raffel, N.; Hoffmann, I.; Beckmann, M.W.; Dittrich, R.; Schrauder, M. Antioxidant effect of aqueous extract of four plants with therapeutic potential on gynecological diseases; Semen persicae, Leonurus cardiaca, Hedyotis diffusa, and Curcuma zedoaria. Eur. J. Med. Res. 2017, 22, 50. [Google Scholar] [CrossRef] [Green Version]
- Thomford, N.E.; Senthebane, D.A.; Rowe, A.; Munro, D.; Seele, P.; Maroyi, A.; Dzobo, K. Natural products for drug discovery in the 21st century: Innovations for novel drug discovery. Int. J. Mol. Sci. 2018, 19, 1578. [Google Scholar] [CrossRef] [Green Version]
- Kabera, J.N.; Semana, E.; Mussa, A.R.; He, X. Plant secondary metabolites: Biosynthesis, classification, function and pharmacological properties. J. Pharm. Pharmacol. 2014, 2, 377–392. [Google Scholar]
- Swallah, M.S.; Sun, H.; Affoh, R.; Fu, H.; Yu, H. Antioxidant Potential Overviews of Secondary Metabolites (Polyphenols) in Fruits. Int. J. Food Sci. 2020, 2020, 9081686. [Google Scholar] [CrossRef]
- Maaliki, D.; Shaito, A.A.; Pintus, G.; El-Yazbi, A.; Eid, A.H. Flavonoids in hypertension: A brief review of the underlying mechanisms. Curr. Opin. Pharmacol. 2019, 45, 57–65. [Google Scholar] [CrossRef] [PubMed]
- Bahramsoltani, R.; Ebrahimi, F.; Farzaei, M.H.; Baratpourmoghaddam, A.; Ahmadi, P.; Rostamiasrabadi, P.; Rasouli Amirabadi, A.H.; Rahimi, R. Dietary polyphenols for atherosclerosis: A comprehensive review and future perspectives. Crit. Rev. Food Sci. Nutr. 2019, 59, 114–132. [Google Scholar] [CrossRef] [PubMed]
- Ji, J.; Yang, X.; Flavel, M.; Shields, Z.P.-I.; Kitchen, B. Antioxidant and anti-diabetic functions of a polyphenol-rich sugarcane extract. J. Am. Coll. Nutr. 2019, 38, 670–680. [Google Scholar] [CrossRef] [PubMed]
- Selvakumar, P.; Badgeley, A.; Murphy, P.; Anwar, H.; Sharma, U.; Lawrence, K.; Lakshmikuttyamma, A. Flavonoids and other polyphenols act as epigenetic modifiers in breast cancer. Nutrients 2020, 12, 761. [Google Scholar] [CrossRef] [Green Version]
- Jeong, H.; Phan, A.N.; Choi, J.-W. Anti-cancer effects of polyphenolic compounds in epidermal growth factor receptor tyrosine kinase inhibitor-resistant non-small cell lung cancer. Pharmacogn. Mag. 2017, 13, 595. [Google Scholar]
- Rahaiee, S.; Assadpour, E.; Esfanjani, A.F.; Silva, A.S.; Jafari, S.M. Application of nano/microencapsulated phenolic compounds against cancer. Adv. Colloid Interface Sci. 2020, 279, 102153. [Google Scholar] [CrossRef]
- Abdollahi, H. A review on history, domestication and germplasm collections of quince (Cydonia oblonga Mill.) in the World. Genet. Resour. Crop Evol. 2019, 66, 1041–1058. [Google Scholar] [CrossRef]
