Potent Tyrosinase Inhibitory Activity of Curcuminoid Analogues and Inhibition Kinetics Studies
Abstract
1. Introduction
2. Materials and Methods
2.1. Chemical Reagents and General Experimental Procedures
2.2. Chemical Modification of Curcuminoids
2.3. Tyrosinase Inhibition Assay
2.4. Kinetic Analysis of Tyrosinase Inhibition Assay
2.5. Statistical Analysis
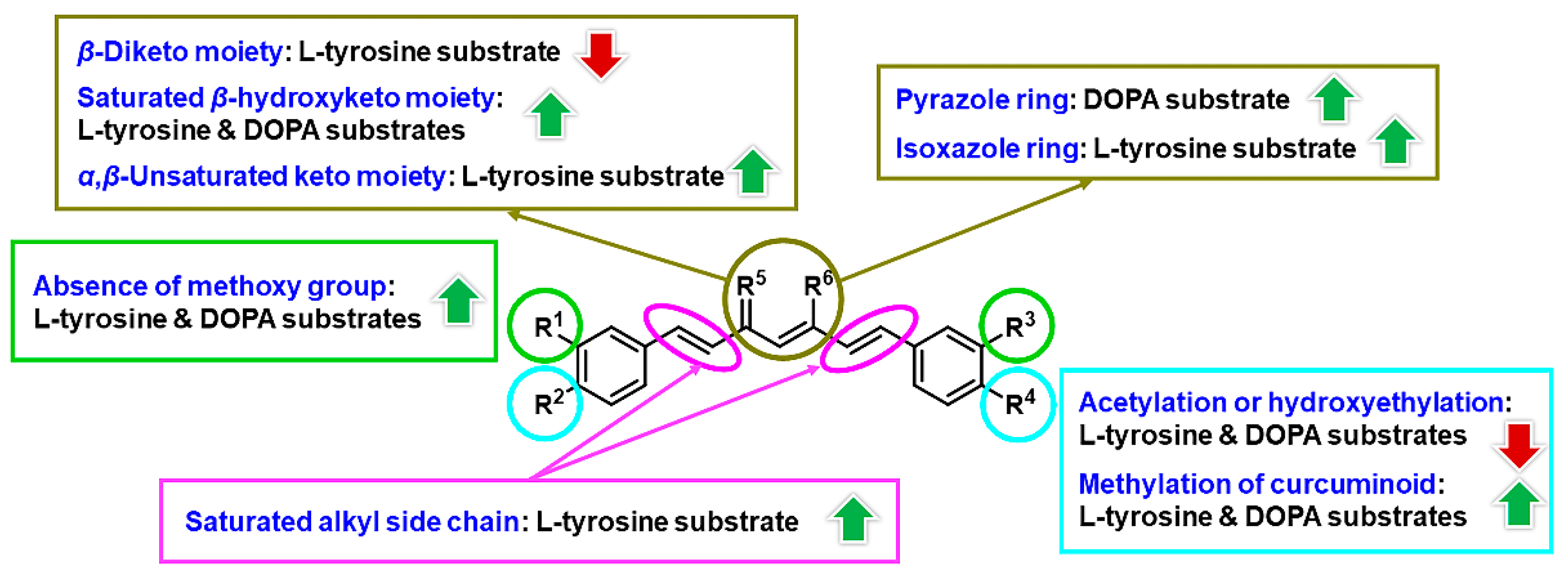
3. Results and Discussion
3.1. Chemistry
3.2. Inhibitory Effects on Tyrosinase
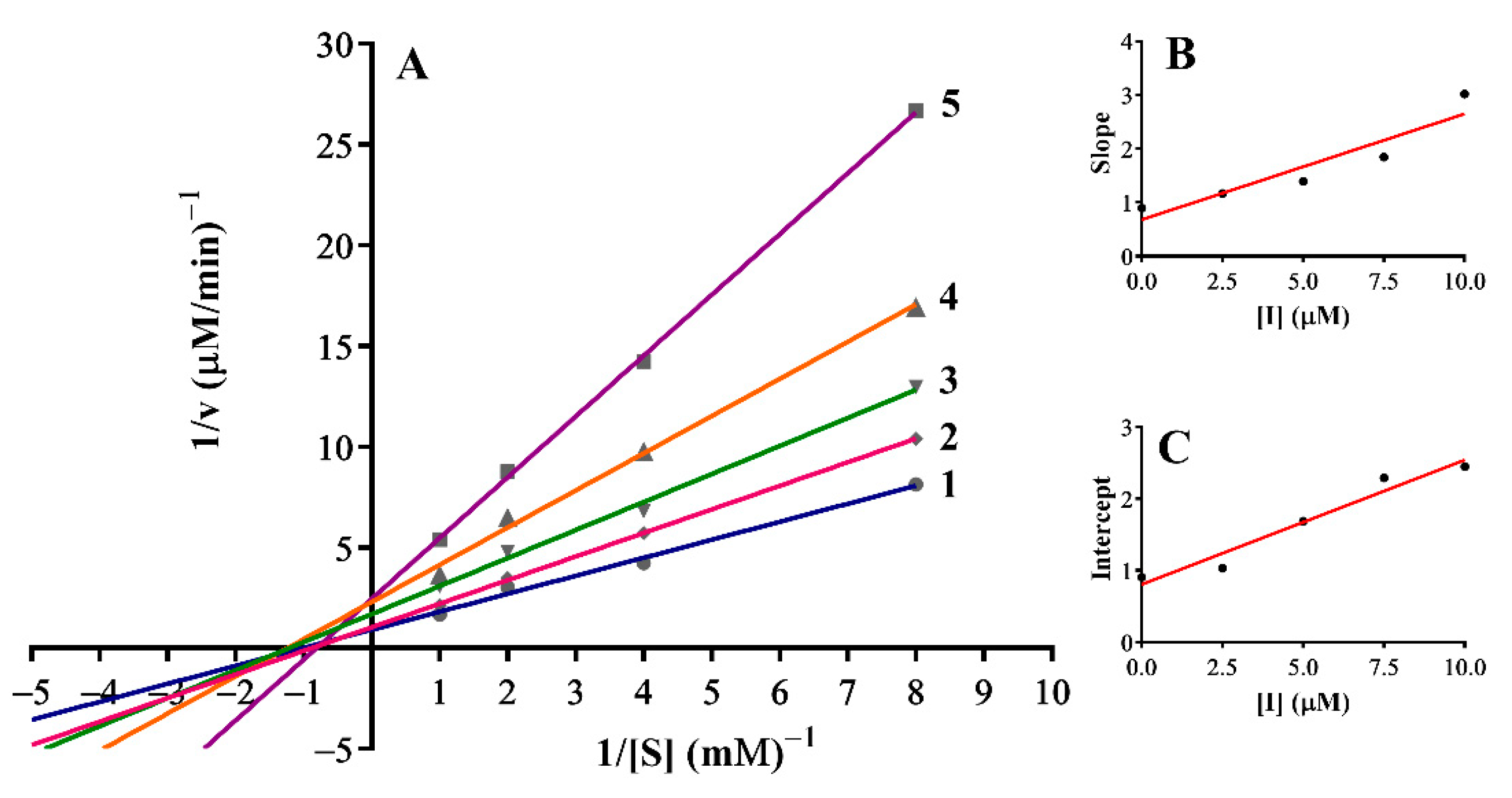
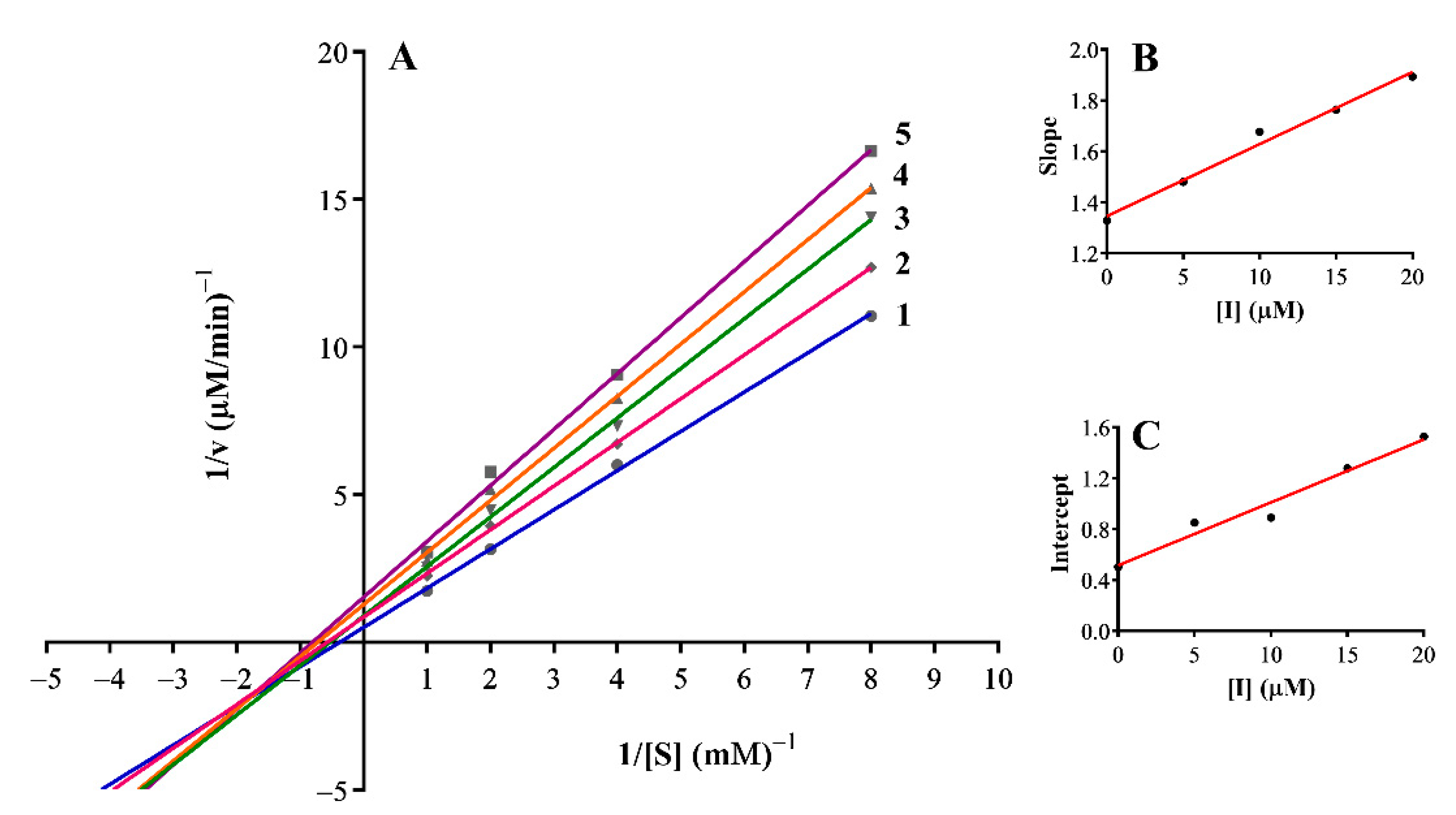
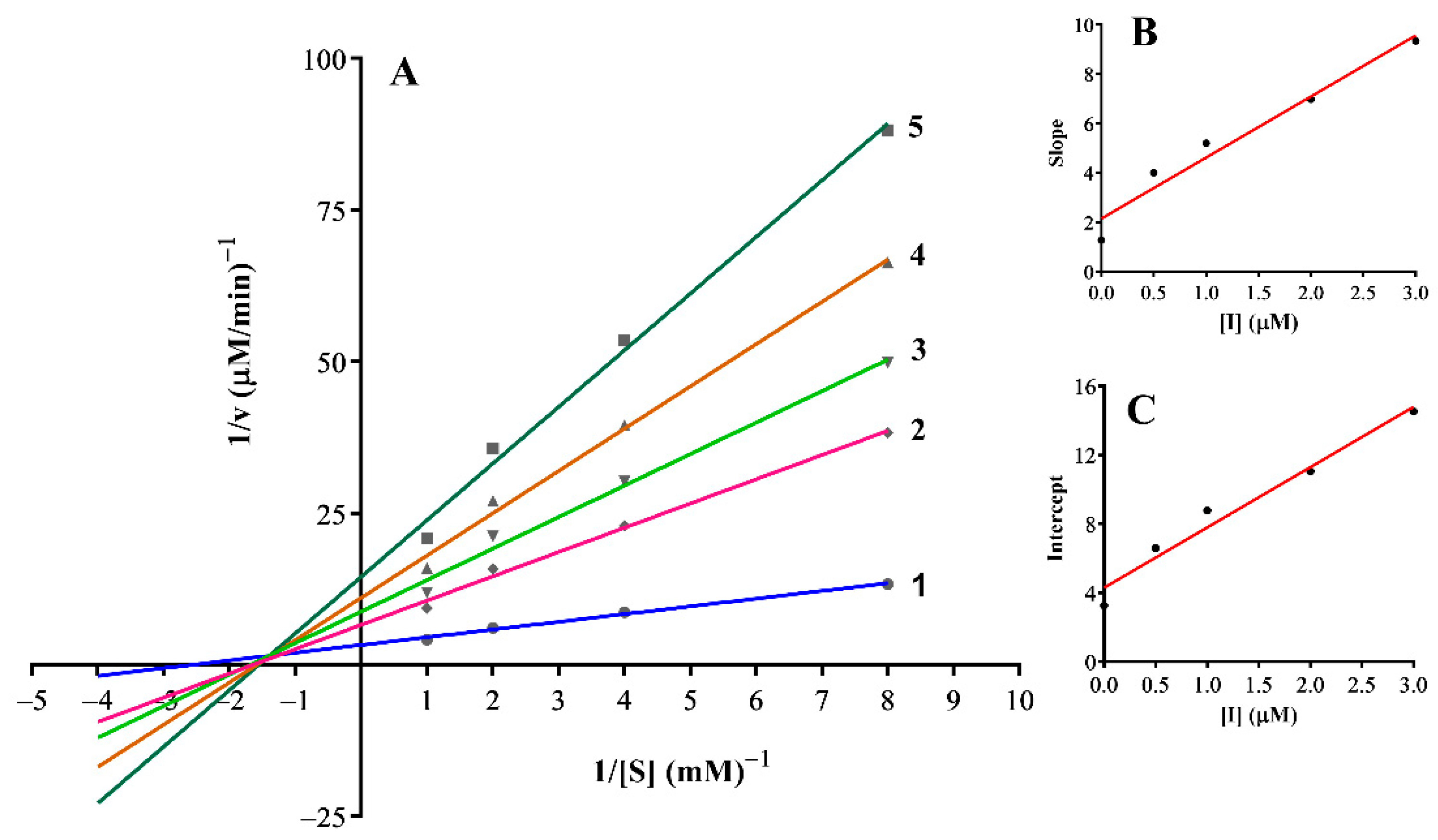
3.3. Inhibition Mechanism on the Mushroom Tyrosinase
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Chang, T.-S. An updated review of tyrosinase inhibitors. Int. J. Mol. Sci. 2009, 10, 2440–2475. [Google Scholar] [CrossRef] [PubMed]
- Sharifi-Rad, J.; Rayess, Y.E.; Rizk, A.A.; Sadaka, C.; Zgheib, R.; Zam, W.; Sestito, S.; Rapposelli, S.; Neffe-Skocińska, K.; Zielińska, D.; et al. Turmeric and its major compound curcumin on health: Bioactive effects and safety profiles for food, pharmaceutical, biotechnological and medicinal applications. Front. Pharmacol. 2020, 11, 01021. [Google Scholar] [CrossRef] [PubMed]
- Jakubczyk, K.; Drużga, A.; Katarzyna, J.; Skonieczna-Żydecka, K. Antioxidant potential of curcumin–A meta-analysis of randomized clinical trials. Antioxidants 2020, 9, 1092. [Google Scholar] [CrossRef] [PubMed]
