Ethnopharmacological Properties and Medicinal Uses of Litsea cubeba
Abstract
1. Introduction
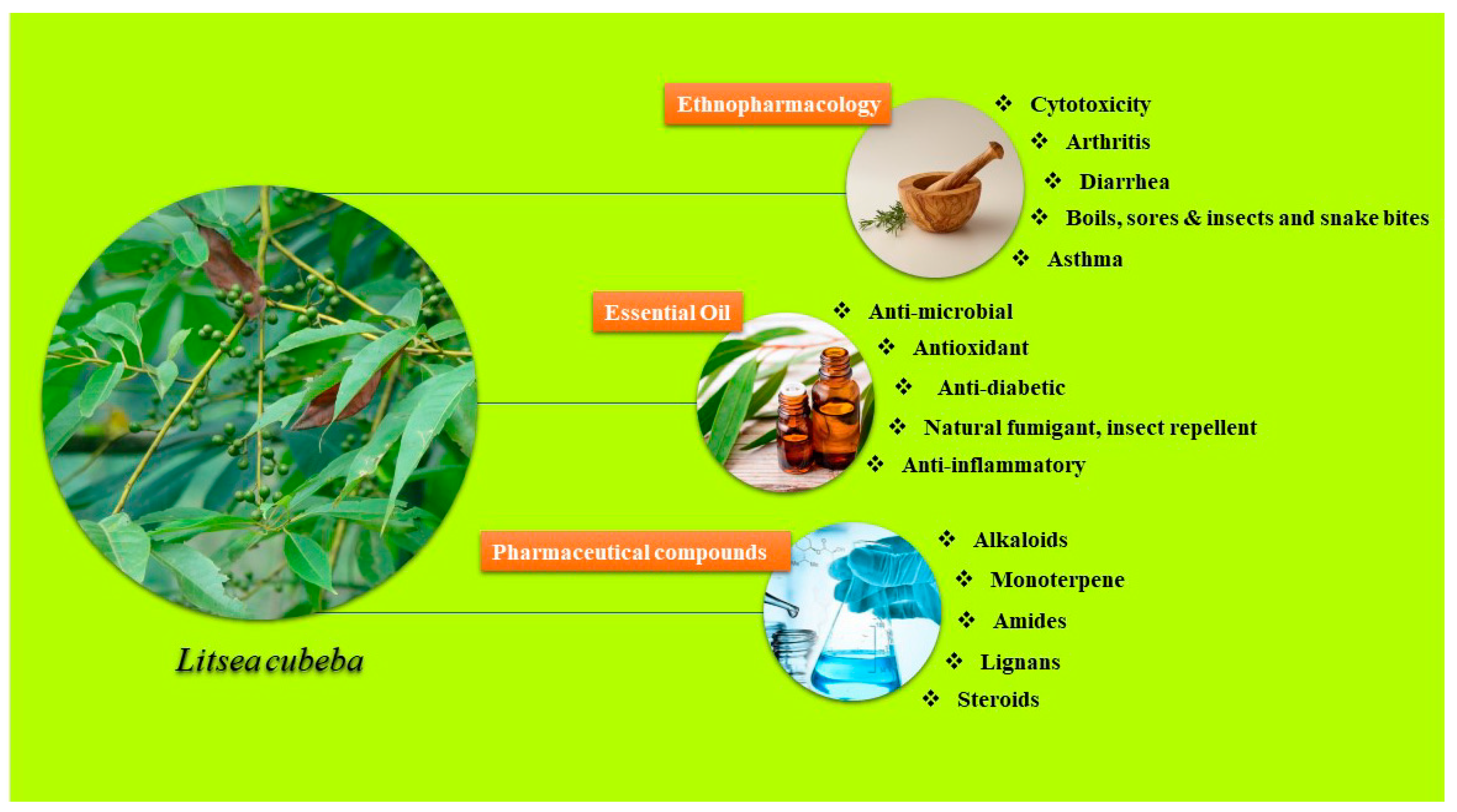
2. Ethnopharmacological Uses of Litsea Species
3. Essential Oils and Their Applications
4. Pharmaceutical Compounds
4.1. Alkaloids
4.2. Monoterpenes
4.3. Sesquiterpenes
4.4. Diterpenes
4.5. Flavonoids
4.6. Amides
4.7. Lignans
4.8. Steroids
4.9. Fatty Acids
5. Functions and Potential Mechanisms of Action
5.1. Anticancer Activity
5.2. Anti-Inflammatory Activity
5.3. Antimicrobial Activity
5.4. Antioxidant Activity
5.5. Antidiabetic Activity
5.6. Anti-HIV Activity
6. Conservation Strategies
7. Conclusions and Future Prospects
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Mabberley, D.J. The Plant-Book: A Portable Dictionary of the Higher Plants; Cambridge University Press: Cambridge, UK, 1993; Volume 581. [Google Scholar]
- Kapoor, L. Handbook of Ayurvedic Medicinal Plants: Herbal Reference Library; Routledge: London, UK, 2017. [Google Scholar]
- Yadav, G.; Goswami, B. Studies on the foliar constituents of food plants of muga silkworm (Antheraea assama Westwood). J. Ecobiol. 1990, 2, 222–228. [Google Scholar]
- Choudhury, S.; Ahmed, R.; Barthel, A.; Leclercq, P.A. Composition of the stem, flower and fruit oils of Litsea cubeba Pers. from two locations of Assam, India. J. Essent. Oil Res. 1998, 10, 381–386. [Google Scholar] [CrossRef]
- Duke, J.A.; Ayensu, E.S. Medicinal Plants of China; Reference Publications: Cambridge, MA, USA, 1985; Volume 2. [Google Scholar]
- Chen, Z.; Bi, H.; Fan, C.; Bao, C. Chemical constituents from the branch of Litsea cubeba (Lour.) Pers. Chem. Ind. For. Prod. 2013, 33, 133–136. [Google Scholar]
- Mao, A. Preliminary report on the folklore botany of Mao Nagas of Manipur India. Ethnobotany 1993, 5, 143–147. [Google Scholar]
- Yao, K.; Yang, C. Using pheasant pepper seed oil for controlling the broad bean weevil Bruchus rufimanus. Acta Ecol. Sin. 1984, 27, 173–181. [Google Scholar]
- Chen, S.-L.; Yu, H.; Luo, H.-M.; Wu, Q.; Li, C.-F.; Steinmetz, A. Conservation and sustainable use of medicinal plants: Problems, progress, and prospects. Chin. Med. 2016, 11, 37. [Google Scholar] [CrossRef]
- Budavari, S.; O’Neil, M.J.; Smith, A.; Heckelman, P.E. The Merck Index; Merck: Rahway, NJ, USA, 1989; Volume 11. [Google Scholar]
- Wang, J.-Q.; Li, J.; Zou, Y.-H.; Cheng, W.-M.; Lu, C.; Zhang, L.; Ge, J.-F.; Huang, C.; Jin, Y.; Lv, X.-W. Preventive effects of total flavonoids of Litsea coreana leve on hepatic steatosis in rats fed with high fat diet. J. Ethnopharmacol. 2009, 121, 54–60. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.-S.; Wen, Z.-Q.; Li, B.-T.; Zhang, H.-B.; Yang, J.-H. Ethnobotany, phytochemistry, and pharmacology of the genus Litsea: An update. J. Ethnopharmacol. 2016, 181, 66–107. [Google Scholar] [CrossRef] [PubMed]
- Feng, T.; Zhang, R.-T.; Tan, Q.-G.; Liu, Y.-P.; Cai, X.-H.; Luo, X.-D. Two new isoquinoline alkaloids from Litsea cubeba. Z. Naturforsch. B 2009, 64, 871–874. [Google Scholar] [CrossRef]
- Wang, H.; Liu, Y. Chemical composition and antibacterial activity of essential oils from different parts of Litsea cubeba. Chem. Biodivers. 2010, 7, 229–235. [Google Scholar] [CrossRef]
- Liu, T.-T.; Yang, T.-S. Antimicrobial impact of the components of essential oil of Litsea cubeba from Taiwan and antimicrobial activity of the oil in food systems. Int. J. Food Microbiol. 2012, 156, 68–75. [Google Scholar] [CrossRef]
- Hwang, J.-K.; Choi, E.-M.; Lee, J.H. Antioxidant activity of Litsea cubeba. Fitoterapia 2005, 76, 684–686. [Google Scholar] [CrossRef] [PubMed]
- Ho, C.-L.; Jie-Ping, O.; Liu, Y.-C.; Hung, C.-P.; Tsai, M.-C.; Liao, P.-C.; Wang, E.I.-C.; Chen, Y.-L.; Su, Y.-C. Compositions and in vitro anticancer activities of the leaf and fruit oils of Litsea cubeba from Taiwan. Nat. Prod. Commun. 2010, 5, 617–620. [Google Scholar] [CrossRef]
