Lake Zeiļu Clay Application Induced Changes in Human Skin Hydration, Elasticity, Transepidermal Water Loss and PH in Healthy Individuals
Abstract
1. Introduction
2. Materials and Methods
- Acne vulgaris or local therapy for acne vulgaris in the past 2 months;
- Localized infection, herpes simplex reactivation, impetigo, psoriasis and rosacea;
- Isoretinoine therapy or photodynamic therapy in the past 6 months;
- Fractional or full facial laser ablation, radiofrequency facial treatment, medium or deep chemical peel and acne scar filling in the past 6 months;
- Decompensated diabetes;
- Light chemical peel, microneedling procedure, mesotherapy and biorevitalization in the past 2 months;
- Systemic antibacterial therapy in the past 2 months;
- Pregnancy or lactation.
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Massaro, M.; Colletti, C.G.; Lazzara, G.; Riela, S. The use of some clay minerals as natural resources for drug carrier applications. J. Funct. Biomater. 2018, 9, 58. [Google Scholar] [CrossRef]
- Forest and Earth Entrails Resources. Available online: https://agris.fao.org/agris-search/search.do?recordID=LV2019000458 (assessed on 19 June 2020).
- Vecstaudža, J.; Stunda-Zujeva, A.; Irbe, Z.; Bērziņa-Cimdiņa, L. Composition of commercial cosmetic clay and suitability of latvian clay for cosmetic purposes. Mater. Sci. Appl. Chem. 2012, 26, 42–48. [Google Scholar]
- Haydel, S.; Remenih, C.; Williams, L. Broad-spectrum in vitro antibacterial activities of clay minerals against antibiotic-susceptible and antibiotic-resistant bacterial pathogens. J. Antimicrob. Chemother. 2008, 61, 353–361. [Google Scholar] [CrossRef]
- Ghiaci, M.; Aghaei, H.; Soleimanian, S.; Sedaghat, S. Enzyme immobilization: Part 1. Modified bentonite as a new and efficient support for immobilization of candida rugosa lipase. Appl. Clay Sci. 2009, 43, 289–295. [Google Scholar] [CrossRef]
- Ferrell, R.E. Medicinal clay and spiritual healing. Clays Clay Min. 2008, 56, 751–760. [Google Scholar] [CrossRef]
- Williams, L.B.; Haydel, S.E. Evaluation of the medicinal use of clay minerals as antibacterial agents. Int. Geol. Rev. 2010, 52, 745–770. [Google Scholar] [CrossRef]
- Carretero, M.I.; Lagaly, G. Clays and health: An introduction. Appl. Clay Sci. 2007, 36, 1–3. [Google Scholar]
- Carretero, M.I. Clay minerals and their beneficial effects upon human health. A review. Appl. Clay Sci. 2002, 21, 155–163. [Google Scholar] [CrossRef]
- Gomes, C.S.F.; Silva, J.B.P. Minerals and clay minerals in medical geology. Appl. Clay Sci. 2007, 36, 4–21. [Google Scholar] [CrossRef]
- Williams, L.B.; Haydel, S.E.; Giese, R.F.; Eberl, D.D. Chemical and mineralogical characteristics of french green clays used for healing. Clays and Clay Miner. 2008, 56, 437–452. [Google Scholar] [CrossRef]
- Carretero, M.I.; Gomes, C.S.F.; Tateo, F. 5 clays and human health. Dev. Clay Sci. 2006, 1, 717–741. [Google Scholar]
- Meier, L.; Stange, R.; Michalsen, A.; Uehleke, B. Clay jojoba oil facial mask for lesioned skin and mild acne–results of a prospective, Observational Pilot Study. Complementary Med. Res. 2012, 19, 75–79. [Google Scholar] [CrossRef]
- Mpuchane, S.F.; Ekosse, G.I.E.; Gashe, B.A.; Morobe, I.; Coetzee, S.H. Microbiological characterisation of southern African medicinal and cosmetic clays. Int. J. Environ. Health Res. 2010, 20, 27–41. [Google Scholar] [CrossRef]
- Viseras, C.; Aguzzi, C.; Cerezo, P.; Lopez-Galindo, A. Uses of clay minerals in semisolid health care and therapeutic products. Appl. Clay Sci. 2007, 36, 37–50. [Google Scholar] [CrossRef]
- Williams, L.B. Geomimicry: Harnessing the antibacterial action of clays. Clay Miner. 2017, 52, 1–24. [Google Scholar] [CrossRef][Green Version]
- Morrison, K.D.; Misra, R.; Williams, L.B. Unearthing the antibacterial mechanism of medicinal clay: A geochemical approach to combating antibiotic resistance. Sci. Rep. 2016, 6, 19043. [Google Scholar] [CrossRef]
