Role of Organic Anions and Phosphatase Enzymes in Phosphorus Acquisition in the Rhizospheres of Legumes and Grasses Grown in a Low Phosphorus Pasture Soil
Abstract
:1. Introduction
2. Results
2.1. Plant Biomass and Plant P Contents
2.2. Rhizosphere pH
2.3. Microbial Biomass P
2.4. Phosphatase Activity
2.5. Organic Anions
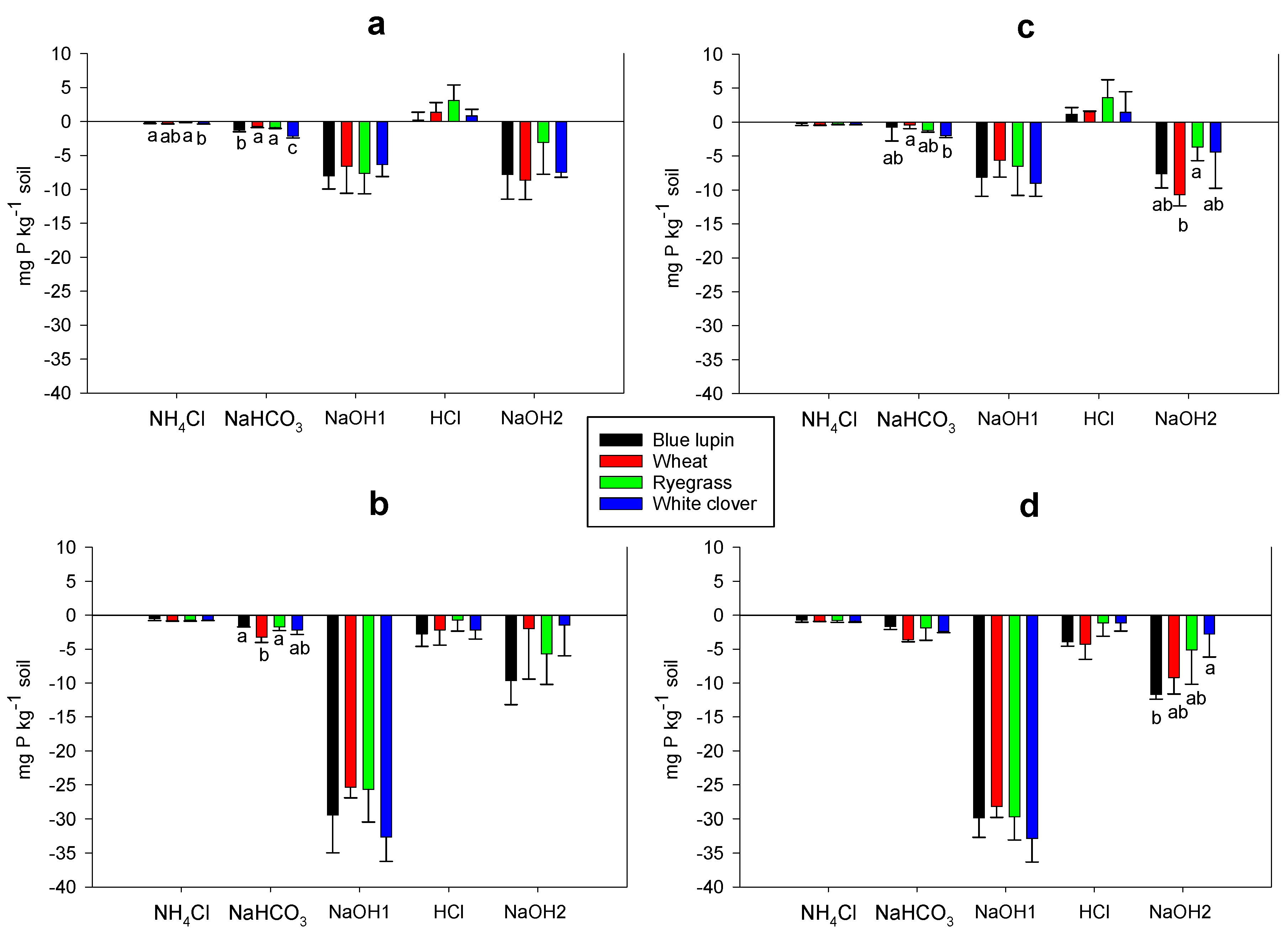
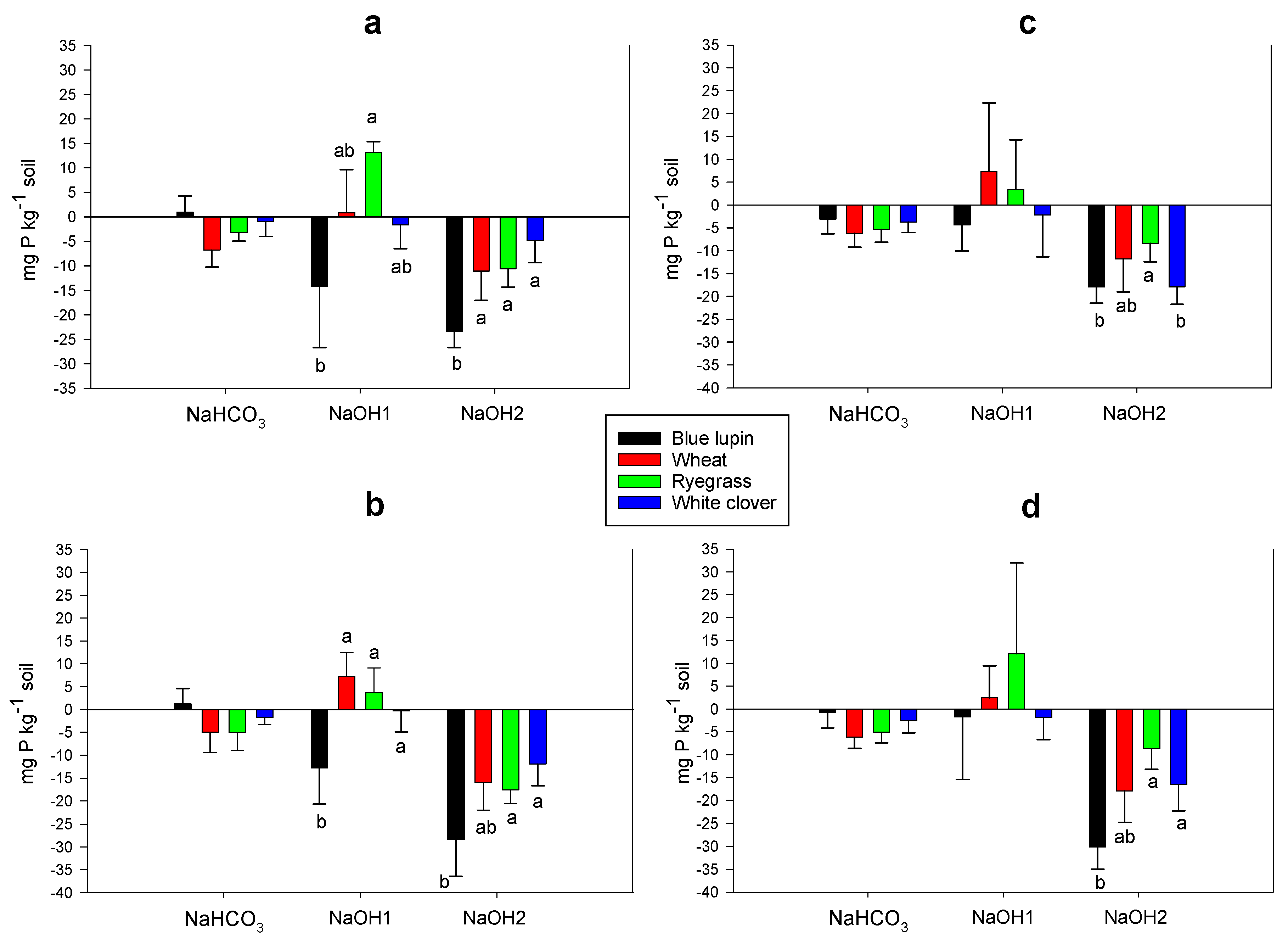
2.6. Rhizosphere P Fractions
3. Discussion
3.1. Plant Responses to Nutrient Addition
3.2. Rhizosphere pH
3.3. Microbial Biomass P
3.4. Phosphatases Activity
3.5. Organic Anions
3.6. Rhizosphere P Fractions and P Acquisition
4. Materials and Methods
4.1. Soil Preparation and Characteristics
4.2. Experimental Design
4.3. Plant and Soil Analyses
4.4. Data Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Butusov, M.; Jernelöv, A. Phosphorus: An element that could have been called Lucifer. In SpringerBriefs in Environmental Science; Springer: New York, NY, USA, 2013; Volume 9, ISBN 978-1-4614-6802-8. [Google Scholar]
- Raghothama, K.G. Phosphorus and Plant Nutrition: An Overview. Phosphorus Agric. Environ. 2005, agronomymonogra, 355–378. [Google Scholar] [CrossRef]
- Holford, I.C.R. Soil phosphorus: its measurement, and its uptake by plants. Soil Res. 1997, 35, 227–240. [Google Scholar] [CrossRef]
- Hedley, M.J.; Stewart, J.W.B.; Chauhan, B.S. Changes in Inorganic and Organic Soil Phosphorus Fractions Induced by Cultivation Practices and by Laboratory Incubations 1. Soil Sci. Soc. Am. J. 1982, 46, 970–976. [Google Scholar] [CrossRef]
- Hinsinger, P. Bioavailability of soil inorganic P in the rhizosphere as affected by root-induced chemical changes: A review. Plant Soil 2001, 237, 173–195. [Google Scholar] [CrossRef]
- Pierzynski, G.M.; McDowell, R.W. Chemistry, cycling, and potential movement of inorganic phosphorus in soils. In Phosphorus: Agriculture and the Environment; Sims, J.T., Sharpley, A.N., Eds.; Agronomy Monograph; American Society of Agronomy, Crop Science Society of America, and Soil Science Society of America: Madison, WI, USA, 2005; pp. 53–86. ISBN 978-0-89118-269-6. [Google Scholar]
- Condron, L.M.; Turner, B.L.; Cade-Menun, B.J. Chemistry and dynamics of soil organic phosphorus. Phosphorus Agric. Environ. 2005, agronomymonogra, 87–121. [Google Scholar] [CrossRef] [Green Version]
- Dalal, R.C. Soil organic phosphorus. In Advances in Agronomy; Brady, N.C., Ed.; Academic Press: Cambridge, MA, USA, 1977; Volume 29, pp. 83–117. [Google Scholar]
- Nash, D.M.; Haygarth, P.M.; Turner, B.L.; Condron, L.M.; McDowell, R.W.; Richardson, A.E.; Watkins, M.; Heaven, M.W. Using organic phosphorus to sustain pasture productivity: A perspective. Geoderma 2014, 221–222, 11–19. [Google Scholar] [CrossRef]
- Richardson, A.E.; Lynch, J.P.; Ryan, P.R.; Delhaize, E.; Smith, F.A.; Smith, S.E.; Harvey, P.R.; Ryan, M.H.; Veneklaas, E.J.; Lambers, H.; et al. Plant and microbial strategies to improve the phosphorus efficiency of agriculture. Plant Soil 2011, 349, 121–156. [Google Scholar] [CrossRef]
- Richardson, A.E.; Simpson, R.J. Soil microorganisms mediating phosphorus availability update on microbial phosphorus. Plant Physiol. 2011, 156, 989–996. [Google Scholar] [CrossRef] [Green Version]
- Yang, X.; Post, W.M. Phosphorus transformations as a function of pedogenesis: A synthesis of soil phosphorus data using Hedley fractionation method. Biogeosciences 2011, 8, 2907–2916. [Google Scholar] [CrossRef] [Green Version]
- Hinsinger, P.; Bengough, A.G.; Vetterlein, D.; Young, I.M. Rhizosphere: biophysics, biogeochemistry and ecological relevance. Plant Soil 2009, 321, 117–152. [Google Scholar] [CrossRef]
- Richardson, A.E.; Hocking, P.J.; Simpson, R.J.; George, T.S. Plant mechanisms to optimise access to soil phosphorus. Crop Pasture Sci. 2009, 60, 124. [Google Scholar] [CrossRef]
- Shen, J.; Yuan, L.; Zhang, J.; Li, H.; Bai, Z.; Chen, X.; Zhang, W.; Zhang, F. Phosphorus dynamics: From soil to plant. Plant Physiol. 2011, 156, 997–1005. [Google Scholar] [CrossRef] [Green Version]
- Vance, C.P.; Uhde-Stone, C.; Allan, D.L. Phosphorus acquisition and use: critical adaptations by plants for securing a nonrenewable resource. New Phytol. 2003, 157, 423–447. [Google Scholar] [CrossRef] [Green Version]