- Lee, K.-H.; Aziz, F.H.; Syahida, A.; Abas, F.; Shaari, K.; Israf, D.A.; Lajis, N.H. Synthesis and biological evaluation of curcumin-like diarylpentanoid analogues for anti-inflammatory, antioxidant and anti-tyrosinase activities. Eur. J. Med. Chem. 2009, 44, 3195–3200. [Google Scholar] [CrossRef]
- Tomeh, M.A.; Hadianamrei, R.; Zhao, X. A review of curcumin and its derivatives as anticancer agents. Int. J. Mol. Sci. 2019, 20, 1033. [Google Scholar] [CrossRef]
- Zheng, D.; Huang, C.; Huang, H.; Zhao, Y.; Khan, M.R.U.; Zhao, H.; Huang, L. Antibacterial mechanism of curcumin: A review. Chem. Biodivers. 2020, 17, e2000171. [Google Scholar] [CrossRef]
- Adamczak, A.; Ożarowski, M.; Karpiński, T.M. Curcumin, a natural antimicrobial agent with strain-specific activity. Pharmaceuticals 2020, 13, 153. [Google Scholar] [CrossRef] [PubMed]
- Jennings, M.R.; Parks, R.J. Curcumin as an antiviral agent. Viruses 2020, 12, 1242. [Google Scholar] [CrossRef]
- Nistico, S.; Tamburi, F.; Bennardo, L.; Dastoli, S.; Schipani, G.; Caro, G.; Fortuna, M.C.; Rossi, A. Treatment of telogen effluvium using a dietary supplement containing Boswellia serrata, Curcuma longa, and Vitis vinifera: Results of an observational study. Dermatol. Ther. 2019, 32, e12842. [Google Scholar] [CrossRef] [PubMed]
