Inorganic–Organic Hybrid Polymer for Fine-Rich Coal Slime Water Treatment: Performance and Interfacial Adsorption Mechanism on Kaolinite Aluminol Surface
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Synthesis of Al-PAM
2.3. Molecular Weight Determination of Al-PAM
2.4. Fourier Transform Infrared Spectroscopy (FTIR) Measurement
2.5. Settling Tests
2.6. Quartz Crystal Microbalance with Dissipation (QCM-D) Measurement
2.7. Microscopic Imaging of Floc Structure
3. Results and Discussion
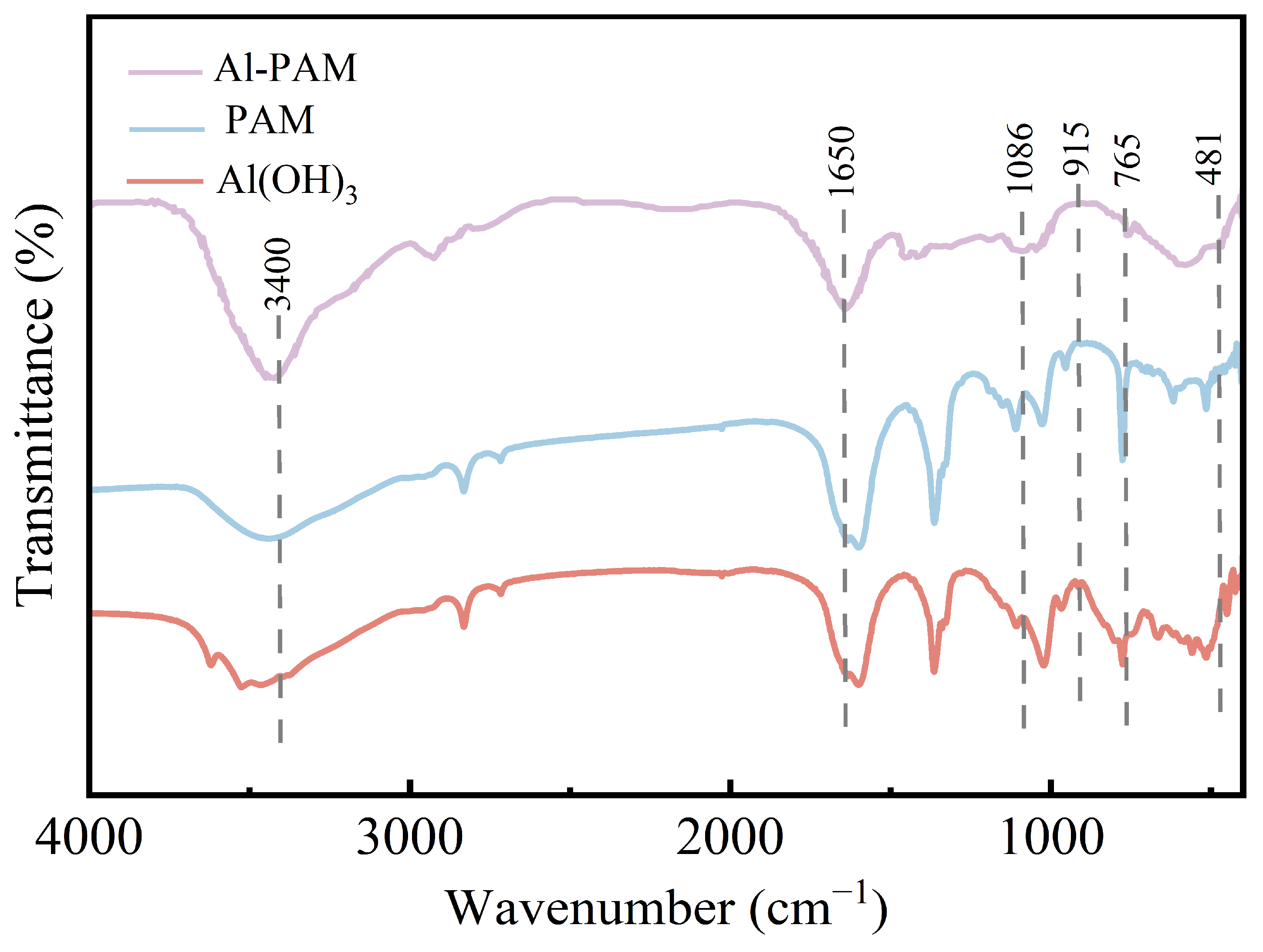
3.1. Characterization of Synthesized Al-PAM
3.2. Settling Performance
3.2.1. Supernatant Turbidity and pH
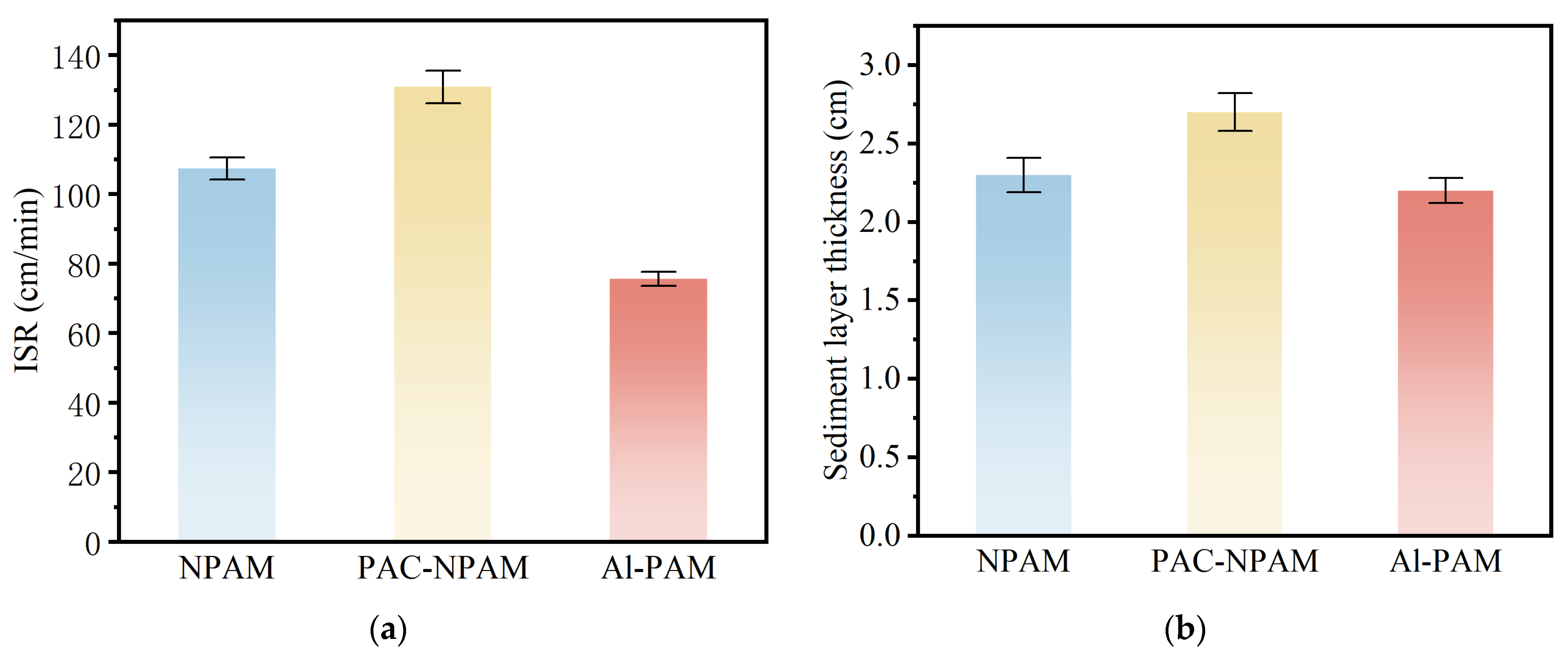
3.2.2. Settling Velocity and Sediment Layer Thickness
3.3. Floc Structure
3.3.1. PAC Flocs
3.3.2. NPAM Flocs
3.3.3. PAC + NPAM Flocs
3.3.4. Al-PAM Flocs
3.4. Adsorption Mechanisms
3.4.1. Adsorption of PAC
3.4.2. Adsorption of NPAM
3.4.3. Adsorption of PAC + NPAM
3.4.4. Adsorption of Al-PAM
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
Abbreviations
| Al-PAM | Aluminum hydroxide-polyacrylamide |
| PAC | Polyaluminum chloride |
| PAM | Polyacrylamide |
| QCM-D | Quartz crystal microbalance with dissipation monitoring |
| SDS | Sodium dodecyl sulfate |
| Al(OH)3-p(NIPAM-co-DMAPMA) | Aluminum Hydroxide-poly(N-isopropylacrylamide-co-N,N-dimethylaminoethyl methacrylate) |
References
- Saletnik, A.; Saletnik, B.; Puchalski, C. Coal as the world’s dominant energy source and its role in the energy transformation and regulations of European Green Deal. J. Environ. Manag. 2025, 392, 126815. [Google Scholar] [CrossRef]
- IEA. Coal Analysis and Forecast to 2027. Available online: https://www.repower.world/news/iea-coal-analysis-and-forecast-to-2027-a-summary (accessed on 15 January 2026).
