Delivery of Iron Oxide Nanoparticles into Primordial Germ Cells in Sturgeon
Abstract
1. Introduction
2. Materials and Methods
2.1. Ethics Statement
2.2. Fish Source, Preparation of Embryos and Sample Collection
2.3. Microinjection of Iron-Oxide Nanoparticles
2.4. Micro-CT Imaging
2.5. Statistical Analysis
3. Results
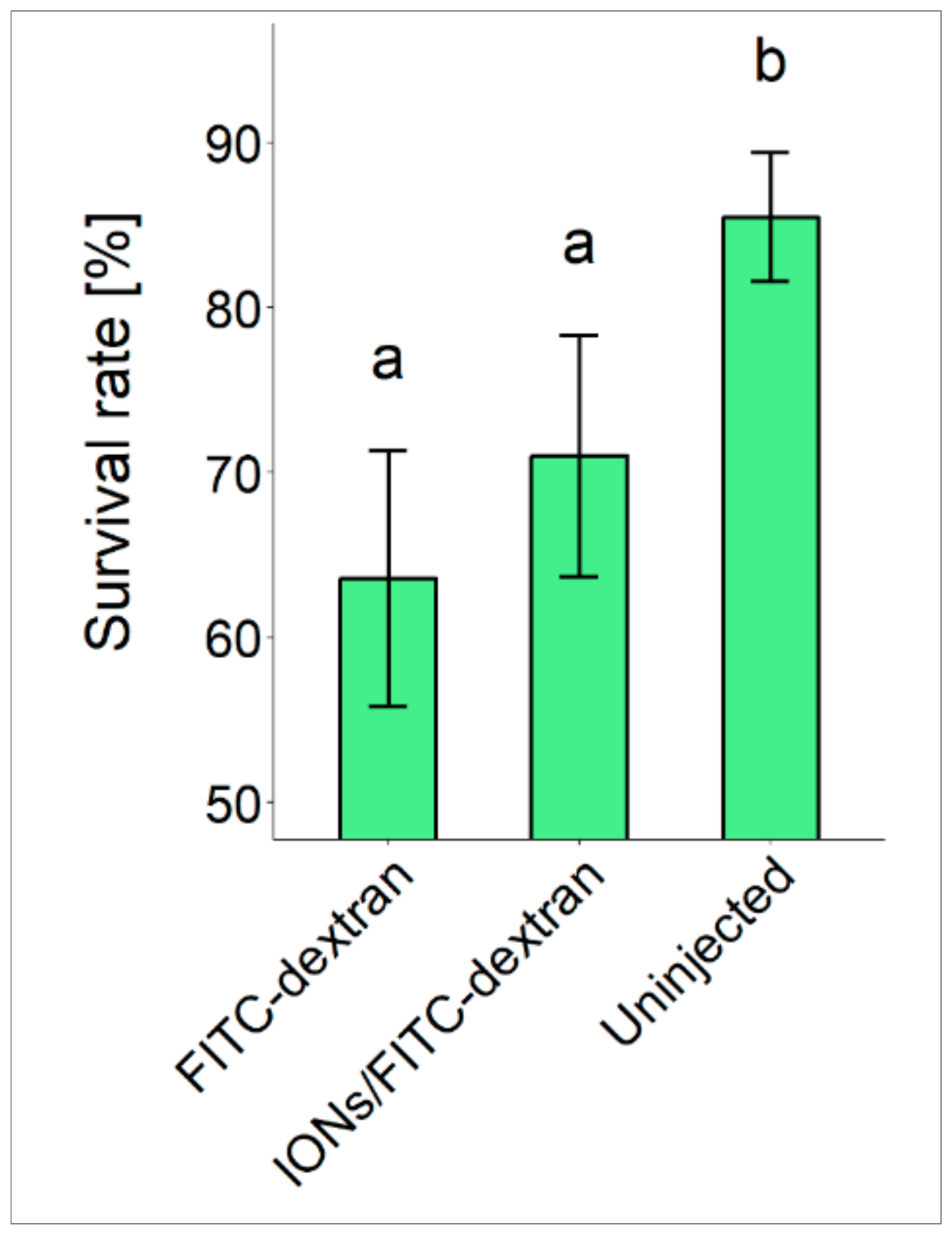
3.1. Fertilization, Hatching and Survival Rates
3.2. Delivery of IONs into PGCs
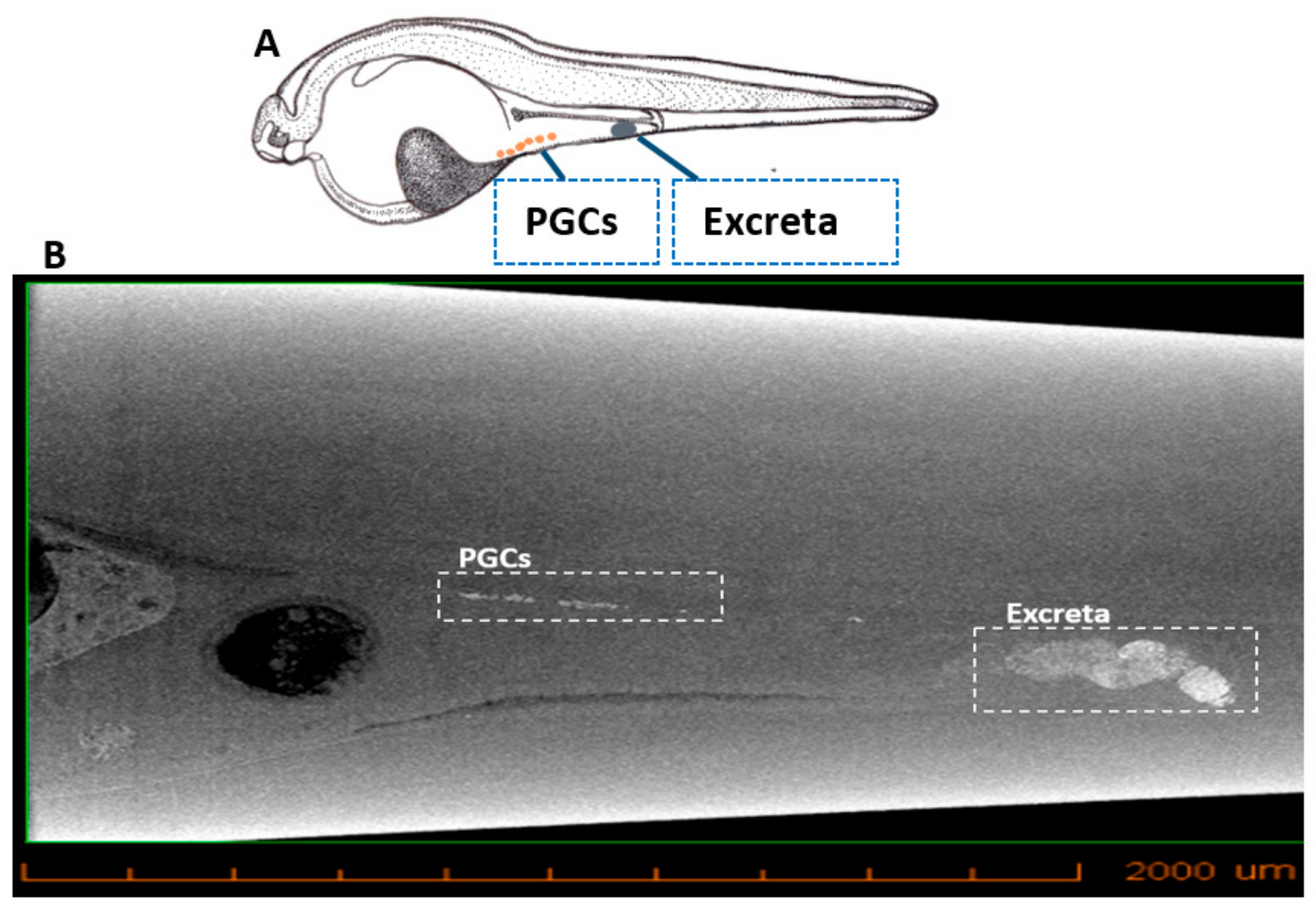
3.3. Micro-CT Imaging
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Johnston, R.L.; Wilcoxon, J.P. Frontiers of Nanoscience; Elsevier: Oxford, UK, 2012. [Google Scholar]
- McNamara, K.; Tofail, S.A.M. Nanosystems: The use of nanoalloys, metallic, bimetallic, and magnetic nanoparticles in biomedical applications. Phys. Chem. Chem. Phys. 2015, 17, 27981–27995. [Google Scholar] [CrossRef] [PubMed]
- Buzea, C.; Pacheco, I.I.; Robbie, K. Nanomaterials and nanoparticles: Sources and toxicity. Biointerphases 2007, 2, MR17–MR71. [Google Scholar] [CrossRef] [PubMed]
- Hassan, S. A review on Nanoparticles: Their synthesis and types. Res. J. Recent. Sci. 2015, 4, 9–11. [Google Scholar]
- Machado, S.; Pacheco, J.; Nouws, H.; Albergaria, J.T.; Delerue-Matos, C.; Nouws, H. Characterization of green zero-valent iron nanoparticles produced with tree leaf extracts. Sci. Total. Environ. 2015, 533, 76–81. [Google Scholar] [CrossRef] [PubMed]
- Oberdörster, G.; Oberdörster, E.; Oberdörster, J. Nanotoxicology: An Emerging Discipline Evolving from Studies of Ultrafine Particles. Environ. Health Perspect. 2005, 113, 823–839. [Google Scholar] [CrossRef] [PubMed]
- Cardoso, V.F.; Francesko, A.; Ribeiro, C.; Bañobre-López, M.; Martins, P.; Lanceros-Mendez, S. Advances in Magnetic Nanoparticles for Biomedical Applications. Adv. Healthc. Mater. 2018, 7, 1–35. [Google Scholar] [CrossRef]
- Khanna, L.; Verma, N.; Tripathi, S. Burgeoning tool of biomedical applications-Superparamagnetic nanoparticles. J. Alloy Compd. 2018, 752, 332–353. [Google Scholar] [CrossRef]
