Plants-Derived Biomolecules as Potent Antiviral Phytomedicines: New Insights on Ethnobotanical Evidences against Coronaviruses
Abstract
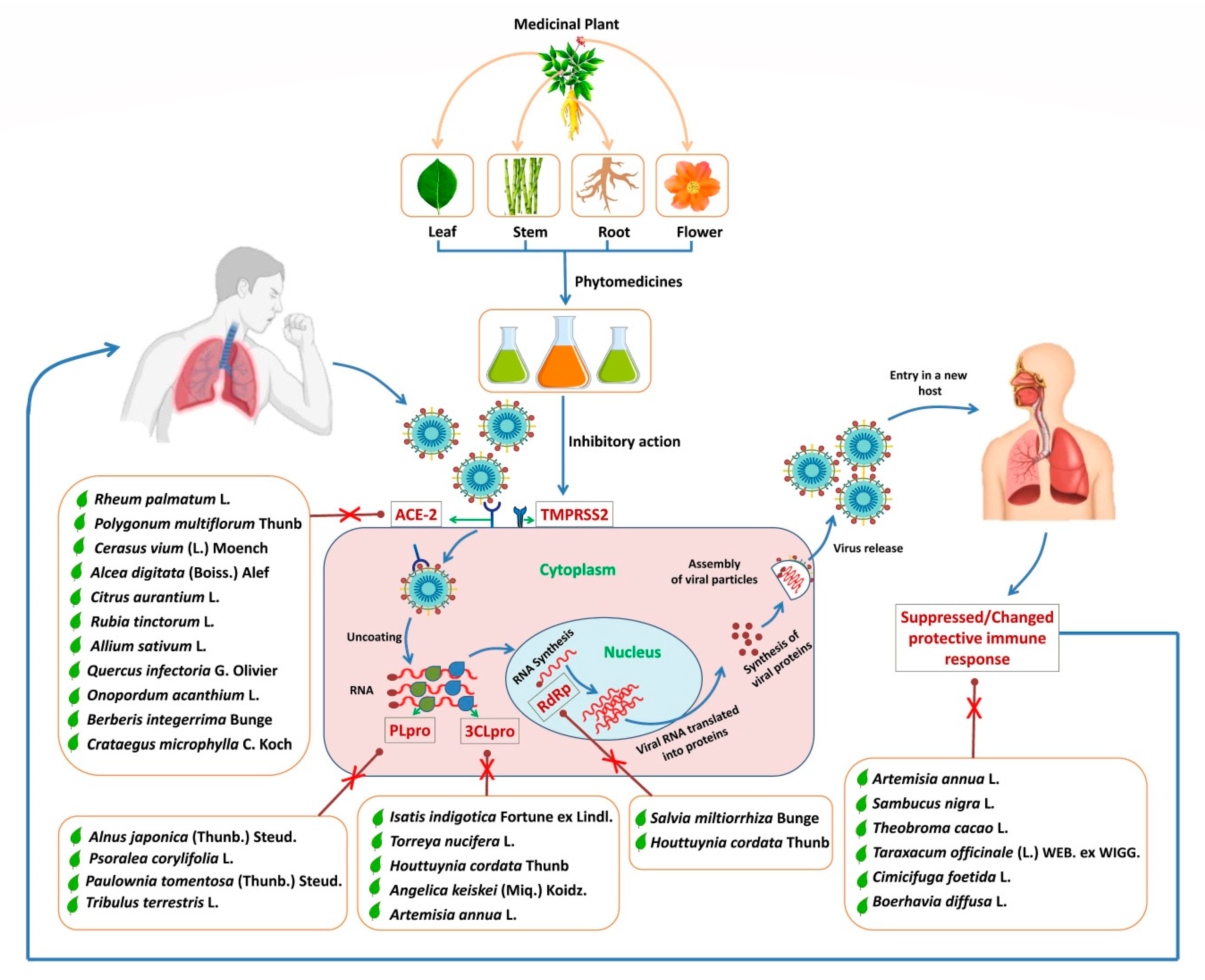
1. Introduction
2. Therapeutic Potential of Medicinal Plants against SARS-CoV and MERS-CoV Infections
2.1. Bupleurum Species
2.2. Lycoris radiate (L’Hér.) Herb.
2.3. Artemisia annua L.
2.4. Pyrrosia lingua (Thunb.) Farw.
2.5. Isatis indigotica Fortune ex Lindl.
2.6. Torreya nucifera L.
2.7. Houttuynia cordata Thunb.
2.8. Lindera aggregate (Sims) Kosterm.
3. Known Medicinal Plants Acting on ACE-2 Receptor
3.1. Rheum palmatum L.
3.2. Polygonum multiflorum Thunb
3.3. Cerasus avium (L.) Moench
3.4. Alcea digitata (Boiss.) Alef
3.5. Citrus aurantium L.
3.6. Rubia tinctorum L.
3.7. Allium sativum L.
3.8. Quercus infectoria G. Olivier
3.9. Onopordum acanthium L.
3.10. Berberis integerrima Bunge
3.11. Crataegus microphylla C. Koch
3.12. Alnus japonica (Thunb.) Steud.
3.13. Psoralea corylifolia L.
3.14. Paulownia tomentosa (Thunb.) Steud.
3.15. Tribulus terrestris L.
4. Other Medicinal Plants in Use against Various Viral Infections and Possibility for the Therapeutic Strategy against COVID-19
4.1. Sambucus nigra L.
4.2. Eleutherococcus senticosus (Rupr. & Maxim.) Maxim.
4.3. Salvia miltiorrhiza Bunge
4.4. Acacia arabica (Lam.) Willd.
4.5. Ocimum sanctum L.
4.6. Ocimum basilicum L.
4.7. Theobroma cacao L.
4.8. Pelargonium sidoides DC.
4.9. Taraxacum officinale (L.) WEB. ex WIGG.
4.10. Illicium oligandrum Merr & Chun
4.11. Glycyrrhiza glabra L.
4.12. Angelica keiskei (Miq.) Koidz.
4.13. Polygala karensium Kurz
4.14. Calophyllum brasiliense Cambess.
4.15. Cimicifuga foetida L.
4.16. Boerhavia diffusa L.
4.17. Terminalia chebula Retz
4.18. Caesalpinia sappan L.
5. Plant-Based Antiviral Drug Discoveries and Future Perspectives
6. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Adhikari, S.P.; Meng, S.; Wu, Y.-J.; Mao, Y.-P.; Ye, R.-X.; Wang, Q.-Z.; Sun, C.; Sylvia, S.; Rozelle, S.; Raat, H.; et al. Epidemiology, causes, clinical manifestation and diagnosis, prevention and control of coronavirus disease (COVID-19) during the early outbreak period: A scoping review. Infect. Dis. Poverty 2020, 9, 29. [Google Scholar] [CrossRef] [PubMed]
- Boopathi, S.; Poma, A.B.; Kolandaivel, P. Novel 2019 coronavirus structure, mechanism of action, antiviral drug promises and rule out against its treatment. J. Biomol. Struct. Dyn. 2020, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Asai, A.; Konno, M.; Ozaki, M.; Otsuka, C.; Vecchione, A.; Arai, T.; Kitagawa, T.; Ofusa, K.; Yabumoto, M.; Hirotsu, T.; et al. COVID-19 drug discovery using intensive approaches. Int. J. Mol. Sci. 2020, 21, 2839. [Google Scholar] [CrossRef] [PubMed]
- Mohammadi, M.; Meskini, M.; Pinto, A.L.D.N. 2019 Novel coronavirus (COVID-19) overview. J. Public Health 2020, 19, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Ye, Z.-W.; Yuan, S.; Yuen, K.-S.; Fung, S.-Y.; Chan, C.-P.; Jin, D.-Y. Zoonotic origins of human coronaviruses. Int. J. Biol. Sci. 2020, 16, 1686–1697. [Google Scholar] [CrossRef] [PubMed]
- Hasöksüz, M.; Kiliç, S.; Saraç, F. Coronaviruses and SARS-COV-2. Turk. J. Med. Sci. 2020, 50, 549–556. [Google Scholar] [CrossRef] [PubMed]
- Hemida, M.G.; Abduallah, M.M.B. The SARS-CoV-2 outbreak from a one health perspective. One Health 2020, 10, 100127. [Google Scholar] [CrossRef]
- WHO. Coronavirus Disease 2019 (COVID-19) Situation Report. 2020. Available online: https://www.who.int/docs/default-source/coronaviruse/situation-reports/20200429-sitrep-100-covid-19.pdf?sfvrsn=bbfbf3d1_2 (accessed on 7 September 2020).
- Siddiqui, A.J.; Jahan, S.; Ashraf, S.A.; Alreshidi, M.; Ashraf, M.S.; Patel, M.; Snoussi, M.; Singh, R.; Adnan, M. Current status and strategic possibilities on potential use of combinational drug therapy against COVID-19 caused by SARS-CoV-2. J. Biomol. Struct. Dyn. 2020, 1–14. [Google Scholar] [CrossRef]
- ECDC. European Centre for Disease Prevention and Control. Available online: https://www.ecdc.europa.eu/en/geographical-distribution-2019-ncov-cases (accessed on 7 September 2020).
- WorldOmeter. Coronavirus WorldOmeter. Available online: https://www.worldometers.info/coronavirus/ (accessed on 7 September 2020).
- Aanouz, I.; Belhassan, A.; El Khatabi, K.; Lakhlifi, T.; El Idrissi, M.; Bouachrine, M. Moroccan medicinal plants as inhibitors against SARS-CoV-2 main protease: Computational investigations. J. Biomol. Struct. Dyn. 2020, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Prasad, A.; Muthamilarasan, M.; Prasad, M. Synergistic antiviral effects against SARS-CoV-2 by plant-based molecules. Plant Cell Rep. 2020, 39, 1109–1114. [Google Scholar] [CrossRef] [PubMed]
- Azad, C.S.; Saxena, M.; Siddiqui, A.J.; Bhardwaj, J.; Puri, S.K.; Dutta, G.P.; Anand, N.; Saxena, A.K.; Anand, N. Synthesis of primaquine glyco-conjugates as potential tissue schizontocidal antimalarial agents. Chem. Biol. Drug Des. 2017, 90, 254–261. [Google Scholar] [CrossRef] [PubMed]
- Bhardwaj, J.; Siddiqui, A.J.; Goyal, M.; Prakash, K.; Soni, A.; Puri, S.K. Repetitive live sporozoites inoculation under arteether chemoprophylaxis confers protection against subsequent sporozoite challenge in rodent malaria model. Acta Trop. 2016, 158, 130–138. [Google Scholar] [CrossRef] [PubMed]
- Prakash, K.; Goyal, M.; Soni, A.; Siddiqui, A.J.; Bhardwaj, J.; Puri, S.K. Molecular cloning and biochemical characterization of iron superoxide dismutase from the rodent malaria parasite Plasmodium vinckei. Parasitol. Int. 2014, 63, 817–825. [Google Scholar] [CrossRef] [PubMed]
- Siddiqui, A.J.; Bhardwaj, J.; Goyal, M.; Prakash, K.; Adnan, M.; Alreshidi, M.M.; Patel, M.; Soni, A.; Redman, W. Immune responses in liver and spleen against Plasmodium yoelii pre-erythrocytic stages in Swiss mice model. J. Adv. Res. 2020, 24, 29–41. [Google Scholar] [CrossRef] [PubMed]
- Siddiqui, A.J.; Bhardwaj, J.; Goyal, M.; Prakash, K.; Soni, A.; Tiwari, V.; Puri, S.K. Assessment of real-time method to detect liver parasite burden under different experimental conditions in mice infected with Plasmodium yoelii sporozoites. Microb. Pathog. 2015, 89, 35–42. [Google Scholar] [CrossRef]
- Siddiqui, A.J.; Bhardwaj, J.; Puri, S.K. mRNA expression of cytokines and its impact on outcomes after infection with lethal and nonlethal Plasmodium vinckei parasites. Parasitol. Res. 2011, 110, 1517–1524. [Google Scholar] [CrossRef]
- Siddiqui, A.J.; Adnan, M.; Jahan, S.; Redman, W.; Saeed, M.; Patel, M. Neurological disorder and psychosocial aspects of cerebral malaria: What is new on its pathogenesis and complications? A minireview. Folia Parasitol. 2020, 67, 67. [Google Scholar] [CrossRef]
- Soni, A.; Goyal, M.; Prakash, K.; Bhardwaj, J.; Siddiqui, A.J.; Puri, S.K. Cloning, expression and functional characterization of heme detoxification protein (HDP) from the rodent malaria parasite Plasmodium vinckei. Gene 2015, 566, 109–119. [Google Scholar] [CrossRef]
- Adnan, M. Bioactive potential of essential oil extracted from the leaves of Eucalyptus globulus (Myrtaceae). J. Pharmacogn. Phytochem. 2019, 8, 213–216. [Google Scholar]
- Patel, M.; Ashraf, M.S.; Siddiqui, A.J.; Ashraf, S.A.; Sachidanandan, M.; Snoussi, M.; Adnan, M.; Hadi, S. Profiling and role of bioactive molecules from puntius sophore (freshwater/brackish fish) skin mucus with its potent antibacterial, antiadhesion, and antibiofilm activities. Biomolecules 2020, 10, 920. [Google Scholar] [CrossRef]
- Reddy, M.N.; Adnan, M.; Alreshidi, M.M.; Saeed, M.; Patel, M. Evaluation of anticancer, antibacterial and antioxidant properties of a medicinally treasured fern tectaria coadunata with its phytoconstituents analysis by HR-LCMS. Anticancer Agents Med. Chem. 2020, 20, 1. [Google Scholar] [CrossRef] [PubMed]
- Adnan, M.; Patel, M.; Deshpande, S.; Alreshidi, M.; Siddiqui, A.J.; Reddy, M.N.; Emira, N.; De Feo, V. Effect of adiantum philippense extract on biofilm formation, adhesion with its antibacterial activities against foodborne pathogens, and characterization of bioactive metabolites: An in vitro-in silico approach. Front. Microbiol. 2020, 11, 1–19. [Google Scholar] [CrossRef] [PubMed]
- Adnan, M.; Patel, M.; Reddy, M.N.; Alshammari, E. Formulation, evaluation and bioactive potential of Xylaria primorskensis terpenoid nanoparticles from its major compound xylaranic acid. Sci. Rep. 2018, 8, 1740. [Google Scholar] [CrossRef] [PubMed]
- Patel, M.; Sachidanandan, M.; Adnan, M. Serine arginine protein kinase 1 (SRPK1): A moonlighting protein with theranostic ability in cancer prevention. Mol. Biol. Rep. 2018, 46, 1487–1497. [Google Scholar] [CrossRef] [PubMed]
- Adnan, M.; Alshammari, E.; Ashraf, S.A.; Patel, K.; Lad, K.; Patel, M. Physiological and molecular characterization of biosurfactant producing endophytic fungi xylaria regalis from the cones of thuja plicata as a potent plant growth promoter with its potential application. BioMed Res. Int. 2018, 2018, 1–11. [Google Scholar] [CrossRef]
- Arunkumar, G.; Mudgal, P.P.; Maity, H.; Dowarha, D.; Devadiga, S.; Nag, S.; Arunkumar, G. Herbal plants and plant preparations as remedial approach for viral diseases. Virusdisease 2015, 26, 225–236. [Google Scholar] [CrossRef]
- Ashraf, S.A.; ElKhalifa, A.E.O.; Siddiqui, A.J.; Patel, M.; AwadElkareem, A.M.; Snoussi, M.; Ashraf, M.S.; Adnan, M.; Hadi, S. Cordycepin for health and wellbeing: A potent bioactive metabolite of an entomopathogenic cordyceps medicinal fungus and its nutraceutical and therapeutic potential. Molecules 2020, 25, 2735. [Google Scholar] [CrossRef]