- Seal, S.; Chatterjee, P.; Bhattacharya, S.; Pal, D.; Dasgupta, S.; Kundu, R.; Mukherjee, S.; Bhattacharya, S.; Bhuyan, M.; Bhattacharyya, P.R. Vapor of volatile oils from Litsea cubeba seed induces apoptosis and causes cell cycle arrest in lung cancer cells. PLoS ONE 2012, 7, 47014. [Google Scholar] [CrossRef]
- Liao, P.-C.; Yang, T.-S.; Chou, J.-C.; Chen, J.; Lee, S.-C.; Kuo, Y.-H.; Ho, C.-L.; Chao, L.K.-P. Anti-inflammatory activity of neral and geranial isolated from fruits of Litsea cubeba Lour. J. Funct. Foods 2015, 19, 248–258. [Google Scholar] [CrossRef]
- Sun, Y.-X.; Lu, Y.-X.; Wang, L.-Y. Study on the mechanism of action of total flavonoids of Litsea coreana for reducing blood glucose level in rat with type 2 diabetes mellitus. Chin. J. Integr. Med. 2010, 30, 617–621. [Google Scholar]
- Chhetri, D.; Parajuli, P.; Subba, G. Antidiabetic plants used by Sikkim and Darjeeling Himalayan tribes, India. J. Ethnopharmacol. 2005, 99, 199–202. [Google Scholar] [CrossRef]
- Yadav, M. Herbal drugs and phytoconstituents useful for the management of diabetes. Int. J. Green Pharm. 2017, 11, S21. [Google Scholar]
- Jiang, Z.; Akhtar, Y.; Bradbury, R.; Zhang, X.; Isman, M.B. Comparative toxicity of essential oils of Litsea pungens and Litsea cubeba and blends of their major constituents against the cabbage looper, Trichoplusia ni. J. Agric. Food Chem. 2009, 57, 4833–4837. [Google Scholar] [CrossRef]
- Seo, S.-M.; Kim, J.; Lee, S.-G.; Shin, C.-H.; Shin, S.-C.; Park, I.-K. Fumigant antitermitic activity of plant essential oils and components from ajowan (Trachyspermum ammi), allspice (Pimenta dioica), caraway (Carum carvi), dill (Anethum graveolens), geranium (Pelargonium graveolens), and litsea (Litsea cubeba) oils against Japanese termite (Reticulitermes speratus Kolbe). J. Agric. Food Chem. 2009, 57, 6596–6602. [Google Scholar]
- Zhang, H.J.; Zheng, L.H.; Zhao, K.; Chen, Y.; Yi, Z. Insecticidal activities of constituents of Litsea cubeba fruit extracts effective against the maize weevil (Coleoptera: Curculionidae). J. Insect Sci. 2017, 17, 103. [Google Scholar] [CrossRef]
- Mohanan, N.; Kumar, E.S. A new species of Litsea (Lauraceae) from India. Nord. J. Bot. 2003, 23, 611–613. [Google Scholar] [CrossRef]
- Guzmán-Gutiérrez, S.; Gómez-Cansino, R.; García-Zebadúa, J.; Jiménez-Pérez, N.; Reyes-Chilpa, R. Antidepressant activity of Litsea glaucescens essential oil: Identification of β-pinene and linalool as active principles. J. Ethnopharmacol. 2012, 143, 673–679. [Google Scholar] [CrossRef] [PubMed]
- Kong, D.-G.; Zhao, Y.; Li, G.-H.; Chen, B.-J.; Wang, X.-N.; Zhou, H.-L.; Lou, H.-X.; Ren, D.-M.; Shen, T. The genus Litsea in traditional Chinese medicine: An ethnomedical, phytochemical and pharmacological review. J. Ethnopharmacol. 2015, 164, 256–264. [Google Scholar] [CrossRef]
- Agrawal, N.; Choudhary, A.S.; Sharma, M.C.; Dobhal, M.P. Chemical constituents of plants from the genus Litsea. Chem. Biodivers. 2011, 8, 223–243. [Google Scholar] [CrossRef]
- Ho, C.-L.; Lin, C.-Y.; Wang, E.I.-C.; Su, Y.-C. Composition, antioxidant and antimicrobial activities of leaf and twig essential oils of Litsea akoensis from Taiwan. Nat. Prod. Commun. 2011, 6, 901–904. [Google Scholar] [CrossRef]
- Cheng, H.-I.; Lin, W.-Y.; Duh, C.-Y.; Lee, K.-H.; Tsai, I.-L.; Chen, I.-S. New cytotoxic butanolides from Litsea acutivena. J. Nat. Prod. 2001, 64, 1502–1505. [Google Scholar] [CrossRef]
- Normile, D. The new face of traditional Chinese medicine. Science 2003, 299, 188–190. [Google Scholar] [CrossRef]
- Xie, Z.; Yu, Y. The Guide of National Chinese Herbal Medicine (I); People’s Medical Publishing House: Beijing, China, 1996. [Google Scholar]
- Kumar, P.B.; Kannan, M.M.; Quine, S.D. Litsea deccanensis ameliorates myocardial infarction in wistar rats: Evidence from biochemical histological studies. J. Young Pharm. 2011, 3, 287–296. [Google Scholar] [CrossRef]
