Live Imaging of Phosphate Levels in Arabidopsis Root Cells Expressing a FRET-Based Phosphate Sensor
Abstract
1. Introduction
2. Results
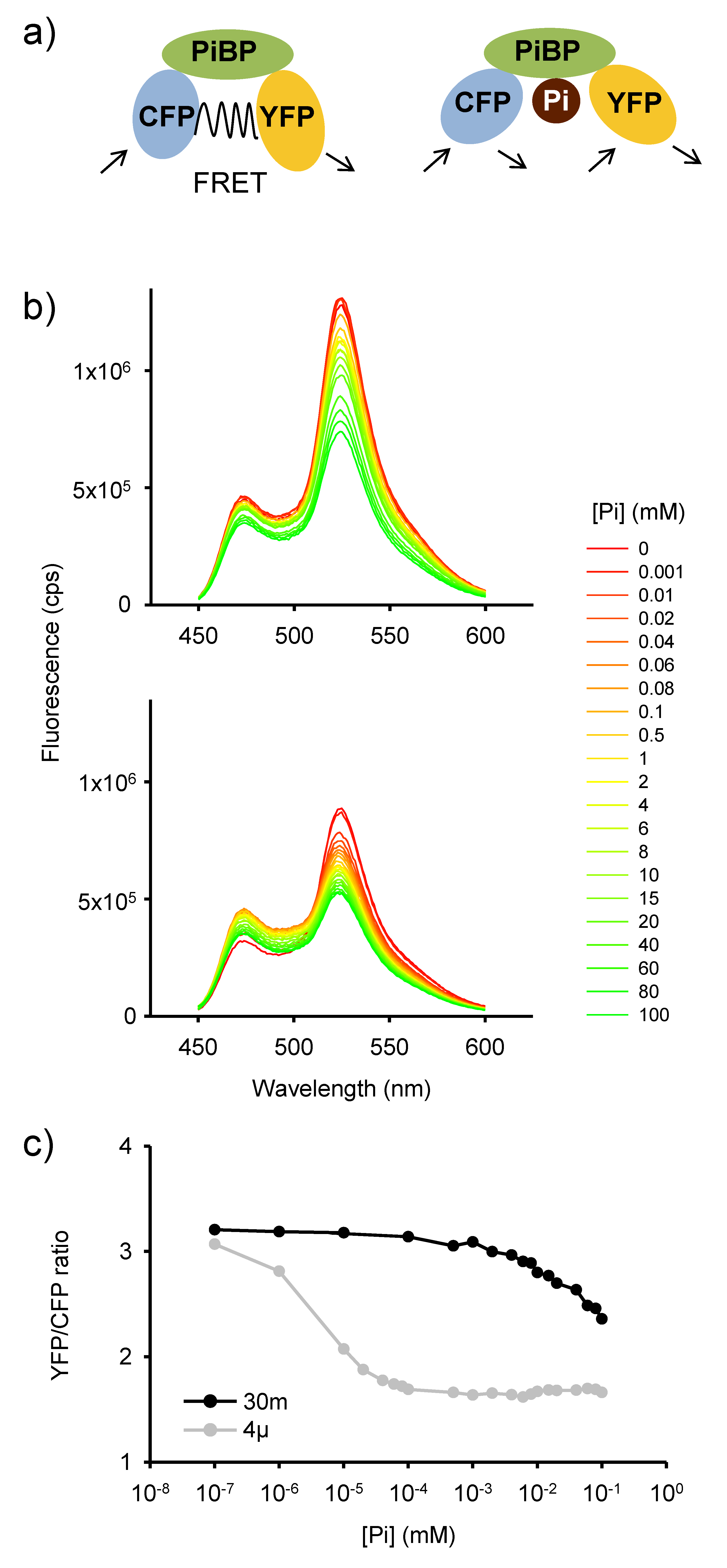
2.1. In Vitro Analysis of Pi-Dependent FRET Response in Purified FLIPPi Sensors
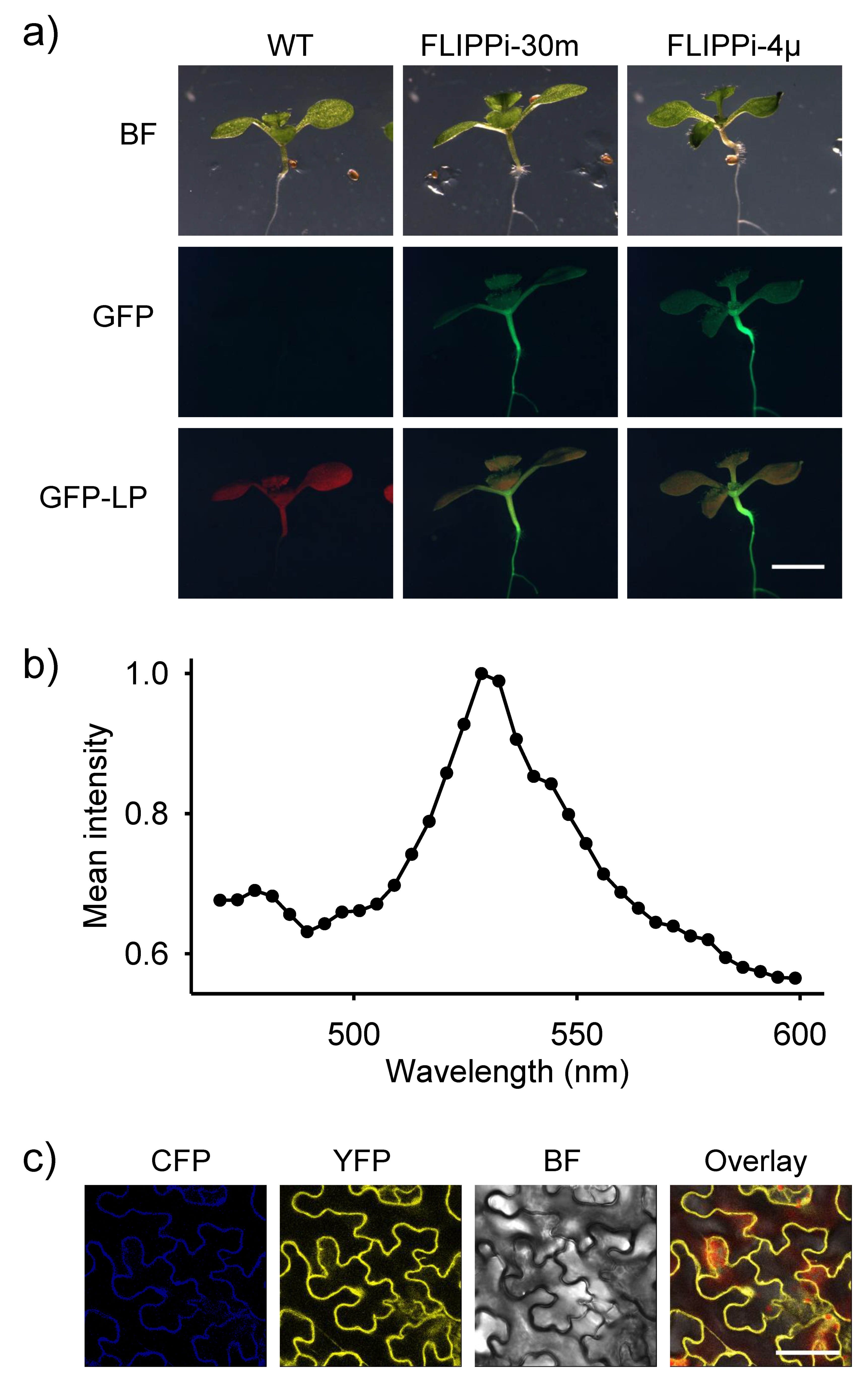
2.2. Expression of FLIPPi Sensors in Wild-Type Arabidopsis
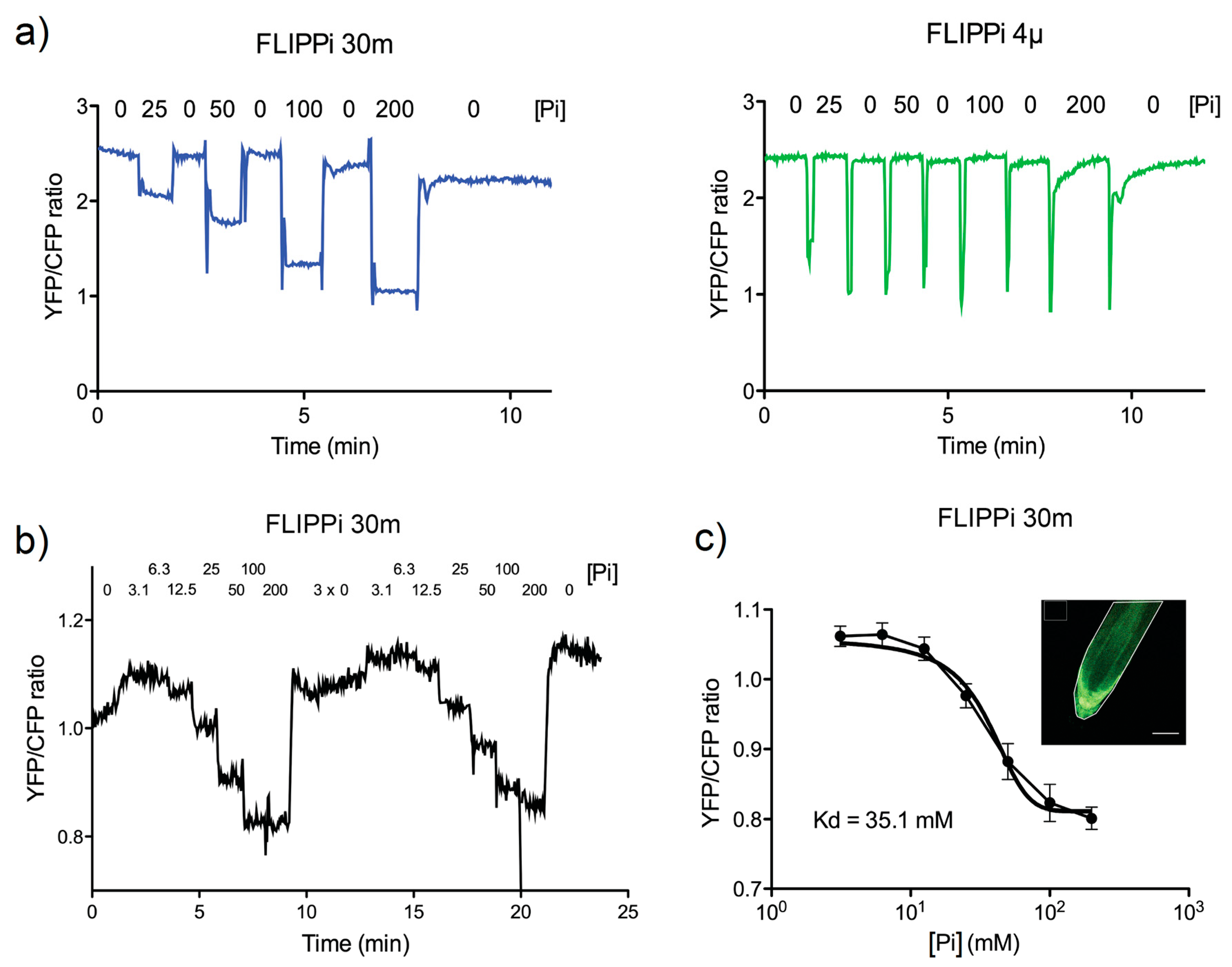
2.3. Live Imaging of FRET Response to Pi Buffer Perfusions in Arabidopsis Roots
3. Discussion
4. Conclusions
5. Materials and Methods
5.1. Plasmid Construction and Plant Transformation
5.2. In Vitro Analysis of FLIPPi Proteins
5.3. Plant Growth Conditions
5.4. Live Imaging and FRET Analysis with Pi Buffer Perfusions
5.5. Data Analysis
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Marschner, H. Mineral Nutrition of Higher Plants, 2nd ed.; Rendig, V.V., Taylor, H.M., Eds.; Academic Press: London, UK; San Diego, CA, USA, 1995. [Google Scholar]
- Desmoulin, F.; Cozzone, P.J.; Canioni, P. Phosphorus-31 nuclear-magnetic-resonance study of phosphorylated metabolites compartmentation, intracellular pH and phosphorylation state during normoxia, hypoxia and ethanol perfusion, in the perfused rat liver. Eur. J. Biochem. 1987, 162, 151–159. [Google Scholar] [CrossRef] [PubMed]
- López-Arredondo, D.L.; Leyva-González, M.A.; González-Morales, S.I.; López-Bucio, J.; Herrera-Estrella, L. Phosphate nutrition: Improving low-phosphate tolerance in crops. Annu. Rev. Plant Biol. 2014, 65, 95–123. [Google Scholar] [CrossRef] [PubMed]