- Vollono, L.; Falconi, M.; Gaziano, R.; Iacovelli, F.; Dika, E.; Terracciano, C.; Bianchi, L.; Campione, E. Potential of curcumin in skin disorders. Nutrients 2019, 11, 2169. [Google Scholar]
- Panahi, Y.; Fazlolahzadeh, O.; Atkin, S.L.; Majeed, M.; Butler, A.E.; Johnston, T.P.; Sahebkar, A. Evidence of curcumin and curcumin analogue effects in skin diseases: A narrative review. J. Cell Physiol. 2019, 234, 1165–1178. [Google Scholar] [CrossRef] [PubMed]
- Du, Z.-Y.; Jiang, Y.-F.; Tang, Z.-K.; Mo, R.-Q.; Xue, G.-H.; Lu, Y.-J.; Zheng, X.; Dong, C.-Z.; Zhang, K. Antioxidant and tyrosinase inhibition of polyphenolic curcumin analogs. Biosci. Biotechnol. Biochem. 2011, 75, 2351–2358. [Google Scholar] [CrossRef]
- Bhullar, K.S.; Jha, A.; Youssef, D.; Rupasinghe, H.P.V. Curcumin and its carbocyclic analogs: Structure-activity in relation to antioxidant and selected biological properties. Molecules 2013, 18, 5389–5404. [Google Scholar] [CrossRef]
- Jiang, Y.; Du, Z.; Xue, G.; Chen, Q.; Lu, Y.; Zheng, X.; Conney, A.H.; Zhang, K. Synthesis and biological evaluation of unsymmetrical curcumin analogues as tyrosinase inhibitors. Molecules 2013, 18, 3948–3961. [Google Scholar] [CrossRef]