- Ageel, A.; Islam, M.; Ginawi, O.; Al-Yahya, M. Evaluation of the aphrodisiac activity of Litsea chinensis (Lauraceae) and Orchis malculata (Orchidaceae) extracts in rats. Phytother. Res. 1994, 8, 103–105. [Google Scholar] [CrossRef]
- Hassan, S.H.A.; Fry, J.R.; Bakar, M.F.A. Antioxidant and phytochemical study on pengolaban (Litsea garciae), an edible underutilized fruit endemic to Borneo. Food Sci. Biotechnol. 2013, 22, 1–7. [Google Scholar] [CrossRef]
- Min, B.S.; Lee, S.Y.; Kim, J.H.; Kwon, O.K.; Park, B.Y.; An, R.B.; Lee, J.K.; Moon, H.I.; Kim, T.J.; Kim, Y.H. Lactones from the Leaves of Litsea j aponica and Their Anti-complement Activity. J. Nat. Prod. 2003, 66, 1388–1390. [Google Scholar] [CrossRef] [PubMed]
- Yoon, W.-J.; Kang, S.C.; Ham, Y.-M.; Kim, K.-N.; Yang, W.H.; Kim, H.-J.; Park, S.-Y.; Jung, Y.-H. Antioxidative and anti-inflammatory activities of Litsea japonica leaves. J. Korean Soc. Appl. Biol. 2010, 53, 27–32. [Google Scholar] [CrossRef]
- Bhatt, B.; Lemtur, M.; Changkija, S.; Sarkar, B. Fuelwood characteristics of important trees and shrubs of Eastern Himalaya. Energ. Sources Part A Recovery Util. Environ. Eff. 2017, 39, 47–50. [Google Scholar] [CrossRef]
- Baul, T.K.; Hossain, M.M.; Mezbahuddin, M.; Mohiuddin, M. Vegetative propagation of Litsea monopetala, a wild tropical medicinal plant: Effects of indole-3-butyric acid (IBA) on stem cuttings. J. For. Res. 2011, 22, 409–416. [Google Scholar] [CrossRef]
- Hua, H.; Xing, F.; Selvaraj, J.N.; Wang, Y.; Zhao, Y.; Zhou, L.; Liu, X.; Liu, Y. Inhibitory effect of essential oils on Aspergillus ochraceus growth and ochratoxin A production. PLoS ONE 2014, 9, 108285. [Google Scholar] [CrossRef]
- Rana, M.P.; Sohel, M.S.I.; Akhter, S.; Islam, M.J. Ethno-medicinal plants use by the Manipuri tribal community in Bangladesh. J. For. Res. 2010, 2, 85–92. [Google Scholar] [CrossRef]
- Ho, C.-L.; Wang, E.I.-C.; Hsu, K.-P.; Lee, P.-Y.; Su, Y.-C. Composition and antimicrobial activity of the leaf essential oil of Litsea kostermansii from Taiwan. Nat. Prod. Commun. 2009, 4, 1123–1236. [Google Scholar] [CrossRef]
- Nawawi, A.A.; Nakamura, N.; Hattori, M.; Kurokawa, M.; Shiraki, K. Inhibitory effects of Indonesian medicinal plants on the infection of herpes simplex virus type 1. Phytother. Res. 1999, 13, 37–41. [Google Scholar] [CrossRef]
- Ghosh, M.; Sinha, B. GC-MS studies on the bark extracts of Litsea polyantha Juss. Middle East J. Sci. Res. 2010, 5, 441–444. [Google Scholar]
- Delectis Florae Reipublicae Popularis Sinicae agendae. In Flora Reipublicae Popularis Sinicae; Academiae Sinicae, Ed.; Science Press: Beijing, China, 1999; Volume 77, pp. 1977–1999. [Google Scholar]
- Kala, C.P. Ethnomedicinal botany of the Apatani in the Eastern Himalayan region of India. J. Ethnobiol. Ethnomed. 2005, 1, 11. [Google Scholar] [CrossRef]
- Holloway, D.M.; Scheinmann, F. Co-occurrence of aporphine and biphenyl constituents in Litsea turfosa. Phytochemistry 1973, 12, 1503–1505. [Google Scholar] [CrossRef]
- Bighelli, A.; Muselli, A.; Casanova, J.; Tam, N.T.; Van Anh, V.; Bessière, J.-M. Chemical variability of Litsea cubeba leaf oil from Vietnam. J. Essent. Oil Res. 2005, 17, 86–88. [Google Scholar] [CrossRef]
- Si, L.; Chen, Y.; Han, X.; Zhan, Z.; Tian, S.; Cui, Q.; Wang, Y. Chemical composition of essential oils of Litsea cubeba harvested from its distribution areas in China. Molecules 2012, 17, 7057–7066. [Google Scholar] [CrossRef]
- Thielmann, J.; Muranyi, P. Review on the chemical composition of Litsea cubeba essential oils and the bioactivity of its major constituents citral and limonene. J. Essent. Oil Res. 2019, 1–18. [Google Scholar] [CrossRef]
- Saikia, A.K.; Chetia, D.; D’Arrigo, M.; Smeriglio, A.; Strano, T.; Ruberto, G. Screening of fruit and leaf essential oils of Litsea cubeba Pers. from north-east India–chemical composition and antimicrobial activity. J. Essent. Oil Res. 2013, 25, 330–338. [Google Scholar] [CrossRef]
- Luo, M.; Jiang, L.-K.; Zou, G.-L. Acute and genetic toxicity of essential oil extracted from Litsea cubeba (Lour.) Pers. J. Food Prot. 2005, 68, 581–588. [Google Scholar] [CrossRef]