- Ben-Shabat, S.; Yarmolinsky, L.; Porat, D.; Dahan, A. Antiviral effect of phytochemicals from medicinal plants: Applications and drug delivery strategies. Drug Deliv. Transl. Res. 2019, 10, 354–367. [Google Scholar] [CrossRef]
- Yao, R.-Y.; Zou, Y.-F.; Chen, X.-F. Traditional use, pharmacology, toxicology, and quality control of species in genus Bupleurum L. Chin. Herb. Med. 2013, 5, 245–255. [Google Scholar] [CrossRef]
- Cheng, P.-W.; Ng, L.-T.; Chiang, L.-C.; Lin, C.-C.; Ng, L.-T. Antiviral effects of saikosaponins on human coronavirus 229E in vitro. Clin. Exp. Pharmacol. Physiol. 2006, 33, 612–616. [Google Scholar] [CrossRef]
- Yang, F.; Dong, X.; Yin, X.; Wang, W.; You, L.; Ni, J. Radix bupleuri: A review of traditional uses, botany, phytochemistry, pharmacology, and toxicology. BioMed Res. Int. 2017, 2017, 1–22. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.; Duan, M.; Zhao, Y.; Ling, F.; Xiao, K.; Li, Q.; Li, B.; Lu, C.; Qi, W.; Zeng, Z.; et al. Saikosaponin A inhibits influenza A virus replication and lung immunopathology. Oncotarget 2015, 6, 42541–42556. [Google Scholar] [CrossRef] [PubMed]
- Tykheev, Z.A.; Taraskin, V.V.; Radnaeva, L.D.; Zhang, F.Q.; Chen, S.L. Total saikosaponin content in some species of Bupleurum L. IOP Conf. Ser. Earth Environ. Sci. 2019, 320, 012055. [Google Scholar] [CrossRef]
- Lamoral-Theys, D.; Decaestecker, C.; Mathieu, V.; Dubois, J.; Kornienko, A.; Kiss, R.; Evidente, A.; Pottier, L. Lycorine and its derivatives for anticancer drug design. Mini Rev. Med. Chem. 2010, 10, 41–50. [Google Scholar] [CrossRef] [PubMed]
- Cedrón, J.C.; Gutiérrez, D.G.; Flores, N.; Ravelo, Á.G.; Estévez-Braun, A. Synthesis and antiplasmodial activity of lycorine derivatives. Bioorg. Med. Chem. 2010, 18, 4694–4701. [Google Scholar] [CrossRef]
- Mikami, M.; Kitahara, M.; Kitano, M.; Ariki, Y.; Mimaki, Y.; Sashida, Y.; Yamazaki, M.; Yui, S. Suppressive activity of lycoricidinol (narciclasine) against cytotoxicity of neutrophil-derived calprotectin, and its suppressive effect on rat adjuvant arthritis model. Biol. Pharm. Bull. 1999, 22, 674–678. [Google Scholar] [CrossRef][Green Version]
- Kretzing, S.; Abraham, G.; Seiwert, B.; Ungemach, F.R.; Krügel, U.; Regenthal, R. Dose-dependent emetic effects of the Amaryllidaceous alkaloid lycorine in beagle dogs. Toxicon 2011, 57, 117–124. [Google Scholar] [CrossRef]
- Ieven, M.; Berghe, D.A.V.D.; Vlietinck, A.J. Plant antiviral agents. IV. Influence of lycorine on growth pattern of three animal viruses. Planta Med. 1983, 49, 109–114. [Google Scholar] [CrossRef]
- Liu, J.-N.; Yang, Y.; Xu, Y.; Ma, C.; Qin, C.; Zhang, L. Lycorine reduces mortality of human enterovirus 71-infected mice by inhibiting virus replication. Virol. J. 2011, 8, 483. [Google Scholar] [CrossRef]
- Wang, H.; Guo, T.; Yang, Y.; Yu, L.; Pan, X.; Li, Y.-H. Lycorine derivative LY-55 inhibits EV71 and CVA16 replication through downregulating autophagy. Front. Microbiol. 2019, 9, 277. [Google Scholar] [CrossRef]
- Mukhtar, M.; Arshad, M.; Ahmad, M.; Pomerantz, R.J.; Wigdahl, B.; Parveen, Z. Antiviral potentials of medicinal plants. Virus Res. 2008, 131, 111–120. [Google Scholar] [CrossRef] [PubMed]
- Efferth, T.; Romero, M.R.; Wolf, D.G.; Stamminger, T.; Marin, J.J.G.; Marschall, M. The antiviral activities of artemisinin and artesunate. Clin. Infect. Dis. 2008, 47, 804–811. [Google Scholar] [CrossRef] [PubMed]
- AleSaeidi, S.; Miraj, S. A systematic review of anti-malarial properties, immunosuppressive properties, anti-inflammatory properties, and anti-cancer properties of artemisia annua. Electron. Physician 2016, 8, 3150–3155. [Google Scholar] [CrossRef] [PubMed]
- Ho, W.E.; Peh, H.Y.; Chan, T.K.; Wong, W.F. Artemisinins: Pharmacological actions beyond anti-malarial. Pharmacol. Ther. 2014, 142, 126–139. [Google Scholar] [CrossRef] [PubMed]
- Lin, L.-T.; Hsu, W.-C.; Lin, C.-C. Antiviral natural products and herbal medicines. J. Tradit. Complement. Med. 2014, 4, 24–35. [Google Scholar] [CrossRef]
- Law, S.; Leung, A.W.; Xu, C. Is the traditional Chinese herb “Artemisia annua” possible to fight against COVID-19? Integr. Med. Res. 2020, 9, 100474. [Google Scholar] [CrossRef]
- Li, S.-Y.; Chen, C.; Zhang, H.-Q.; Guo, H.-Y.; Wang, H.; Wang, L.; Zhang, X.; Hua, S.-N.; Yu, J.; Xiao, P.-G.; et al. Identification of natural compounds with antiviral activities against SARS-associated coronavirus. Antivir. Res. 2005, 67, 18–23. [Google Scholar] [CrossRef]
- Benatouil, C.P.; Reanimator, A. Action of Artemisia Annua on Adaptive Immunity in COVID-19 Infections. Available online: https://lavierebelle.org/action-de-l-artemisia-annua-sur-l?lang=en (accessed on 22 June 2020).
- Gao, D.; Fan, Y.; Feng, H.; Liu, L.; Zhang, Y.; Xin, X. Chemical components and antibacterial activity of the essential oil of six pyrrosia species. Chem. Biodivers. 2020, 10, 1–10. [Google Scholar] [CrossRef]
- Zheng, M. Experimental study of 472 herbs with antiviral action against the herpes simplex virus. Chin. J. Mod. Dev. Tradit. Med. 1990, 10, 39–41. [Google Scholar]
- Xiao, W.; Peng, Y.; Tan, Z.; Lv, Q.; Chan, C.-O.; Yang, J.; Chen, S. Comparative evaluation of chemical profiles of pyrrosiae folium originating from three pyrrosia species by HPLC-DAD combined with multivariate statistical analysis. Molecules 2017, 22, 2122. [Google Scholar] [CrossRef]
- Chen, Y.; Fan, C.-L.; Wang, Y.; Zhang, X.-Q.; Huang, X.-J.; Ye, W.-C. Chemical constituents from roots of Isatis indigotica. China J. Chin. Mater. Med. 2018, 43, 2091–2096. [Google Scholar]
- Zhang, D.; Shi, Y.; Xu, R.; Du, K.; Guo, F.; Chen, K.; Li, Y.; Wang, R. Alkaloid enantiomers from the roots of isatis indigotica. Molecules 2019, 24, 3140. [Google Scholar] [CrossRef] [PubMed]
- Lin, C.-W.; Tsai, F.-J.; Tsai, C.-H.; Lai, C.-C.; Wan, L.; Ho, T.-Y.; Hsieh, C.-C.; Chao, P.-D.L. Anti-SARS coronavirus 3C-like protease effects of Isatis indigotica root and plant-derived phenolic compounds. Antivir. Res. 2005, 68, 36–42. [Google Scholar] [CrossRef] [PubMed]
- Chang, S.-J.; Chang, Y.-C.; Lu, K.-Z.; Tsou, Y.-Y.; Lin, C.-W. Antiviral activity of isatis indigotica extract and its derived indirubin against japanese encephalitis virus. Evid. Based Complement. Altern. Med. 2012, 2012, 1–7. [Google Scholar] [CrossRef]
- Oh, J.; Rho, H.S.; Yang, Y.; Yoon, J.Y.; Lee, J.; Hong, Y.D.; Kim, H.C.; Choi, S.S.; Kim, T.W.; Shin, S.S.; et al. Extracellular signal-regulated kinase is a direct target of the anti-inflammatory compound amentoflavone derived from Torreya nucifera. Mediat. Inflamm. 2013, 2013, 1–11. [Google Scholar] [CrossRef]
- Endo, Y.; Osada, Y.; Kimura, F.; Shirakawa, H.; Fujimoto, K. Effects of Japanese Torreya (Torreya nucifera) seed oil on the activities and mRNA expression of lipid metabolism-related enzymes in rats. Biosci. Biotechnol. Biochem. 2007, 71, 231–233. [Google Scholar] [CrossRef]
- Ryu, Y.B.; Jeong, H.J.; Kim, J.H.; Kim, Y.M.; Park, J.-Y.; Kim, D.; Naguyen, T.T.H.; Park, S.-J.; Chang, J.S.; Park, K.H.; et al. Biflavonoids from Torreya nucifera displaying SARS-CoV 3CLpro inhibition. Bioorg. Med. Chem. 2010, 18, 7940–7947. [Google Scholar] [CrossRef]
- Shingnaisui, K.; Dey, T.; Manna, P.; Kalita, J. Therapeutic potentials of Houttuynia cordata Thunb. against inflammation and oxidative stress: A review. J. Ethnopharmacol. 2018, 220, 35–43. [Google Scholar] [CrossRef]
- Lau, K.-M.; Lee, K.-M.; Koon, C.-M.; Cheung, C.S.-F.; Lau, C.-P.; Ho, H.-M.; Lee, M.Y.-H.; Au, S.W.-N.; Cheng, C.H.K.; Lau, C.B.; et al. Immunomodulatory and anti-SARS activities of Houttuynia cordata. J. Ethnopharmacol. 2008, 118, 79–85. [Google Scholar] [CrossRef]
- Chiow, K.; Phoon, M.; Putti, T.; Tan, B.K.; Chow, V.T.K. Evaluation of antiviral activities of Houttuynia cordata Thunb. extract, quercetin, quercetrin and cinanserin on murine coronavirus and dengue virus infection. Asian Pac. J. Trop. Med. 2016, 9, 1–7. [Google Scholar] [CrossRef]
- Cheng, D.; Sun, L.; Zou, S.; Chen, J.; Mao, H.-Y.; Zhang, Y.; Liao, N.; Zhang, R.-H. Antiviral effects of Houttuynia cordata polysaccharide extract on Murine Norovirus-1 (MNV-1)—A human norovirus surrogate. Molecules 2019, 24, 1835. [Google Scholar] [CrossRef] [PubMed]
- Chen, M.-Y.; Li, H.; Lu, X.-X.; Ling, L.-J.; Weng, H.-B.; Sun, W.; Chen, D.-F.; Zhang, Y.-Y. Houttuynia cordata polysaccharide alleviated intestinal injury and modulated intestinal microbiota in H1N1 virus infected mice. Chin. J. Nat. Med. 2019, 17, 187–197. [Google Scholar] [CrossRef]
- Xu, C.; Yang, B.-X.; Zhu, W.; Li, X.; Tian, J.; Zhang, L. Characterisation of polyphenol constituents of Linderae aggregate leaves using HPLC fingerprint analysis and their antioxidant activities. Food Chem. 2015, 186, 83–89. [Google Scholar] [CrossRef]
- Wei, G.; Chen, H.; Nie, F.; Ma, X.; Jiang, H. 1, 3, 6-trihydroxy-7-methyl-9, 10-anthracenedione isolated from genus lindera with anti-cancer activity. Anticancer Agents Med. Chem. 2017, 17, 1604–1607. [Google Scholar] [CrossRef]
- Xiao, M.; Cao, N.; Fan, J.-J.; Shen, Y.; Xu, Q. Studies on flavonoids from the leaves of Lindera aggregata. J. Chin. Med. Mater. 2011, 34, 62–64. [Google Scholar]
- Jung, S.-H.; Han, J.-H.; Park, H.-S.; Lee, J.-J.; Yang, S.Y.; Kim, Y.; Heo, K.-S.; Myung, C.-S. Inhibition of collagen-induced platelet aggregation by the secobutanolide secolincomolide a from lindera obtusiloba blume. Front. Pharmacol. 2017, 8, 560. [Google Scholar] [CrossRef]
- Extension Gardener. Lycoris Radiata. Available online: https://plants.ces.ncsu.edu/plants/lycoris-radiata/ (accessed on 24 August 2020).