- Bukhari, S.N.; Jantan, I.; Unsal Tan, O.; Sher, M.; Naeem-ul-Hassan, M.; Qin, H.-L. Biological activity and molecular docking studies of curcumin-related α,β-unsaturated carbonyl-based synthetic compounds as anticancer agents and mushroom tyrosinase inhibitors. J. Agric. Food Chem. 2014, 62, 5538–5547. [Google Scholar] [CrossRef] [PubMed]
- Shirota, S.; Miyazaki, K.; Aiyama, R.; Ichioka, M.; Yokokura, T. Tyrosinase inhibitors from crude drugs. Biol. Pharm. Bull. 1994, 17, 266–269. [Google Scholar] [CrossRef][Green Version]
- Nguyen, M.H.; Nguyen, H.X.; Nguyen, M.T.; Nguyen, N.T. Phenolic constituents from the heartwood of Artocapus altilis and their tyrosinase inhibitory activity. Nat. Prod. Commun. 2012, 7, 185–186. [Google Scholar] [CrossRef] [PubMed]
- Changtam, C.; De Koning, H.P.; Ibrahim, H.; Sajid, M.S.; Gould, M.K.; Suksamrarn, A. Curcuminoid analogs with potent activity against Trypanosoma and Leishmania species. Eur. J. Med. Chem. 2010, 45, 941–956. [Google Scholar] [CrossRef]
- Changtam, C.; Hongmanee, P.; Suksamrarn, A. Isoxazole analogs of curcuminoids with highly potent multidrug-resistant antimycobacterial activity. Eur. J. Med. Chem. 2010, 45, 4446–4457. [Google Scholar] [CrossRef]
- Chen, J.; Sun, H.; Tao, X.; Wang, S.; Sun, A. Inhibitory mechanism and kinetics study of apple polyphenols on the activity of tyrosinase. Int. J. Food Prop. 2014, 17, 1694–1701. [Google Scholar] [CrossRef]