- Carretero, M.I.; Pozo, M. Clay and non-clay minerals in the pharmaceutical and cosmetic industries Part II. Active ingredients. Appl. Clay Sci. 2010, 47, 171–181. [Google Scholar] [CrossRef]
- Gubitosa, J.; Rizzi, V.; Fini, P.; Cosma, P. Hair care cosmetics: From traditional shampoo to solid clay and herbal shampoo, a review. Cosmetics 2019, 6, 13. [Google Scholar] [CrossRef]
- Dušenkova, I.; Kusiņa, I.; Mālers, J.; Bērziņa-Cimdiņa, L. Application of latvian illite clays in cosmetic products with sun protection ability. In Proceedings of the 10th International Scientific and Practical Conference “Environment. Technologies. Resources”, Rezekne, Latvia, June 18–20 2015; Rezekne Academy of Technologies: Rezekne, Latvia, 2015. [Google Scholar]
- da Silva Favero, J.; dos Santos, V.; Weiss-Angeli, V.; Gomes, L.B.; Veras, D.G.; Dani, N.; Mexias, A.S.; Bergmann, C.P. Evaluation and characterization of Melo Bentonite clay for cosmetic applications. Appl. Clay Sci. 2019, 175, 40–46. [Google Scholar] [CrossRef]
- Tretjakova, R.; Noviks, G.; Mezinskis, G. Investigation of structure and composition of clay in lakes of Latgale for practical use. In Proceedings of the 12th International Scientific and Practical Conference “Environment. Technologies. Resources”, Rezekne, Latvia, June 18–20 2019; Rezekne Academy of Technologies: Rezekne, Latvia, 2019; pp. 298–303. [Google Scholar]
- Evaluation and Characterization of Melo Bentonite Clay for Cosmetic Applications. Available online: https://www.researchgate.net/publication/332379849_Evaluation_and_characterization_of_Melo_Bentonite_clay_for_cosmetic_applications (assessed on 19 June 2020).
- Dário, G.M.; Da Silva, G.G.; Gonçalves, D.L.; Silveira, P.; Junior, A.T.; Angioletto, E.; Bernardin, A.M. Evaluation of the healing activity of therapeutic clay in rat skin wounds. Mater. Sci. Eng. C 2014, 43, 109–116. [Google Scholar] [CrossRef]
- Moraes, J.D.D.; Bertolinob, S.R.A.; Cuffini, S.L.; Ducart, D.F.; Bretzke, P.E.; Leonardi, G.R. Clay minerals: Properties and applications to dermocosmetic products and perspectives of natural raw materials for therapeutic purposes—A review. Int. J. Pharm. 2017, 534, 213–219. [Google Scholar] [CrossRef]
- Carretero, M.I.; Pozo, M. Clay and non-clay minerals in the pharmaceutical industry: Part I. Excipients and medical applications. Appl. Clay Sci. 2009, 46, 73–80. [Google Scholar] [CrossRef]
- Clijsen, R.; Taeymans, J.; Duquet, W.; Barel, A.; Clarys, P. Changes of skin characteristics during and after local Parafango therapy as used in physiotherapy. Ski. Res. Technol. 2008, 14, 237–242. [Google Scholar] [CrossRef]
- Pan-on, S.; Rujivipat, S.; Ounaroon, A.; Kongkaew, C.; Tiyaboonchai, W. Development, characterization and skin irritation of mangosteen peel extract solid dispersion containing clay facial mask. Int. Appl. Pharm. 2018, 10, 202–208. [Google Scholar] [CrossRef]
- Berardesca, E.; Abril, E.; Rona, C.; Vesnaver, R.; Cenni, A.; Oliva, M. An effective night slimming topical treatment. Int. J. Cosmet. Sci. 2012, 34, 263–272. [Google Scholar] [CrossRef]
- Anthonissen, M.; Daly, D.; Fieuws, S.; Massagé, P.; Van Brussel, M.; Vranckx, J.; Van den Kerckhove, E. Measurement of elasticity and transepidermal water loss rate of burn scars with the dermalab. Burns 2013, 39, 420–428. [Google Scholar] [CrossRef]
- Saknite, I.; Zavorins, A.; Zablocka, I.; Kisis, J.; Spigulis, J. Comparison of a near-infrared reflectance spectroscopy system and skin conductance measurements for in vivo estimation of skin hydration: A clinical study. J. Biomed. Photonics Eng. 2017, 3, 1–6. [Google Scholar] [CrossRef]
- What They Are and How They Work. Available online: https://www.skintherapyletter.com/eczema/how-moisturizers-work/ (accessed on 10 May 2020).