- Xie, W.; Guo, Z.; Gao, F.; Gao, Q.; Wang, D.; Liaw, B.S.; Cai, Q.; Sun, X.; Wang, X.; Zhao, L. Shape-, size-and structure-controlled synthesis and biocompatibility of iron oxide nanoparticles for magnetic theranostics. Theranostics 2018, 8, 3284–3307. [Google Scholar] [CrossRef]
- A Pankhurst, Q.; Connolly, J.; Jones, S.K.; Dobson, J. Applications of magnetic nanoparticles in biomedicine. J. Phys. D Appl. Phys. 2003, 36, R167–R181. [Google Scholar] [CrossRef]
- Huber, D.L. Synthesis, Properties, and Applications of Iron Nanoparticles. Small 2005, 36, 482–501. [Google Scholar] [CrossRef]
- Vorotnikova, E.; Ivkov, R.; Foreman, A.; Tries, M.; Braunhut, S.J. The magnitude and time-dependence of the apoptotic response of normal and malignant cells subjected to ionizing radiation versus hyperthermia. Int. J. Radiat. Biol. 2006, 82, 549–559. [Google Scholar] [CrossRef] [PubMed]
- Harmon, B.; Takano, Y.; Winterford, C.; Gobe, G. The Role of Apoptosis in the Response of Cells and Tumours to Mild Hyperthermia. Int. J. Radiat. Biol. 1991, 59, 489–501. [Google Scholar] [CrossRef] [PubMed]
- Otte, J. Hyperthermia in cancer therapy. Eur. J. Pediatr. 1988, 147, 560–569. [Google Scholar] [CrossRef] [PubMed]
- Issels, R.D. Hyperthermia adds to chemotherapy. Eur. J. Cancer. 2008, 44, 2546–2554. [Google Scholar] [CrossRef] [PubMed]
- Bednarikova, Z.; Marek, J.; Demjen, E.; Dutz, S.; Mocanu, M.M.; Wu, J.W.; Wang, S.S.S.; Gazova, Z.; Mocanu, M.M. Effect of nanoparticles coated with different modifications of dextran on lysozyme amyloid aggregation. J. Magn. Magn. Mater. 2019, 473, 1–6. [Google Scholar] [CrossRef]
- Guzman, R.; Uchida, N.; Bliss, T.M.; He, D.; Christopherson, K.K.; Stellwagen, D.; Capela, A.; Greve, J.; Malenka, R.C.; Moseley, M.E.; et al. Long-term monitoring of transplanted human neural stem cells in developmental and pathological contexts with MRI. Proc. Natl. Acad. Sci. USA 2007, 104, 10211–10216. [Google Scholar] [CrossRef] [PubMed]