- Ashraf, M.U.; Muhammad, G.; Hussain, M.A.; Bukhari, S.N. Cydonia oblonga M., A Medicinal Plant Rich in Phytonutrients for Pharmaceuticals. Front. Pharm. 2016, 7, 163. [Google Scholar] [CrossRef] [Green Version]
- Silva, B.M.; Andrade, P.B.; Ferreres, F.; Domingues, A.L.; Seabra, R.M.; Ferreira, M.A. Phenolic profile of quince fruit (Cydonia oblonga Miller)(pulp and peel). J. Agric. Food Chem. 2002, 50, 4615–4618. [Google Scholar] [CrossRef]
- Wojdyło, A.; Oszmiański, J.; Bielicki, P. Polyphenolic composition, antioxidant activity, and polyphenol oxidase (PPO) activity of quince (Cydonia oblonga Miller) varieties. J. Agric. Food Chem. 2013, 61, 2762–2772. [Google Scholar] [CrossRef]
- Minaiyan, M.; Ghannadi, A.; Etemad, M.; Mahzouni, P. A study of the effects of Cydonia oblonga Miller (Quince) on TNBS-induced ulcerative colitis in rats. Res. Pharm. Sci. 2012, 7, 103. [Google Scholar] [PubMed]
- Zhou, W.; Abdurahman, A.; Umar, A.; Iskander, G.; Abdusalam, E.; Berké, B.; Bégaud, B.; Moore, N. Effects of Cydonia oblonga Miller extracts on blood hemostasis, coagulation and fibrinolysis in mice, and experimental thrombosis in rats. J. Ethnopharmacol. 2014, 154, 163–169. [Google Scholar] [CrossRef] [PubMed]
- Aliasl, F.; Toliyat, T.; Mohammadi, A.; Minaee, B.; Samadi, N.; Aliasl, J.; Sadeghpour, O. Medicinal properties of Cydonia oblonga mill fruit (pulp and peel) in Iranian traditional medicine and modern phytothrapy. Tradit. Integr. Med. 2016, 1, 122–128. [Google Scholar]
- Pacifico, S.; Gallicchio, M.; Fiorentino, A.; Fischer, A.; Meyer, U.; Stintzing, F.C. Antioxidant properties and cytotoxic effects on human cancer cell lines of aqueous fermented and lipophilic quince (Cydonia oblonga Mill.) preparations. Food Chem. Toxicol. 2012, 50, 4130–4135. [Google Scholar] [CrossRef]
- Essafi-Benkhadir, K.; Refai, A.; Riahi, I.; Fattouch, S.; Karoui, H.; Essafi, M. Quince (Cydonia oblonga Miller) peel polyphenols modulate LPS-induced inflammation in human THP-1-derived macrophages through NF-κB, p38MAPK and Akt inhibition. Biochem. Biophys. Res. Commun. 2012, 418, 180–185. [Google Scholar] [CrossRef]
- Adiban, H.; Shirazi, F.H.; Gholami, S.; Kamalinejad, M.; Hosseini, S.H.; Noubarani, M.; Eskandari, M.R. Chemopreventive effect of quince (Cydonia oblonga Mill.) fruit extract on hepatocellular carcinoma induced by diethylnitrosamine in rats. Int. Pharm. Acta 2019, 2, e2. [Google Scholar]
- Parvan, M.; Sajjadi, S.-E.; Minaiyan, M. Protective effect of two extracts of Cydonia oblonga Miller (quince) fruits on gastric ulcer induced by indomethacin in rats. Int. J. Prev. Med. 2017, 8, 58. [Google Scholar]