- Raghothama, K.G. Phosphate Acquisition. Annu. Rev. Plant Physiol. Plant Mol. Biol. 1999, 50, 665–693. [Google Scholar] [CrossRef] [PubMed]
- Schachtman, D.; Reid, R.; Ayling, S. Phosphorus Uptake by Plants: From Soil to Cell. Plant Physiol. 1998, 116, 447–453. [Google Scholar] [CrossRef]
- Franco-Zorrilla, J.M.; González, E.; Bustos, R.; Linhares, F.; Leyva, A.; Paz-Ares, J. The transcriptional control of plant responses to phosphate limitation. J. Exp. Bot. 2004, 55, 285–293. [Google Scholar] [CrossRef]
- Zhang, Z.; Liao, H.; Lucas, W.J. Molecular mechanisms underlying phosphate sensing, signaling, and adaptation in plants. J. Integr. Plant Biol. 2014, 56, 192–220. [Google Scholar] [CrossRef]
- Hammond, J.P.; Broadley, M.R.; White, P.J.; King, G.J.; Bowen, H.C.; Hayden, R.; Meacham, M.C.; Mead, A.; Overs, T.; Spracklen, W.P.; et al. Shoot yield drives phosphorus use efficiency in Brassica oleracea and correlates with root architecture traits. J. Exp. Bot. 2009, 60, 1953–1968. [Google Scholar] [CrossRef]
- Heuer, S.; Gaxiola, R.; Schilling, R.; Herrera-Estrella, L.; López-Arredondo, D.; Wissuwa, M.; Delhaize, E.; Rouached, H. Improving phosphorus use efficiency: A complex trait with emerging opportunities. Plant J. 2017, 90, 868–885. [Google Scholar] [CrossRef]
- Chen, Z.; Truong, T.; Ai, H. Illuminating Brain Activities with Fluorescent Protein-Based Biosensors. Chemosensors 2017, 5, 32. [Google Scholar] [CrossRef]
- Pendin, D.; Greotti, E.; Lefkimmiatis, K.; Pozzan, T. Exploring cells with targeted biosensors. J. Gen. Physiol. 2017, 149, 1–36. [Google Scholar] [CrossRef]