- Jones, A. Coal’s Share in Global Generation Mix to Slip. Available online: https://www.argusmedia.com/en/news-and-insights/latest-market-news/2786338-coal-s-share-in-global-generation-mix-to-slip-iea (accessed on 10 January 2026).
- Sabah, E.; Erkan, Z.E. Interaction mechanism of flocculants with coal waste slurry. Fuel 2006, 85, 350–359. [Google Scholar] [CrossRef]
- Li, L.; Zhang, X.; Luo, J.; Hu, B. Theoretical Analysis of the Movement Law of Top Coal and Overburden in a Fully Mechanized Top-Coal Caving Face with a Large Mining Height. Processes 2022, 10, 2596. [Google Scholar] [CrossRef]
- Chen, X.; Peng, Y. Managing clay minerals in froth flotation—A critical review. Miner. Process. Extr. Metall. Rev. 2018, 39, 289–307. [Google Scholar] [CrossRef]
- Fan, Y.; Ruan, C.; Chang, M.; Dong, X.; Ma, X.; Ye, G. Effect of settling vortex of coal slime flocs with different sizes on the settlement of microfine particles. Powder Technol. 2025, 452, 120422. [Google Scholar] [CrossRef]
- Pan, F.; Ding, S.; Zou, H. Research status of genesis analysis and treatment technology of difficult-to-settle slime water. Coal Process. Compr. Util. 2023, 3, 9–14. [Google Scholar] [CrossRef]
- Wang, L.; Min, F.; Sun, K.; Chen, J.; Cheng, Y.; Song, C. Exploration of interaction between different types of flocculants and coal particles based on experiments and simulations. Surf. Interfaces 2025, 59, 105903. [Google Scholar] [CrossRef]
- Fan, Y.; Chang, T.; Zhang, J.; Xu, H.; Cui, J.; Tian, H.; Yin, Y. Research on influence of CPAM ionic degree on settling characteristics of high muddied coal slime water. Coal Sci. Technol. 2023, 51, 251–260. [Google Scholar] [CrossRef]
- Nguyen, C.V.; Nguyen, A.V.; Doi, A.; Dinh, E.; Nguyen, T.V.; Ejtemaei, M.; Osborne, D. Advanced solid-liquid separation for dewatering fine coal tailings by combining chemical reagents and solid bowl centrifugation. Sep. Purif. Technol. 2021, 259, 118172. [Google Scholar] [CrossRef]
- Eskibalci, M.F.; Ozkan, M.F. Comparison of conventional coagulation and electrocoagulation methods for dewatering of coal preparation plant. Miner. Eng. 2018, 122, 106–112. [Google Scholar] [CrossRef]
- Zhang, W.; Dong, Z.; Han, X. Experimental study on flocculation and settlement of slime water in Guantun Coal Mine Preparation Plant. Coal Process. Compr. Util. 2023, 02, 51–56. [Google Scholar] [CrossRef]
- Han, Y.H. Quantum Chemistry Study on the Surface Properties and Dispersion Mechanism of Kaolinite and Montmorillonite. Ph.D. Thesis, China University of Mining and Technology (Beijing), Beijing, China, 2017. [Google Scholar]
- Zhang, M.Q.; Liu, J.T.; Liu, H.H.; Wang, Y.T. Effects of water hardness on the dispersion of fine coal and montmorillonite. J. China Univ. Min. Technol. 2009, 38, 114–118. [Google Scholar]
- He, M.; Yang, X.; Sun, X. Study on Clay Mineral Characteristics of Soft Rock in Chinese Coal Mines; China Coal Industry Publishing House: Beijing, China, 2006. [Google Scholar]
- Chen, Q.; Li, P.; Wei, X.; Chen, C.; Dang, W.; Nie, H.; Zhang, J. Mineralogy and geochemistry of shale from Shanxi Formation, Southern North China Basin: Implication for organic matter accumulation. Unconv. Resour. 2025, 6, 100151. [Google Scholar] [CrossRef]