- Wang, Y.-S.; Liao, Z.; Zhu, H.-K.; Feng, X.-F.; Huang, R.; Zhu, N.; Yang, J.-H. Megastigmane O-glucopyranosides from Litsea glutinosa. Chem. Nat. Compd. 2012, 48, 346–349. [Google Scholar] [CrossRef]
- Huang, X.-W.; Feng, Y.-C.; Huang, Y.; Li, H.-L. Potential cosmetic application of essential oil extracted from Litsea cubeba fruits from China. J. Essent. Oil Res. 2013, 25, 112–119. [Google Scholar] [CrossRef]
- Noosidum, A.; Prabaripai, A.; Chareonviriyaphap, T.; Chandrapatya, A. Excito-repellency properties of essential oils from Melaleuca leucadendron L. Litsea cubeba (Lour.) Persoon, and Litsea salicifolia (Nees) on Aedes aegypti (L.) mosquitoes. J. Vector Ecol. 2008, 33, 305–313. [Google Scholar] [CrossRef]
- Ko, K.; Juntarajumnong, W.; Chandrapatya, A. Repellency, fumigant and contact toxicities of Litsea cubeba (Lour.) Persoon against Sitophilus zeamais Motschulsky and Tribolium castaneum (Herbst). Kasetsart J. 2009, 43, 56–63. [Google Scholar]
- Zhou, S.; Kong, W.; Cao, J.; Logrieco, A.; Yang, S.; Yang, M. Effect of Aspergillus flavus contamination on the inherent quality of Glycyrrhiza uralensis. World Mycotoxin J. 2014, 7, 83–89. [Google Scholar] [CrossRef]
- Guerra, I.C.D.; de Oliveira, P.D.L.; de Souza Pontes, A.L.; Lúcio, A.S.S.C.; Tavares, J.F.; Barbosa-Filho, J.M.; Madruga, M.S.; de Souza, E.L. Coatings comprising chitosan and Mentha piperita L. or Mentha × villosa Huds essential oils to prevent common postharvest mold infections and maintain the quality of cherry tomato fruit. Int. J. Food Microbiol. 2015, 214, 168–178. [Google Scholar] [CrossRef] [PubMed]
- Ellis, W.; Smith, J.; Simpson, B.; Oldham, J.; Scott, P.M. Aflatoxins in food: Occurrence, biosynthesis, effects on organisms, detection, and methods of control. Crit. Rev. Food Sci. Nutr. 1991, 30, 403–439. [Google Scholar] [CrossRef]
- Kumar, P.; Mahato, D.K.; Kamle, M.; Mohanta, T.K.; Kang, S.G. Aflatoxins: A global concern for food safety, human health and their management. Front. Microbiol. 2017, 7, 2170. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; Jiang, J.; Qimei, L.; Yan, X.; Zhao, J.; Yuan, H.; Qin, Z.; Wang, M. The fungicidal terpenoids and essential oil from Litsea cubeba in Tibet. Molecules 2010, 15, 7075–7082. [Google Scholar] [CrossRef]
- Li, W.-R.; Shi, Q.-S.; Liang, Q.; Xie, X.-B.; Huang, X.-M.; Chen, Y.-B. Antibacterial activity and kinetics of Litsea cubeba oil on Escherichia coli. PLoS ONE 2014, 9, 110983. [Google Scholar] [CrossRef] [PubMed]
- Zhang, A.; Zhang, Y.; Branfman, A.R.; Baldessarini, R.J.; Neumeyer, J.L. Advances in development of dopaminergic aporphinoids. J. Med. Chem. 2007, 50, 171–181. [Google Scholar] [CrossRef]
- Custodio, D.L.; da Veiga Junior, V.F. Lauraceae alkaloids. RSC Adv. 2014, 4, 21864–21890. [Google Scholar] [CrossRef]
- Lee, S.-S.; Chen, C.-K.; Huang, F.-M.; Chen, C.-H. Two dibenzopyrrocoline alkaloids from Litsea cubeba. J. Nat. Prod. 1996, 59, 80–82. [Google Scholar] [CrossRef]
- Zhang, W.; Hu, J.-F.; Lv, W.-W.; Zhao, Q.-C.; Shi, G.-B. Antibacterial, antifungal and cytotoxic isoquinoline alkaloids from Litsea cubeba. Molecules 2012, 17, 12950–12960. [Google Scholar] [CrossRef]
- Tanaka, H.; Yatsuhashi, S.; Yasuda, T.; Sato, M.; Sakai, E.; Xiao, C.; Murata, H.; Murata, J. A new amide from the leaves and twigs of Litsea auriculata. J. Nat. Med. 2009, 63, 331–334. [Google Scholar] [CrossRef] [PubMed]
- Huang, C.-H.; Huang, W.-J.; Wang, S.-J.; Wu, P.-H.; Wu, W.-B. Litebamine, a phenanthrene alkaloid from the wood of Litsea cubeba, inhibits rat smooth muscle cell adhesion and migration on collagen. Eur. J. Pharmacol. 2008, 596, 25–31. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.-J.; Tseng, Y.-H.; Chu, F.-H.; Wen, T.-Y.; Cheng, W.-W.; Chen, Y.-T.; Tsao, N.-W.; Wang, S.-Y. Neuropharmacological activities of fruit essential oil from Litsea cubeba Persoon. J. Wood Sci. 2012, 58, 538–543. [Google Scholar] [CrossRef]
