Pilot Study and Preliminary Results of Biodegradable Intramedullary Nailing of Forearm Fractures in Children
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Design, Background and Rationale
2.2. Consent
2.3. Participants
2.4. Inclusion Criteria
- Diaphyseal forearm fractures (radius or ulna or both).
- Surgical stabilization is required.
- Time between injury and operative treatment not exceeding 14 days.
- Patients from 3 years to 13 years (chronological age).
- The patient and the patient’s legal guardian have signed the informed consent form and are willing to participate in all follow-up visits.
2.5. Exclusion Criteria
- Spiroid fractures, multifragmentary fractures.
- Metaphyseal and epiphyseal fractures.
- Fractures where internal fixation is otherwise contraindicated, e.g., active or potential infection, pathological fracture (malignancy) or when a patient’s cooperation cannot be guaranteed.
2.6. Participant Withdrawal Criteria
- at the request of the patient or his or her legal representative.
- on the part of the study by the investigator if it is considered that continued study participation is not in the best interest of the child.
2.7. Participant Timeline
2.8. Sample Size
2.9. Allocation, Randomization and Blinding
2.10. Intervention
2.11. Outcomes
2.11.1. Definition of Fracture Healing
2.11.2. Primary Outcome Measures
2.11.3. Secondary Outcome Measures
- At some of the study sites, MRI scans at one and two years after surgery will be performed to assess the amount of biodegradation and to detect soft-tissue reaction caused by the implant.
- Cost effectiveness of this treatment will be evaluated in a health technology assessment 1 year after surgery of the last patient included in the study.
- Evaluation of bony union depending on fracture type and immobilization time, return to daily activity, return to sport.
- Safety and performance of operative technique in Monteggia’s lesions.
2.12. Data Collection and Management
2.13. Monitoring
2.14. Harm’s Auditing
2.15. Auditing
2.16. Follow-Up Examination
2.17. Statistical Analysis
- Fracture type determined by X-ray.
- Age.
- Gender.
- BMI.
- Surgical technique.
- Immobilization time.
- Bony union formation.
2.18. Research Ethics Approval
2.19. Protocol Amendments
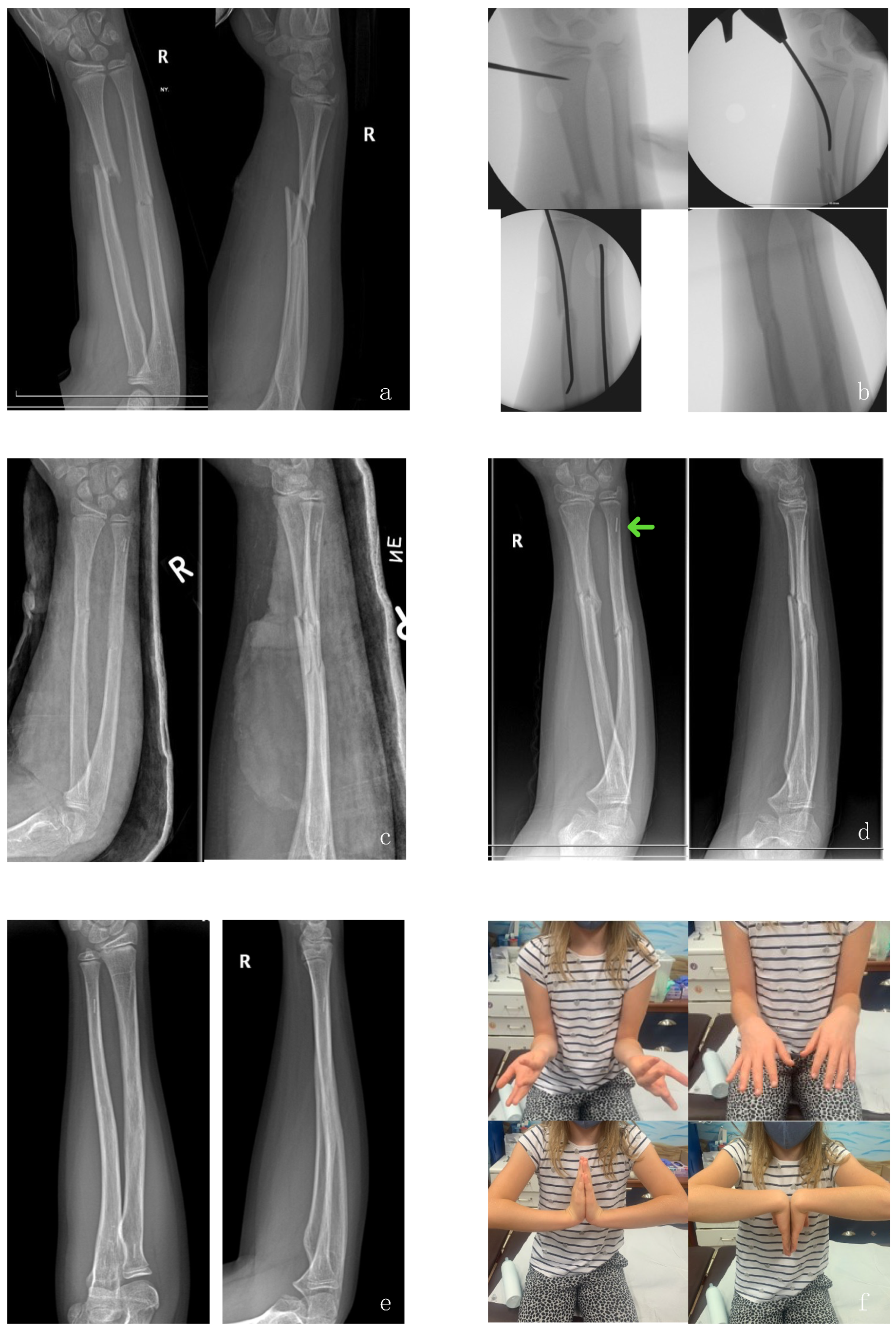
3. Operative Technique and Preliminary Results
3.1. Preliminary Results (Pilot Study)
3.1.1. Complications
3.1.2. Refracture
4. Discussion
4.1. Potential Impact and Significance of the Multicenter Trial
4.2. Strengths and Limitation of the Trial
4.3. Expectations
4.4. Trial Status
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Appendix A
| Study Site | Review Board/Ethics Committee | Approval Number | Date of Approval (mm-dd-yyyy) |
|---|---|---|---|