- Jones, D.L.; Oburger, E. Solubilization of Phosphorus by Soil Microorganisms. In Phosphorus in Action: Biological Processes in Soil Phosphorus Cycling; Bünemann, E., Oberson, A., Frossard, E., Eds.; Soil Biology; Springer: Berlin/Heidelberg, Germany, 2011; pp. 169–198. ISBN 978-3-642-15271-9. [Google Scholar]
- Nannipieri, P.; Giagnoni, L.; Landi, L.; Renella, G. Role of phosphatase enzymes in soil. In Phosphorus in Action; Bünemann, E., Oberson, A., Frossard, E., Eds.; Springer: Berlin/Heidelberg, Germany, 2011; Volume 26, pp. 215–243. ISBN 978-3-642-15270-2. [Google Scholar]
- Chen, C.R.; Condron, L.M.; Davis, M.R.; Sherlock, R.R. Phosphorus dynamics in the rhizosphere of perennial ryegrass (Lolium perenne L.) and radiata pine (Pinus radiata D. Don.). Soil Biol. Biochem. 2002, 34, 487–499. [Google Scholar] [CrossRef]
- Marschner, H. The soil-root interface (rhizosphere) in relation to mineral nutrition. In Mineral Nutrition of Higher Plants, 2nd ed.; Academic Press: London, UK, 1995; pp. 537–595. ISBN 978-0-12-473542-2. [Google Scholar]
- Oburger, E.; Jones, D.L.; Wenzel, W.W. Phosphorus saturation and pH differentially regulate the efficiency of organic acid anion-mediated P solubilization mechanisms in soil. Plant Soil 2011, 341, 363–382. [Google Scholar] [CrossRef]
- Villegas, J.; Fortin, J.A. Phosphorus solubilization and pH changes as a result of the interactions between soil bacteria and arbuscular mycorrhizal fungi on a medium containing NO3- as nitrogen source. Can. J. Bot. 2002, 80, 571–576. [Google Scholar] [CrossRef]
- Jones, D.L. Organic acids in the rhizosphere—A critical review. Plant Soil 1998, 205, 25–44. [Google Scholar] [CrossRef]
- Hocking, P.J. Organic acids exuded from roots in phosphorus uptake and aluminum tolerance of plants in acid soils. In Advances in Agronomy; Academic Press: Cambridge, MA, USA, 2001; Volume 74, pp. 63–97. [Google Scholar]
- Wang, Y.; Whalen, J.K.; Chen, X.; Cao, Y.; Huang, B.; Lu, C.; Shi, Y. Mechanisms for altering phosphorus sorption characteristics induced by low-molecular-weight organic acids. Can. J. Soil Sci. 2016, 96, 289–298. [Google Scholar] [CrossRef] [Green Version]
- Richardson, A.E. Soil microorganisms and phosphorus availability. In Soil biota: Management in Sustainable Farming Systems; Pankhurst, C.E., Doube, B.M., Gupta, V.V.S.R., Grace, P.R., Eds.; CSIRO: Canberra, Australia, 1994; pp. 50–62. [Google Scholar]
- Tabatabai, M.A. Soil Enzymes. In Methods of Soil Analysis: Part 2—Microbiological and Biochemical Properties; Mickelson, S.H., Bigham, J.M., Eds.; Soil Science Society of America: Madison, WI, USA, 1994; pp. 775–833. [Google Scholar]
- Gianfreda, L. Enzymes of importance to rhizosphere processes. J. Soil Sci. Plant Nutr. 2015, 15, 283–306. [Google Scholar] [CrossRef] [Green Version]
- Schefe, C.R.; Watt, M.; Slattery, W.J.; Mele, P.M. Organic anions in the rhizosphere of Al-tolerant and Al-sensitive wheat lines grown in an acid soil in controlled and field environments. Soil Res. 2008, 46, 257. [Google Scholar] [CrossRef]
- Zhou, Y.; Zhu, H.; Yao, Q. Contrasting P acquisition strategies of the bacterial communities associated with legume and grass in subtropical orchard soil. Environ. Microbiol. Rep. 2018, 10, 310–319. [Google Scholar] [CrossRef]
- Giles, C.D.; Richardson, A.E.; Cade-Menun, B.J.; Mezeli, M.M.; Brown, L.K.; Menezes-Blackburn, D.; Darch, T.; Blackwell, M.S.A.; Shand, C.A.; Stutter, M.I.; et al. Phosphorus acquisition by citrate- and phytase-exuding Nicotiana tabacum plant mixtures depends on soil phosphorus availability and root intermingling. Physiol. Plant. 2018, 163, 356–371. [Google Scholar] [CrossRef] [PubMed]
- Darch, T.; Blackwell, M.S.A.; Chadwick, D.; Haygarth, P.M.; Hawkins, J.M.B.; Turner, B.L. Assessment of bioavailable organic phosphorus in tropical forest soils by organic acid extraction and phosphatase hydrolysis. Geoderma 2016, 284, 93–102. [Google Scholar] [CrossRef] [PubMed]
- Clarholm, M.; Skyllberg, U.; Rosling, A. Organic acid induced release of nutrients from metal-stabilized soil organic matter–The unbutton model. Soil Biol. Biochem. 2015, 84, 168–176. [Google Scholar] [CrossRef]
- Cabeza, R.A.; Myint, K.; Steingrobe, B.; Stritsis, C.; Schulze, J.; Claassen, N. Phosphorus fractions depletion in the rhizosphere of young and adult maize and oilseed rape plants. J. Soil Sci. Plant Nutr. 2017, 17, 824–838. [Google Scholar] [CrossRef]
