Anacardium Plants: Chemical,Nutritional Composition and Biotechnological Applications
Abstract
:1. Introduction
2. Habitat and Cultivation of Anacardium Species
3. Nutritional Composition
3.1. Amino Acids
3.2. Vitamins and Minerals
3.3. Lipids and Fatty Acid Profile
3.4. Polysaccharides
3.5. Antinutrients and Heavy Metals
4. Phytochemical Composition of Anacardium Plants: A Brief Overview
4.1. Phenolic Compounds
4.1.1. Flavonoids
4.1.2. Tannins
4.2. Anacardic Acids
4.3. Carotenoids, Carotenoid Esters, and Anthocyanins
4.4. Essential Oils, Volatiles and Aroma Compounds
4.5. Other Compounds
5. Food Preservative Applications of Anacardium Plants
5.1. Food Preservation from Natural Sources
5.2. Anacardium Antioxidant and Antimicrobial Activities and its Potential as Food Preservative
6. Conclusions and Future Perspectives
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Tianlu, M.; Barfod, A. Anacardiaceae. In Flora of China; Wu, Z.Y., Raven, P.H., Hong, D.Y., Eds.; Science Press and Missouri Botanical Garden Press: Beijing, China; St. Louis, MO, USA, 2008; Volume 11, pp. 335–357. [Google Scholar]
- Mitra, R.; Mitchell, B.; Gray, C.; Orbell, J.; Coulepis, T.; Muralitharan, M. Medicinal plants of brazil. Asia Pac. Biotech News 2007, 11, 689–706. [Google Scholar]
- The Plant List. Version 1. Available online: http://www.Theplantlist.Org/ (accessed on 27 September 2018).
- Trox, J.; Vadivel, V.; Vetter, W.; Stuetz, W.; Scherbaum, V.; Gola, U.; Nohr, D.; Biesalski, H.K. Bioactive compounds in cashew nut (anacardium occidentale l.) kernels: Effect of different shelling methods. J. Agric. Food Chem. 2010, 58, 5341–5346. [Google Scholar] [CrossRef] [PubMed]
- Baptista, A.; Goncalves, R.V.; Bressan, J.; Peluzio, M. Antioxidant and antimicrobial activities of crude extracts and fractions of cashew (anacardium occidentale l.), cajui (anacardium microcarpum), and pequi (caryocar brasiliense c.): A systematic review. Oxid. Med. Cell Longev. 2018, 2018, 3753562. [Google Scholar] [CrossRef] [PubMed]
- Adewale, B.; Ibiremo, O.; Odoh, N.; Adeyemi, E. Genetic estimates and trend analysis of some growth parameters of cashew (anacardium occidentale l.) as influenced by nine nutrient combinations. J. Agric. Biotechnol. Sustain. Dev. 2013, 5, 6–11. [Google Scholar] [CrossRef]
- Akinwale, T.; Olubamiwa, O.; Ajav, E. Cottage processing of cashew apple juice in nigeria: Physico-chemical and sensory evaluation of product. J. Food Technol. Afr. 2001, 6, 56–58. [Google Scholar] [CrossRef]
- Adavi, R.D. Molecular Diversity and Phenotyping of Selected Cashew Genotypes of Goa and Physiological Response of cv. Goa-1 to Insitu Moisture Conservation; University of Agricultural Sciences Dharwad: Karnataka, India, 2008. [Google Scholar]
- Adebola, P.; Esan, E. Finlay and wilkinson’s stability parameters and genotype ranks for yield of 12 cashew (anacardium occidentale l.) selections in Nigeria. Trop. Agric. 2002, 79, 137–139. [Google Scholar]
- Adejumo, T.; Otuonye, A. The use of botanicals in the control of inflorescence blight disease of cashew, anacardium occidentale. Nigr. J. Sci. 2002, 36, 75–80. [Google Scholar]
- Batish, D.R.; Kohli, R.; Singh, H.; Saxena, D. Studies on herbicidal activity of parthenin, a constituent of parthenium hysterophorus, towards billgoat weed (ageratum conyzoides). Curr. Sci. 1997, 73, 369–371. [Google Scholar]
- Honorato, T.L.; Rabelo, M.C.; Gonçalves, L.R.B.; Pinto, G.A.S.; Rodrigues, S. Fermentation of cashew apple juice to produce high added value products. World J. Microbiol. Biotechnol. 2007, 23, 1409–1415. [Google Scholar] [CrossRef]
- Yahia, E. Postharvest biology and technology of tropical and subtropical fruits. In Wood Head Publishing; Cambridge University Press: Cambridge, UK, 2011. [Google Scholar]
- Prajapati, N.D. Handbook of Medicinal Plants; Agrobios: Rajasthan, India, 2003. [Google Scholar]
- Tamuno, E.; Onyedikachi, E. Proximate, mineral and functional properties of defatted and undefatted cashew (anacardium occidentale linn.) kernel flour. Eur. J. Food Sci. Technol. 2015, 3, 11–19. [Google Scholar]
- Donkoh, A.; Attoh-Kotoku, V.; Osei Kwame, R.; Gascar, R. Evaluation of nutritional quality of dried cashew nut testa using laboratory rat as a model for pigs. Sci. World J. 2012, 2012, 984249. [Google Scholar] [CrossRef]
- Rico, R.; Bullo, M.; Salas-Salvado, J. Nutritional composition of raw fresh cashew (anacardium occidentale l.) kernels from different origin. Food Sci. Nutr. 2016, 4, 329–338. [Google Scholar] [CrossRef]
- Adou, M.; Tetchi, F.; Gbané, M.; Kouassi, K.; Amani, N. Physico-chemical characterization of cashew apple juice (anacardium occidentale, l.) from yamoussoukro (côte d’ivoire). Innov. Rom. Food Biotechnol. 2012, 11, 32–43. [Google Scholar]
- Adeyeye, E.I.; Asaolu, S.S.; Aluko, A.O. Amino acid composition of two masticatory nuts (cola acuminata and garcinia kola) and a snack nut (anacardium occidentale). Int. J. Food Sci. Nutr. 2007, 58, 241–249. [Google Scholar] [CrossRef]
- Fagbemi, T.N. Effect of processing on chemical composition of cashew nut (anacardium occidentale). J. Food Sci. Technol. 2009, 46, 36–40. [Google Scholar]
- Fetuga, B.; Babatunde, G.; Oyenuga, V. Composition and nutritive value of cashew nut to the rat. J. Agric. Food Chem. 1974, 22, 678–682. [Google Scholar] [CrossRef]
- Chung, K.H.; Shin, K.O.; Hwang, H.J.; Choi, K.S. Chemical composition of nuts and seeds sold in Korea. Nutr. Res. Pract. 2013, 7, 82–88. [Google Scholar] [CrossRef] [Green Version]
- Eliakim-Ikechukwu, C.; Obri, A.; Akpa, O. Phytochemical and micronutrient composition of anacardium occidentale linn (cashew) stem-bark hydroethanolic extract and its effect on the fasting blood glucose levels and body weight of diabetic wistar rats. Int. J. Nutr. Wellness 2010, 10, 1–6. [Google Scholar]
- Belonwu, D.; Ibegbulem, C.; Nwokocha, M.; Chikezie, P. Some phytochemicals and hydrophilic vitamins of anacardium occidentale. Res. J. Phytochem. 2014, 8, 78–91. [Google Scholar]
- Melo-Cavalcante, A.A.; Rubensam, G.; Picada, J.N.; Gomes da Silva, E.; Fonseca Moreira, J.C.; Henriques, J.A.P. Mutagenicity, antioxidant potential, and antimutagenic activity against hydrogen peroxide of cashew (anacardium occidentale) apple juice and cajuina. Environ. Mol. Mutagenesis 2003, 41, 360–369. [Google Scholar] [CrossRef]
