Isolation, Structural Characterization, and In Vitro Antioxidant Activity of Polysaccharides from Cynanchum auriculatum Royle ex Wight
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials and Chemicals
2.2. Preparation of CAP
2.3. Homogeneity and Molecular Weight Analysis
2.4. FT-IR Spectroscopy
2.5. Monosaccharide Composition Analysis
2.6. Methylation Analysis
2.7. NMR Spectroscopy
2.8. Assay of Antioxidant Activity In Vitro
2.8.1. DPPH Radical Scavenging Activity
2.8.2. ABTS Radical Scavenging Activity
2.8.3. Hydroxyl Radical Scavenging Activity
2.9. Statistical Analysis
3. Results and Discussion
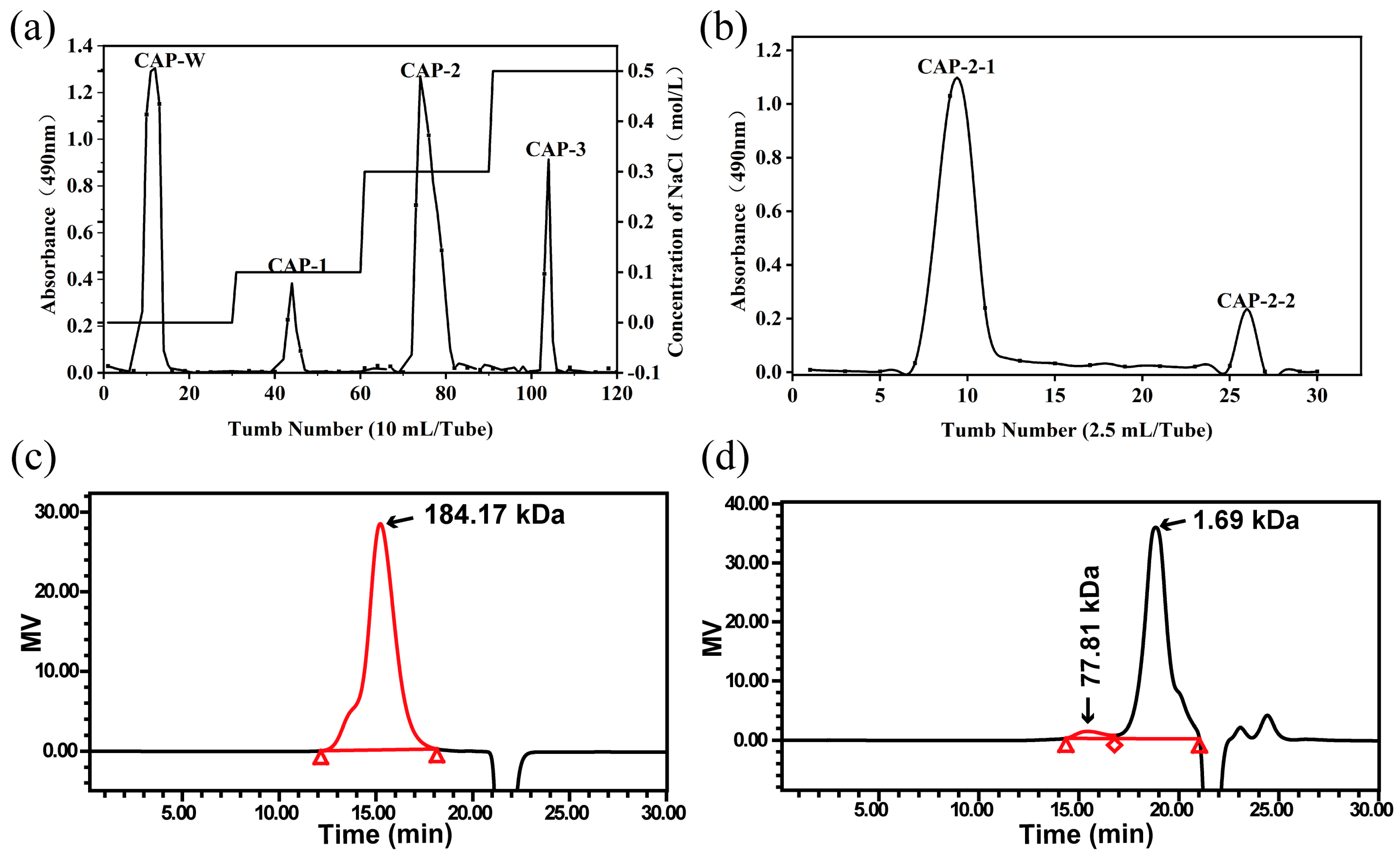
3.1. Purification of CAPs
3.2. The Mw of CAP2-1 and UCAP2-1
3.3. FT-IR Analysis of CAP2-1
3.4. Monosaccharide Composition of CAP2-1
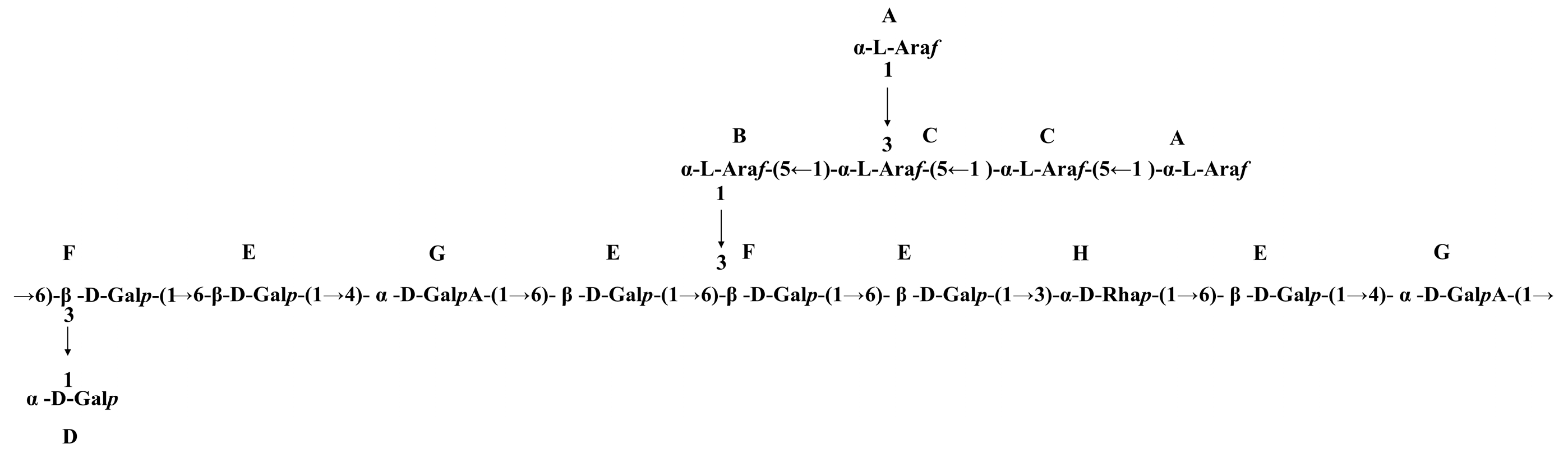
3.5. Linkage Feature of CAP2-1
3.6. NMR Spectral Analysis of CAP2-1
3.7. Proposed Structure of CAP2-1
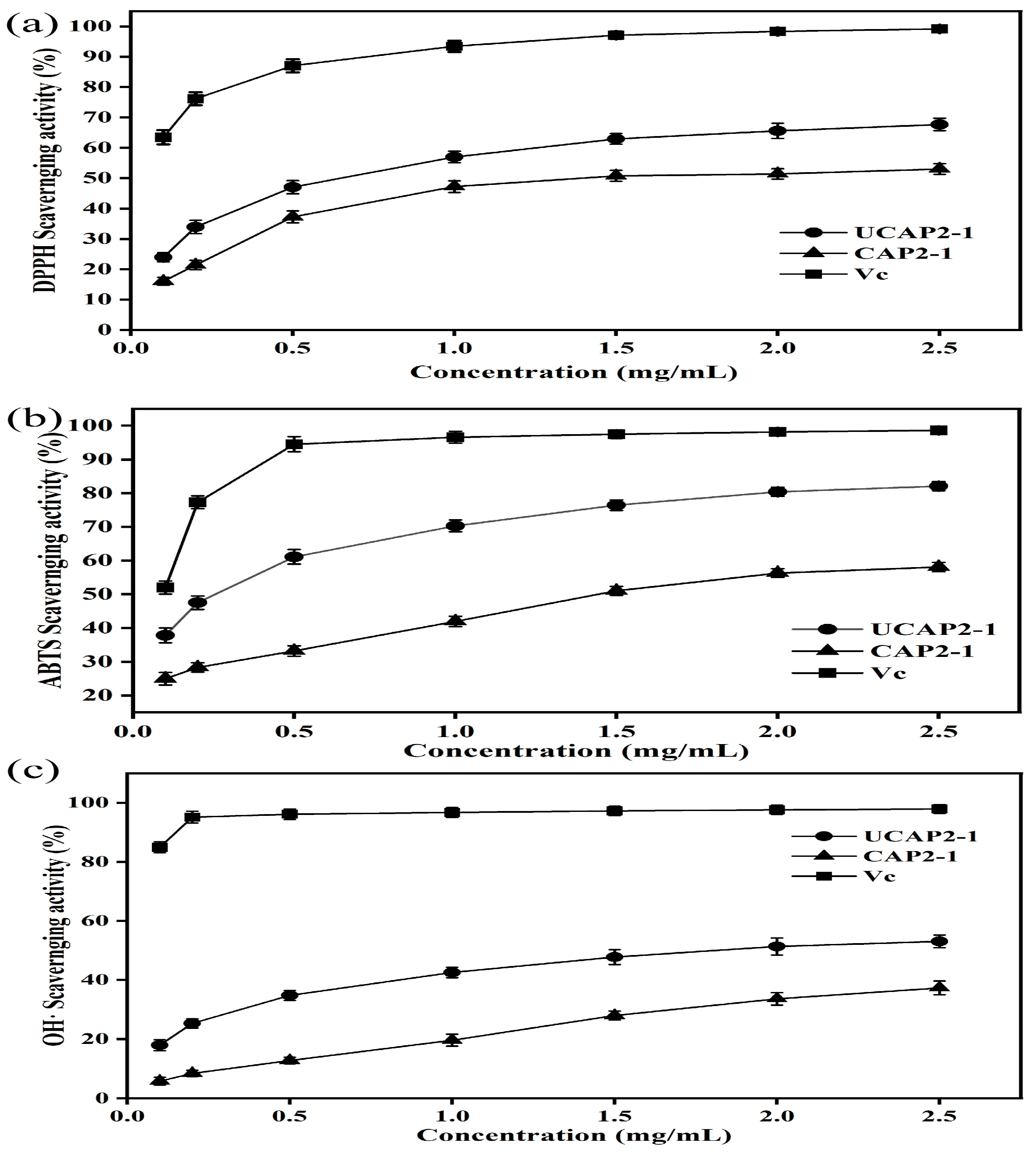
3.8. Antioxidant Activities of CAP2-1 and UCAP2-1
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Van’t Erve, T.J. Strategies to decrease oxidative stress biomarker levels in human medical conditions: A meta-analysis on 8-iso-prostaglandin F(2α). Redox Biol. 2018, 17, 284–296. [Google Scholar] [CrossRef]
- Meng, Q.; Li, J.; Wang, C.; Shan, A. Biological function of resveratrol and its application in animal production: A review. J. Anim. Sci. Biotechnol. 2023, 14, 25. [Google Scholar] [CrossRef]
- Clemente-Suárez, V.J.; Bustamante-Sanchez, A.; Rubio-Zarapuz, A.; Martín-Rodríguez, A.; Tornero-Aguilera, J.F.; Beltrán-Velasco, A.I. Biomimetic Strategies for Nutraceutical Delivery: Advances in Bionanomedicine for Enhanced Nutritional Health. Biomimetics 2025, 10, 426. [Google Scholar] [CrossRef]
- Lee, W.E.; Genetzakis, E.; Figtree, G.A. Novel Strategies in the Early Detection and Treatment of Endothelial Cell-Specific Mitochondrial Dysfunction in Coronary Artery Disease. Antioxidants 2023, 12, 1359. [Google Scholar] [CrossRef]
- Frandsen, J.R.; Narayanasamy, P. Neuroprotection through flavonoid: Enhancement of the glyoxalase pathway. Redox Biol. 2018, 14, 465–473. [Google Scholar] [CrossRef] [PubMed]