- Hoehn, M.; Küstermann, E.; Blunk, J.; Wiedermann, D.; Trapp, T.; Wecker, S.; Föcking, M.; Arnold, H.; Hescheler, J.; Fleischmann, B.K.; et al. Monitoring of implanted stem cell migration in vivo: A highly resolved in vivo magnetic resonance imaging investigation of experimental stroke in rat. Proc. Natl. Acad. Sci. USA 2002, 99, 16267–16272. [Google Scholar] [CrossRef]
- Bemis, W.E.; Kynard, B. Sturgeon rivers: An introduction to acipenseriform biogeography and life history. Environ. Biol. Fishes 1997, 48, 167–183. [Google Scholar] [CrossRef]
- Pikitch, E.K.; Doukakis, P.; Lauck, L.; Chakrabarty, P.; Erickson, D.L. Status, trends and management of sturgeon and paddlefish fisheries. Fish Fish. 2005, 6, 233–265. [Google Scholar] [CrossRef]
- Billard, R.; Guillaume, L. Biology and conservation of sturgeon and paddlefish. Rev. Fish. Biol. Fish. 2001, 1, 355–392. [Google Scholar]
- Ludwig, A.; Belfiore, N.M.; Pitra, C.; Svirsky, V.; Jenneckens, I. Genome duplication events and functional reduction of ploidy levels in sturgeon (Acipenser, Huso and Scaphirhynchus). Genet 2001, 158, 1203–1215. [Google Scholar]
- Zhang, H.; Wei, Q.W.; Kyanrd, B.E.; Du, H.; Yang, D.G.; Chen, X.H. Spatial structure and bottom characteristics of the only remaining spawning area of Chinese sturgeon in the Yangtze River. J. Appl. Ichthyol. 2011, 27, 251–256. [Google Scholar] [CrossRef]
- Hildebrand, L.; McLeod, C.; McKenzie, S. Status and management of white sturgeon in the Columbia River in British Columbia, Canada: An overview. J. Appl. Ichthyol. 1999, 15, 164–172. [Google Scholar] [CrossRef]
- Dettlaff, T.A.; Ginsburg, A.S.; Schmalhausen, O.I. Sturgeon Fishes: Developmental Biology and Aquaculture; Springer: New York, NY, USA, 1993. [Google Scholar]
- Ohinata, Y. A Signaling Principle for the Specification of the Germ Cell Lineage in Mice. Cell 2009, 137, 571–584. [Google Scholar] [CrossRef] [PubMed]
- Saito, T.; Pšenička, M. Novel Technique for Visualizing Primordial Germ Cells in Sturgeons (Acipenser ruthenus, A. gueldenstaedtii, A. baerii, and Huso huso). Biol. Reprod. 2015, 93, 96. [Google Scholar] [PubMed]
