Antidepressive-like Behavior-Related Metabolomic Signatures of Sigma-1 Receptor Knockout Mice
Abstract
:1. Introduction
2. Materials and Methods
2.1. Animals
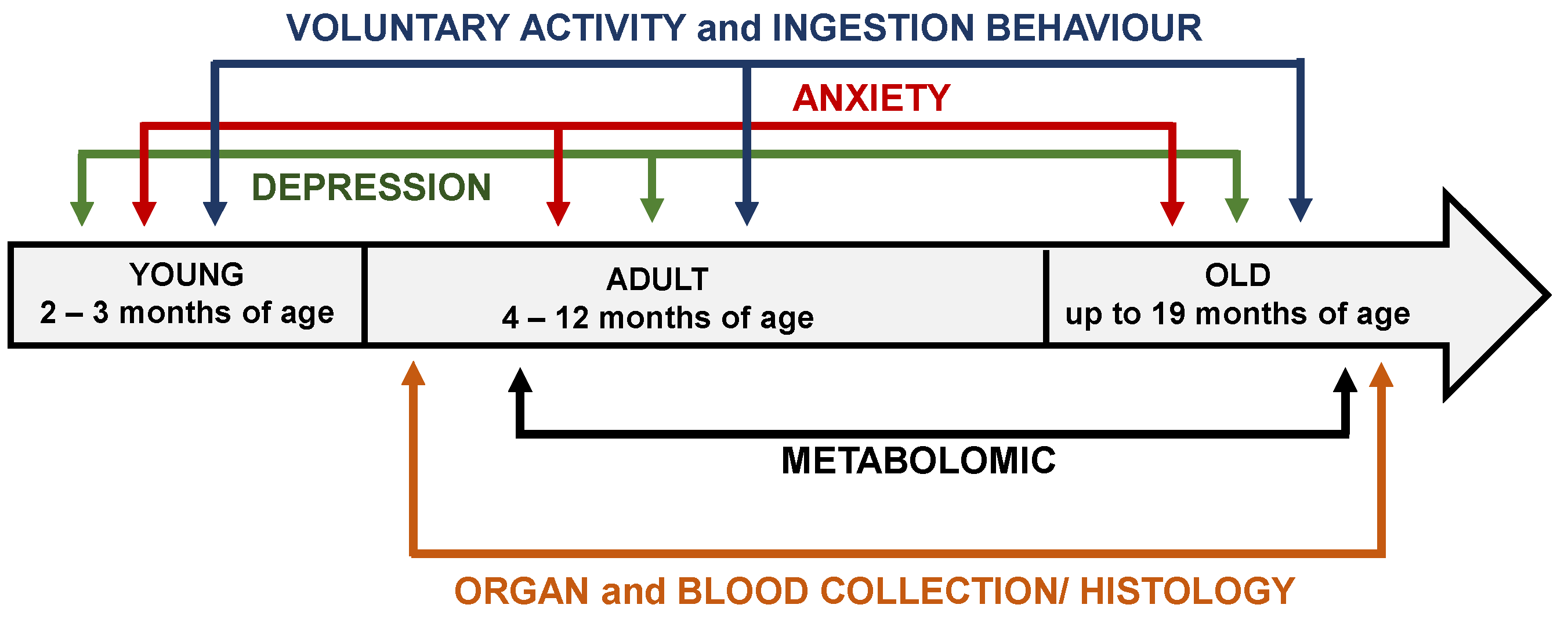
2.2. Experimental design
2.3. Detection of Endogenous Metabolites
2.4. Voluntary Activity and Ingestion Behavior
2.5. Depression-like Behavior
2.5.1. Tail Suspension Test
2.5.2. Sucrose Preference Test
2.6. Anxiety-like Behavior
2.6.1. Zero Maze Test
2.6.2. Open-Field Test
2.7. Quantitative PCR (qPCR) Analysis
2.8. Weight of Organs
2.9. HistologyBrain Tissue Preparation for Histological Analysis
Brain Tissue Preparation for Histological Analysis
2.10. Immunohistochemistry
2.10.1. Staining of Serotonin (5-HT) and Tryptophan Hydroxylase 2 (TPH2)
2.10.2. Image Analysis
2.11. Statistical Analysis
3. Results
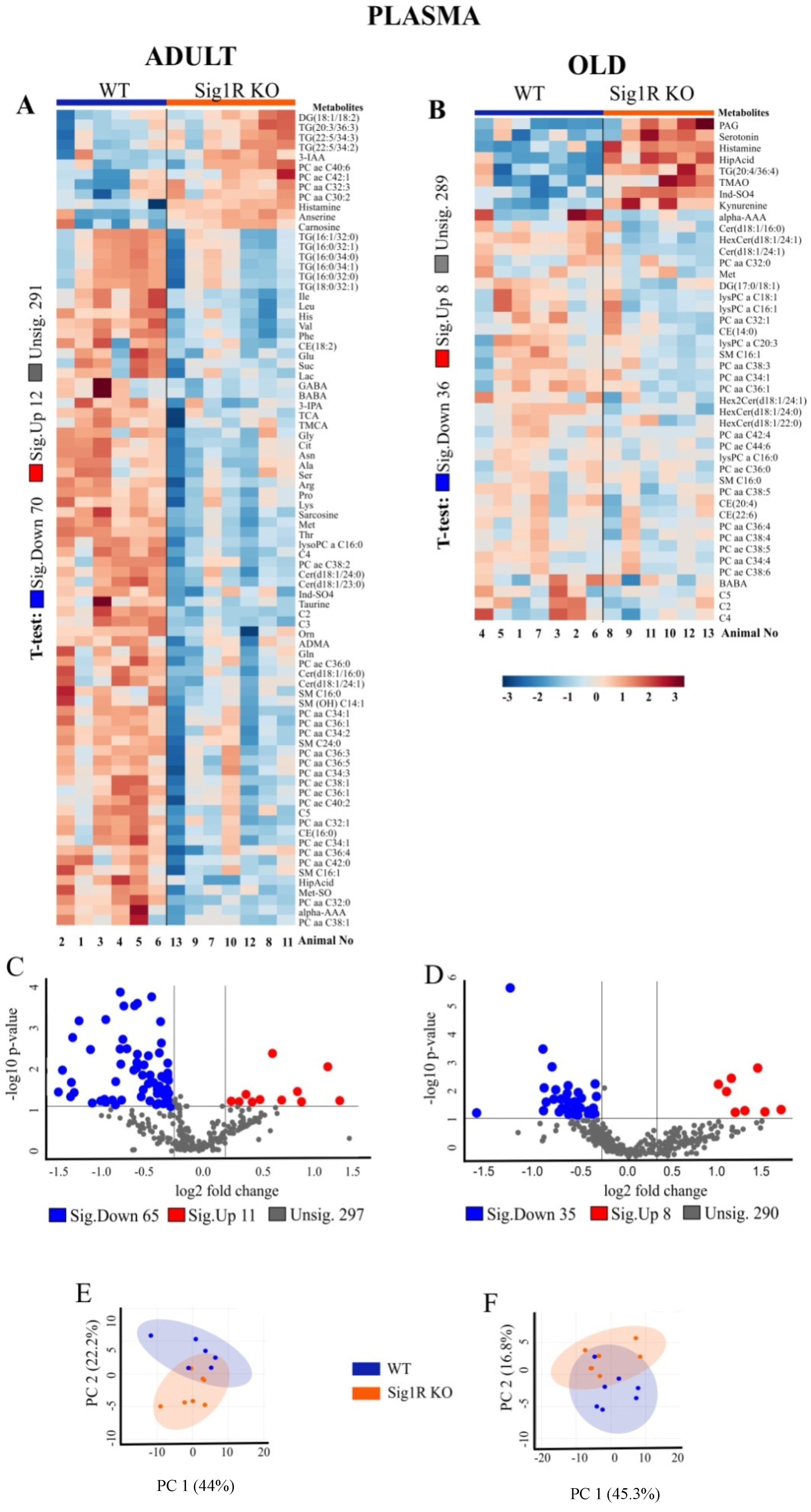
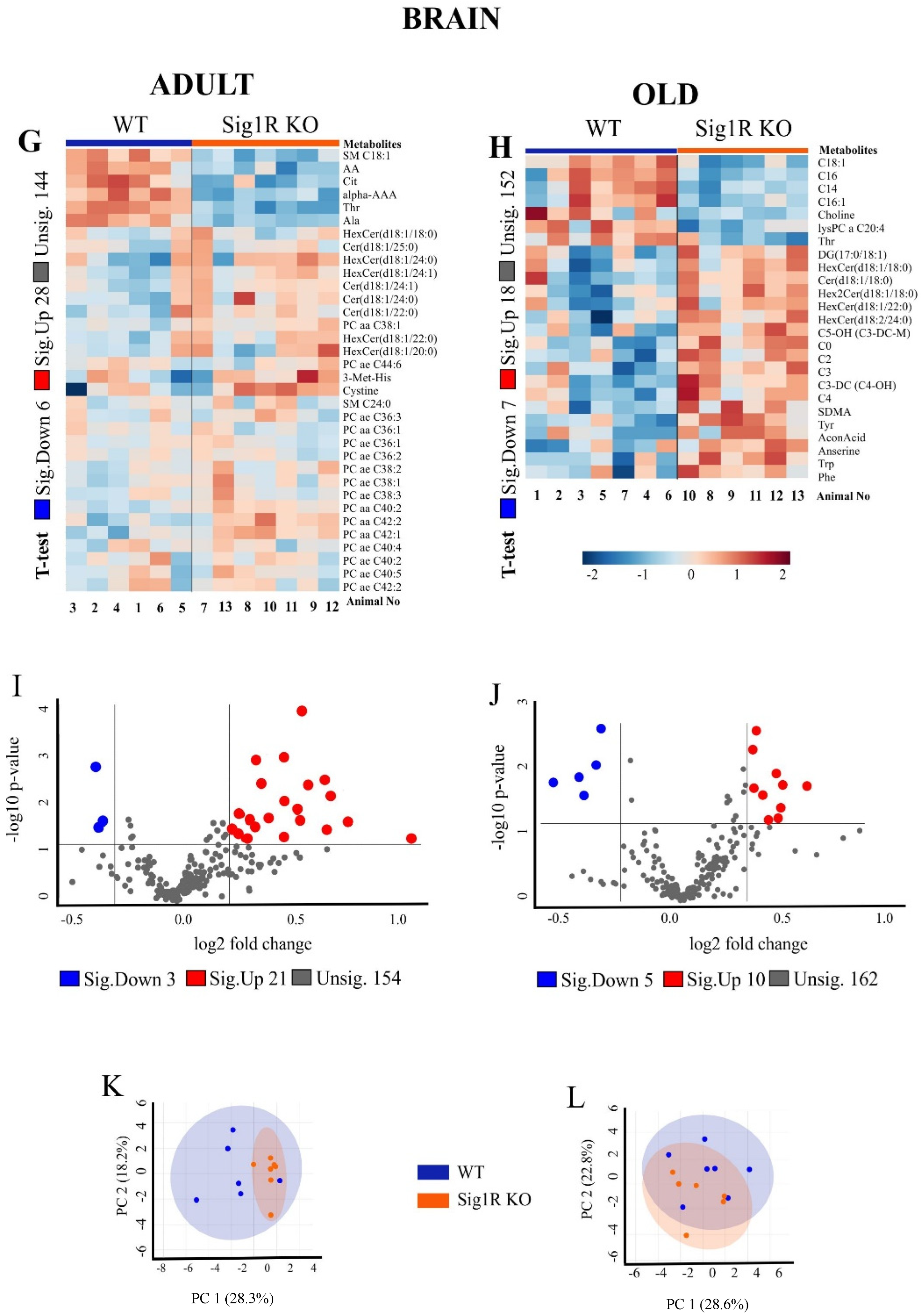
3.1. Age- and Genotype-Related Metabolic Signatures in Plasma and Brain Tissue of Sig1R KO Mice
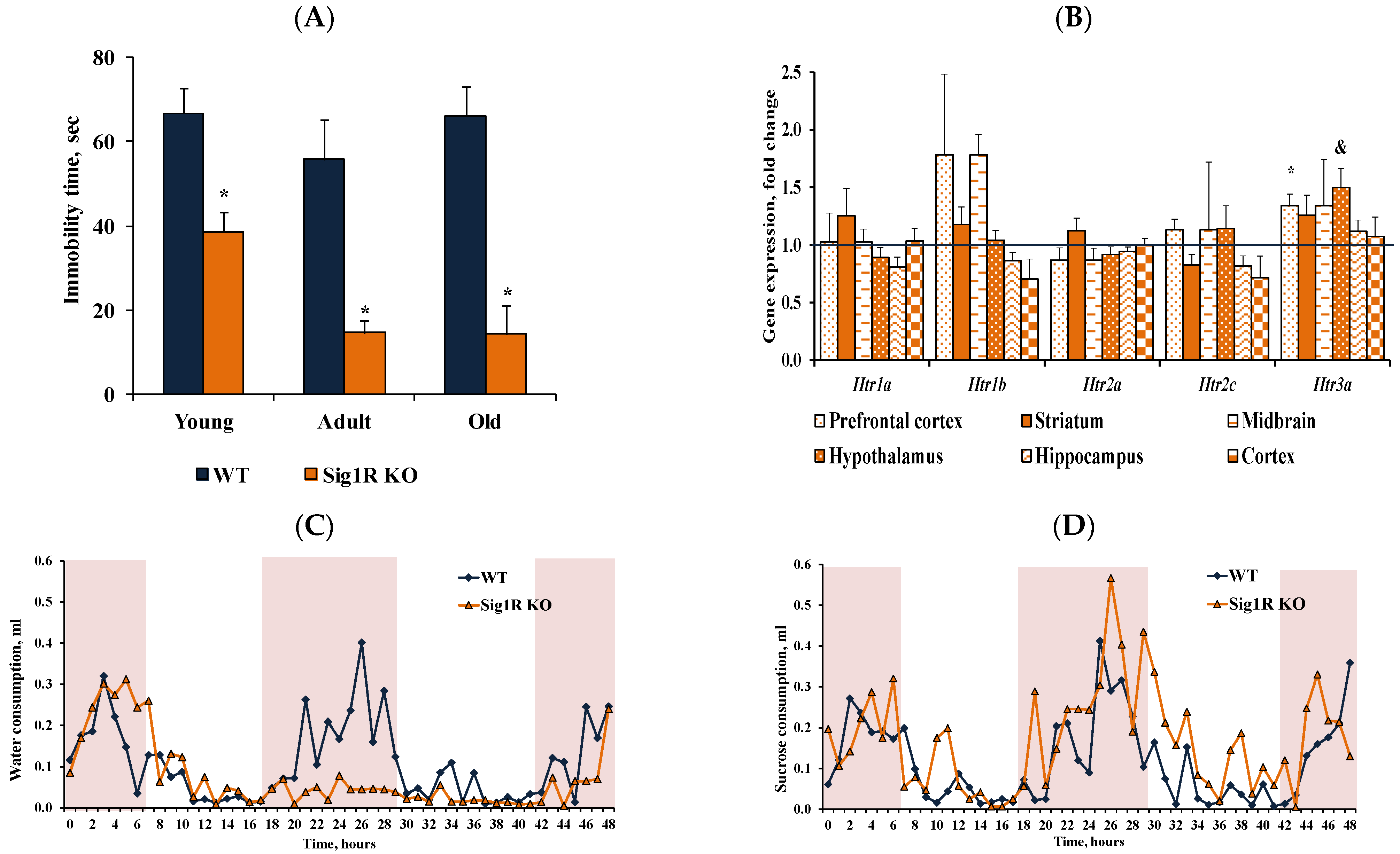
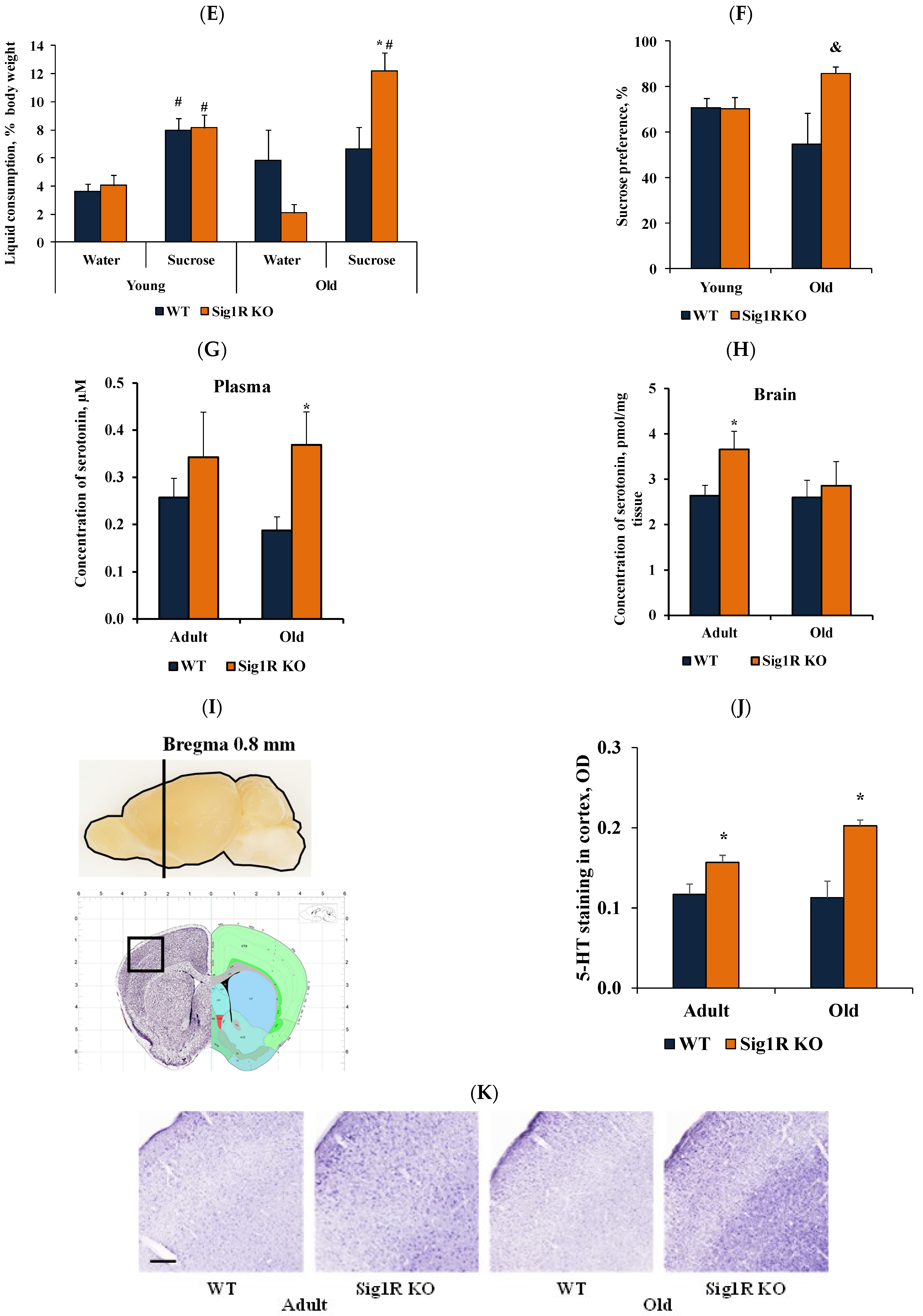
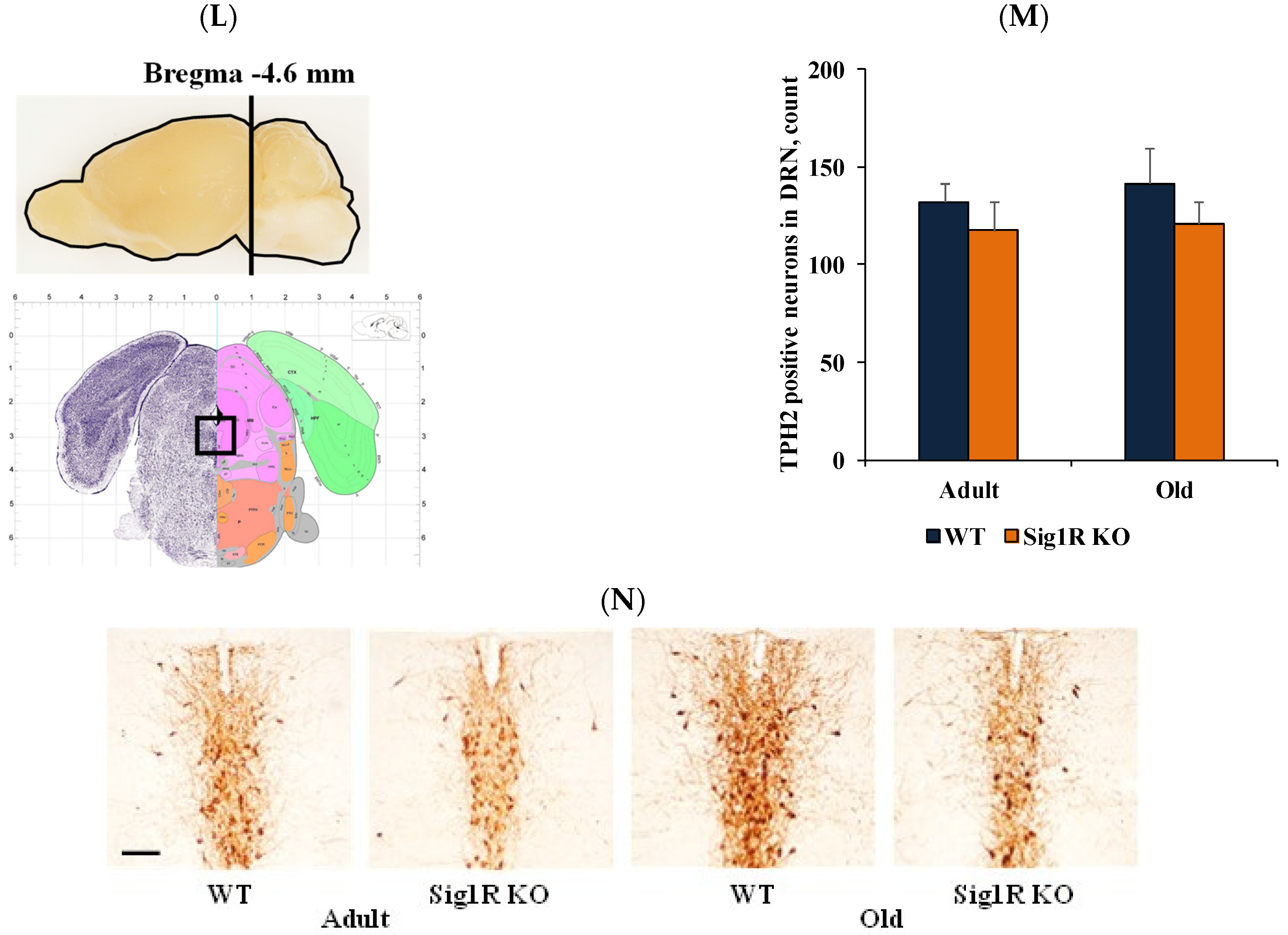
3.2. Reduced Behavioral Despair in Sig1R KO Mice Is Associated with Increased Levels of Serotonin
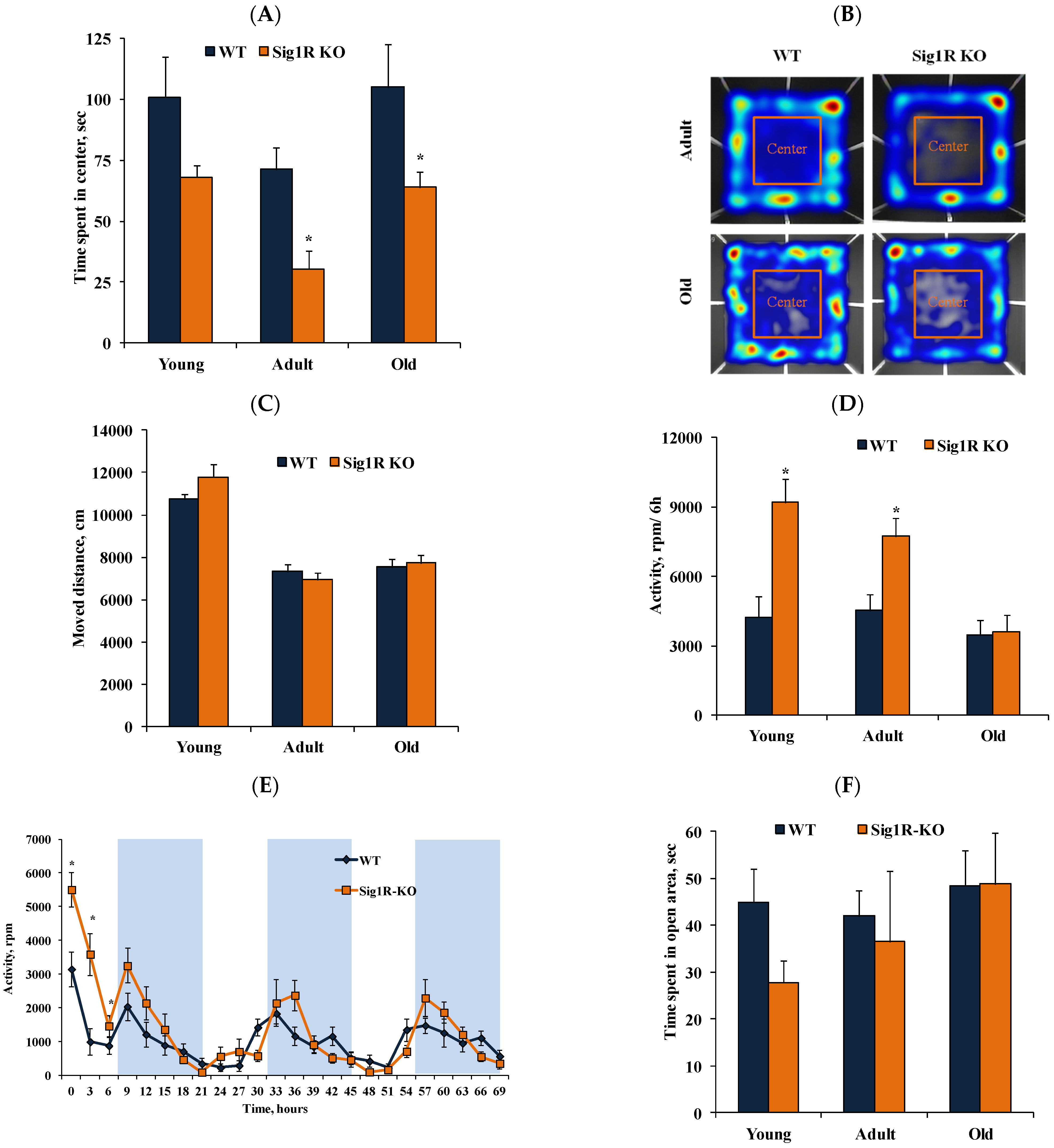
3.3. Anxiety-like Behavior Characteristics of CD-1 Background Sig1R KO Mice
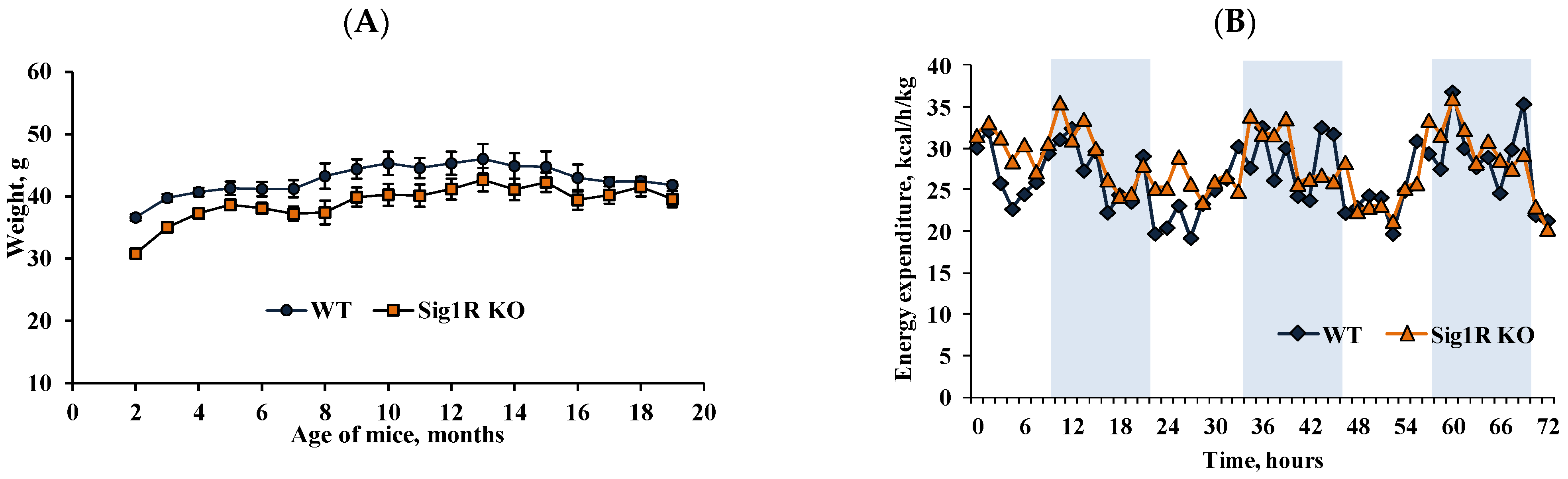
3.4. Physiological and Anatomical Characteristics of Sig1R KO Mice
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Alonso, G.; Phan, V.L.; Guillemain, I.; Saunier, M.; Legrand, A.; Anoal, M.; Maurice, T. Immunocytochemical localization of the sigma1 receptor in the adult rat central nervous system. Neuroscience 2000, 97, 155–170. [Google Scholar] [CrossRef]
- Kitaichi, K.; Chabot, J.G.; Moebius, F.F.; Flandorfer, A.; Glossmann, H.; Quirion, R. Expression of the purported sigma1 (σ1) receptor in the mammalian brain and its possible relevance in deficits induced by antagonism of the NMDA receptor complex as revealed using an antisense strategy. J. Chem. Neuroanat. 2000, 20, 375–387. [Google Scholar] [CrossRef]
- Su, T.P.; Junien, J.L. Sigma receptors: Sigma receptors in the central nervous system and the periphery. In Sigma Receptors: Chemistry, Cell Biology and Clinical Implications., 1st ed.; Itzhak, Y., Ed.; Academic Press: Cambridge, MA, USA, 1994; pp. 1–413. [Google Scholar]
- Hayashi, T.; Su, T.P. Sigma-1 Receptor Chaperones at the ER- Mitochondrion Interface Regulate Ca2+ Signaling and Cell Survival. Cell 2007, 131, 596–610. [Google Scholar] [CrossRef] [Green Version]