- Artennua. Artemisia Annua Characteristics. Available online: https://www.artennua.com/artemisia-annua/ (accessed on 24 August 2020).
- Hardy Fern Foundation. Pyrrosia Lingua Tongue Fern. Available online: https://hardyferns.org/ferns/pyrrosia-lingua/ (accessed on 22 August 2020).
- The Sunlight Experiment. COVID-19: Searching For Potential Treatment Options in Plants. Available online: https://thesunlightexperiment.com/blog/herbal-medicine-covid-19 (accessed on 22 August 2020).
- Torreya Nucifera. Available online: https://alchetron.com/Torreya-nucifera (accessed on 20 August 2020).
- Wikipedia. Houttuynia cordata. Available online: https://en.wikipedia.org/wiki/Houttuynia_cordata (accessed on 20 August 2020).
- Useful Tropical Plants. Lindera Aggregata. Available online: http://tropical.theferns.info/viewtropical.php?id=Lindera+aggregata (accessed on 21 August 2020).
- Ho, T.-Y.; Wu, S.-L.; Chen, J.-C.; Li, C.-C.; Hsiang, C.-Y. Emodin blocks the SARS coronavirus spike protein and angiotensin-converting enzyme 2 interaction. Antivir. Res. 2007, 74, 92–101. [Google Scholar] [CrossRef]
- Lee, J.-C.; Tseng, C.-K.; Wu, S.-F.; Chang, F.-R.; Chiu, C.-C.; Wu, Y.-C. San-Huang-Xie-Xin-Tang extract suppresses hepatitis C virus replication and virus-induced cyclooxygenase-2 expression. J. Viral Hepat. 2011, 18, e315–e324. [Google Scholar] [CrossRef]
- Yang, K.L.; Gao, Y.; Yang, F.W.; Liu, M.; Shi, S.Z.; Chen, Y.M.; Zhang, J.H.; Tian, J.H. Analysis of traditional Chinese medicine from patent information sharing platform of coronavirus disease 2019 (COVID-19). China J. Chin. Mater. Med. 2020, 45, 3001–3006. [Google Scholar] [CrossRef]
- Zhao, M.-J.; Chao, J.; Dai, Y.-T.; Chen, S.-L.; Li, Q.; Fan, Z.-Q.; Wang, D.-D. Quality evaluation of Rhei Radix et Rhizoma decoction. China J. Chin. Mater. Med. 2018, 43, 861–867. [Google Scholar]
- Zheng, L.; Chen, S.; Cao, Y.; Zhao, L.; Gao, Y.; Ding, X.; Wang, X.; Gu, Y.; Wang, S.; Zhu, Z.; et al. Combination of comprehensive two-dimensional prostate cancer cell membrane chromatographic system and network pharmacology for characterizing membrane binding active components from Radix et Rhizoma Rhei and their targets. J. Chromatogr. A 2018, 1564, 145–154. [Google Scholar] [CrossRef] [PubMed]
- Wikipedia. Rheum Palmatum. Available online: https://en.wikipedia.org/wiki/Rheum_palmatum (accessed on 21 August 2020).
- Cheng, W.; Li, Y.; Yang, W.; Wu, S.; Wei, M.; Gao, Y.; Kang, C.; Zhang, S.; Li, Y. Simultaneous determination of 13 constituents of radix polygoni multiflori in rat plasma and its application in a pharmacokinetic study. Int. J. Anal. Chem. 2020, 2020, 4508374. [Google Scholar] [CrossRef]
- He, Q.; Tu, C.; Wang, J.-B.; Liu, Z.-J.; Sha, M.-C.; Zhang, L.; Li, C.-Y.; Xiao, X.-H. Antiplatelet aggregation bioactivity of Polygoni Multiflori Radix with chemical fingerprints and spectrum-effect correlation analysis. China J. Chin. Mater. Med. 2017, 42, 1679–1684. [Google Scholar]
- Lee, B.-J.; Lee, K. Discrimination and proper use of polygoni multiflori radix, cynanchi wilfordii radix, and cynanchi auriculati radix in Korea: A descriptive review. Evid. Based Complement. Altern. Med. 2015, 2015, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Liang, Z.T.; Chen, H.; Yu, Z.-L.; Zhao, Z.-Z. Comparison of raw and processed Radix Polygoni Multiflori (Heshouwu) by high performance liquid chromatography and mass spectrometry. Chin. Med. 2010, 5, 29. [Google Scholar] [CrossRef] [PubMed]
- Lin, L.; Ni, B.; Lin, H.; Zhang, M.; Yan, L.; Qu, C.; Ni, J. Simultaneous determination of 14 constituents of Radix polygoni multiflori from different geographical areas by liquid chromatography-tandem mass spectrometry. Biomed. Chromatogr. 2014, 29, 1048–1055. [Google Scholar] [CrossRef]
- Plants For A Future. Polygonum Multiflorum—Thunb. Available online: https://pfaf.org/user/Plant.aspx?LatinName=Polygonum+multiflorum (accessed on 20 August 2020).
- Console, L.; Giangregorio, N.; Cellamare, S.; Bolognino, I.; Palasciano, M.; Indiveri, C.; Incampo, G.; Campana, S.; Tonazzi, A.; Lara, C.; et al. Human mitochondrial carnitine acylcarnitine carrier: Molecular target of dietary bioactive polyphenols from sweet cherry (Prunus avium L.). Chem. Interact. 2019, 307, 179–185. [Google Scholar] [CrossRef]
- Heidary, F.; Varnaseri, M.; Gharebaghi, R. The potential use of persian herbal medicines against COVID-19 through angiotensin-converting enzyme. Arch. Clin. Infect. Dis. 2020, 15, e102838. [Google Scholar] [CrossRef]
- Kouchmeshky, A.; Jameie, S.B.; Amin, G.; Ziai, S.A. Investigation of angiotensin-convertings enzyme inhibitory effects of medicinal plants used in traditional persian medicine for treatment of hypertension: Screening study. Thrita Stud. J. Med. Sci. 2012, 1, 13–23. [Google Scholar] [CrossRef]
- Serteser, A.; Kargioglu, M.; Gök, V.; Bağci, Y.; Özcan, M.M.; Arslan, D. Determination of antioxidant effects of some plant species wild growing in Turkey. Int. J. Food Sci. Nutr. 2008, 59, 643–651. [Google Scholar] [CrossRef]
- Ziai, S.A.; Heidari, M.R.; Amin, G.H.; Koochemeshki, A.; Heidari, M. Inhibitory effects of germinal angiotensin converting enzyme by medicinal plants used in iranian traditional medicine as antihypertensive. J. Kerman Univ. Med Sci. 2009, 16, 134. [Google Scholar]
- Drug Information of Cerasus Avium. Cerasus Avium. Available online: https://www.rpsi.ir/en/medicinalherb/item/1285/cerasus_avium (accessed on 22 August 2020).
- Ameri, A.; Heydarirad, G.; Rezaeizadeh, H.; Choopani, R.; Ghobadi, A.; Gachkar, L. Evaluation of efficacy of an herbal compound on dry mouth in patients with head and neck cancers. J. Evid. Based Integr. Med. 2015, 21, 30–33. [Google Scholar] [CrossRef]
- Nasser, R.; Jafari, F.; Rezaeizadeh, H.; Nasseri, M.; Kamalinejad, M.; Ghobadi, A.; Shamsipour, M.; Zargaran, A.; Ameri, A. Efficacy of a persian medicine herbal compound (alcea digitataalefandmalva sylvestrisl.) on prevention of radiation induced acutemucositis in patients with head and neck cancer: A pilot study. Int. J. Cancer Manag. 2017, 10, e8642. [Google Scholar] [CrossRef]
- Kew Science. Alcea Digitata. Available online: http://powo.science.kew.org/taxon/urn:lsid:ipni.org:names:558657-1 (accessed on 20 August 2020).
- He, W.; Li, Y.; Liu, M.; Yu, H.; Chen, Q.; Chen, Y.; Ruan, J.; Ding, Z.; Zhang, Y.; Wang, T. Citrus aurantium L. and its flavonoids regulate TNBS-induced inflammatory bowel disease through anti-inflammation and suppressing isolated jejunum contraction. Int. J. Mol. Sci. 2018, 19, 3057. [Google Scholar] [CrossRef] [PubMed]
- Süntar, I.; Khan, H.; Patel, S.; Celano, R.; Rastrelli, L. An overview on Citrus aurantium L.: Its functions as food ingredient and therapeutic agent. Oxidative Med. Cell. Longev. 2018, 2018, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Zhao, H.-Y.; Yang, L.; Wei, J.; Huang, M.; Jiang, J.-G. Bioactivity evaluations of ingredients extracted from the flowers of Citrus aurantium L. var. amara Engl. Food Chem. 2012, 135, 2175–2181. [Google Scholar] [CrossRef] [PubMed]
- Kew Science. Citrus × Aurantium L. Available online: http://www.plantsoftheworldonline.org/taxon/urn:lsid:ipni.org:names:59600-2 (accessed on 20 August 2020).
- Lajkó, E.; Bányai, P.; Zámbó, Z.; Kursinszki, L.; Szőke, É.; Kohidai, L. Targeted tumor therapy by Rubia tinctorum L.: Analytical characterization of hydroxyanthraquinones and investigation of their selective cytotoxic, adhesion and migration modulator effects on melanoma cell lines (A2058 and HT168-M1). Cancer Cell Int. 2015, 15, 1–15. [Google Scholar] [CrossRef]
- Nejad, H.E. Ahmad esalat nejad. Rubia tinctorum L. (Rubiaceae) or madder as one of the living color to dyeing wool. Int. J. Adv. Biol. Biomed. Res. 2013, 1, 1315–1319. [Google Scholar]
- Kew Science. Rubia tinctorum L. Available online: http://www.plantsoftheworldonline.org/taxon/urn:lsid:ipni.org:names:765369-1 (accessed on 20 August 2020).
- Baek, S.C.; Nam, K.H.; Yi, S.; Jo, M.S.; Lee, K.H.; Lee, Y.H.; Lee, J.; Kim, K.H. Anti-adipogenic effect of β-carboline alkaloids from garlic (Allium sativum). Foods 2019, 8, 673. [Google Scholar] [CrossRef]
- Burian, J.P.; Carlos, I.Z.; Sacramento, L.V.S. Fungal infection control by garlic extracts (Allium sativum L.) and modulation of peritoneal macrophages activity in murine model of sporotrichosis. Braz. J. Biol. 2017, 77, 848–855. [Google Scholar] [CrossRef][Green Version]
- Batiha, G.E.-S.; Beshbishy, A.M.; Wasef, L.; Elewa, Y.H.A.; Abdel-Daim, M.; El-Hack, M.; Taha, A.E.; Abd-Elhakim, Y.M.; Devkota, H.P. Chemical constituents and pharmacological activities of garlic (Allium sativum L.): A review. Nutrients 2020, 12, 872. [Google Scholar] [CrossRef] [PubMed]
- Phan, A.D.; Netzel, G.; Chhim, P.; Netzel, M.E.; Sultanbawa, Y. Phytochemical characteristics and antimicrobial activity of australian grown garlic (Allium sativum L.) cultivars. Foods 2019, 8, 358. [Google Scholar] [CrossRef] [PubMed]
- Biomedical Education. Allium sativum L. Available online: http://iprsindh.com.pk/allium-sativum-l/ (accessed on 23 August 2020).
- Chokpaisarn, J.; Urao, N.; Voravuthikunchai, S.P.; Koh, T.J. Quercus infectoria inhibits Set7/NF-κB inflammatory pathway in macrophages exposed to a diabetic environment. Cytokine 2017, 94, 29–36. [Google Scholar] [CrossRef] [PubMed]
- Chusri, S.; Phatthalung, P.N.; Voravuthikunchai, S. Anti-biofilm activity of Quercus infectoria G. Olivier against methicillin-resistant Staphylococcus aureus. Lett. Appl. Microbiol. 2012, 54, 511–517. [Google Scholar] [CrossRef] [PubMed]
- Sharifi, N.; Souri, E.; Ziai, S.A.; Amin, G.; Amanlou, M. Discovery of new angiotensin converting enzyme (ACE) inhibitors from medicinal plants to treat hypertension using an in vitro assay. Daru 2013, 21, 74. [Google Scholar] [CrossRef] [PubMed]
- Shrestha, S.; Kaushik, V.S.; Eshwarappa, R.S.B.; Subaramaihha, S.R.; Ramanna, L.M.; Lakkappa, D.B.; Prasad, S.B.B. Pharmacognostic studies of insect gall of Quercus infectoria Olivier (Fagaceae). Asian Pac. J. Trop. Biomed. 2014, 4, 35–39. [Google Scholar] [CrossRef]
- Natural Medicine Facts. Top Plants Containing Gallic-Acid. Available online: https://www.naturalmedicinefacts.info/plant/quercus-infectoria.html (accessed on 23 August 2020).
- Abusamra, Y.A.-K.; Scuruchi, M.; Habibatni, S.; Maammeri, Z.; Benayache, S.; D’Ascola, A.; Avenoso, A.; Campo, G.M.; Spina, E. Evaluation of putative cytotoxic activity of crude extracts from Onopordum acanthium leaves and Spartium junceum flowers against the U-373 glioblastoma cell line. Pak. J. Pharm. Sci. 2015, 28, 1225–1232. [Google Scholar]
- Csupor-Löffler, B.; Zupkó, I.; Molnár, J.; Forgo, P.; Hohmann, J. Bioactivity-guided isolation of antiproliferative compounds from the roots of Onopordum acanthium. Nat. Prod. Commun. 2014, 9, 337–340. [Google Scholar] [CrossRef]
- Robertovna, G.E.; Alexeevich, K.D.; Alexeevich, S.A.; Petrovna, G.M.; Kenzhebaevna, O.K.; Garsiya, E.R.; Konovalov, D.A.; Шамилoв, A.A.; Glushko, M.P.; Орынбасарoва, K.K. A traditional medicine plant, Onopordum acanthium L. (asteraceae): Chemical composition and pharmacological research. Plants 2019, 8, 40. [Google Scholar] [CrossRef]
- Natural Medicine Facts. Available online: https://www.wikiwand.com/en/Onopordum_acanthium (accessed on 21 August 2020).