- Nagaraja, K.; Nampoothiri, V. Chemical characterization of high-yielding varieties of cashew (anacardium occidentale). Plant Foods Hum. Nutr. 1986, 36, 201–206. [Google Scholar] [CrossRef]
- Trox, J.; Vadivel, V.; Vetter, W.; Stuetz, W.; Kammerer, D.R.; Carle, R.; Scherbaum, V.; Gola, U.; Nohr, D.; Biesalski, H.K. Catechin and epicatechin in testa and their association with bioactive compounds in kernels of cashew nut (anacardium occidentale l.). Food Chem. 2011, 128, 1094–1099. [Google Scholar] [CrossRef]
- Subramanian, S.; Joseph, K.; Nair, A. Polyphenols from anacardium occidentale. Phytochemisty 1969, 8, 673–674. [Google Scholar] [CrossRef]
- Nagaraja, K. Lipids of high-yielding varieties of cashew (anacardium occidentale l.). Plant Foods Hum. Nutr. 1987, 37, 307–311. [Google Scholar] [CrossRef]
- Guo, Q.; Wang, F.; He, F.; Ha, Y.M.; Li, Q.P.; Jin, J.; Deng, Z.X. The impact of technical cashew nut shell liquid on thermally-induced trans isomers in edible oils. J. Food Sci. Technol. 2016, 53, 1487–1495. [Google Scholar] [CrossRef]
- Martínez Aguilar, Y.; Rodríguez, F.S.; Saavedra, M.A.; Hermosilla Espinosa, R.; Yero, O.M. Secondary metabolites and in vitro antibacterial activity of extracts from anacardium occidentale l. (cashew tree) leaves. Rev. Cuba. De Plantas Med. 2012, 17, 320–329. [Google Scholar]
- Fadeyi, O.; Olatunji, G.; Ogundele, V. Isolation and characterization of the chemical constituents of anacardium occidentale cracked bark. Nat. Prod. Chem. Res. 2015, 3, 5. [Google Scholar] [CrossRef]
- De Paula, R.C.M.; Rodrigues, J.F. Composition and rheological properties of cashew tree gum, the exudate polysaccharide from anacardium-occidentale L. Carbohyd. Polym. 1995, 26, 177–181. [Google Scholar] [CrossRef]
- Okonkwo, T.J.; Okorie, O.; Okonta, J.M.; Okonkwo, C.J. Sub-chronic hepatotoxicity of anacardium occidentale (anacardiaceae) inner stem bark extract in rats. Indian J. Pharm. Sci. 2010, 72, 353–357. [Google Scholar] [CrossRef]
- Mubofu, E.B.; Mgaya, J.E. Chemical valorization of cashew nut shell waste. Top. Curr. Chem. 2018, 376, 8. [Google Scholar] [CrossRef]
- Philip, J.Y.; Buchweishaija, J.; Mkayula, L.L.; Ye, L. Preparation of molecularly imprinted polymers using anacardic acid monomers derived from cashew nut shell liquid. J. Agric. Food Chem. 2007, 55, 8870–8876. [Google Scholar] [CrossRef]
- Assuncao, R.B.; Mercadante, A.Z. Carotenoids and ascorbic acid from cashew apple (anacardium occidentale l.): Variety and geographic effects. Food Chem. 2003, 81, 495–502. [Google Scholar] [CrossRef]
- Tedong, L.; Madiraju, P.; Martineau, L.C.; Vallerand, D.; Arnason, J.T.; Desire, D.D.; Lavoie, L.; Kamtchouing, P.; Haddad, P.S. Hydro-ethanolic extract of cashew tree (anacardium occidentale) nut and its principal compound, anacardic acid, stimulate glucose uptake in c2c12 muscle cells. Mol. Nutr. Food Res. 2010, 54, 1753–1762. [Google Scholar] [CrossRef]
- Paramashivappa, R.; Phain, K.; Vithay, A.; Rao, A. Method for isolation of major phenolic consitutents from cashew (anacardium occidentale l.) nutshell liquid. J. Agric. Food Chem. 2001, 49, 2548–2551. [Google Scholar] [CrossRef]
- Stasiuk, M.; Janiszewska, A.; Kozubek, A. Phenolic lipids affect the activity and conformation of acetylcholinesterase from electrophorus electricus (electric eel). Nutrients 2014, 6, 1823–1831. [Google Scholar] [CrossRef]
- Kumar, P.; Paramashivappa, R.; Vithayathil, P.J.; Subba Rao, P.V.; Srinivasa Rao, A. Process for isolation of cardanol from technical cashew (anacardium occidentale l.) nut shell liquid. J. Agric. Food Chem. 2002, 50, 4705–4708. [Google Scholar] [CrossRef]
- Vilar, M.S.; de Souza, G.L.; Vilar Dde, A.; Leite, J.A.; Raffin, F.N.; Barbosa-Filho, J.M.; Nogueira, F.H.; Rodrigues-Mascarenhas, S.; Moura, T.F. Assessment of phenolic compounds and anti-inflammatory activity of ethyl acetate phase of anacardium occidentale l. Bark. Molecules 2016, 21, 1087. [Google Scholar] [CrossRef]
- Marc, A.; Ange, K.; Achille, T.; Georges, A. Phenolic profile of cashew apple juice (anacardium occidentale l.) from yamoussoukro and korhogo (côte d’ivoire). J. Appl. Biosci. 2012, 49, 3331–3338. [Google Scholar]
- Queiroz, C.; da Silva, A.J.R.; Lopes, M.L.M.; Fialho, E.; Valente-Mesquita, V.L. Polyphenol oxidase activity, phenolic acid composition and browning in cashew apple (anacardium occidentale, l.) after processing. Food Chem. 2011, 125, 128–132. [Google Scholar] [CrossRef]
- Konan, N.A.; Bacchi, E.M. Antiulcerogenic effect and acute toxicity of a hydroethanolic extract from the cashew (anacardium occidentale l.) leaves. J. Ethnopharmacol. 2007, 112, 237–242. [Google Scholar] [CrossRef]
- Alexander-Lindo, R.L.; Morrison, E.Y.; Nair, M.G. Hypoglycaemic effect of stigmast-4-en-3-one and its corresponding alcohol from the bark of anacardium occidentale (cashew). Phytother. Res. 2004, 18, 403–407. [Google Scholar] [CrossRef]
- De Brito, E.S.; de Araujo, M.C.P.; Lin, L.Z.; Harnly, J. Determination of the flavonoid components of cashew apple (anacardium occidentale) by lc-dad-esi/ms. Food Chem. 2007, 105, 1112–1118. [Google Scholar] [CrossRef]
- Mohd Shukri, M.; Alan, C. Analysis of phenolics in anacardium occidentale shoot extracts using a reversed-phase high performance liquid chromatography tandem mass spectrometry (rp-hplc-ms). J. Trop. Agric. Food Sci. 2010, 38, 221–230. [Google Scholar]
- Celis, C.; García, A.; Sequeda, G.; Mendez, G.; Torrenegra, R. Antimicrobial activity of extracts obtained from anacardium excelsum againts some pathogenic microorganisms. Emir. J. Food Agric. 2011, 23, 249–257. [Google Scholar]
- Miean, K.H.; Mohamed, S. Flavonoid (myricetin, quercetin, kaempferol, luteolin, and apigenin) content of edible tropical plants. J. Agric. Food Chem. 2001, 49, 3106–3112. [Google Scholar] [CrossRef]
- Ajileye, O.O.; Obuotor, E.M.; Akinkunmi, E.O.; Aderogba, M.A. Isolation and characterization of antioxidant and antimicrobial compounds from anacardium occidentale l. (anacardiaceae) leaf extract. J. King Saud Univ. Sci. 2015, 27, 244–252. [Google Scholar] [CrossRef]