- Aslam, M.; Sial, A.A. Effect of hydroalcoholic extract of cydonia oblonga miller (Quince) on sexual behaviour of wistar rats. Adv. Pharmacol. Sci. 2014, 2014, 282698. [Google Scholar] [CrossRef] [Green Version]
- Zhou, W.; Abdusalam, E.; Abliz, P.; Reyim, N.; Tian, S.; Aji, Q.; Issak, M.; Iskandar, G.; Moore, N.; Umar, A. Effect of Cydonia oblonga Mill. fruit and leaf extracts on blood pressure and blood rheology in renal hypertensive rats. J. Ethnopharmacol. 2014, 152, 464–469. [Google Scholar] [CrossRef]
- Umar, A.; Iskandar, G.; Aikemu, A.; Yiming, W.; Zhou, W.; Berké, B.; Begaud, B.; Moore, N. Effects of Cydonia oblonga Miller leaf and fruit flavonoids on blood lipids and anti-oxydant potential in hyperlipidemia rats. J. Ethnopharmacol. 2015, 169, 239–243. [Google Scholar] [CrossRef]
- Di Sotto, A.; Vecchiato, M.; Abete, L.; Toniolo, C.; Giusti, A.M.; Mannina, L.; Locatelli, M.; Nicoletti, M.; Di Giacomo, S. Capsicum annuum L. var. Cornetto di Pontecorvo PDO: Polyphenolic profile and in vitro biological activities. J. Funct. Foods 2018, 40, 679–691. [Google Scholar] [CrossRef]
- Di Sotto, A.; Durazzi, F.; Sarpietro, M.G.; Mazzanti, G. Antimutagenic and antioxidant activities of some bioflavours from wine. Food Chem. Toxicol. 2013, 60, 141–146. [Google Scholar] [CrossRef] [PubMed]
- Kraus, N.A.; Ehebauer, F.; Zapp, B.; Rudolphi, B.; Kraus, B.J.; Kraus, D. Quantitative assessment of adipocyte differentiation in cell culture. Adipocyte 2016, 5, 351–358. [Google Scholar] [CrossRef] [Green Version]
- Arora, I.; Sharma, M.; Tollefsbol, T.O. Combinatorial epigenetics impact of polyphenols and phytochemicals in cancer prevention and therapy. Int. J. Mol. Sci. 2019, 20, 4567. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Li, X.; Lin, J.; Gao, Y.; Han, W.; Chen, D. Antioxidant activity and mechanism of Rhizoma Cimicifugae. Chem. Cent. J. 2012, 6, 140. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ak, T.; Gülçin, İ. Antioxidant and radical scavenging properties of curcumin. Chem.-Biol. Interact. 2008, 174, 27–37. [Google Scholar] [CrossRef]
- Wojdyło, A.; Oszmiański, J.; Czemerys, R. Antioxidant activity and phenolic compounds in 32 selected herbs. Food Chem. 2007, 105, 940–949. [Google Scholar] [CrossRef]
- Podsędek, A.; Zakłos-Szyda, M.; Polka, D.; Sosnowska, D. Effects of Viburnum opulus fruit extracts on adipogenesis of 3T3-L1 cells and lipase activity. J. Funct. Foods 2020, 73, 104111. [Google Scholar] [CrossRef]
- Hadrich, F.; Sayadi, S. Apigetrin inhibits adipogenesis in 3T3-L1 cells by downregulating PPARγ and CEBP-α. Lipids Health Dis. 2018, 17, 95. [Google Scholar] [CrossRef] [Green Version]