- Ewen-Campen, B.; Schwager, E.E.; Extavour, C.G.; Ewen-Campen, B.; Ewen-Campen, B. The molecular machinery of germ line specification. Mol. Reprod. Dev. 2009, 77, 3–18. [Google Scholar] [CrossRef]
- Baloch, A.R.; Franěk, R.; Saito, T.; Pšenička, M. Dead-end (dnd) protein in fish—A review. Fish Physiol. Biochem. 2019, 1–8. [Google Scholar] [CrossRef]
- Lewis, Z.R.; McClellan, M.C.; Postlethwait, J.H.; Cresko, W.A.; Kaplan, R.H. Female-specific increase in primordial germcells marks sex differentiation in threespine stickleback (Gasterosteus aculeatus). J. Morphol. 2008, 269, 909–921. [Google Scholar] [CrossRef]
- Saito, D.; Morinaga, C.; Aoki, Y.; Nakamura, S.; Mitani, H.; Furu-tani-Seiki, M.; Kondoh, H.; Tanaka, M. Proliferation of germ cells during gonadal sex differentiation in medaka: In-sights from germ cell-depleted mutant zenzai. Dev. Biol. 2007, 310, 280–290. [Google Scholar] [CrossRef]
- Tzung, K.-W.; Goto, R.; Saju, J.M.; Sreenivasan, R.; Saito, T.; Arai, K.; Yamaha, E.; Hossain, M.S.; Calvert, M.E.; Orban, L. Early Depletion of Primordial Germ Cells in Zebrafish Promotes Testis Formation. Stem Cell Rep. 2015, 5, 156. [Google Scholar] [CrossRef]
- Yoshizaki, G.; Lee, S. Production of live fish derived from frozen germ cells via germ cell transplantation. Stem. Cell Res. 2018, 29, 103–110. [Google Scholar] [CrossRef] [PubMed]
- Yamaha, E.; Saito, T.; Goto-Kazeto, R.; Arai, K. Developmental biotechnology for aquaculture, with special reference to surrogate production in teleost fishes. J. Sea Res. 2007, 58, 8–22. [Google Scholar] [CrossRef]
- Saito, T.; Pšenička, M.; Goto, R.; Adachi, S.; Inoue, K.; Arai, K.; Yamaha, E. The Origin and Migration of Primordial Germ Cells in Sturgeons. PLoS ONE 2014, 9, e86861. [Google Scholar] [CrossRef] [PubMed]
- Maeda, H.; Greish, K.; Fang, J. Polymer Therapeutics II. The EPR Effect and Polymeric Drugs: A Paradigm Shift for Cancer Chemotherapy in the 21st Century; Springer: Berlin/Heidelberg, Germany, 2006. [Google Scholar]
- Krøvel, A.V.; Olsen, L.C. Expression of avas:EGFPtransgene in primordialgerm cells of the zebrafish. Mech. Dev. 2002, 116, 141–150. [Google Scholar] [CrossRef]
