Solidago virgaurea L.: A Review of Its Ethnomedicinal Uses, Phytochemistry, and Pharmacological Activities
Abstract
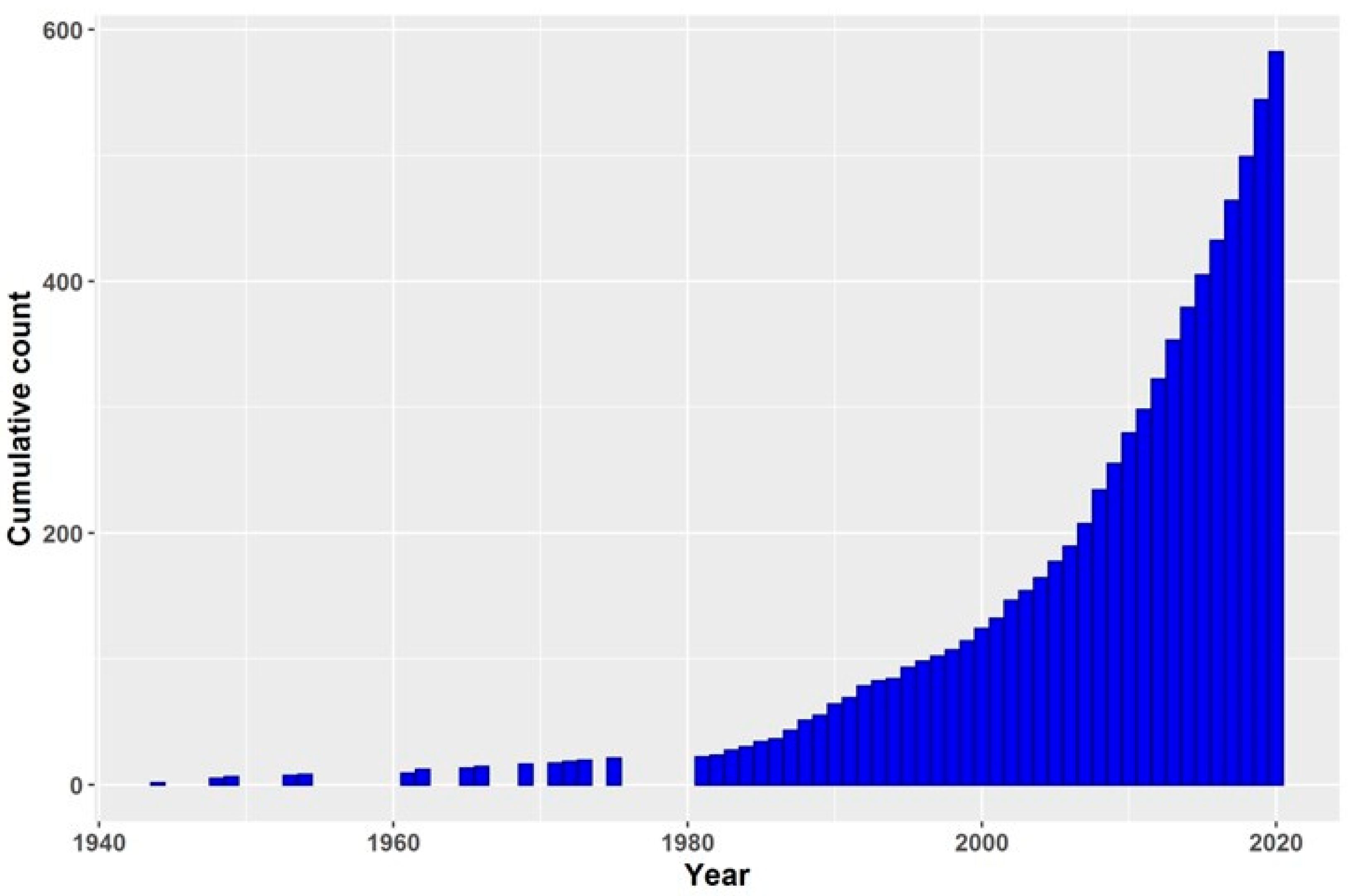
1. Introduction
2. General Description, Taxonomy, and Distribution
3. Synonyms and Common Names
4. Ethnomedicinal Uses
4.1. Germany
4.2. Czech Republic
4.3. Poland
4.4. Russian Federation
4.5. Ukraine
4.6. Bulgaria
4.7. Romania and the Republic of Moldova
4.8. Korea
4.9. China
4.10. Other Uses
5. Phytochemistry
| Chemical Compounds | Place/Country of Collection | References |
|---|---|---|
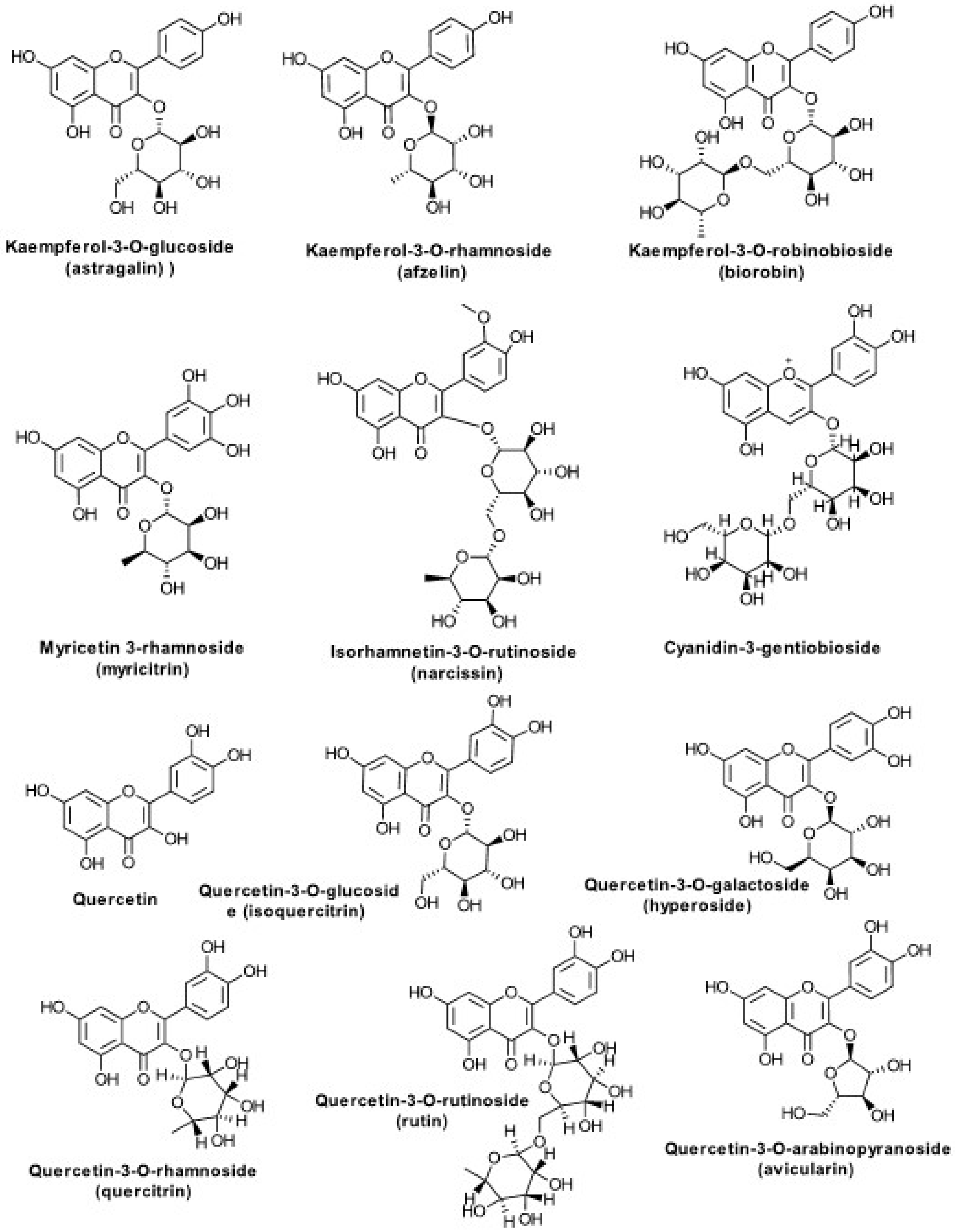
| Flavonoids (Figure 3) | ||
| Quercetin, Quercetin-3-O-glucoside (isoquercitrin) Quercetin-3-O-galactoside (hyperoside) Quercetin-3-O-rhamnoside (quercitrin) Quercetin-3-O-rutinoside (rutin) Quercetin-3-O-arabinopyranoside (avicularin) Kaempferol-3-O-glucoside (astragalin) Kaempferol-3-O-rhamnoside (afzelin) Kaempferol-3-O-rutinoside (nicotiflorin) Kaempferol-3-O-robinobioside (biorobin) Myricetin 3-rhamnoside (myricitrin) Isorhamnetin-3-O-rutinoside (narcissin) Cyanidin-3-gentiobioside mono-C-glycosylflavones (?) di-C-glycosylflavones (?) | Poland, Italy, Hungary, Korea, Romania, Lithuania | Fuchs L. [82], Budzianowski J. et al. [61], Borkowski B. and Skrzypczakowa L. [60], Chodera A. et al. [65], Roslon W. et al. [59], Pietta P. et al. [62], Apáti P. et al. [83], Choi SZ. et al. [49], Tamas M. [63], Dobjanschi L. et al. [45,64], Kraujaliene V. et al. [84] |
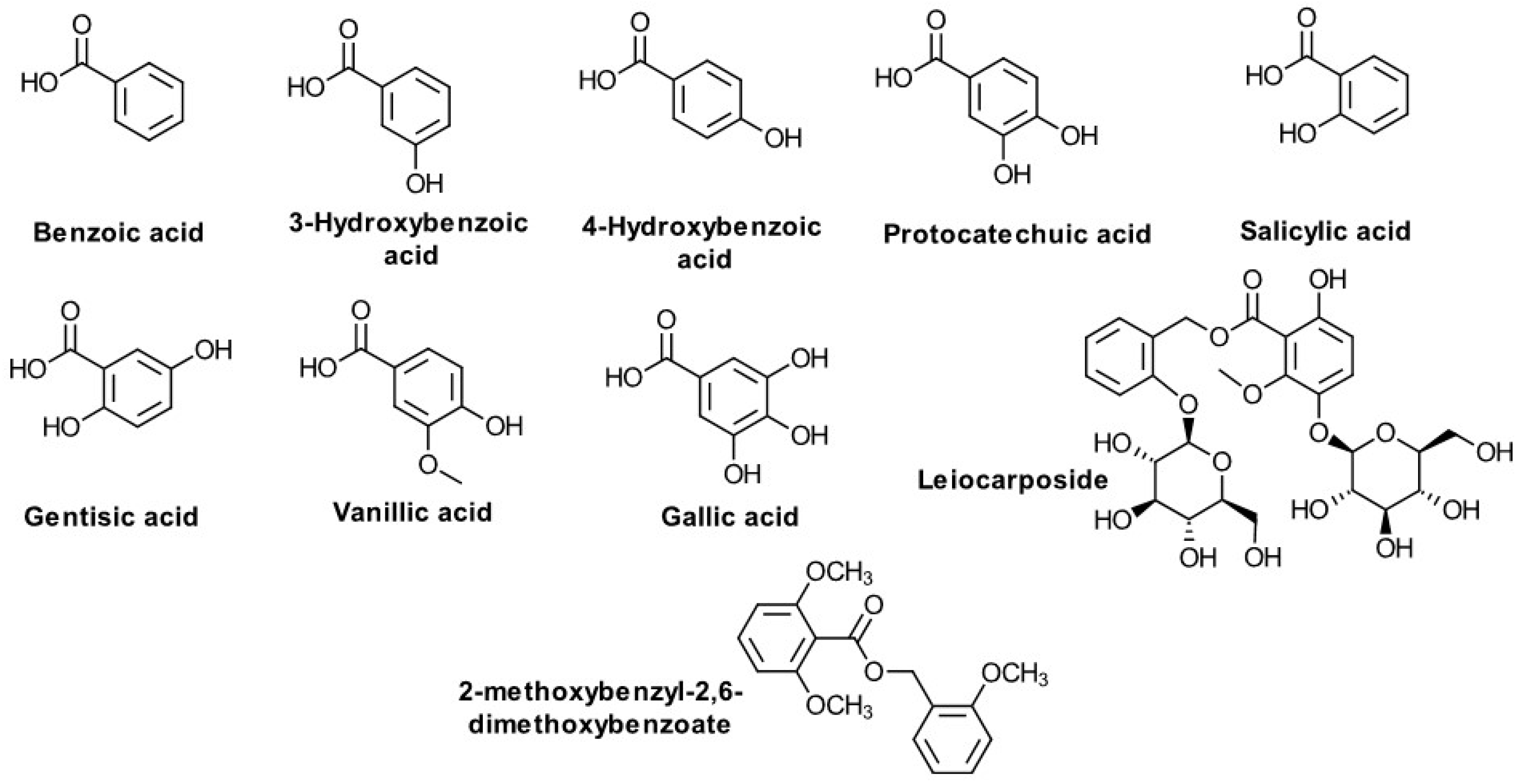
| C6-C1 Compounds (Figure 4) | ||
| Benzoic acid 3-Hydroxybenzoic acid 4-Hydroxybenzoic acid 3,4-Dihydroxybenzoic (protocatechuic) acid Salicylic acid Gentisic acid Vanillic acid Gallic acid Leiocarposide 2-methoxybenzyl-2,6-dimethoxybenzoate | Poland Egypt Korea | Kalemba D. [85], Abdel Motaal A. et al. [10], Choi SZ. et al. [49], Thiem, B. et al. [58], Bajkacz S. et al. [86], Sung JH et al. [50], |
| C6-C2 and C6-C3 Compounds (Figure 5) | ||
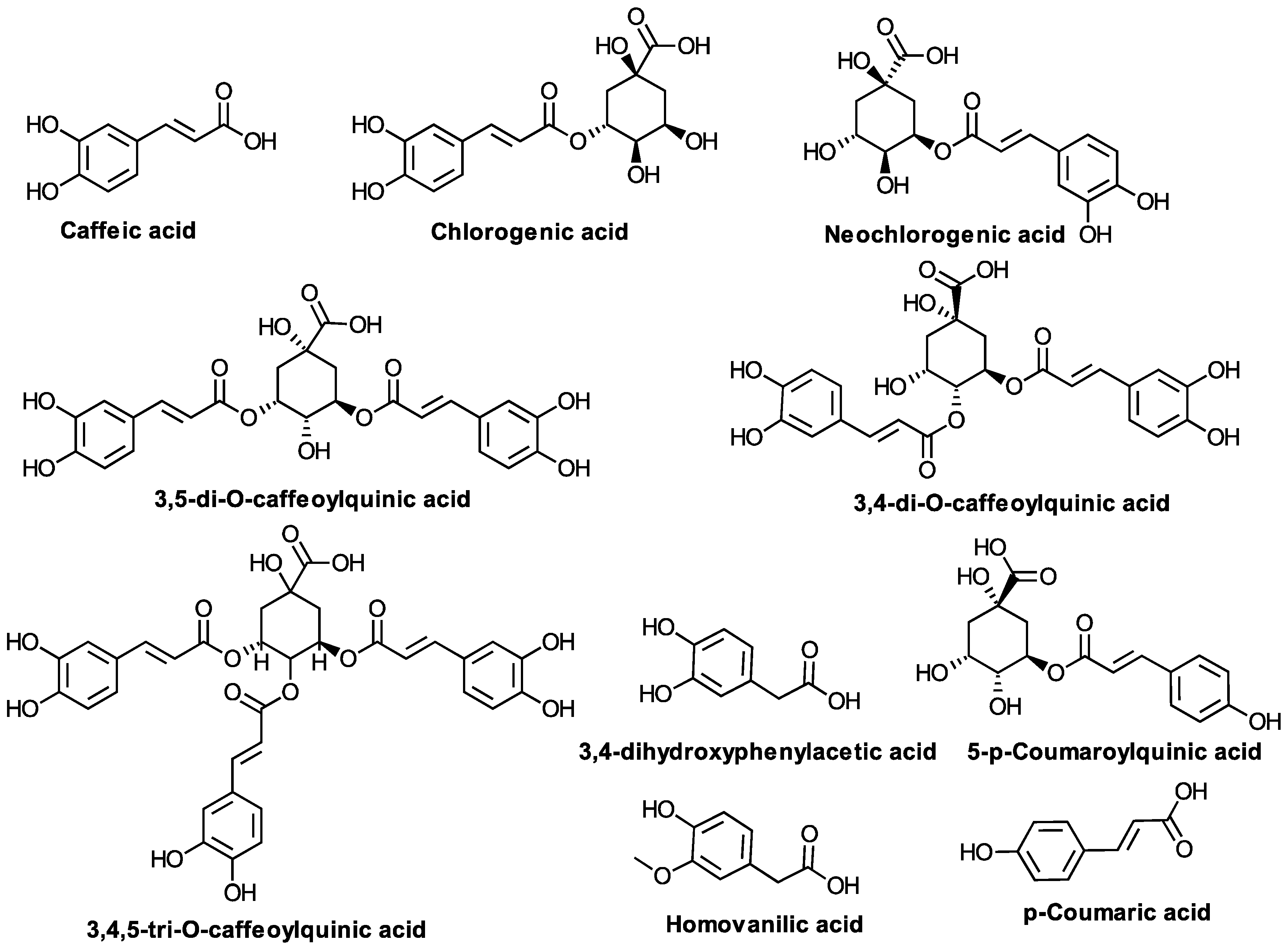