- Muhammed, A.M.; Subbu, R.M.; Jirovetz, L.; Mohamed, S.P. Composition and antimicrobial analysis of the essential oil of Litsea Laevigata nees (Lauraceae). Nat. Prod. Commun. 2008, 3, 1069–1072. [Google Scholar] [CrossRef]
- Ahmad, F.B.; bin Jantan, I.; Bakar, B.A.; Ahmad, A.S.B. A comparative study of the composition of the leaf oils of three Litsea species from Borneo. J. Essent. Oil Res. 2005, 17, 323–326. [Google Scholar] [CrossRef]
- Zhang, H.-J.; Van Hung, N.; Cuong, N.M.; Soejarto, D.D.; Pezzuto, J.M.; Fong, H.H.; Tan, G.T. Sesquiterpenes and butenolides, natural anti-HIV constituents from Litsea verticillata. Planta Med. 2005, 71, 452–457. [Google Scholar] [CrossRef] [PubMed]
- Trisonthi, P.; Sato, A.; Nishiwaki, H.; Tamura, H. A new diterpene from Litsea cubeba fruits: Structure elucidation and capability to induce apoptosis in HeLa cells. Molecules 2014, 19, 6838–6850. [Google Scholar] [CrossRef]
- Cheng, M.-J.; Wang, T.-A.; Lee, S.-J.; Chen, I.-S. A new butanolide and a new secobutanolide from Litsea lii var. nunkao-tahangensis. Nat. Prod. Res. 2010, 24, 647–656. [Google Scholar] [CrossRef]
- Chowdhury, J.U.; Bhuiyan, M.N.I.; Nandi, N.C. Aromatic plants of Bangladesh: Essential oils of leaves and fruits of Litsea glutinosa (Lour.) CB Robinson. Bangladesh J. Bot. 2008, 37, 81–83. [Google Scholar] [CrossRef]
- Chen, L.; Cheng, W.; Hu, C.; Jin, Y.; Li, R.; Li, J. Study on anti-inflammatory effects of total flavonoids of Litsea coreana Leve. Var. Anhui Nongye Daxue Xuebao 2004, 39, 439–442. [Google Scholar]
- Ye, H.; Jin, L.; Yu, J.; Wu, J. Research on the mechanism of antioxidation of flavoniods from leaves of Litsea coreana. J. Wenzhou Med. Coll. 2006, 36, 424–427. [Google Scholar]
- Tang, W.J.; Zhang, Y.L.; Xiao, Q.P.; Huang, C.; Jin, Y.; Li, J. Four flavanocoumarins from the leaves of Litsea coreana LEVL. Chem. Biodivers. 2013, 10, 1128–1132. [Google Scholar] [CrossRef] [PubMed]
- Guo, Q.; Zeng, K.; Gao, X.; Zhu, Z.; Zhang, S.; Chai, X.; Tu, P. Chemical constituents with NO production inhibitory and cytotoxic activities from Litsea cubeba. J. Nat. Med. 2015, 69, 94–99. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.; Zhu, C.; Xu, H.; Ni, X.; Yang, P. Study on chemical constituents of the root of Litsea cubeba II chloroform portion and ethyl acetate portion from methanol extract. Chin. J. Pharm. 2010, 7, 504–508. [Google Scholar]
- Agrawal, N.; Pareek, D.; Dobhal, S.; Sharma, M.C.; Joshi, Y.C.; Dobhal, M.P. Butanolides from methanolic extract of Litsea glutinosa. Chem. Biodivers. 2013, 10, 394–400. [Google Scholar] [CrossRef]
- Tsai, I.L.; Cheng, M.J.; Hung, H.W.; Cheng, H.I.; Chen, I.S. Chemical constituents from the leaves of Litsea acutivena. J. Chin. Chem. Soc. 2007, 54, 503–506. [Google Scholar] [CrossRef]
- Hata, T. Studies on the formosan plant seed oils XVI oil of Litsea cubeba pers. Nippon Kagaku Kaishi 1939, 60, 122–125. [Google Scholar] [CrossRef]
- Liu, R.; Zhang, H.-C.; Zhou, F.; Wang, R.-M.; Tu, Q.; Wang, J.-Y. Flavonoids and alkaloids from the leaves of Litsea fruticosa. Biochem. Syst. Ecol. 2013, 50, 293–295. [Google Scholar] [CrossRef]
- Yan, X.; Wei, X.; Xie, H.; Liu, M.; Zhang, F. Aporphine alkaloids of Litsea rotundifolia and L. rotundifolia var. oblongifolia. J. Trop. Subtrop. Bot. 2000, 8, 324–328. [Google Scholar]
- Jeong, W.-S.; Jun, M.; Kong, A.-N.T. Nrf2: A potential molecular target for cancer chemoprevention by natural compounds. Antioxid. Redox Signal. 2006, 8, 99–106. [Google Scholar] [CrossRef] [PubMed]
- Shen, T.; Chen, X.-M.; Harder, B.; Long, M.; Wang, X.-N.; Lou, H.-X.; Wondrak, G.T.; Ren, D.-M.; Zhang, D.D. Plant extracts of the family Lauraceae: A potential resource for chemopreventive agents that activate the nuclear factor-erythroid 2-related factor 2/antioxidant response element pathway. Planta Med. 2014, 80, 426–434. [Google Scholar] [PubMed]