- Kooch, Y.; Noghre, N. The effect of shrubland and grassland vegetation types on soil fauna and flora activities in a mountainous semi-arid landscape of Iran. Sci. Total. Environ. 2020, 703, 135497. [Google Scholar] [CrossRef]
- Rafiee, F.; Nejati, V.; Heidari, R.; Ashraf, H. Department of biology, faculty of science, urmia university, urmia, iran protective effect of methanolic extract of berberis integerrima bunge. root on carbon tetrachloride-induced testicular injury in wistar rats. Int. J. Reprod. Biomed. 2016, 14, 133–140. [Google Scholar] [CrossRef] [PubMed]
- Sabahi, Z.; Farmani, F.; Soltani, F.; Moein, M. DNA protection, antioxidant and xanthine oxidase inhibition activities of polyphenol-enriched fraction of Berberis integerrima Bunge fruits. Iran. J. Basic Med. Sci 2018, 21, 411–416. [Google Scholar] [PubMed]
- Newsha. Science—Black Barberry. Available online: https://www.newshadrinks.com/about-herbs/details/black-barberry (accessed on 20 August 2020).
- Hosseinimehr, S.J.; Mahmoudzadeh, A.; Azadbakht, M.; Akhlaghpoor, S.; Azadbakht, M. Radioprotective effects of hawthorn against genotoxicity induced by gamma irradiation in human blood lymphocytes. Radiat. Environ. Biophys. 2008, 48, 95–98. [Google Scholar] [CrossRef] [PubMed]
- Melikoğlu, G.; Bitis, L.; Meriçli, A.H. Flavonoids of crataegus microphylla. Nat. Prod. Res. 2004, 18, 211–213. [Google Scholar] [CrossRef]
- Renda, G.; Ozel, A.; Barut, B.; Korkmaz, B.; Yaylı, N. The in vitro protection by Crataegus microphylla extracts against oxidative damage and enzyme inhibition effects. Turk. J. Pharm. Sci. 2018, 15, 77–84. [Google Scholar] [CrossRef]
- Wikipedia. Crataegus Microphylla. Available online: https://en.wikipedia.org/wiki/Crataegus_microphylla (accessed on 21 August 2020).
- Tung, N.H.; Kwon, H.-J.; Kim, J.-H.; Ra, J.C.; Kim, J.A.; Kim, Y. An anti-influenza component of the bark of Alnus japonica. Arch. Pharmacal Res. 2010, 33, 363–367. [Google Scholar] [CrossRef]
- Wikipedia. Alnus japonica. Available online: https://en.wikipedia.org/wiki/Alnus_japonica (accessed on 22 August 2020).
- Park, J.-Y.; Jeong, H.J.; Kim, J.H.; Kim, Y.M.; Park, S.-J.; Kim, M.; Park, K.H.; Lee, W.S.; Ryu, Y.B. Diarylheptanoids from Alnus japonica inhibit papain-like protease of severe acute respiratory syndrome coronavirus. Biol. Pharm. Bull. 2012, 35, 2036–2042. [Google Scholar] [CrossRef]
- Wikipedia. Psoralea Corylifolia. Available online: https://en.wikipedia.org/wiki/Psoralea_corylifolia (accessed on 22 August 2020).
- Alam, F.; Khan, G.N.; Bin Asad, M.H.H. Psoralea corylifolia L: Ethnobotanical, biological, and chemical aspects: A review. Phytother. Res. 2017, 32, 597–615. [Google Scholar] [CrossRef]
- Chopra, B.; Dhingra, A.K.; Dhar, K.L. Psoralea corylifolia L. (Buguchi)—Folklore to modern evidence: Review. Fitoterapia 2013, 90, 44–56. [Google Scholar] [CrossRef]
- Kim, D.W.; Seo, K.H.; Curtis-Long, M.J.; Oh, K.Y.; Oh, J.-W.; Cho, J.K.; Lee, K.H.; Park, K.H. Phenolic phytochemical displaying SARS-CoV papain-like protease inhibition from the seeds of Psoralea corylifolia. J. Enzym. Inhib. Med. Chem. 2013, 29, 59–63. [Google Scholar] [CrossRef]
- Wikipedia. Paulownia tomentosa. Available online: https://en.wikipedia.org/wiki/Paulownia_tomentosa (accessed on 21 August 2020).
- Ali, S.A.; Ibrahim, N.A.; Mohammed, M.M.; El-Hawary, S.; Refaat, E.A. The potential chemo preventive effect of ursolic acid isolated from Paulownia tomentosa, against N-diethylnitrosamine: Initiated and promoted hepatocarcinogenesis. Heliyon 2019, 5, 01769. [Google Scholar] [CrossRef] [PubMed]
- Cho, J.K.; Curtis-Long, M.J.; Lee, K.H.; Kim, D.W.; Ryu, H.W.; Yuk, H.J.; Park, K.H. Geranylated flavonoids displaying SARS-CoV papain-like protease inhibition from the fruits of Paulownia tomentosa. Bioorg. Med. Chem. 2013, 21, 3051–3057. [Google Scholar] [CrossRef] [PubMed]
- Schneiderová, K.; Smejkal, K. Phytochemical profile of Paulownia tomentosa (Thunb). steud. Phytochem. Rev. 2014, 14, 799–833. [Google Scholar] [CrossRef] [PubMed]
- Zima, A.; Hošek, J.; Treml, J.; Muselík, J.; Suchy, P.; Pražanová, G.; Lopes, A.; Žemlička, M. Antiradical and cytoprotective activities of several C-geranyl-substituted flavanones from Paulownia tomentosa fruit. Molecules 2010, 15, 6035–6049. [Google Scholar] [CrossRef] [PubMed]
- Wikipedia. Tribulus Terrestris. Available online: https://en.wikipedia.org/wiki/Tribulus_terrestris (accessed on 22 August 2020).
- Reshma, P.L.; Lekshmi, V.S.; Sankar, V.; Raghu, K.G. Tribulus terrestris (Linn.) attenuates cellular alterations induced by ischemia in H9c2 cells via antioxidant potential. Phytother. Res. 2015, 29, 933–943. [Google Scholar] [CrossRef] [PubMed]
- Song, Y.H.; Kim, D.W.; Curtis-Long, M.J.; Yuk, H.J.; Wang, Y.; Zhuang, N.; Lee, K.H.; Jeon, K.S.; Park, K.H. Papain-Like Protease (PLpro) inhibitory effects of cinnamic amides from Tribulus terrestris fruits. Biol. Pharm. Bull. 2014, 37, 1021–1028. [Google Scholar] [CrossRef] [PubMed]
- Stefanescu, R.; Tero-Vescan, A.; Negroiu, A.; Aurică, E.; Vari, C. A comprehensive review of the phytochemical, pharmacological, and toxicological properties of Tribulus terrestris L. Biomolecules 2020, 10, 752. [Google Scholar] [CrossRef]
- Kil, Y.-S.; Pham, S.T.; Seo, E.K.; Jafari, M. Angelica keiskei, an emerging medicinal herb with various bioactive constituents and biological activities. Arch. Pharm. Res. 2017, 40, 655–675. [Google Scholar] [CrossRef]
- Kweon, M.; Lee, H.; Park, C.; Choi, Y.H.; Ryu, J.-H. A chalcone from Ashitaba (Angelica keiskei) stimulates myoblast differentiation and inhibits dexamethasone-induced muscle atrophy. Nutrients 2019, 11, 2419. [Google Scholar] [CrossRef]
- Park, J.-Y.; Ko, J.-A.; Kim, D.W.; Kim, Y.M.; Kwon, H.-J.; Jeong, H.J.; Kim, C.Y.; Park, K.H.; Lee, W.-S.; Ryu, Y.B. Chalcones isolated from Angelica keiskeiinhibit cysteine proteases of SARS-CoV. J. Enzym. Inhib. Med. Chem. 2015, 31, 23–30. [Google Scholar] [CrossRef]
- Zhang, T.; Wang, Q.; Fredimoses, M.; Gao, G.; Wang, K.; Chen, H.; Wang, T.; Oi, N.; Zykova, T.A.; Reddy, K.; et al. The Ashitaba (Angelica keiskei) chalcones 4-hydroxyderricin and xanthoangelol suppress melanomagenesis by targeting BRAF and PI3-K. Cancer Prev. Res. 2018, 11, 607–620. [Google Scholar] [CrossRef] [PubMed]
- Wikipedia. Ashitaba. Available online: https://en.wikipedia.org/wiki/Ashitaba (accessed on 18 August 2020).
- Akram, M.; Tahir, I.M.; Shah, S.M.A.; Mahmood, Z.; Altaf, A.; Ahmad, K.; Munir, N.; Daniyal, M.; Nasir, S.; Mehboob, H. Antiviral potential of medicinal plants against HIV, HSV, influenza, hepatitis, and coxsackievirus: A systematic review. Phytother. Res. 2018, 32, 811–822. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.; Zuckerman, D.M.; Brantley, S.E.; Sharpe, M.; Childress, K.O.; Hoiczyk, E.; Pendleton, A.R. Sambucus nigra extracts inhibit infectious bronchitis virus at an early point during replication. BMC Vet. Res. 2014, 10, 24. [Google Scholar] [CrossRef] [PubMed]
- Della, V.A.; Ricci, G.; Ralli, M.; Gambacorta, V.; De Lucia, A.; Minni, A.; Pirozzi, C.; Paccone, M.; Pastore, V.; Di Stadio, A. The effects of oral supplements with Sambucus nigra, Zinc, Tyndallized Lactobacillus acidophilus (HA122), Arabinogalactans, vitamin D, vitamin E and vitamin C in otitis media with effusion in children: A randomized controlled trial. Eur. Rev. Med. Pharmacol. Sci. 2019, 23, 6360–6370. [Google Scholar]
- Młynarczyk, K.; Walkowiak-Tomczak, D.; Łysiak, G. Bioactive properties of Sambucus nigra L. as a functional ingredient for food and pharmaceutical industry. J. Funct. Foods 2018, 40, 377–390. [Google Scholar] [CrossRef]
- Porter, R.S.; Bode, R.F. A review of the antiviral properties of black elder (Sambucus nigra L.) products. Phytother. Res. 2017, 31, 533–554. [Google Scholar] [CrossRef]
- Ulbricht, C.; Basch, E.; Cheung, L.; Goldberg, H.; Hammerness, P.; Isaac, R.; Khalsa, K.P.S.; Romm, A.; Rychlik, I.; Varghese, M.; et al. An evidence-based systematic review of elderberry and elderflower (Sambucus nigra) by the natural standard research collaboration. J. Diet. Suppl. 2014, 11, 80–120. [Google Scholar] [CrossRef]
- Zakay-Rones, Z.; Varsano, N.; Zlotnik, M.; Manor, O.; Regev, L.; Schlesinger, M.; Mumcuoglu, M. Inhibition of several strains of influenza virus in vitro and reduction of symptoms by an elderberry extract (Sambucus nigra L.) during an outbreak of influenza B panama. J. Altern. Complement. Med. 1995, 1, 361–369. [Google Scholar] [CrossRef]
- Kew Science. Sambucus nigra L. Available online: http://www.plantsoftheworldonline.org/taxon/urn:lsid:ipni.org:names:30122169-2 (accessed on 19 August 2020).
- Lee, S.; Shin, N.-S.; Oh, K.-B.; Shin, K.H. Antibacterial compounds from the leaves of Acanthopanax senticosus. Arch. Pharm. Res. 2003, 26, 40–42. [Google Scholar] [CrossRef]
- Wang, Z.; Jiang, H.; Xia, Y.-G.; Yang, B.-Y.; Kuang, H.-X. α-glucosidase inhibitory constituents from acanthopanax senticosus harm leaves. Molecules 2012, 17, 6269–6276. [Google Scholar] [CrossRef]
- Yamauchi, Y.; Ge, Y.-W.; Yoshimatsu, K.; Komatsu, K.; Kuboyama, T.; Yang, X.; Tohda, C.; Komastu, K. Memory enhancement by oral administration of extract of Eleutherococcus senticosus leaves and active compounds transferred in the brain. Nutrients 2019, 11, 1142. [Google Scholar] [CrossRef] [PubMed]
- Wikipedia. Eleutherococcus senticosus. Available online: https://en.wikipedia.org/wiki/Eleutherococcus_senticosus (accessed on 20 August 2020).
- Shao, F.; Lu, S. Identification, molecular cloning and expression analysis of five RNA-dependent RNA polymerase genes in salvia miltiorrhiza. PLoS ONE 2014, 9, e95117. [Google Scholar] [CrossRef] [PubMed]
- Wu, B.-W.; Pan, T.-L.; Leu, Y.; Chang, Y.-K.; Tai, P.-J.; Lin, K.-H.; Horng, J.-T. Antiviral effects of Salvia miltiorrhiza (danshen) against enterovirus. Am. J. Chin. Med. 2007, 35, 153–168. [Google Scholar] [CrossRef] [PubMed]
- Yin, Z.-K.; Feng, Z.-M.; Jiang, J.-S.; Zhang, X.; Zhang, P.-C.; Yang, Y.-N. Two new tanshinone derivatives from the rhizomes of Salvia miltiorrhiza and their antiviral activities. J. Asian Nat. Prod. Res. 2019, 22, 24–29. [Google Scholar] [CrossRef] [PubMed]
- Zhang, D.; Guo, J.; Zhang, M.; Liu, X.; Ba, M.; Tao, X.; Yu, L.; Guo, Y.; Dai, J.-G. Oxazole-containing diterpenoids from cell cultures of Salvia miltiorrhiza and their anti-HIV-1 activities. J. Nat. Prod. 2017, 80, 3241–3246. [Google Scholar] [CrossRef]
- Salvia miltiorrhiza Flower. Available online: https://en.wikipedia.org/wiki/Salvia_miltiorrhiza#/media/File:Salviamiltiorrhiza.jpg (accessed on 21 August 2020).