- Fan, Y.; Zhong, X.; Lin, F.; Liu, C.; Yang, L.; Wang, M.; Chen, G.; Chen, Y.; Yang, Y. Responses of soil phosphorus fractions after nitrogen addition in a subtropical forest ecosystem: Insights from decreased Fe and Al oxides and increased plant roots. Geoderma 2019, 337, 246–255. [Google Scholar] [CrossRef]
- Li, G.; Li, H.; Leffelaar, P.A.; Shen, J.; Zhang, F. Dynamics of phosphorus fractions in the rhizosphere of fababean (Vicia faba L.) and maize (Zea mays L.) grown in calcareous and acid soils. Crop Pasture Sci. 2015, 66, 1151–1160. [Google Scholar] [CrossRef]
- Ye, D.; Zhang, X.; Li, T.; Xu, J.; Chen, G. Phosphorus-acquisition characteristics and rhizosphere properties of wild barley in relation to genotypic differences as dependent on soil phosphorus availability. Plant Soil 2018, 423, 503–516. [Google Scholar] [CrossRef]
- Li, L.; Tang, C.; Rengel, Z.; Zhang, F. Chickpea facilitates phosphorus uptake by intercropped wheat from an organic phosphorus source. Plant Soil 2003, 248, 297–303. [Google Scholar] [CrossRef]
- Veneklaas, E.J.; Stevens, J.; Cawthray, G.R.; Turner, S.; Grigg, A.M.; Lambers, H. Chickpea and white lupin rhizosphere carboxylates vary with soil properties and enhance phosphorus uptake. Plant Soil 2003, 248, 187–197. [Google Scholar] [CrossRef]
- Nuruzzaman, M.; Lambers, H.; Bolland, M.D.A.; Veneklaas, E.J. Distribution of Carboxylates and Acid Phosphatase and Depletion of Different Phosphorus Fractions in the Rhizosphere of a Cereal and Three Grain Legumes. Plant Soil 2006, 281, 109–120. [Google Scholar] [CrossRef]
- Lambers, H.; Clements, J.C.; Nelson, M.N. How a phosphorus-acquisition strategy based on carboxylate exudation powers the success and agronomic potential of lupines (Lupinus, Fabaceae). Am. J. Bot. 2013, 100, 263–288. [Google Scholar] [CrossRef] [PubMed]
- Gerke, J. The acquisition of phosphate by higher plants: Effect of carboxylate release by the roots. A critical review. J. Plant Nutr. Soil Sci. 2015, 178, 351–364. [Google Scholar] [CrossRef]
- Wouterlood, M.; Lambers, H.; Veneklaas, E.J. Plant phosphorus status has a limited influence on the concentration of phosphorus-mobilising carboxylates in the rhizosphere of chickpea. Funct. Plant Biol. 2005, 32, 153–159. [Google Scholar] [CrossRef]
- Pearse, S.J.; Veneklaas, E.J.; Cawthray, G.; Bolland, M.D.A.; Lambers, H. Carboxylate composition of root exudates does not relate consistently to a crop species’ ability to use phosphorus from aluminium, iron or calcium phosphate sources. New Phytol. 2007, 173, 181–190. [Google Scholar] [CrossRef]
- Wang, Y.; Krogstad, T.; Clarke, J.L.; Hallama, M.; Øgaard, A.F.; Eich-Greatorex, S.; Kandeler, E.; Clarke, N. Rhizosphere Organic Anions Play a Minor Role in Improving Crop Species’ Ability to Take Up Residual Phosphorus (P) in Agricultural Soils Low in P Availability. Front. Plant Sci. 2016, 7. [Google Scholar] [CrossRef] [Green Version]
- Wang, Y.; Krogstad, T.; Clarke, N.; Øgaard, A.F.; Clarke, J.L. Impact of phosphorus on rhizosphere organic anions of wheat at different growth stages under field conditions. AoB Plants 2017, 9. [Google Scholar] [CrossRef] [Green Version]
- Pandey, R.; Meena, S.K.; Krishnapriya, V.; Ahmad, A.; Kishora, N. Root carboxylate exudation capacity under phosphorus stress does not improve grain yield in green gram. Plant Cell Rep. 2014, 33, 919–928. [Google Scholar] [CrossRef]
- Ryan, P.R.; James, R.A.; Weligama, C.; Delhaize, E.; Rattey, A.; Lewis, D.C.; Bovill, W.D.; McDonald, G.; Rathjen, T.M.; Wang, E.; et al. Can citrate efflux from roots improve phosphorus uptake by plants? Testing the hypothesis with near-isogenic lines of wheat. Physiol. Plant. 2014, 151, 230–242. [Google Scholar] [CrossRef]
- Oburger, E.; Schmidt, H. New Methods to Unravel Rhizosphere Processes. Trends Plant Sci. 2016, 21, 243–255. [Google Scholar] [CrossRef]
- Oburger, E.; Jones, D.L. Sampling root exudates—Mission impossible? Rhizosphere 2018, 6, 116–133. [Google Scholar] [CrossRef]
- Adams, M.A. Phosphatase activity and phosphorus fractions in Karri (Eucalyptus diversicolor F. Muell.) forest soils. Biol. Fertil. Soils 1992, 14, 200–204. [Google Scholar] [CrossRef]