- Su, T.P.; Su, T.C.; Nakamura, Y.; Tsai, S.Y. The Sigma-1 Receptor as a Pluripotent Modulator in Living Systems. Trends Pharmacol. Sci. 2016, 37, 262–278. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Maurice, T.; Su, T.P.; Privat, A. Sigma1 (σ1) receptor agonists and neurosteroids attenuate b25-35-amyloid peptide-induced amnesia in mice through a common mechanism. Neuroscience 1998, 83, 413–428. [Google Scholar] [CrossRef]
- Mavlyutov, T.A.; Epstein, M.L.; Verbny, Y.I.; Huerta, M.S.; Zaitoun, I.; Ziskind-Conhaim, L.; Ruoho, A.E. Lack of sigma-1 receptor exacerbates ALS progression in mice. Neuroscience 2013, 240, 129–134. [Google Scholar] [CrossRef] [Green Version]
- Gris, G.; Merlos, M.; Vela, J.M.; Zamanillo, D.; Portillo-Salido, E. S1RA, a selective sigma-1 receptor antagonist, inhibits inflammatory pain in the carrageenan and complete Freund’s adjuvant models in mice. Behav. Pharmacol. 2014, 25, 226–235. [Google Scholar] [CrossRef]
- Couly, S.; Goguadze, N.; Yasui, Y.; Kimura, Y.; Wang, S.M.; Sharikadze, N.; Wu, H.E.; Su, T.P. Knocking Out Sigma-1 Receptors Reveals Diverse Health Problems. Cell Mol. Neurobiol. 2022, 42, 597–620. [Google Scholar] [CrossRef]
- Aishwarya, R.; Abdullah, C.S.; Morshed, M.; Remex, N.S.; Bhuiyan, M.S. Sigmar1’s Molecular, Cellular, and Biological Functions in Regulating Cellular Pathophysiology. Front. Physiol. 2021, 12, 705575. [Google Scholar] [CrossRef] [PubMed]
- Rosen, D.A.; Seki, S.M.; Fernández-Castañeda, A.; Beiter, R.M.; Eccles, J.D.; Woodfolk, J.A.; Gaultier, A. Modulation of the sigma-1 receptor-IRE1 pathway is beneficial in preclinical models of inflammation and sepsis. Sci. Transl. Med. 2019, 11, eaau5266. [Google Scholar] [CrossRef]
- Vela, J.M. Repurposing Sigma-1 Receptor Ligands for COVID-19 Therapy? Front. Pharmacol. 2020, 11, 582310. [Google Scholar] [CrossRef] [PubMed]
- Kulkarni, S.K.; Dhir, A. σ-1 receptors in major depression and anxiety. Expert Rev. Neurother. 2009, 9, 1021–1034. [Google Scholar] [CrossRef] [PubMed]
- Muglia, P.; Tozzi, F.; Galwey, N.W.; Francks, C.; Upmanyu, R.; Kong, X.Q.; Antoniades, A.; Domenici, E.; Perry, J.; Rothen, S.; et al. Genome-wide association study of recurrent major depressive disorder in two European case-control cohorts. Mol. Psychiatry 2010, 15, 589–601. [Google Scholar] [CrossRef] [PubMed]