- Deuschle, K.; Fehr, M.; Hilpert, M.; Lager, I.; Lalonde, S.; Looger, L.L.; Okumoto, S.; Persson, J.; Schmidt, A.; Frommer, W.B. Genetically encoded sensors for metabolites. Cytometry A 2005, 64, 3–9. [Google Scholar] [CrossRef] [PubMed]
- Germond, A.; Fujita, H.; Ichimura, T.; Watanabe, T.M. Design and development of genetically encoded fluorescent sensors to monitor intracellular chemical and physical parameters. Biophys. Rev. 2016, 8, 121–138. [Google Scholar] [CrossRef] [PubMed]
- Gjetting, S.K.; Schulz, A.; Fuglsang, A.T. Perspectives for using genetically encoded fluorescent biosensors in plants. Front. Plant Sci. 2013, 4, 234. [Google Scholar] [CrossRef] [PubMed]
- Deuschle, K.; Chaudhuri, B.; Okumoto, S.; Lager, I.; Lalonde, S.; Frommer, W.B. Rapid metabolism of glucose detected with FRET glucose nanosensors in epidermal cells and intact roots of Arabidopsis RNA-silencing mutants. Plant Cell 2006, 18, 2314–2325. [Google Scholar] [CrossRef] [PubMed]
- Chaudhuri, B.; Hörmann, F.; Lalonde, S.; Brady, S.M.; Orlando, D.A.; Benfey, P.; Frommer, W.B. Protonophore- and pH-insensitive glucose and sucrose accumulation detected by FRET nanosensors in Arabidopsis root tips. Plant J. 2008, 56, 948–962. [Google Scholar] [CrossRef]
- Chaudhuri, B.; Hörmann, F.; Frommer, W.B. Dynamic imaging of glucose flux impedance using FRET sensors in wild-type Arabidopsis plants. J. Exp. Bot. 2011, 62, 2411–2417. [Google Scholar] [CrossRef]
- Yang, H.; Bogner, M.; Stierhof, Y.-D.; Ludewig, U. H-independent glutamine transport in plant root tips. PLoS ONE 2010, 5, e8917. [Google Scholar] [CrossRef]