- El Gendy, A.E.-N.G.; Al-Mahdy, D.A.M.; El Dine, R.S.; Fahmy, S.; Yassin, A.; Porzel, A.; Brandt, W. Structure activity relationships of antimicrobial gallic acid derivatives from pomegranate and acacia fruit extracts against potato bacterial wilt pathogen. Chem. Biodivers. 2015, 12, 955–962. [Google Scholar] [CrossRef]
- Ghoke, S.S.; Sood, R.; Kumar, N.; Pateriya, A.K.; Bhatia, S.; Mishra, A.; Dixit, R.; Singh, V.K.; Desai, D.; Kulkarni, D.D.; et al. Evaluation of antiviral activity of Ocimum sanctum and Acacia arabica leaves extracts against H9N2 virus using embryonated chicken egg model. BMC Complement. Altern. Med. 2018, 18, 174. [Google Scholar] [CrossRef] [PubMed]
- Hegazy, G.A.; Alnoury, A.M.; Gad, H.G. The role of Acacia Arabica extract as an antidiabetic, antihyperlipidemic, and antioxidant in streptozotocin-induced diabetic rats. Saudi Med. J. 2013, 34, 727–733. [Google Scholar]
- Nutan, N.; Modi, M.; Dezzutti, C.S.; Kulshreshtha, S.; Rawat, A.K.S.; Srivastava, S.K.; Malhotra, S.; Verma, A.; Ranga, U.; Gupta, S.K. Extracts from Acacia catechu suppress HIV-1 replication by inhibiting the activities of the viral protease and Tat. Virol. J. 2013, 10, 309. [Google Scholar] [CrossRef]
- Acacia. Available online: http://www.epharmacognosy.com/2012/10/acacia-acacia-arabica-lam-willd.html (accessed on 21 August 2020).
- Ahirwar, P.; Shashikiran, N.D.; Sundarraj, R.K.; Singhla, S.; Thakur, R.A.; Maran, S. A clinical trial comparing antimicrobial efficacy of “essential oil of Ocimum sanctum” with triple antibiotic paste as an intracanal medicament in primary molars. J. Indian Soc. Pedod. Prev. Dent. 2018, 36, 191–197. [Google Scholar] [CrossRef]
- Baliga, M.S.; Jimmy, R.; Thilakchand, K.R.; Sunitha, V.; Bhat, N.R.; Saldanha, E.; Rao, S.; Rao, P.; Arora, R.B.; Palatty, P.L. Ocimum sanctum L (holy basil or tulsi) and its phytochemicals in the prevention and treatment of cancer. Nutr. Cancer 2013, 65, 26–35. [Google Scholar] [CrossRef] [PubMed]
- Cohen, M.; Cohen, M. Tulsi—Ocimum sanctum: A herb for all reasons. J. Ayurveda Integr. Med. 2014, 5, 251–259. [Google Scholar] [CrossRef] [PubMed]
- Kamyab, A.A.; Eshraghian, A. Anti-inflammatory, gastrointestinal and hepatoprotective effects of Ocimum sanctum Linn: An ancient remedy with new application. Inflamm. Allergy Drug Targets 2013, 12, 378–384. [Google Scholar] [CrossRef] [PubMed]
- Mondal, S.; Mirdha, B.R.; Mahapatra, S.C. The science behind sacredness of Tulsi (Ocimum sanctum Linn.). Indian J. Physiol. Pharmacol. 2010, 53, 291–306. [Google Scholar]
- Pattanayak, P.; Behera, P.; Das, D.; Panda, S.K. Ocimum sanctum Linn. A reservoir plant for therapeutic applications: An overview. Pharmacogn. Rev. 2010, 4, 95–105. [Google Scholar] [CrossRef]
- Penmetsa, G.S.; Pitta, S.R. Efficacy of Ocimum sanctum, Aloe vera and chlorhexidine mouthwash on gingivitis: A randomized controlled comparative clinical study. An. Int. Q. J. Res. Ayurveda 2019, 40, 23–26. [Google Scholar] [CrossRef]
- Prakash, P.; Gupta, N. Therapeutic uses of Ocimum sanctum Linn (Tulsi) with a note on eugenol and its pharmacological actions: A short review. Indian J. Physiol. Pharmacol. 2005, 49, 125–131. [Google Scholar]
- The National Innovation Foundation (NIF)—India. Uses of Ocimum Sanctum L. Available online: http://nif.org.in/OCIMUM-SANCTUM-L (accessed on 23 August 2020).
- Alegría-Herrera, E.; Herrera-Ruiz, M.; Román-Ramos, R.; Zamilpa, A.; Santillán-Urquiza, M.A.; Aguilar, M.I.; Avilés-Flores, M.; Fuentes-Mata, M.; Jiménez-Ferrer, E. Effect of Ocimum basilicum, Ocimum selloi, and rosmarinic acid on cerebral vascular damage in a chronic hypertension model. Biol. Pharm. Bull. 2019, 42, 201–211. [Google Scholar] [CrossRef]
- Ayuob, N.; El Wahab, M.G.A.; Ali, S.S.; Abdel-Tawab, H.S. Ocimum basilicum improve chronic stress-induced neurodegenerative changes in mice hippocampus. Metab. Brain Dis. 2018, 33, 795–804. [Google Scholar] [CrossRef]
- Rashidian, A.; Roohi, P.; Mehrzadi, S.; Ghannadi, A.R.; Minaiyan, M. Protective effect of Ocimum basilicum essential oil against acetic acid–induced colitis in rats. J. Evid. Based Integr. Med. 2016, 21, NP36–NP42. [Google Scholar] [CrossRef]
- Sestili, P.; Ismail, T.; Calcabrini, C.; Guescini, M.; Catanzaro, E.; Turrini, E.; Layla, A.; Akhtar, S.; Fimognari, C. The potential effects of Ocimum basilicum on health: A review of pharmacological and toxicological studies. Expert Opin. Drug Metab. Toxicol. 2018, 14, 679–692. [Google Scholar] [CrossRef] [PubMed]
- Singh, P.; Chakraborty, P.; He, D.-H.; Mergia, A. Extract prepared from the leaves of Ocimum basilicum inhibits the entry of Zika virus. Acta Virol. 2019, 63, 316–321. [Google Scholar] [CrossRef] [PubMed]
- Monaco Nature Encyclopedia. Ocimum basilicum. Available online: https://www.monaconatureencyclopedia.com/ocimum-basilicum/?lang=en (accessed on 23 August 2020).
- Goya, L.; Martin, A.E.; Sarriá, B.; Ramos, S.; Mateos, R.; Bravo-Clemente, L. Effect of cocoa and its flavonoids on biomarkers of inflammation: Studies of cell culture, animals and humans. Nutrients 2016, 8, 212. [Google Scholar] [CrossRef] [PubMed]
- Kamei, M.; Nishimura, H.; Takahashi, T.; Takahashi, N.; Inokuchi, K.; Mato, T.; Takahashi, K. Anti-influenza virus effects of cocoa. J. Sci. Food Agric. 2015, 96, 1150–1158. [Google Scholar] [CrossRef]
- Latif, R. Chocolate/cocoa and human health: A review. Neth. J. Med. 2013, 71, 63–68. [Google Scholar]
- Oyeleke, S.A.; Ajayi, A.M.; Umukoro, S.; Aderibigbe, A.; Ademowo, O.G. Anti-inflammatory activity of Theobroma cacao L. stem bark ethanol extract and its fractions in experimental models. J. Ethnopharmacol. 2018, 222, 239–248. [Google Scholar] [CrossRef]
- Wikipedia. Theobroma Cacao. Available online: https://en.wikipedia.org/wiki/Theobroma_cacao (accessed on 22 August 2020).
- Careddu, D.; Pettenazzo, A. Pelargonium sidoides extract EPs 7630: A review of its clinical efficacy and safety for treating acute respiratory tract infections in children. Int. J. Gen. Med. 2018, 11, 91–98. [Google Scholar] [CrossRef]
- Moyo, M.; Aremu, A.O.; Gruz, J.; Šubrtová, M.; Szüčová, L.; Doležal, K.; Van Staden, J. Conservation strategy for Pelargonium sidoides DC: Phenolic profile and pharmacological activity of acclimatized plants derived from tissue culture. J. Ethnopharmacol. 2013, 149, 557–561. [Google Scholar] [CrossRef]
- Moyo, M.; Van Staden, J. Medicinal properties and conservation of Pelargonium sidoides DC. J. Ethnopharmacol. 2014, 152, 243–255. [Google Scholar] [CrossRef]
- Theisen, L.L.; Muller, C.P. EPs® 7630 (Umckaloabo®), an extract from Pelargonium sidoides roots, exerts anti-influenza virus activity in vitro and in vivo. Antivir. Res. 2012, 94, 147–156. [Google Scholar] [CrossRef]
- Wikipedia. Pelargonium Sidoides. Available online: https://en.wikipedia.org/wiki/Pelargonium_sidoides (accessed on 21 August 2020).
- Abdel-Magied, N.; Fattah, S.M.A.; Elkady, A.A. Differential effect of Taraxacum officinale L. (dandelion) root extract on hepatic and testicular tissues of rats exposed to ionizing radiation. Mol. Biol. Rep. 2019, 46, 4893–4907. [Google Scholar] [CrossRef] [PubMed]
- Choi, J.; Yoon, K.D.; Kim, J. Chemical constituents from Taraxacum officinale and their α-glucosidase inhibitory activities. Bioorg. Med. Chem. Lett. 2018, 28, 476–481. [Google Scholar] [CrossRef] [PubMed]
- Flores-Ocelotl, M.R.; Rosas-Murrieta, N.H.; Moreno, D.A.; Vallejo-Ruiz, V.; Reyes-Leyva, J.; Domínguez, F.; Santos-López, G. Taraxacum officinale and Urtica dioica extracts inhibit dengue virus serotype 2 replication in vitro. BMC Complement. Altern. Med. 2018, 18, 95. [Google Scholar] [CrossRef] [PubMed]
- Han, H.; He, W.; Wang, W.; Gao, B. Inhibitory effect of aqueous dandelion extract on HIV-1 replication and reverse transcriptase activity. BMC Complement. Altern. Med. 2011, 11, 112. [Google Scholar] [CrossRef]
- He, W.; Han, H.; Wang, W.; Gao, B. Anti-influenza virus effect of aqueous extracts from dandelion. Virol. J. 2011, 8, 538. [Google Scholar] [CrossRef] [PubMed]
- Lee, B.-R.; Lee, J.-H.; An, H.-J. Effects of Taraxacum officinale on fatigue and immunological parameters in mice. Molecules 2012, 17, 13253–13265. [Google Scholar] [CrossRef]
- Herbs and Remedies. Medicinal Use of Dandelion—Taraxacum officinale (Asteraceae). Available online: https://herbsandremedies.club/medicinal-plants/medicinal-use-of-dandelion-taraxacum-officinale-asteraceae/ (accessed on 22 August 2020).
- Lü, H.-N.; Ma, S.-G.; Liu, Y.-B.; Qu, J.; Li, Y.; Xu, S.; Zhu, H.; Yu, S.-S. Sesquiterpenes from the roots of Illicium oligandrum. J. Asian Nat. Prod. Res. 2015, 17, 430–438. [Google Scholar] [CrossRef]
- Ma, S.-G.; Gao, R.-M.; Li, Y.-H.; Jiang, J.-D.; Gong, N.-B.; Li, L.; Lu, Y.; Tang, W.-Z.; Liu, Y.-B.; Qu, J.; et al. Antiviral spirooliganones A and B with unprecedented skeletons from the roots of Illicium oligandrum. Org. Lett. 2013, 15, 4450–4453. [Google Scholar] [CrossRef]
- Tang, W.-Z.; Liu, Y.; Yu, S.-S.; Qu, J.; Su, D.-M. New sesquiterpene lactone and neolignan glycosides with antioxidant and anti-inflammatory activities from the fruits of Illicium oligandrum. Planta Med. 2007, 73, 484–490. [Google Scholar] [CrossRef]
- Zhu, Q.; Tang, C.-P.; Ke, C.-Q.; Wang, W.; Zhang, H.-Y.; Ye, Y. Sesquiterpenoids and phenylpropanoids from pericarps of Illicium oligandrum. J. Nat. Prod. 2009, 72, 238–242. [Google Scholar] [CrossRef] [PubMed]
- Burncoose Nurseries. Illicium oligandrum. Available online: https://www.burncoose.co.uk/site/plants.cfm?pl_id=6283 (accessed on 22 August 2020).
- Dao, T.T.; Nguyen, P.H.; Lee, H.S.; Kim, E.; Park, J.; Lim, S.I.; Oh, W. Chalcones as novel influenza A (H1N1) neuraminidase inhibitors from Glycyrrhiza inflata. Bioorg. Med. Chem. Lett. 2011, 21, 294–298. [Google Scholar] [CrossRef] [PubMed]
- Dastagir, G.; Rizvi, M.A. Review—Glycyrrhiza glabra L. (Liquorice). Pak. J. Pharm. Sci. 2016, 29, 1727–1733. [Google Scholar] [PubMed]
- Harding, V.; Stebbing, J. Liquorice: A treatment for all sorts? Lancet Oncol. 2017, 18, 1155. [Google Scholar] [CrossRef]
- Pastorino, G.; Cornara, L.; Soares, S.; Rodrigues, F.; Oliveira, M. Liquorice (Glycyrrhiza glabra): A phytochemical and pharmacological review. Phytother. Res. 2018, 32, 2323–2339. [Google Scholar] [CrossRef] [PubMed]
- Kew Science. Glycyrrhiza glabra L. Available online: http://www.plantsoftheworldonline.org/taxon/urn:lsid:ipni.org:names:496941-1 (accessed on 23 August 2020).
- Dao, T.T.; Dang, T.T.; Nguyen, P.H.; Kim, E.; Thuong, P.T.; Oh, W. Xanthones from Polygala karensium inhibit neuraminidases from influenza A viruses. Bioorg. Med. Chem. Lett. 2012, 22, 3688–3692. [Google Scholar] [CrossRef] [PubMed]
- Le Pogam, P.; Boustie, J. Xanthones of lichen source: A 2016 update. Molecules 2016, 21, 294. [Google Scholar] [CrossRef]
- Panda, S.; Chand, M.; Sakhuja, R.; Jain, S. Xanthones as potential antioxidants. Curr. Med. Chem. 2013, 20, 4481–4507. [Google Scholar] [CrossRef]
- EOL. Polygala Karensium Kurz. Available online: https://eol.org/pages/2886028 (accessed on 23 August 2020).