- Howard, D.M.; Adams, M.J.; Clarke, T.K.; Hafferty, J.D.; Gibson, J.; Shirali, M.; Coleman, J.R.I.; Hagenaars, S.P.; Ward, J.; Wigmore, E.M.; et al. Genome-wide meta-analysis of depression identifies 102 independent variants and highlights the importance of the prefrontal brain regions. Nat. Neurosci. 2019, 22, 343–352. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kishi, T.; Yoshimura, R.; Okochi, T.; Fukuo, Y.; Kitajima, T.; Okumura, T.; Tsunoka, T.; Kawashima, K.; Yamanouchi, Y.; Kinoshita, Y.; et al. Association analysis of SIGMAR1 with major depressive disorder and SSRI response. Neuropharmacology 2010, 58, 1168–1173. [Google Scholar] [CrossRef]
- Mandelli, L.; Wang, S.M.; Han, C.; Lee, S.J.; Patkar, A.A.; Masand, P.S.; Pae, C.U.; Serretti, A. The Impact of a Single Nucleotide Polymorphism in SIGMAR1 on Depressive Symptoms in Major Depressive Disorder and Bipolar Disorder. Adv. Ther. 2017, 34, 713–724. [Google Scholar] [CrossRef]
- Langa, F.; Codony, X.; Tovar, V.; Lavado, A.; Giménez, E.; Cozar, P.; Cozar, P.; Cantero, M.; Dordal, A.; Hernández, E.; et al. Generation and phenotypic analysis of sigma receptor type I (σ1) knockout mice. Eur. J. Neurosci. 2003, 18, 2188–2196. [Google Scholar] [CrossRef]
- Chevallier, N.; Keller, E.; Maurice, T. Behavioural phenotyping of knockout mice for the sigma-1 (σ1) chaperone protein revealed gender-related anxiety, depressive-like and memory alterations. J. Psychopharmacol. 2011, 25, 960–975. [Google Scholar] [CrossRef]
- Sabino, V.; Cottone, P.; Parylak, S.L.; Steardo, L.; Zorrilla, E.P. Sigma-1 receptor knockout mice display a depressive-like phenotype. Behav. Brain Res. 2009, 198, 472–476. [Google Scholar] [CrossRef] [Green Version]
- Sha, S.; Hong, J.; Qu, W.J.; Lu, Z.H.; Li, L.; Yu, W.F.; Chen, L. Sex-related neurogenesis decrease in hippocampal dentate gyrus with depressive-like behaviors in sigma-1 receptor knockout mice. Eur. Neuropsychopharmacol. 2015, 25, 1275–1286. [Google Scholar] [CrossRef]
- Abdullah, C.S.; Alam, S.; Aishwarya, R.; Miriyala, S.; Panchatcharam, M.; Bhuiyan, M.A.N.; Peretik, J.M.; Orr, A.W.; James, J.; Osinska, H.; et al. Cardiac dysfunction in the sigma 1 receptor knockout mouse associated with impaired mitochondrial dynamics and bioenergetics. J. Am. Heart Assoc. 2018, 7, e009775. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Di, T.; Zhang, S.; Hong, J.; Zhang, T.; Chen, L. Hyperactivity of hypothalamic-pituitary-adrenal axis due to dysfunction of the hypothalamic glucocorticoid receptor in sigma-1 receptor knockout mice. Front. Mol. Neurosci. 2017, 10, 287. [Google Scholar] [CrossRef] [PubMed]