- Choi, E.-M.; Hwang, J.-K. Effects of methanolic extract and fractions from Litsea cubeba bark on the production of inflammatory mediators in RAW264. 7 cells. Fitoterapia 2004, 75, 141–148. [Google Scholar] [CrossRef]
- Gogoi, D.; Bezbaruah, R.L.; Bordoloi, M.; Sarmah, R.; Bora, T.C. Insights from the docking analysis of biologically active compounds from plant Litsea Genus as potential COX-2 inhibitors. Bioinformation 2012, 8, 812. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.S.; Oh, O.-J.; Min, H.-Y.; Park, E.-J.; Kim, Y.; Park, H.J.; Han, Y.N.; Lee, S.K. Eugenol suppresses cyclooxygenase-2 expression in lipopolysaccharide-stimulated mouse macrophage RAW264. 7 cells. Life Sci. 2003, 73, 337–348. [Google Scholar] [CrossRef]
- Hong, C.H.; Hur, S.K.; Oh, O.-J.; Kim, S.S.; Nam, K.A.; Lee, S.K. Evaluation of natural products on inhibition of inducible cyclooxygenase (COX-2) and nitric oxide synthase (iNOS) in cultured mouse macrophage cells. J. Ethnopharmacol. 2002, 83, 153–159. [Google Scholar] [CrossRef]
- Ye, H.; Yu, J. The preliminary studies on antioxidation of three kinds of flavoniods from Litsea coreana. Zhong Yao Cai 2004, 27, 113–115. [Google Scholar] [PubMed]
- Choudhury, D.; Ghosal, M.; Das, A.P.; Mandal, P. In vitro antioxidant activity of methanolic leaves and barks extracts of four Litsea plants. Asian J. Plant Sci. Res. 2013, 3, 99–107. [Google Scholar]
- Wong, M.-H.; Lim, L.-F.; bin Ahmad, F.; bin Assim, Z. Antioxidant and antimicrobial properties of Litsea elliptica Blume and Litsea resinosa Blume (Lauraceae). Asian Pac. J. Trop. Biomed. 2014, 4, 386–392. [Google Scholar] [CrossRef] [PubMed]
- Hoang, V.D.; Tan, G.T.; Zhang, H.-J.; Tamez, P.A.; Van Hung, N.; Cuong, N.M.; Soejarto, D.D.; Fong, H.H.; Pezzuto, J.M. Natural anti-HIV agents—Part I:(+)-demethoxyepiexcelsin and verticillatol from Litsea verticillata. Phytochemistry 2002, 59, 325–329. [Google Scholar] [CrossRef]
- Zhang, H.-J.; Tan, G.T.; Hoang, V.D.; Van Hung, N.; Cuong, N.M.; Soejarto, D.D.; Pezzuto, J.M.; Fong, H.H. Natural anti-HIV agents. Part 3: Litseaverticillols A–H, novel sesquiterpenes from Litsea verticillata. Tetrahedron 2003, 59, 141–148. [Google Scholar] [CrossRef]
- McClanahan, T.R. The effect of a seed source on primary succession in a forest ecosystem. Vegetatio 1986, 65, 175–178. [Google Scholar] [CrossRef]
- Urbanska, K.M.; Erdt, S.; Fattorini, M. Seed rain in natural grassland and adjacent ski run in the Swiss Alps: A preliminary report. Restor. Ecol. 1998, 6, 159–165. [Google Scholar] [CrossRef]
- Bossuyt, B.; Hermy, M. Seed bank assembly follows vegetation succession in dune slacks. J. Veg. Sci. 2004, 15, 449–456. [Google Scholar] [CrossRef]
- Pakeman, R.; Small, J. The role of the seed bank, seed rain and the timing of disturbance in gap regeneration. J. Veg. Sci. 2005, 16, 121–130. [Google Scholar] [CrossRef]
- Booth, B.D.; Larson, D.W. The role of seed rain in determining the assembly of a cliff community. J. Veg. Sci. 1998, 9, 657–668. [Google Scholar] [CrossRef]
- Fuller, R.; Del Moral, R. The role of refugia and dispersal in primary succession on Mount St. Helens, Washington. J. Veg. Sci. 2003, 14, 637–644. [Google Scholar] [CrossRef]
- Tackenberg, O.; Stöcklin, J. Wind dispersal of alpine plant species: A comparison with lowland species. J. Veg. Sci. 2008, 19, 109–118. [Google Scholar] [CrossRef]
- Olano, J.; Caballero, I.; Laskurain, N.; Loidi, J.; Escudero, A. Seed bank spatial pattern in a temperate secondary forest. J. Veg. Sci. 2002, 13, 775–784. [Google Scholar] [CrossRef]
- Auld, T.D.; Denham, A.J.; Turner, K. Dispersal and recruitment dynamics in the fleshy-fruited Persoonia lanceolata (Proteaceae). J. Veg. Sci. 2007, 18, 903–910. [Google Scholar] [CrossRef]
- Augusto, L.; Dupouey, J.-L.; Picard, J.-F.; Ranger, J. Potential contribution of the seed bank in coniferous plantations to the restoration of native deciduous forest vegetation. Acta Oecol. 2001, 22, 87–98. [Google Scholar] [CrossRef]