- Kim, S.; Hwang, S.M.; Choi, E.H.; Ahn, S.K.; Lee, S.H. The effect of bentonite and glycolic acid on the stratum conium. Ann Derm. 2001, 13, 205–210. [Google Scholar] [CrossRef][Green Version]
- Maria, V.; Zague, V.; Dario, M.; Nishikawa, D.; Pinto, C.; Almeida, M.; Trossini, G.; Vieira-Coelho, A.; Baby, A. Characterization and short-term clinical study of clay facial mask. J. Basic Appl. Pharm. Sci. 2016, 37, 1–6. [Google Scholar]
- Valenti, D.M.Z.; Silva, J.; Teodoro, W.R.; Velosa, A.P.; Mello, S.B.V. Effect of topical clay application on the synthesis of collagen in skin: An experimental study. Clin. Exp. Dermatol. 2012, 37, 164–168. [Google Scholar] [CrossRef]
- Proksch, E. PH in nature, humans and skin. J. Derm. 2018, 45, 1044–1052. [Google Scholar] [CrossRef] [PubMed]
- About Skin PH and Why it Matters. Available online: www.healthline.com/health/whats-so-important-about-skin-ph (accessed on 10 May 2020).
- Ali, S.M.; Yosipovitch, G. Skin pH: From basic science to basic skin care. Acta Derm. Venereol. 2013, 93, 261–267. [Google Scholar] [CrossRef] [PubMed]
- What is Skin’s PH Level and How to Maintain it? Available online: www.stylecraze.com/articles/what-is-the-importance-of-ph-on-your-skin-and-what-you-can-do-about-it/#WhatIsSkinpHLevel (accessed on 10 May 2020).
- Modabberi, S.; Namayandeh, A.; López-Galindo, A.; Viseras, C.; Setti, M.; Ranjbaran, M. Characterization of Iranian bentonites to be used as pharmaceutical materials. Appl. Clay Sci. 2015, 116, 193–201. [Google Scholar] [CrossRef]