- Taiwo, B.J.; Fatokun, A.A.; Olubiyi, O.O.; Bamigboye-Taiwo, O.T.; van Heerden, F.R.; Wright, C.W. Identification of compounds with cytotoxic activity from the leaf of the nigerian medicinal plant, anacardium occidentale l. (anacardiaceae). Bioorg. Med. Chem. 2017, 25, 2327–2335. [Google Scholar] [CrossRef]
- Schweiggert, R.M.; Vargas, E.; Conrad, J.; Hempel, J.; Gras, C.C.; Ziegler, J.U.; Mayer, A.; Jimenez, V.; Esquivel, P.; Carle, R. Carotenoids, carotenoid esters, and anthocyanins of yellow-, orange-, and red-peeled cashew apples (anacardium occidentale l.). Food Chem. 2016, 200, 274–282. [Google Scholar] [CrossRef]
- Michodjehoun-Mestres, L.; Amraoui, W.; Brillouet, J.M. Isolation, characterization, and determination of 1-o-trans-cinnamoyl-beta-d-glucopyranose in the epidermis and flesh of developing cashew apple (anacardium occidentale l.) and four of its genotypes. J. Agric. Food Chem. 2009, 57, 1377–1382. [Google Scholar] [CrossRef]
- Da Silva, I.M.; Rabelo, M.C.; Rodrigues, S. Cashew juice containing prebiotic oligosaccharides. J. Food Sci. Technol. 2014, 51, 2078–2084. [Google Scholar] [CrossRef]
- Chandrasekara, N.; Shahidi, F. Effect of roasting on phenolic content and antioxidant activities of whole cashew nuts, kernels, and testa. J. Agric. Food Chem. 2011, 59, 5006–5014. [Google Scholar] [CrossRef]
- Cunha, A.G.; Brito, E.S.; Moura, C.F.H.; Ribeiro, P.R.V.; Miranda, M.R.A. Uplc-qtof-ms/ms-based phenolic profile and their biosynthetic enzyme activity used to discriminate between cashew apple (anacardium occidentale l.) maturation stages. J. Chromatogr. B 2017, 1051, 24–32. [Google Scholar] [CrossRef]
- Razali, N.; Razab, R.; Junit, S.M.; Aziz, A.A. Radical scavenging and reducing properties of extracts of cashew shoots (anacardium occidentale). Food Chem. 2008, 111, 38–44. [Google Scholar] [CrossRef]
- Suo, M.R.; Isao, H.; Ishida, Y.; Shimano, Y.; Bi, C.X.; Kato, H.; Takano, F.; Ohta, T. Phenolic lipid ingredients from cashew nuts. J. Nat. Med. Tokyo 2012, 66, 133–139. [Google Scholar] [CrossRef]
- Murthy, S.S.; Anjaneyulu, A.S.; Row, L.R.; Pelter, A.; Ward, R.S. Chemical examination of anacardium occidentale. Planta Med. 1982, 45, 3–10. [Google Scholar] [CrossRef]
- Konan, N.A.; Lincopan, N.; Collantes Díaz, I.E.; de Fátima Jacysyn, J.; Tanae Tiba, M.M.; Pessini Amarante Mendes, J.G.; Bacchi, E.M.; Spira, B. Cytotoxicity of cashew flavonoids towards malignant cell lines. Exp. Toxicol. Pathol. 2012, 64, 435–440. [Google Scholar] [CrossRef]
- Lokeswari, N.; Sriramireddy, D.; Sudhakar, P.; Varaprasad, B. Extraction of tannins from anacardium occidentale and effect of physical parameters. J. Pharm. Res. 2010, 13, 906–908. [Google Scholar]
- Viswanath, V.; Leo, V.V.; Prabha, S.S.; Prabhakumari, C.; Potty, V.P.; Jisha, M.S. Thermal properties of tannin extracted from anacardium occidentale l. Using tga and ft-ir spectroscopy. Nat. Prod. Res. 2016, 30, 223–227. [Google Scholar] [CrossRef]
- Nagaraja, K.V. Biochemical composition of cashew (anacardium occidentale l.) kernel testa. J. Food. Sci. Technol. 2000, 37, 554–556. [Google Scholar]
- Morais, S.M.; Silva, K.A.; Araujo, H.; Vieira, I.G.P.; Alves, D.R.; Fontenelle, R.O.S.; Silva, A.M.S. Anacardic acid constituents from cashew nut shell liquid: Nmr characterization and the effect of unsaturation on its biological activities. Pharmaceuticals 2017, 10, 31. [Google Scholar] [CrossRef]
- Philip, J.Y.; Da Cruz Francisco, J.; Dey, E.S.; Buchweishaija, J.; Mkayula, L.L.; Ye, L. Isolation of anacardic acid from natural cashew nut shell liquid (cnsl) using supercritical carbon dioxide. J. Agric. Food Chem. 2008, 56, 9350–9354. [Google Scholar] [CrossRef]
- Przeworska, E.; Gubernator, J.; Kozubek, A. Formation of liposomes by resorcinolic lipids, single-chain phenolic amphiphiles from anacardium occidentale L. Biochim. Biophys. Acta 2001, 1513, 75–81. [Google Scholar] [CrossRef]
- Maia, J.; Andrade, E.; Zoghbi, M. Volatile constituents of the leaves, fruits and flowers of cashew (anacardium occidentale l.). J. Food Comp. Anal. 2000, 13, 227–232. [Google Scholar] [CrossRef]
- Kossouoh, C.; Moudachirou, M.; Adjakidje, V.; Chalchat, J.; Figuérédo, G. Essential oil chemical composition of anacardium occidentale l. Leaves from benin. J. Essen. Oil Res. 2008, 20, 5–8. [Google Scholar] [CrossRef]
- Egonyu, J.P.; Ekesi, S.; Kabaru, J.; Irungu, L.; Torto, B. Cashew volatiles mediate short-range location responses in pseudotheraptus wayi (heteroptera: Coreidae). Environ. Entomol. 2013, 42, 1400–1407. [Google Scholar] [CrossRef]
- Montanari, R.M.; Barbosa, L.C.A.; Demuner, A.J.; Silva, C.J.; Andrade, N.J.; Ismail, F.M.D.; Barbosa, M.C.A. Exposure to anacardiaceae volatile oils and their constituents induces lipid peroxidation within food-borne bacteria cells. Molecules 2012, 17, 9728–9740. [Google Scholar] [CrossRef]
- Garruti, D.; Franco, M.; da Silva, M.; Janzantti, N.; Alves, G. Evaluation of volatile favour compounds from cashew apple (anacardium occidentale l) juice by the osme gas chromatography/olfactometry technique. J. Sci. Food Agric. 2003, 83, 1455–1462. [Google Scholar] [CrossRef]
- Bicalho, B.; Rezende, C.M. Volatile compounds of cashew apple (anacardium occidentale l). Zeitschrift für Naturforschung C 2001, 56, 35–39. [Google Scholar] [CrossRef]
- Cardeal, Z.; Guimara, E.; Parreira, F. Analysis of volatile compounds in some typical brazilian fruits and juices by spme-gc method. Food Addit. Contam. 2005, 22, 508–513. [Google Scholar] [CrossRef]
- Patel, R.N.; Bandyopadhyay, S.; Ganesh, A. Extraction of cashew (anacardium occidentale) nut shell liquid using supercritical carbon dioxide. Bioresour. Technol 2006, 97, 847–853. [Google Scholar] [CrossRef]
- Sullivan, J.T.; Richards, C.S.; Lloyd, H.A.; Krishna, G. Anacardic acid: Molluscicide in cashew nut shell liquid. Planta Med. 1982, 44, 175–177. [Google Scholar] [CrossRef]
- Velagapudi, R.; Ajileye, O.O.; Okorji, U.; Jain, P.; Aderogba, M.A.; Olajide, O.A. Agathisflavone isolated from anacardium occidentale suppresses sirt1-mediated neuroinflammation in bv2 microglia and neurotoxicity in appswe-transfected sh-sy5y cells. Phytother. Res. 2018, 32, 1957–1966. [Google Scholar] [CrossRef]
- Moronkola, D.; Kasali, A.; Ekundayo, O. Composition of the limonene dominated leaf essential oil of nigerian anacardium occidentale. J. Essen. Oil Res. 2007, 19, 351–353. [Google Scholar] [CrossRef]