- Chen, Q.-X.; Song, K.-K.; Qui, L.; Liu, X.-D.; Huang, H.; Guo, H.-Y. Inhibitory effects on mushroom tyrosinase by p-alkoxybenzoic acids. Food Chem. 2005, 91, 269–274. [Google Scholar] [CrossRef]
- Ghani, U.; Ullah, N. New potent inhibitors of tyrosinase: Novel clues to binding of 1,3,4-thiadiazole-2(3H)-thiones, 1,3,4-oxadiazole-2(3H)-thiones, 4-amino-1,2,4-triazole-5(4H)-thiones, and substituted hydrazides to the dicopper active site. Bioorg. Med. Chem. 2010, 18, 4042–4048. [Google Scholar] [CrossRef] [PubMed]
- Zolghadri, S.; Bahrami, A.; Khan, M.T.H.; Munoz-Munoz, J.; Garcia-Molina, F.; Garcia-Canovas, F.; Saboury, A.A. A comprehensive review on tyrosinase inhibitors. J. Enzym. Inhib. Med. Chem. 2019, 34, 279–309. [Google Scholar] [CrossRef]
- Noh, J.-M.; Lee, Y.-S. Inhibitory activities of hydroxyphenolic acid-amino acid conjugates on tyrosinase. Food Chem. 2011, 125, 953–957. [Google Scholar] [CrossRef]
- Fan, Q.; Jiang, H.; Yuan, E.-D.; Zhang, J.-X.; Ning, Z.-X.; Qi, S.-J.; Wei, Q.-Y. Tyrosinase inhibitory effects and antioxidative activities of novel cinnamoyl amides with amino acid ester moiety. Food Chem. 2012, 134, 1081–1087. [Google Scholar] [CrossRef] [PubMed]
- Radhakrishnan, S.; Shimmon, R.; Conn, C.; Baker, A. Design, synthesis and biological evaluation of hydroxy substituted amino chalcone compounds for antityrosinase activity in B16 cells. Bioorg. Chem. 2015, 62, 117–123. [Google Scholar] [CrossRef]