- Zhang, Z.J.; Liu, J.T. Settling Characteristics Analysis of Coal Slime Water Based on Original Hardness. J. China Coal Soc. 2014, 39, 757–763. [Google Scholar] [CrossRef]
- Tombácz, E.; Szekeres, M. Surface charge heterogeneity of kaolinite in aqueous suspension in comparison with montmorillonite. Appl. Clay Sci. 2006, 34, 105–124. [Google Scholar] [CrossRef]
- Chang, J.; Shao, H.; Liu, B.; Manica, R.; Li, Z.; Liu, Q.; Xu, Z. Control of nanostructures through pH-dependent self-assembly of nanoplatelets. J. Colloid Interface Sci. 2021, 582, 439–445. [Google Scholar] [CrossRef]
- Fan, Y.P.; Dong, X.S.; Li, H. Study on Sedimentation and Dehydration of Oxidized Coal with Hydrogen Peroxide. Coal Technol. 2015, 34, 308–310. [Google Scholar] [CrossRef]
- Li, G.C.; Yan, X.H.; Li, M.M. EDLVO Analysis of Flocculation and Sedimentation of Slime Water under PAC. J. Heilongjiang Univ. Sci. Technol. 2020, 30, 45–49. [Google Scholar]
- Li, Y.; Chen, C.; Xia, W.; Sun, Y.; Qiu, H. Flocculation of sodium alginate combined with AlCl3/PAC on fine calcite: An in-situ experimental and calculation study. Appl. Surf. Sci. 2025, 682, 161692. [Google Scholar] [CrossRef]
- Xu, S.; Shi, J.; Deng, J.; Sun, H.; Wu, J.; Ye, Z. Flocculation and dewatering of the Kaolin slurry treated by single- and dual-polymer flocculants. Chemosphere 2023, 328, 138445. [Google Scholar] [CrossRef]
- Mpofu, P.; Addai-Mensah, J.; Ralston, J. Influence of hydrolyzable metal ions on the interfacial chemistry, particle interactions, and dewatering behavior of kaolinite dispersions. J. Colloid Interface Sci. 2003, 261, 349–359. [Google Scholar] [CrossRef] [PubMed]
- Sun, W.; Long, J.; Xu, Z.; Masliyah, J.H. Study of Al(OH)3−Polyacrylamide-Induced Pelleting Flocculation by Single Molecule Force Spectroscopy. Langmuir 2008, 24, 14015–14021. [Google Scholar] [CrossRef]
- Alagha, L.; Wang, S.; Yan, L.; Xu, Z.; Masliyah, J. Probing Adsorption of Polyacrylamide-Based Polymers on Anisotropic Basal Planes of Kaolinite Using Quartz Crystal Microbalance. Langmuir 2013, 29, 3989–3998. [Google Scholar] [CrossRef]
- Ng, J.; Osborn, I.; Harbottle, D.; Liu, Q.; Masliyah, J.H.; Xu, Z. Stimuli-Responsive Hybrid Polymer for Enhanced Solid–Liquid Separation of Industrial Effluents. Environ. Sci. Technol. 2019, 53, 6436–6443. [Google Scholar] [CrossRef]
- Dutta, S.; Chakraborty, S.; Sengupta, S.; Acharya, S.; Basu, D.; Bandyopadhyay, A. Genesis of an ecofriendly An + B3 hyperbranched polyester from Poly (ethylene glycol) and aconitic acid for application as flocculant. J. Polym. Res. 2024, 31, 39. [Google Scholar] [CrossRef]
- Zhang, Q.; Yin, Y.Q.; Xu, P.C. Experiment on properties of coal slime and its sedimentation under the action of coagulant. Coal Eng. 2022, 54, 181–186. [Google Scholar]
- Wang, J.B. Study on the settling performance and flocculent characteristics of coal slurry water under the action of different flocculants. Shanxi Chem. Ind. 2024, 44, 100–101+117. [Google Scholar] [CrossRef]