| Austria Baden/Mödling | Niederösterreichische Ethikkommission | GS4-EK-3/165-2020 | 11-09-2020 |
| Germany | Ethik-Kommission der Universität Lübeck | 21-258 | 08-05-2021 |
| Hungary Pecs | PTE-KK Regionalis és Intézményi Kutatás—Etikai Bizottság | 8737-PTE2021 | 23-04-2021 |
| Hungary Peterfy | Péterfy Kórház Rendelőintézet, Országos Traumatológiai Intézet, Intézményi Kutatásetikai Bizottság | 4/2020 | 11-05-2020 |
| Denmark | De videnskabsetiske komiteer | H-21011210 | 07-07-2021 |
| France | Comité de Protection des Personnes Est IV (CPP Est IV) | CNRIPH 21.02.01.44305 | 20-12-2021 |
References
- Raney, E.M.; Freccero, D.M.; Dolan, L.A.; Lighter, D.E.; Fillman, R.R.; Chambers, H.G. Evidence-Based Analysis of Removal of Orthopaedic Implants in the Pediatric Population. J. Pediatric Orthop. 2008, 28, 701–704. Available online: https://journals.lww.com/01241398-200810000-00001 (accessed on 16 May 2022). [CrossRef] [PubMed]
- Boulos, A.; DeFroda, S.F.; Kleiner, J.E.; Thomas, N.; Gil, J.A.; Cruz, A.I. Inpatient orthopaedic hardware removal in children: A cross-Sectional study. J. Clin. Orthop. Trauma 2017, 8, 270–275. [Google Scholar] [CrossRef] [PubMed]
- Busam, M.L.; Esther, R.J.; Obremskey, W.T. Hardware Removal: Indications and Expectations. J. Am. Acad. Orthop. Surg. 2006, 14, 113–120. Available online: http://journals.lww.com/00124635-200602000-00006 (accessed on 16 May 2022). [CrossRef] [PubMed] [Green Version]
- Kroeze, R.; Helder, M.; Govaert, L.; Smit, T. Biodegradable Polymers in Bone Tissue Engineering. Materials 2009, 2, 833–856. Available online: https://www.mdpi.com/1996-1944/2/3/833 (accessed on 16 May 2022). [CrossRef] [Green Version]
- Hope, P.; Williamson, D.; Coates, C.; Cole, W. Biodegradable pin fixation of elbow fractures in children. A randomised trial. J. Bone Jt. Surg. Br. Vol. 1991, 73, 965–968. Available online: http://online.boneandjoint.org.uk/doi/10.1302/0301-620X.73B6.1659570 (accessed on 16 May 2022). [CrossRef]
- Comino-Garayoa, R.; Cortés-Bretón Brinkmann, J.; Peláez, J.; López-Suárez, C.; Martínez-González, J.M.; Suárez, M.J. Allergies to Titanium Dental Implants: What Do We Really Know about Them? A Scoping Review. Biology 2020, 9, 404. Available online: https://www.mdpi.com/2079-7737/9/11/404 (accessed on 16 May 2022). [CrossRef]
- Lieber, J.; Dietzel, M.; Scherer, S.; Schäfer, J.; Kirschner, H.; Fuchs, J. Implant removal associated complications after ESIN osteosynthesis in pediatric fractures. Eur. J. Trauma Emerg. Surg. 2021, 1–8. [Google Scholar] [CrossRef]
- Gortzak, Y.; Mercado, E.; Atar, D.; Weisel, Y. Pediatric Olecranon Fractures. J. Pediatric Orthop. 2006, 26, 39–42. Available online: https://journals.lww.com/01241398-200601000-00008 (accessed on 16 May 2022). [CrossRef]