- Sampaio, K.L.; Garruti, D.S.; Franco, M.R.; Janzantti, N.S.; Da Silva, M.A. Aroma volatiles recovered in the water phase of cashew apple (anacardium occidentale l.) juice during concentration. J. Sci. Food Agric. 2011, 91, 1801–1809. [Google Scholar] [CrossRef]
- Valim, M.F.; Rouseff, R.L.; Lin, J. Gas chromatographic-olfactometric characterization of aroma compounds in two types of cashew apple nectar. J. Agric. Food Chem. 2003, 51, 1010–1015. [Google Scholar] [CrossRef]
- Agila, A.; Barringer, S.A. Volatile profile of cashews (anacardium occidentale l.) from different geographical origins during roasting. J. Food Sci. 2011, 76, C768–C774. [Google Scholar] [CrossRef]
- Sampaio, K.L.; Biasoto, A.C.; Da Silva, M.A. Comparison of techniques for the isolation of volatiles from cashew apple juice. J. Sci. Food Agric. 2015, 95, 299–312. [Google Scholar] [CrossRef]
- De Godoy Alves Filho, E.; Rodrigues, T.H.S.; Fernandes, F.A.N.; Pereira, A.L.F.; Narain, N.; de Brito, E.S.; Rodrigues, S. Chemometric evaluation of the volatile profile of probiotic melon and probiotic cashew juice. Food Res. Int. 2017, 99, 461–468. [Google Scholar] [CrossRef]
- Fancelli, M.; Borges, M.; Laumann, R.A.; Pickett, J.A.; Birkett, M.A.; Blassioli-Moraes, M.C. Attractiveness of host plant volatile extracts to the asian citrus psyllid, diaphorina citri, is reduced by terpenoids from the non-host cashew. J. Chem. Ecol. 2018, 44, 397–405. [Google Scholar] [CrossRef]
- Aguilar-Ortigoza, C.J.; Sosa, V.; Aguilar-Ortigoza, M. Toxic phenols in various anacardiaceae species. Econ. Bot. 2003, 57, 354–364. [Google Scholar] [CrossRef]
- Toyomizu, M.; Sugiyama, S.; Jin, R.L.; Nakatsu, T. Alpha-glucosidase and aldose reductase inhibitorsconstituents of cashew, anacardium-occidentale, nut shell liquids. Phytother. Res. 1993, 7, 252–254. [Google Scholar] [CrossRef]
- Trevisan, M.T.S.; Pfundstein, B.; Haubner, R.; Würtele, G.; Spiegelhalder, B.; Bartsch, H.; Owen, R.W. Characterization of alkyl phenols in cashew (anacardium occidentale) products and assay of their antioxidant capacity. Food Chem. Toxicol. 2006, 44, 188–197. [Google Scholar] [CrossRef]
- Kubo, I.; Komatsu, S.; Ochi, M. Molluscicides from the cashew anacardium occidentale and their large-scale isolation. J. Agric. Food Chem. 1986, 34, 970–973. [Google Scholar] [CrossRef]
- Kubo, I.; Kinst-Hori, I.; Yokokawa, Y. Tyrosinase inhibitors from anacardium occidentale fruits. J. Nat. Prod. 1994, 57, 545–551. [Google Scholar] [CrossRef]
- Kubo, I.; Masuoka, N.; Ha, T.J.; Tsujimoto, K. Antioxidant activity of anacardic acids. Food Chem. 2006, 99, 555–562. [Google Scholar] [CrossRef]
- Kubo, I.; Nihei, K.; Tsujimoto, K. Antibacterial action of anacardic acids against methicillin resistant Staphylococcus aureus (MRSA). J. Agric. Food Chem. 2003, 51, 7624–7628. [Google Scholar] [CrossRef]
- Ferreira, A.; Vinagre, A.; Basrbastefano, V.; Cola, M.; Hiruma, C.; Serra, C.; Vilegas, W.; Souza, A. Estudio de la actividad antiulcerogénica del extracto bruto metanólico de anacardium humile st. Hil. (anacardiaceae). Rev. Cuba. Plant Med. 2004, 9, 69. [Google Scholar]
- Ramos, F.; Osorio, C.; Duque, C.; Cordero, C.; Aristizába, L.; Garzon, F.; Fujimoto, Y. Estudio químico de la nuez del marañón gigante (anacardium giganteum). Rev. Acad. Colomb. Cienc 2004, 28, 565–576. [Google Scholar]
- Leung, E.M.; Tang, P.N.; Ye, Y.; Chan, W. Analysis of 2-alkylcyclobutanones in cashew nut, nutmeg, apricot kernel, and pine nut samples: Re-evaluating the uniqueness of 2-alkylcyclobutanones for irradiated food identification. J. Agric. Food Chem. 2013, 61, 9950–9954. [Google Scholar] [CrossRef]
- Doyle, M.P.; Buchanan, R.L. (Eds.) Food Microbiology: Fundamentals and Frontiers, 4th ed.; ASM Press: Washington, DC, USA, 2013; pp. 765–801. [Google Scholar]
- Prakash, B.; Kedia, A.; Mishra, P.K.; Dubey, N.K. Plant essential oils as food preservatives to control moulds, mycotoxin contamination and oxidative deterioration of agri-food commodities-potentials and challenges. Food Control 2015, 47, 381–391. [Google Scholar] [CrossRef]
- Mogos, G.D. CMS Announces Proposed Rule on Physician Fee Schedule Calculation Method; MGMA Connex: Hobart, Australia, 2006; pp. 12–14. [Google Scholar]
- Prakash, B.; Kujur, A.; Singh, P.P.; Kumar, A.; Yadav, A. Plants-derived bioactive compounds as functional food ingredients and food preservative. Nutr. Food Sci. 2017, 2, 1–7. [Google Scholar]
- Gutiérrez-del-Río, I.; Fernández, J.; Lombó, F. Plant nutraceuticals as antimicrobial agents in food preservation: Terpenoids, polyphenols and thiols. Int. J. Antimicrob. Agents 2018, 52, 309–315. [Google Scholar] [CrossRef]
- Pisoschi, A.M.; Pop, A.; Georgescu, C.; Turcuş, V.; Olah, N.K.; Mathe, E. An overview of natural antimicrobials role in food. Eur. J. Med. Chem. 2018, 143, 922–935. [Google Scholar] [CrossRef]
- Salehi, B.; Valussi, M.; Jugran, A.K.; Martorell, M.; Ramírez-Alarcón, K.; Stojanović-Radić, Z.Z.; Antolak, H.; Kręgiel, D.; Mileski, K.S.; Sharifi-Rad, M. Nepeta species: From farm to food applications and phytotherapy. Trends Food Sci. Technol. 2018, 80, 104–122. [Google Scholar] [CrossRef]
- Sharifi-Rad, M.; Ozcelik, B.; Altın, G.; Daşkaya-Dikmen, C.; Martorell, M.; Ramírez-Alarcón, K.; Alarcón-Zapata, P.; Morais-Braga, M.F.B.; Carneiro, J.N.P.; Alves Borges Leal, A.L.; et al. Salvia spp. Plants-from farm to food applications and phytopharmacotherapy. Trends Food Sci. Technol. 2018, 80, 242–263. [Google Scholar] [CrossRef]
- Salehi, B.; Stojanović-Radić, Z.; Matejić, J.; Sharopov, F.; Antolak, H.; Kręgiel, D.; Sen, S.; Sharifi-Rad, M.; Acharya, K.; Sharifi-Rad, R.; et al. Plants of genus mentha: From farm to food factory. Plants 2018, 7, 70. [Google Scholar] [CrossRef]
- Rai, M.; Pandit, R.; Gaikwad, S.; Kövics, G. Antimicrobial peptides as natural bio-preservative to enhance the shelf-life of food. J. Food Sci. Technol. 2016, 53, 3381–3394. [Google Scholar] [CrossRef] [Green Version]
- Pandey, A.K.; Kumar, P.; Singh, P.; Tripathi, N.N.; Bajpai, V.K. Essential oils: Sources of antimicrobials and food preservatives. Front. Microbiol. 2017, 7, 1–14. [Google Scholar] [CrossRef]