- Xu, D.F.; Xu, B.B.; Chang, J.J.; Zhen, J.Q.; Ma, X.M.; Zhang, C.; Wang, J. Research on Adaptive Flocculants for Flocculation and Sedimentation of Difficult-to-Settle Coal Slurry. Coal Prep. Technol. 2025, 53, 25–31. [Google Scholar] [CrossRef]
- Wang, J.B. Influence of flocculant type variation on settling and dewatering performance of coal slurry water. China Coal Vis. 2023, 1, 154–156. [Google Scholar]
- Lu, Y.J.; Ma, X.M.; Fan, Y.P.; Dong, X.S.; Hao, X.B.; Zhang, C. Synthesis of cationic flocculant CS-g-ADM and experimental study on coal slurry water sedimentation. Coal Sci. Technol. 2025, 53, 503–513. [Google Scholar]
- Yang, J.N.; Zhang, Z.J. Effect of a new flocculant TG on sedimentation performance of coal slime water. Clean Coal Technol. 2024, 30, 615–621. [Google Scholar] [CrossRef]
- Zhang, L.F.; Pang, Z.R. Application research of new coal slurry water purifying agent BK819A in a coal preparation plant in Inner Mongolia. Coal Process. Compr. Util. 2023, 11, 16–19. [Google Scholar] [CrossRef]
- Zhang, P.P.; Zhu, X.B.; Li, W.; Fang, C.J. Study on synthesis and properties of polysilicate aluminum ferric sulfate flocculant. Clean. World 2023, 39, 63–67+71. [Google Scholar]
- Liu, C.; Ding, X.G. Study of the coal slime water sedimentation effect obtained with the use of compound agent. Coal Prep. Technol. 2021, 5, 50–53. [Google Scholar] [CrossRef]
- Chang, J.; Xue, J.; Liang, S.; Zhao, W.; Li, Z. Flocculation Performance and Interfacial Adsorption Mechanism of Aluminum Hydroxide–Polyacrylamide in Coal Slime Water Treatment. Polymers 2026, 18, 458. [Google Scholar] [CrossRef] [PubMed]
- Zou, W.J. Selective Flocculation-Flotation Separation of Coking Coal’s Middlings. Ph.D. Thesis, China University of Mining and Technology, Xuzhou, China, 2014. [Google Scholar]
- Yan, L. Study of Anisotropic Surface Property of Phyllosilicates by Atomic Force Microscopy. Ph.D. Thesis, University of Alberta, Edmonton, AB, Canada, 2013. [Google Scholar]
- Richter, R.P.; Brisson, A. QCM-D on Mica for Parallel QCM-DAFM Studies. Langmuir 2004, 20, 4609–4613. [Google Scholar] [CrossRef]
- Ma, X.M. Study on Adsorption Behavior of Solid-Liquid Separation Chemicals at Coal Tailings Interface. Ph.D. Thesis, Taiyuan University of Technology, Taiyuan, China, 2019. [Google Scholar]
- Li, X.; Wang, Z.; Zhang, Y.; Zhang, W.; Zhang, H.; Liu, P.; Lei, T. Hydrogen-rich gas formation from catalytic pyrolysis of biomass tar by aluminum dross coupled HZSM-5 co-loaded Ni-Fe bimetallic catalysts: Influence of co-carrier characteristics. J. Environ. Manag. 2025, 389, 126016. [Google Scholar] [CrossRef]
- Munk, P.; Aminabhavi, T.M.; Williams, P.; Hoffman, D.E.; Chmelir, M. Some Solution Properties of Polyacrylamide. Macromolecules 1980, 13, 871–876. [Google Scholar] [CrossRef]
- Voinova, M.V.; Rodahl, M.; Jonson, M.; Kasemo, B. Viscoelastic Acoustic Response of Layered Polymer Films at Fluid-Solid Interfaces: Continuum Mechanics Approach. Phys. Scr. 1999, 59, 391. [Google Scholar] [CrossRef]