- Luzuriaga, A.L.; Escudero, A.; Olano, J.M.; Loidi, J. Regenerative role of seed banks following an intense soil disturbance. Acta Oecol. 2005, 27, 57–66. [Google Scholar] [CrossRef]
- Xie, G.; Li, X.; Li, L.; Jiang, Y.; Zheng, Y.; Wang, W. Wild Energy Plant Resources, Conservation and Sustainable Use of Sanqingshan in Eastern China. IERI Procedia 2013, 5, 10–13. [Google Scholar] [CrossRef]
- Mao, A.; Wetten, A.; Fay, M.; Caligari, P. In vitro propagation of Litsea cubeba (Lours.) Pers., a multipurpose tree. Plant Cell Rep. 2000, 19, 263–267. [Google Scholar] [CrossRef] [PubMed]
| Country | Species | Plant Parts | Ethnopharmacological Properties | Solvent/Ratio/Dose Administered | Reference |
|---|---|---|---|---|---|
| Taiwan | L. akoensis Hayata | Stem bark | Cytotoxicity, antimicrobial activity | 15–30 μL of the oil dissolved in dimethylsulfoxide (DMSO) inoculated to plates with test microorganisms | [30] |
| China, Taiwan, Indochina | L. acutivena Hayata | Leaves and twigs | Oil for antimicrobial | 50 μL of 1 mg/mL MTT administered against A549 and HT-29 cells | [31] |
| India, China, Taiwan, Indonesia, and other parts of Southeast Asia | L. cubeba (Lour.) Pers. | Fruits | Pain reliever, promotes blood circulation, relieves stomach distension, asthma, demesia, diarrhea, turbid urine, and traumatic injury | - | [28,32,33,34] |
| Roots | Relieves cold, stomachache, headache, dermatophytosis, and arthralgia | - | |||
| Leaves | Promotes blood circulation, cures mammitis, heals hemostasis, sores furuncle, insect and snake bites, cures myocardial infarction in Wistar rats | 100 to 200 mg/kg of extract daily for a period of 21 days in rats | |||
| India | L. chinensis (Gaertn.) Sonner. | Fruit, leaves, stem | Activates sexual behavior | 500 mg/kg of extract to male rats | [35] |
| Malaysia, Indonesia, Philippines, Taiwan | L. garciae S. Vidal | Fruits | Antifungal, antioxidant | Samples (0.1 g) extracted for 2 h with 80% methanol | [36] |
| Southern Korea, Japan | L. japonica (Thunb.) Jussieu | Leaves | Antioxidative, anti-inflammatory | Assay with IC50 values of 149 and 58 μM | [37,38] |
| India (Eastern Himalaya) | L. laeta (Nees) Hook. f. | Leaves | Fuelwood | - | [39] |
| Nepal, India, Bangladesh, Burma, China | L. monopetala (Roxb.) Pers | Barks, leaves, roots, trunk | Cures gonorrhea, skin diseases, boil, diarrhea, and dislocation, antimicrobial | Fungal growth inhibition at 150–250 µL/L with fumigation | [40,41,42] |
| Taiwan | L. nakaii Hayata | Leaves | Antimicrobial | 15–30 μL of the oil in DMSO applied to microbial plates | [43] |
| Indonesia | L. odorifera Val. | Leaves | Anti HSV-1 | - | [44] |
| India | L. polyantha Juss. | Barks and roots | Effective in pains, bruises, fractures, diarrhea | - | [45] |
| China | L. rotundifolia Hemsl. | Roots | Treating rheumatic pain | - | [33] |
| India, Nepal, Bhutan, Vietnam, Bangladesh, Myanmar, China | L. salicifolia (J. Roxb. ex Nees) Hook. f. | - | Fruits for bone fracture, stomach disorder | - | [46,47] |
| Malaysia (Sarawak) | L. turfusa Kosterm. | Ground barks | Antifungal, antitumor | - | [48] |
| Compounds | Function | Reference |
|---|---|---|
| Alkaloids | ||
| (–)-8-O-Methyloblongine; (–)-Litcubine; (–)-Litcubinine; (–)-Magnocurarine; (–)-Oblongine; (+)-Isoboldine β-N-oxide;(+)-8-Methoxyisolaurenine-N-oxide; (+)-N-(Methoxycarbonyl)-N-(norboldine/norglaucine/norlauroscholtzine/norglaucine/norbulbodione/nordicentrin/norisocorydione/norpredicentrine); Actinodaphnine; Isoboldine; Atheroline; Boldine; Cassameridine; Cassythicine; Coclaurine; Corydine; Corytuberine; Dicentrine; Dicentrinone; Glaucine; Glaziovine; Isocorydine; Isodomesticine; Juziphine; Laetanine; Laetine; Lancifoliaine; Laurelliptine; Laurolitsine; Laurotetanine; Lindcarpine; Litebamine; Litsedine; Litseferine; Litseglutine B; Magnoflorine; N,O-Dimethylharnovine; N-Acetyllaurolitsine; N-Allyllaurolitsine; N-Methylcoclaurine; N-Methyllaurotetanine; N-Methyllindcarpine; Norcorydine; Nordicentrine; Norisoboldine; Norisocorydine; Norjuziphine; Oxoushinsunine; Pallidine; Phanostenine; Predicentrine; Reticuline; Sebiferine; Ushinsunine; Xanthoplanine; Butanolides and Butenolactone | Antioxidant, antiplatelet, antitumor, anticonvulsant, and antiplasmodial effects | [12,13,66,67,68,69] |