- Gamoudi, S.; Srasra, E. Characterization of Tunisian clay suitable for pharmaceutical and cosmetic applications. Appl. Clay Sci. 2017, 146, 162–166. [Google Scholar] [CrossRef]
- Yosipovitch, G.; Hu, J. The importance of skin pH. Ski. Aging 2003, 11, 88–93. [Google Scholar]
- Korting, H.C.; Kober, M.; Mueller, M.; Braun-Falco, O. Influence of repeated washings with soap and synthetic detergents on pH and resident flora of the skin of forehead and forearm. Acta Derm. Venereol. 1987, 67, 41–47. [Google Scholar]
- Moldovan, M.; Nanu, A. Influence of cleansing product type on several skin parameters after single use. Farmacia 2010, 58, 29–37. [Google Scholar]
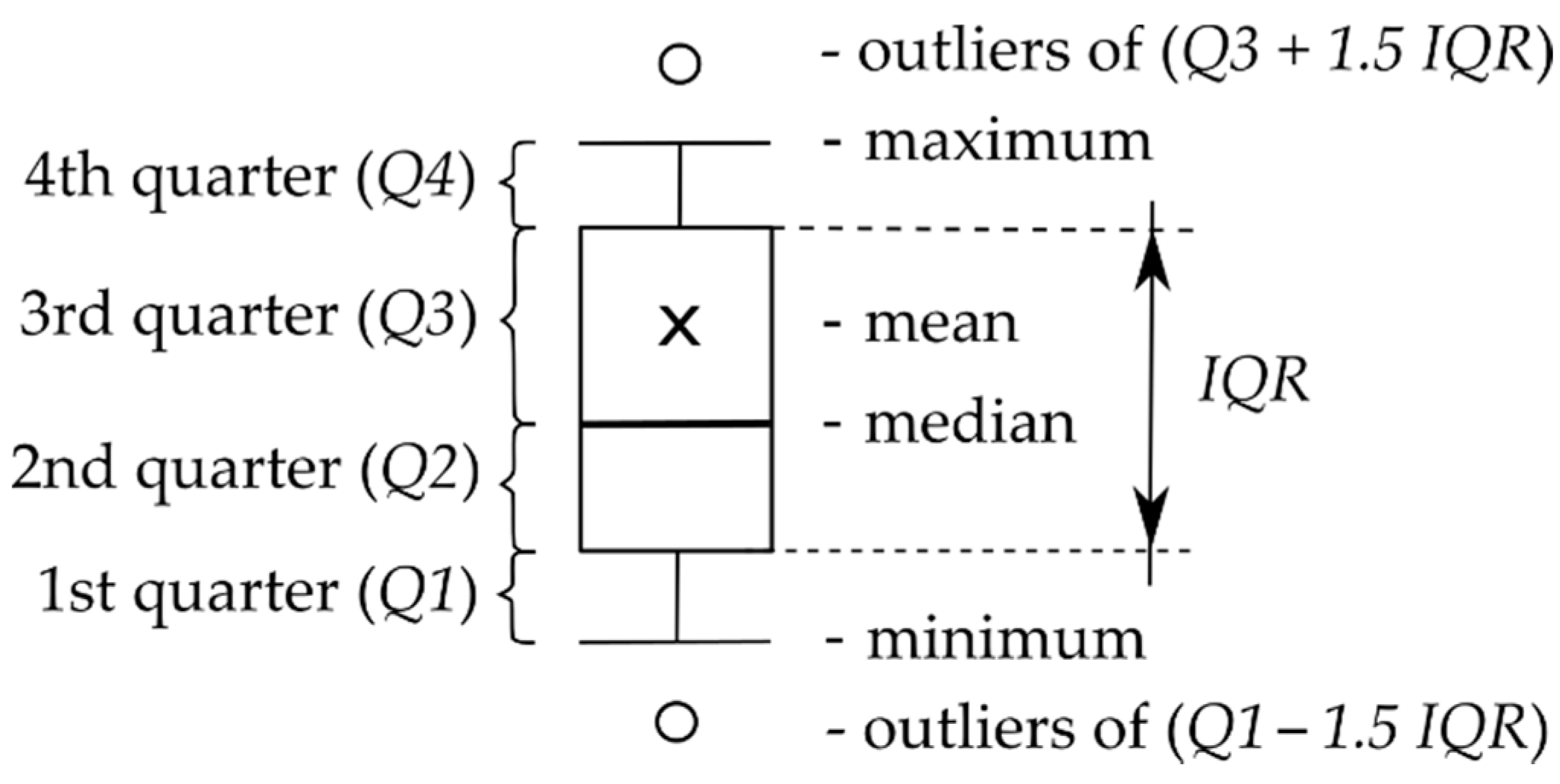
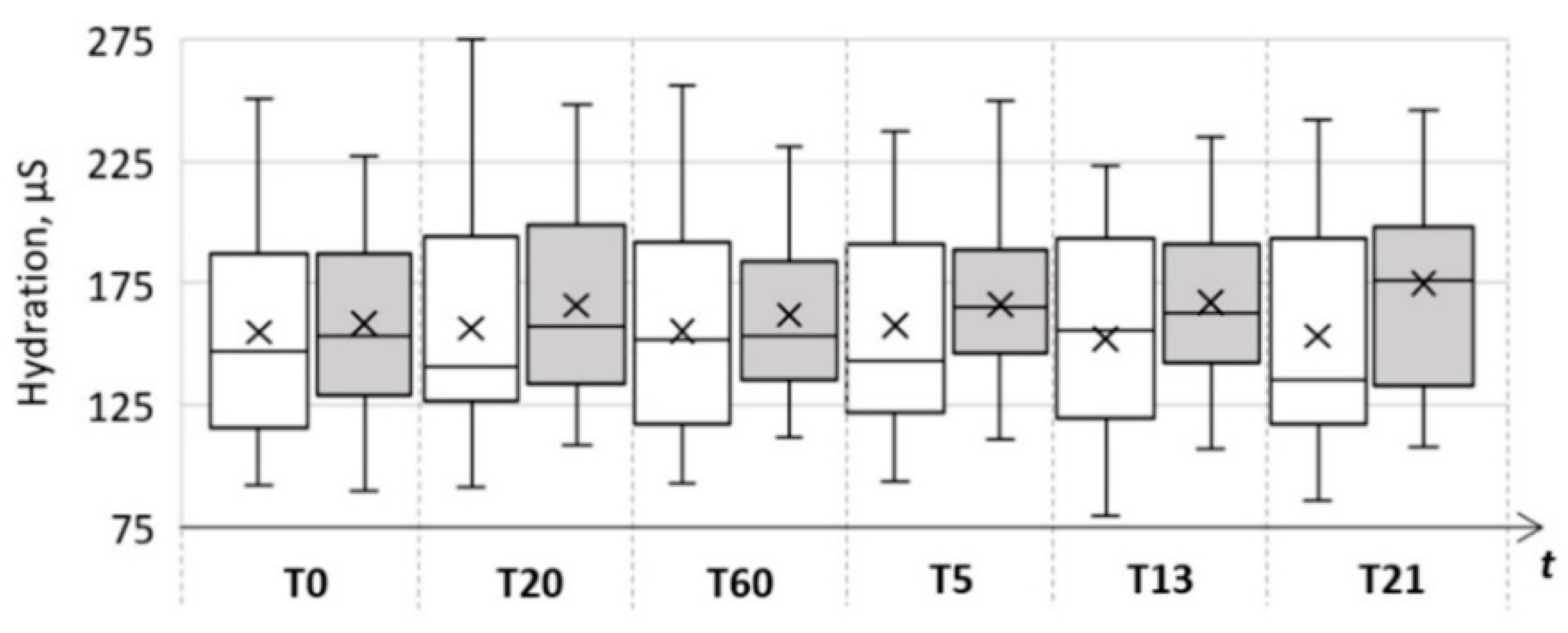