- Aranaz, P.; Navarro-Herrera, D.; Zabala, M.; Miguéliz, I.; Romo-Hualde, A.; López-Yoldi, M.; Martínez, J.A.; Vizmanos, J.L.; Milagro, F.I.; González-Navarro, C.J. Phenolic compounds inhibit 3T3-L1 adipogenesis depending on the stage of differentiation and their binding affinity to PPARγ. Molecules 2019, 24, 1045. [Google Scholar] [CrossRef] [Green Version]
- Sudhakar, M.; Sasikumar, S.J.; Silambanan, S.; Natarajan, D.; Ramakrishnan, R.; Nair, A.J.; Kiran, M.S. Chlorogenic acid promotes development of brown adipocyte-like phenotype in 3T3-L1 adipocytes. J. Funct. Foods 2020, 74, 104161. [Google Scholar] [CrossRef]
- Kong, L.; Xu, M.; Qiu, Y.; Liao, M.; Zhang, Q.; Yang, L.; Zheng, G. Chlorogenic acid and caffeine combination attenuates adipogenesis by regulating fat metabolism and inhibiting adipocyte differentiation in 3T3-L1 cells. J. Food Biochem. 2021, 45, e13795. [Google Scholar] [CrossRef] [PubMed]
- Jiang, H.; Horiuchi, Y.; Hironao, K.-y.; Kitakaze, T.; Yamashita, Y.; Ashida, H. Prevention effect of quercetin and its glycosides on obesity and hyperglycemia through activating AMPKα in high-fat diet-fed ICR mice. J. Clin. Biochem. Nutr. 2020, 67, 20–47. [Google Scholar] [CrossRef] [PubMed]
- Jang, Y.S.; Wang, Z.; Lee, J.-M.; Lee, J.-Y.; Lim, S.S. Screening of Korean natural products for anti-adipogenesis properties and isolation of kaempferol-3-O-rutinoside as a potent anti-adipogenetic compound from Solidago virgaurea. Molecules 2016, 21, 226. [Google Scholar] [CrossRef] [Green Version]
- Abliz, A.; Aji, Q.; Abdusalam, E.; Sun, X.; Abdurahman, A.; Zhou, W.; Moore, N.; Umar, A. Effect of Cydonia oblonga Mill. leaf extract on serum lipids and liver function in a rat model of hyperlipidaemia. J. Ethnopharmacol. 2014, 151, 970–974. [Google Scholar] [CrossRef]

| Antioxidant Assay | Purified Extract | Unpurified Extract | Positive Control |
|---|---|---|---|
| DPPH # scavenging activity | 3.316 ± 0.34 *** | 62.32 ± 0.20 *** | 1.189 ± 0.13 |
| ABTS # scavenging activity | 36.38 ± 0.42 *** | 69.59 ± 0.37 *** | 15.74 ± 0.21 |
| No. | Analytes | RT a (min) | Ion Mode | Precursor Ion (m/z) b | Product Ions (m/z) b | Cone Voltage (cV) | Collision Energy (cE) |
|---|---|---|---|---|---|---|---|
| 1 | Neochlorogenic acid | 2.48 | ESI- | 353.096 | 190.93, 134.958 | 38 | 18 |
| 2 | Chlorogenic acid | 3.26 | ESI- | 353.096 | 190.93, 134.958 | 38 | 32 |
| 3 | Cryptochlorogenic acid | 3.39 | ESI- | 353 | 191, 135 | 40 | 25 |
| 4 | Caffeic acid | 3.82 | ESI- | 179 | 135, 107 | 54 | 20 |
| 5 | Quercetin 3-rutinoside | 4.40 | ESI- | 609 | 271,300 | 66 | 65 |
| 6 | Quercetin 3-galactoside | 5.09 | ESI- | 463.16 | 299.988, 270.909 | 8 | 28 |
| 7 | Quercetin 3-glucoside | 5.39 | ESI- | 463 | 271, 300 | 8 | 36 |