- Tanaka, M.; Kinoshita, M.; Kobayashi, D.; Nagahama, Y. Establishment ofmedaka (Oryzias latipes) transgenic lines with the expression of greenfluorescent protein fluorescence exclusively in germ cells: A useful modelto monitor germ cells in a live vertebrate. Proc. Natl. Acad. Sci. USA 2001, 98, 2544–2549. [Google Scholar] [CrossRef]
- Yoshizaki, G.; Takeuchi, Y.; Sakatani, S.; Takeuchi, T. Germ cell-specific expression of green fluorescent protein in transgenic rainbow trout under control of the rainbow trout vasa-like gene promoter. Int. J. Dev. Biol. 2000, 44, 323–326. [Google Scholar] [PubMed]
- Zhu, X.; Tian, S.; Cai, Z. Toxicity Assessment of Iron Oxide Nanoparticles in Zebrafish (Danio rerio) Early Life Stages. PLoS ONE 2012, 7, e46286. [Google Scholar] [CrossRef]
- Di Corato, R.; Espinosa, A.; Lartigue, L.; Tharaud, M.; Chat, S.; Pellegrino, T.; Ménager, C.; Gazeau, F.; Wilhelm, C. Magnetic hyperthermia efficiency in the cellular environment for different nanoparticle designs. Biomaterials 2014, 35, 6400–6411. [Google Scholar] [CrossRef]
- Kolosnjaj-Tabi, J.; Javed, Y.; Lartigue, L.; Volatron, J.; Elgrabli, D.; Marangon, I.; Pugliese, G.; Caron, B.; Figuerola, A.; Luciani, N.; et al. The One Year Fate of Iron Oxide Coated Gold Nanoparticles in Mice. ACS Nano 2015, 9, 7925–7939. [Google Scholar] [CrossRef]
- Hedayatnasab, Z.; Abnisa, F.; Wan Daud, W.M.A. Investigation properties of superparamagnetic nanoparticles and magnetic field-dependent hyperthermia therapy. IOP Conf. Ser. Mater. Sci. Eng. 2017, 334, 012042. [Google Scholar] [CrossRef]
- Grandi, G.; Chicca, M. Histological and ultrastructural investigation of early gonad development and sex differentiation in Adriatic sturgeon (Acipenser naccarii, Acipenseriformes, Chondrostei). J. Morphol. 2008, 269, 1238–1262. [Google Scholar] [CrossRef]
- Linhartová, Z.; Saito, T.; Kaspar, V.; Rodina, M.; Praskova, E.; Hagihara, S.; Pšenička, M. Sterilization of sterlet Acipenser ruthenus by using knock-down agent, antisense morpholino oligonucleotide, against dead end gene. Theriogenology 2015, 84, 1246–1255. [Google Scholar] [CrossRef]