- Domeneghetti, L.; Demarchi, I.G.; Caitano, J.Z.; Pedroso, R.B.; Silveira, T.G.V.; Lonardoni, M.V.C. Calophyllum brasiliense modulates the immune response and promotes Leishmania amazonensis intracellular death. Mediat. Inflamm. 2018, 2018, 1–9. [Google Scholar] [CrossRef]
- Jeong, H.J.; Kim, Y.M.; Kim, J.H.; Kim, J.Y.; Park, J.-Y.; Park, S.-J.; Ryu, Y.B.; Lee, W.S. Homoisoflavonoids from Caesalpinia sappan displaying viral neuraminidases inhibition. Biol. Pharm. Bull. 2012, 35, 786–790. [Google Scholar] [CrossRef]
- Kudo, E.; Taura, M.; Matsuda, K.; Shimamoto, M.; Kariya, R.; Goto, H.; Hattori, S.; Kimura, S.; Okada, S. Inhibition of HIV-1 replication by a tricyclic coumarin GUT-70 in acutely and chronically infected cells. Bioorg. Med. Chem. Lett. 2013, 23, 606–609. [Google Scholar] [CrossRef]
- Wikipedia. Calophyllum Brasiliense. Available online: https://en.wikipedia.org/wiki/Calophyllum_brasiliense (accessed on 24 August 2020).
- Dai, X.; Yi, X.; Sun, Z.; Ruan, P. Cimicifuga foetida L. plus adefovir effectively inhibits the replication of hepatitis B virus in patients with chronic hepatitis B. Biomed. Rep. 2016, 4, 493–497. [Google Scholar] [CrossRef] [PubMed]
- Gai, Y.-Y.; Liu, W.; Sha, C.-J.; Wang, Y.-L.; Sun, Y.-T.; Li, X.-J.; Fawcett, J.P.; Gu, J. Pharmacokinetics and bioavailability of cimicifugosides after oral administration of Cimicifuga foetida L. extract to rats. J. Ethnopharmacol. 2012, 143, 249–255. [Google Scholar] [CrossRef] [PubMed]
- Wang, K.-C.; Chang, J.-S.; Lin, L.-T.; Chiang, L.-C.; Lin, C.-C. Antiviral effect of cimicifugin from cimicifuga foetida against human respiratory syncytial virus. Am. J. Chin. Med. 2012, 40, 1033–1045. [Google Scholar] [CrossRef] [PubMed]
- Zhu, G.-L.; Zhu, D.-F.; Wan, L.-S.; Peng, X.-R.; Bao, N.-M.; Zhang, Z.-R.; Zhou, L.; Qiu, M.-H. Six new 9,19-cycloartane triterpenoids from Cimicifuga foetida L. Nat. Prod. Bioprospect. 2016, 6, 187–193. [Google Scholar] [CrossRef] [PubMed]
- Efloras. Cimicifuga Foetida. Available online: http://www.efloras.org/object_page.aspx?object_id=108347&flora_id=800 (accessed on 24 August 2020).
- Anbazhagan, G.K.; Palaniyandi, S.; Joseph, B. Antiviral plant extracts. Plant Extr. 2019, 1–10. [Google Scholar] [CrossRef]
- Bose, M.; Kamra, M.; Mullick, R.; Bhattacharya, S.; Das, S.; Karande, A.A. A plant-derived dehydrorotenoid: A new inhibitor of hepatitis C virus entry. FEBS Lett. 2017, 591, 1305–1317. [Google Scholar] [CrossRef]
- Manu, K.; Kuttan, G. Effect of punarnavine, an alkaloid from boerhaavia diffusa, on cell-mediated immune responses and TIMP-1 in B16F-10 metastatic melanoma-bearing mice. Immunopharmacol. Immunotoxicol. 2007, 29, 569–586. [Google Scholar] [CrossRef]
- Mishra, S.; Aeri, V.; Gaur, P.K.; Jachak, S.M. Phytochemical, therapeutic, and ethnopharmacological overview for a traditionally important herb: Boerhavia diffusaLinn. BioMed Res. Int. 2014, 2014, 1–19. [Google Scholar] [CrossRef]
- Wikipedia. Boerhavia Diffusa. Available online: https://en.wikipedia.org/wiki/Boerhavia_diffusa (accessed on 24 August 2020).
- Bag, A.; Bhattacharyya, S.K.; Chattopadhyay, R.R.; Rashid, R.A. The development of Terminalia chebula Retz. (Combretaceae) in clinical research. Asian Pac. J. Trop. Biomed. 2013, 3, 244–252. [Google Scholar] [CrossRef]
- Kesharwani, A.; Polachira, S.K.; Nair, R.; Agarwal, A.; Mishra, N.N.; Gupta, S.K. Anti-HSV-2 activity of Terminalia chebula Retz extract and its constituents, chebulagic and chebulinic acids. BMC Complement. Altern. Med. 2017, 17, 110. [Google Scholar] [CrossRef]
- Lin, L.-T.; Chen, T.-Y.; Lin, S.-C.; Chung, C.-Y.; Lin, T.-C.; Wang, G.-H.; Anderson, R.; Lin, C.-C.; Richardson, C.D. Broad-spectrum antiviral activity of chebulagic acid and punicalagin against viruses that use glycosaminoglycans for entry. BMC Microbiol. 2013, 13, 187. [Google Scholar] [CrossRef] [PubMed]
- Nigam, M.; Mishra, A.P.; Adhikari-Devkota, A.; Dirar, A.I.; Hassan, M.; Adhikari, A.; Belwal, T.; Devkota, H.P. Fruits of Terminalia chebula Retz.: A review on traditional uses, bioactive chemical constituents and pharmacological activities. Phytother. Res. 2020, 10, 1–9. [Google Scholar] [CrossRef]
- Sheng, Z.; Zhao, J.; Muhammad, I.; Zhang, Y. Optimization of total phenolic content from Terminalia chebula Retz. fruits using response surface methodology and evaluation of their antioxidant activities. PLoS ONE 2018, 13, e0202368. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; He, L.; Lu, Q.; Li, D. Pharmacological activity of Terminalia chebula. China J. Chin. Mater. Med. 2016, 41, 619–623. [Google Scholar] [CrossRef]
- Promila, P.; Madan, V.K. Therapeutic & phytochemical profiling of Terminalia chebula Retz. (harad): A review. J. Med. Plants Stud. 2018, 6, 25–31. [Google Scholar]
- Liu, A.; Shu, S.-H.; Qin, H.-L.; Lee, S.; Wang, Y.-T.; Du, G. in vitro anti-influenza viral activities of constituents from Caesalpinia sappan. Planta Med. 2009, 75, 337–339. [Google Scholar] [CrossRef] [PubMed]
- Tewtrakul, S.; Chaniad, P.; Pianwanit, S.; Karalai, C.; Ponglimanont, C.; Yodsaoue, O. Anti-HIV-1 integrase activity and molecular docking study of compounds from Caesalpinia sappan L. Phytother. Res. 2015, 29, 724–729. [Google Scholar] [CrossRef]
- Yang, F.; Zhou, W.-L.; Liu, A.; Qin, H.-L.; Lee, S.M.; Wang, Y.-T.; Du, G. The protective effect of 3-deoxysappanchalcone on in vitro influenza virus-induced apoptosis and inflammation. Planta Med. 2012, 78, 968–973. [Google Scholar] [CrossRef]
- Natures Beauty Creations. Caesalpinia sappan L. Available online: https://www.asia-medicinalplants.info/caesalpinia-sappan-l/ (accessed on 23 August 2020).
- Wagner, H.; Bauer, R.; Melchart, D.; Xiao, P.-G.; Staudinger, A. Radix et Rhizoma Rhei—Dahuang. In Chromatographic Fingerprint Analysis of Herbal Medicines: Thin-Layer and High Performance Liquid Chromatography of Chinese Drugs; Wagner, H., Bauer, R., Melchart, D., Xiao, P.-G., Staudinger, A., Eds.; Springer Vienna: Vienna, Austria, 2011; pp. 857–874. [Google Scholar]
- Su, B.; Li, X.-B. Advance in studies on effect of Glycyrrhizae Radix et Rhizoma in relieving purgative activity of Rhei Radix et Rhizoma. China J. Chin. Mater. Med. 2015, 40, 577–581. [Google Scholar]
- Wei, Y.; Liu, M.; Liu, J.; Li, H. Influence factors on the hepatotoxicity of polygoni multiflori Radix. Evid. Based Complement. Altern. Med. 2019, 2019, 5482896. [Google Scholar] [CrossRef]
- Kim, Y.-J.; Lee, J.Y.; Kim, H.-J.; Kim, D.-H.; Lee, T.H.; Kang, M.S.; Choi, Y.-K.; Lee, H.L.; Kim, J.; An, H.-J.; et al. Inhibitory effect of emodin on raw 264.7 activated with double stranded rna analogue poly I:C. Afr. J. Tradit. Complement. Altern. Med. 2017, 14, 157–166. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Liang, L.; Xu, J.; Liang, Z.-T.; Dong, X.-P.; Chen, H.; Zhao, Z.-Z. Tissue-specific analysis of secondary metabolites creates a reliable morphological criterion for quality grading of polygoni multiflori Radix. Molecules 2018, 23, 1115. [Google Scholar] [CrossRef]
- Budak, N.H. Bioactive components of Prunus avium L. black gold (red cherry) and Prunus avium L. stark gold (white cherry) juices, wines and vinegars. J. Food Sci. Technol. 2016, 54, 62–70. [Google Scholar] [CrossRef] [PubMed]
- Shen, C.-Y.; Jiang, J.-G.; Zhu, W.; Ou-Yang, Q. Anti-inflammatory effect of essential oil from Citrus aurantium L. var. amara Engl. J. Agric. Food Chem. 2017, 65, 8586–8594. [Google Scholar] [CrossRef] [PubMed]
- Stohs, S.J. Safety, efficacy, and mechanistic studies regarding citrus aurantium (bitter orange) extract andp-synephrine. Phytother. Res. 2017, 31, 1463–1474. [Google Scholar] [CrossRef]
- Pimenta, F.C.F.; Alves, M.F.; Melo, S.A.L.; De Almeida, A.A.F.; Leite, J.R.; Pordeus, L.C.D.M.; Diniz, M.D.F.F.M. Anxiolytic effect of citrus aurantium L. on patients with chronic myeloid leukemia. Phytother. Res. 2016, 30, 613–617. [Google Scholar] [CrossRef]
- Marhoume, F.Z.; Laaradia, M.A.; Zaid, Y.; Laadraoui, J.; Oufquir, S.; Aboufatima, R.; Chait, A.; Bagri, A.; Zaid, Y.; Oufkir, S. Anti-aggregant effect of butanolic extract of Rubia tinctorum L. on platelets in vitro and ex vivo. J. Ethnopharmacol. 2019, 241, 111971. [Google Scholar] [CrossRef]
- Xiong, Y.; Yang, Y.; Xiong, W.; Yao, Y.; Wu, H.; Zhang, M. Network pharmacology-based research on the active component and mechanism of the antihepatoma effect of Rubia cordifolia L. J. Cell. Biochem. 2019, 120, 12461–12472. [Google Scholar] [CrossRef]
- Shang, A.; Cao, S.-Y.; Xu, X.-Y.; Gan, R.-Y.; Tang, G.-Y.; Corke, H.; Mavumengwana, V.; Li, H.-B. Bioactive compounds and biological functions of garlic (Allium sativum L.). Foods 2019, 8, 246. [Google Scholar] [CrossRef]
- Martins, N.; Petropoulos, S.A.; Ferreira, I.C. Chemical composition and bioactive compounds of garlic (Allium sativum L.) as affected by pre- and post-harvest conditions: A review. Food Chem. 2016, 211, 41–50. [Google Scholar] [CrossRef]
- Kim, S.; Kim, D.-B.; Jin, W.; Park, J.; Yoon, W.; Lee, Y.; Kim, S.; Lee, S.; Kim, S.; Lee, O.-H.; et al. Comparative studies of bioactive organosulphur compounds and antioxidant activities in garlic (Allium sativum L.), elephant garlic (Allium ampeloprasum L.) and onion (Allium cepa L.). Nat. Prod. Res. 2017, 32, 1193–1197. [Google Scholar] [CrossRef] [PubMed]
- Chavan, R.D.; Shinde, P.; Girkar, K.; Madage, R.; Chowdhary, A. Assessment of anti-influenza activity and hemagglutination inhibition of plumbago indica and Allium sativum extracts. Pharmacogn. Res. 2016, 8, 105–111. [Google Scholar] [CrossRef] [PubMed]
- Tayel, A.A.; El-Sedfy, M.A.; Ibrahim, A.I.; Moussa, S.H. Application of Quercus infectoria extract as a natural antimicrobial agent for chicken egg decontamination. Rev. Argent. Microbiol. 2018, 50, 391–397. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, A.; Salih, F.A. Quercus infectoria gall extracts reduce quorum sensing-controlled virulence factors production and biofilm formation in Pseudomonas aeruginosa recovered from burn wounds. BMC Complement. Altern. Med. 2019, 19, 177. [Google Scholar] [CrossRef]
- Motamedi, H.; Azizi, A.; Ahmadi, M. Nutritive value of treated Quercus infectoria and Quercus libani leaves with the tannin-degrading bacterium Klebsiella pneumoniae for ruminant feeding in vitro. J. Appl. Microbiol. 2019, 127, 1339–1348. [Google Scholar] [CrossRef]
- Kheirandish, F.; Delfan, B.; Mahmoudvand, H.; Moradi, N.; Ezatpour, B.; Ebrahimzadeh, F.; Rashidipour, M. Antileishmanial, antioxidant, and cytotoxic activities of Quercus infectoria Olivier extract. Biomed. Pharmacother. 2016, 82, 208–215. [Google Scholar] [CrossRef]
- Qaderi, M.M.; Cavers, P.B.; Bernards, M.A. Isolation and structural characterization of a water-soluble germination inhibitor from Scotch thistle (Onopordum acanthium) cypselas. J. Chem. Ecol. 2003, 29, 2425–2438. [Google Scholar] [CrossRef]
- Tung, N.H.; Kwon, H.-J.; Kim, J.-H.; Ra, J.C.; Ding, Y.; Kim, J.A.; Kim, Y. Anti-influenza diarylheptanoids from the bark of Alnus japonica. Bioorg. Med. Chem. Lett. 2010, 20, 1000–1003. [Google Scholar] [CrossRef]
- Won, T.H.; Song, I.-H.; Kim, K.-H.; Yang, W.-Y.; Lee, S.K.; Oh, D.-C.; Oh, W.K.; Oh, K.-B.; Shin, J. Bioactive metabolites from the fruits of psoralea corylifolia. J. Nat. Prod. 2015, 78, 666–673. [Google Scholar] [CrossRef]