- Bin Heyat, M.B.; Akhtar, F.; Abbas, S.J.; Al-Sarem, M.; Alqarafi, A.; Stalin, A.; Abbasi, R.; Muaad, A.Y.; Lai, D.; Wu, K. Wearable Flexible Electronics Based Cardiac Electrode for Researcher Mental Stress Detection System Using Machine Learning Models on Single Lead Electrocardiogram Signal. Biosensors 2022, 12, 427. [Google Scholar] [CrossRef]
- Pathak, C.; Jaiswal, Y.K.; Vinayak, M. Queuine promotes antioxidant defence system by activating cellular antioxidant enzyme activities in cancer. Biosci. Rep. 2008, 28, 73–81. [Google Scholar] [CrossRef] [PubMed]
- Yusri, N.M.; Chan, K.W.; Iqbal, S.; Ismail, M. Phenolic Content and Antioxidant Activity of Hibiscus cannabinus L. Seed Extracts after Sequential Solvent Extraction. Molecules 2012, 17, 12612–12621. [Google Scholar] [CrossRef]
- Fernandes, P.A.R.; Coimbra, M.A. The antioxidant activity of polysaccharides: A structure-function relationship overview. Carbohyd. Polym. 2023, 314, 120965. [Google Scholar] [CrossRef] [PubMed]
- Fu, Y.-P.; Li, C.-Y.; Peng, X.; Zou, Y.-F.; Rise, F.; Paulsen, B.S.; Wangensteen, H.; Inngjerdingen, K.T. Polysaccharides from Aconitum carmichaelii leaves: Structure, immunomodulatory and anti-inflammatory activities. Carbohyd. Polym. 2022, 291, 119655. [Google Scholar] [CrossRef]
- Wang, N.; Zhang, X.; Wang, S.; Guo, Q.; Li, Z.; Liu, H.; Wang, C. Structural characterisation and immunomodulatory activity of polysaccharides from white asparagus skin. Carbohyd. Polym. 2020, 227, 115314. [Google Scholar] [CrossRef] [PubMed]
- Zong, A.; Cao, H.; Wang, F. Anticancer polysaccharides from natural resources: A review of recent research. Carbohyd. Polym. 2012, 90, 1395–1410. [Google Scholar] [CrossRef]
- Chen, F.; Huang, G. Antioxidant activity of polysaccharides from different sources of ginseng. Int. J. Biol. Macromol. 2019, 125, 906–908. [Google Scholar] [CrossRef]
- Hu, Y.; Zhou, L.; Yang, J.; Bai, R.; Marchioni, E.; Zhao, M.; Zhou, L. Anti-inflammatory mechanism of Houttuynia cordata polysaccharides against ulcerative colitis based on multi-omics conjoint analysis. Int. J. Biol. Macromol. 2024, 283, 137311. [Google Scholar] [CrossRef]
- Nie, C.; Zhu, P.; Ma, S.; Wang, M.; Hu, Y. Purification, characterization and immunomodulatory activity of polysaccharides from stem lettuce. Carbohyd. Polym. 2018, 188, 236–242. [Google Scholar] [CrossRef]