| 8 | Kaempferol 3-rutinoside | 5.96 | ESI- | 593.149 | 254.955, 284.885 | 78 | 66 |
| 9 | Kaempferol 3-glucoside | 6.32 | ESI- | 477 | 255, 248 | 58 | 38 |
| 10 | Isochlorogenic acid | 6.36 | ESI- | 515 | 353, 191 | 50 | 15 |
| No. | Analytes | Calibration Curves a | R2 | Range (ng/mL) | LOD (ng/mL) | LOQ (ng/mL) |
|---|---|---|---|---|---|---|
| 1 | Neochlorogenic acid | y = 75.69x − 6838 | 0.998 | 19.53–10,000 | 0.87 | 2.65 |
| 2 | Chlorogenic acid | y = 122.68x + 24332 | 0.999 | 29.3–15,000 | 0.58 | 1.76 |
| 3 | Cryptochlorogenic acid | y = 19.976x − 2298.1 | 0.998 | 19.53–10,000 | 1.65 | 4.98 |
| 4 | Caffeic acid | y = 64.629x + 235 | 0.999 | 2.92–1500 | 1.64 | 4.99 |
| 5 | Quercetin 3-rutinoside | y = 176.09x + 12675 | 0.999 | 24.41–12,500 | 0.42 | 1.28 |
| 6 | Quercetin 3-galactoside | y = 157.88x + 1499 | 0.999 | 19.53–10,000 | 1.48 | 4.50 |
| 7 | Quercetin 3-glucoside | y = 176.52x + 12273 | 0.997 | 19.53–10,000 | 0.55 | 1.69 |
| 8 | Kaempferol 3-rutinoside | y = 352.82x + 24426 | 0.996 | 7.32–3750 | 0.11 | 0.34 |
| 9 | Kaempferol 3-glucoside | y = 342.62x + 12438 | 0.996 | 7.32–3750 | 0.49 | 1.47 |
| 10 | Isochlorogenic acid | y = 27.534x − 2162.3 | 0.999 | 14.64–7500 | 1.27 | 3.85 |
| No. | Analytes | Precision a | Repeatability | Stability | |
|---|---|---|---|---|---|
| Intra-Day | Inter-Day | ||||
| RSD b % (n = 6) | RSD % (n = 3) | RSD % (n = 6) | RSD % (n = 3) | ||
| 1 | Neochlorogenic acid | 1.12 | 3.37 | 1.93 | 2.31 |
| 2 | Chlorogenic acid | 1.84 | 2.69 | 1.84 | 2.79 |
| 3 | Cryptochlorogenic acid | 1.88 | 2.58 | 1.78 | 2.55 |
| 4 | Caffeic acid | 0.91 | 1.17 | 1.89 | 3.53 |
| 5 | Quercetin 3-rutinoside | 1.38 | 2.18 | 1.60 | 2.93 |
| 6 | Quercetin 3-galactoside | 1.61 | 3.26 | 1.52 | 3.97 |
| 7 | Quercetin 3-glucoside | 1.31 | 2.66 | 2.14 | 3.01 |
| 8 | Kaempferol 3-rutinoside | 2.97 | 3.41 | 2.59 | 3.37 |
| 9 | Kaempferol 3-glucoside | 2.45 | 1.90 | 2.57 | 3.05 |
| 10 | Isochlorogenic acid | 1.68 | 2.46 | 2.26 | 3.00 |
| No. | Compounds | Low (n = 3) | Medium (n = 3) | High (n = 3) | |||
|---|---|---|---|---|---|---|---|
| Recovery (%) a | RSD (%) | Recovery (%) | RSD (%) | Recovery (%) | RSD (%) | ||
| 1 | Neochlorogenic acid | 96.44 | 0.91 | 96.65 | 0.47 | 97.41 | 0.87 |
| 2 | Chlorogenic acid | 98.3 | 0.09 | 98.42 | 1.27 | 98.82 | 0.05 |
| 3 | Cryptochlorogenic acid | 99.55 | 0.97 | 99.56 | 0.03 | 99.95 | 0.33 |
| 4 | Caffeic acid | 96.61 | 1.51 | 97.09 | 0.65 | 97.58 | 0.35 |
| 5 | Quercetin 3-rutinoside | 97.54 | 1.33 | 97.81 | 1.07 | 97.98 | 0.52 |
| 6 | Quercetin 3-galactoside | 98.72 | 0.99 | 99.03 | 0.53 | 99.08 | 0.62 |