- Clarholm, M. Microbial biomass P, labile P, and acid phosphatase activity in the humus layer of a spruce forest, after repeated additions of fertilizers. Biol. Fertil. Soils 1993, 16, 287–292. [Google Scholar] [CrossRef]
- Starnes, L.D.; Padmanabhan, P.; Sahi, S.V. Effect of P sources on growth, P accumulation and activities of phytase and acid phosphatases in two cultivars of annual ryegrass (Lolium multiflorum L.). Plant Physiol. Biochem. 2008, 46, 580–589. [Google Scholar] [CrossRef]
- Spohn, M.; Kuzyakov, Y. Distribution of microbial- and root-derived phosphatase activities in the rhizosphere depending on P availability and C allocation—Coupling soil zymography with 14C imaging. Soil Biol. Biochem. 2013, 67, 106–113. [Google Scholar] [CrossRef]
- Wasaki, J.; Sakaguchi, J.; Yamamura, T.; Ito, S.; Shinano, T.; Osaki, M.; Kandeler, E. P and N deficiency change the relative abundance and function of rhizosphere microorganisms during cluster root development of white lupin (Lupinus albus L.). Soil Sci. Plant Nutr. 2018, 64, 686–696. [Google Scholar] [CrossRef]
- Wei, X.; Hu, Y.; Razavi, B.S.; Zhou, J.; Shen, J.; Nannipieri, P.; Wu, J.; Ge, T. Rare taxa of alkaline phosphomonoesterase-harboring microorganisms mediate soil phosphorus mineralization. Soil Biol. Biochem. 2019, 131, 62–70. [Google Scholar] [CrossRef]
- Cordell, D.; Drangert, J.-O.; White, S. The story of phosphorus: Global food security and food for thought. Glob. Environ. Chang. 2009, 19, 292–305. [Google Scholar] [CrossRef]
- Maltais-Landry, G. Legumes have a greater effect on rhizosphere properties (pH, organic acids and enzyme activity) but a smaller impact on soil P compared to other cover crops. Plant Soil 2015, 394, 139–154. [Google Scholar] [CrossRef]
- Pang, J.; Ryan, M.H.; Lambers, H.; Siddique, K.H. Phosphorus acquisition and utilisation in crop legumes under global change. Curr. Opin. Plant Biol. 2018, 45, 248–254. [Google Scholar] [CrossRef]
- Marklein, A.R.; Houlton, B.Z. Nitrogen inputs accelerate phosphorus cycling rates across a wide variety of terrestrial ecosystems. New Phytol. 2012, 193, 696–704. [Google Scholar] [CrossRef] [PubMed]
- Zang, H.; Blagodatskaya, E.; Wang, J.; Xu, X.; Kuzyakov, Y. Nitrogen fertilization increases rhizodeposit incorporation into microbial biomass and reduces soil organic matter losses. Biol. Fertil. Soils 2017, 53, 419–429. [Google Scholar] [CrossRef]
- Chen, H.; Chen, M.; Li, D.; Mao, Q.; Zhang, W.; Mo, J. Responses of soil phosphorus availability to nitrogen addition in a legume and a non-legume plantation. Geoderma 2018, 322, 12–18. [Google Scholar] [CrossRef]
- Olander, L.P.; Vitousek, P.M. Regulation of soil phosphatase and chitinase activityby N and P availability. Biogeochemistry 2000, 49, 175–191. [Google Scholar] [CrossRef]
- Vitousek, P.M.; Porder, S.; Houlton, B.Z.; Chadwick, O.A. Terrestrial phosphorus limitation: mechanisms, implications, and nitrogen–phosphorus interactions. Ecol. Appl. 2010, 20, 5–15. [Google Scholar] [CrossRef] [Green Version]
- Deng, Q.; Hui, D.; Dennis, S.; Reddy, K.C. Responses of terrestrial ecosystem phosphorus cycling to nitrogen addition: A meta-analysis. Glob. Ecol. Biogeogr. 2017, 26, 713–728. [Google Scholar] [CrossRef]
- Liu, E.; Yan, C.; Mei, X.; He, W.; Bing, S.H.; Ding, L.; Liu, Q.; Liu, S.; Fan, T. Long-term effect of chemical fertilizer, straw, and manure on soil chemical and biological properties in northwest China. Geoderma 2010, 158, 173–180. [Google Scholar] [CrossRef]
- Yang, K.; Zhu, J.; Gu, J.; Yu, L.; Wang, Z. Changes in soil phosphorus fractions after 9 years of continuous nitrogen addition in a Larix gmelinii plantation. Ann. For. Sci. 2015, 72, 435–442. [Google Scholar] [CrossRef]
- Fan, Y.; Lin, F.; Yang, L.; Zhong, X.; Wang, M.; Zhou, J.; Chen, Y.; Yang, Y. Decreased soil organic P fraction associated with ectomycorrhizal fungal activity to meet increased P demand under N application in a subtropical forest ecosystem. Biol. Fertil. Soils 2018, 54, 149–161. [Google Scholar] [CrossRef]
- Mat Hassan, H.; Marschner, P.; McNeill, A.; Tang, C. Growth, P uptake in grain legumes and changes in rhizosphere soil P pools. Biol. Fertil. Soils 2012, 48, 151–159. [Google Scholar] [CrossRef]