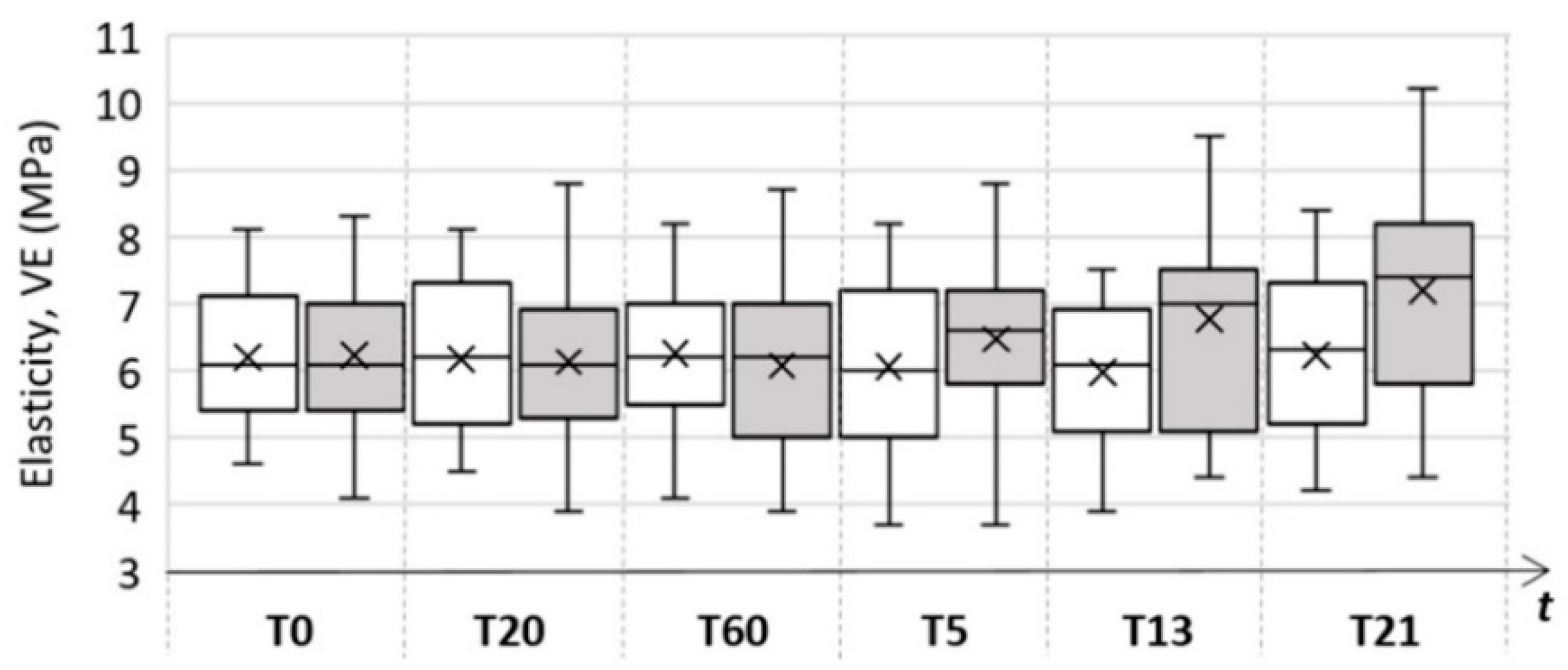
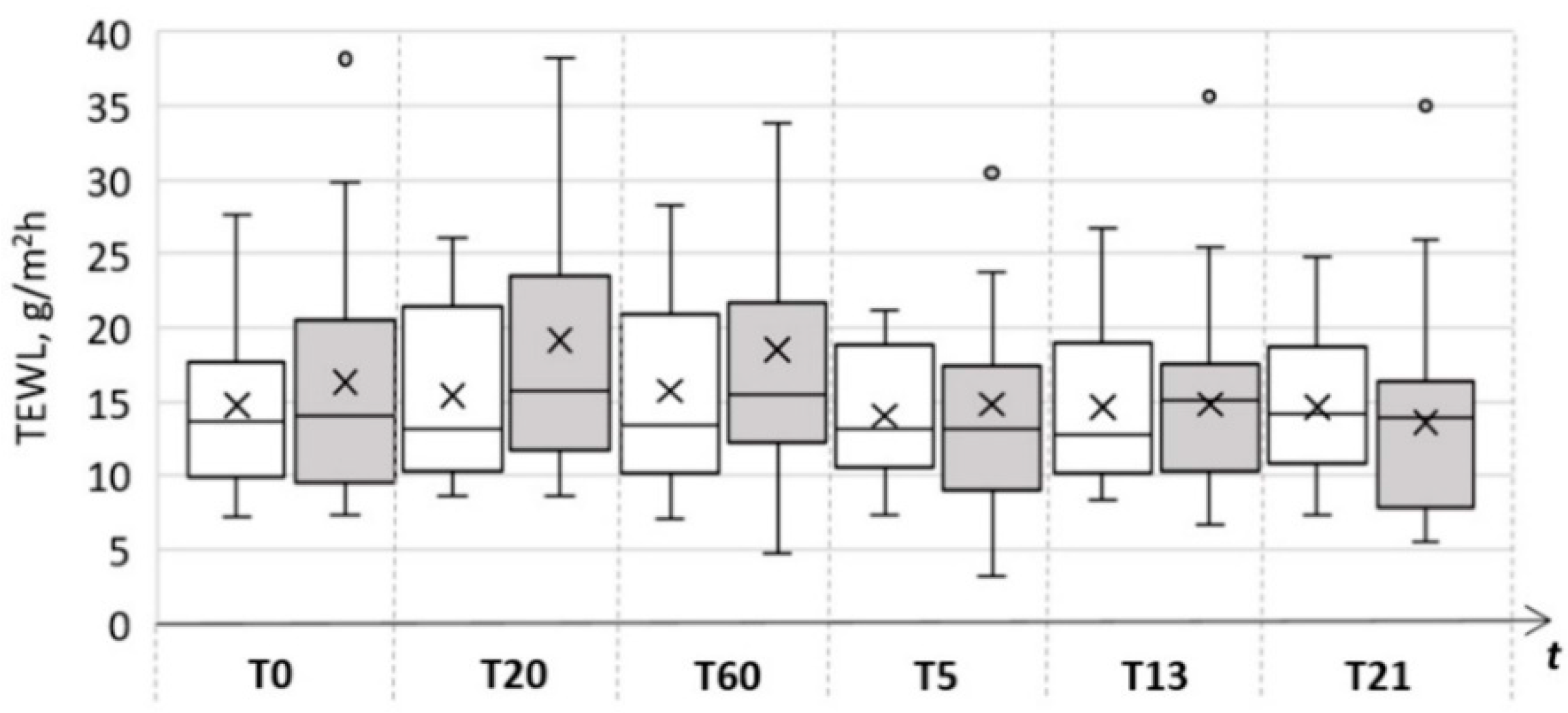
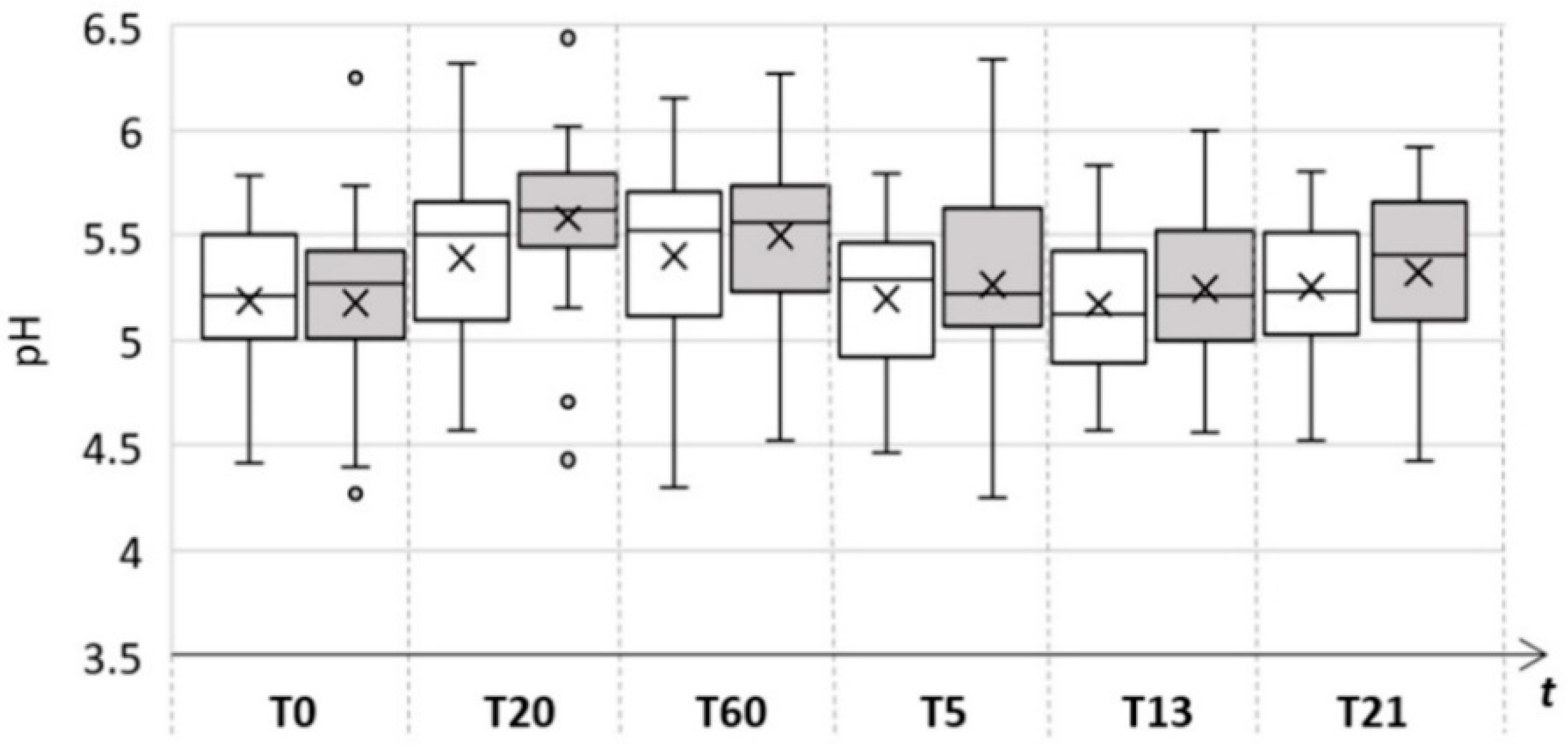
| Skin Parameter | Control Area | Test Area | ||||
|---|---|---|---|---|---|---|
| T0 | T60 | T21 | T0 | T60 | T21 | |
| Elasticity (MPa) | 6.2 ± 1.0 | 6.3 ± 1.1 | 6.2 ± 1.2 | 6.2 ± 1.2 | 6.1 ± 1.2 | 7.2 ± 1.5 |
| Hydration (µS) | 154.9 ± 42.6 | 155.6 ± 42.1 | 153.4 ± 45.9 | 158.4 ± 35.2 | 162.0 ± 35.1 | 174.8 ± 40.3 |
| TEWL (g/m2 h) | 14.7 ± 5.7 | 15.7 ± 6.6 | 14.6 ± 4.7 | 16.3 ± 8.1 | 18.5 ± 11.7 | 13.6 ± 6.4 |
| pH | 5.2 ± 0.4 | 5.4 ± 0.4 | 5.3 ± 0.4 | 5.2 ± 0.4 | 5.5 ± 0.4 | 5.3 ± 0.4 |
| Pair | Elasticity | Hydration | TEWL | PH |
|---|---|---|---|---|
| T0control and T0test | 0.70060 | 0.42080 | 0.17850 | 0.67410 |