| Monoterpenes | ||
| Camphene; Bornylacetate; DL-Carvone; 1,8-Cineole; Citronellal; Citronellol; p-Cymene; Geranial; Geranyl acetate; Geraniol; Limonene; Linalool; β-Myrcene; Neral; Nerol; Neryl acetate; (E)-β-Ocimene; (Z)-β-Ocimene; β-Phellandrene; α-Pinene; β-Pinene; α-a-Isopulegol; Sabinene; cis-Sabinene hydrate; α-Terpineol; Terpinen-4-ol; Terpinolene; α-Terpinylacetate; Litseacubebic acid | Antibacterial activity | [29,62,70] |
| Sesquiterpenes | ||
| α-Amorphene; Aphanamol II; Aromadendrene; Bulnesol; α-Cadinene; β-Cadinene; γ-Cadinene; δ-Cadinene; α-Cadinol; β-Caryophyllene; Chromolaevanedione; α-Copaene; Isocurcumol; Elemol; β-Elemene; γ-Elemene; α-Eudesmol; β-Eudesmol; γ-Eudesmol; Germacrene; α-Humulene; Humulene oxide; Indonesiol; Ledene | Defensive roles | [29,71,72,73] |
| Diterpenes | ||
| Cubelin ((þ)-6-(4-hydroxy-4-methyl-2-pentenoyl)-4,6-dimethyl-5-(3-methyl 2-butenyl)-1,3-cyclohexadienecarbaldehyde);trans-Phytol | Antioxidative, antifungal, antiasthmatic, anti-anaphylactic properties | [74,75,76] |
| Flavonoids | ||
| Flavones; flavanols; flavanones; flavanonols; anthocyanidins; chalcones; flavan-3-ols | Anti-inflammatory, antioxidant, and hepatoprotective activities | [11,77,78,79] |
| Amides | ||
| cis-N-Feruloyl-3-methoxytyramine; N-Feruloyl-3-methoxytyramine; 3-Methoxy-N-sinapoyltyramine; N-trans-3,4-methylenecinnamoyl-3-methoxytyramine; Cubebamine A; 1,2-dihydro-6,8-dimethoxy-7-1-(3,5-dimethoxy-4-hydroxyphenyl)-N1,N2-bis-(2-(4-hydroxyphenyl)ethyl)-2,3-naphthalene dicarboxamide; N-cis-3,4-methylenedioxycinnamoyl-3-methoxytyramine | Anticancer effects | [6,68,80,81] |
| Lignans | ||
| Eugenol; syringaresinol; 9,9′-O-di-(E)-feruloyl-(+)-secoisolariciresinol; 9,9′-O-di-(E)-feruloyl-5,5′-(+)-dimethoxysecoisolariciresinol; balanophonin B; (+)-medioresinol; Lancifolin A; cyclolignan; Dehydrodieugenol; Dehydrodiisougenol; Grandisin; (+)-Eudesmin; (+)-Epiexcelsin; Biseugenol A, B; syringaresinol; Glochidioboside | Antioxidant and anticancer effects | [80,82] |
| Steroids | ||
| β-sitostenone; Daucosterol; β-Sitosterol; Sepesteonol, 5,6-Epoxystigmastan-3-ol; Stigmasterol; 6-O-Palmitoyl-β-sitosteryl-D-glucoside | [6,83] | |
| Fatty acids | ||
| Capric acid; cis-Dec-4-enoic acid; cis-Dodec-4-enoic acid (Linderic acid); cis-Tetradec-4-enoic acid (Tsuzuic acid); Hexadecenoic acid; Lignoceric acid; Lauric acid; Linoleic acid; Myristic acid; Oleic acid; Palmitic acid; Ethyl palmitate; Stearic acid; Ethyl stearate; Litseacubebic acid; 2,6-Dimethyl-6-hydroxy-2E,4E-hepta-2,4-dienal; 6,7-Dihydroxy-3,7-dimethyl-oct-2-enoic acid | Antidiabetic effects | [62,81,84] |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kamle, M.; Mahato, D.K.; Lee, K.E.; Bajpai, V.K.; Gajurel, P.R.; Gu, K.S.; Kumar, P. Ethnopharmacological Properties and Medicinal Uses of Litsea cubeba. Plants 2019, 8, 150. https://doi.org/10.3390/plants8060150
Kamle M, Mahato DK, Lee KE, Bajpai VK, Gajurel PR, Gu KS, Kumar P. Ethnopharmacological Properties and Medicinal Uses of Litsea cubeba. Plants. 2019; 8(6):150. https://doi.org/10.3390/plants8060150
Chicago/Turabian StyleKamle, Madhu, Dipendra K. Mahato, Kyung Eun Lee, Vivek K. Bajpai, Padam Raj Gajurel, Kang Sang Gu, and Pradeep Kumar. 2019. "Ethnopharmacological Properties and Medicinal Uses of Litsea cubeba" Plants 8, no. 6: 150. https://doi.org/10.3390/plants8060150
APA StyleKamle, M., Mahato, D. K., Lee, K. E., Bajpai, V. K., Gajurel, P. R., Gu, K. S., & Kumar, P. (2019). Ethnopharmacological Properties and Medicinal Uses of Litsea cubeba. Plants, 8(6), 150. https://doi.org/10.3390/plants8060150