- Wang, Z.; Zhou, X.; Shu, Z.; Zheng, Y.; Hu, X.; Zhang, P.; Huang, H.; Sheng, L.; Zhang, P.; Wang, Q.; et al. Regulation strategy, bioactivity, and physical property of plant and microbial polysaccharides based on molecular weight. Int. J. Biol. Macromol. 2023, 244, 125360. [Google Scholar] [CrossRef]
- Zhou, H.; Fan, Z.; Li, Y.; Liu, X.; Wang, B.; Xing, J.; He, J.; Zheng, R.; Li, J. Structure–Antioxidant Activity Relationship of Polysaccharides Isolated by Microwave/Ultrasonic-Assisted Extraction from Pleurotus ferulae. Antioxidants 2025, 14, 91. [Google Scholar] [CrossRef] [PubMed]
- Lo, T.C.-T.; Chang, C.A.; Chiu, K.-H.; Tsay, P.-K.; Jen, J.-F. Correlation evaluation of antioxidant properties on the monosaccharide components and glycosyl linkages of polysaccharide with different measuring methods. Carbohyd. Polym. 2011, 86, 320–327. [Google Scholar] [CrossRef]
- Tang, Y.; He, X.; Liu, G.; Wei, Z.; Sheng, J.; Sun, J.; Li, C.; Xin, M.; Li, L.; Yi, P. Effects of different extraction methods on the structural, antioxidant and hypoglycemic properties of red pitaya stem polysaccharide. Food Chem. 2023, 405, 134804. [Google Scholar] [CrossRef]
- Jing, Y.; Yan, M.; Zhang, H.; Liu, D.; Qiu, X.; Hu, B.; Zhang, D.; Zheng, Y.; Wu, L. Effects of Extraction Methods on the Physicochemical Properties and Biological Activities of Polysaccharides from Polygonatum sibiricum. Foods 2023, 12, 2088. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Y.; Feng, X.; Guo, J.; Wang, L.; Guo, X.; Zhu, X. A review of extraction, purification, structural properties and biological activities of legumes polysaccharides. Front. Nutr. 2022, 9, 1021448. [Google Scholar] [CrossRef]
- Yuan, X.-H.; Tu, T.-T.; Mao, Y.-H.; Wang, Y.-C.; Huang, M.-Q.; Wang, L.; Luo, Y.; Wang, C.-X.; Qiu, S.-Y.; Deng, B.; et al. Characterization and bifidogenic effects of a low-molecular weight polysaccharide isolated from a Chinese herb, Polygonatum kingianum Coll. et Hemsl (Huangjing) rhizome. Carbohyd. Polym. 2026, 373, 124678. [Google Scholar] [CrossRef]
- Guo, Q.; Huang, X.; Kang, J.; Ding, H.; Liu, Y.; Wang, N.; Cui, S.W. Immunomodulatory and antivirus activities of bioactive polysaccharides and structure-function relationship. Bioact. Carbohydr. Diet. Fibre 2022, 27, 100301. [Google Scholar] [CrossRef]