| T0control and T60control | 0.61970 | 0.55560 | 0.04910 | 0.00145 |
| T0control and T21control | 0.35290 | 0.46130 | 0.85700 | 0.35490 |
| T0test and T60test | 0.10280 | 0.60240 | 0.06373 | 0.00006 |
| T60test and T60control | 0.49250 | 0.15620 | 0.07541 | 0.06600 |
| T0test and T21test | 0.00015 | 0.00096 | 0.00325 | 0.02878 |
| T21control and T21test | 0.00091 | 0.00034 | 0.03449 | 0.32170 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Misiņa, S.M.; Tretjakova, R.; Kodors, S.; Zavorins, A. Lake Zeiļu Clay Application Induced Changes in Human Skin Hydration, Elasticity, Transepidermal Water Loss and PH in Healthy Individuals. Cosmetics 2020, 7, 51. https://doi.org/10.3390/cosmetics7030051
Misiņa SM, Tretjakova R, Kodors S, Zavorins A. Lake Zeiļu Clay Application Induced Changes in Human Skin Hydration, Elasticity, Transepidermal Water Loss and PH in Healthy Individuals. Cosmetics. 2020; 7(3):51. https://doi.org/10.3390/cosmetics7030051
Chicago/Turabian StyleMisiņa, Samanta Marija, Rasma Tretjakova, Sergejs Kodors, and Aleksejs Zavorins. 2020. "Lake Zeiļu Clay Application Induced Changes in Human Skin Hydration, Elasticity, Transepidermal Water Loss and PH in Healthy Individuals" Cosmetics 7, no. 3: 51. https://doi.org/10.3390/cosmetics7030051
APA StyleMisiņa, S. M., Tretjakova, R., Kodors, S., & Zavorins, A. (2020). Lake Zeiļu Clay Application Induced Changes in Human Skin Hydration, Elasticity, Transepidermal Water Loss and PH in Healthy Individuals. Cosmetics, 7(3), 51. https://doi.org/10.3390/cosmetics7030051