| Caffeic acid, Chlorogenic acid 5-O-caffeoylquinic (neochlorogenic) acid 3,5-di-O-caffeoylquinic acid 3,4-di-O-caffeoylquinic acid 4,5-di-O-caffeoylquinic acid 3,4,5-tri-O-caffeoylquinic acid Methyl 3,5-di-O-caffeoylquinate 3-hydroxyphenylacetic acid 3,4-dihydroxyphenylacetic acid 5-p-Coumaroylquinic acid Homovanilic acid p-Coumaric acid Ferulic acid Sinapic acid Rosmarinic acid | Poland Egypt Korea Iran | Kalemba D. [85], Abdel Motaal A. et al. [10], Choi SZ. et al. [49], Thiem B. et al. [58], Bajkacz S. et al. [86], Haghi G., Hatami A. [87], Roslon W. et al. [59], Kraujalienė V et al. [84], M. Marksa et al. [88], D. Fraisse et al. [89], Borkowski B. and Skrzypczakowa L. [60], Jaiswal R. et al. [90] |
| Coumarins | ||
| 7-hydroxy-coumarin (umbelliferone) | Czech Republic | Dobias P. et al. [91] |
| Terpene Derivatives (Figure 6) | ||
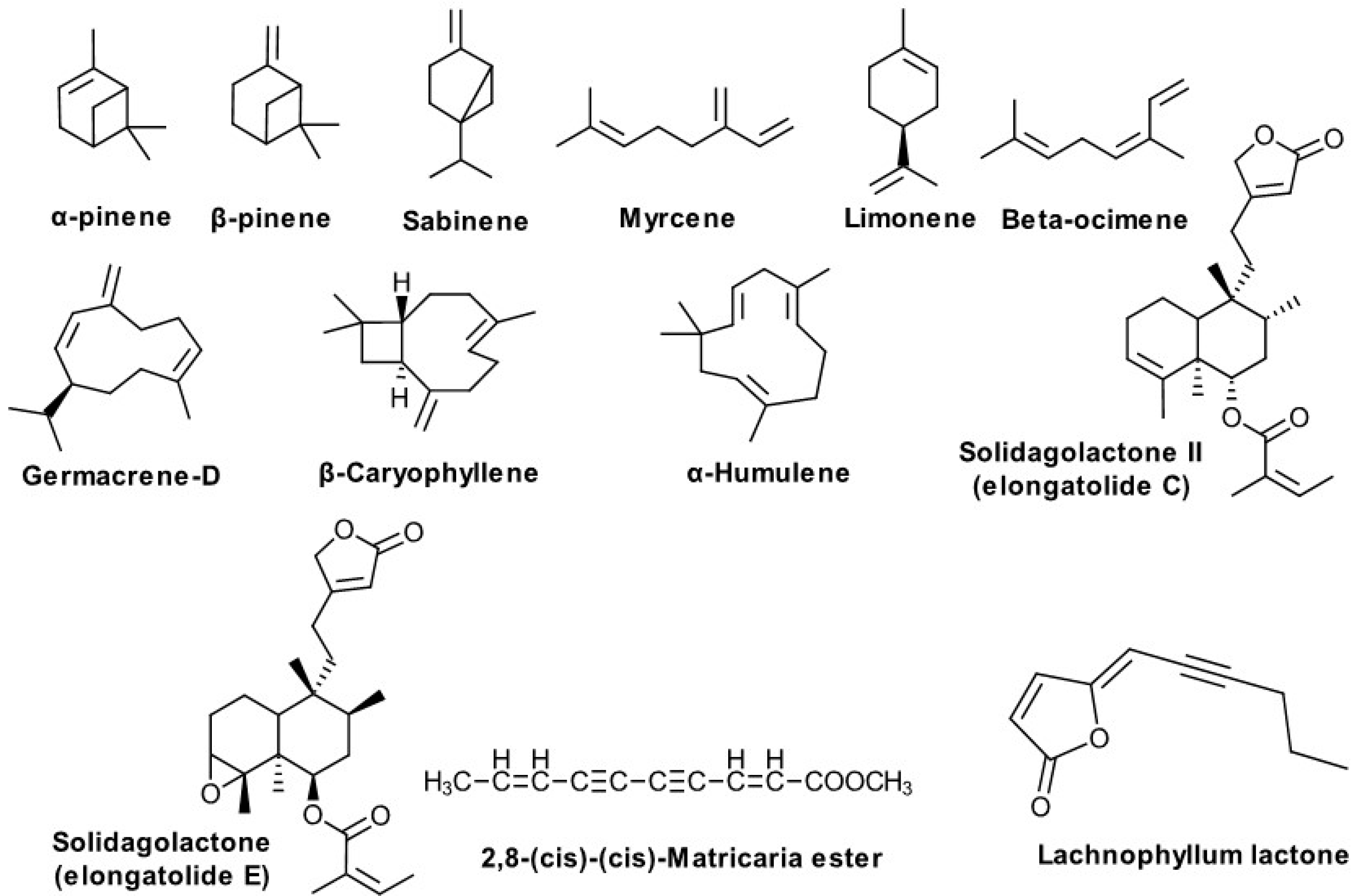
| α-Pinene, β-Pinene, Sabinene Myrcene Limonene β-Ocimene Germacrene-D, β-Caryophyllene, α-Humulene, Clerodane diterpenes 2,8-(cis)-(cis)-Matricaria ester Matricaria γ-lactones Lachnophyllum lactone ent-germacra-4(15),5,10(14)-trien-1β-ol β-dictyopterol | Poland, Japan, Italy, Russian Federation, USA Denmark Korea | Kalemba D. [74], Kalemba D. and Thiem B. [75], Fujita S. [76], Bertoli A. et al. [77], Tkachev AV. et al. [35], Goswami A et al. [92], Starks CM. et al. [78], Lam J. [80], Choi S. [49] |
| Saponins (Figure 7) | ||
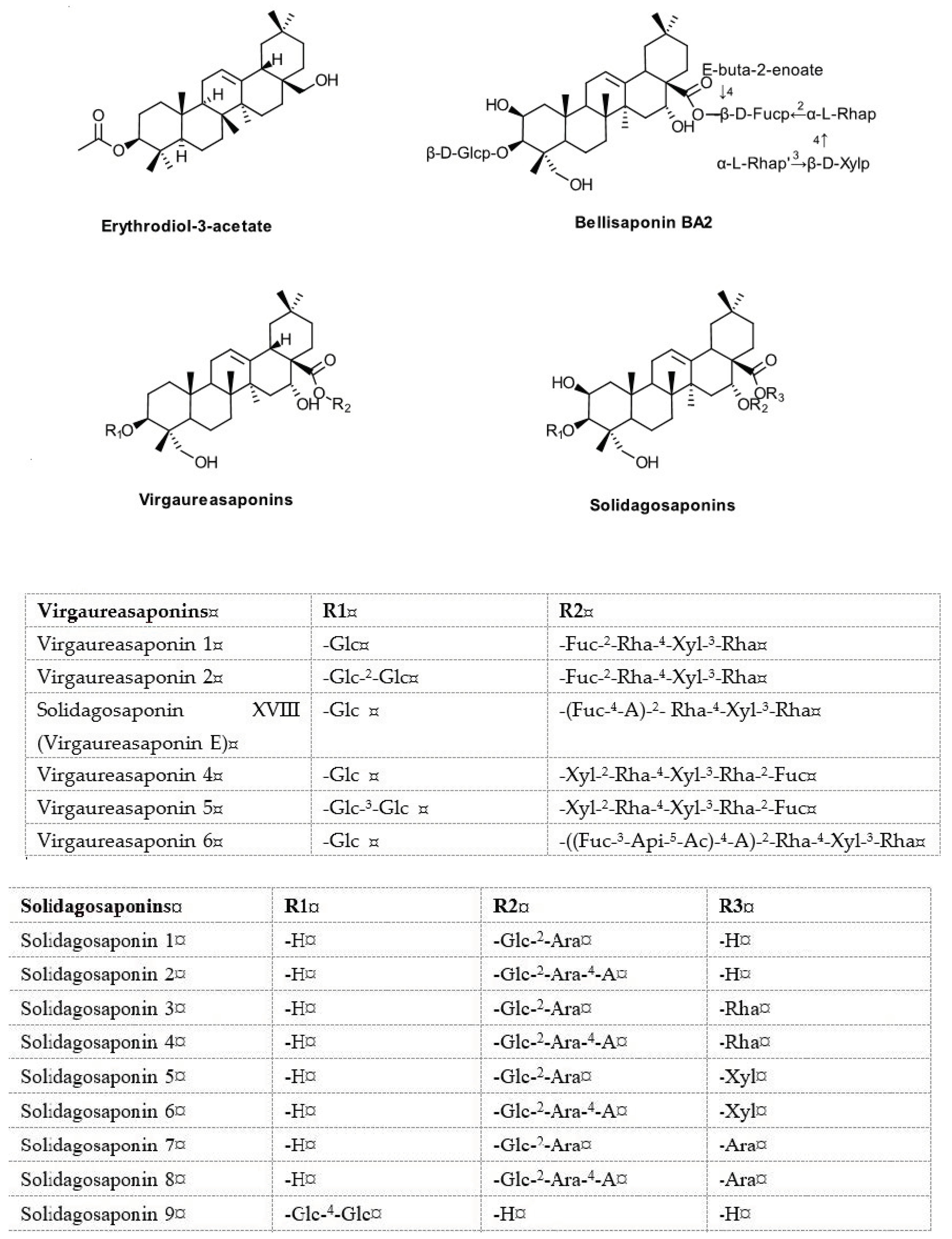
| Virgaureasaponins 1–6 Solidagosaponins X-XXIX Bellisaponin BA2 Erythrodiol-3-acetate | Germany, France, Romania Japan | Bader G. et al. [34,56,57,58,59], Chevalier M. et al. [86], Laurençon L. et al. [61], Dobjanschi L. et al. [85], Inose Y. et al. [60], Sung JH et al. [50] |
| Carbohydrates and Other Compounds | ||
| Polysaccharides α-tocopherol quinone 2-phyten-1-ol | Russian Federation Korea | Pychenkova PA. [66], Sung JH et al. [50] |
5.1. Flavonoids
5.2. C6-C1 Compounds
5.3. C6-C2 and C6-C3 Compounds
5.4. Coumarins
5.5. Terpene Derivatives
5.6. Saponins
5.7. Polysaccharides
6. Pharmacology
6.1. Antioxidant Properties