- Barberis, S.; Quiroga, H.G.; Barcia, C.; Talia, J.M.; Debattista, N. Natural Food Preservatives Against Microorganisms; Elsevier: Amsterdam, The Netherlands, 2018; pp. 621–658. [Google Scholar]
- Satoh, M.; Takeuchi, N.; Nishimura, T.; Ohta, T.; Tobinaga, S. Synthesis of anacardic acids, 6-[8(z),11(z)-pentadecadienyl]salicylic acid and 6-[8(z),11(z),14-pentadecatrienyl]salicylic acid. Chem. Pharm. Bull. 2001, 49, 18–22. [Google Scholar] [CrossRef]
- Narasimhan, B.; Panghal, A.; Singh, N.; Dhake, A.S. Efficiency of anacardic acid as preservative in tomato products. J. Food Process. Preserv. 2008, 32, 600–609. [Google Scholar] [CrossRef]
- Himejima, M.; Kubo, I. Antibacterial agents from the cashew anacardium occidentale (anacardiaceae) nut shell oil. J. Agric. Food Chem. 1991, 39, 418–421. [Google Scholar] [CrossRef]
- Yuttana, S.; Kanittada, T.; Suwannahong, K. Antioxidant, antibacterial, and cytotoxicity activities of cashew. Int. J. Green Pharm. 2018, 2018, 229–234. [Google Scholar]
- Alia, A.H.N.A.; Shukri, M.A.M.; Razali, M. Antimicrobial potency of essential oil from cashew (anacardium occidentale linn.) clones. J. Trop. Agric. 2016, 44, 73–80. [Google Scholar]
- Da Silva, R.A.; Liberio, S.A.; do Amaral, F.M.M.; do Nascimento, F.R.F.; Torres, L.M.B.; Neto, V.M.; Guerra, R.N.M. Antimicrobial and antioxidant activity of anacardium occidentale l. Flowers in comparison to bark and leaves extracts. J. Biosci. Med. 2016, 4, 87–89. [Google Scholar]
- Andrade, T.d.J.A.d.S.; Araújo, B.Q.; Citó, A.M.d.G.L.; da Silva, J.; Saffi, J.; Richter, M.F.; Ferraz, A.d.B.F. Antioxidant properties and chemical composition of technical cashew nut shell liquid (tcnsl). Food Chem. 2011, 126, 1044–1048. [Google Scholar] [CrossRef]
- Calo, J.R.; Crandall, P.G.; O’Bryan, C.A.; Ricke, S.C. Essential oils as antimicrobials in food systems—A review. Food Control 2015, 545, 111–119. [Google Scholar] [CrossRef]
- Chaves, M.H.; Citó, A.M.d.G.L.; Lopes, J.A.D.; da Costa, D.A.; de Oliveira, C.A.A.; e Francisco, A.F.C.; Brito Júnior, E.M. Fenóis totais, atividade antioxidante e constituintes químicos de extratos de anacardium occidentale l., anacardiaceae. Braz. J. Pharmacogn. 2010, 20, 106–112. [Google Scholar] [CrossRef]
- Ferreira, P.R.B.; Mendes, C.S.O.; Rodrigues, C.G.; Rocha, J.C.M.; Royo, V.d.A.; Valério, H.M.; Oliveira, D.A.d. Antibacterial activity tannin-rich fraction from leaves of anacardium humile. Ciência Rural 2012, 42, 1861–1864. [Google Scholar] [CrossRef]
- Barbosa-Filho, V.M.; Waczuk, E.P.; Kamdem, J.P.; Abolaji, A.O.; Lacerda, S.R.; da Costa, J.G.M.; de Menezes, I.R.A.; Boligon, A.A.; Athayde, M.L.; da Rocha, J.B.T.; et al. Phytochemical constituents, antioxidant activity, cytotoxicity and osmotic fragility effects of caju (anacardium microcarpum). Ind. Crop. Prod. 2014, 55, 280–288. [Google Scholar] [CrossRef]
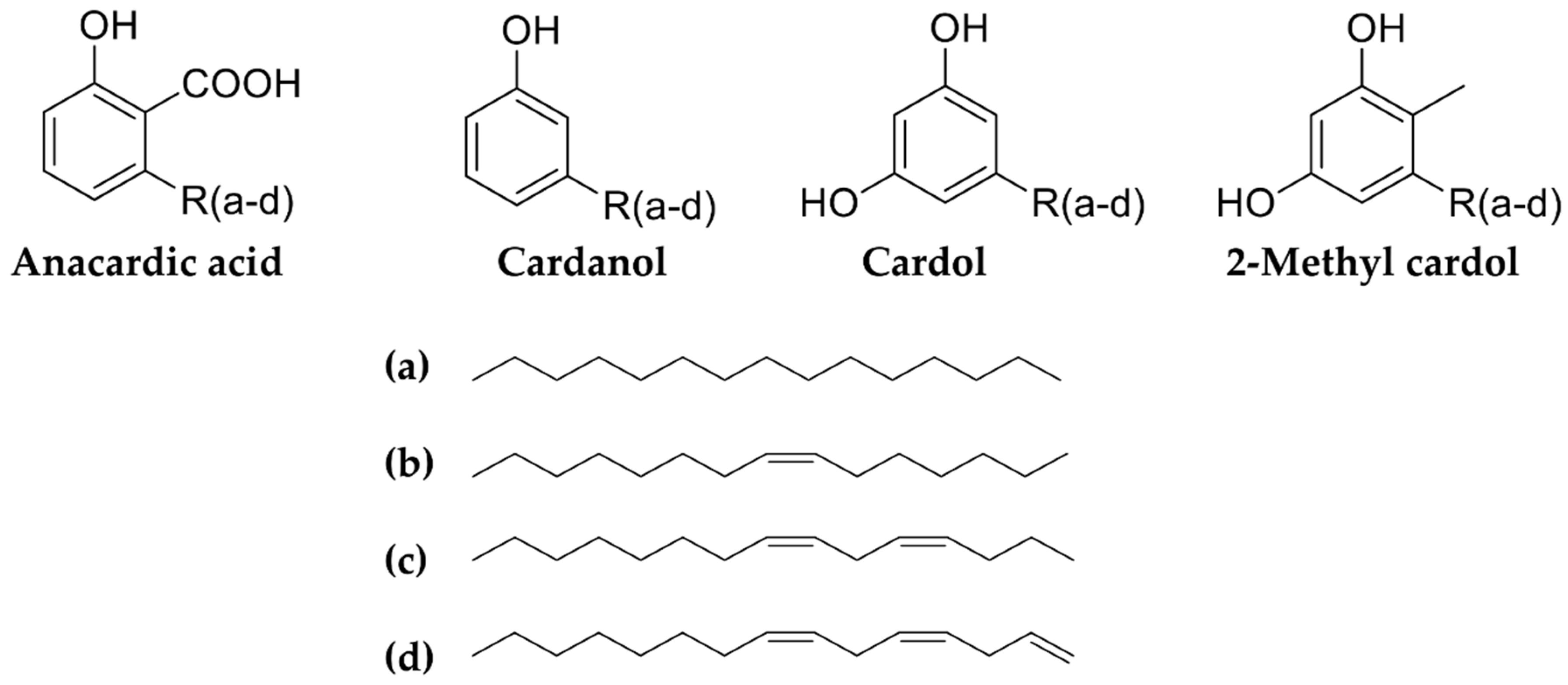
| Country/Area | Amino Acid | Plant Part/Culture/Extract | References |
|---|---|---|---|
| Nigeria, India, Spain | arginine alanine aspartic acid cysteine/cystine glutamic acid glycine histidine isoleucine leucine lysine methionine phenylalanine proline serine threonine tryptophan tyrosin evaline | Good grade and discarded cashew nut meal, cashew nuts, whole and defatted cashew nut flours, Vietnamese, Indian (Kerala origin) Brazilian, and Ivory Coast cashew kernels | [17,20,21,22] |
| Variety/Cultivar | Country/Area | Vitamins/Functional Biofactors | Plant Part/Culture/Extract | References |
|---|---|---|---|---|
| - | Brazil Spain | vitamin C + dehydroascorbic acid) | fresh and processed apple juice, Vietnamese, Indian (Kerala origin) Brazilian, & Ivory Coast cashew kernels | [17,25] |
| M 6/1, Bla 256-1, M 10/4 and M 44/3, Red & Yellow fruited species | Nigeria | vitamin C | cashew apples and kernels, fruit, leaves, stem bark and roots | [24,26] |
| - | Spain | vitamins B1, B5 (pantothenic acid, microbiological) vitamin B6, B8 (biotin, microbiological), B9 (total folate, microbiological), and B12 | Vietnamese, Indian (Kerala origin) Brazilian, and Ivory Coast cashew kernels | [17] |
| Red & Yellow fruited species | Nigeria Spain | vitamins B2 and B3 | fruit, leaves, stem bark and roots, Vietnamese, Indian (Kerala origin) Brazilian, and Ivory Coast cashew kernels | [17,24] |
| - | Indonesia, Spain | vitamin E (tocopherol/α-tocopherol/γ-tocopherol/δ-tocopherol) | kernels, kernels of cashew nut, Vietnamese, Indian (Kerala origin) Brazilian, and Ivory Coast cashew kernels | [4,17,27] |
| - | Spain | vitamin K1 | Vietnamese, Indian (Kerala origin) Brazilian, and Ivory Coast cashew kernels | [17] |
| - | India | m-digallic acid | flowers | [28] |
| - | India | ethylgullute methyl gallute leucocyanidin leucodelphinidin | leaves | [28] |
| - | Indonesia | lutein zeaxanthin | kernels | [4,27] |
| Red & Yellow fruited species | Indonesia, Nigeria | thiamine | kernels, fruit, leaves, stem bark and roots | [4,24,27] |
| Country/Area | Mineral | Plant Part/Culture/Extract | Reference |
|---|---|---|---|
| Spain | calcium iron magnesium potassium phosphorus sodium zinc | Vietnamese, Indian (Kerala origin) Brazilian, and Ivory Coast cashew kernels | [17] |
| Country/Area | Fatty Acids and Esters | Plant Part/Culture/Extract | References |
|---|---|---|---|
| Spain, Indonesia | C18:0 stearic acid | whole and defatted cashew nut flours, kernels, Vietnamese, Indian (Kerala origin) Brazilian, and Ivory Coast cashew kernels | [4,17,20,27] |
| Spain | saturated fatty acid C17:0 heptadecanoic acid C20:0 arachidic acid C22:0 behenic acid C24:0 lignoceric acid monounsaturated fatty acid C20:1 gadoleic acid polyunsaturated fatty acid C18:3n3 linolenic acid trans fatty acid C18:1n9t elaidic acid C18:1n7t vaccenic acid | Vietnamese, Indian (Kerala origin) Brazilian, and Ivory Coast cashew kernels | [17] |
| China | C18:1t trans-oleic acid | cashew nut shell liquid | [30] |
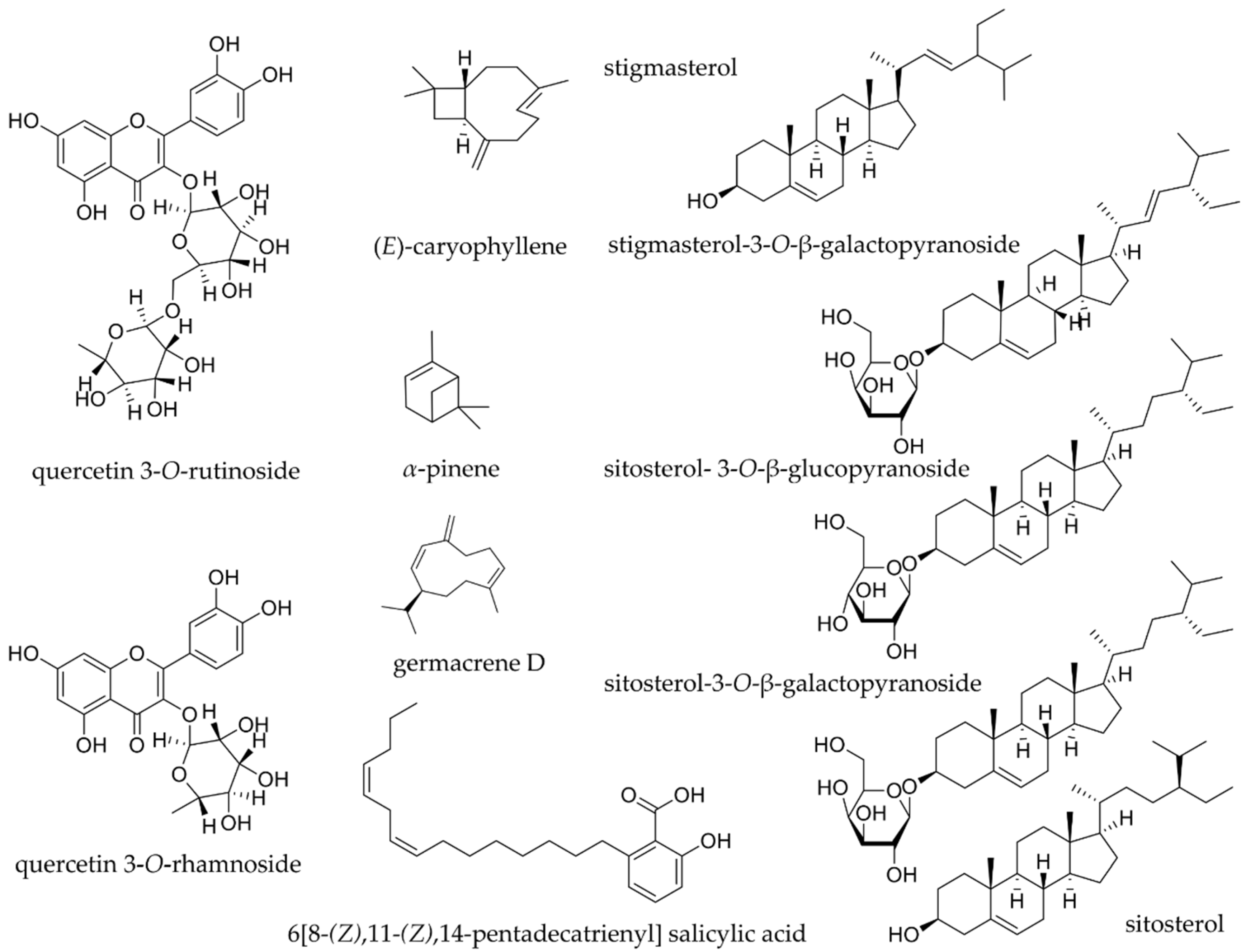
| India | β-sitosterol | leaves and shoot powder, tender leaves | [28,31] |
| India | stigmasterol | leaves and shoot powder | [31] |
| Nigeria | 1-cyclohexylnonene 2-trifluoroacetoxydodecane 3-[(trimethylsilyl)oxy]-17-[o-(phenyl methyl)oxime]-(3α,5α)-androstan -11,17-dione 5-methylbut-2-en-1-yl 3-hydroxy-5-methoxy cyclohexane carboxylate cis-oleic acid cyclohexane carboxylic acid cyclohexanecarboxylic acid, decyl ester decyl ester | cracked bark | [32] |