| Compound | Structure | Tyrosinase Inhibitory Activity (IC50; µM) 1 | |
|---|---|---|---|
| L-tyrosine | DOPA | ||
| 1 | 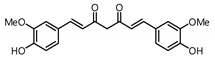 | 326.5 | 94.8 |
| 2 | 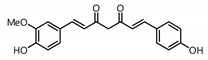 | 470.0 | 41.1 |
| 3 |  | 46.5 | 34.8 |
| 4 | 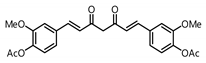 | Inactive 2 | Inactive 2 |
| 5 | 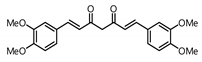 | 86.2 | 8.0 |
| 6 | 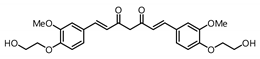 | Inactive 2 | Inactive 2 |
| 7 |  | 66.9 | 54.6 |
| 8 | 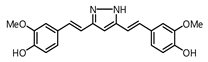 | 677.1 | 21.9 |
| 9 | 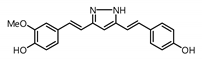 | 154.4 | 19.7 |
| 10 | 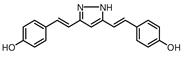 | 129.1 | 19.2 |
| 11 | 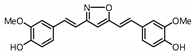 | 24.3 | 330.9 |
| 12 | 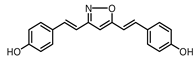 | 8.3 | 86.2 |
| 13 | 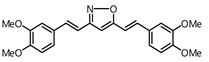 | Inactive 2 | Inactive 2 |
| 14 | 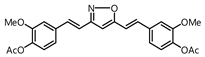 | Inactive 2 | Inactive 2 |
| 15 | 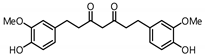 | 51.6 | 283.1 |
| 16 |  | 14.6 | 25.4 |
| 17 | 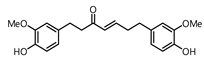 | 9.4 | 63.5 |
| Kojic acid 3 | 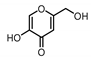 | 173.2 | 132.8 |
| Compound | Inhibition Type | Inhibition Constants (µM) | |