6.2. Anti-Inflammatory Effects
6.3. Analgesic Activity
6.4. Spasmolytic and Antihypertensive Activity
6.5. Diuretic Effects and Benefits in Other Urinary Tract Conditions
6.6. Antibacterial Activity
6.7. Antifungal Activity
6.8. Antiparasite Activity
6.9. Cytotoxic and Antitumor Activity
6.10. Antimutagenic Activity
6.11. Antiadipogenic and Antidiabetic Activities
6.12. Cardioprotective Effects
6.13. Antisenescence Effects
7. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- World Flora Online. Consortium WFO (2020): World Flora Online. Available online: http://www.worldfloraonline.org (accessed on 3 August 2020).
- Beck, J.B.; Semple, J.C.; Brull, J.M.; Lance, S.L.; Phillips, M.M.; Hoot, S.B.; Meyer, G.A. Genus-Wide Microsatellite Primers for the Goldenrods (Solidago; Asteraceae). Appl. Plant Sci. 2014, 2, 1300093. [Google Scholar] [CrossRef] [PubMed]
- Toiu, A.; Vlase, L.; Vodnar, D.C.; Gheldiu, A.-M.; Oniga, I. Solidago graminifolia L. Salisb. (Asteraceae) as a Valuable Source of Bioactive Polyphenols: HPLC Profile, In Vitro Antioxidant and Antimicrobial Potential. Molecules 2019, 24, 2666. [Google Scholar] [CrossRef] [PubMed]
- Woźniak, D.; Ślusarczyk, S.; Domaradzki, K.; Dryś, A.; Matkowski, A. Comparison of Polyphenol Profile and Antimutagenic and Antioxidant Activities in Two Species Used as Source of Solidaginis herba—Goldenrod. Chem. Biodivers. 2018, 15, e1800023. [Google Scholar] [CrossRef] [PubMed]
- Goulart, S.; Moritz, M.I.G.; Lang, K.L.; Liz, R.; Schenkel, E.P.; Fröde, T.S. Anti-inflammatory evaluation of Solidago chilensis Meyen in a murine model of pleurisy. J. Ethnopharmacol. 2007, 113, 346–353. [Google Scholar] [CrossRef] [PubMed]
- Tutin, T.G.; Heywood, V.H.; Burges, N.A.; Moore, D.M.; Valentine, D.H.; Walters, S.M.; Webb, D.A. Flora Europaea. Volume 4. Plantaginaceae to Compositae (and Rubiaceae); Zenodo: Geneva, Switzerland, 1976. [Google Scholar]
- Native Plant Trust. Go Botany. Available online: https://gobotany.nativeplanttrust.org/species/euthamia/graminifolia/ (accessed on 1 September 2020).