| Country/Area | Polysaccharides | Plant Part/Culture/Extract | Reference |
|---|---|---|---|
| Brazil | arabinose galactose glucose glucuronic acid mannose rhamnose | crude gum | [33] |
| Variety/Cultivar | Country/Area | Chemical Constituents | Plant Part/Extract | References |
|---|---|---|---|---|
| Phenolic compounds | ||||
| Elongated yellow, elongated red and rounded red | Brazil, Cameroon | 2-hydroxy-6-pentadecylbenzoic acid | leaves, cashew apple, nuts | [37,38] |
| Elongated yellow, elongated red and rounded red | Brazil | 2,6-dihydroxybenzoic acid | cashew apple | [37] |
| - | India | cyaniding peonidin | cashew nut shell liquid | [39] |
| - | India, Cameroon, Poland, Tanzania | anacardic acid | cashew nut shell liquid, cashew seed | [35,38,39,40] |
| - | India, Cameroon, Poland, Tanzania | cardanol (decarboxylated anacardic acid) | cashew nut shell liquid, nuts | [35,38,39,40,41] |
| - | India, Poland, Tanzania | cardol | cashew nut shell liquid | [35,39,40,41] |
| - | Cameroon | salicyclic acid | nuts | [38] |
| - | India | ethyl gallate | flowers | [28] |
| - | India | hyperoside (quercetin 3-galactoside) | leaves, flower | [28] |
| Yellow and red | Brazil, Cote d’Ivoire | gallic acid | cashew apple juice, juice, stem bark | [42,43,44] |
| - | Brazil | 5-hydroxymethylfurfural cinnamic acid protocatechuic acid | cashew apple juice | [44] |
| - | Indonesia, Brazil | (+)-catechin | kernels of cashew nut, bark | [27,42] |
| Yellow and red | Cote d’Ivoire | caffeic acid coumaric acid ferulic acid naringenin | juice | [43] |
| - | Brazil | amentoflavone derivate | leaves | [45] |
| - | Jamaica | stigmast-4-en-3-one stigmast-4-en-3-ol | bark | [46] |
| Yellow and red | Brazil, Malaysia | myricetin-O-glycoside | cashew apple, young tender leaves | [47,48] |
| Yellow and red | Malaysia | kaempferol-3-O-arabinofuranoside kaempferol-3-O-arabinopyranoside kaempferol-3-O-xyloside quercetin-3-O-xyloside unknown quercetin pentose unknown quercetin conjugate | young tender leaves | [48] |
| Yellow and red | Brazil, Malaysia | kaempferol-3-O-glucoside quercetin 3-O-arabinofuranoside | cashew apple, freeze dried, young tender leaves | [47,48] |
| - | Colombia * | 1,2,3-benzenetriol 2-methyl-2-propenoic acid 2,2′-methylenebis(6-(1,1-dimethylethyl)-4-ethyl-pheno 2,2′,6,6′-tetramethyl-4,4′-methylenediphenol hexadecane 2,4-dioctylphenol 2,6-di[p-cyanophenyl]-4-picoline 4-methyl)furan-3-carboxylate 9-octadecenoic acid,12-hydroxy-13- methyl ester methyl 2-[2-(2-ethyl-1,3-dioxolan-2-ylmethyl)-1-hydroxipent-4-enyl]-N-(benzyl) benzenesulfonamide | integument | [49] |
| - | Colombia * | 2-(1,1-dimethylethyl)-4-(1,1,3,3-tetramethylbutyl)phenol | integument, extracts and fractions | [49] |
| - | Malasiya, Brazil | myricetin | cashew shoots, cashew apple | [47,50] |
| Yellow and red | India, Malasiya, Brazil, Cote d’Ivoire | quercetin | flowers, cashew shoots, leaves, cashew apple, juice | [28,43,45,47,50] |
| - | Brazil | 3-O-hexoside of methyl-cyanidin 5-methylcyanidin chloride delphinidin chloride kaempferol rhamnetin O-glycoside quercetin O-glycoside | cashew apple | [47] |
| - | India Brazil | delphinidin | cashew nut shell liquid, cashew apple | [39,47] |
| - | Nigeria | agathisflavone | leaves | [51,52] |
| - | Nigeria | quercetin 3-O-rutinoside | leaves | [51] |
| Yellow and red | Brazil, Malaysia, Nigeria | quercetin 3-O-rhamnoside | cashew apple, freeze dried cashew apple, young tender leaves | [47,48,51] |
| - | Brazil | 5-methylcyanidin 3-O-hexoside myricetin 3-O-arabinofuranoside myricetin 3-O-arabinopyranoside myricetin 3-O-galactoside myricetin 3-O-glucoside myricetin 3-O-xylopyranoside quercetin 3-O-xylo-pyranoside rhamnetin | cashew apple, freeze dried cashew apple | [47] |
| Yellow and red | Brazil, Malaysia | myricetin 3-O-rhamnoside quercetin 3-O-arabino-pyranoside quercetin 3-O-galactoside quercetin 3-O-glucoside | cashew apple, freeze dried, young tender leaves | [47,48] |
| - | Brazil, Costa Rica | quercetin 3-O-arabinopyrannoside | freeze dried cashew apple | [47] |
| Yellow, orange, and red | - | 7-O-methylcyanidin 3-O-β-d-galactopyranoside | peeled cashew apples | [53] |
| - | Spain, Costa Rica | provitamin A | Vietnamese, Indian (Kerala origin) Brazilian, and Ivory Coast cashew kernels, yellow-, orange-, and red-peeled cashew apples | [17,53] |
| clones CCP 76 and EMBRAPA 50 | Brazil | (-)-epicatechin (-)-epigallocatechin | cashew apples, stem bark | [42,54] |
| clones CCP | Brazil | (-)-epicatechin-3-O-gallate (-)-epigallocatechin-O-gallate | cashew apples | [54] |
| Phenolic lipids | ||||
| - | Poland, Tanzania | 2-methyl cardol | cashew nut shell liquid | [35,40] |
| - | Brazil | lycopene | cashew apple | [55] |
| Carotenoids, carotenoid esters | ||||
| - | Indonesia, Brazil, Spain, Costa Rica | β-carotene | kernels, cashew apple, Vietnamese, Indian (Kerala origin) Brazilian, and Ivory Coast cashew kernels, yellow-, orange-, and red-peeled cashew apples | [4,17,27,53] |
| Yellow, orange, and red | Costa Rica | β-cryptoxanthin palmitate | peeled cashew apples | [53] |
| Variety/Cultivar | Country/Area | Chemical Constituents | Plant Part/Culture/Extract | References |
|---|---|---|---|---|
| Red cashew | Nigeria, Bénin Kenya | (E)-β-ocimene | leaves, oil, cashew leaves | [68,69,70] |
| Red cashew | Nigeria, Bénin Brazil | δ-cadinene | leaves, oil, aerial parts | [68,69,71] |
| Red cashew, yellow cashew | Nigeria Spain | palmitic acid | fruits, whole and defatted cashew nut flours, Vietnamese, Indian (Kerala origin) Brazilian, and Ivory Coast cashew kernels | [17,20,68] |
| Red cashew | Nigeria | palmitic oleic acid | fruits | [68] |
| Yellow cashew | Nigeria | 4-hydroxydodecanoic acid lactone (E)-hex-2-enol (Z)-hex-3-enol | fruits | [68] |
| Red cashew | Nigeria | benzyl tiglate methyl salicylate | flowers | [68] |
| - | China | trans-linoleic acid | cashew nut shell liquid | [30] |
| - | Bénin Spain, Indonesia, Brazil | C18:2 linoleic acid | leaves oil, whole and defatted cashew nut flours, kernels of cashew nut, Vietnamese, Indian (Kerala origin) Brazilian, and Ivory Coast cashew kernels | [4,17,20,27,69] |
| - | China | trans-linolenic acid isomers 9 t,11 t-CLA and 10 t,12 t- CLA isomers | cashew nut shell liquid | [30] |