- Sugihara, S.; Tomita, Y.; Nishigaki, T.; Kilasara, M.; Wasaki, J.; Funakawa, S. Effects of different phosphorus-efficient legumes and soil texture on fractionated rhizosphere soil phosphorus of strongly weathered soils. Biol. Fertil. Soils 2016, 52, 367–376. [Google Scholar] [CrossRef]
- Pinxterhuis, I.B.; Edwards, J.P. Comparing nitrogen management on dairy farms – Canterbury case studies. J. N. Z. Grassl. 2018, 80, 201–206. [Google Scholar] [CrossRef]
- Kidd, D.R.; Ryan, M.H.; Haling, R.E.; Lambers, H.; Sandral, G.A.; Yang, Z.; Culvenor, R.A.; Cawthray, G.R.; Stefanski, A.; Simpson, R.J. Rhizosphere carboxylates and morphological root traits in pasture legumes and grasses. Plant Soil 2016, 402, 77–89. [Google Scholar] [CrossRef]
- Egle, K.; Römer, W.; Keller, H. Exudation of low molecular weight organic acids by Lupinus albus L., Lupinus angustifolius L. and Lupinus luteus L. as affected by phosphorus supply. Agronomie 2003, 23, 511–518. [Google Scholar] [CrossRef]
- Wang, B.; Shen, J.; Tang, C.; Rengel, Z. Root Morphology, Proton Release, and Carboxylate Exudation in Lupin in Response to Phosphorus Deficiency. J. Plant Nutr. 2008, 31, 557–570. [Google Scholar] [CrossRef]
- Funayama-Noguchi, S.; Noguchi, K.; Terashima, I. Comparison of the response to phosphorus deficiency in two lupin species, Lupinus albus and L. angustifolius, with contrasting root morphology. Plant Cell Environ. 2015, 38, 399–410. [Google Scholar] [CrossRef]
- Rietra, R.P.J.J.; Heinen, M.; Dimkpa, C.O.; Bindraban, P.S. Effects of Nutrient Antagonism and Synergism on Yield and Fertilizer Use Efficiency. Commun. Soil Sci. Plant Anal. 2017, 48, 1895–1920. [Google Scholar] [CrossRef] [Green Version]
- Marschner, H.; Römheld, V. In vivo Measurement of Root-induced pH Changes at the Soil-Root Interface: Effect of Plant Species and Nitrogen Source. Z. Für Pflanzenphysiol. 1983, 111, 241–251. [Google Scholar] [CrossRef]
- Hinsinger, P.; Plassard, C.; Tang, C.; Jaillard, B. Origins of root-mediated pH changes in the rhizosphere and their responses to environmental constraints: A review. Plant Soil 2003, 248, 43–59. [Google Scholar] [CrossRef]
- Hinsinger, P.; Betencourt, E.; Bernard, L.; Brauman, A.; Plassard, C.; Shen, J.; Tang, X.; Zhang, F. P for Two, Sharing a Scarce Resource: Soil Phosphorus Acquisition in the Rhizosphere of Intercropped Species. Plant Physiol. 2011, 156, 1078–1086. [Google Scholar] [CrossRef] [Green Version]
- Li, H.; Shen, J.; Zhang, F.; Clairotte, M.; Drevon, J.J.; Le Cadre, E.; Hinsinger, P. Dynamics of phosphorus fractions in the rhizosphere of common bean (Phaseolus vulgaris L.) and durum wheat (Triticum turgidum durum L.) grown in monocropping and intercropping systems. Plant Soil 2008, 312, 139–150. [Google Scholar] [CrossRef]
- Kreuzeder, A.; Santner, J.; Scharsching, V.; Oburger, E.; Hoefer, C.; Hann, S.; Wenzel, W.W. In situ observation of localized, sub-mm scale changes of phosphorus biogeochemistry in the rhizosphere. Plant Soil 2018, 424, 573–589. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tang, C.; Barton, L.; McLay, C.D.A. A comparison of proton excretion of twelve pasture legumes grown in nutrient solution. Aust. J. Exp. Agric. 1997, 37, 563–570. [Google Scholar] [CrossRef]
- Tang, C.; Rengel, Z. Role of Plant Cation/Anion Uptake Ratio in Soil Acidification. In Handbook of Soil Acidity; Rengel, Z., Ed.; CRC Press: Boca Raton, FL, USA, 2003; ISBN 978-0-8247-0890-0. [Google Scholar]
- Dodd, R.J.; McDowell, R.W.; Condron, L.M. Manipulation of fertiliser regimes in phosphorus enriched soils can reduce phosphorus loss to leachate through an increase in pasture and microbial biomass production. Agric. Ecosyst. Environ. 2014, 185, 65–76. [Google Scholar] [CrossRef]
- Heuck, C.; Weig, A.; Spohn, M. Soil microbial biomass C:N:P stoichiometry and microbial use of organic phosphorus. Soil Biol. Biochem. 2015, 85, 119–129. [Google Scholar] [CrossRef]
- Spohn, M.; Widdig, M. Turnover of carbon and phosphorus in the microbial biomass depending on phosphorus availability. Soil Biol. Biochem. 2017, 113, 53–59. [Google Scholar] [CrossRef]
- Skujins, J.J.; Braal, L.; McLaren, A.D. Characterization of Phosphatase in a Terrestrial Soil Sterilized with an Electron Beam. Enzymol. Neth. Superseded Mol. Cell Biochem. 1962, 25, 125–133. [Google Scholar]