- Lucki, I.; Dalvi, A.; Mayorga, A.J. Sensitivity to the effects of pharmacologically selective antidepressants in different strains of mice. Psychopharmacology 2001, 155, 315–322. [Google Scholar] [CrossRef] [PubMed]
- Stelfa, G.; Vavers, E.; Svalbe, B.; Serzants, R.; Miteniece, A.; Lauberte, L.; Grinberga, S.; Gukalova, B.; Dambrova, M.; Zvejniece, L. Reduced GFAP expression in bergmann glial cells in the cerebellum of sigma-1 receptor knockout mice determines the neurobehavioral outcomes after traumatic brain injury. Int. J. Mol. Sci. 2021, 22, 11611. [Google Scholar] [CrossRef]
- Boivin, J.R.; Piekarski, D.J.; Wahlberg, J.K.; Wilbrecht, L. Age, sex, and gonadal hormones differently influence anxiety- and depression-related behavior during puberty in mice. Psychoneuroendocrinology 2017, 85, 78–87. [Google Scholar] [CrossRef]
- Gonzalez, F.J.; Fang, Z.Z.; Ma, X. Transgenic mice and metabolomics for study of hepatic xenobiotic metabolism and toxicity. Expert Opin. Drug Metab. Toxicol. 2015, 11, 869–881. [Google Scholar] [CrossRef] [Green Version]
- Pal, A.; Fontanilla, D.; Gopalakrishnan, A.; Chae, Y.K.; Markley, J.L.; Ruoho, A.E. The sigma-1 receptor protects against cellular oxidative stress and activates antioxidant response elements. Eur. J. Pharmacol. 2012, 682, 12–20. [Google Scholar] [CrossRef] [Green Version]
- Percie du Sert, N.; Hurst, V.; Ahluwalia, A.; Alam, S.; Avey, M.T.; Baker, M.; Browne, W.J.; Clark, A.; Cuthill, I.C.; Dirnagl, U.; et al. The ARRIVE guidelines 2.0, Updated guidelines for reporting animal research. Br. J. Pharmacol. 2020, 177, 3617–3624. [Google Scholar] [CrossRef]
- Entrena, J.M.; Cobos, E.J.; Nieto, F.R.; Cendán, C.M.; Gris, G.; Del Pozo, E.; Zamanillo, D.; Baeyens, J.M. Sigma-1 receptors are essential for capsaicin-induced mechanical hypersensitivity: Studies with selective sigma-1 ligands and sigma-1 knockout mice. Pain 2009, 143, 252–261. [Google Scholar] [CrossRef]
- Gougoula, C.; Bielfeld, A.P.; Pour, S.J.; Sager, M.; Krüssel, J.S.; Benten, W.P.M.; Baston-Büst, D.M. Metabolic and behavioral parameters of mice with reduced expression of Syndecan-1. PLoS ONE 2019, 14, e0219604. [Google Scholar] [CrossRef] [Green Version]