- Móricz, Á.M.; Jamshidi-Aidji, M.; Krüzselyi, D.; Darcsi, A.; Böszörményi, A.; Csontos, P.; Béni, S.; Ott, P.G.; Morlock, G.E. Distinction and valorization of 30 root extracts of five goldenrod (Solidago) species. J. Chromatogr. A 2020, 1611, 460602. [Google Scholar] [CrossRef]
- European Medicines Agency. Assessment Report on Solidago virgaurea L., Herba; European Medicines Agency: London, UK, 2008. [Google Scholar]
- Abdel Motaal, A.; Ezzat, S.M.; Tadros, M.G.; El-Askary, H.I. In vivo anti-inflammatory activity of caffeoylquinic acid derivatives from Solidago virgaurea in rats. Pharm. Biol. 2016, 54, 2864–2870. [Google Scholar] [CrossRef]
- Yarnell, E. Botanical medicines for the urinary tract. World J. Urol. 2002, 20, 285–293. [Google Scholar] [CrossRef]
- Cai, T.; Caola, I.; Tessarolo, F.; Piccoli, F.; D’Elia, C.; Caciagli, P.; Nollo, G.; Malossini, G.; Nesi, G.; Mazzoli, S.; et al. Solidago, orthosiphon, birch and cranberry extracts can decrease microbial colonization and biofilm development in indwelling urinary catheter: A microbiologic and ultrastructural pilot study. World J. Urol. 2014, 32, 1007–1014. [Google Scholar] [CrossRef]
- Calalb, T.; Fursenco, C.; Ionita, O.; Ghendov, V. The morpho-anatomical study of Solidago virgaurea L. species from the flora of Republic of Moldova. East Eur. Sci. J. 2018, 2-1, 4–13. [Google Scholar]
- Szymura, M.; Wolski, K. Leaf Epidermis Traits as Tools to Identify Solidago L. Taxa in Poland. Acta Biol. Crac. Ser. Bot. 2011, 53. [Google Scholar] [CrossRef]
- Dobjanschi, L.; Vostinaru, O.; Tamas, M. Criteria for Solidago Species Differentiation. Acta Univ. Cibiniensis Ser. F Chem. 2005, 8, 41–44. [Google Scholar]
- Buynov, M.; Goryachkina, Y.; Fedoseyeva, G. Otlichitel’nyye Morfologicheskiye i Anatomo-Diagnosticheskiye Priznaki Vidov Zolotarnikov—Kanadskogo, Obyknovennogo, Daurskogo: Uchebnoye Posobiye; IGMU: Irkutsk, Russia, 2013. [Google Scholar]
- Souza, D.M.F.D.; Sá, R.D.; Araújo, E.L.; Randau, K.P. Anatomical, phytochemical and histochemical study of Solidago chilensis Meyen. An. Acad. Bras. Ciênc. 2018, 90, 2107–2120. [Google Scholar] [CrossRef]
- Fedotova, V.; Chelombit’sh, V. Anatomo-diagnosticheskoye izucheniye zolotarnika kavkazskogo (Solidago caucasica Kem.-Nath.) flory Severnogo Kavkaza. In Razrabotka, Issledovaniye i Marketing Novoy Farmatsevticheskoy Produktsii: Sb. nauch. tr.; Vyp. 67; The Publisher of Medicine and Pharmacy Institute of Pyatigorsk: Pyatigorsk, Russia, 2012. [Google Scholar]
- Bley, L.F. Ueber Verwechselung der herba Solidaginis virgaureae mit herba Senecion. nemorensis. Arch. Pharm. 1850, 113, 34–37. [Google Scholar] [CrossRef][Green Version]
- Semple, J. An intuitive phylogeny and summary of chromosome number variation in the goldenrod genus Solidago (Asteraceae: Astereae). Phytoneuron 2016, 32, 1–9. [Google Scholar]
- Sakaguchi, S.; Kimura, T.; Kyan, R.; Maki, M.; Nishino, T.; Ishikawa, N.; Nagano, A.J.; Honjo, M.N.; Yasugi, M.; Kudoh, H.; et al. Phylogeographic analysis of the East Asian goldenrod (Solidago virgaurea complex, Asteraceae) reveals hidden ecological diversification with recurrent formation of ecotypes. Ann. Bot. 2018, 121, 489–500. [Google Scholar] [CrossRef]
- Kiełtyk, P.; Mirek, Z. Taxonomy of the Solidago virgaurea Group (Asteraceae) in Poland, with Special Reference to Variability along an Altitudinal Gradient. Folia Geobot. 2014, 49, 259–282. [Google Scholar] [CrossRef][Green Version]
- Yuzepchuk, S. Zolotarnik–Solidago L. In Flora SSSR 25; Shishkin, B., Ed.; Izdatel’stvo Akademii Nauk SSSR: Moscow, Russia; Saint Petersburg, Russia, 1959; pp. 31–50. [Google Scholar]
- Slavík, B. Solidago L.–zlatobýl. In Květena České Republiky 7 (Flora of the Czech Republic 7); Slavík, B., Štěpánková, J., Eds.; Academia: Prague, Czech Republic, 2004; pp. 114–123. [Google Scholar]
- Botanical Society of Britain and Ireland; Biological Records Center; UK Center for Ecology and Hydrology; Joint Nature Conservation Committee. Online Atlas of the British and Irish Flora—Solidago virgaurea. Available online: https://www.brc.ac.uk/plantatlas/plant/solidago-virgaurea (accessed on 28 August 2020).
- Sakaguchi, S.; Horie, K.; Ishikawa, N.; Nagano, A.J.; Yasugi, M.; Kudoh, H.; Ito, M. Simultaneous evaluation of the effects of geographic, environmental and temporal isolation in ecotypic populations of Solidago virgaurea. New Phytol. 2017, 216, 1268–1280. [Google Scholar] [CrossRef]
- Laphitz, R.M.L.; Semple, J.C. A Multivariate Morphometric Analysis of the Solidago chilensis Group in South America and Related Taxa in North America (Asteraceae, Astereae) 1. Ann. Mo. Bot. Gard. 2015, 100, 423–441. [Google Scholar] [CrossRef]
- American Botanical Council. Solidago virgaurea L. Available online: http://cms.herbalgram.org/MedPlantID/BotanicalEntries/Solidago_virgaurea.html (accessed on 28 August 2020).
- European Medicines Agency. Community Herbal Monograph on Solidago virgaurea L. Herba, EMEA/HMPC/285758/2007 2008. Available online: https://www.ema.europa.eu/en/documents/herbal-monograph/final-community-herbal-monograph-solidago-virgaurea-l-herba_en.pdf (accessed on 28 August 2020).
- Blum, H. Goldenrod has increasing importance as a herbal drug. Gemuse 1999, 35, 431–432. [Google Scholar]
- Borchert, V.E.; Czyborra, P.; Fetscher, C.; Goepel, M.; Michel, M.C. Extracts from Rhois aromatica and Solidaginis virgaurea inhibit rat and human bladder contraction. Naunyn-Schmiedeberg’s Arch. Pharmacol. 2004, 369, 281–286. [Google Scholar] [CrossRef]
- Móricz, Á.M.; Ott, P.G.; Häbe, T.T.; Darcsi, A.; Böszörményi, A.; Alberti, Á.; Krüzselyi, D.; Csontos, P.; Béni, S.; Morlock, G.E. Effect-Directed Discovery of Bioactive Compounds Followed by Highly Targeted Characterization, Isolation and Identification, Exemplarily Shown for Solidago virgaurea. Anal. Chem. 2016, 88, 8202–8209. [Google Scholar] [CrossRef]
- Melzig, M.F. Goldenrod—A classical exponent in the urological phytotherapy. Wien. Med. Wochenschr. 2004, 154, 523–527. [Google Scholar] [CrossRef]
- Gross, S.C.; Goodarzi, G.; Watabe, M.; Bandyopadhyay, S.; Pai, S.K.; Watabe, K. Antineoplastic activity of Solidago virgaurea on prostatic tumor cells in an SCID mouse model. Nutr. Cancer 2002, 43, 76–81. [Google Scholar] [CrossRef]
- Tkachev, A.V.; Korolyuk, E.A.; Letchamo, W. Volatile Oil-Bearing Flora of Siberia VIII: Essential Oil Composition and Antimicrobial Activity of Wild Solidago virgaurea L. from the Russian Altai. J. Essent. Oil Res. 2006, 18, 46–50. [Google Scholar] [CrossRef]
- Heger, M. Entzündliche Erkrankungen der Urogenitalorgane. In Naturheilverfahren in der Urologie; Springer: Berlin/Heidelberg, Germany, 2003; pp. 55–102. ISBN 978-3-662-08920-0. [Google Scholar]
- Bader, G.; Wray, V.; Hiller, K. The Main Saponins from the Aerial Parts and the Roots of Solidago virgaurea subsp. virgaurea *. Planta Med. 1995, 61, 158–161. [Google Scholar] [CrossRef]
- A. Vogel. Plant Encylopedia Solidago virgaurea L. Available online: https://www.avogel.com/plant-encyclopaedia/solidago_virgaurea.php (accessed on 28 August 2020).
- Tománková, V.; Vlčková, A.; Anzenbacher, P.; Bachleda, P.; Anzenbacherová, E. Užívání přípravků ze zlatobýlu obecného (Solidago virgaurea) neovlivňuje metabolizmus současně podávaných léčiv. Klin Farm. Farm 2016, 30, 11–15. [Google Scholar]
- Nawłoć pospolita (Solidago virgaurea). Available online: https://www.plantago-sklep.pl/blog/nawloc-pospolita/ (accessed on 28 August 2020).
- Malankina, Y. Zolotarnik: Lekarstvennyye svoystva i primeneniye. Available online: https://www.greeninfo.ru/grassy/solidago_canadensis.html/Article/_/aID/5959 (accessed on 28 August 2020).