- Nannipieri, P.; Kandeler, E.; Ruggiero, P. Enzyme activities and microbiological and biochemical processes in soil. In Enzymes in the Environment; Burns, R.G., Dick, R.P., Eds.; Marcel Dekker: New York, NY, USA, 2002; pp. 1–33. [Google Scholar]
- Badri, D.V.; Vivanco, J.M. Regulation and function of root exudates. Plant Cell Environ. 2009, 32, 666–681. [Google Scholar] [CrossRef]
- Finkel, O.M.; Salas-González, I.; Castrillo, G.; Spaepen, S.; Law, T.F.; Teixeira, P.J.P.L.; Jones, C.D.; Dangl, J.L. The effects of soil phosphorus content on plant microbiota are driven by the plant phosphate starvation response. PLoS Biol. 2019, 17, e3000534. [Google Scholar] [CrossRef] [Green Version]
- Ragot, S.A.; Kertesz, M.A.; Mészáros, É.; Frossard, E.; Bünemann, E.K. Soil phoD and phoX alkaline phosphatase gene diversity responds to multiple environmental factors. FEMS Microbiol. Ecol. 2017, 93. [Google Scholar] [CrossRef] [Green Version]
- Wu, L.; Kobayashi, Y.; Wasaki, J.; Koyama, H. Organic acid excretion from roots: a plant mechanism for enhancing phosphorus acquisition, enhancing aluminum tolerance, and recruiting beneficial rhizobacteria. Soil Sci. Plant Nutr. 2018, 64, 697–704. [Google Scholar] [CrossRef]
- Margenot, A.J.; Nakayama, Y.; Parikh, S.J. Methodological recommendations for optimizing assays of enzyme activities in soil samples. Soil Biol. Biochem. 2018, 125, 350–360. [Google Scholar] [CrossRef]
- Neumann, G.; Römheld, V. Root excretion of carboxylic acids and protons in phosphorus-deficient plants. Plant Soil 1999, 211, 121–130. [Google Scholar] [CrossRef]
- Bolan, N.S.; Naidu, R.; Mahimairaja, S.; Baskaran, S. Influence of low-molecular-weight organic acids on the solubilization of phosphates. Biol. Fertil. Soils 1994, 18, 311–319. [Google Scholar] [CrossRef]
- Earl, K.D.; Syers, J.K.; McLaughlin, J.R. Origin of the Effects of Citrate, Tartrate, and Acetate on Phosphate Sorption by Soils and Synthetic Gels 1. Soil Sci. Soc. Am. J. 1979, 43, 674–678. [Google Scholar] [CrossRef]
- Pearse, S.J.; Veneklaas, E.J.; Cawthray, G.R.; Bolland, M.D.A.; Lambers, H. Carboxylate release of wheat, canola and 11 grain legume species as affected by phosphorus status. Plant Soil 2006, 288, 127–139. [Google Scholar] [CrossRef]
- Condron, L.M.; Black, A.; Wakelin, S.A. Effects of long-term fertiliser inputs on the quantities of organic carbon in a soil profile under irrigated grazed pasture. N. Z. J. Agric. Res. 2012, 55, 161–164. [Google Scholar] [CrossRef] [Green Version]
- Wakelin, S.A.; Macdonald, L.M.; O’Callaghan, M.; Forrester, S.T.; Condron, L.M. Soil functional resistance and stability are linked to different ecosystem properties. Austral Ecol. 2014, 39, 522–531. [Google Scholar] [CrossRef]
- Wakelin, S.A.; Condron, L.M.; Gerard, E.; Dignam, B.E.A.; Black, A.; O’Callaghan, M. Long-term P fertilisation of pasture soil did not increase soil organic matter stocks but increased microbial biomass and activity. Biol. Fertil. Soils 2017, 53, 511–521. [Google Scholar] [CrossRef]
- Spohn, M.; Schleuss, P.-M. Addition of inorganic phosphorus to soil leads to desorption of organic compounds and thus to increased soil respiration. Soil Biol. Biochem. 2019, 130, 220–226. [Google Scholar] [CrossRef]
- Nannipieri, P.; Giagnoni, L.; Landi, L.; Renella, G. Role of Phosphatase Enzymes in Soil. In Phosphorus in Action: Biological Processes in Soil Phosphorus Cycling; Bünemann, E., Oberson, A., Frossard, E., Eds.; Soil Biology; Springer: Berlin/Heidelberg, Germany, 2011; pp. 215–243. ISBN 978-3-642-15271-9. [Google Scholar]
- Jakobsen, I.; Leggett, M.E.; Richardson, A.E. Rhizosphere microorganisms and plant phosphorus uptake. Phosphorus Agric. Environ. 2005, agronomymonogra, 437–494. [Google Scholar] [CrossRef] [Green Version]
- Blackwell, M.S.A.; Brookes, P.C.; de la Fuente-Martinez, N.; Gordon, H.; Murray, P.J.; Snars, K.E.; Williams, J.K.; Bol, R.; Haygarth, P.M. Chapter 1 - Phosphorus solubilization and potential transfer to surface waters from the soil microbial biomass following drying–rewetting and freezing–thawing. In Advances in Agronomy; Sparks, D.L., Ed.; Advances in Agronomy v106; Academic Press: Cambridge, MA, USA, 2010; Volume 106, pp. 1–35. [Google Scholar]