- Can, A.; Dao, D.T.; Terrillion, C.E.; Piantadosi, S.C.; Bhat, S.; Gould, T.D. The tail suspension test. J. Vis. Exp. 2011, 2011, e3769. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Koehl, M.; Ladevèze, E.; Catania, C.; Cota, D.; Abrous, D.N. Inhibition of mTOR signaling by genetic removal of p70 S6 kinase 1 increases anxiety-like behavior in mice. Transl. Psychiatry 2021, 11, 165. [Google Scholar] [CrossRef] [PubMed]
- Zvejniece, L.; Svalbe, B.; Vavers, E.; Makrecka-Kuka, M.; Makarova, E.; Liepins, V.; Kalvinsh, I.; Liepinsh, E.; Dambrova, M. S-phenylpiracetam, a selective DAT inhibitor, reduces body weight gain without influencing locomotor activity. Pharmacol. Biochem. Behav. 2017, 160, 21–29. [Google Scholar] [CrossRef] [PubMed]
- Ye, J.; Coulouris, G.; Zaretskaya, I.; Cutcutache, I.; Rozen, S.; Madden, T.L. Primer-BLAST: A tool to design target-specific primers for polymerase chain reaction. BMC Bioinformatics 2012, 13, 134. [Google Scholar] [CrossRef] [Green Version]
- Vavers, E.; Zvejniece, B.; Stelfa, G.; Svalbe, B.; Vilks, K.; Kupats, E.; Mezapuke, R.; Lauberte, L.; Dambrova, M.; Zvejniece, L. Genetic inactivation of the sigma-1 chaperone protein results in decreased expression of the R2 subunit of the GABA-B receptor and increased susceptibility to seizures. Neurobiol. Dis. 2021, 150, 105244. [Google Scholar] [CrossRef]
- Pu, J.; Liu, Y.; Zhang, H.; Tian, L.; Gui, S.; Yu, Y.; Chen, X.; Chen, Y.; Yang, L.; Ran, Y.; et al. An integrated meta-analysis of peripheral blood metabolites and biological functions in major depressive disorder. Mol. Psychiatry 2021, 26, 4265–4276. [Google Scholar] [CrossRef] [Green Version]
- Kalinichenko, L.S.; Gulbins, E.; Kornhuber, J.; Müller, C.P. Serotonin–lipid interactions and their role in behavior. Handb. Behav. Neurosci. 2020, 31, 289–308. [Google Scholar]
- Sharp, T.; Barnes, N.M. Central 5-HT receptors and their function; present and future. Neuropharmacology 2020, 177, 108155. [Google Scholar] [CrossRef]
- Kornhuber, J.; Müller, C.P.; Becker, K.A.; Reichel, M.; Gulbins, E. The ceramide system as a novel antidepressant target. Trends Pharmacol. Sci. 2014, 35, 293–304. [Google Scholar] [CrossRef]
- Kurz, J.; Parnham, M.J.; Geisslinger, G.; Schiffmann, S. Ceramides as Novel Disease Biomarkers. Trends Mol. Med. 2019, 25, 20–32. [Google Scholar] [CrossRef]
- Dinoff, A.; Herrmann, N.; Lanctôt, K.L. Ceramides and depression: A systematic review. J. Affect. Disord. 2017, 213, 35–43. [Google Scholar] [CrossRef] [PubMed]