|---|---|---|---|
| KI | KIS | ||
| 5 | Mixed-II type | 4.64 | 3.44 |
| 12 | Non-competitive | 0.12 | 0.08 |
| 17 | Mixed-I type | 0.01 | 0.05 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Athipornchai, A.; Niyomtham, N.; Pabuprapap, W.; Ajavakom, V.; Duca, M.; Azoulay, S.; Suksamrarn, A. Potent Tyrosinase Inhibitory Activity of Curcuminoid Analogues and Inhibition Kinetics Studies. Cosmetics 2021, 8, 35. https://doi.org/10.3390/cosmetics8020035
Athipornchai A, Niyomtham N, Pabuprapap W, Ajavakom V, Duca M, Azoulay S, Suksamrarn A. Potent Tyrosinase Inhibitory Activity of Curcuminoid Analogues and Inhibition Kinetics Studies. Cosmetics. 2021; 8(2):35. https://doi.org/10.3390/cosmetics8020035
Chicago/Turabian StyleAthipornchai, Anan, Nattisa Niyomtham, Wachirachai Pabuprapap, Vachiraporn Ajavakom, Maria Duca, Stéphane Azoulay, and Apichart Suksamrarn. 2021. "Potent Tyrosinase Inhibitory Activity of Curcuminoid Analogues and Inhibition Kinetics Studies" Cosmetics 8, no. 2: 35. https://doi.org/10.3390/cosmetics8020035
APA StyleAthipornchai, A., Niyomtham, N., Pabuprapap, W., Ajavakom, V., Duca, M., Azoulay, S., & Suksamrarn, A. (2021). Potent Tyrosinase Inhibitory Activity of Curcuminoid Analogues and Inhibition Kinetics Studies. Cosmetics, 8(2), 35. https://doi.org/10.3390/cosmetics8020035