- Gjetting, K.S.K.; Ytting, C.K.; Schulz, A.; Fuglsang, A.T. Live imaging of intra- and extracellular pH in plants using pHusion, a novel genetically encoded biosensor. J. Exp. Bot. 2012, 63, 3207–3218. [Google Scholar] [CrossRef]
- Gu, H.; Lalonde, S.; Okumoto, S.; Looger, L.L.; Scharff-Poulsen, A.M.; Grossman, A.R.; Kossmann, J.; Jakobsen, I.; Frommer, W.B. A novel analytical method for in vivo phosphate tracking. FEBS Lett. 2006, 580, 5885–5893. [Google Scholar] [CrossRef]
- Mukherjee, P.; Banerjee, S.; Wheeler, A.; Ratliff, L.A.; Irigoyen, S.; Garcia, L.R.; Lockless, S.W.; Versaw, W.K. Live Imaging of Inorganic Phosphate in Plants with Cellular and Subcellular Resolution. Plant Physiol. 2015, 167, 628–638. [Google Scholar] [CrossRef]
- Banerjee, S.; Versaw, W.K.; Garcia, L.R. Imaging Cellular Inorganic Phosphate in Caenorhabditis elegans Using a Genetically Encoded FRET-Based Biosensor. PLoS ONE 2015, 10, e0141128. [Google Scholar] [CrossRef] [PubMed]
- Rebeille, F.; Bligny, R.; Martin, J.B.; Douce, R. Relationship between the cytoplasm and the vacuole phosphate pool in Acer pseudoplatanus cells. Arch. Biochem. Biophys. 1983, 225, 143–148. [Google Scholar] [CrossRef]
- Mimura, T.; Sakano, K.; Shimmen, T. Studies on the distribution, re-translocation and homeostasis of inorganic phosphate in barley leaves. Plant Cell Environ. 1996, 19, 311–320. [Google Scholar] [CrossRef]
- Pratt, J.; Boisson, A.-M.; Gout, E.; Bligny, R.; Douce, R.; Aubert, S. Phosphate (Pi) starvation effect on the cytosolic Pi concentration and Pi exchanges across the tonoplast in plant cells: An in vivo 31P-nuclear magnetic resonance study using methylphosphonate as a Pi analog. Plant Physiol. 2009, 151, 1646–1657. [Google Scholar] [CrossRef]