- Suleymanova, F.S.; Nesterova, O.V. The historical background and prospects of Canadian goldenrod (Solidago canadensis L.) herb medicinal use. J. Sci. Artic. Health Educ. Millenn. 2017, 19, 142–149. [Google Scholar] [CrossRef]
- Kolosova, V. Name–Text–Ritual: The Role of Plant Characteristics in Slavic Folk Medicine. Folklorika 2005, X, 44–61. [Google Scholar] [CrossRef]
- Kiselova, Y.; Ivanova, D.; Chervenkov, T.; Gerova, D.; Galunska, B.; Yankova, T. Correlation between theIn Vitro antioxidant activity and polyphenol content of aqueous extracts from bulgarian herbs. Phytother. Res. 2006, 20, 961–965. [Google Scholar] [CrossRef]
- Dobjanschi, L.; Paltinean, R.; Vlase, L.; Babota, M.; Fritea, L.; Tamas, M. Comparative phytochemical research of Solidago genus: S. graminifolia. Note I. Flavonoids. Acta Biol. Marisiensis 2018, 1, 18–26. [Google Scholar]
- Calalb, T.; Bodrug, M. Botanica Faramaceutică; CEP Medicina: Chisinau, Moldova, 2009. [Google Scholar]
- Tamas, M.; Toader, S. Acţiunea diuretică a unor specii de Solidago. Clujul Med. 1989, 62, 75–79. [Google Scholar]
- Petran, M.; Dragos, D.; Gilca, M. Historical ethnobotanical review of medicinal plants used to treat children diseases in Romania (1860s–1970s). J. Ethnobiol. Ethnomed. 2020, 16, 15. [Google Scholar] [CrossRef]
- Choi, S.Z.; Choi, S.U.; Lee, K.R. Pytochemical constituents of the aerial parts from Solidago virga-aurea var. gigantea. Arch. Pharmacal. Res. 2004, 27, 164–168. [Google Scholar] [CrossRef]
- Sung, J.H.; Lee, J.O.; Son, J.K.; Park, N.S.; Kim, M.R.; Kim, J.G.; Moon, D.C. Cytotoxic constituents from Solidago virga-aurea var. gigantea MIQ. Arch. Pharmacal. Res. 1999, 22, 633–637. [Google Scholar] [CrossRef]
- Inose, Y.; Miyase, T.; Ueno, A. Studies on the Constituents of Solidago virga-aurea L. I. Structural Elucidation of Saponins in the Herb. Chem. Pharm. Bull. 1991, 39, 2037–2042. [Google Scholar] [CrossRef]
- Dmuchowski, W.; Gozdowski, D.; Brągoszewska, P.; Baczewska, A.H.; Suwara, I. Phytoremediation of zinc contaminated soils using silver birch (Betula pendula Roth). Ecol. Eng. 2014, 71, 32–35. [Google Scholar] [CrossRef]
- Bielecka, A.; Królak, E. The accumulation of Mn and Cu in the morphological parts of Solidago canadensis under different soil conditions. PeerJ 2019, 7, e8175. [Google Scholar] [CrossRef]
- Bielecka, A.; Królak, E. Solidago canadensis as a bioaccumulator and phytoremediator of Pb and Zn. Environ. Sci. Pollut Res. Int. 2019, 26, 36942–36951. [Google Scholar] [CrossRef]
- Oldroyd, B.P. What’s Killing American Honey Bees? PLoS Biol. 2007, 5, e168. [Google Scholar] [CrossRef]
- Metzner, J.; Hirschelmann, R.; Hiller, K. Antiphlogistic and analgesic effects of leiocarposide, a phenolic bisglucoside of Solidago virgaurea L. Pharmazie 1984, 39, 869–870. [Google Scholar]
- Chodera, A.; Dabrowska, K.; Bobkiewicz-Kozłowska, T.; Tkaczyk, J.; Skrzypczak, L.; Budzianowski, J. Effect of leiocarposide on experimental urinary calculi in rats. Acta Pol. Pharm. 1988, 45, 181–186. [Google Scholar] [PubMed]
- Thiem, B.; Wesołowska, M.; Skrzypczak, L.; Budzianowski, J. Phenolic compounds in two Solidago L. species from in vitro culture. Acta Pol. Pharm. 2001, 58, 277–281. [Google Scholar] [PubMed]
- Roslon, W.; Osinska, E.; Mazur, K.; Geszprych, A. Chemical characteristics of European goldenrod (Solidago virgaurea L. subsp. virgaurea) from natural sites in central and Eastern Poland. Acta Sci. Pol. Hortorum Cultus 2014, 13, 55–65. [Google Scholar]
- Borkowski, B.; Skrzypczakowa, L. Polyphenolic Compounds in Herbs of the Species Solidago L. Acta Pol. Pharm. 1962, 19, 491–495. [Google Scholar]
- Budzianowski, J.; Skrzypczak, L.; Wesolowska, M. Flavonoid patterns in the genus Solidago. Pharm. Weekbl. Method. 1987, 9, 230. [Google Scholar]
- Pietta, P.; Gardana, C.; Mauri, P.; Zecca, L. High-performance liquid chromatographic analysis of flavonol glycosides of Solidago virgaurea. J. Chromatogr. A 1991, 558, 296–301. [Google Scholar] [CrossRef]
- Tamas, M. Cercetări chemotaxonomice la genul Solidago. Contrib. Bot. (Cluj-Napoca) 1986, 110–113. [Google Scholar]
- Dobjanschi, L.; Fritea, L.; Patay, E.B.; Tamas, M. Comparative study of the morphological and phytochemical characterization of Romanian Solidago species. Pak. J. Pharm. Sci. 2019, 32, 1571–1579. [Google Scholar]
- Chodera, A.; Dabrowska, K.; Sloderbach, A.; Skrzypczak, L.; Budzianowski, J. Effect of flavonoid fractions of Solidago virgaurea L. on diuresis and levels of electrolytes. Acta Pol. Pharm. 1991, 48, 35–37. [Google Scholar]
- Bader, G.; Binder, K.; Hiller, K.; Ziegler-Böhme, H. The antifungal action of triterpene saponins of Solidago virgaurea L. Die Pharm. 1987, 42, 140. [Google Scholar]
- Bader, G.; Kulhanek, Y.; Ziegler-Böhme, H. Zur antimyzetischen Wirksamkeit von polygalasäureglycosiden. Pharmazie 1990, 45, 618–620. [Google Scholar] [PubMed]
- Bader, G.; Plohmann, B.; Franz, G. Saponin from Solidago virgaurea L. Possible Agents for Therapy of Cancer? In Proceedings of the 44th Annual Congress of the Society for Medicinal Plant Research and a Joint Meeting with the Czech Biotechnology Society, Prague, Czech Republic, 3–7 September 1996; Abstracts of Lectures and Poster Presentations. pp. 3–7. [Google Scholar]
- Bader, G.; Wray, V.; Just, U.; Hiller, K. Enzymatic hydrolysis of the cytotoxic triterpenoid glycoside virgaureasaponin 1. Phytochemistry 1998, 49, 153–156. [Google Scholar] [CrossRef]
- Inose, Y.; Miyase, T.; Ueno, A. Studies on the Constituents of Solidago virga-aurea L. II. Structures of Solidagosaponins X-XX. Chem. Pharm. Bull. 1992, 40, 946–953. [Google Scholar] [CrossRef][Green Version]
- Dobjanschi, L.; Zdrinca, M.; Muresan, M.; Vicas, S.; Antonescu, A. The thin layer chromatography analysis of saponins belonging to Solidago species. Fasc. Prot. Mediu. 2013, 21, 56–60. [Google Scholar]
- Chevalier, M.; Medioni, E.; Prêcheur, I. Inhibition of Candida albicans yeast–hyphal transition and biofilm formation by Solidago virgaurea water extracts. J. Med. Microbiol. 2012, 61, 1016–1022. [Google Scholar] [CrossRef]
- Laurençon, L.; Sarrazin, E.; Chevalier, M.; Prêcheur, I.; Herbette, G.; Fernandez, X. Triterpenoid saponins from the aerial parts of Solidago virgaurea alpestris with inhibiting activity of Candida albicans yeast-hyphal conversion. Phytochemistry 2013, 86, 103–111. [Google Scholar] [CrossRef]
- Kalemba, D. Constituents of the essential oil of Solidago virgaurea L. Flavour Fragr. J. 1998, 13, 373–376. [Google Scholar] [CrossRef]
- Kalemba, D.; Thiem, B. Constituents of the essential oils of four micropropagated Solidago species. Flavour Fragr. J. 2004, 19, 40–43. [Google Scholar] [CrossRef]
- Fujita, Y.; Fujita, S.; Hayama, Y. Miscellaneous contributions to the essential oils of plants from various territories. Components of the essential oils of Solidago virgaurea Linn. ssp. Nippon Nogei Kagaku Kaishi 1990, 64, 1729–1732. (In Japanese) [Google Scholar] [CrossRef][Green Version]
- Bertoli, A.; Cioni, P.L.; Flamini, G.; Morelli, I.; Spinelli, G.; Tomei, P.E. Constituents of the Essential Oil of Solidago litoralis, an Endemic Plant from Northern Tuscany (Italy). J. Essent. Oil Res. 1999, 11, 215–216. [Google Scholar] [CrossRef]
- Starks, C.M.; Williams, R.B.; Goering, M.G.; O’Neil-Johnson, M.; Norman, V.L.; Hu, J.-F.; Garo, E.; Hough, G.W.; Rice, S.M.; Eldridge, G.R. Antibacterial clerodane diterpenes from Goldenrod (Solidago virgaurea). Phytochemistry 2010, 71, 104–109. [Google Scholar] [CrossRef]
- Pychenkova, P.A. Dynamics of the amount and characteristics of the polysaccharides of Solidago virgaurea. Chem. Nat. Compd. 1987, 23, 246–247. [Google Scholar] [CrossRef]
- Lam, J. Polyacetylenes of Solidago virgaurea: Their seasonal variation and NMR long-range spin coupling constants. Phytochemistry 1971, 10, 647–653. [Google Scholar] [CrossRef]
- Council of Europe. European Pharmacopoeia (Ph. Eur.), 10th ed.; Council of Europe: Strasbourg, France, 2019. [Google Scholar]
- Fuchs, L. Isolierung Von Quercitrin Aus Solidago Virga Aurea L., S. Serotina Ait und S. Canadensis L. Sci. Pharm. 1949, 17, 128–131. [Google Scholar] [PubMed]
- Apáti, P.; Szentmihályi, K.; Kristó, S.T.; Papp, I.; Vinkler, P.; Szoke, É.; Kéry, Á. Herbal remedies of Solidago—correlation of phytochemical characteristics and antioxidative properties. J. Pharm. Biomed. Anal. 2003, 32, 1045–1053. [Google Scholar] [CrossRef]