- Demirkan, A.; Isaacs, A.; Ugocsai, P.; Liebisch, G.; Struchalin, M.; Rudan, I.; Wilson, J.F.; Pramstaller, P.P.; Gyllensten, U.; Campbell, H.; et al. Plasma phosphatidylcholine and sphingomyelin concentrations are associated with depression and anxiety symptoms in a Dutch family-based lipidomics study. J. Psychiatr. Res. 2013, 47, 357–362. [Google Scholar] [CrossRef] [PubMed]
- Ramachandran, S.; Chu, U.B.; Mavlyutov, T.A.; Pal, A.; Pyne, S.; Ruoho, A.E. The sigma1 receptor interacts with N-alkyl amines and endogenous sphingolipids. Eur. J. Pharmacol. 2009, 609, 19–26. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hayashi, T.; Fujimoto, M. Detergent-resistant microdomains determine the localization of σ-1 receptors to the endoplasmic reticulum-mitochondria junction. Mol. Pharmacol. 2010, 77, 517–528. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hayashi, T.; Hayashi, E.; Fujimoto, M.; Sprong, H.; Su, T.P. The lifetime of UDP-galactose: Ceramide galactosyltransferase is controlled by a distinct endoplasmic reticulum-associated degradation (ERAD) regulated by sigma-1 receptor chaperones. J. Biol. Chem. 2012, 287, 43156–43169. [Google Scholar] [CrossRef] [Green Version]
- Cruciani-Guglielmacci, C.; López, M.; Campana, M.; le Stunff, H. Brain ceramide metabolism in the control of energy balance. Front. Physiol. 2017, 8, 787. [Google Scholar] [CrossRef] [Green Version]
- Gulbins, E.; Palmada, M.; Reichel, M.; Lüth, A.; Böhmer, C.; Amato, D.; Müller, C.P.; Tischbirek, C.H.; Groemer, T.W.; Tabatabai, G.; et al. Acid sphingomyelinase-ceramide system mediates effects of antidepressant drugs. Nat. Med. 2013, 19, 934–938. [Google Scholar] [CrossRef] [Green Version]
- Zoicas, I.; Huber, S.E.; Kalinichenko, L.S.; Gulbins, E.; Müller, C.P.; Kornhuber, J. Ceramides affect alcohol consumption and depressive-like and anxiety-like behavior in a brain region- and ceramide species-specific way in male mice. Addict. Biol. 2020, 25, e12847. [Google Scholar] [CrossRef] [Green Version]
- Aldinger, K.A.; Sokoloff, G.; Rosenberg, D.M.; Palmer, A.A.; Millen, K.J. Genetic variation and population substructure in outbred CD-1 mice: Implications for genome-wide association studies. PLoS ONE 2009, 4, e4729. [Google Scholar] [CrossRef] [Green Version]
- Rice, M.C.; O’Brien, S.J. Genetic variance of laboratory outbred Swiss mice. Nature 1980, 283, 157–161. [Google Scholar] [CrossRef]