- Palmqvist, L.; Holmberg, K. Dispersant Adsorption and Viscoelasticity of Alumina Suspensions Measured by Quartz Crystal Microbalance with Dissipation Monitoring and in Situ Dynamic Rheology. Langmuir 2008, 24, 9989–9996. [Google Scholar] [CrossRef]
- Dunér, G.; Thormann, E.; Dėdinaitė, A. Quartz Crystal Microbalance with Dissipation (QCM-D) studies of the viscoelastic response from a continuously growing grafted polyelectrolyte layer. J. Colloid Interface Sci. 2013, 408, 229–234. [Google Scholar] [CrossRef] [PubMed]
- Yang, W.Y.; Qian, J.W.; Shen, Z.Q. A novel flocculant of Al(OH)3–polyacrylamide ionic hybrid. J. Colloid Interface Sci. 2004, 273, 400–405. [Google Scholar] [CrossRef] [PubMed]
- Yang, W.Y. Preparation, Dilute Solution Behavior and Flocculation Performance of Aluminum Hydroxide-Polyacrylamide Hybrid Materials. Ph.D. Thesis, Zhejiang University, Hangzhou, China, 2005. [Google Scholar]
- Xue, J. Study on Coagulation Performance and Adsorption Mechanism of Novel Efficient Coagulant Al-PAM in Coal Slime Water. Master’s Thesis, Xi’an University of Science and Technology, Xi’an, China, 2025. [Google Scholar]
- Bartosiewicz, H.; Curcio, P. Coal Handling and Preparation Plant (CHPP) Corrosion Control and Management. Available online: https://acarp.com.au/abstracts.aspx?repId=C12055 (accessed on 16 January 2026).
- Litvinenko, V. XVIII International Coal Preparation Congress; Springer: Berlin/Heidelberg, Germany, 2016. [Google Scholar]
- Yao, C.; Song, Q.; Meng, L.; Zhong, H.; Cao, W.; Li, H.; Sun, C.Q.; Pang, S.; Zhang, L. Interfacial polarization and lattice hydrogenation enable accelerated aluminum combustion with hydrogen-rich fluoropolymers. Chem. Eng. J. 2025, 521, 167068. [Google Scholar] [CrossRef]







| Flocculant Type | Flocculant Name | Flocculant Dosage | Coal Slime Water Properties/ Concentration (C *) | Settling Velocity (cm∙min−1) | Sediment Properties | Supernatant Clarity | Ref. * | |
|---|---|---|---|---|---|---|---|---|
| TR * | Inorganic Coagulant | Polyaluminum Chloride (PAC) | 15 kg∙t−1 | C = 40 g∙L−1 | Approx. 6 | Not specified | Low (Superior to PAS and PFS) | [30] |
| Anionic Organic Flocculant | Anionic Polyacrylamide (APAM, MW 6 million) | 100 g∙t−1 | D * < 0.074 mm, C = 20 g∙L−1 | 79 | Large floc particle size; Fractal dimension 1.635 | Transmittance 78% | [31] | |
| Cationic Organic Flocculant | Cationic Polyacrylamide (CPAM) | 50 g∙t−1 | d50 = 26.61 μm, Ash 49.58%, contains kaolinite, quartz, calcite | 72.08 | Sediment height 5.1 cm | Transmittance 89.1% | [32] | |
| Cationic Organic Flocculant | Cationic Polyacrylamide (CPAM, MW 6 million) | 250 g∙t−1 | d < 0.074 mm, C = 20 g∙L−1 | 18.38 | Small floc particle size; Fractal dimension 1.602 | Transmittance 56% | [33] | |
| Non-ionic Organic Flocculant | Non-ionic Polyacrylamide (NPAM) | 90 g∙t−1 | d50 = 26.61 μm, Ash 49.58%, contains kaolinite, quartz, calcite | Low | Sediment height 6.5 cm | Transmittance 68% | [32] | |
| Non-ionic Organic Flocculant | Non-ionic Polyacrylamide (NPAM, MW 6 million) | 250 g∙t−1 | d < 0.074 mm, C = 20 g∙L−1 | Low | Medium floc particle size; Fractal dimension 1.618 | Transmittance 67% | [33] | |