- Schneiderová, K.; Šlapetová, T.; Hrabal, R.; Dvorakova, H.; Prochazkova, P.; Novotna, J.; Urbanova, M.; Cvačka, J.; Smejkal, K. Tomentomimulol and mimulone B: Two new C geranylated flavonoids from Paulownia tomentosa fruits. Nat. Prod. Res. 2013, 27, 613–618. [Google Scholar] [CrossRef]
- Tian, C.; Zhang, Z.; Wang, H.; Guo, Y.; Zhao, J.; Liu, M. Extraction technology, component analysis, and in vitro antioxidant and antibacterial activities of total flavonoids and fatty acids from Tribulus terrestris L. fruits. Biomed. Chromatogr. 2019, 33, e4474. [Google Scholar] [CrossRef] [PubMed]
- Hawkins, J.; Baker, C.; Cherry, L.; Dunne, E. Black elderberry (Sambucus nigra) supplementation effectively treats upper respiratory symptoms: A meta-analysis of randomized, controlled clinical trials. Complement. Ther. Med. 2019, 42, 361–365. [Google Scholar] [CrossRef] [PubMed]
- Shahsavandi, S.; Ebrahimi, M.M.; Farahani, A.H. Interfering with lipid raft association: A mechanism to control influenza virus infection by Sambucus nigra. Iran. J. Pharm. Res. 2017, 16, 1147–1154. [Google Scholar] [PubMed]
- Zhang, S.; Guo, S.-L.; Wang, Q.-B.; Liu, Y.; Shen, H.-W.; Wang, Z.-Y. Effects of fungi fraction on growth and anti-oxidative activity of Eleutherococcus senticosus. China J. Chin. Mater. Med. 2019, 44, 1517–1523. [Google Scholar]
- Li, T.; Ferns, K.; Yan, Z.-Q.; Yin, S.-Y.; Kou, J.-J.; Li, D.; Zeng, Z.; Yin, L.; Wang, X.; Bao, H.-X.; et al. Acanthopanax senticosus: Photochemistry and anticancer potential. Am. J. Chin. Med. 2016, 44, 1543–1558. [Google Scholar] [CrossRef]
- Jin, L.; Schmiech, M.; El Gaafary, M.; Zhang, X.; Syrovets, T.; Simmet, T. A comparative study on root and bark extracts of Eleutherococcus senticosus and their effects on human macrophages. Phytomedicine 2020, 68, 153181. [Google Scholar] [CrossRef]
- Zhou, H.; Xing, J.; Liu, S.; Song, F.; Caib, Z.; Pi, Z.; Liu, Z.; Liu, S. Screening and determination for potential α-glucosidase inhibitors from leaves of acanthopanax senticosus harms by using UF-LC/MS and ESI-MSn. Phytochem. Anal. 2011, 23, 315–323. [Google Scholar] [CrossRef]
- Wang, L.; Zhang, R.-M.; Liu, G.-Y.; Wei, B.-L.; Wang, Y.; Cai, H.-Y.; Li, F.-S.; Xu, Y.-L.; Zheng, S.-P.; Wang, G. Chinese herbs in treatment of influenza: A randomized, double-blind, placebo-controlled trial. Respir. Med. 2010, 104, 1362–1369. [Google Scholar] [CrossRef]
- Jia, Q.; Zhu, R.; Tian, Y.; Chen, B.; Li, R.; Li, L.; Wang, L.; Che, Y.; Zhao, D.; Mo, F.; et al. Salvia miltiorrhiza in diabetes: A review of its pharmacology, phytochemistry, and safety. Phytomedicine 2019, 58, 152871. [Google Scholar] [CrossRef]
- Wang, L.; Ma, R.; Liu, C.; Liu, H.; Zhu, R.; Guo, S.; Tang, M.; Li, Y.; Niu, J.; Fu, M.; et al. Salvia miltiorrhiza: A potential red light to the development of cardiovascular diseases. Curr. Pharm. Des. 2017, 23, 1077–1097. [Google Scholar] [CrossRef]
- Rahuman, A.A.; Bagavan, A.; Kamaraj, C.; Vadivelu, M.; Zahir, A.A.; Elango, G.; Pandiyan, G.; Kamaraj, C. Evaluation of indigenous plant extracts against larvae of culex quinquefasciatus say (diptera: Culicidae). Parasitol. Res. 2008, 104, 637–643. [Google Scholar] [CrossRef]
- Vlachojannis, J.; Erne, P.; Zimmermann, B.; Chrubasik-Hausmann, S. The impact of cocoa flavanols on cardiovascular health. Phytother. Res. 2016, 30, 1641–1657. [Google Scholar] [CrossRef] [PubMed]
- Wickramasuriya, A.M.; Dunwell, J.M. Cacao biotechnology: Current status and future prospects. Plant Biotechnol. J. 2017, 16, 4–17. [Google Scholar] [CrossRef] [PubMed]
- Wirngo, F.E.; Lambert, M.N.; Jeppesen, P.B. The physiological effects of dandelion (Taraxacum officinale) in type 2 diabetes. Rev. Diabet. Stud. 2016, 13, 113–131. [Google Scholar] [CrossRef] [PubMed]
- Schütz, K.; Carle, R.; Schieber, A. Taraxacum—A review on its phytochemical and pharmacological profile. J. Ethnopharmacol. 2006, 107, 313–323. [Google Scholar] [CrossRef]
- Wang, K.C.; Chang, J.S.; Chiang, L.C.; Lin, C.C. Cimicifuga foetida L. inhibited human respiratory syncytial virus in HEp-2 and A549 cell lines. Am. J. Chin. Med. 2012, 40, 151–162. [Google Scholar] [CrossRef]
- Thomford, N.E.; Awortwe, C.; Dzobo, K.; Adu, F.; Chopera, D.; Wonkam, A.; Skelton, M.; Blackhurst, D.; Dandara, C. Inhibition of CYP2B6 by medicinal plant extracts: Implication for use of efavirenz and nevirapine-based Highly Active Anti-Retroviral Therapy (HAART) in resource-limited settings. Molecules 2016, 21, 211. [Google Scholar] [CrossRef]
- Mandeville, A.; Cock, I.E. Terminalia chebula Retz. fruit extracts inhibit bacterial triggers of some autoimmune diseases and potentiate the activity of tetracycline. Indian J. Microbiol. 2018, 58, 496–506. [Google Scholar] [CrossRef]
- Li, K.; Han, X.; Li, R.; Xu, Z.; Pan, T.; Liu, J.; Li, B.; Wang, S.; Diao, Y.; Liu, X. Composition, antivirulence activity, and active property distribution of the fruit of terminalia chebula Retz. J. Food Sci. 2019, 84, 1721–1729. [Google Scholar] [CrossRef]
- El Sayed, K.A. Natural products as antiviral agents. Stud. Nat. Prod. Chem. 2000, 24, 473–572. [Google Scholar] [CrossRef]
- Creagh, T.; Ruckle, J.L.; Tolbert, D.T.; Giltner, J.; Eiznhamer, D.A.; Dutta, B.; Flavin, M.T.; Xu, Z.-Q. Safety and pharmacokinetics of single doses of (+) − calanolide a, a novel, naturally occurring nonnucleoside reverse transcriptase inhibitor, in healthy, human immunodeficiency virus-negative human subjects. Antimicrob. Agents Chemother. 2001, 45, 1379–1386. [Google Scholar] [CrossRef] [PubMed]
- Currens, M.J.; Gulakowski, R.J.; Mariner, J.M.; Moran, R.A.; Buckheit, R.W.; Gustafson, K.R.; McMahon, J.B.; Boyd, M.R. Antiviral activity and mechanism of action of calanolide A against the human immunodeficiency virus type-1. J. Pharmacol. Exp. Ther. 1996, 279, 645–651. [Google Scholar] [PubMed]
- Ubillas, R.; Jolad, S.; Bruening, R.; Kernan, M.; King, S.; Sesin, D.; Barrett, M.; Stoddart, C.; Flaster, T.; Kuo, J.; et al. SP-303, an antiviral oligomeric proanthocyanidin from the latex of Croton lechleri (Sangre de Drago). Phytomedicine 1994, 1, 77–106. [Google Scholar] [CrossRef]
- Wyde, P.R.; Ambrose, M.W.; Meyerson, L.R.; Gilbert, B.E. The antiviral activity of SP-303, a natural polyphenolic polymer, against respiratory syncytial and parainfluenza type 3 viruses in cotton rats. Antivir. Res. 1993, 20, 145–154. [Google Scholar] [CrossRef]
| Botanical Name and Vegetal Part Use for Medicinal Purpose | Picture | Bioactive Compounds | Biological Activities | Therapeutic Effect against Diseases | References |
|---|---|---|---|---|---|
| Medicinal Plants Against Coronavirus Related Infections | |||||
| Bupleurum species (Root) |  | SSa, SSb2, SSc and SSd | Antiviral, anti-inflammatory, anti-tumor, neuro-modulation, immune-regulation | CoVs and Influenza virus | [32,34,35] |
| Lycoris radiate (L’Hér.) Herb. (Flower and stem cortex) |  | Lycorine | Antiviral effects, anticancer, anti-malarial, anti-inflammatory, induction of nausea and emesis | SARS-CoV-1, poliovirus, HIV, HSV and coxsackie virus | [41,42,43,44,71] |
| Artemisia annua L. (Whole plant) |  | Quercetin, flavonoid, polyphenols, triterpenes, sterols, saponins, polysaccharides, dicaffeoylquinic acid | Anti-malarial, antiviral, anticancer, bronchitis, haemorrhoids | SARS-CoV-1, MERS-CoV, Poliovirus, HIV, RSV, HSV1, hepatitis C, type 2 dengue virus and human cytomegalovirus | [29,45,48,49,50,72] |
| Pyrrosia lingua(Thunb.) Farw. (Leaves) |  | Flavonoids, (mangiferin, isomangiferin, trifolin, astragalin), chlorogenic acid, mangiferin, isomangiferin, astragalin, and trifolin | Antiviral, antioxidant, antibacterial, anticancer | HIV, SARS-CoV-1 | [48,50,52,54,73] |
| Isatis indigotica Fortune ex Lindl. (Leaf and root) |  | Indigo, indirubin, indican, β-sitosterol, sinigrin, hesperetin, aloe-emodin | Antiviral, antioxidant, antibacterial | SARS-CoV-1 3CLpro, HSV1, Influenza virus, coxsackie virus B3 | [48,55,57,58,74] |
| Torreya nucifera L. (Leaves) |  | Biflavonoid amentoflavone | Antiviral | SARS-CoV-1 3CLpro, stomachache, hemorrhoids, and rheumatoid arthritis | [48,59,61,75] |
| Houttuynia cordata Thunb. (Leaves) |  | Volatile oils, organic acids, flavonoids cordarine, kalium sulfuricum, potassium, zinc, iron, copper, amino acid, vitamins and manganese | Antiviral, anti-inflammatory, anti-allergic, anti-oxidant, Immunomodulatory and anticancer | SARS-CoV-1 3CLpro and RdRp, cough, lung abscess, phlegm, dyspnea, pneumonia, refractory hemoptysis | [48,57,63,65,66,76] |
| Lindera aggregate (Sims) Kosterm. (Root) |  | Flavonoids, isoquinoline alkaloids, sesquiterpene lactones and tannins | Antiviral, anti-tumor, anti-inflammatory, antimicrobial and anti-diabetic | SARS-CoV-1, chest pain, inflammation, indigestion, cold hernia | [48,50,68,69,77] |
| Rheum palmatum L. (Root) |  | Emodin, physcion, chrysophanol, rhein, and aloe-emodin | Antiviral, anti-pyretic, anti-neoplastic, anti-pasmolytic, antibacterial, laxative, hemostatic, and anti-spasmodic | SARS-CoV-1 ACE2, laxative or astringent, stomachicum, hemorrhoids, liver bile disease or gastroenteritis | [78,79,80,81,82,83] |
| Polygonum multiflorum Thunb. (Root) |  | Polygonumosides A, B, C, and D, resveratrol, chrysophanol, polydatin, emodin-1,6-dimethyl ether, rhaponticoside, emodin, 2-acetylemodin, physcion, rhein, citreorosein, apigenin, fallacinol, tricin, rutin, quercetin, luteolin, kaempferol, iso-orientin, hyperoside, vitexin, quercetin-3-O-arabinoside, polygonflavanol A, hexadecanoic acid ethyl ester, phosphatidylethanolamine, hexanoic acid, copaene, eicosane, squalene, catechin, epicatechin, 3-O-galloyl-procyanidin B2, β-sitosterol, gallic acid, methyl gallate, daucosterol, and schizandrin | Anti-CoVs, antioxidant, immunomodulation, anti-hyperlipidemia, anticancer, hepato-protection, anti-inflammation, | SARS-CoV-1 ACE2, rubella, scrofula, waist and knee pain, paralysis, vaginal discharge, hypercholesterolemia (liver and kidney), malaria, neuro-protective | [78,84,85,86,87,88,89] |
| Cerasus avium (L.) Moench (Stem) |  | Polyphenols, carotenoids, vitamins, minerals | Antioxidant, antimicrobial and antiviral | SARS-CoV-1 ACE2, oxidative stress, tooth aches and mouth diseases | [78,90,91,92,93,94,95] |
| Alcea digitata (Boiss.) Alef (Flower) |  | Unknown | Antiviral, antioxidant, anti-inflammatory, antimicrobial, anti-tussive, expectorant and laxative | SARS-CoV-1 ACE2, lung respiratory disorder, head and neck cancer and lubrication of throat | [91,92,96,97,98] |
| Citrus aurantium L. (Fruit) |  | Phenolics (flavanone glycosides, hydroxycinnamic acids), vitamin C, and carotenoids | Antiviral, antioxidant, anticancer | SARS-CoV-1 ACE2, anxiety, lung related disease, obesity, gastrointestinal disorder and prostate cancer | [91,92,99,100,101,102] |
| Rubia tinctorum L. (Root) |  | Anthraquinone, Alizarin and pseudopurpurin | Antiviral, antimicrobial | SARS-CoV-1 ACE2, kidney, bladder stone, menstrual and urinary disorder | [91,92,103,104,105] |
| Allium sativum L. (Cloves) |  | Alliin, allicin, ajoenes, vinyldithiins, and flavonoids | Antiviral, antimicrobial, antioxidant, anti-inflammatory, and anticancer | SARS-CoV-1 ACE2, inflammation, cancer and bacterial infection | [91,92,94,106,107,108,109,110] |