- Chai, Z.; Huang, W.; Zhao, X.; Wu, H.; Zeng, X.; Li, C. Preparation, characterization, antioxidant activity and protective effect against cellular oxidative stress of polysaccharide from Cynanchum auriculatum Royle ex Wight. Int. J. Biol. Macromol. 2018, 119, 1068–1076. [Google Scholar] [CrossRef]
- Wang, X.-J.; Li, Z.-L.; Lv, X.-H.; Zuo, Q.-Y.; Zhao, Y.-M.; Ding, Y.-F.; Pu, S.-B.; Qian, S.-H.; Peng, Y.-R. Anti-tumor evaluation and multiple analysis on different extracted fractions of the root of Cynanchum auriculatum Royle ex Wight. J. Sep. Sci. 2017, 40, 3054–3063. [Google Scholar] [CrossRef] [PubMed]
- Nasiru, M.M.; Sun, Y.-E.; Zhao, L.; Bunhok, T.; Roth, C.M.; Sovath, S.; Pharith, H.; Wang, W.; Li, C. Isolation, Purification, and Antioxidant Activity of Polyphenols from Cynanchum auriculatum Royle ex Wight. Separations 2024, 11, 316. [Google Scholar] [CrossRef]
- Wang, Y.; Han, J.; Yue, Y.; Wu, Y.; Zhang, W.; Xia, W.; Wu, M. Purification, structure identification and immune activity of a neutral polysaccharide from Cynanchum auriculatum. Int. J. Biol. Macromol. 2023, 237, 124142. [Google Scholar] [CrossRef] [PubMed]
- Xu, Y.; Wu, C.; Wang, L.; Wu, S.; Chen, Y.; Ding, X.; Wang, L.; Yu, Y.; Du, W.; Zhang, Y.; et al. Phytochemical and chemotaxonomic investigations on the aerial parts of Cynanchum auriculatum Royle ex Wight. Biochem. Syst. Ecol. 2023, 107, 104609. [Google Scholar] [CrossRef]
- Ding, X.; Wang, L.; Xu, Y.; Zheng, S.; Wang, S.; Wang, L.; Qin, M.; Wu, S.; Yu, Y.; Hong, J.; et al. Chemical constituents from the flowers of Cynanchum auriculatum Royle ex Wight. Biochem. Syst. Ecol. 2023, 106, 104562. [Google Scholar] [CrossRef]
- Wu, Z.; Wang, Y.; Meng, X.; Wang, X.; Li, Z.; Qian, S.; Wei, Y.; Shu, L.; Ding, Y.; Wang, P.; et al. Total C-21 steroidal glycosides, isolated from the root tuber of Cynanchum auriculatum Royle ex Wight, attenuate hydrogen peroxide-induced oxidative injury and inflammation in L02 cells. Int. J. Mol. Med. 2018, 42, 3157–3170. [Google Scholar] [CrossRef]
- Peng, Y.R.; Li, Y.B.; Liu, X.D.; Zhang, J.F.; Duan, J.A. Anti-tumor activity of C-21 steroidal glycosides from Cynanchum auriculatum Royle ex Wight. Phytomedicine 2008, 15, 1016–1020. [Google Scholar] [CrossRef]
- Liu, Z.; Liu, Z.; Li, L.; Zhang, J.; Zhao, Q.; Lin, N.; Zhong, W.; Jiang, M. Immunomodulatory effects of the polysaccharide from Sinonovacula constricta on RAW264.7 macrophage cells. Food Sci. Nutr. 2022, 10, 1093–1102. [Google Scholar] [CrossRef]