| Organic/Inorganic Composite | Polyaluminum Chloride (PAC) + Cationic Polyacrylamide (CPAM) | PAC: 50 g∙t−1 CPAM: 50 g∙t−1 | d50 = 26.61 μm | 75.38 | Sediment height 4.5 cm | Transmittance 96.2% | [32] | |
| NF * | Cationic Organic Flocculant | Chitosan-acrylamide-dimethyldiallylammonium chloride graft copolymer (CS-g-ADM) | 6 mg∙L−1 | C = 20 g∙L−1, pH 8.74 | 63 | Sediment height 2.2 cm; Filter cake moisture 21.95% | Transmittance 93% | [34] |
| Cationic Organic Flocculant | P(DMDAAC-AM) (PDA) copolymer | Optimal dosage ≤ 200 g∙t−1 | Fine coal slime water | Highly improved | Filter cake moisture increased; Larger flocs, higher fractal dimension (1.86) | Specific values not provided | [9] | |
| Novel Flocculant (TG) alone | Novel Flocculant TG | 70 g∙t−1 | C = 40 g∙L−1, 74.88% d < 0.045 mm | 33.33 | Sediment height 2.9 cm | 111 NTU | [35] | |
| Novel Flocculant (TG) + CPAM | Novel Flocculant TG + Cationic Polyacrylamide (CPAM) | TG:60 g∙t−1 CPAM:20 g∙t−1 | C = 40 g∙L−1, 74.88% d < 0.045 mm | 50 | Sediment height 2.3 cm | 39 NTU | [35] | |
| Organic/ Inorganic Composite | Novel Purifying Agent (BK819A, containing N-(2-aminopropyl)acrylamide fragment) | 5 mg∙L−1 | C = 20 g∙L−1 | 16.8 mL∙min−1 | - | 91 NTU | [36] | |
| Inorganic Composite Flocculant | Poly-silicic aluminum ferric sulfate (PSAFS) | 0.2 g∙L−1 (relative to slime water) | Initial turbidity 133.8 NTU | Large flocs, easy to settle | Not specified | Turbidity removal rate > 95% | [37] | |
| Composite Agent | Sodium polyacrylate-polyacrylamide copolymer (flocculant) + Diallyl dimethyl ammonium chloride (coagulant) | Optimal ratio 5:1 | C = 21.7 g∙L−1 | Highly improved | Not specified | Circulating water C = 0.5 g∙L−1 | [38] | |
| Organic-inorganic Composite Flocculant | Aluminum Hydroxide–Polyacrylamide (Al-PAM) | 6 mg∙L−1 | 62.49% d < 0.045 mm, C = 30 g∙L−1 | 84 | 2.3 cm | 45.77 NTU | [39] | |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Chang, J.; Zhao, H.; Liang, S.; Feng, X.; Xue, J.; Zhao, W. Inorganic–Organic Hybrid Polymer for Fine-Rich Coal Slime Water Treatment: Performance and Interfacial Adsorption Mechanism on Kaolinite Aluminol Surface. Separations 2026, 13, 99. https://doi.org/10.3390/separations13030099
Chang J, Zhao H, Liang S, Feng X, Xue J, Zhao W. Inorganic–Organic Hybrid Polymer for Fine-Rich Coal Slime Water Treatment: Performance and Interfacial Adsorption Mechanism on Kaolinite Aluminol Surface. Separations. 2026; 13(3):99. https://doi.org/10.3390/separations13030099
Chicago/Turabian StyleChang, Jing, Hang Zhao, Shizhen Liang, Xihao Feng, Jia Xue, and Wei Zhao. 2026. "Inorganic–Organic Hybrid Polymer for Fine-Rich Coal Slime Water Treatment: Performance and Interfacial Adsorption Mechanism on Kaolinite Aluminol Surface" Separations 13, no. 3: 99. https://doi.org/10.3390/separations13030099
APA StyleChang, J., Zhao, H., Liang, S., Feng, X., Xue, J., & Zhao, W. (2026). Inorganic–Organic Hybrid Polymer for Fine-Rich Coal Slime Water Treatment: Performance and Interfacial Adsorption Mechanism on Kaolinite Aluminol Surface. Separations, 13(3), 99. https://doi.org/10.3390/separations13030099