- Carneiro, A.M.D.; Airey, D.C.; Thompson, B.; Zhu, C.; Bin, L.L.; Chesler, E.J.; Chesler, E.J.; Erikson, K.M.; Blakely, R.D. Functional coding variation in recombinant inbred mouse lines reveals multiple serotonin transporter-associated phenotypes. Proc. Natl. Acad. Sci. USA 2009, 106, 2047–2052. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhao, H.; Zhang, B.L.; Yang, S.J.; Rusak, B. The role of lateral habenula-dorsal raphe nucleus circuits in higher brain functions and psychiatric illness. Behav. Brain Res. 2015, 277, 89–98. [Google Scholar] [CrossRef] [PubMed]
- Metzger, M.; Bueno, D.; Lima, L.B. The lateral habenula and the serotonergic system. Pharmacol. Biochem. Behav. 2017, 162, 22–28. [Google Scholar] [CrossRef] [PubMed]
- Browne, C.A.; Hammack, R.; Lucki, I. Dysregulation of the Lateral Habenula in Major Depressive Disorder. Front. Synaptic Neurosci. 2018, 10, 46. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bahi, A.; Schwed, J.S.; Walter, M.; Stark, H.; Sadek, B. Anxiolytic and antidepressant-like activities of the novel and potent non-imidazole histamine H3 receptor antagonist ST-1283. Drug Des. Dev. Ther. 2014, 8, 627–637. [Google Scholar]
- Haas, H.L.; Sergeeva, O.A.; Selbach, O. Histamine in the nervous system. Physiol. Rev. 2008, 88, 1183–1241. [Google Scholar] [CrossRef]
- Sanna, M.D.; Ghelardini, C.; Thurmond, R.L.; Masini, E.; Galeotti, N. Behavioural phenotype of histamine H4 receptor knockout mice: Focus on central neuronal functions. Neuropharmacology 2017, 114, 48–57. [Google Scholar] [CrossRef] [PubMed]
- Acevedo, S.F.; Ohtsu, H.; Benice, T.S.; Rizk-Jackson, A.; Raber, J. Age-dependent measures of anxiety and cognition in male histidine decarboxylase knockout (Hdc-/-) mice. Brain Res. 2006, 1071, 113–123. [Google Scholar] [CrossRef]
- Wu, G. Amino acids: Metabolism, functions, and nutrition. Amino Acids. 2009, 37, 1–17. [Google Scholar] [CrossRef]
- Yang, J.; Yan, B.; Zhao, B.; Fan, Y.; He, X.; Yang, L.; Ma, Q.; Zheng, J.; Wang, W.; Bai, L.; et al. Assessing the causal effects of human serum metabolites on 5 major psychiatric disorders. Schizophr Bull. 2020, 46, 804–813. [Google Scholar] [CrossRef]
- Crouzier, L.; Denus, M.; Richard, E.M.; Maurice, T.; Delprat, B.; Tavernier, A.; Tavernier, A.; Diez, C.; Cubedo, N. Sigma-1 receptor is critical for mitochondrial activity and unfolded protein response in larval zebrafish. Int. J. Mol. Sci. 2021, 22, 11049. [Google Scholar] [CrossRef] [PubMed]

| Gene Name | Sigmar1 | Pgrmc1 | Tph2 | Gabbr2 | Drd1 | Drd2 | Dnajc14 (Drip78) |
|---|---|---|---|---|---|---|---|
| Protein Name | Sigma-1 Receptor | Sigma-2 Receptor | Tryptophan 5-Hydroxylase 2 | GABA-B Receptor Subunit 2 | D(1A) Dopamine Receptor | D(2) Dopamine Receptor | Dopamine Receptor Interacting Protein |