- DuBois, M.; Gilles, K.A.; Hamilton, J.K.; Rebers, P.A.; Smith, F. Colorimetric Method for Determination of Sugars and Related Substances. Anal. Chem. 1956, 28, 350–356. [Google Scholar] [CrossRef]
- Li, Q.; Li, Y.; Niu, H.; Wang, E.; Jiao, L.; Li, H.; Wu, W. Structure Elucidation and Immunoactivity Study of Armillaria mellea Fruiting Body Polysaccharides. Separations 2024, 11, 3. [Google Scholar] [CrossRef]
- Yang, L.; Zhang, H.; Zhao, Y.; Huang, J.; Zhu, D.; Wang, S.; Zhu, L.; Chen, L.; Xu, X.; Liu, H. Chemical structure, chain conformation and rheological properties of pectic polysaccharides from soy hulls. Int. J. Biol. Macromol. 2020, 148, 41–48. [Google Scholar] [CrossRef] [PubMed]
- Li, B.; Dobruchowska, J.M.; Hoogenkamp, M.A.; Gerwig, G.J. Structural investigation of an extracellular polysaccharide produced by the cariogenic bacterium Streptococcus mutans strain UA159. Carbohyd. Polym. 2012, 90, 675–682. [Google Scholar] [CrossRef]
- Tang, W.; Dong, M.; Wang, W.; Han, S.; Rui, X.; Chen, X.; Jiang, M.; Zhang, Q.; Wu, J.; Li, W. Structural characterization and antioxidant property of released exopolysaccharides from Lactobacillus delbrueckii ssp. bulgaricus SRFM-1. Carbohyd. Polym. 2017, 173, 654–664. [Google Scholar] [CrossRef]
- Seedevi, P.; Moovendhan, M.; Sudharsan, S.; Sivasankar, P.; Sivakumar, L.; Vairamani, S.; Shanmugam, A. Isolation and chemical characteristics of rhamnose enriched polysaccharide from Grateloupia lithophila. Carbohyd. Polym. 2018, 195, 486–494. [Google Scholar] [CrossRef]
- Zhu, Y.-M.; Pan, L.-C.; Zhang, L.-J.; Yin, Y.; Zhu, Z.-Y.; Sun, H.-Q.; Liu, C.-Y. Chemical structure and antioxidant activity of a polysaccharide from Siraitia grosvenorii. Int. J. Biol. Macromol. 2020, 165, 1900–1910. [Google Scholar] [CrossRef]
- Xie, M.; Hu, B.; Wang, Y.; Zeng, X. Grafting of Gallic Acid onto Chitosan Enhances Antioxidant Activities and Alters Rheological Properties of the Copolymer. J. Agr. Food Chem. 2014, 62, 9128–9136. [Google Scholar] [CrossRef]
- Li, C.; Huang, Q.; Fu, X.; Yue, X.-J.; Liu, R.H.; You, L.-J. Characterization, antioxidant and immunomodulatory activities of polysaccharides from Prunella vulgaris Linn. Int. J. Biol. Macromol. 2015, 75, 298–305. [Google Scholar] [CrossRef] [PubMed]