- Kraujalienė, V.; Pukalskas, A.; Venskutonis, P.R. Biorefining of goldenrod (Solidago virgaurea L.) leaves by supercritical fluid and pressurized liquid extraction and evaluation of antioxidant properties and main phytochemicals in the fractions and plant material. J. Funct. Foods 2017, 37, 200–208. [Google Scholar] [CrossRef]
- Kalemba, D. Phenolic acids in four Solidago species. Pharmazie 1992, 47, 471–472. [Google Scholar]
- Bajkacz, S.; Baranowska, I.; Buszewski, B.; Kowalski, B.; Ligor, M. Determination of Flavonoids and Phenolic Acids in Plant Materials Using SLE-SPE-UHPLC-MS/MS Method. Food Anal. Methods 2018, 11, 3563–3575. [Google Scholar] [CrossRef]
- Haghi, G.; Hatami, A. Simultaneous Quantification of Flavonoids and Phenolic Acids in Plant Materials by a Newly Developed Isocratic High-Performance Liquid Chromatography Approach. J. Agric. Food Chem. 2010, 58, 10812–10816. [Google Scholar] [CrossRef]
- Marksa, M.; Zymone, K.; Ivanauskas, L.; Radušienė, J.; Pukalskas, A.; Raudone, L. Antioxidant profiles of leaves and inflorescences of native, invasive and hybrid Solidago species. Ind. Crops Prod. 2020, 145, 112123. [Google Scholar] [CrossRef]
- Fraisse, D.; Felgines, C.; Texier, O.; Lamaison, J.-L. Caffeoyl Derivatives: Major Antioxidant Compounds of Some Wild Herbs of the Asteraceae Family. Food Nutr. Sci. 2011, 02, 181–192. [Google Scholar] [CrossRef]
- Jaiswal, R.; Kiprotich, J.; Kuhnert, N. Determination of the hydroxycinnamate profile of 12 members of the Asteraceae family. Phytochemistry 2011, 72, 781–790. [Google Scholar] [CrossRef] [PubMed]
- Dobiáš, P.; Pavlíková, P.; Adam, M.; Eisner, A.; Beňová, B.; Ventura, K. Comparison of pressurised fluid and ultrasonic extraction methods for analysis of plant antioxidants and their antioxidant capacity. Open Chem. 2010, 8, 87–95. [Google Scholar] [CrossRef]
- Goswami, A.; Barua, R.N.; Sharma, R.P.; Baruah, J.N.; Kulanthaivel, P.; Herz, W. Clerodanes from Solidago virgaurea. Phytochemistry 1984, 23, 837–841. [Google Scholar] [CrossRef]
- Melzig, M.; Löser, B.; Bader, G.; Papsdorf, G. European goldenrod as an anti-inflammatory drug: Investigations into the cyto-and molecular pharmacology for a better understanding of the anti-inflammatory activity of preparations from Solidago virgaurea. Z. Phytother. 2000, 21, 67–70. [Google Scholar]
- Vuolo, M.M.; Lima, V.S.; Junior, M.R.M. Phenolic compounds: Structure, classification, and antioxidant power. In Bioactive Compounds; Elsevier: Amsterdam, The Netherlands, 2019; pp. 33–50. [Google Scholar]
- Demir, H.; Acik, L.; Bali, E.B.; Koç, L.Y.; Kaynak, G. Antioxidant and antimicrobial activities of Solidago virgaurea extracts. Afr. J. Biotechnol. 2009, 8, 2. [Google Scholar]
- Meyer, B.; Schneider, W.; Elstner, E. Antioxidative properties of alcoholic extracts from Fraxinus excelsior, Populus tremula and Solidago virgaurea. Arzneimittel-forschung 1995, 45, 174. [Google Scholar] [PubMed]
- Duarte, J.; Utrilla, P.; Jimenez, J.; Tamargo, J.; Zarzuelo, A.; Pérez Vizcaíno, F. Vasodilatory effects of flavonoids in rat aortic smooth muscle. Structure-activity relationships. Gen. Pharmacol. 1993, 24, 857–862. [Google Scholar] [CrossRef]
- Kruk, J.; Baranowska, I.; Buszewski, B.; Bajkacz, S.; Kowalski, B.; Ligor, M. Flavonoids enantiomer distribution in different parts of goldenrod (Solidago virgaurea L.), lucerne (Medicago sativa L.) and phacelia (Phacelia tanacetifolia Benth.). Chirality 2019, 31, 138–149. [Google Scholar] [CrossRef]
- Bjorkman, O.; Holmgren, P. Preliminary Observations on Anthocyanins and Other Flavonoid Compounds and Respiration Rates in Different Ecotypes of Solidago virgaurea. Physiol. Plant. 1958, 11, 154–157. [Google Scholar] [CrossRef]
- Jang, Y.; Wang, Z.; Lee, J.-M.; Lee, J.-Y.; Lim, S. Screening of Korean Natural Products for Anti-Adipogenesis Properties and Isolation of Kaempferol-3-O-rutinoside as a Potent Anti-Adipogenetic Compound from Solidago virgaurea. Molecules 2016, 21, 226. [Google Scholar] [CrossRef] [PubMed]
- Hiller, K.; Dube, G.; Zeigan, D. Virgaureoside aa new bisdesmosidic phenol glycoside from Solidago virgaurea. Die Pharm. 1985, 40, 795–796. [Google Scholar]
- Paun, G.; Neagu, E.; Albu, C.; Radu, G.L. Verbascum phlomoides and Solidago virgaureae herbs as natural source for preventing neurodegenerative diseases. J. Herb. Med. 2016, 6, 180–186. [Google Scholar] [CrossRef]
- Skrzypczak, L.; Wesołowska, M.; Thiem, B.; Budzianowski, J. Solidago L. Species (Goldenrod): In Vitro Regeneration and Biologically Active Secondary Metabolites. In Medicinal and Aromatic Plants XI; Bajaj, Y.P.S., Ed.; Biotechnology in Agriculture and Forestry; Springer: Berlin/Heidelberg, Germany, 1999; Volume 43, pp. 384–403. ISBN 978-3-642-08363-1. [Google Scholar]
- Bader, G.; Janka, M.; Hannig, H.; Hiller, K. Zur quantitativen Bestimmung von Leiocaposid in Solidago virgaurea L. Die Pharm. 1990, 45, 380–381. [Google Scholar]
- Nkuimi Wandjou, J.G.; Quassinti, L.; Gudžinskas, Z.; Nagy, D.U.; Cianfaglione, K.; Bramucci, M.; Maggi, F. Chemical composition and antiproliferative effect of essential oils of four Solidago species (S. canadensis, S. gigantea, S. virgaurea and S.× niederederi). Chem. Biodivers. 2020. [Google Scholar] [CrossRef]
- Hiller, K.; Genzel, S.; Murach, M.; Franke, P. Zur Kenntnis der Saponine der Gattung Solidago. 1 Mitteilung: Uber die Saponine von Solidago virgaurea L. Pharmazie 1975, 30, 188–190. [Google Scholar] [PubMed]
- Bader, G.; Wray, V.; Hiller, K. Virgaureasaponin 3, a 3,28-bisdesmosidic triterpenoid saponin from Solidago virgaurea. Phytochemistry 1992, 31, 621–623. [Google Scholar] [CrossRef]
- Miyase, T.; Inose, Y.; Ueno, A. Studies on the Constituents of Solidago virga-aurea L. III. Structures of Solidagosaponins XXI-XXIX. Chem. Pharm. Bull. 1994, 42, 617–624. [Google Scholar] [CrossRef][Green Version]
- Schöpke, T.; Wray, V.; Rzazewska, B.; Hiller, K. Bellissaponins BA1 and BA2, acylated saponins from Bellis perennis. Phytochemistry 1991, 30, 627–631. [Google Scholar] [CrossRef]
- Saluk-Juszczak, J.; Pawlaczyk, I.; Olas, B.; Kołodziejczyk, J.; Ponczek, M.; Nowak, P.; Tsirigotis-Wołoszczak, M.; Wachowicz, B.; Gancarz, R. The effect of polyphenolic-polysaccharide conjugates from selected medicinal plants of Asteraceae family on the peroxynitrite-induced changes in blood platelet proteins. Int. J. Biol. Macromol. 2010, 47, 700–705. [Google Scholar] [CrossRef] [PubMed]
- Salehi, B.; Martorell, M.; Arbiser, J.L.; Sureda, A.; Martins, N.; Maurya, P.K.; Sharifi-Rad, M.; Kumar, P.; Sharifi-Rad, J. Antioxidants: Positive or Negative Actors? Biomolecules 2018, 8, 124. [Google Scholar] [CrossRef] [PubMed]
- Woo, J.H.; Shin, S.L.; Jeong, H.S.; Lee, C.H. Influence of Applied Pressure and Heat Treatment on Antioxidant Activities of Young Leaves from Achillea alpina and Solidago virgaurea subsp. gigantea. Korean J. Plant Res. 2010, 23, 123–130. [Google Scholar]
- Kim, Y.-J.; Kim, H.-Y.; Choe, J.-H.; Park, J.-H.; Ham, Y.-K.; Yeo, E.-J.; Hwang, K.-E.; Kim, C.-J. Antioxidant activity of goldenrod (Solidago virgaurea) leaf and stem powder on raw ground pork during chilled storage. Korean J. Food Sci. Anim. Resour. 2013, 33, 1–8. [Google Scholar] [CrossRef][Green Version]
- Choe, J.; Choi, Y.; Kim, H.; Han, D.; Kim, H.; Kim, Y.; Park, J.; Chung, H.; Kim, C. Effects of goldenrod (Solidago virgaurea) leaf and stem extracts on oxidative stability in cooked ground pork during chilled storage. In Proceedings of the International Congress of Meat Science and Technology, Ghent, Belgium, 7–12 August 2011; pp. 7–12. [Google Scholar]
- Jacker, H.; Voigt, G.; Hiller, K. Zum antiexsudativen verhalten einiger triterpensaponine. Pharmazie 1982, 37, 380–382. [Google Scholar]
- Choi, S.Z.; Choi, S.U.; Bae, S.Y.; Neung Pyo, S.; Lee, K.R. Immunobioloical activity of a new benzyl benzoate from the aerial parts of Solidago virga-aurea var. gigantea. Arch. Pharmacal. Res. 2005, 28, 49. [Google Scholar] [CrossRef]
- El-Ghazaly, M.; Khayyal, M.; Okpanyi, S.; Arens-Corell, M. Study of the anti-inflammatory activity of Populus tremula, Solidago virgaurea and Fraxinus excelsior. Arzneimittel-forschung 1992, 42, 333–336. [Google Scholar]
- Strehl, E.; Schneider, W.; Elstner, E.F. Inhibition of dihydrofolate reductase activity by alcoholic extracts from Fraxinus excelsior, Populus tremula and Solidago virgaurea. Arzneimittel-forschung 1995, 45, 172–173. [Google Scholar]
- ESCOP Solidago virgaurea. In Monographs on the Medicinal Uses of Plant Drugs; European Scientific Cooperative on Phytotherapy Thieme: Exeter, UK; Stuttgart, Germany; New York, NY, USA, 2003; pp. 487–491. ISBN 1-58890-233-1.