| Prefrontal cortex | - | 1 ± 0.0 | 1.1 ± 0.1 | 1.1 ± 0.1 | 0.9 ± 0.2 | 0.7 ± 0.3 | 0.7 ± 0.2 |
| Striatum | - | 0.9 ± 0.1 | 1.5 ± 0.2 | 1.6 ± 0.3 | 1 ± 0.1 | 1 ± 0.2 | 1.1 ± 0.5 |
| Hypothalamus | - | 1 ± 0.1 | 0.9 ± 0.2 | 1 ± 0.1 | 1.1 ± 0.2 | 1 ± 0.1 | 0.6 ± 0.1 |
| Midbrain | - | 1.1 ± 0.2 | 0.8 ± 0.2 | 0.8 ± 0.1 | 1.8 ± 0.6 | 1 ± 0.1 | 1.1 ± 0.2 |
| Hippocampus | - | 0.8 ± 0.1 | 1.3 ± 0.2 | 0.9 ± 0.1 | 0.9 ± 0.1 | 0.9 ± 0.1 | 0.8 ± 0.0 |
| Cortex | - | 0.8 ± 0.3 | 1 ± 0.1 | 1.5 ± 0.2 * | 0.7 ± 0.2 | 0.7 ± 0.3 | 1 ± 0.2 |
| Organs/Group | Organs Weights, % of Body Weight | |||
|---|---|---|---|---|
| 4 M | 6 M | 18 M | ||
| Brain | WT | 1.2 ± 0.02 | 1.2 ± 0.05 | 1.3 ± 0.02 |
| Sig1R KO | 1.3 ± 0.03 * | 1.3 ± 0.08 * | 1.4 ± 0.03 * | |
| Heart | WT | 0.5 ± 0.01 | 0.5 ± 0.03 | 0.5 ± 0.02 |
| Sig1R KO | 0.4 ± 0.01 | 0.4 ± 0.01 * | 0.4 ± 0.05 * | |
| Kidney | WT | 1.2 ± 0.05 | 1.6 ± 0.07 | 1.6 ± 0.06 |
| Sig1R KO | 1.2 ± 0.03 | 1.3 ± 0.03 * | 1.3 ± 0.05 * | |
| Pancreas | WT | 0.7 ± 0.07 | 1.0 ± 0.11 | 0.9 ± 0.07 |
| Sig1R KO | 0.8 ± 0.06 | 0.5 ± 0.08 * | 0.6 ± 0.05 * | |
| Liver | WT | 4.5 ± 0.09 | 5.2 ± 0.43 | 5.2 ± 0.33 |
| Sig1R KO | 4.4 ± 0.10 | 4.8 ± 0.35 | 4.9 ± 0.35 | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Svalbe, B.; Zvejniece, B.; Stelfa, G.; Vilks, K.; Vavers, E.; Vela, J.M.; Dambrova, M.; Zvejniece, L. Antidepressive-like Behavior-Related Metabolomic Signatures of Sigma-1 Receptor Knockout Mice. Biomedicines 2022, 10, 1572. https://doi.org/10.3390/biomedicines10071572
Svalbe B, Zvejniece B, Stelfa G, Vilks K, Vavers E, Vela JM, Dambrova M, Zvejniece L. Antidepressive-like Behavior-Related Metabolomic Signatures of Sigma-1 Receptor Knockout Mice. Biomedicines. 2022; 10(7):1572. https://doi.org/10.3390/biomedicines10071572
Chicago/Turabian StyleSvalbe, Baiba, Baiba Zvejniece, Gundega Stelfa, Karlis Vilks, Edijs Vavers, José Miguel Vela, Maija Dambrova, and Liga Zvejniece. 2022. "Antidepressive-like Behavior-Related Metabolomic Signatures of Sigma-1 Receptor Knockout Mice" Biomedicines 10, no. 7: 1572. https://doi.org/10.3390/biomedicines10071572
APA StyleSvalbe, B., Zvejniece, B., Stelfa, G., Vilks, K., Vavers, E., Vela, J. M., Dambrova, M., & Zvejniece, L. (2022). Antidepressive-like Behavior-Related Metabolomic Signatures of Sigma-1 Receptor Knockout Mice. Biomedicines, 10(7), 1572. https://doi.org/10.3390/biomedicines10071572