- Jiang, J.; Kong, F.; Li, N.; Zhang, D.; Yan, C.; Lv, H. Purification, structural characterization and in vitro antioxidant activity of a novel polysaccharide from Boshuzhi. Carbohyd. Polym. 2016, 147, 365–371. [Google Scholar] [CrossRef]
- Mkadmini Hammi, K.; Hammami, M.; Rihouey, C.; Le Cerf, D.; Ksouri, R.; Majdoub, H. Optimization extraction of polysaccharide from Tunisian Zizyphus lotus fruit by response surface methodology: Composition and antioxidant activity. Food Chem. 2016, 212, 476–484. [Google Scholar] [CrossRef]
- Luo, Q.-L.; Tang, Z.-H.; Zhang, X.-F.; Zhong, Y.-H.; Yao, S.-Z.; Wang, L.-S.; Lin, C.-W.; Luo, X. Chemical properties and antioxidant activity of a water-soluble polysaccharide from Dendrobium officinale. Int. J. Biol. Macromol. 2016, 89, 219–227. [Google Scholar] [CrossRef]
- Xu, Y.; Cai, F.; Yu, Z.; Zhang, L.; Li, X.; Yang, Y.; Liu, G. Optimisation of pressurised water extraction of polysaccharides from blackcurrant and its antioxidant activity. Food Chem. 2016, 194, 650–658. [Google Scholar] [CrossRef] [PubMed]
- Zha, S.; Zhao, Q.; Chen, J.; Wang, L.; Zhang, G.; Zhang, H.; Zhao, B. Extraction, purification and antioxidant activities of the polysaccharides from maca (Lepidium meyenii). Carbohyd. Polym. 2014, 111, 584–587. [Google Scholar] [CrossRef] [PubMed]
- Ye, Z.; Wang, W.; Yuan, Q.; Ye, H.; Sun, Y.; Zhang, H.; Zeng, X. Box–Behnken design for extraction optimization, characterization and in vitro antioxidant activity of Cicer arietinum L. hull polysaccharides. Carbohyd. Polym. 2016, 147, 354–364. [Google Scholar] [CrossRef]
- Chen, Q.; Xue, G.; Ni, Q.; Wang, Y.; Gao, Q.; Zhang, Y.; Xu, G. Physicochemical and rheological characterization of pectin-rich polysaccharides from Gardenia jasminoides J. Ellis flower. Food Sci. Nutr. 2020, 8, 3335–3345. [Google Scholar] [CrossRef]
- Wu, H.; Shu, L.; Liang, T.; Li, Y.; Liu, Y.; Zhong, X.; Xing, L.; Zeng, W.; Zhao, R.; Wang, X. Extraction optimization, physicochemical property, antioxidant activity, and α-glucosidase inhibitory effect of polysaccharides from lotus seedpods. J. Sci. Food Agric. 2022, 102, 4065–4078. [Google Scholar] [CrossRef]