- Partio, E.K.; Merikanto, J.; Heikkilä, J.T.; Ylinen, P.; Mäkelä, E.A.; Vainio, J.; Törmälä, P.; Rokkanen, P. Totally absorbable screws in fixation of subtalar extra articular arthrodesis in children with spastic neuromuscular disease: Preliminary report of a randomized prospective study of fourteen arthrodeses fixed with absorbable or metallic screws. J. Pediatric Orthop. 1992, 12, 646–650. Available online: http://www.ncbi.nlm.nih.gov/pubmed/1517428 (accessed on 16 May 2022). [CrossRef]
- Sinikumpu, J.-J.; Keränen, J.; Haltia, A.-M.; Serlo, W.; Merikanto, J. A new mini-invasive technique in treating pediatric diaphyseal forearm fractures by bioabsorbable elastic stable intramedullary nailing: A preliminary technical report. Scand. J. Surg. 2013, 102, 258–264. Available online: http://journals.sagepub.com/doi/10.1177/1457496913490459 (accessed on 16 May 2022). [CrossRef] [Green Version]
- Korhonen, L.; Perhomaa, M.; Kyrö, A.; Pokka, T.; Serlo, W.; Merikanto, J.; Sinikumpu, J.-J. Intramedullary nailing of forearm shaft fractures by biodegradable compared with titanium nails: Results of a prospective randomized trial in children with at least two years of follow-up. Biomaterials 2018, 185, 383–392. Available online: https://linkinghub.elsevier.com/retrieve/pii/S0142961218306422 (accessed on 16 May 2022). [CrossRef] [PubMed]
- Makki, D.; Kheiran, A.; Gadiyar, R.; Ricketts, D. Refractures following removal of plates and elastic nails from paediatric forearms. J. Pediatric Orthop. B 2014, 23, 221–226. Available online: https://journals.lww.com/01202412-201405000-00003 (accessed on 16 May 2022). [CrossRef] [PubMed]
- Makki, D.; Matar, H.E.; Webb, M.; Wright, D.M.; James, L.A.; Ricketts, D.M. Elastic stable intramedullary nailing in paediatric forearm fractures: The rate of open reduction and complications. J. Pediatric Orthop. B 2017, 26, 412–416. Available online: https://journals.lww.com/01202412-201709000-00004 (accessed on 16 May 2022). [CrossRef] [PubMed]
- Martus, J.E.; Preston, R.K.; Schoenecker, J.G.; Lovejoy, S.A.; Green, N.E.; Mencio, G.A. Complications and Outcomes of Diaphyseal Forearm Fracture Intramedullary Nailing. J. Pediatric Orthop. 2013, 33, 598–607. Available online: https://journals.lww.com/01241398-201309000-00003 (accessed on 16 May 2022). [CrossRef] [PubMed]
- Kruppa, C.; Bunge, P.; Schildhauer, T.A.; Dudda, M. Low complication rate of elastic stable intramedullary nailing (ESIN) of pediatric forearm fractures. Medicine 2017, 96, e6669. Available online: https://journals.lww.com/00005792-201704210-00057 (accessed on 16 May 2022). [CrossRef]