- Psenicka, M.; Saito, T.; Rodina, M.; Dzyuba, B. Cryopreservation of early stage Siberian sturgeon Acipenser baerii germ cells, comparison of whole tissue and dissociated cells. Cryobiology 2016, 72, 119–122. [Google Scholar] [CrossRef]
- Saito, T.; Guralp, H.; Iegorova, V.; Rodina, M.; Psenicka, M. Elimination of primordial germ cells in sturgeon embryos by ultraviolet irradiation. Biol. Reprod. 2018, 99, 556–564. [Google Scholar] [CrossRef]
- Baloch, A.R.; Franěk, R.; Tichopád, T.; Fučíková, M.; Rodina, M.; Pšenička, M. Dnd1 Knockout in Sturgeons by CRISPR/Cas9 Generates Germ Cell Free Host for Surrogate Production. Animals 2019, 9, 174. [Google Scholar] [CrossRef]
- Xie, X.; Li, P.; Pšenička, M.; Ye, H.; Steinbach, C.; Li, C.; Wei, Q. Optimization of In Vitro Culture Conditions of Sturgeon Germ Cells for Purpose of Surrogate Production. Animals 2019, 9, 106. [Google Scholar] [CrossRef]

© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Baloch, A.R.; Fučíková, M.; Rodina, M.; Metscher, B.; Tichopád, T.; Shah, M.A.; Franěk, R.; Pšenička, M. Delivery of Iron Oxide Nanoparticles into Primordial Germ Cells in Sturgeon. Biomolecules 2019, 9, 333. https://doi.org/10.3390/biom9080333
Baloch AR, Fučíková M, Rodina M, Metscher B, Tichopád T, Shah MA, Franěk R, Pšenička M. Delivery of Iron Oxide Nanoparticles into Primordial Germ Cells in Sturgeon. Biomolecules. 2019; 9(8):333. https://doi.org/10.3390/biom9080333
Chicago/Turabian StyleBaloch, Abdul Rasheed, Michaela Fučíková, Marek Rodina, Brian Metscher, Tomáš Tichopád, Mujahid Ali Shah, Roman Franěk, and Martin Pšenička. 2019. "Delivery of Iron Oxide Nanoparticles into Primordial Germ Cells in Sturgeon" Biomolecules 9, no. 8: 333. https://doi.org/10.3390/biom9080333
APA StyleBaloch, A. R., Fučíková, M., Rodina, M., Metscher, B., Tichopád, T., Shah, M. A., Franěk, R., & Pšenička, M. (2019). Delivery of Iron Oxide Nanoparticles into Primordial Germ Cells in Sturgeon. Biomolecules, 9(8), 333. https://doi.org/10.3390/biom9080333