- Wang, Y.; Xiong, X.; Huang, G. Ultrasound-assisted extraction and analysis of maidenhairtree polysaccharides. Ultrason. Sonochemistry 2023, 95, 106395. [Google Scholar] [CrossRef]
- Li, W.; Tang, W.; Ji, J.; Xia, X.; Rui, X.; Chen, X.; Jiang, M.; Zhou, J.; Dong, M. Characterization of a novel polysaccharide with anti-colon cancer activity from Lactobacillus helveticus MB2-1. Carbohydr. Res. 2015, 411, 6–14. [Google Scholar] [CrossRef]
- Zhang, Y.; Zhou, T.; Wang, H.; Cui, Z.; Cheng, F.; Wang, K.-P. Structural characterization and in vitro anti-tumor activity of an acidic polysaccharide from Angelica sinensis (Oliv.) Diels. Carbohyd. Polym. 2016, 147, 401–408. [Google Scholar] [CrossRef]
- Wang, X.; Zhang, H.; Wang, Z.; Bai, H. Optimization of ultrasonic-assisted alkaline extraction of polysaccharides from Phellodendron amurense Rupr. pollen using response surface methodology and its structure features. RSC Adv. 2015, 5, 106800–106808. [Google Scholar] [CrossRef]
- Zhang, Q.; Xu, Y.; Zou, S.; Zhang, X.; Cao, K.; Fan, Q. Novel functional polysaccharides from Radix Polygoni Multiflori water extracted residue: Preliminary characterization and immunomodulatory activity. Carbohyd. Polym. 2016, 137, 625–631. [Google Scholar] [CrossRef] [PubMed]
- Petersen, B.O.; Hindsgaul, O.; Paulsen, B.S.; Redondo, A.R.; Skovsted, I.C. Structural elucidation of the capsular polysaccharide from Streptococcus pneumoniae serotype 47A by NMR spectroscopy. Carbohydr. Res. 2014, 386, 62–67. [Google Scholar] [CrossRef] [PubMed]
- Wu, Y.-T.; Huo, Y.-F.; Xu, L.; Xu, Y.-Y.; Wang, X.-L.; Zhou, T. Purification, characterization and antioxidant activity of polysaccharides from Porphyra haitanensis. Int. J. Biol. Macromol. 2020, 165, 2116–2125. [Google Scholar] [CrossRef]


| Methylated Sugars | Linkages | Major Mass Fragments (m/z) | Molar Ratio |
|---|---|---|---|
| 2,3,5-Me3-Araf | α-Araf-(1→ | 43, 71, 87, 102, 118, 129, 161 | 2.13 |
| 2,3-Me2-Araf | →5)-α-Araf-(1→ | 43, 87, 102, 118, 129, 189 | 1.76 |
| 2-Me-Araf | →3,5)-α-Araf-(1→ | 43, 88, 101, 117, 130, 143, 190 | 0.86 |
| 2,3,4,6-Me4-Galp | α-Galp-(1→ | 43, 45, 71, 87, 102, 118, 129, 145, 161, 205 | 0.99 |
| 2,3,4-Me3-Galp | →6)-β-Galp-(1→ | 43, 45, 87, 99, 102, 113, 118, 129, 162, 173, 233 | 2.25 |
| 2,4-Me2-Galp | →3,6)-β-Galp-(1→ | 43, 87, 101, 118, 129, 160, 234 | 0.89 |