| Quercus infectoria G. Olivier (Gall) |  | Phenolic compound (p-hydroxybenzoic acid, catechol, caffeine, pyrogallol, catechein, e-vanillic acid, 3-hydroxytyrosol cinnamic, p-Coumaric, gallic acids and resveratrol), flavonoid compounds | Antiviral, anti-fungal, antibacterial, antioxidant, anti-inflammatory, anti-diabetic, anti-parasitic, anti-venom | SARS-CoV-1 ACE2, diarrhea, menorrhagia, dysentery, gonorrhea, tonsillitis, impetigo and internal hemorrhages | [111,112,113,114,115] |
| Onopordum acanthium L. (Leaf, flower, stem and root) |  | Flavonoids, sesquiterpene lactones, lignans, phenylpropanoids, triterpenoids, and sterols | Antiviral, anti-tumor, anti-inflammatory, antioxidant and cardio-tonic agent | SARS-CoV-1 ACE2, cancer, treat nervousness | [113,116,117,118,119] |
| Berberis integerrim Bunge (Root) |  | Berbamine, berberuin, palmatine, oxyacanthine, malic acid, ascorbic acid, caffeic acid, ursolic acid, coumarin, beta carotene, and tannin | Antiviral, anti-inflammatory, anti-hyperglycemic, anti-hyperlipidemic, anticancer, antioxidant | SARS-CoV-1 ACE2, alleviate insomnia, bronchial diseases, and liver disorder | [113,120,121,122,123] |
| Crataegus microphylla C. Koch (Leaves, flower, stem and root) |  | Phenols, phenolic acids, procyanidins, flavonoids, triterpenes, polysaccharides, catecho-lamines | Antiviral, antioxidant, anti-inflammatory and anti-diabetic | SARS-CoV-1 ACE2, heart muscle cells activation, coronary dilation, regulated blood flow | [113,120,124,125,126,127] |
| Alnus japonica (Thunb.) Steud. (Bark) |  | Hirsutenone, oregonin, rubranoside rubranoside B, rubranol, and hirsutanonol | Antiviral, anticancer, anti-inflammatory, antioxidant and induction of lymphatic and gastroenteric disorders. | SARS-CoV-PLpro fever, cancer, blood and lymphatic disorders, gastroenteric disorders | [128,129,130] |
| Psoralea corylifolia L. (Seed) |  | Neobavaisoflavone, isobavachalcone, Bavachinin, 40 –O-methyl bavachalcone, corylifol A and psoralidin | Antiviral, antioxidant, antibacterial and anti-depressant activities | SARS-CoV-PLpro leukoderma, psoriasis, vitiligo, asthma, ulcers, kidney disorders | [131,132,133,134] |
| Paulownia tomentosa (Thunb.) Steud. (Fruit) |  | Tomentin A, tomentin B, tomentin C, tomentin D, tomentin E, geranylated flavonones | Antiviral, antioxidant and antibacterial | SARS-CoV-PLpro inflammatory bronchitis, upper respiratory tract infection, asthma, tonsillitis, gonorrhea, traumatic bleeding, enteritis, bacteriological diarrhea, erysipelas, swelling, bronchopneumonia, conjunctivitis, and hemorrhoid | [135,136,137,138,139] |
| Tribulus terrestris L. (Fruit) |  | Flavonoid and alkaloids | Antiviral, anti-inflammatory, antioxidant, anti-tumor, anti-diabetic and anti-urolithic | SARS-CoV-PLpro hypertension, premature ejaculation, erectile dysfunction, vitiligo, and kidney | [140,141,142,143] |
| Angelica keiskei (Miq.) Koidz. (Leaves) |  | Chalcones, flavanones and coumarins, coumarins phenolic, acetylenes, sesquiterpene, diterpene, and triterpenes | Antiviral, antibacterial, anti-diabetic, anticancer, anti-inflammatory, antioxidative, anti-coagulant, anti-obesity, anti-tumor, anti-mutagenic and hepato-protective | SARS-CoV-1 3CLpro, bacterial treatment, cancer and diabetes | [144,145,146,147,148] |
| Medicinal Plants against Other Viral Infections | |||||
| Sambucus nigra L. (Leaf, flower and fruit) |  | Flavonoids, lectins, anthocyanin, peptic polysaccharides, polyphenolic compound | Antiviral, Immunomodulatory activity, anti-inflammatory | Common cold, HIV, HSV1, influenza, urinary tract infection, edema, rheumatic | [149,150,151,152,153,154,155,156] |
| Eleutherococcus senticosus (Rupr. & Maxim.) Maxim. (Leaf and root) |  | Phenols, lignans, coumarins, phenylpropanoids, flavonoids, hyperin, rutin, afzelin, quercetin, kaempferol, phenolic acids, triterpenic acids, and anthocyanin | Antiviral, anti-diabetic, anticancer, antioxidant | Influenza virus, chronic coughing, fatigue and infection, ischemic heart disease, diabetic, cancer, altitude sickness, neurodegenerative disorder | [101,149,157,158,159,160] |
| Salvia miltiorrhiza Bunge (Root) |  | Lipophilic diterpenoids, flavonoids, triterpenoids and hydrophilic phenolic compound | Antiviral | HIV, enterovirus removing blood stasis, improving blood circulation, atherosclerosis, thrombosis, angina pectoris, cardiovascular disease | [161,162,163,164,165] |
| Acacia arabica (Lam.) Willd (Leaves) |  | Methyl 3,4,5 tri hydroxyl benzoate, ferulic acid, p -coumaroylquinic acid, isoferulic acid, p-coumaroyl glucoside, epicatechin-3-gallate, ascorbic acid, quercetin, oleic acid, myristic acid, palmitic acid and steroidal sapogenin aglycone | Antiviral, antimicrobial, anti-diabetic, antioxidant | HIV, influenza virus, Newcastle disease, vaccinia virus, bursal disease virus, skin disease | [149,166,167,168,169,170] |
| Ocimum sanctum L. (Leaves) |  | Flavonoids, tannins, saponins, alkaloids, phenols, anthocyanine, triterpenoids, | Antiviral, antimicrobial, anti-cataract, anti-inflammatory, anti-diabetic, anti-hypercholesterolemia, anti-hypertensive, anti-carcinogenic, anti-pyretic, anti-allergic, immunomodulatory, anti-asthmatic, anti-tussive, anti-fertility, anti-ulcer, anti-emetic, anti-spasmodic, anti-arthritic, adaptogenic, anti-leukodermal, anti-coagulant activities | H9N2 influenza disease anxiety, cough, asthma, diarrhea, fever, skin disease, dysentery, arthritis, eye diseases, otalgia, indigestion, hiccups, vomiting, gastric, cardiac and genitourinary disorders, back pain, skin diseases, ringworm, insect, snake and scorpion bites, malaria and antioxidant | [167,171,172,173,174,175,176,177,178,179] |
| Ocimum basilicum L. (Whole plant) |  | Phenolic compounds, flavonoids and anthocyanins | Antiviral, anti-inflammatory, antioxidant and antibacterial | HIV infection and bacterial infection | [149,180,181,182,183,184,185] |
| Theobroma cacao L. (Seed) |  | Polyphenol, theobromine and flavonoids (heobromine, lignin, dietary fiber, free fatty acid, minerals, zinc, copper, iron) | Antiviral, antioxidant, anti-inflammatory | Influenza virus | [186,187,188,189,190] |
| Pelargonium sidoides DC (Root) |  | Methoxycoumarin, proanthocyanidins, EPs 7639 and prodelphinidins | Antiviral and antioxidant | Influenza virus, tuberculosis, respiratory disease, cough, gastrointestinal infection, viral disease | [191,192,193,194,195] |
| Taraxacum officinale (L.) WEB. ex WIGG. (Aerial part and root) |  | Terpenes, flavonoids, phenolic compounds, terpenoids, triterpenoids, steroids, coumarins, phenols, saponins, flavones, flavonols, chalcones, phlobatannins, and cardiac glycosides | Antiviral, antibacterial, choleretic, anti-diabetic, anti-inflammatory, antioxidant, hepato-protective, diuretic and antifungal | HIV, influenza virus, kidney related disease, lung related disease, tumor of breast, diabetic, uterus related infection, digestive system related abnormality | [48,196,197,198,199,200,201,202] |
| Illicium oligandrum Merr and Chun (Root) |  | Sesquiterpene lactones, neolignan glycosides, phenolic diglycosides and prenylated compounds | Antiviral | HSV, coxsackie virus and influenza virus, rheumatoid arthritis, neurotoxic and neurotrophic effects. | [203,204,205,206,207] |
| Glycyrrhiza glabra L. (Root) |  | Flavonoids, glycyrrhizic acid, triterpenoid, saponins | Antiviral, anti-inflammatory, antimicrobial, antioxidant, anti-tumorigenic and anti-ulcer | CoVs, HIV, influenza virus | [208,209,210,211,212,213] |
| Polygala karensium Kurz (Root) |  | Xanthones | Antiviral, antimicrobial, antioxidant, cytotoxicity activity | Influenza virus, cough, bronchitis, neurasthenia, inflammation and amnesia | [214,215,216,217] |
| Calophyllum brasiliense Cambess (Leaves) |  | Tricyclic coumarin | Antiviral, antibacterial, anti-protozoal and antifungal | HIV, parasitic diseases, bacterial and fungal disease | [218,219,220,221] |
| Cimicifuga foetida L. (Rhizomes) |  | Cimicifugin, cycloartane triterpenoids and glycosides | Antiviral, anti-tumor, anti-inflammatory | Respiratory Syncytial Virus, fever, headache, sore throat, toothache, uterine prolapse and inflammation | [222,223,224,225,226] |
| Boerhavia diffusa L. (Leaf, stem and root) |  | Flavonoids, triterpenoids, alkaloids, hypoxanthine, steroids, lipids, lignins, ursolic acid, boeravinone, punarnavoside | Antiviral, anti-fibrinolytic, anti-convulsant, antibacterial, anti-hepatotoxic, anti-asthmatic and anti-nematodal activity | Hepatitis C virus, abdominal pain, jaundice, dyspepsia, release the stress, spleen enlargement, liver | [227,228,229,230,231] |
| Terminalia chebula Retz (Leaf, bark and fruit) |  | Flavonoids, polyphenols, terpenes, anthocyanins, glycosides, gallic acid, chebulagic acid, punicalagin, chebulanin, corilagin, neochebulinic acid, ellagic acid, chebulinic acid, alkaloids | Antiviral, antioxidant, antibacterial, antifungal, anti-protozoal, anti-carcinogenic, anti-mutagenic, anti-diabetic, reno-protective, anti-inflammatory, anti-arthritic, anti-anaphylactic, anti-caries, anti-allergic, immunomodulatory, anti-ulcer, anti-spasmodic | Human cytomegalovirus, hepatitis C virus, dengue virus, measles virus, respiratory syncytial virus, irregular fevers, urinary diseases, diabetes, skin diseases, heart diseases, constipation, ulcers, vomiting, colic pain, hemorrhoids, digestive diseases | [232,233,234,235,236,237,238] |
| Caesalpinia sappan L. (Root) |  | Xanthone, sappanchalcone, coumarin, chalcones, flavones, homoisoflavonoids, and brazilin | Antiviral, anti-inflammatory, antioxidant, antibacterial, antifungal, anti-complementary | HIV, Influenza virus, tuberculosis, diarrhea, dysentery, skin infections and anemia | [219,239,240,241,242] |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Siddiqui, A.J.; Danciu, C.; Ashraf, S.A.; Moin, A.; Singh, R.; Alreshidi, M.; Patel, M.; Jahan, S.; Kumar, S.; Alkhinjar, M.I.M.; et al. Plants-Derived Biomolecules as Potent Antiviral Phytomedicines: New Insights on Ethnobotanical Evidences against Coronaviruses. Plants 2020, 9, 1244. https://doi.org/10.3390/plants9091244
Siddiqui AJ, Danciu C, Ashraf SA, Moin A, Singh R, Alreshidi M, Patel M, Jahan S, Kumar S, Alkhinjar MIM, et al. Plants-Derived Biomolecules as Potent Antiviral Phytomedicines: New Insights on Ethnobotanical Evidences against Coronaviruses. Plants. 2020; 9(9):1244. https://doi.org/10.3390/plants9091244
Chicago/Turabian StyleSiddiqui, Arif Jamal, Corina Danciu, Syed Amir Ashraf, Afrasim Moin, Ritu Singh, Mousa Alreshidi, Mitesh Patel, Sadaf Jahan, Sanjeev Kumar, Mulfi I. M. Alkhinjar, and et al. 2020. "Plants-Derived Biomolecules as Potent Antiviral Phytomedicines: New Insights on Ethnobotanical Evidences against Coronaviruses" Plants 9, no. 9: 1244. https://doi.org/10.3390/plants9091244
APA StyleSiddiqui, A. J., Danciu, C., Ashraf, S. A., Moin, A., Singh, R., Alreshidi, M., Patel, M., Jahan, S., Kumar, S., Alkhinjar, M. I. M., Badraoui, R., Snoussi, M., & Adnan, M. (2020). Plants-Derived Biomolecules as Potent Antiviral Phytomedicines: New Insights on Ethnobotanical Evidences against Coronaviruses. Plants, 9(9), 1244. https://doi.org/10.3390/plants9091244