- Earley, K.W.; Haag, J.R.; Pontes, O.; Opper, K.; Juehne, T.; Song, K.; Pikaard, C.S. Gateway-compatible vectors for plant functional genomics and proteomics. Plant J. 2006, 45, 616–629. [Google Scholar] [CrossRef]
- Clough, S.J.; Bent, A.F. Floral dip: A simplified method for Agrobacterium-mediated transformation of Arabidopsis thaliana. Plant J. 1998, 16, 735–743. [Google Scholar] [CrossRef]
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Assunção, A.G.L.; Gjetting, S.K.; Hansen, M.; Fuglsang, A.T.; Schulz, A. Live Imaging of Phosphate Levels in Arabidopsis Root Cells Expressing a FRET-Based Phosphate Sensor. Plants 2020, 9, 1310. https://doi.org/10.3390/plants9101310
Assunção AGL, Gjetting SK, Hansen M, Fuglsang AT, Schulz A. Live Imaging of Phosphate Levels in Arabidopsis Root Cells Expressing a FRET-Based Phosphate Sensor. Plants. 2020; 9(10):1310. https://doi.org/10.3390/plants9101310
Chicago/Turabian StyleAssunção, Ana G. L., Sisse K. Gjetting, Michael Hansen, Anja T. Fuglsang, and Alexander Schulz. 2020. "Live Imaging of Phosphate Levels in Arabidopsis Root Cells Expressing a FRET-Based Phosphate Sensor" Plants 9, no. 10: 1310. https://doi.org/10.3390/plants9101310
APA StyleAssunção, A. G. L., Gjetting, S. K., Hansen, M., Fuglsang, A. T., & Schulz, A. (2020). Live Imaging of Phosphate Levels in Arabidopsis Root Cells Expressing a FRET-Based Phosphate Sensor. Plants, 9(10), 1310. https://doi.org/10.3390/plants9101310