- Schweitzer, B.I.; Dicker, A.P.; Bertino, J.R. Dihydrofolate reductase as a therapeutic target. FASEB J. 1990, 4, 2441–2452. [Google Scholar] [CrossRef]
- Bonaterra, G.A.; Schwarzbach, H.; Kelber, O.; Weiser, D.; Kinscherf, R. Anti-inflammatory effects of Phytodolor® (STW 1) and components (poplar, ash and goldenrod) on human monocytes/macrophages. Phytomedicine 2019, 58, 152868. [Google Scholar] [CrossRef]
- Von Kruedener, S.; Schneider, W.; Elstner, E. A combination of Populus tremula, Solidago virgaurea and Fraxinus excelsior as an anti-inflammatory and antirheumatic drug. A short review. Arzneimittel-forschung 1995, 45, 169–171. [Google Scholar]
- Sampson, J.H.; Phillipson, J.D.; Bowery, N.G.; O’Neill, M.J.; Houston, J.G.; Lewis, J.A. Ethnomedicinally selected plants as sources of potential analgesic compounds: Indication ofin vitro biological activity in receptor binding assays. Phytother. Res. 2000, 14, 24–29. [Google Scholar] [CrossRef]
- Westendorf, J.; Vahlensieck, W. Spasmolytische und kontraktile Einflüsse eines pflanzlichen Kombinations-präparates auf die glatte Muskulatur des isolierten Meerschweinchendarms. Arzneimittel-forschung/Drug Res. 1981, 31, 40–43. [Google Scholar]
- Rácz, G.; Rácz-Kotilla, E.; Józsa, J. Hypotensive Activity-A Possible Pharmacotaxonomic Character of Solidago L. In Proceedings of the II International Symposium on Spices and Medicinal Plants, Budapest, Hungary, 16–22 July 1979; Volume 96, pp. 15–18. [Google Scholar]
- Raczkotilla, E.; Racz, G. Hypotensive and sedative effect of extracts obtained from Soldidago virgaurea L. [golden rod, drug plants]. Planta Med. 1978, 33, 300. [Google Scholar]
- Lasserre, B.; Kaiser, R.; Chanh, P.H.; Ifansyah, N.; Gleye, J.; Moulis, C. Effects on rats of aqueous extracts of plants used in folk medicine as antihypertensive agents. Naturwissenschaften 1983, 70, 95–96. [Google Scholar] [CrossRef] [PubMed]
- Mangiafico, S.; Costello-Boerrigter, L.C.; Andersen, I.A.; Cataliotti, A.; Burnett, J.C. Neutral endopeptidase inhibition and the natriuretic peptide system: An evolving strategy in cardiovascular therapeutics. Eur. Heart J. 2013, 34, 886–893. [Google Scholar] [CrossRef] [PubMed]
- Kaspers, U.; Poetsch, F.; Nahrstedt, A.; Chatterjee, S. Diuretic effects of extracts and fractions obtained from different Solidago species. Naunyn-Schmiedebergs Arch. Pharmacol. 1998, 358, R495. [Google Scholar]
- Chodera, A.; Dabrowska, K.; Seńczuk, M.; Wasik-Olejnik, A.; Skrzypczak, L.; Budzianowski, J.; Ellnain-Wojtaszek, M. Diuretic effect of the glycoside from a plant of the Solidago L. genus. Acta Pol. Pharm. 1985, 42, 199–204. [Google Scholar]
- Chodera, A.; Dąbrowska, K.; Skrzypczak, L. Biological activity of Leiocarposide from Solidago L. Acta Agron. Acad. Sci. Hung 1985, 34, 112. [Google Scholar]
- Budzianowski, J. Die urologische Wirkung des Leiocarposids. Drogenreport 1999, 12, 20–21. [Google Scholar]
- Edwards, S.E.; Rocha, I.; Williamson, E.M.; Heinrich, M. Phytopharmacy: An Evidence-Based Guide to Herbal Medical Products; Wiley Blackwell: Chichester, West Sussex, UK; Hoboken, NJ, USA, 2015; ISBN 978-1-118-54345-0. [Google Scholar]
- Kolodziej, B.; Kowalski, R.; Kędzia, B. Antibacterial and antimutagenic activity of extracts aboveground parts of three Solidago species: Solidago virgaurea L., Solidago canadensis L. and Solidago gigantea Ait. J. Med. Plants Res. 2011, 5, 6770–6779. [Google Scholar]
- Bussmann, R.W.; Malca-García, G.; Glenn, A.; Sharon, D.; Chait, G.; Díaz, D.; Pourmand, K.; Jonat, B.; Somogy, S.; Guardado, G.; et al. Minimum inhibitory concentrations of medicinal plants used in Northern Peru as antibacterial remedies. J. Ethnopharmacol. 2010, 132, 101–108. [Google Scholar] [CrossRef] [PubMed]
- Silva, A.C.O.; Santana, E.F.; Saraiva, A.M.; Coutinho, F.N.; Castro, R.H.A.; Pisciottano, M.N.C.; Amorim, E.L.C.; Albuquerque, U.P. Which approach is more effective in the selection of plants with antimicrobial activity? Evid.-Based Complementary Altern. Med. ECAM 2013, 2013, 308980. [Google Scholar] [CrossRef]
- Saraiva, M. In vitro evaluation of antioxidant, antimicrobial and toxicity properties of extracts of Schinopsis brasiliensis Engl. (Anacardiaceae). Afr. J. Pharm. Pharmacol. 2011, 5, 1724–1731. [Google Scholar] [CrossRef]
- Bueno, J. In Vitro Antimicrobial Activity of Natural Products Using Minimum Inhibitory Concentrations: Looking for New Chemical Entities or Predicting Clinical Response. Med. Aromat. Plants 2012, 1. [Google Scholar] [CrossRef]
- Thiem, B.; Goślińska, O. Antimicrobial activity of Solidago virgaurea L. from in vitro cultures. Fitoterapia 2002, 73, 514–516. [Google Scholar] [CrossRef]
- Brantner, A.; Grein, E. Antibacterial activity of plant extracts used externally in traditional medicine. J. Ethnopharmacol. 1994, 44, 35–40. [Google Scholar] [CrossRef]
- Brantner, A. Die antimikrobielle Wirkung von Solidago- haltigen Phytourologica. Drogen-Report 1999, 12, 27–28. [Google Scholar]
- Nair, R.; Kalariya, T.; Chanda, S. Antibacterial Activity of Some Plant Extracts Used in Folk Medicine. J. Herb. Pharmacother. 2008, 7, 191–201. [Google Scholar] [CrossRef]
- Alves, T.M.D.A.; Silva, A.F.; Brandão, M.; Grandi, T.S.M.; Smânia, E.D.F.A.; Smânia Júnior, A.; Zani, C.L. Biological screening of Brazilian medicinal plants. Memórias Do Inst. Oswaldo Cruz 2000, 95, 367–373. [Google Scholar] [CrossRef]
- Kalemba, D. Constituents and biological activity of the essential oils of some Solidago and Artemisia species. Sci. Pap. Tech. Univ. Lodz 2000, 857, 1–118. [Google Scholar]
- Xie, Y.; Yang, W.; Tang, F.; Chen, X.; Ren, L. Antibacterial activities of flavonoids: Structure-activity relationship and mechanism. Curr. Med. Chem. 2015, 22, 132–149. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Du, C.; Beaman, H.T.; Monroe, M.B.B. Characterization of Phenolic Acid Antimicrobial and Antioxidant Structure–Property Relationships. Pharmaceutics 2020, 12, 419. [Google Scholar] [CrossRef] [PubMed]
- Guimarães, A.C.; Meireles, L.M.; Lemos, M.F.; Guimarães, M.C.C.; Endringer, D.C.; Fronza, M.; Scherer, R. Antibacterial Activity of Terpenes and Terpenoids Present in Essential Oils. Molecules 2019, 24, 2471. [Google Scholar] [CrossRef]
- Wińska, K.; Mączka, W.; Łyczko, J.; Grabarczyk, M.; Czubaszek, A.; Szumny, A. Essential Oils as Antimicrobial Agents—Myth or Real Alternative? Molecules 2019, 24, 2130. [Google Scholar] [CrossRef]
- Bader, G.; Seibold, M.; Tintelnot, K.; Hiller, K. Cytotoxicity of triterpenoid saponins. Part 2: Relationships between the structures of glycosides of polygalacic acid and their activities against pathogenic Candida species. Pharmazie 2000, 55, 72–74. [Google Scholar]
- Pepeljnjak, S.; Kuštrak, D.; Vukušić, I. Investigation of the Antimycotic Activities of Solidago virgaurea and Solidago gigantea Exstracts. In Proceedings of the 45th Annual Congress of the Society for Medicinal Plant Research, Regensburg, Germany, 7–12 September 1997; Program and Abstracts. p. K18. [Google Scholar]
- Matsuda, H. Studies on chemical structures and pharmacological activities of triterpene saponins. Nat. Med. 1999, 53, 223–228. [Google Scholar]
- Derda, M.; Hadaś, E.; Thiem, B. Plant extracts as natural amoebicidal agents. Parasitol. Res. 2009, 104, 705–708. [Google Scholar] [CrossRef]
- Váradyová, Z.; Pisarčíková, J.; Babják, M.; Hodges, A.; Mravčáková, D.; Kišidayová, S.; Königová, A.; Vadlejch, J.; Várady, M. Ovicidal and larvicidal activity of extracts from medicinal-plants against Haemonchus contortus. Exp. Parasitol. 2018, 195, 71–77. [Google Scholar] [CrossRef]
- Plohmann, B.; Bader, G.; Hiller, K.; Franz, G. Immunomodulatory and antitumoral effects of triterpenoid saponins. Die Pharm. 1997, 52, 953–957. [Google Scholar]
- Bader, G.; Plohmann, B.; Hiller, K.; Franz, G. Cytotoxicity of triterpenoid saponins. Part 1: Activities against tumor cells in vitro and hemolytical index. Pharmazie 1996, 51, 414–417. [Google Scholar]
- Hu, W.; Shen, T.; Wang, M.-H. Cell cycle arrest and apoptosis induced by methyl 3,5-dicaffeoyl quinate in human colon cancer cells: Involvement of the PI3K/Akt and MAP kinase pathways. Chem.-Biol. Interact. 2011, 194, 48–57. [Google Scholar] [CrossRef]
- Jang, Y.S.; Kim, H.-Y.; Zuo, G.; Lee, E.H.; Kang, S.K.; Lim, S.S. Constituents from Solidago virgaurea var. gigantea and their inhibitory effect on lipid accumulation. Fitoterapia 2020, 146, 104683. [Google Scholar] [CrossRef]
- Wang, Z.; Kim, J.H.; Jang, Y.S.; Kim, C.H.; Lee, J.-Y.; Lim, S.S. Anti-obesity effect of Solidago virgaurea var. g igantea extract through regulation of adipogenesis and lipogenesis pathways in high-fat diet-induced obese mice (C57BL/6N). Food Nutr. Res. 2017, 61, 1273479. [Google Scholar] [CrossRef]
- Sanad, F.A.-A.; Ahmed, S.F.; El-Tantawy, W.H. Antidiabetic and hypolipidemic potentials of Solidago virgaurea extract in alloxan-induced diabetes type 1. Arch. Physiol. Biochem. 2020, 1–8. [Google Scholar] [CrossRef]
- El-Tantawy, W.H. Biochemical effects of Solidago virgaurea extract on experimental cardiotoxicity. J. Physiol. Biochem. 2014, 70, 33–42. [Google Scholar] [CrossRef]