| 2,3,6-Me3-Galp | →4)-α-GalpA-(1→ | 43, 71, 87, 99, 102, 113, 118, 129, 131, 162, 173, 233 | 1.57 |
| 2,4-Me-Rhap | →3)-α-Rhap-(1→ | 43, 57, 71, 87, 99, 102, 118, 129, 162, 189, 233 | 0.53 |
| Residues | C-1/H-1 | C-2/H-2 | C-3/H-3 | C-4/H-4 | C-5/H-5ab | C-6/H-6ab | |
|---|---|---|---|---|---|---|---|
| A | α-L-Araf-(1→ | 107.60/5.11 | 81.09/4.09 | 76.70/3.88 | 81.45/3.77 | 61.26/3.54 or 3.50 | |
| B | →5)-α-L-Araf-(1→ | 109.32/5.18 | 82.46/4.18 | 77.80/3.98 | 81.50/4.08 | 68.58/3.37 or 3.83 | |
| C | →3,5)-α-L-Araf-1→ | 107.06/5.17 | 81.22/4.17 | 84.15/4.06 | 81.40/4.26 | 68.58/3.88 or 3.87 | |
| D | α-D-Galp-(1→ | 102.76/5.04 | 74.61/4.00 | 76.37/3.90 | 73.27/3.77 | 75.22/3.54 | 73.32/3.33 or 3.21 |
| E | →6)-β-D-Galp-(1→ | 104.50/4.56 | 74.61/3.63 | 73.60/3.74 | 70.88/3.67 | 70.19/3.88 | 69.04/3.57 or 3.72 |
| F | →3,6)-β-D-Galp-1→ | 103.28/4.53 | 75.22/3.32 | 81.50/3.52 | 74.47/3.23 | 70.42/3.49 | 69.45/3.64 or 3.68 |
| G | →4)-α-GalpA-(1→ | 106.55/5.01 | 68.16/3.52 | 68.76/3.65 | 77.82/4.02 | 74.61/4.25 | 175.23/- |
| H | →3)-α-D-Rhap-(1→ | 100.86/5.08 | 79.26/4.24 | 70.18/3.87 | 73.45/3.83 | 66.24/3.98 | 16.66/1.18 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Zhu, H.; Nasiru, M.M.; Jiang, S.; Sun, Y.; Liu, D.; Li, C. Isolation, Structural Characterization, and In Vitro Antioxidant Activity of Polysaccharides from Cynanchum auriculatum Royle ex Wight. Separations 2026, 13, 41. https://doi.org/10.3390/separations13020041
Zhu H, Nasiru MM, Jiang S, Sun Y, Liu D, Li C. Isolation, Structural Characterization, and In Vitro Antioxidant Activity of Polysaccharides from Cynanchum auriculatum Royle ex Wight. Separations. 2026; 13(2):41. https://doi.org/10.3390/separations13020041
Chicago/Turabian StyleZhu, Hequan, Mustapha Muhammad Nasiru, Sijia Jiang, Yuetao Sun, Dan Liu, and Chunyang Li. 2026. "Isolation, Structural Characterization, and In Vitro Antioxidant Activity of Polysaccharides from Cynanchum auriculatum Royle ex Wight" Separations 13, no. 2: 41. https://doi.org/10.3390/separations13020041
APA StyleZhu, H., Nasiru, M. M., Jiang, S., Sun, Y., Liu, D., & Li, C. (2026). Isolation, Structural Characterization, and In Vitro Antioxidant Activity of Polysaccharides from Cynanchum auriculatum Royle ex Wight. Separations, 13(2), 41. https://doi.org/10.3390/separations13020041