- Li, G.-X.; Wu, X.-Q.; Ye, J.-R.; Yang, H.-C. Characteristics of Organic Acid Secretion Associated with the Interaction between Burkholderia multivorans WS-FJ9 and Poplar Root System. BioMed Res. Int. 2018, 2018, 1–12. [Google Scholar] [CrossRef] [Green Version]
- Vu, D.T.; Tang, C.; Armstrong, R.D. Changes and availability of P fractions following 65 years of P application to a calcareous soil in a Mediterranean climate. Plant Soil 2008, 304, 21–33. [Google Scholar] [CrossRef]
- Chen, C.R.; Condron, L.M.; Davis, M.R.; Sherlock, R.R. Seasonal changes in soil phosphorus and associated microbial properties under adjacent grassland and forest in New Zealand. For. Ecol. Manag. 2003, 177, 539–557. [Google Scholar] [CrossRef]
- George, T.S.; Giles, C.D.; Menezes-Blackburn, D.; Condron, L.M.; Gama-Rodrigues, A.C.; Jaisi, D.; Lang, F.; Neal, A.L.; Stutter, M.I.; Almeida, D.S.; et al. Organic phosphorus in the terrestrial environment: a perspective on the state of the art and future priorities. Plant Soil 2018, 427, 191–208. [Google Scholar] [CrossRef]
- McDowell, R.W.; Condron, L.M. Phosphorus and the Winchmore trials: review and lessons learnt. N. Z. J. Agric. Res. 2012, 55, 119–132. [Google Scholar] [CrossRef]
- Condron, L.M.; Goh, K.M. Effects of long-term phosphatic fertilizer applications on amounts and forms of phosphorus in soils under irrigated pasture in New Zealand. J. Soil Sci. 1989, 40, 383–395. [Google Scholar] [CrossRef]
- Condron, L.M.; Cornforth, I.S.; Davis, M.R.; Newman, R.H. Influence of conifers on the forms of phosphorus in selected New Zealand grassland soils. Biol. Fertil. Soils 1996, 21, 37–42. [Google Scholar] [CrossRef]
- Jin, J.; Tang, C.; Armstrong, R.; Butterly, C.; Sale, P. Elevated CO2 temporally enhances phosphorus immobilization in the rhizosphere of wheat and chickpea. Plant Soil 2013, 368, 315–328. [Google Scholar] [CrossRef]
- Saeed, M.T.; Wahid, M.A.; Saleem, M.F.; Aziz, T. Enhancing Phosphorus use Efficiency by Supplementing through Soil Applications and Seed Phosphorus Reserves in Maize (Zea mays). Int. J. Agric. Biol. 2017, 19, 7. [Google Scholar]
- Julia, C.C.; Rose, T.J.; Pariasca-Tanaka, J.; Jeong, K.; Matsuda, T.; Wissuwa, M. Phosphorus uptake commences at the earliest stages of seedling development in rice. J. Exp. Bot. 2018, 69, 5233–5240. [Google Scholar] [CrossRef] [PubMed]
- Pang, J.; Yang, J.; Lambers, H.; Tibbett, M.; Siddique, K.H.M.; Ryan, M.H. Physiological and morphological adaptations of herbaceous perennial legumes allow differential access to sources of varyingly soluble phosphate. Physiol. Plant. 2015, 154, 511–525. [Google Scholar] [CrossRef] [PubMed]
- Wu, S.; Feng, X.; Wittmeier, A. Microwave Digestion of Plant and Grain Reference Materials in Nitric Acid or a Mixture of Nitric Acid or a Mixture of Nitric Acid and Hydrogen Peroxide for the Determination of Multi-elements by Inductively Coupled Plasma Mass Spectrometry. J. Anal. At. Spectrom. 1997, 12, 797–806. [Google Scholar] [CrossRef]
- Brookes, P.C.; Powlson, D.S.; Jenkinson, D.S. Measurement of microbial biomass phosphorus in soil. Soil Biol. Biochem. 1982, 14, 319–329. [Google Scholar] [CrossRef]
- Morel, C.; Tiessen, H.; Stewart, J.W.B. Correction for P-sorption in the measurement of soil microbial biomass P by CHCl3 fumigation. Soil Biol. Biochem. 1996, 28, 1699–1706. [Google Scholar] [CrossRef]
- Murphy, J.; Riley, J.P. A modified single solution method for the determination of phosphate in natural waters. Anal. Chim. Acta 1962, 27, 31–36. [Google Scholar] [CrossRef]
- Cheng, L.; Tang, X.; Vance, C.P.; White, P.J.; Zhang, F.; Shen, J. Interactions between light intensity and phosphorus nutrition affect the phosphate-mining capacity of white lupin (Lupinus albus L.). J. Exp. Bot. 2014, 65, 2995–3003. [Google Scholar] [CrossRef] [Green Version]
- Wang, Y.-L.; Almvik, M.; Clarke, N.; Eich-Greatorex, S.; Øgaard, A.F.; Krogstad, T.; Lambers, H.; Clarke, J.L. Contrasting responses of root morphology and root-exuded organic acids to low phosphorus availability in three important food crops with divergent root traits. AoB Plants 2015, 7, plv097. [Google Scholar] [CrossRef]