- Lämmermann, I.; Terlecki-Zaniewicz, L.; Weinmüllner, R.; Schosserer, M.; Dellago, H.; de Matos Branco, A.D.; Autheried, D.; Sevcnikar, B.; Kleissl, L.; Berlin, I.; et al. Blocking negative effects of senescence in human skin fibroblasts with a plant extract. NPJ Aging Mech. Dis. 2018, 4, 1–10. [Google Scholar] [CrossRef]

| Compound | Proportion (%) | Reference(s) |
|---|---|---|
| α-Pinene | 0.47–36.5 | [35,76,77,78] |
| Camphene | 0.02–0.6 | [35,76,77,78] |
| Sabinene | 0.06–11.8 | [35,76,77,78] |
| Myrcene | 0.05–17.9 | [35,76,77,78] |
| β-Pinene | 0.16–13.3 | [35,76,77,78] |
| 3-Carene | 0.1–0.7 | [35,76] |
| α-Terpinene | Tr. *–0.3 | [76] |
| Limonene | 0.07–14.8 | [35,76,77,78] |
| p-Cymene | Tr. *–0.77 | [76,77,78] |
| (E)-β-Ocimene | 0.02–4.7 | [35,76,78,79] |
| Linalol | 0.3–0.8 | [76] |
| Nonanal | Tr. **–1.4 | [76,77] |
| trans-Verbenol | Tr. *–0.7 | [76,77] |
| trans-Pinocarveol | 0.09–0.2 | [76,77] |
| Decanal | 0.04–0.7 | [35,76,77] |
| Terpinen-4-ol | 0.1–1.1 | [76,77,78] |
| Borneol | Tr. * | [76,77] |
| α-Terpineol | 0.13–1.89 | [76,77,78] |
| γ-Terpineol | 0.04–0.2 | [76] |
| p-Cymen-8-ol | 0.03–0.51 | [76,78] |
| trans-Carveol | 0.05–0.3 | [76,77] |
| Myrtenal | Tr. *–0.06 | [76,77] |
| Geraniol | 0.02–0.45 | [76,78] |
| Verbenone | Tr. *–0.6 | [76,77] |
| α-Cubebene | Tr.*–2.35 | [76,77,78] |
| δ-Elemene | Tr. *–9.38 | [35,76,77,78] |
| Bornyl acetate | 0.13–4.52 | [35,76,77,78] |
| Carvone | Tr. *–0.4 | [76,77] |
| α-Copaene | Tr. *–0.64 | [35,76,77,78] |
| β-Bourbonene | 0.2–7.28 * | [76,77,78] |
| β-Cubebene | [76,77,78] | |
| β-Elemene | [35,76,77,78] | |
| Geranyl acetate | Tr. *–0.2 | [35,76] |
| Isobutyl benzoate | Tr. * | [76] |
| (Z)-β-Farnesene | Tr. *–0.6 | [76,77,78] |
| β-Caryophyllene | 0.1–10.5 | [35,76,77,78] |
| α-Humulene | 0.1–4.1 | [35,76,77,78] |
| γ-Muurolene | Tr. *–1.86 | [35,76,77,78] |
| Germacrene-D | 0.1–17.68 | [35,76,77,78] |
| Isoamyl benzoate | 0.08–0.4 | [35,76] |
| α-Muurolene | Tr. *–3.6 | [35,76,77,78] |
| Bicyclogermacrene | Tr. *–0.9 | [35,76,77] |
| γ-Cadinene | Tr. *–0.7 | [35,76] |
| Nerolidol | 0.07–0.6 | [35,76,77,78] |
| Calamenene A | Tr. *–0.2 | [76,77,78] |
| Caryophyllene epoxide | 0.4–1.6 | [76,77] |
| Spathulenol | 0.29–11.33 | [76,78] |
| (Z)-hex-3-enyl benzoate | 0.08–0.8 | [76,77] |
| Torreyol | Tr. *–0.6 | [76,77] |
| T-Muurolol | 0.2–1.16 | [76,77] |
| Humulene epoxide | 0.2–0.5 | [76,77] |
| α-Cadinol | Tr. **–3.06 | [35,76,77,78] |
| (Z)-hex-3-enyl salicylate | 0.09–0.3 | [76,77] |
| Eudesma-4(15),7-dien | 0.1–0.2 | [76] |
| Mintsulphide | Tr. * | [76] |
| Cyclocolorenone | Tr. *–0.3 | [76,77] |
| Benzyl benzoate | Tr. *–57.0 | [35,76,77,78] |
| Geranyl benzoate | Tr. *–0.1 | [76] |
| Benzyl salicylate | 0.02–1.14 | [35,76,77,78] |
| β-Phenylethyl salicylate | 0.1–0.6 | [76,77] |
| α-Thujene | Tr. * | [77] |
| Linalool | 0.23–2.0 | [77,78] |
| Perillene | 0.3 | [77] |
| Campholene aldehyde | 0.6 | [77] |
| Pinocarvone | 0.5 | [77] |
| ar-Curcumene | 0.5 | [77] |
| Spathulenol | 1.6 | [77] |
| Salvial-4(14)-en-1-one | 0.1 | [77] |
| Torilenol | 0.8 | [77] |
| Junenol | 0.2 | [77] |
| Eudesma-4(15),7-dien-1β-ol | 0.1 | [77] |
| Acetone | 0.02–0.62 | [78] |
| Ethyl acetate | 0.02–2.27 | [78] |
| Ethyl alcohol | 0.03–1.62 | [78] |
| α-Phellandren | Tr. *–1.12 | [78] |
| β-Phellandren | 0.07–0.26 *** | [35,78] |
| Terpinolene | Tr. *–0.2 | [35,78] |
| n-Hexanol | 0.02–0.10 | [78] |
| cis-3-hexen-1-ol | 0.16–1.52 | [78] |
| trans-Linalool-oxide | Tr. *–0.11 | [78] |
| trans-sabinen-hydrate | Tr. *–0.26 | [78] |
| cis-Linalool-oxide | Tr. *–0.06 | [78] |
| Benzaldehyde | 0.13–0.40 | [78] |
| γ-Elemene | Tr.*–0.06 | [78] |
| Aromadendrene | 0.03–0.20 | [78] |
| Acetophenone | Tr. *–0.19 | [78] |
| Salicyl aldehide | Tr. *–0.02 | [78] |
| trans-β-Farnesene | 0.07–4.80 | [35,78] |
| Germacrene-B | 0.03–16.63 | [78] |
| cis-α-Farnesene | Tr. *–0.41 | [78] |
| δ-Cadinene | 0.2–7.87 | [35,78] |
| Cubenene | 0.02–0.25 | [78] |
| Benzyl alcohol | 0.19–1.80 | [78] |
| 2-Phenyl ethyl alcohol | 0.09–1.18 | [78] |
| o-Methoxy benzaldehyde | 0.02–0.38 | [78] |
| Caryophyllene oxide | 0.1–4.30 | [35,78] |
| Epi-cubenol | 0.04–2.79 | [78] |
| o-Methoxy benzyl alcohol | 0.07–1.29 | [78] |
| T-Cadinol | 0.12–0.76 | [78] |
| T-Muurolol | 0.25–1.16 | [78] |
| δ-Cadinol | 0.15–0.73 | [78] |
| n-Tetracosane | Tr. *–0.24 | [78] |
| n-Pentacosane | Tr. *–0.50 | [78] |
| Phytol | 0.02–1.07 | [78] |
| n-hexacosane | Tr. *–0.30 | [78] |
| Myristic acid | Tr. *–1.30 | [78] |
| β-Ocimene-Y/(Z)- β-ocimene | 0.02–3.0 | [35,78,79] |
| γ-Terpinene | Tr. ** | [35] |
| 1-undecene | Tr. **–0.1 | [35] |
| 4,8-dimethyl-1,3,7-nonatriene | 0.1 | [35] |
| Camphor | Tr. **–0.2 | [35] |
| Zingiberene | 0.4–1.1 | [35] |
| Germacrene A | 0.1–0.7 | [35] |
| (E,E)- α -farnesene | 1.0–2.7 | [35] |
| β-sesquiphellandrene | 0.1–0.2 | [35] |
| (Z)-3-hexenyl benzoate | 0.1–0.4 | [35] |
| β-Eudesmol | Tr. **–0.1 | [35] |
| Neophytadiene | 0.1–0.2 | [35] |
| 2-phenylethyl benzoate | Tr. **–0.4 | [35] |
| Pharmacological Properties | Evidence | Chemical Compounds/Fraction to Which the Property is Ascribed | Critical Assessment of Study Results |
|---|---|---|---|
| Antioxidant effects | In vitro | Caffeoylquinic acids | Moderate antioxidant effects (11th position among 23 herbal products) |
| Antiinflammatory effects | In vitro and in vivo (at least 4 rat studies) | Triterpenes, leiocarposide, rutin and quercetin, caffeoylquinic derivatives | Effect not superior to conventional NSAIDs (phenylbutazone, indomethacin) |
| Analgesic activity | In vivo (one study in mice) | Leiocarposide | Similar effects to aminophenazone, but very short duration (one hour) |
| Spasmolytic activity | In vitro or ex vivo | NA | Modest spasmolytic effect (less than 15% of the papaverine effect) |
| Antihypertensive activity | In vivo (one study in dogs and one in rats) | Antihypertensive activity attributed to flavonols | Contradictory results on blood pressure |
| Diuretic effects | In vitro, in vivo (at least three rat studies) | Flavonoid fraction (quercetin and its derivatives), hydroxycinnamic acid fraction, saponin fraction, leiocarposide | Effects comparable to those of furosemide or slightly inferior for several fractions |
| Control of overactive bladder symptoms | In vitro data showing antimuscarinic effects and two clinical, open, un-controlled data | NA | Low quality, but promising data. Contradictory findings between reduction of urination frequency seen in clinical data and diuretic effects reported in non-clinical studies |
| Antilithiatic effects | One rat study | Leiocarposide | Additional confirmation needed |
| Antibacterial effects | In vitro data only | Matricaria ester isomers, clerodane diterpenes (but their effects are rather modest) | Modest effects in the majority of studies. One disc diffusion study reported encouraging results on S. aureus and E. fecalis for a methanol extract. |
| Antifungal activity | In vitro data only | Triterpene saponins | Modest effects for most fungi tested up to date. Inhibition of inhibition of Candida biofilm formation and of yeast-hyphal switch seems more promising, but the clinical relevance is unclear. |
| Antiparasite activity | One in vivo study for Acanthamoeba, one in vitro study for Haemonchus contortus | NA | Promising results for Acanthamoeba (additional confirmation needed). No effect against H. contortus |
| Cytotoxic and antitumor activity | Mostly in vitro data, one in vivo study (mice) | Saponins, α-tocopherol quinone, 2-phyten-l-ol, a protein, β–dictyopterol, methyl 3,5-dicaffeoyl quinate | Moderate effects against some cancer cell lines. More research needed. |
| Antimutagenic activity | In vitro (one study) | Hexane soluble fraction | Effects observed at a quite high concentration (2.5 mg/mL). |
| Antiadipogenic effects | In vitro (two studies) and in vivo (one murine study) | 5-di-O-caffeoylquinic acid | Promising results, further data necessary |
| Antidiabetic effects | One rat model | Hydro-alcoholic fraction | More data necessary |
| Cardioprotective effects | One rat model | NA | More data necessary |
| Antisenescence effects | One in vitro study | NA | More data necessary |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fursenco, C.; Calalb, T.; Uncu, L.; Dinu, M.; Ancuceanu, R. Solidago virgaurea L.: A Review of Its Ethnomedicinal Uses, Phytochemistry, and Pharmacological Activities. Biomolecules 2020, 10, 1619. https://doi.org/10.3390/biom10121619
Fursenco C, Calalb T, Uncu L, Dinu M, Ancuceanu R. Solidago virgaurea L.: A Review of Its Ethnomedicinal Uses, Phytochemistry, and Pharmacological Activities. Biomolecules. 2020; 10(12):1619. https://doi.org/10.3390/biom10121619
Chicago/Turabian StyleFursenco, Cornelia, Tatiana Calalb, Livia Uncu, Mihaela Dinu, and Robert Ancuceanu. 2020. "Solidago virgaurea L.: A Review of Its Ethnomedicinal Uses, Phytochemistry, and Pharmacological Activities" Biomolecules 10, no. 12: 1619. https://doi.org/10.3390/biom10121619
APA StyleFursenco, C., Calalb, T., Uncu, L., Dinu, M., & Ancuceanu, R. (2020). Solidago virgaurea L.: A Review of Its Ethnomedicinal Uses, Phytochemistry, and Pharmacological Activities. Biomolecules, 10(12), 1619. https://doi.org/10.3390/biom10121619