| - | Brazil | 1-butanol 1-hexanol 1-pentanol 2-butoxy-ethanol 2-methyl-2-pentenal 2-methyl butanoic acid 2-hexanol 3-hexanol 3-hydroxy-2-butanone 3-methyl-1-butanol 3-methyl butanoic acid 4-ethyl benzaldehyde 6-methyl 5-hepten-2-one ethyl 2-hydroxy-4-methylpentanoate ethyl 3-methylbutanoate ethyl 3-methylpentanoate ethyl acetate ethyl butanoate ethyl propanoate ethyl trans-2-butenoate ethyl trans-2-hexenoate ethyl trans-3-hexenoate isoamyl acetate methyl 2-butenoate methyl 2-methylene butanoate methyl 2-hexenoate methyl 3-methyl butanoate methyl 3-methyl pentanoate methyl butanoate N-amyl acetate octanal trans-2-hexenal γ-hexalactone δ-octalactone | apple juice | [72] |
| Nanum | Brazil | benzalde hydeethyl pentanoate hexanal | cashew apple, apple juice | [72,73] |
| - | Brazil Bénin | cis-3-hexenol | apple juice, leaves oil | [69,72] |
| Nanum | Brazil Bénin | nonanal | cashew apple, apple juice, leaves oil | [69,72,73] |
| - | Brazil | acetic acid | apple juice, cashew nut, fruit, cashew nut, juice | [72,74] |
| Nanum red cashew | Brazil Nigeria, Bénin | α -copaene | cashew apple, leaves, leaves oil, aerial parts | [68,69,71,73] |
| - | Brazil | (E)-caryophyllene | aerial parts, leaves oil | [71] |
| - | Bénin, Brazil | ɑ-humulene α-muurolene β -selinene δ-selinene aromadendrene bicyclogermacrene germacrene B germacrene D | aerial parts, leaves oil | [69,71] |
| - | Brazil | α-amorphene β-camigrene δ-cadinol | aerial parts | [71] |
| Nanum | Bénin Brazil | ɑ -cadinol γ-cadinene | cashew apple leaves oil, aerial parts | [69,71,73] |
| - | Kenya | (Z)-ocimene alloocimene | cashew leaves | [70] |
| - | India | cardanol (C13) cardanol (C15) cardanol (C17) cardol diethyl phthalate dimethyl anacardate | nut shell liquid | [75] |
| - | Brazil | anacardic acid diene anacardic acid monoene anacardic acid triene cardanol diene cardanol triene cardol diene cardol monoene cardol triene | nut shell liquid | [65] |
| - | USA | 6-n-C15 alkylsalicylic acid | nut shell | [76] |
| - | Nigeria. | agathisflavone | leaves | [77] |
| Esters | ||||
| Nanum | Brazil | butyl 2-methyl-2-butenoate ethyl 2-butenoate ethyl 2-hexenoate ethyl 2-hydroxy-4-methylpentanoate ethyl 2-methyl-2-butenoate ethyl 2-octenoate ethyl 3-methylbutanoate ethyl benzenoacetate ethyl decanoate ethyl hexadecanoate ethyl tetradecanoate hexyl 2-methyl-2-butenoate hexyl benzoatemethyl 2-methyl-2-butenoate methyl 3-methylbutanoate methyl benzoate pentyl isopentanoate E-ethyl 3-hexenoate E-ethyl cinnamate E-methyl cinnamate Z-ethyl 3-hexenoate Z-ethyl cinnamate | cashew apple | [73] |
| Nanum | Brazil | ethyl 2-methylbutanoate ethyl benzoate ethyl hexanoate ethyl octanoate methyl hexanoate | cashew apple, apple juice | [72,73] |
| Lactones | ||||
| Nanum | Brazil | γ -dodecalactone γ-nonalactone | cashew apple | [73] |
| Carboxylic acids | ||||
| Nanum | Brazil | C8:0 octanoic acid C9:0 nonanoic acid C10:0 decanoic acid C14:0 tetradecanoic acid C18:0 octadecanoic acid | cashew apple | [73] |
| Nanum | Brazil Spain | C12: 0 dodecanoic acid | cashew apple Vietnamese, Indian (Kerala origin) Brazilian, and Ivory Coast cashew kernels | [17,73] |
| Nanum | Brazil Bénin Nigeria Spain | C16: 0 hexadecanoic acid | cashew apple, leaves oil, aerial parts, cracked bark, Vietnamese, Indian (Kerala origin) Brazilian, and Ivory Coast cashew kernels | [17,32,69,71,73] |
| - | Nigeria | octadecanoic acid, 2,3-dihydroxypropyl ester | cracked bark | [32] |
| Aldehydes and ketones | ||||
| Yellow cashew Nanum | Nigeria, Brazil | furfural | fruits, cashew apple | [68,73] |
| Nanum | Brazil | 6-methyl-5-hepten-2-one acetophenone decanal phenylacetaldehyded E-2-decenal | cashew apple | [73] |
| Alcohols | ||||
| Nanum | Brazil | l-octanol | apple juice, cashew apple | [72,73] |
| Yellow cashew, Nanum | Nigeria, Brazil | hexadecanol | flowers, cashew apple | [68,73] |
| Nanum | Brazil | octadecanol | cashew apple | [73] |
| Terpenes | ||||
| Nanum | Brazil | 6-cadinene curcufenol geranylketone norisoprenoids o-cymene ɑ-calacorene ɑ-fram-bergamotene ɑ-muurulol ɑ-tujene β-caryophyllene γ-muurulene | cashew apple | [73] |
| Bénin | p-cymenene trans-ɑ-bergamotene | leaves oil | [69] | |
| Nanum | Brazil, Bénin | ɑ-cubebene β-bisabolene cis-ɑ-bergamotene | cashew apple, leaves oil | [69,73] |
| Hydrocarbons | ||||
| Nanum | Brazil | eicosane heptadecane hexadecane naphthalene nonadecane octadecane pentadecane tridecane | cashew apple | [73] |
| Nanum | Brazil | tetradecane | cashew apple, aerial parts | [71,73] |
| EMBRAPA | Brazil | 1-O-trans-cinnamoyl-D-glucopyranose | cashew apple juice | [54] |
| - | Brazil Nigeria Bénin | ɑ-pinene | cashew nut juice, fresh leaves oil, leaves oil | [69,74,78] |
| - | Brazil Bénin | β-pinene | cashew nut, juice, leaves oil | [69,74] |
| Nanum | Bénin, Nigeria Brazil | limonene | cashew apple, fresh leaves oil leaves oil, aerial parts | [69,71,73,78] |
| - | Bénin | 1-epi-cubenol 1-octen-3-ol 1,2-benzene dicarboxylic acid 1,4-dimethylbenzene 1,8-cineole 1,10-diepi-cubenol 2-nonadecanone 3-carene 4a-H,10a-H-guaia-1(5)-6-diene 4b-H,10a-H-guaia-1(5)-6-diene 5-epi-neointermedeol 6,10,14-trimethyl-2-pentadecanone 7,9-diterbutyl-1-oxaspiro-[4,5]-deca-6,9-diene-2,8-dione 7-octadiene-3,6-diol 9-epi-β-caryophyllene 10-epi-cubenol 10-epi-γ-eudesmol 16-kaurene allo-aromadendrene aristolone benzyl benzoate benzyl salycilate borneol bornyl acetate cadina-1,4-diene cadina-3,5-diene camphene camphor carvenone caryophyllene alcohol caryophyllene oxide citronellal citronellol cis-calamenene cis-linalool oxide cis-p-menth-2-en-1-ol chavicol cryptone cubenol cuminaldehyde cyclohexadecane dihydroedulan I dodecanophenone elemol epi-globulol epi-ɑ-cadinol epi-ɑ-muurolol eremophilene ethylbenzene farnesol geraniol geranyl formate germacrene A globulol guaia-6,9-diene hexyl tiglate humulene epoxide II intermedeol juniper camphor linalool m-mentha-1(7),8-diene manoyl oxide mentha-1,4-dien-7-ol menthone methyl chavicol methyl eugenol methyl linolenate mesitylene mintsulfide myrcene p-anisaldehyde p-cymen-7-ol p-cymene p-menth-1-en-9-ol p-mentha-1,5-dien-7-ol p-mentha-1,5-dien-8-ol p-mentha-1(7),2-dien-8-ol p-mentha-1(7),4(8)-diene piperitone phellandral phytol sabinene santolina alcohol salvial-4(14)-en-1-one selina-3,5-diene selina-3,7(11)-diene selina-4(15),7(11)-diene spathulenol terpinen-4-ol terpinolene thymol trans-calamenene trans-carveol trans-2-cyclohexen-1-ol trans-linalool oxide trans-p-menth-2-en-1-ol trans-piperitol trans-phytol trans-sabinol valencene viridiflorol ylanga-2,4(15)-diene zonarene ɑ-cadinene ɑ-fenchol ɑ-fenchyl acetate ɑ-muurolol ɑ-phellandrene ɑ-terpinene ɑ-terpineol ɑ-thujene ɑ-selinene β-acoradiene β-bourbonene β-bulnesene β-cedrene β-cubebene β-elemene β-phellandrene β-terpineol δ-amorphene δ-elemene γ-2-cadinene γ-amorphene γ-elemene γ-gurjunene γ-muurolene γ-pinocarveol γ-terpinene (E)-2-decenal 2,6-dimethyl-1 (E)-3-hexenyl butyrate (E)-3-hexenyl isovalerate (E)-β-farnesene (E)-β-ionone (E,E)-ɑ-farnesene (Z)-3-hexenyl-2-methyl butyrate (Z)-3-hexenyl benzoate (Z)-3-hexenyl isovalerate (Z)-3-hexenyl (Z)-3-hexenoate | leaves oil | [69] |