- Kidd, D.R.; Ryan, M.H.; Hahne, D.; Haling, R.E.; Lambers, H.; Sandral, G.A.; Simpson, R.J.; Cawthray, G.R. The carboxylate composition of rhizosheath and root exudates from twelve species of grassland and crop legumes with special reference to the occurrence of citramalate. Plant Soil 2018, 424, 389–403. [Google Scholar] [CrossRef] [Green Version]
- Condron, L.M.; Newman, S. Revisiting the fundamentals of phosphorus fractionation of sediments and soils. J. Soils Sediments 2011, 11, 830–840. [Google Scholar] [CrossRef]
- Yang, X.; Post, W.M. Phosphorus transformations as a function of pedogenesis: A synthesis of soil phosphorus data using Hedley fractionation method. Biogeosciences 2011, 8, 2907–2916. [Google Scholar] [CrossRef] [Green Version]
- Olsen, S.R.; Sommers, L.E. Determination of available phosphorus. In Methods of soil analysis. Part 2. Chemical and Microbiological Properties.; Page, A.L., Miller, R.H., Keeney, D.R., Eds.; Agronomy Monograph; American Society of Agronomy, Soil Science Society of America: Madison, WI, USA, 1982; pp. 403–430. [Google Scholar]
- Dick, W.A.; Tabatabai, M.A. An Alkaline Oxidation Method for Determination of Total Phosphorus in Soils 1. Soil Sci. Soc. Am. J. 1977, 41, 511–514. [Google Scholar] [CrossRef]
- He, Z.; Honeycutt, C.W. A Modified Molybdenum Blue Method for Orthophosphate Determination Suitable for Investigating Enzymatic Hydrolysis of Organic Phosphates. Commun. Soil Sci. Plant Anal. 2005, 36, 1373–1383. [Google Scholar] [CrossRef]
- do Nascimento, C.A.C.; Pagliari, P.H.; Schmitt, D.; He, Z.; Waldrip, H. Phosphorus Concentrations in Sequentially Fractionated Soil Samples as Affected by Digestion Methods. Sci. Rep. 2015, 5, 17967. [Google Scholar] [CrossRef] [Green Version]



| Root Biomass | Shoot Biomass | Root P | Shoot P | Plant P Content | |
|---|---|---|---|---|---|
| Blue Lupin | |||||
| Control | 1.2 ± 0.1 b 1 | 2.2 ± 0.1 b | 1.0 ± 0.0 b | 1.1 ± 0.0 c | 3.54 ± 0.25 c |
| P | 1.7 ± 0.2 a | 2.4 ± 0.1 a | 1.1 ± 0.1 a | 1.3 ± 0.1 b | 4.84 ± 0.69 a |
| N | 1.4 ± 0.3 b | 2.1 ± 0.1 b | 0.9 ± 0.0 c | 1.0 ± 0.1 d | 3.26 ± 0.49 c |
| NP | 1.5 ± 0.2 a | 2.6 ± 0.2 a | 1.0 ± 0.1 b | 1.4 ± 0.1 a | 4.67 ± 0.70 b |
| Wheat | |||||
| Control | 0.9 ± 0.0 c | 1.1 ± 0.0 c | 0.8 ± 0.1 b | 0.4 ± 0.0 c | 1.25 ± 0.08 c |
| P | 1.9 ± 0.1 b | 1.4 ± 0.0 b | 1.1 ± 0.0 a | 0.8 ± 0.0 a | 3.21 ± 0.09 a |
| N | 0.9 ± 0.1 c | 1.1 ± 0.1 c | 0.8 ± 0.1 b | 0.4 ± 0.0 c | 1.28 ± 0.13 c |
| NP | 2.2 ± 0.2 a | 2.0 ± 0.2 a | 0.9 ± 0.1 b | 0.6 ± 0.0 b | 3.08 ± 0.24 b |
| Ryegrass | |||||
| Control | 0.4 ± 0.0 c | 0.2 ± 0.0 c | 1.2 ± 0.0 b | 0.7 ± 0.0 b | 0.58 ± 0.04 c |
| P | 1.0 ± 0.1 b | 0.8 ± 0.1 b | 1.3 ± 0.0 a | 1.1 ± 0.0 a | 1.65 ± 0.13 b |
| N | 0.4 ± 0.2 c | 0.3 ± 0.1 c | 1.2 ± 0.1 b | 0.7 ± 0.0 b | 0.73 ± 0.27 c |
| NP | 1.9 ± 0.2 a | 1.1 ± 0.0 a | 1.0 ± 0.1 c | 0.6 ± 0.0 c | 2.52 ± 0.25 a |
| White clover | |||||
| Control | 0.2 ± 0.0 1 c | 0.2 ± 0.0 b | 1.6 ± 0.1 a | 0.7 ± 0.0 b | 0.38 ± 0.03 b |
| P | 1.0 ± 0.1 b | 0.7 ± 0.1 a | 1.5 ± 0.0 b | 0.9 ± 0.0 a | 2.03 ± 0.17 a |
| N | 0.3 ± 0.0 c | 0.2 ± 0.0 b | 1.5 ± 0.0 b | 0.7 ± 0.0 b | 0.46 ± 0.08 b |
| NP | 1.5 ± 0.1 a | 0.9 ± 0.0 a | 1.3 ± 0.0 c | 0.8 ± 0.1 a | 2.38 ± 0.39 a |
| Soil pH | Total C | Total N | Total P | Olsen P | Exchangeable Potassium | Exchangeable Al | Soil Texture (%) | ||
|---|---|---|---|---|---|---|---|---|---|
| g C kg−1 soil | g N kg−1 soil | mg P kg−1 soil | mg P kg−1 soil | me/100 g soil | mg kg−1 soil | Sand | Silt | Clay | |
| 5.4 | 44 | 3.9 | 1236 | 4 | 0.16 | 7.2 | 12.6 | 37.8 | 49.6 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Touhami, D.; McDowell, R.W.; Condron, L.M. Role of Organic Anions and Phosphatase Enzymes in Phosphorus Acquisition in the Rhizospheres of Legumes and Grasses Grown in a Low Phosphorus Pasture Soil. Plants 2020, 9, 1185. https://doi.org/10.3390/plants9091185
Touhami D, McDowell RW, Condron LM. Role of Organic Anions and Phosphatase Enzymes in Phosphorus Acquisition in the Rhizospheres of Legumes and Grasses Grown in a Low Phosphorus Pasture Soil. Plants. 2020; 9(9):1185. https://doi.org/10.3390/plants9091185
Chicago/Turabian StyleTouhami, Driss, Richard W. McDowell, and Leo M. Condron. 2020. "Role of Organic Anions and Phosphatase Enzymes in Phosphorus Acquisition in the Rhizospheres of Legumes and Grasses Grown in a Low Phosphorus Pasture Soil" Plants 9, no. 9: 1185. https://doi.org/10.3390/plants9091185