- Dinçer, R.; Köse, A.; Topal, M.; Öztürk, İ.A.; Engin, M.Ç. Surgical treatment of pediatric forearm fractures with intramedullary nails: Is it a disadvantage to leave the tip exposed? J. Pediatric Orthop. B 2020, 29, 158–163. Available online: https://journals.lww.com/10.1097/BPB.0000000000000635 (accessed on 16 May 2022). [CrossRef]
- Fernandez, F.F.; Langendörfer, M.; Wirth, T.; Eberhardt, O. Failures and complications in intramedullary nailing of children’s forearm fractures. J. Child. Orthop. 2010, 4, 159–167. [Google Scholar] [CrossRef] [Green Version]
- Kim, B.S.; Lee, Y.S.; Park, S.Y.; Nho, J.H.; Lee, S.G.; Kim, Y.H. Flexible intramedullary nailing of forearm fractures at the distal metadiaphyseal junction in adolescents. CiOS Clin. Orthop. Surg. 2017, 9, 101–108. [Google Scholar] [CrossRef]
- Wall, L.; O’Donnell, J.C.; Schoenecker, P.L.; Keeler, K.A.; Dobbs, M.B.; Luhmann, S.J.; Gordon, J.E. Titanium elastic nailing radius and ulna fractures in adolescents. J. Pediatric Orthop. B 2012, 21, 482–488. Available online: https://journals.lww.com/01202412-201209000-00021 (accessed on 16 May 2022). [CrossRef]
- Antabak, A.; Luetic, T.; Ivo, S.; Karlo, R.; Cavar, S.; Bogovic, M.; Medacic, S.S. Treatment outcomes of both-bone diaphyseal paediatric forearm fractures. Injury 2013, 44, S11–S15. Available online: https://linkinghub.elsevier.com/retrieve/pii/S0020138313701906 (accessed on 16 May 2022). [CrossRef]
- Sahin, N. ESIN and K-wire fixation has similar results in both bone pediatric forearm fractures. Turk. J. Trauma Emerg. Surg. 2017, 23, 415–420. Available online: https://www.journalagent.com/travma/pdfs/UTD_23_5_415_420.pdf (accessed on 16 May 2022). [CrossRef] [PubMed]
- Kelly, B.A.; Miller, P.; Shore, B.J.; Waters, P.M.; Bae, D.S. Exposed Versus Buried Intramedullary Implants for Pediatric Forearm Fractures. J. Pediatric Orthop. 2014, 34, 749–755. Available online: https://journals.lww.com/01241398-201412000-00001 (accessed on 16 May 2022). [CrossRef] [PubMed]
- Lascombes, P.; Haumont, T.; Journeau, P. Use and Abuse of Flexible Intramedullary Nailing in Children and Adolescents. J. Pediatric Orthop. 2006, 26, 827–834. Available online: https://journals.lww.com/01241398-200611000-00031 (accessed on 16 May 2022). [CrossRef] [PubMed]
- Rousset, M.; Mansour, M.; Samba, A.; Pereira, B.; Canavese, F. Risk factors for re-fracture in children with diaphyseal fracture of the forearm treated with elastic stable intramedullary nailing. Eur. J. Orthop. Surg. Traumatol. 2016, 26, 145–152. Available online: http://link.springer.com/10.1007/s00590-015-1715-1 (accessed on 16 May 2022). [CrossRef] [PubMed]
- Amerstorfer, F.; Fischerauer, E.; Pearce, S.; Schmidz, B. Paediatric diaphyseal forearm refractures after greenstick fractures: Operative management with ESIN. Injury 2009, 40, 414–725. [Google Scholar]

| Country | Facility | Department | Adress | Facility Contact | Site Recruitment Status | Contact |