| - | Bénin, Kenya | (Z)-3-hexenyl butyrate | leaves oil, cashew leaves | [69,70] |
| - | Nigeria Bénin | ɑ-ylangene | fresh leaves oil, leaves oil | [69,78] |
| - | Brazil | 2-furan-methanol butanoic acid | cashew nut, fruit, cashew nut juice | [74] |
| - | Brazil | 1,3-dihydroxy-2-propanone 2-octil-cyclopropaneoctanal acetic acid 1-butanol ester acetic acid 2-propen-1-ol ester acetic acid 3-hexen-1-ol ester amfibina benzoic acid butanoic acid ethyl ester canfene hexanoic acid 2-propenyl ester hexanoic acid ethyl ester oxalic acid ammonium ester pentanoic acid 3-methylbutyl ester pentanoic acid ethyl ester sulphuric dioxide trans-caryophyllene | cashew nut, juice | [74] |
| - | Nigeria | 17-octadecynoic acid rostane steroid 3-[(trimethylsilyl)oxy]-17-[o-(phenylmethyl)oxime]-(3α,5α)-androstane-11,17-dione | cracked bark | [32] |
| Species | Variety/Cultivar | Country/Area | Chemical Constituents | Plant Part/Culture/Extract | Reference |
|---|---|---|---|---|---|
| Hydrocarbon | |||||
| A. fraxinifolium | - | Brazil | myrcene α-terpinolene δ-2-carene (E)-β-ocimene (Z)-β-ocimene | aerial parts | [71] |
| A. fraxinifolium A. humile | - | Brazil | α-pinene | aerial parts | [71] |
| A. humile | - | Brazil | β-pinene | aerial parts | [71] |
| A. fraxinifolium A. humile A. occidentale | - | Brazil | limonene | aerial parts | [71] |
| Hydrocarbon Sesquiterpenes | |||||
| A. fraxinifolium | - | Brazil | viridiflorene α-amorphene (E,E)-α-farnesene | aerial parts | [71] |
| A. fraxinifolium A. humile | - | Brazil | alloaromadendrene aromadendrene germacrene D α-copaene γ-cadinene | aerial parts | [71] |
| A. humile | - | Brazil | bicyclogermacrene germacrene A α-gurjunene α-humulene α-muurolene β-selinene δ-cadinene (E)-caryophyllene | aerial parts | [71] |
| A. occidentale | - | Brazil | β-camigrene | aerial parts | [71] |
| Oxygenated Sesquiterpenes | |||||
| A. fraxinifolium | - | Brazil | ledol spathulenol | aerial parts | [71] |
| A. fraxinifolium A. humile | - | Brazil | globulol viridiflorol | aerial parts | [71] |
| A. humile | - | Brazil | epiglobulol α-cadinol β-caryophyllene oxide | aerial parts | [71] |
| A. humile | - | Brazil | α-pinene (E)-caryophyllene | leaves oil | [71] |
| Country/Area | Chemical Constituents | Plant Part/Extract | References |
|---|---|---|---|
| Indonesia | 2-methyl-5-pentadecylresorcinol 2-methyl-5[8(2), 11(2)-pentadecadienyll resorcinol 2-methyl-5[8(2), 11(2),14-pentadecatrienyll resorcinol 2-methyl-5[8(Z)-pentadecenyl] resorcinol3-pentadecylphenol 3-[8(2), 11 (2)-pentadecadienyll-phenol 3-[8(Z)-pentadecenylIphenol 3-18(2), 1 1(2), 14-pentadecatrienyllphenol 5-pentadecylresorcinol5-[8(2), 11(2)-pentadecadienyllresorcinol 5-[8(2), 11(Z),1 4-pentadecatrienyllresorcinol 6-pentadecylsalicylic acid 6-[8(2), 11 (21,14-pentadecatrienyllsalicylic acid 6-[8(Z)-pentadeccnyl]salicylic acid 6-18(2), 11(2)-pentadecadienyl]salicylic acid 14,5-[8(Z)-pentadecenyl]-resorcinol | cashew nut shell | [86] |
| Brazil | anacardic acid | fruit extracts, fruit | [87,88,89,90] |
| Brazil | acid 6-pentadec(en) salicylic | fruit | [90] |
| Brazil | alkyl phenols cardols | fruit | [87] |
| Brazil | acid 6-alqu(en) ilsalicylic | bark extract, fruit | [91] |
| Brazil | cardanols | fruit extracts, fruit | [87,88,89] |
| Brazil | cardol 2-methyl-cardol | fruit extracts | [88] |
| Colombia * | bi flavonoids | total extract | [92] |
| Colombia ** | acid 6-alkilsalic alkyl resorcinols | fruit extracts | [93] |
| India | occidentoside β-sitosterol | nut shells | [60] |
| Hong Kong | 2-alkylcyclobutanones | cashew nut samples | [94] |
| Cytotoxic compounds | |||
| Nigeria | anacardicin zoapatanolide A [1,2-bis(2,6-dimethoxy-4-methoxycarbonylphenyl)ethane] methyl gallate | leaves | [52] |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Salehi, B.; Gültekin-Özgüven, M.; Kırkın, C.; Özçelik, B.; Morais-Braga, M.F.B.; Carneiro, J.N.P.; Bezerra, C.F.; Silva, T.G.d.; Coutinho, H.D.M.; Amina, B.; et al. Anacardium Plants: Chemical,Nutritional Composition and Biotechnological Applications. Biomolecules 2019, 9, 465. https://doi.org/10.3390/biom9090465
Salehi B, Gültekin-Özgüven M, Kırkın C, Özçelik B, Morais-Braga MFB, Carneiro JNP, Bezerra CF, Silva TGd, Coutinho HDM, Amina B, et al. Anacardium Plants: Chemical,Nutritional Composition and Biotechnological Applications. Biomolecules. 2019; 9(9):465. https://doi.org/10.3390/biom9090465
Chicago/Turabian StyleSalehi, Bahare, Mine Gültekin-Özgüven, Celale Kırkın, Beraat Özçelik, Maria Flaviana Bezerra Morais-Braga, Joara Nalyda Pereira Carneiro, Camila Fonseca Bezerra, Teresinha Gonçalves da Silva, Henrique Douglas Melo Coutinho, Benabdallah Amina, and et al. 2019. "Anacardium Plants: Chemical,Nutritional Composition and Biotechnological Applications" Biomolecules 9, no. 9: 465. https://doi.org/10.3390/biom9090465
APA StyleSalehi, B., Gültekin-Özgüven, M., Kırkın, C., Özçelik, B., Morais-Braga, M. F. B., Carneiro, J. N. P., Bezerra, C. F., Silva, T. G. d., Coutinho, H. D. M., Amina, B., Armstrong, L., Selamoglu, Z., Sevindik, M., Yousaf, Z., Sharifi-Rad, J., Muddathir, A. M., Devkota, H. P., Martorell, M., Jugran, A. K., ... Cho, W. C. (2019). Anacardium Plants: Chemical,Nutritional Composition and Biotechnological Applications. Biomolecules, 9(9), 465. https://doi.org/10.3390/biom9090465