| 7 | Quercetin 3-glucoside | 98.48 | 0.65 | 98.59 | 1.06 | 98.71 | 1.50 |
| 8 | Kaempferol 3-rutinoside | 96.31 | 0.71 | 96.74 | 0.38 | 96.69 | 0.69 |
| 9 | Kaempferol 3-glucoside | 95.6 | 1.88 | 96.17 | 2.21 | 96.62 | 2.25 |
| 10 | Isochlorogenic acid | 97.56 | 2.11 | 97.73 | 0.52 | 97.81 | 0.57 |
| No. | Compound | Purified Extract | Unpurified Extract |
|---|---|---|---|
| 1 | Neochlorogenic acid | 56.77 ± 1.48 | 1.16 ± 0.01 |
| 2 | Chlorogenic acid | 160.67 ± 2.51 | 2.28 ± 0.01 |
| 3 | Cryptochlorogenic acid | 242.44 ± 0.73 | 3.37 ± 0.01 |
| 4 | Caffeic acid | 1.46 ± 0.05 | 0.03 ± 0.00 |
| 5 | Quercetin 3-rutinoside | 186.33 ± 1.32 | 3.13 ± 0.04 |
| 6 | Quercetin 3-galactoside | 40.24 ± 1.46 | 0.34 ± 0.01 |
| 7 | Quercetin 3-glucoside | 26.72 ± 0.82 | 0.22 ± 0.00 |
| 8 | Kaempferol 3-rutinoside | 3.10 ± 0.04 | 0.02 ± 0.00 |
| 9 | Kaempferol 3-glucoside | 1.92 ± 0.06 | 0.02 ± 0.00 |
| 10 | Isochlorogenic acid | 21.74 ± 0.57 | 0.05 ± 0.01 |
| Total (Ʃ) | 741.41 ± 9.04 | 10.61 ± 0.09 | |
| No. | Contents | Purified Extract | Unpurified Extract |
|---|---|---|---|
| 1 | Phenolic acids | 483.10 ± 5.16 | 7.89 ± 0.02 |
| 2 | Flavonoids | 258.32 ± 3.02 | 2.73 ± 0.03 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Khan, N.; Maihemuti, N.; Nuer, M.; Abudurousuli, K.; Simayi, J.; Talihati, Z.; Han, M.; Hailati, S.; Zhou, W.; Wumaier, A. Analysis of Major Polyphenolic Compounds of Cydonia oblonga Miller (Quince) Fruit Extract by UPLC-MS/MS and Its Effect on Adipogenesis in 3T3-L1 Cells. Separations 2022, 9, 167. https://doi.org/10.3390/separations9070167
Khan N, Maihemuti N, Nuer M, Abudurousuli K, Simayi J, Talihati Z, Han M, Hailati S, Zhou W, Wumaier A. Analysis of Major Polyphenolic Compounds of Cydonia oblonga Miller (Quince) Fruit Extract by UPLC-MS/MS and Its Effect on Adipogenesis in 3T3-L1 Cells. Separations. 2022; 9(7):167. https://doi.org/10.3390/separations9070167
Chicago/Turabian StyleKhan, Nawaz, Nulibiya Maihemuti, Muhadaisi Nuer, Kayisaier Abudurousuli, Jimilihan Simayi, Ziruo Talihati, Mengyuan Han, Sendaer Hailati, Wenting Zhou, and Ainiwaer Wumaier. 2022. "Analysis of Major Polyphenolic Compounds of Cydonia oblonga Miller (Quince) Fruit Extract by UPLC-MS/MS and Its Effect on Adipogenesis in 3T3-L1 Cells" Separations 9, no. 7: 167. https://doi.org/10.3390/separations9070167
APA StyleKhan, N., Maihemuti, N., Nuer, M., Abudurousuli, K., Simayi, J., Talihati, Z., Han, M., Hailati, S., Zhou, W., & Wumaier, A. (2022). Analysis of Major Polyphenolic Compounds of Cydonia oblonga Miller (Quince) Fruit Extract by UPLC-MS/MS and Its Effect on Adipogenesis in 3T3-L1 Cells. Separations, 9(7), 167. https://doi.org/10.3390/separations9070167