|---|---|---|---|---|---|---|
| Austria | Landesklinikum Baden/Mödling | Department of Orthopedics and Trauma | Sr. Maria Restitutagasse 12, 2340 Mödling | Christoph Röder | Recruiting | christoph.roeder@moedling.lknoe.at +43-2236-90040 |
| Denmark | Hvidovre Hospital | Department of Orthopedics | Ketttegaard alle 30, 2650 Hvidovre | Louise Klingenberg | Recruiting | louise.klingenberg.03@regionh.dk +45-40884943 |
| France | Jeanne de Flandre Hospital | Department of Pediatric Orthopedic Surgery | Avenue Eugene Avinée, 59000 LILLE | Federico Canavese | Recruiting | federico.canavese@chru-lille.fr +33-3-20446867 |
| Germany | University Medical Center Schleswig-Holstein, Campus Lübeck | Department of Pediatric Surgery | Ratzeburger Allee 160, 23538 Lübeck | Ludger Tüshaus | Recruiting | ludger.tueshaus@uksh.de +49-451-50042611 |
| Germany | Universitätsmedizin Mainz | Department of Orthopedics and Trauma Surgery | Langenbeckstraße 1 55131 Mainz | Erol Gercek | Not yet Recruiting | erol.gercek@web.de +49-6131-177292 |
| Hungary | Dr. Manninger Jenő Baleseti Közpon | Department of Pediatric Trauma Surgery | 1081 Fiumei út 17 Budapest | Marcell Varga | Recruiting | drvmarcell@gmail.com +36-70-9323027 |
| Hungary | Pécs University Hospital | Department of Pediatrics | Jozsef Attila u. 7, 7623 Pécs | Gergo Jozsa | Recruiting | dr.jozsa.gergo@gmail.com +36-72-535900 |
| Portugal | Hospital Pediátrico-CHUC, EPE | Department of Pediatric Orthopaedics | Av Afonso Romão 3000-602 Coimbra | Cristina Alves | Not yet Recruiting | 6443@chuc.min-saude.pt +351-239-480355 |
| Switzerland | Centre Hospitalier Universitaire Vaudois | Service de Chirurgie de l’enfant et de l’adolescent-SCEA | BH-11 Rue du Bugnon CH-1010 Lausanne | Nicolas Lutz | Not yet Recruiting | nicolas.lutz@chuv.ch +41-21-3148538 |
| Time Point | Prior to Inclusion | During Surgery | 2 Weeks after Surgery | 4 Weeks | 12 Weeks | 1 Year | 2 Years (Optional) |
|---|---|---|---|---|---|---|---|
| Eligibility | x | ||||||
| Informed consent | x | ||||||
| Type of implant | x | ||||||
| Radiological evaluation | x | x | x | x | |||
| MRI scan (optional) | x | x | |||||
| Duration of x-ray | x | ||||||
| Time of operation | x | ||||||
| Length of hospital stay | x | ||||||
| Pain | x | x | x | x | x | ||
| ROM of elbow and wrist | x | x | x | x | x | ||
| Complications | x | x | x | x | x | x | |
| (Serious) adverse events | x | x | x | x | x | x | |
| Cost-effectiveness | x |
| AE | SAE |
|---|---|
| Intra-operative complications related to fixation hardware | Device malfunction |
| Need for secondary reduction due to fracture instability | Hospitalization |
| Mechanical implant failure | Medically important events |
| Incidence of infection | Accidental exposure |
| Osteolysis | Life-threatening events |
| All Patients | Patients > 8 Months Follow Up | Patients > 12 Months Follow Up | |
|---|---|---|---|
| Number of patients (n) | 76 (100%) | 41 (54.0%) | 16 (21.0%) |
| Age (mean; sd; range) (years) | 8.87; ±2.41; 4–12 | 8.6; ±2.6; 4–12 | 8.9; ±2.1; 5–12 |
| Age group (3–7 years) (n) | 23 (30.3%) | 16 (39.0%) | 5 (31.3%) |
| Age group (8–10 years) (n) | 29 (38.2%) | 14 (34.2%) | 8 (50.0%) |
| Age group (11–13 years) (n) | 24 (31.6%) | 11 (26.8%) | 3 (18.8%) |
| Female (n) | 31 (40.8%) | 18 (43.9%) | 8 (50.0%) |
| Male (n) | 45 (59.2%) | 23 (56.1%) | 8 (50.0%) |
| Left (n) | 36 (47.4%) | 23 (56.1%) | 9 (56.2%) |
| Right (n) | 40 (52.6%) | 18 (43.9%) | 7 (43.8%) |
| Follow up (mean; sd; range) (months) | 8.9; ±5.1; 0.2–18.6 | 12.7; ±3.3; 8.2–18.6 | 16.4; ±2.2; 12.1–18.6 |
| Time from trauma to treatment (mean; sd; range) (hours) | 44.1; ±49.8; 2.7–261.0 | 31.3; ±32.5; 2.7–170.3 | 26.0; ±38.1; 2.7–170.3 |
| Number of patients treated within 24 h | 35 (46.1%) | 23 (56.1%) | 12 (75.0%) |
| Operation time (incision to wound closure) (mean; sd; range) (min) | 58.9; ±22.9; 15–119 | 57.2; ± 21.4; 20–105 | 63.3; ±20.4; 35–105 |
| Hospital stay (mean; sd; range) (days) | 1.6; ±0.8; 1–3 | 1.8; ±0.8; 1–3 | 2.4; ±0.6; 1–3 |
| Refracture rate (n) | 1 (1.3%) | 1 (2.4%) | 1 (6.2%) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Roeder, C.; Alves, C.; Balslev-Clausen, A.; Canavese, F.; Gercek, E.; Kassai, T.; Klestil, T.; Klingenberg, L.; Lutz, N.; Varga, M.; et al. Pilot Study and Preliminary Results of Biodegradable Intramedullary Nailing of Forearm Fractures in Children. Children 2022, 9, 754. https://doi.org/10.3390/children9050754
Roeder C, Alves C, Balslev-Clausen A, Canavese F, Gercek E, Kassai T, Klestil T, Klingenberg L, Lutz N, Varga M, et al. Pilot Study and Preliminary Results of Biodegradable Intramedullary Nailing of Forearm Fractures in Children. Children. 2022; 9(5):754. https://doi.org/10.3390/children9050754
Chicago/Turabian StyleRoeder, Christoph, Cristina Alves, Andreas Balslev-Clausen, Federico Canavese, Erol Gercek, Tamás Kassai, Thomas Klestil, Louise Klingenberg, Nicolas Lutz, Marcell Varga, and et al. 2022. "Pilot Study and Preliminary Results of Biodegradable Intramedullary Nailing of Forearm Fractures in Children" Children 9, no. 5: 754. https://doi.org/10.3390/children9050754
APA StyleRoeder, C., Alves, C., Balslev-Clausen, A., Canavese, F., Gercek, E., Kassai, T., Klestil, T., Klingenberg, L., Lutz, N., Varga, M., Jozsa, G., Weinberg, A., & Tüshaus, L. (2022). Pilot Study and Preliminary Results of Biodegradable Intramedullary Nailing of Forearm Fractures in Children. Children, 9(5), 754. https://doi.org/10.3390/children9050754








